Temperature mapping and autoclave validation specialists
|
|
|
- Whitney Johnston
- 6 years ago
- Views:
Transcription
1 Temperature mapping and autoclave validation specialists
2 Who are we? We are Thermal Compliance, specialists in temperature mapping and autoclave validation. Founded in 2007, we are an independent validation service provider to the pharmaceutical, biotechnology and healthcare sectors internationally. We provide validation, calibration, equipment hire and temperature mapping services. Our autoclave validation services include: EN285 testing HTM2010 testing installation qualification operational qualification cycle development performance qualification troubleshooting. 2 Our temperature mapping and validation services are used for equipment and environments such as: fridges freezers incubators ovens warehouses storage rooms humidity cabinets stability rooms. We have a wide range of equipment available to hire including Kaye Validator 2000s and thermal testing equipment. We also offer products including the 3M Electronic Test System, biological indicators, thermocouples, and autoclave tape.
3 Temperature mapping We provide temperature mapping, thermal validation and humidity mapping services for temperature critical equipment. We perform testing over a range of -196 to 400 C, and 0 to 95% RH. We use GE Kaye Validator 2000 and wireless data loggers capable of logging every second to give a detailed analysis of your temperature critical environment, performing work to either our Standard Operating Procedures (SOPs) and protocols, or your own documentation. We also offer onsite calibration services for temperature and pressure. We perform temperature mapping on various healthcare, process, laboratory and storage equipment, including: Laboratory: incubators; ovens; humidity chambers; autoclaves Process: freeze driers; steam-inplace; depyrogenation tunnels; autoclaves Product storage: fridges; freezers; cold stores; warehouses; liquid nitrogen tanks; stability rooms Our knowledge of US and EU industry guidelines such as the Orange guide, ICH guidelines and ASTM E2500 is key to providing you with a clear and compliant validation approach. 3
4 Autoclave validation At Thermal Compliance we specialise in autoclave validation to UK, EU and international standards for the pharmaceutical, biotechnology and healthcare sectors. Our experience and expertise will give you a clear, compliant and transparent autoclave validation package. Awareness of current EU and US regulatory requirements is essential. We can qualify your autoclave to EN 285:2006, EN and PDA Technical Report 01 to meet MHRA, IMB and FDA expectations. Validation Services: Installation/Operation Qualification (IQ, OQ) Cycle/Load Development (CD) Performance Qualification (PQ) and Re-Qualification (RQ) EN 285:2006 testing Pressure and temperature air detector setup Audits and consultancy support Steam quality testing HTM testing HTM 2010 testing. 4
5 Equipment hire Our GE Kaye Validator thermal validation systems are available for hire with current calibration certificates. We also offer thermal baths from -80 C to 400 C and IRTD-400s. There is no minimum hire period; all of our equipment is delivered in protective cases with current calibration certificates; additional validation accessories are also available. Items for hire include: Kaye Validator 2000 (versions 3.6, 3.5, 3.1, 2x) including 3 Sensor Input Modules (SIMS), power lead, serial cable, USB cable, and calibration certificates Kaye LTR-140 (-40), temperature range -40 C to 140 C Kaye LTR-140 (-25), temperature range -25 C to 140 C Kaye M2801 / IRTD-400, temperature range -196 C to 400 C Kaye HTR-400, temperature range 50 C to 400 C, accuracy ±0.2 C to ±0.4 C. Other equipment that we have available to hire includes: entry glands, pressure transmitters and temperature and humidity dataloggers. 5
6 Products At Thermal Compliance, our approach is to identify and embrace forward-thinking solutions. We find innovative solutions to improve performance through timesaving, and by increasing quality of data and ease of use. Our products include: 3M Electronic Test Systems (ETS) We distribute the 3M Electronic Test System (ETS) a paper free electronic Bowie & Dick Test. We provide validation support to assist your move to a paperless system. ETS provides comprehensive, objective, independent information about the critical variables of the steam sterilization process. 6
7 Biological indicators 3M Attest Biological Indicators for Ethylene Oxide (EtO) Sterilization. 3M Attest Rapid Readout Biological Indicator for Steam. 3M Attest Biological Indicators for Steam Sterilization. Autoclave tape 3M Comply Steam Indicator Tape. Individually wrapped. 3M Comply Ethylene Oxide Indicator Tape. Individually wrapped. Standard Towel Packs For use in HTM2010 & EN285 Testing. Bespoke Thermocouples Bespoke for use with Kaye Validator Class 1 (Premium) Type T cable Colour coded to IEC FEP Encapsulation & Welded Tips Numbered. 7
8 Contact us: Thermal Compliance Limited The Granary Streatlam Home Farm County Durham DL12 8TZ UK Call +44 (0) You can also find us online: Thermal Compliance Limited Thermal Compliance Limited is a company registered in England. Registered number: Registered address: The Granary, Streatlam Home Farm, Barnard Castle, County Durham, DL12 8TZ. VAT Number: GB
TMI-Orion Pharmaceutical Medical
 INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
TMI-Orion Pharmaceutical Medical
 INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
TMI-Orion Pharmaceutical Medical
 INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
TMI-Orion Pharmaceutical Medical
 INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
INDUSTRY FLYER TMI-Orion Pharmaceutical Medical HIGH TECH DATA LOGGING SOLUTIONS www.tmi-orion.com TMI-Orion Pharmaceutical Medical TMI-Orion presents a full range of high tech data loggers designed for
Validator Accessories Kaye Product Line. Amphenol Advanced Sensors
 Validator Accessories Kaye Product Line Amphenol Advanced Sensors Dear valued customer, Type T thermocouples are most accurate in the temperature range of -200 to 400 C, providing more accurate, repeatable
Validator Accessories Kaye Product Line Amphenol Advanced Sensors Dear valued customer, Type T thermocouples are most accurate in the temperature range of -200 to 400 C, providing more accurate, repeatable
Thermal Validation Solutions. TrackSense Pro. Pro. The ultimate solution in wireless thermal validation
 TrackSense Pro TrackSense Pro The ultimate solution in wireless thermal validation Ultimate Solution in Wireless Thermal Validation Ellab has incorporated the latest in electronic technology and innovation
TrackSense Pro TrackSense Pro The ultimate solution in wireless thermal validation Ultimate Solution in Wireless Thermal Validation Ellab has incorporated the latest in electronic technology and innovation
Calibration, Qualification and Preventive Maintenance Services Overview
 , Qualification and Preventive Maintenance Services Overview PVSR Corporation, recently certified as an ISO 9001 organization is emerging as an innovator providing the Pharmaceutical, Medical Devices and
, Qualification and Preventive Maintenance Services Overview PVSR Corporation, recently certified as an ISO 9001 organization is emerging as an innovator providing the Pharmaceutical, Medical Devices and
Wireless Data Logger System
 Wireless Data Logger System for Thermal Process Validation Medical / CSSD / Pharmaceutical / Food Flexible Data Logger System For Thermal Validations ebro offers a flexible measurement and documentation
Wireless Data Logger System for Thermal Process Validation Medical / CSSD / Pharmaceutical / Food Flexible Data Logger System For Thermal Validations ebro offers a flexible measurement and documentation
EBI 12 The new data logger generation
 EBI 12 The new data logger generation 3 EBI 12 The new data logger generation High quality stainless steel housing Application range from -90 C to 150 C High temperature accuracy up to 0.05 C Extended
EBI 12 The new data logger generation 3 EBI 12 The new data logger generation High quality stainless steel housing Application range from -90 C to 150 C High temperature accuracy up to 0.05 C Extended
Data Logger Systems. Medical CSSD Pharmaceutical Food
 Data Logger Systems for Thermal Process Validation Medical CSSD Pharmaceutical Food Flexible Data Logger Systems For Thermal Validations ebro offers flexible measurement and documentation systems for validating
Data Logger Systems for Thermal Process Validation Medical CSSD Pharmaceutical Food Flexible Data Logger Systems For Thermal Validations ebro offers flexible measurement and documentation systems for validating
SPECIALIZING IN: Validation Calibration Preventive Maintenance Installation EQUIPMENT AND INSTRUMENT SERVICES
 SPECIALIZING IN: Validation Calibration Preventive Maintenance Installation EQUIPMENT AND INSTRUMENT SERVICES VWRCATALYST helps you simplify equipment and instrument services, in and around the laboratory.
SPECIALIZING IN: Validation Calibration Preventive Maintenance Installation EQUIPMENT AND INSTRUMENT SERVICES VWRCATALYST helps you simplify equipment and instrument services, in and around the laboratory.
THERMAL AND RELATIVE HUMIDITY MAPPING OF A SAMPLING ROOM
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Gangadharappa et al. SJIF Impact Factor 6.041 Volume 5, Issue 4, 1563-1572 Research Article ISSN 2278 4357 THERMAL AND RELATIVE HUMIDITY MAPPING OF
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Gangadharappa et al. SJIF Impact Factor 6.041 Volume 5, Issue 4, 1563-1572 Research Article ISSN 2278 4357 THERMAL AND RELATIVE HUMIDITY MAPPING OF
Kaye Product Line Calibration and Maintenance Service Solutions Rental
 Kaye Product Line Calibration and Maintenance Service Solutions Rental Amphenol Advanced Sensors EUROPE Value-added services that enhance your productivity Your Validation and Monitoring equipment is
Kaye Product Line Calibration and Maintenance Service Solutions Rental Amphenol Advanced Sensors EUROPE Value-added services that enhance your productivity Your Validation and Monitoring equipment is
Validation Master Plan
 Master Plan TITLE: SITE VALIDATION MASTER PLAN 2001 Quality Assurance Document No. XXXXX Rev.no. Page 1 of 15 Subject of Latest Revision: - CONTENTS 1. INTRODUCTION 2. OBJECTIVE 3. DEFINITIONS 4. SCOPE
Master Plan TITLE: SITE VALIDATION MASTER PLAN 2001 Quality Assurance Document No. XXXXX Rev.no. Page 1 of 15 Subject of Latest Revision: - CONTENTS 1. INTRODUCTION 2. OBJECTIVE 3. DEFINITIONS 4. SCOPE
HISTORY AND MILESTONES
 HISTORY AND MILESTONES HISTORY AND MILESTONES DOC S.r.l., Documentation Organization & Consultancy, was established in 1997 by MASCO group C.E.O. Eng. Alberto Borella and Eng. Paolo Curtò who became Managing
HISTORY AND MILESTONES HISTORY AND MILESTONES DOC S.r.l., Documentation Organization & Consultancy, was established in 1997 by MASCO group C.E.O. Eng. Alberto Borella and Eng. Paolo Curtò who became Managing
Thermal Validation Solutions. TrackSense Pro. Pro. The ultimate solution in wireless thermal validation
 Thermal Validation Solutions TrackSense Pro TrackSense Pro The ultimate solution in wireless thermal validation The Ultimate Solution in Wireless Thermal Validation TrackSense Pro Ellab has incorporated
Thermal Validation Solutions TrackSense Pro TrackSense Pro The ultimate solution in wireless thermal validation The Ultimate Solution in Wireless Thermal Validation TrackSense Pro Ellab has incorporated
ebro Validation-Set for large Steam Sterilizers SL 2100 The Set contains:
 ebro Validation-Set for large Steam Sterilizers SL 2100 The Set contains: 1 x Pressure-Temperature Data Logger EBI 10-TP321 5 x Temperature Data Logger EBI 10-T441 1 x Silicone Protection Box AL 101 5
ebro Validation-Set for large Steam Sterilizers SL 2100 The Set contains: 1 x Pressure-Temperature Data Logger EBI 10-TP321 5 x Temperature Data Logger EBI 10-T441 1 x Silicone Protection Box AL 101 5
Steam Quality Testing
 Steam Quality Testing Take the easy route to compliance Assured and compliant results from the outset To qualify plant/utility/clean/pure steam generators, steam distribution systems and steam supplies
Steam Quality Testing Take the easy route to compliance Assured and compliant results from the outset To qualify plant/utility/clean/pure steam generators, steam distribution systems and steam supplies
Innovative. to the pharmaceutical, biotechnology and advanced technology. and equipment. Our learning management
 Innovative S o l u t i o n s Pharma-Tech Process and Facilities Services is an industry leader in providing innovative technical consulting and staff-augmentation services to the pharmaceutical, biotechnology
Innovative S o l u t i o n s Pharma-Tech Process and Facilities Services is an industry leader in providing innovative technical consulting and staff-augmentation services to the pharmaceutical, biotechnology
Validation and Complete Routine Control Sets
 Validation and Routine Control Sets Complete Validation Sets Complete Routine Control Sets Description: Validation data loggers with evaluation software and an extensive range of accessories German TÜV
Validation and Routine Control Sets Complete Validation Sets Complete Routine Control Sets Description: Validation data loggers with evaluation software and an extensive range of accessories German TÜV
Vaisala viewlinc Monitoring System Services
 Vaisala viewlinc Monitoring System Services Integrated Service for Maximum Compliance, Safety and Performance Vaisala viewlinc Life Cycle Maintenance Agreement Software Maintenance Technical Support Training
Vaisala viewlinc Monitoring System Services Integrated Service for Maximum Compliance, Safety and Performance Vaisala viewlinc Life Cycle Maintenance Agreement Software Maintenance Technical Support Training
Data loggers and software solutions to monitor and control your industrial processes
 www.tmi-orion.com VALIDATE, MONITOR, CONTROL YOUR PROCESSES Validate, monitor, control your industrial processes TMI-Orion has acquired since the introduction of the first pressure and temperature data
www.tmi-orion.com VALIDATE, MONITOR, CONTROL YOUR PROCESSES Validate, monitor, control your industrial processes TMI-Orion has acquired since the introduction of the first pressure and temperature data
Specialists in autoclave, sterilizer and decontamination equipment
 E S T S European Sterilizer & Technical Services Specialists in autoclave, sterilizer and decontamination equipment Portfolio Our portfolio of clients comes from the following industry sectors: Hospitals
E S T S European Sterilizer & Technical Services Specialists in autoclave, sterilizer and decontamination equipment Portfolio Our portfolio of clients comes from the following industry sectors: Hospitals
AQUALAB 4TE, 4TEV, 4TE DUO, 4TEV DUO BENCHTOP WATER ACTIVITY
 AQUALAB 4TE, 4TEV, 4TE DUO, 4TEV DUO BENCHTOP WATER ACTIVITY Installation Qualification + Operational Qualification Protocols and Instructions TABLE OF CONTENTS 1. Introduction.. 3 2. Installation Qualification
AQUALAB 4TE, 4TEV, 4TE DUO, 4TEV DUO BENCHTOP WATER ACTIVITY Installation Qualification + Operational Qualification Protocols and Instructions TABLE OF CONTENTS 1. Introduction.. 3 2. Installation Qualification
STEAM QUALITY TEST KIT SQ1
 STEAM QUALITY TEST KIT SQ1 Superheat Test Dryness Test Non-Condensable Gas Test ( 주 ) 바이오시스텍 www.biosystech.com Tel. 031 898 8870 Fax. 031 898 8871 446-916 경기도용인시기흥구동백중앙로 16번길 16-4 에이스동백타워 1동 1708호 Steam
STEAM QUALITY TEST KIT SQ1 Superheat Test Dryness Test Non-Condensable Gas Test ( 주 ) 바이오시스텍 www.biosystech.com Tel. 031 898 8870 Fax. 031 898 8871 446-916 경기도용인시기흥구동백중앙로 16번길 16-4 에이스동백타워 1동 1708호 Steam
Introduction and Background
 Introduction and Background Validation of Biotech Facilities Pacific Biotech Alliance Robert J. Mackey Pacific Biotech Alliance is an established consulting firm in the Pacific Northwest that specializes
Introduction and Background Validation of Biotech Facilities Pacific Biotech Alliance Robert J. Mackey Pacific Biotech Alliance is an established consulting firm in the Pacific Northwest that specializes
Contents. Contents (13) 1 Production (23)
 1 Production (23) 1.A Sanitation (27) 1.A.1 Organisational prerequisites (27) 1.A.2 Sources of contamination (28) 1.A.3 Responsibilities and implementation (29) 1.B Personnel hygiene (31) 1.B.1 Clothing
1 Production (23) 1.A Sanitation (27) 1.A.1 Organisational prerequisites (27) 1.A.2 Sources of contamination (28) 1.A.3 Responsibilities and implementation (29) 1.B Personnel hygiene (31) 1.B.1 Clothing
Mason Technology. Service Solutions. The Largest Irish Based Scientific Service Solution Provider
 Mason Technology Service Solutions The Largest Irish Based Scientific Service Solution Provider Are we the largest Scientific Service Solution Provider in Ireland? Our numbers prove it. Dedicated team
Mason Technology Service Solutions The Largest Irish Based Scientific Service Solution Provider Are we the largest Scientific Service Solution Provider in Ireland? Our numbers prove it. Dedicated team
Professional Data Logger
 Professional Data Logger FOR VALIDATION, ROUTINE CONTROL AND PROCESS MONITORING MEDICINE FOOD PHARMACEUTICAL LABORATORY 2018 50 years of ebro Freiburg 01.04.1968. The Breisgau-founded company ebro Electronic
Professional Data Logger FOR VALIDATION, ROUTINE CONTROL AND PROCESS MONITORING MEDICINE FOOD PHARMACEUTICAL LABORATORY 2018 50 years of ebro Freiburg 01.04.1968. The Breisgau-founded company ebro Electronic
Validation Strategies for Equipment from Multiple Vendors
 David Heiger, Ph.D. Worldwide Compliance & NDS Services Manager 10. December 2002 Validation Strategies for Equipment from Multiple Vendors Time: 3.00 pm Central European Time Telephone Number: +44 20
David Heiger, Ph.D. Worldwide Compliance & NDS Services Manager 10. December 2002 Validation Strategies for Equipment from Multiple Vendors Time: 3.00 pm Central European Time Telephone Number: +44 20
Medical and Pharmaceutical
 Medical and Pharmaceutical Data Logging Solutions Sterilization & Facilities Monitoring Simplifying How the World Measures & Records Data MadgeTech, Inc. is a global company, based in New England and founded
Medical and Pharmaceutical Data Logging Solutions Sterilization & Facilities Monitoring Simplifying How the World Measures & Records Data MadgeTech, Inc. is a global company, based in New England and founded
Medical and Pharmaceutical
 Medical and Pharmaceutical Data Logging Solutions Sterilization & Facilities Monitoring Simplifying How the World Measures & Records Data MadgeTech, Inc. is a global company, based in New England and founded
Medical and Pharmaceutical Data Logging Solutions Sterilization & Facilities Monitoring Simplifying How the World Measures & Records Data MadgeTech, Inc. is a global company, based in New England and founded
A guideline to good manufacturing practice (GMP) requirements
 A guideline to good manufacturing practice (GMP) requirements Part 2: Validation Get all guidelines on www.pharmaguideline.com Email- info@pharmaguideline.com Page 1 of 96 1. Abbreviations EP: GMP: MF:
A guideline to good manufacturing practice (GMP) requirements Part 2: Validation Get all guidelines on www.pharmaguideline.com Email- info@pharmaguideline.com Page 1 of 96 1. Abbreviations EP: GMP: MF:
Managing Validation. Paperless Recorders
 Managing Validation Paperless Recorders Multitrend Plus Validation Background The past five years has seen an increase in the use of computerized systems and products in the pharmaceutical and bio-pharmaceutical
Managing Validation Paperless Recorders Multitrend Plus Validation Background The past five years has seen an increase in the use of computerized systems and products in the pharmaceutical and bio-pharmaceutical
FITOCLIMA WALK-IN STABILITY
 FITOCLIMA WALK-IN TEMPERATURE AND HUMIDITY CHAMBERS ICH, GMP, WHO, FDA COMPLIANT Common applications include: Pharmaceuticals Food and Beverages Cosmetics Veterinary Storage and Conservation Quality control
FITOCLIMA WALK-IN TEMPERATURE AND HUMIDITY CHAMBERS ICH, GMP, WHO, FDA COMPLIANT Common applications include: Pharmaceuticals Food and Beverages Cosmetics Veterinary Storage and Conservation Quality control
FITOCLIMA WALK-IN STABILITY
 FITOCLIMA WALK-IN TEMPERATURE AND HUMIDITY ROOMS ICH, GMP, WHO, FDA COMPLIANT FITOCLIMA WALK-IN CHAMBERS Common applications include: ICH Stability Testing Pharmaceuticals Food and Beverages Cosmetics
FITOCLIMA WALK-IN TEMPERATURE AND HUMIDITY ROOMS ICH, GMP, WHO, FDA COMPLIANT FITOCLIMA WALK-IN CHAMBERS Common applications include: ICH Stability Testing Pharmaceuticals Food and Beverages Cosmetics
All measurement data. Always available. On any device. The data logger system testo Saveris 2. Analysis & Reports. Graphical presentation
 Analysis & Reports Coldstore east 14.09.2014 to 16.09.2014 Graphical presentation 14/09/2014 15/09/2014 16/09/2014 Data logger 1 ( C) Data logger 2 ( C) Data logger 3 ( C) Tabular presentation Alarms All
Analysis & Reports Coldstore east 14.09.2014 to 16.09.2014 Graphical presentation 14/09/2014 15/09/2014 16/09/2014 Data logger 1 ( C) Data logger 2 ( C) Data logger 3 ( C) Tabular presentation Alarms All
The Significance of Validation to US CSSD Managers
 The Significance of Validation to US CSSD Managers Jonathan A. Wilder, Ph.D. President, H & W Technology, LLC/Stericert Co. Managing Director, Quality Processing Resource Group, LLC 1 Introduction This
The Significance of Validation to US CSSD Managers Jonathan A. Wilder, Ph.D. President, H & W Technology, LLC/Stericert Co. Managing Director, Quality Processing Resource Group, LLC 1 Introduction This
Could Poor Temperature Data Management be Putting Your GxP Facility at Risk for Data Integrity Violations? we prove it.
 1 How to Meet New MHRA, FDA and WHO Data Integrity Guidelines WHITE PAPER Could Poor Temperature Data Management be Putting Your GxP Facility at Risk for Data Integrity Violations? we prove it. 2 How to
1 How to Meet New MHRA, FDA and WHO Data Integrity Guidelines WHITE PAPER Could Poor Temperature Data Management be Putting Your GxP Facility at Risk for Data Integrity Violations? we prove it. 2 How to
Installation Qualification/Operational Qualification Protocols and Instructions
 Installation Qualification/Operational Qualification Protocols and Instructions AquaLab PRE Decagon Devices, Inc July 20, 2016 4:05 PM 1 Table of Contents Section 1 Section 2 Section 2.1 Section 2.2 Section
Installation Qualification/Operational Qualification Protocols and Instructions AquaLab PRE Decagon Devices, Inc July 20, 2016 4:05 PM 1 Table of Contents Section 1 Section 2 Section 2.1 Section 2.2 Section
Steam Quality Test Kit
 Steam Quality Test Kit Page 1 The Steam Quality Test Kit is equipped with all the component parts that are necessary to perform the required tests and report the results in accordance with ISO EN 285,
Steam Quality Test Kit Page 1 The Steam Quality Test Kit is equipped with all the component parts that are necessary to perform the required tests and report the results in accordance with ISO EN 285,
Vaisala Life Science Services Integrated Service for Maximum Compliance, Safety and Performance LIFE SCIENCE SOLUTIONS FOR REAL LIFE
 Vaisala Life Science Services Integrated Service for Maximum Compliance, Safety and Performance LIFE SCIENCE SOLUTIONS FOR REAL LIFE Get Your Controlled Environment Under Control Simplified Compliance
Vaisala Life Science Services Integrated Service for Maximum Compliance, Safety and Performance LIFE SCIENCE SOLUTIONS FOR REAL LIFE Get Your Controlled Environment Under Control Simplified Compliance
Sterilization - validation, qualification requirements. Sterilization - Overview
 Sterilization - validation, qualification requirements Dawn Tavalsky 1 ISPE Boston Chapter Framingham MA 19Sep2013 Sterilization - Overview Objectives Discuss definition of Sterile Briefly describe sterilization
Sterilization - validation, qualification requirements Dawn Tavalsky 1 ISPE Boston Chapter Framingham MA 19Sep2013 Sterilization - Overview Objectives Discuss definition of Sterile Briefly describe sterilization
TOP FIVE TIPS. Autoclave Validation. Create & Maintain Clear Validation Protocols. Begin Your Regimen with a Bowie-Dick Test
 Autoclave Validation TOP FIVE TIPS Time and Money Saving Validation Strategies 1 2 Create & Maintain Clear Validation Protocols Begin Your Regimen with a Bowie-Dick Test 3 Use Autoclave-Friendly Data Loggers
Autoclave Validation TOP FIVE TIPS Time and Money Saving Validation Strategies 1 2 Create & Maintain Clear Validation Protocols Begin Your Regimen with a Bowie-Dick Test 3 Use Autoclave-Friendly Data Loggers
Gary Hartnett, Bausch & Lomb
 What we do Mark is a very personable guy. He has a fantastic drive for results, is very customer focused, creative and leverages from a deep understanding of the pharma industry. He inspires these qualities
What we do Mark is a very personable guy. He has a fantastic drive for results, is very customer focused, creative and leverages from a deep understanding of the pharma industry. He inspires these qualities
The procurement, qualification and calibration of lab instruments: An Overview
 The procurement, qualification and calibration of lab instruments: An Overview The reliability of analytical data generated from chemical and physical analyses is critically dependent on three factors:
The procurement, qualification and calibration of lab instruments: An Overview The reliability of analytical data generated from chemical and physical analyses is critically dependent on three factors:
Options for load release
 Approaches to Release of Sterile Goods from Steam Sterilization: Parametric Release and Routine Monitoring Craig Wallace Senior Technical Specialist 3M Infection Prevention Division Today s Topics 1. Options
Approaches to Release of Sterile Goods from Steam Sterilization: Parametric Release and Routine Monitoring Craig Wallace Senior Technical Specialist 3M Infection Prevention Division Today s Topics 1. Options
Steam Quality Testing
 Offering the highest quality services by providing the knowledge and experience necessary for compliance within regulated industries, ultimately resulting in safe and healthy consumers. Commissioning /
Offering the highest quality services by providing the knowledge and experience necessary for compliance within regulated industries, ultimately resulting in safe and healthy consumers. Commissioning /
New Data Integrity Regulations and Practical Advice for Life Science Laboratories. we prove it.
 1 How to Meet New MHRA, FDA, EMA and WHO Data Integrity Guidelines white paper New Data Integrity Regulations and Practical Advice for Life Science Laboratories we prove it. 2 How to Avoid Poor Temperature
1 How to Meet New MHRA, FDA, EMA and WHO Data Integrity Guidelines white paper New Data Integrity Regulations and Practical Advice for Life Science Laboratories we prove it. 2 How to Avoid Poor Temperature
المملكة العربية السعودية وزارة الصحة اإلدارة العامة لمكافحة عدوى المنشئات الصحية
 1. POLICY STATEMENT: Sterilization process should be strictly monitored using, physical, chemical and biological indicator and the results of indicators should be supervised by infection control on daily
1. POLICY STATEMENT: Sterilization process should be strictly monitored using, physical, chemical and biological indicator and the results of indicators should be supervised by infection control on daily
Temperature & Humidity Monitoring Solutions for Cold Chain
 Temperature & Humidity Monitoring Solutions for Cold Chain 1 Our history WE DESIGN AND DEVELOP BRAND NEW SOLUTIONS Tecnosoft, founded in 1989, is an Italian research and development company of technical-scientific
Temperature & Humidity Monitoring Solutions for Cold Chain 1 Our history WE DESIGN AND DEVELOP BRAND NEW SOLUTIONS Tecnosoft, founded in 1989, is an Italian research and development company of technical-scientific
Model FD 23 Drying and heating chambers Classic.Line with forced convection
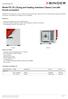 Model FD 23 Drying and heating chambers Classic.Line with forced convection A BINDER FD series heating oven is always used when fast drying and sterilization is required. Thanks to its fully homogeneous
Model FD 23 Drying and heating chambers Classic.Line with forced convection A BINDER FD series heating oven is always used when fast drying and sterilization is required. Thanks to its fully homogeneous
INSTRUMENT CLEANING & STERILISATION FOAMING BATH FORMULATION. Surgistain. Rapid Multi-Enzyme Cleaner. Kimguard Sterile Wrap. Instrument brushes $32.
 1Ltr Rapid Multi-Enzyme Cleaner Ÿ Proprietary combination enzyme Ÿ concentrate Ÿ Unparalleled combination of speed Ÿ and efficacy Ÿ Fully effective against proteins, Ÿ carbohydrates, lipids and Ÿ mucopolysaccharides
1Ltr Rapid Multi-Enzyme Cleaner Ÿ Proprietary combination enzyme Ÿ concentrate Ÿ Unparalleled combination of speed Ÿ and efficacy Ÿ Fully effective against proteins, Ÿ carbohydrates, lipids and Ÿ mucopolysaccharides
TrackSense Pro. The ultimate wireless data logger
 TrackSense Pro The ultimate wireless data logger The Ultimate Wireless Data Logger TrackSense Pro is the most accurate data logger available on the market today Ellab has incorporated the latest in electronic
TrackSense Pro The ultimate wireless data logger The Ultimate Wireless Data Logger TrackSense Pro is the most accurate data logger available on the market today Ellab has incorporated the latest in electronic
Current Moist Heat Sterilization and Validation Seminar
 Current Moist Heat Sterilization and Validation Seminar Keith Shuttleworth is the Senior Consultant at Keith Shuttleworth & Associates Ltd, which provides a range of specialist products, consultancy and
Current Moist Heat Sterilization and Validation Seminar Keith Shuttleworth is the Senior Consultant at Keith Shuttleworth & Associates Ltd, which provides a range of specialist products, consultancy and
MANAGEMENT AND REQUALIFICATION OF STERILISATION PROCESSES
 SUMMARY The Annexe 1 requirement for the annual requalification of sterilization processes is a huge resource requirement as well as a significant amount of downtime for many sites running complex sterilisation
SUMMARY The Annexe 1 requirement for the annual requalification of sterilization processes is a huge resource requirement as well as a significant amount of downtime for many sites running complex sterilisation
Tank Scale Service Checklist
 Tank Scale Service Checklist Specifying Service for Optimized Weighing Processes Selecting the right weighing equipment is an important first step to ensuring that your weighing processes are able to meet
Tank Scale Service Checklist Specifying Service for Optimized Weighing Processes Selecting the right weighing equipment is an important first step to ensuring that your weighing processes are able to meet
Advanced 3D and 4D Temperature Mapping Capabilities
 Advanced 3D and 4D Temperature Mapping Capabilities Background Temperature recording is used in a wide variety of US FDA regulated firms from food canneries to medical device manufacturers to drug manufacturers
Advanced 3D and 4D Temperature Mapping Capabilities Background Temperature recording is used in a wide variety of US FDA regulated firms from food canneries to medical device manufacturers to drug manufacturers
Regulatory Expectations, Standards & Guidelines
 Regulatory Expectations, Standards & Guidelines Regulatory Requirements Pharmacopeias Good Automated Manufacturing Practice (GAMP) 21 CFR Part 11 and Annex 11 Consequences of Non-Compliance 22 Regulatory
Regulatory Expectations, Standards & Guidelines Regulatory Requirements Pharmacopeias Good Automated Manufacturing Practice (GAMP) 21 CFR Part 11 and Annex 11 Consequences of Non-Compliance 22 Regulatory
Water purification in the pharmaceutical industry
 ABB MEASUREMENT & ANALYTICS APPLICATION DESCRIPTION Water purification in the pharmaceutical industry Providing independent verification and validation of the water purification process for compliance
ABB MEASUREMENT & ANALYTICS APPLICATION DESCRIPTION Water purification in the pharmaceutical industry Providing independent verification and validation of the water purification process for compliance
 Rev. 1 May 2018 Terragene offers a range of effective and easy-to-handle products to control the washing, hygiene, disinfection and sterilization processes in medical diagnostic laboratories. Available
Rev. 1 May 2018 Terragene offers a range of effective and easy-to-handle products to control the washing, hygiene, disinfection and sterilization processes in medical diagnostic laboratories. Available
Standard Monitoring System
 Cleanroom-Requirements according to Annex 1, EU-GMP-Guidelines Strict requirements for compliance with the environmental conditions in the relative cleanroom classes Continuous data recording and archiving
Cleanroom-Requirements according to Annex 1, EU-GMP-Guidelines Strict requirements for compliance with the environmental conditions in the relative cleanroom classes Continuous data recording and archiving
It s simple. USB data logger testo 184. Easy temperature monitoring and uninterrupted quality documentation in the transportation of pharmaceuticals.
 It s simple. USB data logger testo 184. Easy temperature monitoring and uninterrupted quality documentation in the transportation of pharmaceuticals. testo 184 Reliable monitoring of temperature, humidity
It s simple. USB data logger testo 184. Easy temperature monitoring and uninterrupted quality documentation in the transportation of pharmaceuticals. testo 184 Reliable monitoring of temperature, humidity
Training Prospectus Public Course Dates
 Training Prospectus 2018 Public Course Dates Honeyman Group Assuring Compliance in Life Science Operating extensively within the global pharmaceutical, biotech, medical device and related healthcare industries,
Training Prospectus 2018 Public Course Dates Honeyman Group Assuring Compliance in Life Science Operating extensively within the global pharmaceutical, biotech, medical device and related healthcare industries,
Melting Point M-565 Technical data sheet
 Melting Point M-565 Technical data sheet The video camera equipped Melting Point M-565 for the automatic determination of melting and boiling points is designed for true, professional quality control.
Melting Point M-565 Technical data sheet The video camera equipped Melting Point M-565 for the automatic determination of melting and boiling points is designed for true, professional quality control.
Mediprene 500M. Transparent TPE Series
 Mediprene 500M Transparent TPE Series CONTENTS INTRODUCTION KEY PROPERTIES RAW MATERIAL SELECTION TYPICAL APPLICATIONS COLOURED & TRANSPARENT COMPOUNDS GRADE TABLE PROCESSING CONTACTS INTRODUCTION The
Mediprene 500M Transparent TPE Series CONTENTS INTRODUCTION KEY PROPERTIES RAW MATERIAL SELECTION TYPICAL APPLICATIONS COLOURED & TRANSPARENT COMPOUNDS GRADE TABLE PROCESSING CONTACTS INTRODUCTION The
GETINGE HS55 HOSPITAL STERILIZER. Always with you
 GETINGE HS55 HOSPITAL STERILIZER Always with you 2 Getinge HS55 GETINGE HS55 The most versatile, reliable and EAsy-to-use hospital sterilizer in the world The Getinge HS55 is a fully automatic steam sterilizer.
GETINGE HS55 HOSPITAL STERILIZER Always with you 2 Getinge HS55 GETINGE HS55 The most versatile, reliable and EAsy-to-use hospital sterilizer in the world The Getinge HS55 is a fully automatic steam sterilizer.
Model BF 720 Incubators Avantgarde.Line with forced convection
 Model BF 720 Incubators Avantgarde.Line with forced convection The BINDER incubator of the BF Avantgarde.Line series with forced convection is suitable for all gentle incubation applications, particularly
Model BF 720 Incubators Avantgarde.Line with forced convection The BINDER incubator of the BF Avantgarde.Line series with forced convection is suitable for all gentle incubation applications, particularly
QbD1200 TOTAL ORGANIC CARBON ANALYZER RELIABLE MEASUREMENT MADE SIMPLE.
 QbD1200 TOTAL ORGANIC CARBON ANALYZER RELIABLE MEASUREMENT MADE SIMPLE. The NEW QbD1200 delivers the highest performance with unparalleled ease of operation and maintenance Reliable data Stop throwing
QbD1200 TOTAL ORGANIC CARBON ANALYZER RELIABLE MEASUREMENT MADE SIMPLE. The NEW QbD1200 delivers the highest performance with unparalleled ease of operation and maintenance Reliable data Stop throwing
FlexAct BT (Bag Tester) Flexboy 2D November, 2015
 FlexAct BT (Bag Tester) Flexboy 2D November, 2015 Agenda 1 2 3 4 Container Closure Integrity for SUS FlexAct BT Value Proposition FlexAct BT Instruments FlexAct BT SU Flexboy 2D 5 FlexAct BT Validation
FlexAct BT (Bag Tester) Flexboy 2D November, 2015 Agenda 1 2 3 4 Container Closure Integrity for SUS FlexAct BT Value Proposition FlexAct BT Instruments FlexAct BT SU Flexboy 2D 5 FlexAct BT Validation
POL: 08:VP:003:03:NIBT PAGE : 1 of 7
 POL: 08:VP:003:03:NIBT PAGE : 1 of 7 Northern Ireland Blood Transfusion Service POLICY DOCUMENT Document Details Document Number: POL:08:VP:003:03:NIBT No. of Appendices: 2 Supersedes Number: 08:02:VP:003:NIBT
POL: 08:VP:003:03:NIBT PAGE : 1 of 7 Northern Ireland Blood Transfusion Service POLICY DOCUMENT Document Details Document Number: POL:08:VP:003:03:NIBT No. of Appendices: 2 Supersedes Number: 08:02:VP:003:NIBT
21CFR11 / GAMP4 Compliant Measurement Data
 ComSoft 3.3 Version 21CFR11 21CFR11 / GAMP4 Compliant Measurement Data Security in pharmaceutical and chemical production processes due to validation compatible software Certified by Authentic Well proven
ComSoft 3.3 Version 21CFR11 21CFR11 / GAMP4 Compliant Measurement Data Security in pharmaceutical and chemical production processes due to validation compatible software Certified by Authentic Well proven
21CFR11 / GAMP4 Compliant Measurement Data
 21CFR11 / GAMP4 Compliant Measurement Data Security in pharmaceutical and chemical production processes due to validation compatible software Certified by Authentic Well proven Unequivocal Fulfill FDA
21CFR11 / GAMP4 Compliant Measurement Data Security in pharmaceutical and chemical production processes due to validation compatible software Certified by Authentic Well proven Unequivocal Fulfill FDA
Helping Make Safe & Reliable Products Because people count on your products everyday
 Helping Make Safe & Reliable Products Because people count on your products everyday Stability Chambers and Rooms www.cszproducts.com Stability Chambers and Rooms Stability chambers are ideal for ICH Q1A
Helping Make Safe & Reliable Products Because people count on your products everyday Stability Chambers and Rooms www.cszproducts.com Stability Chambers and Rooms Stability chambers are ideal for ICH Q1A
Model FD 115 Drying and heating chambers Avantgarde.Line with forced convection
 Model FD 115 Drying and heating chambers Avantgarde.Line with forced convection A BINDER FD series Avantgarde.Line heating oven is always used when fast drying and sterilization is required. Thanks to
Model FD 115 Drying and heating chambers Avantgarde.Line with forced convection A BINDER FD series Avantgarde.Line heating oven is always used when fast drying and sterilization is required. Thanks to
VALIDATION QUALIFICATION. Pharmaceutical Consultancy Services, All rights reserved. 1. JaapKoster
 VALIDATION QUALIFICATION 1 JaapKoster AGENDA Introduction Validation & Qualification Pre-requisites Process Validation (PV) Analytical Methods Validation (AMV) Shipping Validation / Supply Chain Re-validation
VALIDATION QUALIFICATION 1 JaapKoster AGENDA Introduction Validation & Qualification Pre-requisites Process Validation (PV) Analytical Methods Validation (AMV) Shipping Validation / Supply Chain Re-validation
Getinge HS55 Hospital sterilizer
 Getinge HS55 Hospital sterilizer The most versatile, reliable and Easy-to-use hospital sterilizer in the world The Getinge HS55 is a fully automatic steam sterilizer. It has pre-set programs for the most
Getinge HS55 Hospital sterilizer The most versatile, reliable and Easy-to-use hospital sterilizer in the world The Getinge HS55 is a fully automatic steam sterilizer. It has pre-set programs for the most
Using Empower SystemsQT Qualification Tool
 Using Empower SystemsQT Qualification Tool for Waters Modular HPLC Systems A summary of Empower SystemsQT Qualification Tool features and benefits, with emphasis on the technical attributes of the qualification
Using Empower SystemsQT Qualification Tool for Waters Modular HPLC Systems A summary of Empower SystemsQT Qualification Tool features and benefits, with emphasis on the technical attributes of the qualification
Temperature Mapping Study of Walk-in incubator in a Pharma Industry
 Research Article Temperature Mapping Study of Walk-in incubator in a Pharma Industry Harish Gowda G.S, Gangadharappa H.V*, Kiran H.C, Naveen Kumar V Department of Pharmaceutics (Pharmaceutical Quality
Research Article Temperature Mapping Study of Walk-in incubator in a Pharma Industry Harish Gowda G.S, Gangadharappa H.V*, Kiran H.C, Naveen Kumar V Department of Pharmaceutics (Pharmaceutical Quality
QUALIFICATION and VALIDATION Open Discussion Workshop QP Forum 21 st April 2016
 QUALIFICATION and VALIDATION Open Discussion Workshop QP Forum 21 st April 2016 Representatives from cross section of companies were at this workshop namely: Abbott, Stiefel, Abbvie, Pfizer, Helsinn, BMS,
QUALIFICATION and VALIDATION Open Discussion Workshop QP Forum 21 st April 2016 Representatives from cross section of companies were at this workshop namely: Abbott, Stiefel, Abbvie, Pfizer, Helsinn, BMS,
Mediclave Autoclaves 6, 18 & 23 Litre B, S & N Class
 Mediclave Autoclaves 6, 18 & 23 Litre B, S & N Class LTE Mediclave range of autoclaves are some of the most comprehensively equipped medical steam sterilizers available. Designed to incorporate features
Mediclave Autoclaves 6, 18 & 23 Litre B, S & N Class LTE Mediclave range of autoclaves are some of the most comprehensively equipped medical steam sterilizers available. Designed to incorporate features
LAB DIVISION. STERILIZATION cglp Process Equipment for Research and Microbiological Laboratories
 LAB DIVISION STERILIZATION cglp Process Equipment for Research and Microbiological Laboratories LAB division Company LAST Technology is the worldwide provider for cleaning, decontamination, disinfection
LAB DIVISION STERILIZATION cglp Process Equipment for Research and Microbiological Laboratories LAB division Company LAST Technology is the worldwide provider for cleaning, decontamination, disinfection
GAMP 4/5 Prospective / retrospective Qualification of water treatment plants) P. Dony
 GAMP 4/5 Prospective / retrospective Qualification of water treatment plants) P. Dony 31.03.2008 Table of contents Slide 2 1. GAMP 4 and GAMP 5 differences 2. CHRIST prospective Qualification strategy
GAMP 4/5 Prospective / retrospective Qualification of water treatment plants) P. Dony 31.03.2008 Table of contents Slide 2 1. GAMP 4 and GAMP 5 differences 2. CHRIST prospective Qualification strategy
Blackbox for sensitive freight.
 Blackbox for sensitive freight. Data logger testo 184: Temperatur monitoring and quality data documentation in the transport of foods and pharmaceutical goods. testo 184 Optimum temperatures for sensitive
Blackbox for sensitive freight. Data logger testo 184: Temperatur monitoring and quality data documentation in the transport of foods and pharmaceutical goods. testo 184 Optimum temperatures for sensitive
General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT
 General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (16) 86 R2 MANAGEMENT OF ENVIRONMENTAL CONDITIONS Full document title and reference Document type Management of Environmental
General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (16) 86 R2 MANAGEMENT OF ENVIRONMENTAL CONDITIONS Full document title and reference Document type Management of Environmental
Drug Quality Assurance: Systems at ChemCon Author: Dr. Peter Gockel
 Drug Quality Assurance: Systems at ChemCon Author: Dr. Peter Gockel On February 13th, 2006, the FOOD AND DRUG ADMINISTRATION (FDA) implemented a revision to the Compliance Program Guidance Manual for active
Drug Quality Assurance: Systems at ChemCon Author: Dr. Peter Gockel On February 13th, 2006, the FOOD AND DRUG ADMINISTRATION (FDA) implemented a revision to the Compliance Program Guidance Manual for active
It s simple. USB data logger testo 184. Easy temperature monitoring and uninterrupted quality documentation in the transportation of pharmaceuticals.
 It s simple. USB data logger testo 184. Easy temperature monitoring and uninterrupted quality documentation in the transportation of pharmaceuticals. testo 184 Reliable monitoring of temperature, humidity,
It s simple. USB data logger testo 184. Easy temperature monitoring and uninterrupted quality documentation in the transportation of pharmaceuticals. testo 184 Reliable monitoring of temperature, humidity,
Temperature Mapping Report
 Document Status: Page: 1 of 13 Temperature Mapping Report Company Name & Site Warehouse Start Date: DD Month YYYY End Date: DD Month YYYY Document Status: Page: 2 of 13 Table of Contents 1 Approvals...
Document Status: Page: 1 of 13 Temperature Mapping Report Company Name & Site Warehouse Start Date: DD Month YYYY End Date: DD Month YYYY Document Status: Page: 2 of 13 Table of Contents 1 Approvals...
Losing Everything: Is a Chart Recorder Enough to Protect Critical Assets? we prove it. - Reliable - Flexible - Compliant
 WHITE PAPER IS A CHART RECORDER ENOUGH TO PROTECT CRITICAL ASSETS 1 - Reliable - Flexible - Compliant white PAPer Losing Everything: Is a Chart Recorder Enough to Protect Critical Assets? we prove it.
WHITE PAPER IS A CHART RECORDER ENOUGH TO PROTECT CRITICAL ASSETS 1 - Reliable - Flexible - Compliant white PAPer Losing Everything: Is a Chart Recorder Enough to Protect Critical Assets? we prove it.
DETAILED PROCEDURE AND GUIDELINES TEMPERATURE MAPPING STUDY & QUALIFICATION OF COLD ROOMS, WAREHOUSES, VANS, TRUCKS, REEFERS, REFRIGERATORS & BOXES
 DETAILED PROCEDURE AND GUIDELINES ON TEMPERATURE MAPPING STUDY & QUALIFICATION OF COLD ROOMS, WAREHOUSES, VANS, TRUCKS, REEFERS, REFRIGERATORS & BOXES FOR PHARMACEUTICAL INDUSTRY Revision History: Revision
DETAILED PROCEDURE AND GUIDELINES ON TEMPERATURE MAPPING STUDY & QUALIFICATION OF COLD ROOMS, WAREHOUSES, VANS, TRUCKS, REEFERS, REFRIGERATORS & BOXES FOR PHARMACEUTICAL INDUSTRY Revision History: Revision
Optimizing Performance Bench Scale Service Checklist
 Optimizing Performance Bench Scale Specifying Service For optimized weighing processes Selecting the right weighing equipment is an important first step to ensuring that your weighing processes are able
Optimizing Performance Bench Scale Specifying Service For optimized weighing processes Selecting the right weighing equipment is an important first step to ensuring that your weighing processes are able
PERFORMANCE QUALIFICATION PROTOCOL HVAC SYSTEM
 Page 1 of 24 PERFORMANCE QUALIFICATION PROTOCOL FOR HVAC SYSTEM Signing of this Performance Qualification Protocol indicates agreement with the Validation Master Plan approach of the equipment. Further
Page 1 of 24 PERFORMANCE QUALIFICATION PROTOCOL FOR HVAC SYSTEM Signing of this Performance Qualification Protocol indicates agreement with the Validation Master Plan approach of the equipment. Further
Installation Qualification/Operational Qualification Protocols and Instructions
 Installation Qualification/Operational Qualification Protocols and Instructions AquaLab VSA Vapor Sorption Analyzer Decagon Devices, Inc June 28, 2016 11:03 AM 1 Table of Contents Section 1 Section 2 Section
Installation Qualification/Operational Qualification Protocols and Instructions AquaLab VSA Vapor Sorption Analyzer Decagon Devices, Inc June 28, 2016 11:03 AM 1 Table of Contents Section 1 Section 2 Section
E-Val Pro. The next generation in real-time thermal validation
 E-Val Pro The next generation in real-time thermal validation E-Val Pro Thermal Validation System E-Val Pro 40 channel module The E-Val Pro Thermal Validation System is designed for validation applications
E-Val Pro The next generation in real-time thermal validation E-Val Pro Thermal Validation System E-Val Pro 40 channel module The E-Val Pro Thermal Validation System is designed for validation applications
Validation Strategies for Equipment from Multiple Vendors
 Welcome to our E-Seminar: Validation Strategies for Equipment from Multiple Vendors Page 1 Content FDA guidelines and inspectional observations Validation & Qualification Approaches Instrument Qualification
Welcome to our E-Seminar: Validation Strategies for Equipment from Multiple Vendors Page 1 Content FDA guidelines and inspectional observations Validation & Qualification Approaches Instrument Qualification
McKesson Sterilization Products
 McKesson Sterilization Products Table of Contents Introduction Introduction... 3 Biological Indicators and Incubators Steam Integrator... 4 Sterilization Biological Indicator Vial...4 Self-Contained Biological
McKesson Sterilization Products Table of Contents Introduction Introduction... 3 Biological Indicators and Incubators Steam Integrator... 4 Sterilization Biological Indicator Vial...4 Self-Contained Biological
Freeze drying and sterilisation processes R&D Laboratory
 Freeze drying and sterilisation processes R&D Laboratory Telstar makes its Process Laboratory available to you Current manufacturing regulations require medicines to be qualified and prepared following
Freeze drying and sterilisation processes R&D Laboratory Telstar makes its Process Laboratory available to you Current manufacturing regulations require medicines to be qualified and prepared following
Warehouse Mapping of GxP Storage Facilities: Why it s Not an Option. we prove it. A GxP Compliant Thermal Mapping Overview
 1 A GxP Compliant Thermal Mapping Overview white paper Warehouse Mapping of GxP Storage Facilities: Why it s Not an Option we prove it. 2 Warehouse Mapping of GxP Storage Facilities: Why It s Not an Option
1 A GxP Compliant Thermal Mapping Overview white paper Warehouse Mapping of GxP Storage Facilities: Why it s Not an Option we prove it. 2 Warehouse Mapping of GxP Storage Facilities: Why It s Not an Option
Conducting Supplier Audits: Ensure Validation Compliance
 Conducting Supplier Audits: Ensure Validation Compliance Presented by: Gamal Amer, Ph. D. Principal Premier Compliance Services, Inc. All rights reserved. Do not copy without permission. 1 Outline Why
Conducting Supplier Audits: Ensure Validation Compliance Presented by: Gamal Amer, Ph. D. Principal Premier Compliance Services, Inc. All rights reserved. Do not copy without permission. 1 Outline Why
KINGSMANN CARE GROUP
 PHARMA CONSULTANTS KINGSMANN CARE GROUP KINGSMANN CONSULTANCY SERVICES Thank you for taking interest in Kingsmann Consultancy Services. Kingsmann Consultancy (KC) is a leading business development-consulting
PHARMA CONSULTANTS KINGSMANN CARE GROUP KINGSMANN CONSULTANCY SERVICES Thank you for taking interest in Kingsmann Consultancy Services. Kingsmann Consultancy (KC) is a leading business development-consulting
