Integra PLUS SUTURABLE. Limit uncertainty with proven solutions for CSF leak prevention and natural dural repair.
|
|
|
- Bertha Tucker
- 6 years ago
- Views:
Transcription
1 DuraGen Family Dural Regeneration Matrices DuraGen PLUS Limit uncertainty with proven solutions for CSF leak prevention and natural dural repair. SUTURABLE DuraGen Products for sale in Europe, Middle-East and Africa only.
2 An innovation in Dural Repair A Pioneer in Collagen Regeneration Matrices For almost forty years, Integra and its predecessors have been leading the development and manufacturing of collagen implants and advanced regenerative technologies. In the early 1970 s the first collagen matrix to improve skin restoration of severely burned patients was developed. This advanced matrix led to a revolution in understanding the science behind the biological response and role of extracellular matrices in tissue regeneration. This science along with expertise in collagen processing and manufacturing resulted in DuraGen dural regeneration matrix. Integra pioneered regenerative medicine. Over 10 million collagen implants. 1 million implants and counting. Integra created the onlay dural graft paradigm. What is? Integra DuraGen Family Dural Regeneration Matrices Conforms intimately to complex surfaces. How does the Duragen matrix work? The onlay nature of DuraGen matrix revolutionized duraplasty. The highly porous collagen scaffold promotes rapid fibrin clot formation. DuraGen matrix rapidly provides watertight closure to prevent CSF leakage while promoting natural dural growth. DuraGen matrix is one of the safest and most effective onlay grafts for the restoration and repair of dura mater. DuraGen matrix is made from a controlled collagen source and is treated with a proprietary process designed to remove antigenic components, yielding our Ultra Pure Collagen Technology. It is conformable and contours instantly and effectively to the complex surfaces of the brain and spinal cord, rapidly forming a biological seal to protect against cerebrospinal fluid (CSF) leakage: 1 day post-implantation, a fibrin clot has formed within the matrix creating a watertight barrier. 2 Brochure Duragen Matrix
3 The Science that Seals The DuraGen matrix was engineered with the IDEAL balance of strength and flexibility to ensure OPTIMAL handling, sealing, and resorption-leaving the patient with a natural dural repair. It Starts with Ultra Pure Collagen Technology Harvested, processed, and purified to limit the risk of infection, immunological response, and foreign body reaction, thereby reducing the chance of fibrotic encapsulation. In almost 40 years of Ultra Pure Collagen Technology use, there have been no confirmed reports of foreignbody reactions or rejections. Integra s Ultra Pure Collagen Technology is the foundation of DuraGen matrix. Precisely Engineered Porosity Platelets infiltrate the matrix and initiate fibrin clot formation, forming an effective layer that prevents CSF leakage and initiates the dural repair process. The pore size is optimized to allow fibroblasts to rapidly enter the matrix and lay down natural collagen fibers. The optimized 99% porosity, even distribution, and pore interconnectivity promote uniform tissue regeneration throughout the matrix. Excellent Conformability and Handling Offers optimal conformability and ease of handling. DuraGen matrix is quickly and easily hydrated prior to implantation or in situ. Upon hydration, DuraGen matrix becomes a pliable membrane that conforms to the existing dura and remains in place through surface tension and fibrin clot formation, eliminating the need for sutures. Integra s Dural Regeneration Matrices are specially designed to meet your cranial and spinal needs. Safety Profile Better safety profile than synthetic dural substitute 1-6 minimization of postoperative complications. Infection rate comparable to other methods of dural closure. 1-5 Effectiveness proved against CSF leakage. 1,3,4,6 Inhibition of fibrosis and prevention of adhesion. 8 Incidence of infection Heuer et al. 2 Messing-Jünger et al. 5 von Wild 3 Raul et al. 4 Malliti et al Percentage of patients Integra Dural Regeneration Matrix Heuer et al. 2 Reyes-Moreno et al. 7 Messing-Jünger et al. 5 Raul et al. 4 Synthetic substitute Percentage of patients Autograft 3 Brochure Duragen Matrix
4 The Science That Seals Scientific Superiority 1 Excellent Conformability and Adherence The hydrated graft conforms closely to the complex surfaces of the exposed brain or spinal cord. Matrix rapidly fills with the patient s blood and plasma exudate. 2 Rapid CSF Leak Prevention Type 1 collagen matrix rapidly initiates platelet aggregation. Upon contact with the collagen matrix, platelets degranulate and release clotting factors that initiate fibrin clot formation. The fibrin clot creates a watertight barrier and binds the implanted matrix to the patient s dura. In Vivo Dural Repair Continuum By 60-days post-implantation, new dural tissue has formed. Erythrocytes Red Blood Cells Macrophages White Blood Cells Erythrocytes Red Blood Cells Fully Hydrated Collagen Fibers Collagen Fibers Spaces Filled with Fibrin Clot Fibroblasts 1 Day Post Implantation Fibrin clot formed within matrix creating watertight barrier 3 Days Post Implantation Fibroblasts infiltrate and attach to graft matrix to lay down new collagen 4 Brochure Duragen Matrix
5 The Science That Seals 3 Rapid Fibroblast Infiltration Ultra Pure Collagen Technology, in combination with the open pore structures, promotes fibroblast activity and acts as a scaffold for cells to deposit new collagen. The graft structure features pores of 50 to 150 microns, within the optimal size for rapid fibroblast infiltration. Fibroblasts begin to migrate into the matrix 2 to 3 days after implantation and start the process of laying down new collagen. 4 Uniform Tissue Formation Within two weeks of implantation, a neodural membrane has formed between the dural margins to permanently close the dural defect. After 6-8 weeks, the implant is resorbed and replaced by dura. After 1 year, the neodura has developed into mature dura. Bone Matrix Filled with Human Collagen Outer Layer Regenerated Dura with Appropriate Inner and Outer Layers Regenerated Dura Inner Layer Arachnoid Cortex Fibroblasts Blood Vessels Cranial Margin 60 Days Post Implantation Collagen matrix has been resorbed and replaced with dura 26 Weeks Post Implantation Dura as seen in secondary cranioplasty procedure 6-months after decomressive hemicraniectomy* *G. Heuer, M. Stiefel, E. Maloney-Wilensky, S. Danish, C. Dolinskas, P. LeRoux. DuraGen is an Effective Dural Substitute: Clinical Experience in 100 Patients. American Association of Neurological Surgeons Annual Meeting, April Brochure Duragen Matrix
6 The Leading Family of Dural Repair Products Limit uncertainty with one of the most innovative line of products for optimal dural repair: PLUS: Reliability DuraGen Plus matrix has an excellent safety record and provides industry-leading conformability and resorption. The improved consistency of DuraGen Plus matrix* offers increased tensile strength for optimized handling during challenging neurosurgery cases. Clinically proven to limit CSF leakage, DuraGen Plus matrix is also indicated to be used as an adhesion barrier in spinal procedures. *compared with DuraGen matrix, the 1 st generation of Integra Dural Regeneration Matrices. SUTURABLE: Adaptability Suturable DuraGen matrix provides the benefits of DuraGen Plus matrix with the added versatility of accommodating both suture and onlay techniques. Suturable DuraGen matrix is a bilayer collagen graft with enhanced strength and support, providing the ability to suture without losing the conformability and resorption you demand from a dural graft. DuraGen Plus Indications DuraGen Plus Adhesion Barrier Matrix is indicated as an onlay graft for the repair and restoration of dural defects in cranial and spinal surgical procedures. DuraGen Plus matrix is also indicated as an adhesion barrier for the inhibition of post-surgical peridural fibrosis. DuraGen Plus matrix readily conforms to the surface of the brain, spinal cord and overlying tissues. DuraGen Plus matrix may be used to close dural defects following traumatic injury, excision, retraction or shrinkage. DuraGen Plus matrix may be used to supplement primary closure. DuraGen Plus Contraindications DuraGen Plus matrix is not designed, sold or intended for use except as described in the indications for use and is contraindicated in the following situations: For patients with a known history of hypersensitivity to bovine - derived materials. For primary repair of spinal neural tube defects; anterior spinal surgery with dural resection (e.g., transoral surgery). Should be used with caution in infected regions. Not recommended to cover dural defects involving mastoid air cells. Not recommended for large defects at the skull base following surgery. Suturable Duragen Indications Suturable DuraGen is indicated as a dural substitute for the repair and restoration of dural defects in cranial and spinal surgical procedures. Suturable DuraGen readily conforms to the surface of the brain and overlying tissues. Suturable DuraGen may be used to close dural defects following traumatic injury, excision, retraction or shrinkage. Suturable DuraGen may be used to supplement primary closure. Suturable Duragen Contraindications Suturable DuraGen is not designed, sold or intended for use except as described in the indications for use and is contraindicated in the following situations: For patients with a known history of hypersensitivity to bovine derived materials. Should be used with caution in infected regions. 6 Brochure Duragen Matrix
7 Product Sizes to Meet Your Surgical Needs Sizes shown in inches at actual size DuraGen Plus Dural Regeneration Matrix Reference Size Units DP-1011-l 1 in x 1 in (2.5 cm x 2.5 cm) 1 DP-5011-l 1 in x 1 in (2.5 cm x 2.5 cm) 5 DP-1013-l 1 in x 3 in (2.5 cm x 7.5 cm) 1 DP-5013-l 1 in x 3 in (2.5 cm x 7.5 cm) 5 DP-1022-l 2 in x 2 in (5 cm x 5 cm) 1 DP-5022-l 2 in x 2 in (5 cm x 5 cm) 5 DP-1033-l 3 in x 3 in (7.5 cm x 7.5 cm) 1 DP-5033-l 3 in x 3 in (7.5 cm x 7.5 cm) 5 DP-1045-l 4 in x 5 in (10 cm x 12.5 cm) 1 DP-1057-l 5 in x 7 in (12.5 cm x 17.5 cm) 1 Suturable DuraGen Dural Regeneration Matrix Reference Size Units DURS-1391-ITL 1 in x 3 in (2.5 cm x 7.5 cm) 1 DURS-2291-ITL 2 in x 2 in (5 cm x 5 cm) 1 DURS-3391-ITL 3 in x 3 in (7.5 cm x 7.5 cm) 1 DURS-4591-ITL 4 in x 5 in (10 cm x 12.5 cm) 1 References 1. G. Heuer, M. Stiefel, E. Maloney-Wilensky, S. Danish, C. Dolinskas, P. LeRoux. DuraGen is an Effective Dural Substitute: Clinical Experience in 100 Patients. American Association of Neurological Surgeons Annual Meeting, April K. von Wild. Examination of the safety and efficacy of an absorbable dura mater substitute (Dura Patch) in normal applications in neurosurgery. Surg Neurol 52: , JS. Raul, J. Godard, F. Arbez-Gindre, A. Czorny. Use of polyester urethane (Neuro-Patch) as a dural substitute. Prospective study of 70 cases. Neurochirurgie 2003 May;49(2-3 Pt1): AM. Messing-Jünger, J. Ibáñez, F. Calbucci, M. Choux, G. Lena, I. Mohsenipour, F. Van Calenbergh. Effectiveness and handling characteristics of a three-layer polymer dura substitute: a prospective multicenter clinical study. J Neurosurg Dec;105(6): M. Malliti, P. Page, C. Gury, E. Chomette, F. Nataf, FX. Roux. Comparison of deep wound infection rates using a synthetic dural substitute (neuro-patch) or pericranium graft for dural closure: a clinical review of 1 year. Neurosurgery 2004 Mar;54(3): I. Reyes-Moreno, R. Verheggen. Time-sparing and effective procedure for dural closure in the posterior fossa using a vicryl mesh (Ethisorb). Neurocirugia 2006 Dec;17(6): I. Haq, Y. Cruz-Almeida, EB. Siqueira, M. Norenberg, BA. Green, AD. Levi. Postoperative fibrosis after surgical treatment of the porcine spinal cord: a comparison of dural substitutes. J Neurosurg Spine 2:50 54, P. Narotam, A. Gousseau, G. McGinn. Collagen Matrix (DuraGen) for duraplasty following cranial and spinal surgery. 35 th Canadian Congress of Neurological Sciences, Ottawa, Canada, June Brochure Duragen Matrix
8 Unsurpassed Clinical Data Over 1,400 patients in 10 published clinical studies 0% Foreign body response 0% - 5.6% Infection rate 0% - 7.1% Leakage rate Integra s DuraGen products have more published human clinical data than any other collagenbased dural graft. Clinical studies have shown effective protection against CSF leakage with sutureless closure and no reports of foreign body reactions or graft rejections. Summary outcome statistics derived from the following 1o clinical studies: 1. Danish SF, et al: Experience with acellular human dura and bovine collagen matrix for duraplasty after posterior fossa decompression for Chiari malformations.j Neurosurg Pediatrics 104:16-20, Harvey RJ, et al: Closure of large skull base defects after endoscopic transnasal craniotomy. J Neurosurg 111 (2) : , Horaczek JA, et al: Collagen matrix in decompressive hemicraniectomy. Neurosurgery 63 (1 suppl.1): ONS 176-ONS 181, Lee JH, et al: Dural reconstruction in meningioma surgery in, Lee JH (ed): Meningiomas: Diagnosis, Treatment an Outcome. London: Springer, 2009, pp Litvack ZN, et al: Dural augmentation: Part I: evaluation of collagen matrix allografts for dural defect after craniotomy. Neurosurgery 65 (5): , Narotam PK, et al: Collagen matrix (DuraGen) in dural repair: analysis of a new modified technique. Spine 29 (24) : , Narotam PK, et al: Collagen matrix duraplasty for cranial and spinal surgery: a clinical and imaging study. J Neurosurg 106 (1): 45 51, Narotam PK, et al: Collagen matrix duraplasty for posterior fossa surgery: evaluation of surgical technique in 52 adult patients. J Neurosurg 111 (2): , Sade B, et al: Non-watertight dural reconstruction in meningioma surgery: results in 439 consecutive patients and a review of the literature. J Neurosurg 114 (3) : , Stendel R, et al: Efficacy and safety of a collagen matrix for cranial and spinal dural reconstruction using different fixation techniques. J Neurosurg 109 (2): , DuraGen Family Regenerative Matrices Made from Ultra Pure Collagen Technology. Precisely engineered porosity for complete and natural repair. Excellent conformability & handling. 1 Million Implants and Counting Integra DuraGen graft provides the confidence of utilizing a dural matrix which has been implanted over one million times. Integra LifeSciences Services (France) SAS Sales & Marketing EMEA Immeuble Séquoia 2 97 allée Alexandre Borodine Parc technologique de la Porte des Alpes Saint Priest FRANCE Phone: +33 (0) Fax: +33 (0) emea.info@integralife.com integralife.eu Distributed by Customer Services International: +33 (0) (0) (Fax) csemea@integralife.com France: +33 (0) (0) (Fax) custsvcfrance@integralife.com United Kingdom: +44 (0) (0) (Fax) custsvcuk@integralife.com Germany: +49 (0) (0) (Fax) custsvcgermany@integralife.com Benelux: +32 (0) (0) (Fax) custsvcbenelux@integralife.com Switzerland: +41 (0) (0) (Fax) custsvcsuisse@integralife.com Integra LifeSciences Corporation 311 Enterprise Drive Plainsboro, NJ USA 2012 Integra LifeSciences Corporation. All rights reserved Availability of these products might vary from a given country or region to another, as a result of specific local regulatory approval or clearance requirements for sale in such country or region. Always refer to the appropriate instructions for use for complete clinical instructions. Non contractual document. The manufacturer reserves the right, without prior notice, to modify the products in order to improve their quality. WARNING: Applicable laws restrict these products to sale by or on the order of a physician. Duragen, Integra and the Integra logo are registered trademarks of Integra Lifesciences Corporation in the United States and/or other countries. Duragen Plus, Suturable Duragen and Ultra Pure Collagen are trademarks of Integra Lifesciences Corporation. All the references numbers mentioned on this document are CE marked according to European council directive 93/42/EEC on medical devices and its relatives, unless specifically identified as NOT CE MARKED. Last modification: 07/2012 Class III BSI
DuraGen Secure. Dural Regeneration Matrix
 DuraGen Secure Dural Regeneration Matrix DESCRIPTION DuraGen Secure Dural Regeneration Matrix is an absorbable implant for the repair of dural defects. DuraGen Secure Dural Regeneration Matrix is an easy
DuraGen Secure Dural Regeneration Matrix DESCRIPTION DuraGen Secure Dural Regeneration Matrix is an absorbable implant for the repair of dural defects. DuraGen Secure Dural Regeneration Matrix is an easy
EVIDENCE FOR SYNTHECEL DURA REPAIR. Value Analysis Brief: Introduction. Methods
 Value Analysis Brief: EVIDENCE FOR SYNTHECEL DURA REPAIR Introduction This value analysis brief presents evidence for SYNTHECEL Dura Repair in the repair of dura mater (duraplasty) during cranial surgery,
Value Analysis Brief: EVIDENCE FOR SYNTHECEL DURA REPAIR Introduction This value analysis brief presents evidence for SYNTHECEL Dura Repair in the repair of dura mater (duraplasty) during cranial surgery,
Safe Harbor Statement
 May 2004 A leader in the research, manufacturing and marketing of medical devices used in neuro-trauma and neurosurgery, plastic and reconstructive surgery and general surgery Safe Harbor Statement Certain
May 2004 A leader in the research, manufacturing and marketing of medical devices used in neuro-trauma and neurosurgery, plastic and reconstructive surgery and general surgery Safe Harbor Statement Certain
What is the future of ACL reconstruction?
 What is the future of ACL reconstruction? Charles J. Gatt, Jr., MD Chair, Department of Orthopaedic Surgery Rutgers Robert Wood Johnson Medical School New Brunswick, NJ Clinical question Do patients with
What is the future of ACL reconstruction? Charles J. Gatt, Jr., MD Chair, Department of Orthopaedic Surgery Rutgers Robert Wood Johnson Medical School New Brunswick, NJ Clinical question Do patients with
SurgiMend. Product Summary
 SurgiMend Collagen Matrix for Soft Tissue Reconstruction Product Summary Because we are committed to limiting uncertainty, Integra offers SurgiMend in 1, 2, 3 or 4 mm thicknesses and in over sixty configurations,
SurgiMend Collagen Matrix for Soft Tissue Reconstruction Product Summary Because we are committed to limiting uncertainty, Integra offers SurgiMend in 1, 2, 3 or 4 mm thicknesses and in over sixty configurations,
Ventrio ST Hernia Patch featuring Sepra Technology
 Ventrio ST Hernia Patch featuring Sepra Technology Sepra Technology An extensively studied barrier with more than 10 publications and used clinically since 2007. Unique hydrogel barrier swells to minimize
Ventrio ST Hernia Patch featuring Sepra Technology Sepra Technology An extensively studied barrier with more than 10 publications and used clinically since 2007. Unique hydrogel barrier swells to minimize
SCIENCE. VALUE. INNOVATION.
 SCIENCE. VALUE. INNOVATION. OUR STORY TELA Bio, a surgical reconstruction company, was created out of the desire to do things differently. Combining the drive and innovation of a start-up with a seasoned
SCIENCE. VALUE. INNOVATION. OUR STORY TELA Bio, a surgical reconstruction company, was created out of the desire to do things differently. Combining the drive and innovation of a start-up with a seasoned
Instructions for Use READY TO USE
 READY TO USE Instructions for Use LifeCell Corporation One Millennium Way Branchburg, NJ 08876-3876 1.908.947.1215 1.800.367.5737 Fax: 1.908.947.1089 E-mail: customerservicegroup@lifecell.com www.lifecell.com
READY TO USE Instructions for Use LifeCell Corporation One Millennium Way Branchburg, NJ 08876-3876 1.908.947.1215 1.800.367.5737 Fax: 1.908.947.1089 E-mail: customerservicegroup@lifecell.com www.lifecell.com
D URING the past war many articles 4'~,6'7,8'1~ were published on the
 EXPERIMENTAL OBSERVATIONS ON THE USE OF STAINLESS STEEL FOR CRANIOPLASTY A COMPARISON WITH TANTALUM* MICHAEL SCOTT, M.D., A~D HENRY T. WYCIS, M.D. Department of Neurosurgery, Temple University Hospital
EXPERIMENTAL OBSERVATIONS ON THE USE OF STAINLESS STEEL FOR CRANIOPLASTY A COMPARISON WITH TANTALUM* MICHAEL SCOTT, M.D., A~D HENRY T. WYCIS, M.D. Department of Neurosurgery, Temple University Hospital
A review of Dr. Dinakar Golla s clinical research with AdMatrix surgical grafts for soft tissue repair
 TEAMeffort: Using aggressive surgical techniques in combination with AdMatrix (Lattice Biologics acellular dermal scaffold product) to heal difficult and persistent wounds A review of Dr. Dinakar Golla
TEAMeffort: Using aggressive surgical techniques in combination with AdMatrix (Lattice Biologics acellular dermal scaffold product) to heal difficult and persistent wounds A review of Dr. Dinakar Golla
UNIT CELL PROCESSES UNDERLYING TISSUE ENGINEERING AND REGENERATIVE MEDICINE
 Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/20.441/HST522J UNIT CELL PROCESSES UNDERLYING TISSUE ENGINEERING AND REGENERATIVE
Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/20.441/HST522J UNIT CELL PROCESSES UNDERLYING TISSUE ENGINEERING AND REGENERATIVE
LITe LIF Less invasive posterior lumbar interbody fusion procedure
 LITe LIF Less invasive posterior lumbar interbody fusion procedure LITe LIF Access LITe Midline Retractor Fixation Xia CT Interbody Tritanium PL Cage Instruments LITe Decompression 2.0 Power CD3 and RemB
LITe LIF Less invasive posterior lumbar interbody fusion procedure LITe LIF Access LITe Midline Retractor Fixation Xia CT Interbody Tritanium PL Cage Instruments LITe Decompression 2.0 Power CD3 and RemB
The Ventrio ST Hernia Patch Featuring Sepra Technology. Efficient:
 Ventrio ST Hernia Patch featuring SEPRA Technology NEW Sepra Technology Based on the technology used in Seprafilm with more than 13 years of proven clinical success. Unique hydrogel barrier swells to minimise
Ventrio ST Hernia Patch featuring SEPRA Technology NEW Sepra Technology Based on the technology used in Seprafilm with more than 13 years of proven clinical success. Unique hydrogel barrier swells to minimise
Disclosure-Yaszemski
 Current and Future Uses of Additive Manufacturing in Neurologic, Musculoskeletal, Spinal, and Oncologic Surgery Michael J. Yaszemski, M.D., Ph.D. John & Posy Krehbiel Endowed Professor of Orthopedic Surgery
Current and Future Uses of Additive Manufacturing in Neurologic, Musculoskeletal, Spinal, and Oncologic Surgery Michael J. Yaszemski, M.D., Ph.D. John & Posy Krehbiel Endowed Professor of Orthopedic Surgery
Affinity. A Paradigm Shift in Skeletal Reconstruction
 Affinity A Paradigm Shift in Skeletal Reconstruction INTRODUCING TRS AFFINITY SKELETAL RECONSTRUCTION BREAKTHROUGH Tissue Regeneration Systems (TRS ) is a start-up medical device company commercializing
Affinity A Paradigm Shift in Skeletal Reconstruction INTRODUCING TRS AFFINITY SKELETAL RECONSTRUCTION BREAKTHROUGH Tissue Regeneration Systems (TRS ) is a start-up medical device company commercializing
Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020
 Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020 Reference Code: GDMECC0884DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020 Reference Code: GDMECC0884DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
Advancing the Open Ventral Hernia Repair Experience
 Advancing the Open Ventral Hernia Repair Experience SOFT TISSUE REPAIR Right Procedure. Right Product. Right Outcome. Hernia Repair Fixation Absorbable Fixation System Optimized Design for Open Ventral
Advancing the Open Ventral Hernia Repair Experience SOFT TISSUE REPAIR Right Procedure. Right Product. Right Outcome. Hernia Repair Fixation Absorbable Fixation System Optimized Design for Open Ventral
Specifically designed for abdominal wall repair STRUCTURAL
 Specifically designed for abdominal wall repair STRUCTURAL The Better Approach To a Better Allograft Better Standards: Board of Directors comprised primarily of surgeons 3-years run by surgeons, for surgeons
Specifically designed for abdominal wall repair STRUCTURAL The Better Approach To a Better Allograft Better Standards: Board of Directors comprised primarily of surgeons 3-years run by surgeons, for surgeons
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 2004 CPMP/BPWG/1089/00 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON THE CLINICAL INVESTIGATION OF PLASMA
European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 2004 CPMP/BPWG/1089/00 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON THE CLINICAL INVESTIGATION OF PLASMA
Anika Therapeutics, Inc.
 Anika Therapeutics, Inc. Jefferies Global Healthcare Conference June 4, 2015 Safe Harbor Statement The statements made in this presentation that are not statements of historical fact are forward looking
Anika Therapeutics, Inc. Jefferies Global Healthcare Conference June 4, 2015 Safe Harbor Statement The statements made in this presentation that are not statements of historical fact are forward looking
Presenting SURGICEL Powder
 SURGICEL Powder Presenting SURGICEL Powder Built to stop continuous, broad-surface oozing fast1,2 The next generation of SURGICEL Absorbable Hemostats SURGICEL Powder efficiently and effectively controls
SURGICEL Powder Presenting SURGICEL Powder Built to stop continuous, broad-surface oozing fast1,2 The next generation of SURGICEL Absorbable Hemostats SURGICEL Powder efficiently and effectively controls
ALLODERM SELECT ALLODERM SELECT RESTORE
 ALLODERM SELECT ALLODERM SELECT RESTORE Regenerative Tissue Matrix Instructions for Use Processed from donated human tissue by: LifeCell Corporation One Millennium Way Branchburg, NJ 08876-3876 DESCRIPTION
ALLODERM SELECT ALLODERM SELECT RESTORE Regenerative Tissue Matrix Instructions for Use Processed from donated human tissue by: LifeCell Corporation One Millennium Way Branchburg, NJ 08876-3876 DESCRIPTION
Biological Prostheses
 SAPIENZA UNIVERSITA DI ROMA EHS Dipartimento di Chirurgia Generale Paride Stefanini U.O.C. di Chirurgia Generale D e Day Surgery (Direttore: Prof. P. Negro) PRG M Chirurgia della Parete Addominale (Prof.
SAPIENZA UNIVERSITA DI ROMA EHS Dipartimento di Chirurgia Generale Paride Stefanini U.O.C. di Chirurgia Generale D e Day Surgery (Direttore: Prof. P. Negro) PRG M Chirurgia della Parete Addominale (Prof.
DermaMatrix Acellular Dermis. Comparative testing.
 DermaMatrix Acellular Dermis. Comparative testing. Natural, like native tissue Minimally processed Strong and flexible DermaMatrix Acellular Dermis. Human dermal collagen matrix. Unique process DermaMatrix
DermaMatrix Acellular Dermis. Comparative testing. Natural, like native tissue Minimally processed Strong and flexible DermaMatrix Acellular Dermis. Human dermal collagen matrix. Unique process DermaMatrix
A.P.A.G. Activated Plasma Albumin Gel
 A.P.A.G. Activated Plasma Albumin Gel How Does This Work In a new wound, platelets are the first cells which arrive. They aggregate to stop the bleeding and then release substances (growth factors, cytokines,
A.P.A.G. Activated Plasma Albumin Gel How Does This Work In a new wound, platelets are the first cells which arrive. They aggregate to stop the bleeding and then release substances (growth factors, cytokines,
Finally! Everything you need when supporting soft tissue; nothing you don t. Strength and Beauty. Inside and Out.
 alatea Surgical Bioresorbable Scaffold Collection a Strength and Beauty. Inside and Out. 3D 3-Dimensional Biologically Derived Monofilament Strong Bioresorbable Finally! Everything you need when supporting
alatea Surgical Bioresorbable Scaffold Collection a Strength and Beauty. Inside and Out. 3D 3-Dimensional Biologically Derived Monofilament Strong Bioresorbable Finally! Everything you need when supporting
More than surface deep
 Tritanium PL Posterior Lumbar Cage More than surface deep Featuring Tritanium In-Growth Technology: 1 TM Built to fuse Tritanium In-Growth Technology1 Stryker s proprietary Tritanium In-Growth Technology,
Tritanium PL Posterior Lumbar Cage More than surface deep Featuring Tritanium In-Growth Technology: 1 TM Built to fuse Tritanium In-Growth Technology1 Stryker s proprietary Tritanium In-Growth Technology,
FORWARD LOOKING STATEMENT
 FORWARD LOOKING STATEMENT Statements made in this presentation that look forward in time or that express management's beliefs, expectations, hopes or predictions of the future are forward-looking statements
FORWARD LOOKING STATEMENT Statements made in this presentation that look forward in time or that express management's beliefs, expectations, hopes or predictions of the future are forward-looking statements
United Kingdom Tissue Engineered - Skin Substitutes Market Outlook to 2020
 United Kingdom Tissue Engineered - Skin Substitutes Market Outlook to Reference Code: GDMECC0860DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
United Kingdom Tissue Engineered - Skin Substitutes Market Outlook to Reference Code: GDMECC0860DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
TRELLIS COLLAGEN RIBBON
 TRELLIS COLLAGEN RIBBON 147321-1 English (en) The following languages are included in this packet: M Wright Medical Technology, Inc. 5677 Airline Rd. Arlington, TN 38002 USA www.wmt.com August 2012 Printed
TRELLIS COLLAGEN RIBBON 147321-1 English (en) The following languages are included in this packet: M Wright Medical Technology, Inc. 5677 Airline Rd. Arlington, TN 38002 USA www.wmt.com August 2012 Printed
Visualizing the invisible
 Visualizing the invisible Intraoperative Multi-Modal Imaging in traoperative visualization technology SonoWand Invite allows the surgeon to identify and distinguish lesions from normal brain tissue and
Visualizing the invisible Intraoperative Multi-Modal Imaging in traoperative visualization technology SonoWand Invite allows the surgeon to identify and distinguish lesions from normal brain tissue and
Biologic Prosthetics & Beyond
 & Beyond & Beyond: Overview Introduction Prosthetics Overview UCSF Postgraduate Course in General Surgery San Francisco, CA May 18, 2013 Review of Clinical Data Summary Recommendations Hobart W. Harris,
& Beyond & Beyond: Overview Introduction Prosthetics Overview UCSF Postgraduate Course in General Surgery San Francisco, CA May 18, 2013 Review of Clinical Data Summary Recommendations Hobart W. Harris,
Lyset BOOST YOUR CELL CULTURE TODAY FOR THE EXPERIMENTS OF TOMORROW
 Lyset BOOST YOUR CELL CULTURE TODAY FOR THE EXPERIMENTS OF TOMORROW Lyset, the human platelet derived supplement for cell culture Among the different alternatives to animal serum, platelet derived preparations
Lyset BOOST YOUR CELL CULTURE TODAY FOR THE EXPERIMENTS OF TOMORROW Lyset, the human platelet derived supplement for cell culture Among the different alternatives to animal serum, platelet derived preparations
A Primer for budding entrepreneurs: I think I have a great idea, now what? Background on Confluent The Primer Discussion
 A Primer for budding entrepreneurs: I think I have a great idea, now what? Background on Confluent The Primer Discussion Company Overview Platform technology of in situ polymerized biomaterials and delivery
A Primer for budding entrepreneurs: I think I have a great idea, now what? Background on Confluent The Primer Discussion Company Overview Platform technology of in situ polymerized biomaterials and delivery
CRANIOS REINFORCED BONE VOID FILLER
 CRANIOS REINFORCED BONE VOID FILLER The tougher bone cement TECHNICAL MONOGRAPH INTRODUCTION CRANIOS REINFORCED Bone Void Filler contains reinforcing fibers that impart an increase in toughness* to reduce
CRANIOS REINFORCED BONE VOID FILLER The tougher bone cement TECHNICAL MONOGRAPH INTRODUCTION CRANIOS REINFORCED Bone Void Filler contains reinforcing fibers that impart an increase in toughness* to reduce
CAUTION: U.S. Federal law restricts this device to sale by or on the order of a licensed physician.
 TM CAUTION: U.S. Federal law restricts this device to sale by or on the order of a licensed physician. TABLE OF CONTENTS Section Port Styles 4 Description 5 Indications 5 Contraindications 5-6 Information
TM CAUTION: U.S. Federal law restricts this device to sale by or on the order of a licensed physician. TABLE OF CONTENTS Section Port Styles 4 Description 5 Indications 5 Contraindications 5-6 Information
Alex A. Aimetti, PhD Sr. Director, Medical Education October 29, InVivo Therapeutics
 Translation of Biomaterial-based Therapies for the Treatment of Acute and Chronic Spinal Cord Injury: The Neuro-Spinal Scaffold and Bioengineered Neural Trails Alex A. Aimetti, PhD Sr. Director, Medical
Translation of Biomaterial-based Therapies for the Treatment of Acute and Chronic Spinal Cord Injury: The Neuro-Spinal Scaffold and Bioengineered Neural Trails Alex A. Aimetti, PhD Sr. Director, Medical
Histoacryl A revolution in mesh fixation
 Histoacryl A revolution in mesh fixation Closure Technologies Histoacryl A revolution in mesh fixation A new indication for a classic product Histoacryl has been used for more than 40 years in operating
Histoacryl A revolution in mesh fixation Closure Technologies Histoacryl A revolution in mesh fixation A new indication for a classic product Histoacryl has been used for more than 40 years in operating
The Biomechanics of ProLayer Acellular Dermal Matrix: suture retention strength
 The Biomechanics of ProLayer Acellular Dermal Matrix: suture retention strength Reginald Stilwell, B.S., C.T.B.S., Ryan Delaney, M.S. ProLayer, Centennial, CO Basic science volume 2 ProLayer Acellular
The Biomechanics of ProLayer Acellular Dermal Matrix: suture retention strength Reginald Stilwell, B.S., C.T.B.S., Ryan Delaney, M.S. ProLayer, Centennial, CO Basic science volume 2 ProLayer Acellular
T HE DEMONSTRATION of the effectiveness of fibrin foam as an agent for
 FURTHER USES OF GELATIN FOAM IN NEUROSURGERY* EDGAR F. FINCHER, M.D. Department of Surgery, Emory University School of Medicine, Atlanta, Georgia (Received for publication October 12, 1946) T HE DEMONSTRATION
FURTHER USES OF GELATIN FOAM IN NEUROSURGERY* EDGAR F. FINCHER, M.D. Department of Surgery, Emory University School of Medicine, Atlanta, Georgia (Received for publication October 12, 1946) T HE DEMONSTRATION
Spinal duraplasty materials and hydrostasis: a biomechanical study
 J Neurosurg Spine 15:422 427, 2011 Spinal duraplasty materials and hydrostasis: a biomechanical study Laboratory investigation Michael A. Finn, M.D., 1 Nathan D. Faulkner, M.D., 2 Scott J. Hetzel, M.S.,
J Neurosurg Spine 15:422 427, 2011 Spinal duraplasty materials and hydrostasis: a biomechanical study Laboratory investigation Michael A. Finn, M.D., 1 Nathan D. Faulkner, M.D., 2 Scott J. Hetzel, M.S.,
White Paper: Designing Functional Fabrics for Today s Heart Valves and Beyond
 White Paper: Designing Functional Fabrics for Today s Heart Valves and Beyond Ryan Heniford Business Development Director Peter Gabriele Director, Emerging Technology How fabrics can be used in heart valves,
White Paper: Designing Functional Fabrics for Today s Heart Valves and Beyond Ryan Heniford Business Development Director Peter Gabriele Director, Emerging Technology How fabrics can be used in heart valves,
B i o m a t e r i a l s E n g i n e e r i n g
 B i o m a t e r i a l s E n g i n e e r i n g Collagen, a key factor for clinical success INTRODUCTION A REVOLUTIONARY INNOVATION Tecnoss exclusive manufacturing process is able to neutralize the antigenic
B i o m a t e r i a l s E n g i n e e r i n g Collagen, a key factor for clinical success INTRODUCTION A REVOLUTIONARY INNOVATION Tecnoss exclusive manufacturing process is able to neutralize the antigenic
TISSUE ENGINEERING AND REGENERATION: TECHNOLOGIES AND GLOBAL MARKETS
 TISSUE ENGINEERING AND REGENERATION: TECHNOLOGIES AND GLOBAL MARKETS HLC101B August 2014 Yojana Jeevane Project Analyst ISBN: 1-56965-894-3 BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA
TISSUE ENGINEERING AND REGENERATION: TECHNOLOGIES AND GLOBAL MARKETS HLC101B August 2014 Yojana Jeevane Project Analyst ISBN: 1-56965-894-3 BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA
Presented by LINXObere Medizintechnik GmbHH
 Advanced Wound Closure Solutions By LINXOBERE Surgical Products Presented by LINXObere Medizintechnik GmbHH Needle Type Structure Symbol Main Applications Round Needles (Taper Point) Smallest possible
Advanced Wound Closure Solutions By LINXOBERE Surgical Products Presented by LINXObere Medizintechnik GmbHH Needle Type Structure Symbol Main Applications Round Needles (Taper Point) Smallest possible
Ask your surgeon for more information about Natrelle and visit GummyImplants.com
 The #1 surgeon-preferred gummy implant collection in the US* Two-stage breast reconstruction starts with Natrelle 133 Series Tissue Expanders Natrelle 133 Series Tissue Expanders are made to match Natrelle
The #1 surgeon-preferred gummy implant collection in the US* Two-stage breast reconstruction starts with Natrelle 133 Series Tissue Expanders Natrelle 133 Series Tissue Expanders are made to match Natrelle
InVivo Therapeutics. Developing Innovative Products for Spinal Cord Injury
 1 Developing Innovative Products for Spinal Cord Injury 2 Forward-Looking Statements Before we begin, we would like to remind everyone that during our presentation, we will be making forward-looking statements
1 Developing Innovative Products for Spinal Cord Injury 2 Forward-Looking Statements Before we begin, we would like to remind everyone that during our presentation, we will be making forward-looking statements
BIOTAPE XM TISSUE MATRIX The following languages are included in this packet:
 BIOTAPE XM TISSUE MATRIX 150866-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 -Chinese (sch)
BIOTAPE XM TISSUE MATRIX 150866-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 -Chinese (sch)
FDA Regulatory, Compliance and Policy Developments: 361 HCT/Ps
 FDA Regulatory, Compliance and Policy Developments: 361 HCT/Ps September 27, 2018 Presentation by: Elaine H. Tseng Partner FDA and Life Sciences Group King & Spalding Discussion with: Thomas Poché Vice
FDA Regulatory, Compliance and Policy Developments: 361 HCT/Ps September 27, 2018 Presentation by: Elaine H. Tseng Partner FDA and Life Sciences Group King & Spalding Discussion with: Thomas Poché Vice
Intercytex Group plc
 Intercytex Group plc 0 Disclaimer This information contained in this document is being supplied to you solely for your information and may not be copied, reproduced or further distributed to any person
Intercytex Group plc 0 Disclaimer This information contained in this document is being supplied to you solely for your information and may not be copied, reproduced or further distributed to any person
MIIG INJECTABLE Graft The following languages are included in this packet:
 MIIG INJECTABLE Graft 128801-10 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch)
MIIG INJECTABLE Graft 128801-10 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch)
Kuros Biosciences AG «A new era in Bone Regeneration»
 Kuros Biosciences AG «A new era in Bone Regeneration» A new era in Orthobiologics through unique Biologics & Clinically proven portfolio Ivan Cohen-Tanugi MD CEO Harry Welten - CFO DISCLAIMER: This document
Kuros Biosciences AG «A new era in Bone Regeneration» A new era in Orthobiologics through unique Biologics & Clinically proven portfolio Ivan Cohen-Tanugi MD CEO Harry Welten - CFO DISCLAIMER: This document
BAXTER UNVEILS DESIGN ENHANCEMENTS TO FLOSEAL AND TISSEEL HEMOSTATIC AGENTS AT AORN MEETING
 FOR IMMEDIATE RELEASE Media Contact Beth Mueller, (224) 948-5353 media@baxter.com Investor Contact Clare Trachtman, (224) 948-3085 BAXTER UNVEILS DESIGN ENHANCEMENTS TO FLOSEAL AND TISSEEL HEMOSTATIC AGENTS
FOR IMMEDIATE RELEASE Media Contact Beth Mueller, (224) 948-5353 media@baxter.com Investor Contact Clare Trachtman, (224) 948-3085 BAXTER UNVEILS DESIGN ENHANCEMENTS TO FLOSEAL AND TISSEEL HEMOSTATIC AGENTS
New creative textile construction using innovative braiding technology and materials
 White Paper New creative textile construction using innovative braiding technology and materials DSM and Meister Provide Foundation for Medical Device Design Philippe Gédet, Engineer, Meister & Cie AG
White Paper New creative textile construction using innovative braiding technology and materials DSM and Meister Provide Foundation for Medical Device Design Philippe Gédet, Engineer, Meister & Cie AG
strength of science Now with sizes up to 25 cm x 45 cm XCM BIOLOGIC Tissue Matrix
 strength of science XCM BIOLOGIC Tissue Matrix Now with sizes up to 25 cm x 45 cm Your trusted partner providing a complete portfolio of biologic meshes. DePuy Synthes Companies of Johnson & Johnson offer
strength of science XCM BIOLOGIC Tissue Matrix Now with sizes up to 25 cm x 45 cm Your trusted partner providing a complete portfolio of biologic meshes. DePuy Synthes Companies of Johnson & Johnson offer
Surgical Adhesives in Facial Plastic Surgery
 Surgical Adhesives in Facial Plastic Surgery Dean M. Toriumi, Victor K. Chung, Quintin M. Cappelle University of Illinois at Chicago Chicago, IL Supplement to Toriumi DM, Chung VK, Cappelle QM. Surgical
Surgical Adhesives in Facial Plastic Surgery Dean M. Toriumi, Victor K. Chung, Quintin M. Cappelle University of Illinois at Chicago Chicago, IL Supplement to Toriumi DM, Chung VK, Cappelle QM. Surgical
Regenerative Technology Investor Day. December 14, 2016 Plainsboro, NJ
 Regenerative Technology Investor Day December 14, 2016 Plainsboro, NJ Welcome Angela Steinway GLOBAL HEAD OF STRATEGIC INITIATIVES AND INVESTOR RELATIONS Safe Harbor Certain statements made in this presentation
Regenerative Technology Investor Day December 14, 2016 Plainsboro, NJ Welcome Angela Steinway GLOBAL HEAD OF STRATEGIC INITIATIVES AND INVESTOR RELATIONS Safe Harbor Certain statements made in this presentation
BUILT TO CONCENTRATE. Magellan is an autologous concentration system that delivers concentrated platelets and cells at the point of care.
 BUILT TO CONCENTRATE Magellan is an autologous concentration system that delivers concentrated platelets and cells at the point of care. DELIVER PERSONALIZED MEDICINE Every patient has a unique biology
BUILT TO CONCENTRATE Magellan is an autologous concentration system that delivers concentrated platelets and cells at the point of care. DELIVER PERSONALIZED MEDICINE Every patient has a unique biology
Ceramics, Cracow, Poland
 Chemically and physically functionalized carbon composites a prospective material for tissue treatment S.Blazewicz 1*, E.Stodolak 1, E. Staszkow 2, T.Mikolajczyk 3 1 AGH University of Science and Technology,
Chemically and physically functionalized carbon composites a prospective material for tissue treatment S.Blazewicz 1*, E.Stodolak 1, E. Staszkow 2, T.Mikolajczyk 3 1 AGH University of Science and Technology,
FORWARD-LOOKING STATEMENTS
 FORWARD-LOOKING STATEMENTS The statements made by Kensey Nash and its representatives in this presentation will include certain forward-looking statements, including financial forecasts, that are based
FORWARD-LOOKING STATEMENTS The statements made by Kensey Nash and its representatives in this presentation will include certain forward-looking statements, including financial forecasts, that are based
INUED DISCONTINUED DISCONTINUED DISCON MAKING THE IMPOSSIBLE POSSIBLE CENTER FOR REGENERATIVE MEDICINE
 INUED DISCONTINUED DISCONTINUED DISCON MAKING THE IMPOSSIBLE POSSIBLE CENTER FOR > SOLUTIONS AND HOPE Millions of people worldwide suffer from deadly diseases, chronic conditions and congenital disorders
INUED DISCONTINUED DISCONTINUED DISCON MAKING THE IMPOSSIBLE POSSIBLE CENTER FOR > SOLUTIONS AND HOPE Millions of people worldwide suffer from deadly diseases, chronic conditions and congenital disorders
Trabecular Metal Technology
 Trabecular Metal Technology Alexandre Nehme Departement of Orthopedic Surgery. Saint Georges University Hospital. Beirut. The cellular structure of Tantallum (Trabecular Metal) resembles bone and approximates
Trabecular Metal Technology Alexandre Nehme Departement of Orthopedic Surgery. Saint Georges University Hospital. Beirut. The cellular structure of Tantallum (Trabecular Metal) resembles bone and approximates
Skin Grafting. Biomaterials. Denis Suljendic Christian van der Pol
 Skin Grafting Biomaterials Denis Suljendic Christian van der Pol Overview Structure and function of skin History Reasons for a skin graft Traditional methods Modern solutions Future advancements 2 Structure
Skin Grafting Biomaterials Denis Suljendic Christian van der Pol Overview Structure and function of skin History Reasons for a skin graft Traditional methods Modern solutions Future advancements 2 Structure
Dressing Material Moisture facilitate
 Wound Healing Concept Dressing Material นพ.ส ทธ ส ทธ ส ณห ส ณห จ นทร ขจร ศ ลยศาสตร ตกแต ง โรงพยาบาลพระมงก ฎเกล า Moisture facilitate - Epidermal migration - Angiogenesis - Connective tissue synthesis -
Wound Healing Concept Dressing Material นพ.ส ทธ ส ทธ ส ณห ส ณห จ นทร ขจร ศ ลยศาสตร ตกแต ง โรงพยาบาลพระมงก ฎเกล า Moisture facilitate - Epidermal migration - Angiogenesis - Connective tissue synthesis -
Platelet Concentrate in Total Knees BIOLOGICS. This brochure is for International use only. It is not for distribution in the United States.
 Platelet Concentrate in Total Knees BIOLOGICS This brochure is for International use only. It is not for distribution in the United States. Platelet Concentrate in Total Knees Total Knee Arthroplasty Total
Platelet Concentrate in Total Knees BIOLOGICS This brochure is for International use only. It is not for distribution in the United States. Platelet Concentrate in Total Knees Total Knee Arthroplasty Total
VENTRAL HERNIA REPAIR SYSTEM
 VENTRAL HERNIA REPAIR SYSTEM Parietex Optimized Composite (x) Mesh and AbsorbaTack Fixation Device ECEPTIONAL COMBINATION MEDTRONIC HERNIA REPAIR SOLUTIONS Through innovative biomedical engineering, Medtronic
VENTRAL HERNIA REPAIR SYSTEM Parietex Optimized Composite (x) Mesh and AbsorbaTack Fixation Device ECEPTIONAL COMBINATION MEDTRONIC HERNIA REPAIR SOLUTIONS Through innovative biomedical engineering, Medtronic
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM INTRAPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted
INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM INTRAPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted
Deutsche Bank Health Care Conference
 Deutsche Bank Health Care Conference May 8, 2018 NASDAQ: ANIK0 Safe Harbor Statement The statements made in, and during the course of, this presentation that are not statements of historical fact, including
Deutsche Bank Health Care Conference May 8, 2018 NASDAQ: ANIK0 Safe Harbor Statement The statements made in, and during the course of, this presentation that are not statements of historical fact, including
BAXTER ANNOUNCES AGREEMENT TO BROADEN PORTFOLIO OF INNOVATIVE SURGICAL PRODUCTS
 FOR IMMEDIATE RELEASE Media Contact: Beth Mueller, (224) 948-5353 media@baxter.com Investor Contact: Clare Trachtman, (224) 948-3085 BAXTER ANNOUNCES AGREEMENT TO BROADEN PORTFOLIO OF INNOVATIVE SURGICAL
FOR IMMEDIATE RELEASE Media Contact: Beth Mueller, (224) 948-5353 media@baxter.com Investor Contact: Clare Trachtman, (224) 948-3085 BAXTER ANNOUNCES AGREEMENT TO BROADEN PORTFOLIO OF INNOVATIVE SURGICAL
NEXT GENERATION ECM-BASED ALLOGRAFT TECHNOLOGY:
 NEXT GENERATION ECM-BASED ALLOGRAFT TECHNOLOGY: Potent biological scaffolds strategically control stem cell fate and function, allowing our allografts to harness the regenerative potential of patient s
NEXT GENERATION ECM-BASED ALLOGRAFT TECHNOLOGY: Potent biological scaffolds strategically control stem cell fate and function, allowing our allografts to harness the regenerative potential of patient s
Translation of Biomaterial-based Therapies for the Treatment of Spinal Cord Injury: The Neuro-Spinal Scaffold and Bioengineered Neural Trails
 Translation of Biomaterial-based Therapies for the Treatment of Spinal Cord Injury: The Neuro-Spinal Scaffold and Bioengineered Neural Trails Alex A. Aimetti, PhD Sr. Director, Medical Education October
Translation of Biomaterial-based Therapies for the Treatment of Spinal Cord Injury: The Neuro-Spinal Scaffold and Bioengineered Neural Trails Alex A. Aimetti, PhD Sr. Director, Medical Education October
Meso BioMatrix Acellular Peritoneum Matrix RECOMMENDED TECHNIQUE GUIDE. Recreating harmonious bodies for a new vision of rebirth.
 Meso BioMatrix Acellular Peritoneum Matrix RECOMMENDED TECHNIQUE GUIDE Recreating harmonious bodies for a new vision of rebirth. 1 DISCOVER SEBBIN Sebbin is a 30 years-established company with a peerless
Meso BioMatrix Acellular Peritoneum Matrix RECOMMENDED TECHNIQUE GUIDE Recreating harmonious bodies for a new vision of rebirth. 1 DISCOVER SEBBIN Sebbin is a 30 years-established company with a peerless
PRINCIPLES AND PRACTICE OF TISSUE ENGNEERING:
 Harvard-MIT Division of Health Sciences and Technology HST.535: Principles and Practice of Tissue Engineering Instructor: Myron Spector Massachusetts Institute of Technology Harvard Medical School Brigham
Harvard-MIT Division of Health Sciences and Technology HST.535: Principles and Practice of Tissue Engineering Instructor: Myron Spector Massachusetts Institute of Technology Harvard Medical School Brigham
February 22, Dear Dr. Cox:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Integra LifeSciences Corp.
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Integra LifeSciences Corp.
Highly-ordered and hierarchical porosity scaffolds for nerve repair
 Highly-ordered and hierarchical porosity scaffolds for nerve repair J. SAKAMOTO 1 (PH. D.), D. LYNAM 1 (PH. D.), D. SHAHRIARI 1, K. KOFFLER 2 (PH. D.), P. WALTON (SC. D.) C. CHAN 1 (PH. D.), AND M.H. TUSZYNSKI
Highly-ordered and hierarchical porosity scaffolds for nerve repair J. SAKAMOTO 1 (PH. D.), D. LYNAM 1 (PH. D.), D. SHAHRIARI 1, K. KOFFLER 2 (PH. D.), P. WALTON (SC. D.) C. CHAN 1 (PH. D.), AND M.H. TUSZYNSKI
MEDPOR. Orbital Floor
 MEDPOR Orbital Floor Orbital Fracture Repair Available in a variety of shapes, sizes, and configurations, MEDPOR and MEDPOR TITAN Implants provide surgeons with an expanding range of options for reconstruction
MEDPOR Orbital Floor Orbital Fracture Repair Available in a variety of shapes, sizes, and configurations, MEDPOR and MEDPOR TITAN Implants provide surgeons with an expanding range of options for reconstruction
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: B I O M A T E R I A L en English INSTRUCTIONS FOR USE FOR GORE SYNECOR INTRAPERITONEAL BIOMATERIAL INDICATIONS The GORE SYNECOR Intraperitoneal Biomaterial device is intended
INSTRUCTIONS FOR USE FOR: B I O M A T E R I A L en English INSTRUCTIONS FOR USE FOR GORE SYNECOR INTRAPERITONEAL BIOMATERIAL INDICATIONS The GORE SYNECOR Intraperitoneal Biomaterial device is intended
ADVANCED WOUND CARE MANAGEMENT
 POREX, FILTRATION GROUP ADVANCED WOUND CARE MANAGEMENT Innovative medical grade foams for acute and chronic wounds POREX Wound Care Foams The ideal wound care solution Our high performance medical grade
POREX, FILTRATION GROUP ADVANCED WOUND CARE MANAGEMENT Innovative medical grade foams for acute and chronic wounds POREX Wound Care Foams The ideal wound care solution Our high performance medical grade
CO 2 Laser Fascia to Dura Soldering for Pig Dural Defect Reconstruction
 CO 2 Laser Fascia to Dura Soldering for Pig Dural Defect Reconstruction Boaz Forer, M.D., 1 TamarVasileyev, 2 Ziv Gil, M.D., Ph.D., 1 Tamar Brosh, 3 Naam Kariv, 4 Abraham Katzir, 2 and Dan M. Fliss, M.D.
CO 2 Laser Fascia to Dura Soldering for Pig Dural Defect Reconstruction Boaz Forer, M.D., 1 TamarVasileyev, 2 Ziv Gil, M.D., Ph.D., 1 Tamar Brosh, 3 Naam Kariv, 4 Abraham Katzir, 2 and Dan M. Fliss, M.D.
Introduction to Cell/ Biomaterial Engineering
 Introduction to Cell/ Biomaterial Engineering Module 3, Lecture 1! 20.109 Spring 2011! Topics for Lecture 1 Introduction to tissue engineering! motivation! basic principles + examples! Introduction to
Introduction to Cell/ Biomaterial Engineering Module 3, Lecture 1! 20.109 Spring 2011! Topics for Lecture 1 Introduction to tissue engineering! motivation! basic principles + examples! Introduction to
CHAPTER 4 CONCLUSIONS AND FUTURE PERSPECTIVES
 CHAPTER 4 CONCLUSIONS AND FUTURE PERSPECTIVES Wound infections are the major challenges in the wound care management. Infection can be either due to microbial attack or due to some diseases. This infection
CHAPTER 4 CONCLUSIONS AND FUTURE PERSPECTIVES Wound infections are the major challenges in the wound care management. Infection can be either due to microbial attack or due to some diseases. This infection
A Legacy of Serving the Surgical Community
 Precisely. A Legacy of Serving the Surgical Community For over half a century, Stryker has been developing products based on the expressed needs of leading practitioners. You will often find a Stryker
Precisely. A Legacy of Serving the Surgical Community For over half a century, Stryker has been developing products based on the expressed needs of leading practitioners. You will often find a Stryker
BIOFIBER BIOFIBER CM AMBIENT TEMPERATUR. Absorbable Scaffold. Collagen Coated. Absorbable Scaffold
 BIOFIBER Absorbable Scaffold BIOFIBER CM Collagen Coated Absorbable Scaffold A B S O R B A B L E AMBIENT TEMPERATUR B I O L O G I C S C AAND F F OCRYOPRESERVE L D I N G S OSTORAGE L U T I O N S OPTION
BIOFIBER Absorbable Scaffold BIOFIBER CM Collagen Coated Absorbable Scaffold A B S O R B A B L E AMBIENT TEMPERATUR B I O L O G I C S C AAND F F OCRYOPRESERVE L D I N G S OSTORAGE L U T I O N S OPTION
Tornier Medical Education
 Tornier Medical Education D E C E M B E R 7, 2 0 0 9 Orthopaedic Uses of Tissue Matrices in Tendon Repair: Why All Tissue Matrices are Not Created Equal John R. Harper, Ph.D, Research Department, LifeCell
Tornier Medical Education D E C E M B E R 7, 2 0 0 9 Orthopaedic Uses of Tissue Matrices in Tendon Repair: Why All Tissue Matrices are Not Created Equal John R. Harper, Ph.D, Research Department, LifeCell
CapSure Permanent Fixation System
 CapSure Permanent Fixation System Permanent Fixation Redefined Advancing the Fixation Experience Recipient of 2015 SLS Innovations of the Year recognition. SOFT TISSUE REPAIR Right Procedure. Right Product.
CapSure Permanent Fixation System Permanent Fixation Redefined Advancing the Fixation Experience Recipient of 2015 SLS Innovations of the Year recognition. SOFT TISSUE REPAIR Right Procedure. Right Product.
Anika Therapeutics, Inc.
 Anika Therapeutics, Inc. Canaccord Genuity 34th Annual Growth Conference August 14 th, 2014 Safe Harbor Statement The statements made in this presentation, which are not statements of historical fact,
Anika Therapeutics, Inc. Canaccord Genuity 34th Annual Growth Conference August 14 th, 2014 Safe Harbor Statement The statements made in this presentation, which are not statements of historical fact,
Regenerative Strategies for Vascular and Lung Tissues. Laura E Niklason MD, PhD
 Regenerative Strategies for Vascular and Lung Tissues Laura E Niklason MD, PhD Disclosure: Some of this work in this presentation is from Humacyte Inc. Niklason is a founder of Humacyte, and holds stock
Regenerative Strategies for Vascular and Lung Tissues Laura E Niklason MD, PhD Disclosure: Some of this work in this presentation is from Humacyte Inc. Niklason is a founder of Humacyte, and holds stock
Harvesting a New Standard of Care
 Harvesting a New Standard of Care The handoff. Be confident what you re passing along is of the highest quality. What do you need to deliver the optimal conduit? Is it knowing that you controlled hemostasis?
Harvesting a New Standard of Care The handoff. Be confident what you re passing along is of the highest quality. What do you need to deliver the optimal conduit? Is it knowing that you controlled hemostasis?
CUSA EXcel + Ultrasonic Aspirator tender package
 CUSA EXcel + Ultrasonic Aspirator tender package Limits uncertainty with Selectivity and Power CUSA EXcel + Table Of Contents 1 Integra, the company...03 2 CUSA EXcel + ultrasonic aspirators applications...03
CUSA EXcel + Ultrasonic Aspirator tender package Limits uncertainty with Selectivity and Power CUSA EXcel + Table Of Contents 1 Integra, the company...03 2 CUSA EXcel + ultrasonic aspirators applications...03
Processed from Donated Human Tissue by LifeCell Corporation for KCI USA, Inc.
 for wounds instructions for use Processed from Donated Human Tissue by LifeCell Corporation for KCI USA, Inc. marketed by KCI USA, Inc. San Antonio, TX 78219 USA 800.275.4524 www.kci1.com www.graftjacketbykci.com
for wounds instructions for use Processed from Donated Human Tissue by LifeCell Corporation for KCI USA, Inc. marketed by KCI USA, Inc. San Antonio, TX 78219 USA 800.275.4524 www.kci1.com www.graftjacketbykci.com
D-Stat Dry Topical Hemostat Proven to Reduce Time-to-Hemostasis 1,2
 D-Stat Dry Topical Hemostat Proven to Reduce Time-to-Hemostasis 1,2 Clinical Data to Support a True Hemostat Results are from a 376-patient, five-center, prospective randomized U.S. clinical study evaluating
D-Stat Dry Topical Hemostat Proven to Reduce Time-to-Hemostasis 1,2 Clinical Data to Support a True Hemostat Results are from a 376-patient, five-center, prospective randomized U.S. clinical study evaluating
Biocompatibility and War and Peace
 Professor David Williams, D.Sc.,F.R.Eng., Professor and Director of International Affairs, Wake Forest Institute of Regenerative Medicine, North Carolina, USA Editor-in-Chief, Biomaterials President-elect,
Professor David Williams, D.Sc.,F.R.Eng., Professor and Director of International Affairs, Wake Forest Institute of Regenerative Medicine, North Carolina, USA Editor-in-Chief, Biomaterials President-elect,
Equity. The Finest Equine Pericardium Matrix. 0,4 mm the thinnest
 Equity The Finest Equine Pericardium Matrix 0,4 mm the thinnest Equity why Equity The evolution of living organisms has selected highl performance animal tissues. From the Eocene, 50 million years ago,
Equity The Finest Equine Pericardium Matrix 0,4 mm the thinnest Equity why Equity The evolution of living organisms has selected highl performance animal tissues. From the Eocene, 50 million years ago,
THE SCIENCE OF PLATELET RICH PLASMA
 THE SCIENCE OF PLATELET RICH PLASMA Cheri A. Ong, MD FACS ONG INSTITUTE FOR PLASTIC SURGERY & HEALTH WHAT IS PRP BLOOD PLASMA 55% of the body s today body volume 45% blood cells:- Red blood cells, White
THE SCIENCE OF PLATELET RICH PLASMA Cheri A. Ong, MD FACS ONG INSTITUTE FOR PLASTIC SURGERY & HEALTH WHAT IS PRP BLOOD PLASMA 55% of the body s today body volume 45% blood cells:- Red blood cells, White
Adipose rabbit mesenchymal stem cells for the treatment of the chronic scar tissue of the vocal cords
 Adipose rabbit mesenchymal stem cells for the treatment of the chronic scar tissue of the vocal cords Dr. Vasiliki E Kalodimou, Head of Flow Cytometry-Research and Regenerative Medicine Department, IASO-Maternity
Adipose rabbit mesenchymal stem cells for the treatment of the chronic scar tissue of the vocal cords Dr. Vasiliki E Kalodimou, Head of Flow Cytometry-Research and Regenerative Medicine Department, IASO-Maternity
Bone Marrow Concentration. For customized cellular concentrations of platelet-rich plasma from bone marrow aspirate
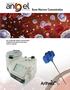 ARTHREX TM SYSTEM Bone Marrow Concentration For customized cellular concentrations of platelet-rich plasma from bone marrow aspirate Arthrex Angel System Technology is what sets Angel apart from the competition.
ARTHREX TM SYSTEM Bone Marrow Concentration For customized cellular concentrations of platelet-rich plasma from bone marrow aspirate Arthrex Angel System Technology is what sets Angel apart from the competition.
COATING SYSTEMS BIOLINE COATING
 COATING SYSTEMS BIOLINE COATING BIOLINE COATING MIMICKING HUMAN TISSUE MAQUET THE GOLD STANDARD MAQUET is an international synonym for innovative and technological advances in operating rooms and intensive
COATING SYSTEMS BIOLINE COATING BIOLINE COATING MIMICKING HUMAN TISSUE MAQUET THE GOLD STANDARD MAQUET is an international synonym for innovative and technological advances in operating rooms and intensive
Cellular Matrix HYDRATION & REGENERATION. Autologous Platelet Rich Plasma combined with Hyaluronic Acid for Skin Regeneration
 Cellular Matrix HYDRATION & REGENERATION Autologous Platelet Rich Plasma combined with Hyaluronic Acid for Skin Regeneration A-PRP & HA Autologous Platelet Rich Plasma Hyaluronic Acid Cellular Matrix is
Cellular Matrix HYDRATION & REGENERATION Autologous Platelet Rich Plasma combined with Hyaluronic Acid for Skin Regeneration A-PRP & HA Autologous Platelet Rich Plasma Hyaluronic Acid Cellular Matrix is
Force Fiber Sutures and Braids
 Force Fiber s and Braids Force Fiber Fusion page 2 Force Fiber OrthoTape Braid page 4 Force Fiber page 6 Work With The Experts 1 Teleflex Medical OEM Force Fiber Fusion PRESSURE DISTRIBUTION FiberTape
Force Fiber s and Braids Force Fiber Fusion page 2 Force Fiber OrthoTape Braid page 4 Force Fiber page 6 Work With The Experts 1 Teleflex Medical OEM Force Fiber Fusion PRESSURE DISTRIBUTION FiberTape
Project Proposal Ultrasound Mediated Tissue Engineering Project Team 21
 Project Proposal Ultrasound Mediated Tissue Engineering Project Team 21 Nicholas Calistri, Aimon Iftikhar, Yvonne Nanakos and Kelly Stratton Project 40 for Dr. Yusuf Khan University of Connecticut Health
Project Proposal Ultrasound Mediated Tissue Engineering Project Team 21 Nicholas Calistri, Aimon Iftikhar, Yvonne Nanakos and Kelly Stratton Project 40 for Dr. Yusuf Khan University of Connecticut Health
