Nature and origin of gap filaments in striated muscle
|
|
|
- Elfreda Little
- 5 years ago
- Views:
Transcription
1 Nature and origin of gap filaments in striated muscle K. TROMBITAS, P. H. W. W. BAATSEN, M. S. Z. KELLERMAYER and G. H. POLLACK Bioengineering WD-12, University of Washington, Seattle, Washington 98195, USA Summary Immunoelectron microscopy was used to study the nature and origin of 'gap' filaments in frog semitendinosus muscle. Gap filaments are fine longitudinal filaments observable only in sarcomeres stretched beyond thick/thin filament overlap: they occupy the gap between the tips of thick and thin filaments. To test whether the gap filaments are part of the titin-filament system, we employed monoclonal antibodies to titin (T-ll, Sigma) and observed the location of the epitope at a series of sarcomere lengths. At resting sarcomere length, the epitope was positioned in the I-band approximately 50 run beyond the apparent ends of the thick filament. The location did not change perceptibly with increasing sarcomere length up to 3.6 fan. Above 3.6 fan, the span between the epitope and the end of the A-band abruptly increased, and above 4 fan, the antibodies could be seen to decorate the gap filaments. Between 5 and 6 fan, the epitope remained approximately in the middle of the gap. Even with this high degree of stretch, the label remained more or less aligned across the myofibril. The abrupt increase of span beyond 3.6 fan implies that the A-band domain of titin is pulled free of its anchor points along the thick filament, and moves toward the gap. Although this domain is functionally inextensible at physiological sarcomere length, the epitope movement in extremely stretched muscle shows that it is intrinsically elastic. Thus, the evidence confirms that gap filaments are clearly part of the titin-filament system. They are derived not only from the I-band domain of titin, but also from its A-band domain. Key words: elastic filament, titin, skeletal muscle, immunoelectron microscopy. Introduction The discovery of titin (also called connectin), a giant structural protein, led eventually to the conclusion that the sarcomere contained, in addition to thick and thin filaments, a third longitudinal filament system composed of titin (Wang, 1985; Maruyama, 1986). On the basis of immunolocalization studies, it was suggested that titin formed a continuous filament connecting the M-line to the Z-line (Wang et al ; Fiirst et al. 1988; Itoh et al. 1988; Pierobon-Bormioli et al. 1990). These studies (see also Trombitas et al ) further implied that at physiological sarcomere length only part of the titin strand was free and elastic: the I-band region could stretch with increases of sarcomere length, but the A-band domain appeared to be tethered to the thick filament. Logically, as the sarcomere is extended, the I-band domain of titin should be extended concomitantly. As the sarcomere is extended beyond thick and thin filament overlap, a segment of the titin filament should become visualizable in the gap. Although gap filaments have long been observable, there has been no direct evidence that they are made of titin. In earlier studies, the misalignment of titin epitopes that occurs at longer sarcomere length has made it impossible to obtain regular gap-filament labelling (Itoh et al. 1988; Ramirez-Mitchell et al. 1990; Wang, 1985). The present study was designed to define the nature and origin of gap filaments. To achieve this, we monitored the relative positions of a titin epitope that ordinarily lies Journal of Cell Science 100, (1991) Printed in Great Britain The Company of Biologists Limited 1991 close to the A-I junction, as the sarcomere was stretched. We confirm that the gap filament is indeed made of titin. However, the filament is derived not from titin's I-band domain exclusively: increasing tension apparently rips titin from its A-band tether, allowing a segment of the A- band domain to be translated into the gap. Materials and methods Single fibres from frog semitendinosus muscle were mechanically skinned by peeling off the sarcolemma in relaxing solution. The solution contained (HIM): calcium proprionate, 0.035; magnesium proprionate, 6; potassium proprionate 5; K 2 EGTA, 15; Mops, 117; Na 2 ATP, 4.4; Na 2 CP, 15.6; KOH, 57; pca9.2, ionic strength 0.2, ph7.0). One end of each fibre was pinned down with a minutien pin (Fine Science Tools Inc., Cat. no ) to a Sylgard elastomer dissection base on the bottom of a small Petri dish. The fibres were then stretched in small increments to the appropriate sarcomere length. Sarcomere length was set from 2.5 jjia to 6.0 fim. Then the fibres were fixed in a freshly prepared fomaldehyde/pbs fixative (3.7 % paraformaldehyde, 2.7 mm KC1, 1.5 mm KH 2 PO 4,137 DIM NaCl, 8 mm Na 2 HPO 4, ph 7.2) for 15 min at 4 C, and washed three times in PBS for 30min per wash. The unspecific binding sites were blocked using PBS/1% BSA solution for 30 min. Conventionally available monoclonal titin antibody (Til, Sigma) was used for the immunolabelling experiments. The test fibres were then incubated for 24 h in the primary antibody solution (25^gml -1 mouse anti-titin IgG in PBS/BSA) at 4 C. After washing the preparations three times with PBS/BSA for 30 min, the fibres were treated with secondary antibody solution (50 fig ml" 1 rabbit anti-mouse IgG (Sigma) in 809
2 PBS/BSA) for 24h, at 4 C. Control fibres were incubated only in the secondary antibody solution under the same conditions as the test fibres. In addition, the overstretched fibres were labelled either with polyclonal tropomyosin antibody (Lemanski, 1979) as described earlier (Trombitas et al. 1990a), or with monoclonal troponin-t antibody (Sigma), in a manner similar to that used with antititin. The unbound secondary antibody was removed from the fibres by washing in PBS, for 30 min per wash, three times. Then the PBS was replaced with Mops buffer solution (20 mm K-Mops, 5mM MgCl2, ph6.8), and the fibres were fixed in 2.5% glutaraldehyde, 0.2% tannic acid, 10 mm MgCl2, 5mM EGTAand 20 mm K-Mops at ph6.8 for 30min at 4 C, according to the method of Reedy and Reedy (1985). Subsequently, the fibres were washed in Mops buffer and in 100 mm potassium phosphate buffer. Then they were postfixed in 1% OsO4, 100 mm potassium phosphate buffer at ph 6.0 for 30 min at 0 C, washed in the same buffer twice for 15 min each, and in water in the same way. Fibres were stained en bloc with 2 % aqueous uranyl acetate, dehydrated in a graded ethanol series, and embedded in Araldite. Ultrathin sections were cut with an LKB Ultratome III, stained with potassium permanganate and lead citrate, and observed and photographed with a Philips 420 electron microscope. Results In muscle stretched above a sarcomere length (SL) of 3.6 nm, a gap appears between the thick and thin filaments. In such a case, sarcomere continuity is maintained by the gap filaments (Fig. 1). Gap filaments associate with thick filaments in the A-band (arrows) and run across the gap into the I-band. They are obviously different from the thin filaments, having much smaller diameter. A fine periodicity of about nm is occasionally observable along the gap filament both in the gap itself, and in the I-band (Fig. 1, arrowheads). Furthermore, immunolabelling experiments show that although both the anti-tropomyosin antibodies (Fig. 1) and the antitroponin-t antibodies (Fig. 2) label the I-segment, there is no labelling of the gap filaments. Thus, the gap filaments are not displaced thin filaments. To study the nature of the gap filaments further, we performed immunolabelling experiments using a monoclonal titin antibody that was reported to label chickenbreast myofibrils near the A-I junction. Although this T11 antibody was reported to cross-react with muscles from several different species, frog striated muscle was not checked. We therefore carried out pilot studies on isolated myofibrils from frog leg muscle using immunofluorescence microscopy. The results (data not shown) revealed labelling at the A-I junction, the same region in which chickenbreast muscle was labelled. Experiments performed using immunoelectron microscopy showed excellent antibody labelling (Fig. 3B). The sarcomeres visualized in this field are at a relatively short sarcomere length, 2.5 /an. The epitope was distinctly labelled, forming a stripe perpendicular to the fibre axis, situated some 50 nm from the visually detectable end of the thick filament. The 50-nm separation is the same as originally reported in chicken muscle (Fiirst et al. 1988). Unexpectedly, the 50-nm separation did not change as the unactivated sarcomere was stretched, at least moderately. Fig. 3C shows the labelling in a sarcomere fixed at 3.2 /on, while Fig. 3D shows labelling in a sarcomere stretched to the verge of no overlap. In both cases the span between the two epitopes straddling the A-band was approximately 1.7 /an, the same as at the shorter lengths. Note that the label pattern remains approximately normal to the filament axis in spite of the large degree of stretch. At sarcomere lengths beyond 3.6/on, where the gap Fig. 1. Anti-tropomyosin-labelled overstretched fibres; SL (sarcomere length)=4.0/un. Labelling is restricted to the I-segment. Each thick filament continues in a single gap filament (arrows). Fine periodicity can occasionally be detected along the gap filaments (arrowheads). Bar, 0.5 fan. Fig. 2. Anti-troponin-T-labelled highly stretched fibre (SL=4.2 fan). I-segments are heavily labelled, while gap filaments remain unlabelled. Bar, 0.5 /an. 810 K. Trombitds et al.
3 Fig. 3. Immunoelectron microscopical localization of T-11 titin epitope in sarcomeres of different length. (A) Control muscle, unlabelled. (B) Antibody-labelled fibre at short sarcomere length (SL=2.5/OTI). The distance between the epitope and the end of the A-band is about 0.05/.an. (C) SL=3.2/an. The epitope position is unchanged relative to B. (D) SL=3.6. Epitope position is again unchanged. (E) SL=3.9/an. The gap between the thick and thin filaments has just appeared. Distance between label and edge of A-band is abruptly increased. Note that the epitope distribution has become slightly irregular. (F) SL=5.4^m. At sarcomere lengths between 5 and 6 /an, the label is found in the middle of the gap, /an from the end of the thick filaments. (G) Overstretched control muscle. Bar, 0.5 fan. between thick and thin filaments just appears, the span between epitopes no longer remains constant; it increases abruptly (Fig. 3E). In this Figure, which shows sarcomeres at 3.9 /on, not only is the span increased (to 1.9 /an), but it is also slightly less regular than at shorter lengths. In this case the epitope is not in the gap; it is localized approximately at the thin filament tips. Fig. 3F shows the labelling pattern well beyond overlap. Here, the label is clearly within the gap. Even with this extreme degree of stretch, the label remains more or less aligned across the myofibril. The epitope-epitope span increased with increasing sarcomere length. The label remained approximately in the middle of the gap. At sarcomere lengths between 5 and 6/an, the epitope is found from 0.4 to 0.6 /an from the edge of the A-band. In instances in which the myofibril was skewed, the label followed the angle of skew of the A-I junction or the Z-line (Fig. 4). This shows that the antibodies are not deposited Gap filaments 811
4 Fig. 4. Labelling pattern in highly stretched sarcomere. The label follows the skew of the striated pattern. d, 3rd A Sarcomere length(^m) Fig. 5. Pattern of stretch of titin epitope T-ll. Continuous line represents the epitope-epitope distance across the A-band (d A ); broken line, the epitope-epitope distance across the Z-line (d r ). Bar, 0.5/an. accidentally. Furthermore, since the epitope lies near the A-I junction at physiological sarcomere lengths, in overstretched muscle it evidently separates titin's A-band and I-band domains. Thus, both domains are seen within the gap. The results obtained at a series of sarcomere lengths are shown in Fig. 5. Data are averaged from 25 fibres. Continuous lines represent the epitope-epitope span across the A-band, broken lines the epitope epitope span across the Z-line. The former does not change until the sarcomere is stretched to approximately the point of no overlap (3.6 \im); then it changes linearly with stretch. The latter increases steadily with stretch, first with steep slope, and later, as the stretch is shared by A-band-freed titin, with shallower slope. Discussion A now classical method of demonstrating that a continuous filament runs effectively from Z-line to Z-line is to stretch the sarcomere beyond thick and thin filament overlap and examine the structure in the electron microscope. A gap then appears between the tips of thick and thin filaments. However, the gap is not empty. Very thin, so-called 'gap filaments' run longitudinally across 812 K. Trombitds et al.
5 the gap (Huxley and Peachey, 1961; Sjostrand, 1962; Carlsen et al. 1965; Locker and Leet, 1975). Such morphological experiments have not yet revealed the full nature of these filaments. The gap filaments have been shown to be distinct from thin filaments (Magid et al. 1984; Trombitas et al. 1990a) and this study also presents both morphological and immunological evidence showing that the gap filaments are not thin filaments that had been drawn out of the I-band (Figs 1 and 2). The filaments' thickness, fine periodicity and immunological properties reveal a third type of filament system. A reasonable postulate has been that the gap filament is a segment of the titin (also known as connectin) filament (Maruyama et al. 1984; Locker, 1984; Wang, 1985). Earlier tests of this postulate have proved supportive but less than conclusive: although anti-titin labelling studies have revealed label in the gap near the A-band-gap junction (Itoh et al. 1988), near the thin filament tips (Wang, 1985; Ramirez-Mitchell et al. 1990), or in the gap (La Salle et al. 1983; Gassner, 1986), the labelling pattern was generally sparse and semi-random. In this study we present direct evidence for a titin epitope in the gap. Labelling was consistent, and, considering the degree of stretch, reasonably regular across the myofibril. The location of the epitope could be followed at each of a series of sarcomere lengths, conferring reassurance that the labelling could not have been of a non-specific variety. Thus, the gap filament appears to contain titin. The titin origin is supported also by the gap filaments' fine periodicity. At a sarcomere length of 4 fan, the I-band domain of the titin filament is elongated to about three times its resting length. Since the titin axial periodicity was reported to be nm in isolated unstrained strands (Trinick et al. 1984; Wang et al. 1984), at a sarcomere length of 4 fan, the observed nm titin periodicity is expected (Fig. 1). One surprise was the sarcomere-length-dependent behavior of the titin epitope. Our initial hypothesis was that the gap filament was a stretched segment of titin's I- band domain. We therefore selected from among antibodies available to us one that lodged in the I-band, very near to the tip of the thick filament, in the hope that this locus would manifest itself in the gap when the sarcomere was overextended. Thus, the T-ll antibody (Furst et al. 1988) appeared suitable. However, we were surprised to find that with moderate stretch, up to 3.6 fan, this epitope failed to move away from the A-band (Fig. 3). We interpret this in one of two ways. The first possibility is that a short segment of titin emanating from the end of the thick filament is stiff, unlike the rest of the filament. A second and simpler possibility is that the epitope does not really lie in the I- band proper, but lies in the zone of the thick filament's tapered ends, where the lower electron density confers an I-band appearance. If the epitope remains bound to the thick filament during stretch, the absence of translation would be explained. Above 3.6 fim, this epitope translated abruptly from the thick filament, toward the Z-line (Fig. 3E,F). There was no perceptible length change in the A-band, implying that the ends of the thick filament are not disassembled during extension (Wang and Wright, 1988). The abruptness of the translation implies that some or all of the A-band titin becomes 'unglued' from the thick filament, and is pulled into the gap. Presumably, the unbinding is a consequence of the increasing tension associated with stretch. The gap filament, then, is not only a segment of I-band titin; it consists, at least in part, of A-band titin pulled from the thick filament. On the other hand, the fact that the epitope - ordinarily at the A-I junction - remains approximately in the center of the gap (Fig. 3F,G) implies that the gap contains a section of I-band titin as well. The antibody is convenient in that it separates A- and I-band domains of titin in the gap. The stretch-dependent translation of the epitope reveals some clues as to the relative stiffness of the two zones of titin. One clear implication is that the segment that comes unbound is not rigid (Maruyama et al. 1989; Wang and Wright, 1988). The finding that the epitope remains near the middle of the gap as the sarcomere is stretched implies that zones on either side of the epitope are intrinsically stretchable. More quantitative conclusions are assumption-dependent. If it is assumed, for example, that: (1) it is the entire A-band domain of titin that comes unbound, leaving a tether only at the M-line (Pierobon-Bormioli et al. 1990); (2) the slack length of the I-band domain of titin is about 0.3,um (assuming the resting sarcomere length is 2.2 fan); and (3) the A-band domain of titin is bound while in the unstrained condition. At sarcomere length of 5.2 fan (Fig. 5) the A-band domain is 1.2 fan long, or 1.5 times its original length, while the I-band domain is 1.4 fan, or about five times its original length. Thus, the A-band domain of titin would appear to be stiffer than the I-band domain (Itoh et al. 1988). On the other hand, if one of the above assumptions were erroneous, this conclusion might not hold. For example, if only a distal section of A-band titin comes loose rather than the entire strand (Wang and Wright, 1988), so that the freed segment has shorter slack length, the observed epitope behavior might imply a similar compliance for this segment as for the I-band segment. More definitive conclusions can be drawn through the use of antibodies bound simultaneously to several epitopes along the titin strand. Uncertainties notwithstanding, the results demonstrate several points unambiguously. The gap filament clearly contains titin - as has previously been suggested. The titin in the gap is recruited from two zones, the edge of the A- band and the adjoining edge of the I-band - not purely from the I-band, as generally believed. Finally, the A-band segment of titin, which is ordinarily bound along the thick filament, is not rigid; it is at least moderately compliant. References CARLSEN, F., KNAPPEIS, G. G. AND BUCHTAL, F. (1961). Ultrastructure of the resting and contracted striated muscle fibre at different degrees of stretch. J. biophys. biochem. Cytol. 11, FURST, D. 0., OSBORN, M., NAVE, R AND WEBER, K. (1988). The organization of titin filaments in the half sarcomere revealed by monoclonal antibodies in immunoelectron microscopy a map of ten nonrepetitive epitopes starting at the Z-line extends close to the M- line J. Cell Biol. 106, GASSNER, D. (1986). Myofibrillar interaction of blot immunoaffinitypunfied antibodies against native titin as studied by direct immunofluorescence and immunogold staining Eur J. Cell Biol. 40, HUXLEY, A. F AND PEAOHEY, L. D. (1961). The maximum length for contraction in vertebrate striated muscle. J. Physwl. 156, ITOH, Y., SUZUKI, T., KIMURA, S., OHASHI, K., HIGUCHI, H., SAWADA, H., SHIMIZU, T., SHIBATA, M. AND MARUYAMA, K. (1988). Extensible and less-extensible domains of connectin filaments in stretched vertebrate skeletal muscle as detected by immunofluorescence and immunoelectron microscopy using monoclonal antibodies. J. Biochem. (Tokyo) 104, Gap filaments 813
6 LA SALLE, F., ROBSON, R. M., LUSBY, M. L., PARRISH, F. C, STROMER, M. H. AND HUIATT, T. W. (1983). Localization of titin in bovine skeletal muscle. J. Cell Biol. 97, 258a. LEMANSKI, L. F. (1979). Role of tropomyosin in actin filament formation in embryonic salamander heart cells. J. Cell Biol. 82, LOOKER, R H. (1984). The role of gap filaments in muscle and meat. Food Microstructure, vol. 3, pp SEM Inc., Chicago. LOCKER, R. H. AND LEET, N. G. (1975). Histology of highly-stretched single fibres. J. Ultrastruct. Res. 52, MAGID, A., TING-BEALL, H. P., CARVELL, M., KONTIS, T. AND LUCAVECHE, C. (1984). Connecting filaments, core filaments and side-struts, a proposal to add three new load-bearing structures to the sliding filament model. In Contractile Mechanism in Muscle (ed. Pollack, G. H. and Sugi, H.), pp Plenum Press, New York. MARUYAMA, K. (1986). Connectin, an elastic filamentous protein of striated muscle. Int. Rev Cytol. 104, MARUYAMA, K, MATSUNO, A., HIGUCHI, H., SHIMAOKA, S., KIMURA, S. AND SHIMIZU, T. (1989). Behavior of connectin (titin) and nebulin in skinned muscle fibres released after extreme stretch as revealed by immunoelectron microscopy. J. Muscle Res. Cell Motil. 10, MARUYAMA, K., SAWADA, H., KIMURA, S., OHASHI, K., HIGUCHI, H. AND UMAZUME, Y. (1984). Connectin filaments in stretched skinned fibres of frog skeletal muscle. J. Cell Biol. 99, PIEROBON-BORMIOLI, S., BETTO, R. AND SALVIATI, G. (1990). The organization of titin (connectin) and nebulin in the sarcomere: an immunolocalization study. J. Muscle Res. Cell Motil. 10, RAMIREZ-MITCHELL, R., WRIGHT, J. AND WANG, K. (1990). Image processing of immunogold-labelled titin filaments in skeletal muscle. Proc. Int. Congr. Electron Microsc. XII vol. 3, pp San Francisco Press, San Francisco REEDY, M. K. AND REEDY, M. C. (1985). Rigor cross-bridge structure in tilted single filament layers and flared-x formations from insect flight muscle. J. molec. Biol. 185, SJOSTRAND, F. (1962). The connections between A- and I-band filaments in striated frog muscle. J Ultrastruct. Res. 7, TRINICK, J. A., KNIGHT, P. AND WHITING, A. (1984). Purification and properties of native titin. J. molec. Biol. 180, TROMBITAS, K., BAATSEN, P. H. W. W., LIN, J J. C, LEMANSKI, L. F. AND POLLACK, G. H. (1990a). Immunoelectron microscopic observations on tropomyosin localization in striated muscle. J. Muscle Res. Cell Motil. 11, TROMBITAS, K., POLLACK, G. H., WRIGHT, J. AND WANG, K (19906). Elastic behavior and arrangement of titin filaments in the I-band. Proc. Int. Congr. Electron Microsc. XII. vol. 3, pp San Francisco Press, San Francisco. WANG, K. (1985). Sarcomere associated cytoskeletal lattices in striated muscle. In Cell and Muscle Motility (ed. by Shay, J. W.), pp New York, Plenum Press. WANG, K., RAMIREZ-MITCHELL, R. AND PALTER, D. (1984a) Titin is an extraordinarily long, flexible and slender myofibrillar protein. Proc. natn. Acad. Sci. U.S.A. 81, WANG, K. AND WRIGHT, J. (1988). Sarcomere matrix of skeletal muscle: The role of thick filaments in the segmental extensibility of elastic titin filaments. Biophys. J. 53, 25a. WANG, K., WRIGHT, J. AND RAMIREZ-MITCHELL, R. (19846). Architecture of the titin/nebulin containing cytoskeletal lattice of the striated muscle sarcomere. Evidence of elastic and inelastic domains of the bipolar filaments. J. Cell Biol. 99, 435a (Received 13 May Accepted 19 August 1991) 814 K. Trombitds et al.
Elastic Behavior of Cormectin Filaments during Thick Filament Movement in Activated Skeletal Muscle
 Elastic Behavior of Cormectin Filaments during Thick Filament Movement in Activated Skeletal Muscle Robert Horowits,* Koscak Maruyama,* and Richard J. Podolsky* * National Institute of Arthritis and Musculoskeletal
Elastic Behavior of Cormectin Filaments during Thick Filament Movement in Activated Skeletal Muscle Robert Horowits,* Koscak Maruyama,* and Richard J. Podolsky* * National Institute of Arthritis and Musculoskeletal
of Gigantic Proportions Frederick C. Parrish, Jr.*, Leader Bruce C. Paterson, Coordinator Jeffery M. Paxhia
 RECIP ROCATION SESSlON Titin: A Myofibrillar/Cytoskeletal Protein of Gigantic Proportions Frederick C. Parrish, Jr.*, Leader Bruce C. Paterson, Coordinator Jeffery M. Paxhia Wang (1988) carried out studies
RECIP ROCATION SESSlON Titin: A Myofibrillar/Cytoskeletal Protein of Gigantic Proportions Frederick C. Parrish, Jr.*, Leader Bruce C. Paterson, Coordinator Jeffery M. Paxhia Wang (1988) carried out studies
A survey of interactions made by the giant protein titin
 Journal of Cell Science 104, 119-123 (1993) Printed in Great Britain The Company of Biologists Limited 1993 119 A survey of interactions made by the giant protein titin A. Soteriou, M. Gamage and J. Trinick*
Journal of Cell Science 104, 119-123 (1993) Printed in Great Britain The Company of Biologists Limited 1993 119 A survey of interactions made by the giant protein titin A. Soteriou, M. Gamage and J. Trinick*
Towards a Molecular Understanding of the Elasticity of Titin
 J. Mol. Biol. (1996) 261, 62 71 Towards a Molecular Understanding of the Elasticity of Titin Wolfgang A. Linke 1 *, Marc Ivemeyer 1, Nicoletta Olivieri 2 Bernhard Kolmerer 2, J. Caspar Rüegg 1 and Siegfried
J. Mol. Biol. (1996) 261, 62 71 Towards a Molecular Understanding of the Elasticity of Titin Wolfgang A. Linke 1 *, Marc Ivemeyer 1, Nicoletta Olivieri 2 Bernhard Kolmerer 2, J. Caspar Rüegg 1 and Siegfried
Titin is an extraordinarily long, flexible, and slender myofibrillar protein
 Proc. Nati. Acad. Sci. USA Vol. 81, pp. 3685-3689, June 1984 Biochemistry Titin is an extraordinarily long, flexible, and slender myofibrillar protein (striated muscle/cytoskeletal lattice/sarcomere/elastic
Proc. Nati. Acad. Sci. USA Vol. 81, pp. 3685-3689, June 1984 Biochemistry Titin is an extraordinarily long, flexible, and slender myofibrillar protein (striated muscle/cytoskeletal lattice/sarcomere/elastic
Bridgelike Interconnections between Thick Filaments in Stretched Skeletal Muscle Fibers Observed by the Freeze-Fracture Method
 Bridgelike Interconnections between Thick Filaments in Stretched Skeletal Muscle Fibers Observed by the Freeze-Fracture Method Suechika Suzuki and Gerald H. Pollack Center for Bioengineering, University
Bridgelike Interconnections between Thick Filaments in Stretched Skeletal Muscle Fibers Observed by the Freeze-Fracture Method Suechika Suzuki and Gerald H. Pollack Center for Bioengineering, University
The cytoskeleton. The cytoskeleton, the motor proteins, the muscle and its regulation. The cytoskeleton. The cytoskeleton.
 , the motor proteins, the muscle and its regulation Dept. of Biophysics, University of Pécs Zoltán Ujfalusi January-February 2012 Dynamic framework of the Eukaryotes Three main filament-class: 1. Intermedier
, the motor proteins, the muscle and its regulation Dept. of Biophysics, University of Pécs Zoltán Ujfalusi January-February 2012 Dynamic framework of the Eukaryotes Three main filament-class: 1. Intermedier
Studies on the interaction between titin and myosin
 Histol Histopath (1992) 7: 333-337 Histology and Histopathology Studies on the interaction between titin and myosin Seu-Mei Wang, Chung-Jiuan Jeng and Mu-Chien Sun Department of Anatomy, College of Medicine,
Histol Histopath (1992) 7: 333-337 Histology and Histopathology Studies on the interaction between titin and myosin Seu-Mei Wang, Chung-Jiuan Jeng and Mu-Chien Sun Department of Anatomy, College of Medicine,
The length of cooperative units on the thin filament in rabbit psoas muscle fibres
 The length of cooperative units on the thin filament in rabbit psoas muscle fibres To elicit a biological function, multiple molecules of the same or similar kind are often assembled together, and they
The length of cooperative units on the thin filament in rabbit psoas muscle fibres To elicit a biological function, multiple molecules of the same or similar kind are often assembled together, and they
Does Thin Filament Compliance Diminish the Cross-Bridge Kinetics? A Study in Rabbit Psoas Fibers
 978 Biophysical Journal Volume 76 February 1999 978 984 Does Thin Filament Compliance Diminish the Cross-Bridge Kinetics? A Study in Rabbit Psoas Fibers Gang Wang, Wei Ding, and Masataka Kawai Department
978 Biophysical Journal Volume 76 February 1999 978 984 Does Thin Filament Compliance Diminish the Cross-Bridge Kinetics? A Study in Rabbit Psoas Fibers Gang Wang, Wei Ding, and Masataka Kawai Department
A polymorphism peculiar to bipolar actin bundles
 gmemm A polymorphism peculiar to bipolar actin bundles Noreen R. Francis and David J. DeRosier Rosenstiel Basic Medical Sciences Research Center, Brandeis University, Waltham, Massachusetts 2254 USA Both
gmemm A polymorphism peculiar to bipolar actin bundles Noreen R. Francis and David J. DeRosier Rosenstiel Basic Medical Sciences Research Center, Brandeis University, Waltham, Massachusetts 2254 USA Both
Limits of titin extension in single cardiac myofibrils
 Journal of Muscle Research and Cell Motility 17, 425--438 (1996). Limits of titin extension in single cardiac myofibrils WOLFGANG A. LINKE l*, MARC L. BARTOO 2, MARC IVEMEYER 1 and GERALD H. POLLACK 2
Journal of Muscle Research and Cell Motility 17, 425--438 (1996). Limits of titin extension in single cardiac myofibrils WOLFGANG A. LINKE l*, MARC L. BARTOO 2, MARC IVEMEYER 1 and GERALD H. POLLACK 2
J. Biochem. 99, (1986) Di Hua HU, Sumiko KIMURA, and Koscak MARUYAMA
 J. Biochem. 99, 1485-1492 (1986) Sodium Dodecyl Sulfate Gel Electrophoresis Studies of Connectin-Like High Molecular Weight Proteins of Various Types of Vertebrate and Invertebrate Muscles I Di Hua HU,
J. Biochem. 99, 1485-1492 (1986) Sodium Dodecyl Sulfate Gel Electrophoresis Studies of Connectin-Like High Molecular Weight Proteins of Various Types of Vertebrate and Invertebrate Muscles I Di Hua HU,
CORRELATION BETWEEN FIBER LENGTH, ULTRASTRUCTURE, AND THE LENGTH-TENSION RELATIONSHIP OF MAMMALIAN SMOOTH MUSCLE
 CORRELATION BETWEEN FIBER LENGTH, ULTRASTRUCTURE, AND THE LENGTH-TENSION RELATIONSHIP OF MAMMALIAN SMOOTH MUSCLE PETER H. COOKE and FREDRIC S. FAY From the Department of Muscle Research, Boston Biomedical
CORRELATION BETWEEN FIBER LENGTH, ULTRASTRUCTURE, AND THE LENGTH-TENSION RELATIONSHIP OF MAMMALIAN SMOOTH MUSCLE PETER H. COOKE and FREDRIC S. FAY From the Department of Muscle Research, Boston Biomedical
IV. Observation of the Internal Structural Arrangement
 THE MYOSIN FILAMENT IV. Observation of the Internal Structural Arrangement FRANK A. PEPE and BARBARA DRUCKER From the Department of Anatomy, The School of Medicine, University of Pennsylvania, Philadelphia,
THE MYOSIN FILAMENT IV. Observation of the Internal Structural Arrangement FRANK A. PEPE and BARBARA DRUCKER From the Department of Anatomy, The School of Medicine, University of Pennsylvania, Philadelphia,
Molecular cloning, expression, and protein interaction of avian muscle titin
 Retrospective Theses and Dissertations 1993 Molecular cloning, expression, and protein interaction of avian muscle titin Kuan Onn Tan Iowa State University Follow this and additional works at: http://lib.dr.iastate.edu/rtd
Retrospective Theses and Dissertations 1993 Molecular cloning, expression, and protein interaction of avian muscle titin Kuan Onn Tan Iowa State University Follow this and additional works at: http://lib.dr.iastate.edu/rtd
A method for determining the periodicity of a troponin component in isolated insect flight muscle thin filaments by gold/fab labelling
 Journal of Cell Science 101, 503-508 (1992) Printed in Great Britain The Company of Biologists Limited 1992 503 A method for determining the periodicity of a troponin component in isolated insect flight
Journal of Cell Science 101, 503-508 (1992) Printed in Great Britain The Company of Biologists Limited 1992 503 A method for determining the periodicity of a troponin component in isolated insect flight
Immunofluorescence and phalloidin labeling of mammalian cells
 Immunofluorescence and phalloidin labeling of mammalian cells 2 Contents Materials for immunofluorescence and phalloidin labeling of mammalian cells...1 Immunofluorescence-labelling on cultivated adherent
Immunofluorescence and phalloidin labeling of mammalian cells 2 Contents Materials for immunofluorescence and phalloidin labeling of mammalian cells...1 Immunofluorescence-labelling on cultivated adherent
H zone narrows; light band narrows; outer darker regions of A / dark band widen; 2 max
 M. (a) (i) A / dark band is mainly due to myosin filaments; H zone only myosin filaments; darker band has both types of filament; light band has only actin filaments; max H zone narrows; light band narrows;
M. (a) (i) A / dark band is mainly due to myosin filaments; H zone only myosin filaments; darker band has both types of filament; light band has only actin filaments; max H zone narrows; light band narrows;
Method for Isolation of Intact Titin (Connectin) Molecules from Mammalian Cardiac Muscle
 ISSN 0006-2979, Biochemistry (Moscow), 2013, Vol. 78, No. 5, pp. 455-462. Pleiades Publishing, Ltd., 2013. Original Russian Text I. M. Vikhlyantsev, A. D. Okuneva, U. V. Shumilina, N. N. Salmov, A. G.
ISSN 0006-2979, Biochemistry (Moscow), 2013, Vol. 78, No. 5, pp. 455-462. Pleiades Publishing, Ltd., 2013. Original Russian Text I. M. Vikhlyantsev, A. D. Okuneva, U. V. Shumilina, N. N. Salmov, A. G.
Immuno-Labelling Cryosections
 Thin sections of biological material, mounted on nickel or gold grids, can be labelled by floating them, section-side down, on small, 10 µl, droplets of antibody. This process is conveniently carried out
Thin sections of biological material, mounted on nickel or gold grids, can be labelled by floating them, section-side down, on small, 10 µl, droplets of antibody. This process is conveniently carried out
Studies with Fluorescent Antibody Staining
 THE M BAND Studies with Fluorescent Antibody Staining ELLEN KUNDRAT and FRANK A. PEPE From the Department of Anatomy, University of Pennsylvania Medical School, Philadelphia, Pennsylvania 19104 ABSTRACT
THE M BAND Studies with Fluorescent Antibody Staining ELLEN KUNDRAT and FRANK A. PEPE From the Department of Anatomy, University of Pennsylvania Medical School, Philadelphia, Pennsylvania 19104 ABSTRACT
SDS-PAGE and Western Blot. Molecular Basis of Evolution
 1 SDS-PAGE and Western Blot Molecular Basis of Evolution Homology high level of DNA and protein sequence similarity due to common ancestry. Evidence Genomes of related organisms are very similar. Even
1 SDS-PAGE and Western Blot Molecular Basis of Evolution Homology high level of DNA and protein sequence similarity due to common ancestry. Evidence Genomes of related organisms are very similar. Even
The Dot-Spot Test. a simple method to monitor immunoreagent activity and influence of fixation on antigen recognition. Aurion Newsletter 4
 Aurion Newsletter 4 The Dot-Spot Test a simple method to monitor immunoreagent activity and influence of fixation on antigen recognition Peter F.E.M. van de Plas AURION Costerweg 5 6702 AA Wageningen The
Aurion Newsletter 4 The Dot-Spot Test a simple method to monitor immunoreagent activity and influence of fixation on antigen recognition Peter F.E.M. van de Plas AURION Costerweg 5 6702 AA Wageningen The
Passive Tension and Stiffness of Vertebrate Skeletal and Insect Flight Muscles: The Contribution of Weak Cross-bridges and Elastic Filaments
 Biophysical Journal Volume 65 November 1993 2141-2159 Passive Tension and Stiffness of Vertebrate Skeletal and Insect Flight Muscles: The Contribution of Weak Cross-bridges and Elastic Filaments 2141 Henk
Biophysical Journal Volume 65 November 1993 2141-2159 Passive Tension and Stiffness of Vertebrate Skeletal and Insect Flight Muscles: The Contribution of Weak Cross-bridges and Elastic Filaments 2141 Henk
Myosin and Actin from Escherichia coli K12 C600
 /. Biochem. 84, 1453-1458 (1978) Myosin and Actin from Escherichia coli K12 C600 Kayoko NAKAMURA, 1 Shizuo WATANABE Koui TAKAHASHI,* and Department of Chemistry, Faculty of Science, Tokyo Institute of
/. Biochem. 84, 1453-1458 (1978) Myosin and Actin from Escherichia coli K12 C600 Kayoko NAKAMURA, 1 Shizuo WATANABE Koui TAKAHASHI,* and Department of Chemistry, Faculty of Science, Tokyo Institute of
Lab 1 Microdissection 1. Microdissection
 Lab 1 Microdissection 1 Lab 1. Microdissection The goal of this techniques lab is to understand how to dissect and process tissues that have been incubated in bromodeoxyuridine (BrdU). BrdU is a synthetic
Lab 1 Microdissection 1 Lab 1. Microdissection The goal of this techniques lab is to understand how to dissect and process tissues that have been incubated in bromodeoxyuridine (BrdU). BrdU is a synthetic
SUPPLEMENTAL MATERIAL. Supplemental Methods:
 SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
Mammalian Cardiac Muscle Thick Filaments: Their Periodicity and Interactions with Actin
 Biophysical Journal Volume 82 March 2002 1497 1508 1497 Mammalian Cardiac Muscle Thick Filaments: Their Periodicity and Interactions with Actin Robert W. Kensler Department of Anatomy, University of Puerto
Biophysical Journal Volume 82 March 2002 1497 1508 1497 Mammalian Cardiac Muscle Thick Filaments: Their Periodicity and Interactions with Actin Robert W. Kensler Department of Anatomy, University of Puerto
The globular head domain of titin extends into the center of the sarcomeric M band
 Journal of Cell Science 106, 319-330 (1993) Printed in Great Britain The Company of Biologists Limited 1993 319 The globular head domain of titin extends into the center of the sarcomeric M band cdna cloning,
Journal of Cell Science 106, 319-330 (1993) Printed in Great Britain The Company of Biologists Limited 1993 319 The globular head domain of titin extends into the center of the sarcomeric M band cdna cloning,
me239 mechanics of the cell me239 mechanics of the cell 3.1 biopolymers - motivation 3.2 biopolymers - polymerization
 4. the cytoskeleton - fiber bundle models bathe, heussinger, claessens, bausch, frey [2008] me239 mechanics of the cell 1 me239 mechanics of the cell 2 biopolymers polymerization of actin and tubulin Figure
4. the cytoskeleton - fiber bundle models bathe, heussinger, claessens, bausch, frey [2008] me239 mechanics of the cell 1 me239 mechanics of the cell 2 biopolymers polymerization of actin and tubulin Figure
MICROTUBULES IN PROTOZOAN CELLS. III. ULTRASTRUCTURAL CHANGES DURING DISINTEGRATION AND REFORMATION OF HELIOZOAN MICROTUBULES
 J. Cell Sci. 32, 87-98 (1978) 87 Printed in Great Britain Company of Biologist! Limited 1978 MICROTUBULES IN PROTOZOAN CELLS. III. ULTRASTRUCTURAL CHANGES DURING DISINTEGRATION AND REFORMATION OF HELIOZOAN
J. Cell Sci. 32, 87-98 (1978) 87 Printed in Great Britain Company of Biologist! Limited 1978 MICROTUBULES IN PROTOZOAN CELLS. III. ULTRASTRUCTURAL CHANGES DURING DISINTEGRATION AND REFORMATION OF HELIOZOAN
RODIGER NAVE and KLAUS WEBER*
 A myofibrillar protein of insect muscle related to vertebrate titin connects Z ba a A ba: purification a molecular characterization of invertebrate mini-titin RODIGER NAVE a KLAUS WEBER* Max Planck Institute
A myofibrillar protein of insect muscle related to vertebrate titin connects Z ba a A ba: purification a molecular characterization of invertebrate mini-titin RODIGER NAVE a KLAUS WEBER* Max Planck Institute
THE BIOCHEMICAL BASIS OF MUSCLE CONTRACTION 1
 THE BIOCHEMICAL BASIS OF MUSCLE CONTRACTION 1 Summary: Early microscopic data concerning the appearance of relaxed and contracted muscles is reviewed. This is correlated with modern biochemical studies
THE BIOCHEMICAL BASIS OF MUSCLE CONTRACTION 1 Summary: Early microscopic data concerning the appearance of relaxed and contracted muscles is reviewed. This is correlated with modern biochemical studies
FORMATION IN RELAXED MUSCLE FIBERS AT VARIOUS IONIC STRENGTHS
 X-RAY DIFFRACTION EVIDENCE FOR CROSS-BRIDGE FORMATION IN RELAXED MUSCLE FIBERS AT VARIOUS IONIC STRENGTHS BERNHARD BRENNER National Institute ofarthritis, Diabetes, and Digestive and Kidney Diseases, National
X-RAY DIFFRACTION EVIDENCE FOR CROSS-BRIDGE FORMATION IN RELAXED MUSCLE FIBERS AT VARIOUS IONIC STRENGTHS BERNHARD BRENNER National Institute ofarthritis, Diabetes, and Digestive and Kidney Diseases, National
BME Engineering Molecular Cell Biology. The Cytoskeleton (I): Actin The Cytoskeleton (II): Microtubule & Intermediate Filament
 BME 42-620 Engineering Molecular Cell Biology Lecture 09: The Cytoskeleton (I): Actin The Cytoskeleton (II): Microtubule & Intermediate Filament BME42-620 Lecture 09, September 27, 2011 1 Outline Overviewofcytoskeletal
BME 42-620 Engineering Molecular Cell Biology Lecture 09: The Cytoskeleton (I): Actin The Cytoskeleton (II): Microtubule & Intermediate Filament BME42-620 Lecture 09, September 27, 2011 1 Outline Overviewofcytoskeletal
Oblique Section 3-D Reconstruction of Relaxed Insect Flight Muscle Reveals the Cross-Bridge Lattice in Helical Registration
 1 620 Biophysical Journal Volume 67 October 1994 1620-1633 Oblique Section 3-D Reconstruction of Relaxed Insect Flight Muscle Reveals the Cross-Bridge Lattice in Helical Registration Holger Schmitz, Carmen
1 620 Biophysical Journal Volume 67 October 1994 1620-1633 Oblique Section 3-D Reconstruction of Relaxed Insect Flight Muscle Reveals the Cross-Bridge Lattice in Helical Registration Holger Schmitz, Carmen
SEM Immunocytochemistry for Cells & Materials
 SEM Immunocytochemistry for Cells & Materials R. Geoff Richards AO Research Institute, AO Foundation, Davos, Switzerland. Immunohistochemistry Immunocytochemistry can be performed on a biological specimen,
SEM Immunocytochemistry for Cells & Materials R. Geoff Richards AO Research Institute, AO Foundation, Davos, Switzerland. Immunohistochemistry Immunocytochemistry can be performed on a biological specimen,
Caffeine Contracture in Frog Cardiac Muscle after Exposure
 Short Communication Japanese Journal of Physiology, 30, 137-141, 1980 Caffeine Contracture in Frog Cardiac Muscle after Exposure to High Concentrations of Calcium Moto MATSUMURA and Kazuhiko NARITA Department
Short Communication Japanese Journal of Physiology, 30, 137-141, 1980 Caffeine Contracture in Frog Cardiac Muscle after Exposure to High Concentrations of Calcium Moto MATSUMURA and Kazuhiko NARITA Department
Segments of the obstructed intestinal loops were fixed in 4% paraformaldehyde
 Supplementary text Supplementary materials and methods Histopathological examination Segments of the obstructed intestinal loops were fixed in 4% paraformaldehyde (PFA) and embedded in paraffin wax with
Supplementary text Supplementary materials and methods Histopathological examination Segments of the obstructed intestinal loops were fixed in 4% paraformaldehyde (PFA) and embedded in paraffin wax with
Immunofluorescence Confocal Microscopy of 3D Cultures Grown on Alvetex
 Immunofluorescence Confocal Microscopy of 3D Cultures Grown on Alvetex 1.0. Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigen-specific antibodies coupled
Immunofluorescence Confocal Microscopy of 3D Cultures Grown on Alvetex 1.0. Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigen-specific antibodies coupled
Introduction to Electron Microscopy Andres Kaech
 Center for Microscopy and Image Analysis Introduction to Electron Microscopy Andres Kaech The types of electron microscopes Transmission electron microscope (TEM) Scanning electron microscope (SEM) 1 The
Center for Microscopy and Image Analysis Introduction to Electron Microscopy Andres Kaech The types of electron microscopes Transmission electron microscope (TEM) Scanning electron microscope (SEM) 1 The
Cross-Bridge Number, Position, and Angle in Target Zones of Cryofixed Isometrically Active Insect Flight Muscle
 Biophysical Journal Volume 86 May 2004 3009 3019 3009 Cross-Bridge Number, Position, and Angle in Target Zones of Cryofixed Isometrically Active Insect Flight Muscle Richard T. Tregear,* Mary C. Reedy,
Biophysical Journal Volume 86 May 2004 3009 3019 3009 Cross-Bridge Number, Position, and Angle in Target Zones of Cryofixed Isometrically Active Insect Flight Muscle Richard T. Tregear,* Mary C. Reedy,
NANOGOLD PRODUCT INFORMATION
 NANOGOLD 95 Horse Block Road, Yaphank NY 11980-9710 Tel: (877) 447-6266 (Toll-Free in US) or (631) 205-9490 Fax: (631) 205-9493 Tech Support: (631) 205-9492 tech@nanoprobes.com www.nanoprobes.com PRODUCT
NANOGOLD 95 Horse Block Road, Yaphank NY 11980-9710 Tel: (877) 447-6266 (Toll-Free in US) or (631) 205-9490 Fax: (631) 205-9493 Tech Support: (631) 205-9492 tech@nanoprobes.com www.nanoprobes.com PRODUCT
THE STRUCTURE OF A SIMPLE Z LINE
 Published Online: 1 September, 1973 Supp Info: http://doi.org/10.1083/jcb.58.3.630 Downloaded from jcb.rupress.org on November 8, 2018 THE STRUCTURE OF A SIMPLE Z LINE CLARA FRANZINI-ARMSTRONG From the
Published Online: 1 September, 1973 Supp Info: http://doi.org/10.1083/jcb.58.3.630 Downloaded from jcb.rupress.org on November 8, 2018 THE STRUCTURE OF A SIMPLE Z LINE CLARA FRANZINI-ARMSTRONG From the
Involvement in Cardiac Z-lines and Z-line
 Evidence for Actin Analogues Involvement in Cardiac Z-lines and Z-line M. YAMAGUCHI, R. M. ROBSON,* and M. H. STROMER* Department of Veterinary Anatomy, The Ohio State University, College of Veterinary
Evidence for Actin Analogues Involvement in Cardiac Z-lines and Z-line M. YAMAGUCHI, R. M. ROBSON,* and M. H. STROMER* Department of Veterinary Anatomy, The Ohio State University, College of Veterinary
LOCALIZATION OF ACTIN AND MICROFILAMENT-ASSOCIATED PROTEINS INTESTINAL BRUSH BORDER BY IMMUNOFLUORESCENCE MICROSCOPY
 LOCALIZATION OF ACTIN AND MICROFILAMENT-ASSOCIATED PROTEINS 1N THE MICROVILLI AND TERMINAL WEB OF THE INTESTINAL BRUSH BORDER BY IMMUNOFLUORESCENCE MICROSCOPY ANTHONY BRETSCHER and KLAUS WEBER. From the
LOCALIZATION OF ACTIN AND MICROFILAMENT-ASSOCIATED PROTEINS 1N THE MICROVILLI AND TERMINAL WEB OF THE INTESTINAL BRUSH BORDER BY IMMUNOFLUORESCENCE MICROSCOPY ANTHONY BRETSCHER and KLAUS WEBER. From the
Supporting Online Material Material and Methods
 Supporting Online Material Material and Methods Live-cell DIC recordings PtK 1 cells (ATCC, Manassas, VA) were filmed on a Nikon (Nikon Instruments, Melville, NY) inverted microscope equipped with 60x
Supporting Online Material Material and Methods Live-cell DIC recordings PtK 1 cells (ATCC, Manassas, VA) were filmed on a Nikon (Nikon Instruments, Melville, NY) inverted microscope equipped with 60x
Specificity: Induced Fit
 Specificity: Induced Fit Conformational changes may occur upon ligand binding (Daniel Koshland in 1958) This adaptation is called the induced fit Induced fit allows for tighter binding of the ligand Induced
Specificity: Induced Fit Conformational changes may occur upon ligand binding (Daniel Koshland in 1958) This adaptation is called the induced fit Induced fit allows for tighter binding of the ligand Induced
The Intriguing Dual Lattices of the Myosin Filaments in Vertebrate Striated Muscles: Evolution and Advantage
 Biology 2014, 3, 846-865; doi:10.3390/biology3040846 Review OPEN ACCESS biology ISSN 2079-7737 www.mdpi.com/journal/biology The Intriguing Dual Lattices of the Myosin Filaments in Vertebrate Striated Muscles:
Biology 2014, 3, 846-865; doi:10.3390/biology3040846 Review OPEN ACCESS biology ISSN 2079-7737 www.mdpi.com/journal/biology The Intriguing Dual Lattices of the Myosin Filaments in Vertebrate Striated Muscles:
A COMPARATIVE STUDY OF NUCLEOCYTOPLASMIC INTERACTIONS
 A COMPARATIVE STUDY OF NUCLEOCYTOPLASMIC INTERACTIONS CARL M. FELDHERR. From the Department of Anatomical Sciences, University of Florida, Gainesville, Florida 32601 It is evident that the pores of the
A COMPARATIVE STUDY OF NUCLEOCYTOPLASMIC INTERACTIONS CARL M. FELDHERR. From the Department of Anatomical Sciences, University of Florida, Gainesville, Florida 32601 It is evident that the pores of the
Titin Actin Interaction in Mouse Myocardium: Passive Tension Modulation and Its Regulation by Calcium/S100A1
 Biophysical Journal Volume 81 October 2001 2297 2313 2297 Titin Actin Interaction in Mouse Myocardium: Passive Tension Modulation and Its Regulation by Calcium/S100A1 R. Yamasaki,* M. Berri, Y. Wu,* K.
Biophysical Journal Volume 81 October 2001 2297 2313 2297 Titin Actin Interaction in Mouse Myocardium: Passive Tension Modulation and Its Regulation by Calcium/S100A1 R. Yamasaki,* M. Berri, Y. Wu,* K.
THE MOLECULAR BASIS OF CROSS-BRIDGE FUNCTION
 THE MOLECULAR BASIS OF CROSS-BRIDGE FUNCTION Kenneth C. Holmes 1. INTRODUCTION Our understanding of the physiology of muscle depends critically on the resolution of the available anatomy. Early insight
THE MOLECULAR BASIS OF CROSS-BRIDGE FUNCTION Kenneth C. Holmes 1. INTRODUCTION Our understanding of the physiology of muscle depends critically on the resolution of the available anatomy. Early insight
ImmunolocQlizQtion of Type VI Collagen in Developing and Healing Rabbit Cornea
 Investigative Ophthalmology & Visual Science, Vol. 31, No. 6, June 1990 Copyright Association for Research in Vision and Ophthalmology ImmunolocQlizQtion of Type VI Collagen in Developing and Healing Rabbit
Investigative Ophthalmology & Visual Science, Vol. 31, No. 6, June 1990 Copyright Association for Research in Vision and Ophthalmology ImmunolocQlizQtion of Type VI Collagen in Developing and Healing Rabbit
Human CD36/SR-B3 ELISA
 K-ASSAY Human CD36/SR-B3 ELISA For the quantitative determination of CD36/SR-B3 in human cell culture supernates, serum and plasma (heparin, EDTA) Cat. No. KT-1200 For Research Use Only. 1 Rev. 12842343
K-ASSAY Human CD36/SR-B3 ELISA For the quantitative determination of CD36/SR-B3 in human cell culture supernates, serum and plasma (heparin, EDTA) Cat. No. KT-1200 For Research Use Only. 1 Rev. 12842343
Methodology for Immunohistochemistry. Learning Objectives:
 Proteomics Methodology for Immunohistochemistry Methodology for Immunohistochemistry A staining process for identifying the proteins location in cells, tissues by using antigen-antibody property. Immuno
Proteomics Methodology for Immunohistochemistry Methodology for Immunohistochemistry A staining process for identifying the proteins location in cells, tissues by using antigen-antibody property. Immuno
ANAT Cell Biology Lecture 11 School of Medical Sciences The University of New South Wales. UNSW Copyright Notice
 ANAT3231 - Cell Biology Lecture 11 School of Medical Sciences The University of New South Wales The actin cytoskeleton Prof Peter Gunning Oncology Research Unit Room 502A Wallace Wurth Building Email:
ANAT3231 - Cell Biology Lecture 11 School of Medical Sciences The University of New South Wales The actin cytoskeleton Prof Peter Gunning Oncology Research Unit Room 502A Wallace Wurth Building Email:
PROTEIN L AGAROSE BEADS
 DESCRIPTION Protein L is an immunoglobulin-binding protein that was isolated from the bacteria Peptostreptococcus magnus and provides a convenient way to separate immunoglobulins from a variety of sources.
DESCRIPTION Protein L is an immunoglobulin-binding protein that was isolated from the bacteria Peptostreptococcus magnus and provides a convenient way to separate immunoglobulins from a variety of sources.
Characterization of a 5.4 kb cdna fragment from the Z-line region of rabbit cardiac titin reveals phosphorylation sites for proline-directed kinases
 Journal of Cell Science 108, 3029-3037 (1995) Printed in Great Britain The Company of Biologists Limited 1995 3029 Characterization of a 5.4 kb cdna fragment from the Z-line region of rabbit cardiac titin
Journal of Cell Science 108, 3029-3037 (1995) Printed in Great Britain The Company of Biologists Limited 1995 3029 Characterization of a 5.4 kb cdna fragment from the Z-line region of rabbit cardiac titin
Anti-Piscirickettsia salmonis monoclonal antibody. Product no: P05
 Anti-Piscirickettsia salmonis monoclonal antibody Product no: P05 Product Description The monoclonal antibody (Mab) against Piscirickettsia salmonis is specific for this bacterium. The specificity of the
Anti-Piscirickettsia salmonis monoclonal antibody Product no: P05 Product Description The monoclonal antibody (Mab) against Piscirickettsia salmonis is specific for this bacterium. The specificity of the
T H E J O U R N A L O F C E L L B I O L O G Y
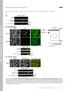 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Nakajima and Tanoue, http://www.jcb.org/cgi/content/full/jcb.201104118/dc1 Figure S1. DLD-1 cells exhibit the characteristic morphology
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Nakajima and Tanoue, http://www.jcb.org/cgi/content/full/jcb.201104118/dc1 Figure S1. DLD-1 cells exhibit the characteristic morphology
DENATURATION OF T4 DNA BY AN IN VITRO PROCESSED GENE 3ZPROTEIN
 DENATURATION OF T4 DNA BY AN IN VITRO PROCESSED GENE 3ZPROTEIN Junko HOSODA*, Bela TAKACS and Christine BRACK Department of Microbiology, Biozentrum University of Basel, Klingelbergstrasse 70, CH - 4056
DENATURATION OF T4 DNA BY AN IN VITRO PROCESSED GENE 3ZPROTEIN Junko HOSODA*, Bela TAKACS and Christine BRACK Department of Microbiology, Biozentrum University of Basel, Klingelbergstrasse 70, CH - 4056
Methods Western blot analysis of plg Quantification of plasminogen accumulation by ELISA Immunohistochemical analysis
 Methods Western blot analysis of plg Wild-type mice first received a standardized burn wound and then were intravenously administered 2 mg of human plg (Omnio AB, Umeå, Sweden). 24 hours after wounding
Methods Western blot analysis of plg Wild-type mice first received a standardized burn wound and then were intravenously administered 2 mg of human plg (Omnio AB, Umeå, Sweden). 24 hours after wounding
THE BASICS OF IMMUNOHISTOCHEMISTRY
 THE BASICS OF IMMUNOHISTOCHEMISTRY Introduction Immunohistochemistry (IHC) identifies specific tissue components by means of a specific antigen/antibody reaction tagged with a visible label. IHC makes
THE BASICS OF IMMUNOHISTOCHEMISTRY Introduction Immunohistochemistry (IHC) identifies specific tissue components by means of a specific antigen/antibody reaction tagged with a visible label. IHC makes
of titin and fibronectin
 Proc. Natl. Acad. Sci. USA Vol. 91, pp. 10114-10118, October 1994 Biophysics Reversible unfolding of fibronectin type III and immunoglobulin domains provides the structural basis for stretch and elasticity
Proc. Natl. Acad. Sci. USA Vol. 91, pp. 10114-10118, October 1994 Biophysics Reversible unfolding of fibronectin type III and immunoglobulin domains provides the structural basis for stretch and elasticity
(From the Division of Biological and Medical Research, Argonne National Laboratory, Lemont, Illinois) Methods and Materials
 OBSERVATIONS ON FIBRILLOGENESIS IN THE CONNECTIVE TISSUE OF THE CHICK EMBRYO WITH THE AID OF SILVER IMPREGNATION* BY F. WASSERMANN, M.D., AND L. KUBOTA (From the Division of Biological and Medical Research,
OBSERVATIONS ON FIBRILLOGENESIS IN THE CONNECTIVE TISSUE OF THE CHICK EMBRYO WITH THE AID OF SILVER IMPREGNATION* BY F. WASSERMANN, M.D., AND L. KUBOTA (From the Division of Biological and Medical Research,
ELECTRON-MICROSCOPIC OBSERVATIONS OF POLYOMA VIRUS-TRANSFORMED MOUSE CELLS TREATED WITH SPECIFIC IMMUNE SERUM
 J. Cell Sci. 7, 711-718(1970) 7II Printed in Great Britain ELECTRON-MICROSCOPIC OBSERVATIONS OF POLYOMA VIRUS-TRANSFORMED MOUSE CELLS TREATED WITH SPECIFIC IMMUNE SERUM G.NEGRONI AND RITA TILLY Department
J. Cell Sci. 7, 711-718(1970) 7II Printed in Great Britain ELECTRON-MICROSCOPIC OBSERVATIONS OF POLYOMA VIRUS-TRANSFORMED MOUSE CELLS TREATED WITH SPECIFIC IMMUNE SERUM G.NEGRONI AND RITA TILLY Department
Technical Manual No Version
 TUNEL Apoptosis Detection Kit Cat. No. L00301 (For Cryopreserved Tissue Sections, FITC-labled POD) Technical Manual No. 0269 Version 01132011 I Description. 1 II Key Features.... 1 III Kit Contents.. 1
TUNEL Apoptosis Detection Kit Cat. No. L00301 (For Cryopreserved Tissue Sections, FITC-labled POD) Technical Manual No. 0269 Version 01132011 I Description. 1 II Key Features.... 1 III Kit Contents.. 1
Cdc42 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
Department of Mechanical Engineering, University of Washington, Seattle, WA, 98195
 Supplementary information Contact Angle Changes Induced by Immunocomplex Formation Jong-Hoon Kim, a Amy Q. Shen, a Kyong-Hoon Lee, b Gerard A. Cangelosi c and Jae-Hyun Chung* a a Department of Mechanical
Supplementary information Contact Angle Changes Induced by Immunocomplex Formation Jong-Hoon Kim, a Amy Q. Shen, a Kyong-Hoon Lee, b Gerard A. Cangelosi c and Jae-Hyun Chung* a a Department of Mechanical
TUNEL Universal Apoptosis Detection Kit (FITC-labeled)
 TUNEL Universal Apoptosis Detection Kit (FITC-labeled) Cat. No. L00427 Technical Manual No. TM0623 Version 03102011 I Description. 1 II Key Features.... 1 III Kit Contents.. 1 IV Storage.. 2 V Protocol..
TUNEL Universal Apoptosis Detection Kit (FITC-labeled) Cat. No. L00427 Technical Manual No. TM0623 Version 03102011 I Description. 1 II Key Features.... 1 III Kit Contents.. 1 IV Storage.. 2 V Protocol..
THE EFFECTS OF CYTOCHALASIN B ON THE MICROFILAMENTS OF BABY HAMSTER KIDNEY (BHK-~21) CELLS
 THE EFFECTS OF CYTOCHALASIN B ON THE MICROFILAMENTS OF BABY HAMSTER KIDNEY (BHK-~21) CELLS ROBERT D. GOLDMAN From the Department of Biology, Case Western Reserve University, Cleveland, Ohio 44106 ABSTRACT
THE EFFECTS OF CYTOCHALASIN B ON THE MICROFILAMENTS OF BABY HAMSTER KIDNEY (BHK-~21) CELLS ROBERT D. GOLDMAN From the Department of Biology, Case Western Reserve University, Cleveland, Ohio 44106 ABSTRACT
Phalloidin-induced actin polymerization in the cytoplasm of
 Proc. Nati. Acad. Sci. USA Vol. 74, No. 12, pp. 5613-5617, December 1977 Cell Biology Phalloidin-induced actin polymerization in the cytoplasm of cultured cells interferes with cell locomotion and growth
Proc. Nati. Acad. Sci. USA Vol. 74, No. 12, pp. 5613-5617, December 1977 Cell Biology Phalloidin-induced actin polymerization in the cytoplasm of cultured cells interferes with cell locomotion and growth
Buffer (20X) Detergent (10X) NADH Reagent 1 (20X) Reagent 2 (100X) 96-well microplate (12 strips) 1
 PROTOCOL Complex I Enzyme Activity Microplate Assay Kit 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 MS141 Rev.1 DESCRIPTION Complex I Enzyme Activity Microplate Assay Kit Included in this kit is
PROTOCOL Complex I Enzyme Activity Microplate Assay Kit 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 MS141 Rev.1 DESCRIPTION Complex I Enzyme Activity Microplate Assay Kit Included in this kit is
2-step or indirect immunofluorescence 1. Substrate on which cells are plated: plastic vs. glass; coating vs. non
 Variables in standard immunostaining protocol 2-step or indirect immunofluorescence 1. Substrate on which cells are plated: plastic vs. glass; coating vs. non 2. Plating density: sparse vs. confluent 3.
Variables in standard immunostaining protocol 2-step or indirect immunofluorescence 1. Substrate on which cells are plated: plastic vs. glass; coating vs. non 2. Plating density: sparse vs. confluent 3.
Human Fibronectin ELISA
 K-ASSAY Human Fibronectin ELISA For the quantitative determination of Fibronectin in human cell culture supernates, serum and plasma (heparin, EDTA, citrate) Cat. No. KT-1269 For Research Use Only. Not
K-ASSAY Human Fibronectin ELISA For the quantitative determination of Fibronectin in human cell culture supernates, serum and plasma (heparin, EDTA, citrate) Cat. No. KT-1269 For Research Use Only. Not
IHC staining protocol. Paraffin, frozen and free-floating sections
 IHC staining protocol Paraffin, frozen and free-floating sections IHC staining protocol Contents Paraffin and frozen sections Immunostaining free-floating sections Signal amplification Paraffin and frozen
IHC staining protocol Paraffin, frozen and free-floating sections IHC staining protocol Contents Paraffin and frozen sections Immunostaining free-floating sections Signal amplification Paraffin and frozen
Online Supplement ALVEOLAR CELL SENESCENCE IN PATIENTS WITH PULMONARY EMPHYSEMA. Takao Tsuji, Kazutetsu Aoshiba, and Atsushi Nagai
 Online Supplement ALVEOLAR CELL SENESCENCE IN PATIENTS WITH PULMONARY EMPHYSEMA Takao Tsuji, Kazutetsu Aoshiba, and Atsushi Nagai MATERIALS AND METHODS Immunohistochemistry Deparaffinized tissue sections
Online Supplement ALVEOLAR CELL SENESCENCE IN PATIENTS WITH PULMONARY EMPHYSEMA Takao Tsuji, Kazutetsu Aoshiba, and Atsushi Nagai MATERIALS AND METHODS Immunohistochemistry Deparaffinized tissue sections
T HE stable and ordered arrangement of thick and
 The Structure of the Sarcomeric M Band: Localization of Defined Domains of Myomesin, M-protein, and the 250-kD Carboxy-terminal Region of Titin by Immunoelectron Microscopy Wolfgang M.J. Obermann,* Mathias
The Structure of the Sarcomeric M Band: Localization of Defined Domains of Myomesin, M-protein, and the 250-kD Carboxy-terminal Region of Titin by Immunoelectron Microscopy Wolfgang M.J. Obermann,* Mathias
Preliminary Biochemical Investigations of the Intermediate Filaments 1
 1970 by Academic Press, Inc. J. IJLTRASTRUCTURE RESEARCH 33, 399--407 (1970) 399 Preliminary Biochemical Investigations of the Intermediate Filaments 1 JOHN E. RASH, ~ JERRY W. SHAY, z AND JOHN J. BIESELE
1970 by Academic Press, Inc. J. IJLTRASTRUCTURE RESEARCH 33, 399--407 (1970) 399 Preliminary Biochemical Investigations of the Intermediate Filaments 1 JOHN E. RASH, ~ JERRY W. SHAY, z AND JOHN J. BIESELE
Supplementary Materials for
 www.advances.sciencemag.org/cgi/content/full/1/1/e1400205/dc1 Supplementary Materials for Myosin-binding protein C corrects an intrinsic inhomogeneity in cardiac excitation-contraction coupling Michael
www.advances.sciencemag.org/cgi/content/full/1/1/e1400205/dc1 Supplementary Materials for Myosin-binding protein C corrects an intrinsic inhomogeneity in cardiac excitation-contraction coupling Michael
Supplementary Note DNA Duplex Design and Construction
 Supplementary Note DNA Duplex Design and Construction Double-stranded DNA was amplified from a M13mp18 plasmid (Bayou Biolabs) by Autosticky PCR 1, which in our hands gave higher yields than asymmetric
Supplementary Note DNA Duplex Design and Construction Double-stranded DNA was amplified from a M13mp18 plasmid (Bayou Biolabs) by Autosticky PCR 1, which in our hands gave higher yields than asymmetric
Role of Ca 2 and Cross-Bridges in Skeletal Muscle Thin Filament Activation Probed with Ca 2 Sensitizers
 2166 Biophysical Journal Volume 76 April 1999 2166 2176 Role of Ca 2 and Cross-Bridges in Skeletal Muscle Thin Filament Activation Probed with Ca 2 Sensitizers Philip A. Wahr and Joseph M. Metzger Department
2166 Biophysical Journal Volume 76 April 1999 2166 2176 Role of Ca 2 and Cross-Bridges in Skeletal Muscle Thin Filament Activation Probed with Ca 2 Sensitizers Philip A. Wahr and Joseph M. Metzger Department
Which hydrogel preparation for immunostaining protocol should I use?
 Protocol: Preparation of TissueSpec hydrogels for immunostaining This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec matrix hydrogels.
Protocol: Preparation of TissueSpec hydrogels for immunostaining This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec matrix hydrogels.
PROTOCOL TO PREPARE PLANTAR FOOTSKIN FOR MORPHOMETRY. I. Removal and Fixation of Plantar Skin (see video)
 PROTOCOL TO PREPARE PLANTAR FOOTSKIN FOR MORPHOMETRY I. Removal and Fixation of Plantar Skin (see video) 1. Sacrifice the animal a. Anaesthetize the animal by placing in a closed chamber with isoflurane.
PROTOCOL TO PREPARE PLANTAR FOOTSKIN FOR MORPHOMETRY I. Removal and Fixation of Plantar Skin (see video) 1. Sacrifice the animal a. Anaesthetize the animal by placing in a closed chamber with isoflurane.
Anti-Cobia (Rachycentron canadum) monoclonal antibody. Product no: F18
 Anti-Cobia (Rachycentron canadum) monoclonal antibody Product no: F18 Product Description This monoclonal antibody (Mab) reacts with Cobia (Rachycentron canadum) immunoglobulin M (IgM). The Mab is of an
Anti-Cobia (Rachycentron canadum) monoclonal antibody Product no: F18 Product Description This monoclonal antibody (Mab) reacts with Cobia (Rachycentron canadum) immunoglobulin M (IgM). The Mab is of an
T H E J O U R N A L O F C E L L B I O L O G Y
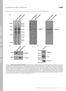 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Bays et al., http://www.jcb.org/cgi/content/full/jcb.201309092/dc1 Figure S1. Specificity of the phospho-y822 antibody. (A) Total cell
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Bays et al., http://www.jcb.org/cgi/content/full/jcb.201309092/dc1 Figure S1. Specificity of the phospho-y822 antibody. (A) Total cell
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Integrative Biology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Table S1. Definition of quantitative cellular features
Electronic Supplementary Material (ESI) for Integrative Biology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Table S1. Definition of quantitative cellular features
(phosphatase tensin) domain is shown in dark gray, the FH1 domain in black, and the
 Supplemental Figure 1. Predicted Domain Organization of the AFH14 Protein. (A) Schematic representation of the predicted domain organization of AFH14. The PTEN (phosphatase tensin) domain is shown in dark
Supplemental Figure 1. Predicted Domain Organization of the AFH14 Protein. (A) Schematic representation of the predicted domain organization of AFH14. The PTEN (phosphatase tensin) domain is shown in dark
Materials and Methods Materials Required for Fixing, Embedding and Sectioning
 Page 1 Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigenspecific antibodies coupled to fluorophores. Depending on the antibody or dye used, proteins,
Page 1 Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigenspecific antibodies coupled to fluorophores. Depending on the antibody or dye used, proteins,
Supplemental data. Supplemental Materials and Methods
 Supplemental data Supplemental Materials and Methods Transfection of plasmid. Transfection of plasmids into FRTL5 cells was performed using Lipofectamine LTX with Plus reagent (Invitrogen) according to
Supplemental data Supplemental Materials and Methods Transfection of plasmid. Transfection of plasmids into FRTL5 cells was performed using Lipofectamine LTX with Plus reagent (Invitrogen) according to
Anti-Bluefin Tuna (Thunnus thynnus) monoclonal antibody. Product no: F19
 Anti-Bluefin Tuna (Thunnus thynnus) monoclonal antibody Product no: F19 Product Description This monoclonal antibody (Mab) reacts with Bluefin Tuna (Thunnus thynnus) immunoglobulin M (IgM). The Mab is
Anti-Bluefin Tuna (Thunnus thynnus) monoclonal antibody Product no: F19 Product Description This monoclonal antibody (Mab) reacts with Bluefin Tuna (Thunnus thynnus) immunoglobulin M (IgM). The Mab is
WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits
 WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits Code N221-KIT N220-KIT Description WesternMAX Chemiluminescent AP Kit, Anti-Mouse Includes: Alkaline Phosphatase (AP) Conjugated Anti-Mouse
WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits Code N221-KIT N220-KIT Description WesternMAX Chemiluminescent AP Kit, Anti-Mouse Includes: Alkaline Phosphatase (AP) Conjugated Anti-Mouse
Technical Note. Tissue Section Imaging. Published August The most recent version of this Technical Note is posted at licor.com/bio/support.
 Technical Note Tissue Section Imaging Published August 2017. The most recent version of this Technical Note is posted at licor.com/bio/support. Page 2 - Tissue Section Imaging Table of Contents Page I.
Technical Note Tissue Section Imaging Published August 2017. The most recent version of this Technical Note is posted at licor.com/bio/support. Page 2 - Tissue Section Imaging Table of Contents Page I.
Supporting Protocols
 Supporting Protocols This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec ECM Hydrogels. Introduction Cells and organoids may form complex
Supporting Protocols This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec ECM Hydrogels. Introduction Cells and organoids may form complex
A PRODUCT FROM KANTAFLEX (INDIA) PVT LIMITED
 ELASTOMERIC BRIDGE BEARING TO LATEST IRC: 83-015 (PART - II) Kanta System of Elastomeric bridge bearing is made out of Poly chloroprene rubber having low crystallization rates and adequate shelf life,
ELASTOMERIC BRIDGE BEARING TO LATEST IRC: 83-015 (PART - II) Kanta System of Elastomeric bridge bearing is made out of Poly chloroprene rubber having low crystallization rates and adequate shelf life,
Cellular UV-Induced DNA Damage Staining Kit (CPD)
 Cellular UV-Induced DNA Damage Staining Kit (CPD) For the rapid detection of CPDs in genomic DNA of cultured cells Cat. No. KT-918 For Research Use Only. Not for use in diagnostic procedures. Page 1 of
Cellular UV-Induced DNA Damage Staining Kit (CPD) For the rapid detection of CPDs in genomic DNA of cultured cells Cat. No. KT-918 For Research Use Only. Not for use in diagnostic procedures. Page 1 of
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Craft et al., http://www.jcb.org/cgi/content/full/jcb.201409036/dc1 Figure S1. GFP -tubulin interacts with endogenous -tubulin. Ciliary
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Craft et al., http://www.jcb.org/cgi/content/full/jcb.201409036/dc1 Figure S1. GFP -tubulin interacts with endogenous -tubulin. Ciliary
Supplementary Figure 1. Antibody-induced cargo release studied by native PAGE. A clear band corresponding to the cargo strand (lane 1) is visible.
 Supplementary Figure 1. Antibody-induced cargo release studied by native PAGE. A clear band corresponding to the cargo strand (lane 1) is visible. Because SYBR Gold is less sensitive to single stranded
Supplementary Figure 1. Antibody-induced cargo release studied by native PAGE. A clear band corresponding to the cargo strand (lane 1) is visible. Because SYBR Gold is less sensitive to single stranded
