Rapid identification of candida species by TaqMan PCR
|
|
|
- Anabel Johns
- 5 years ago
- Views:
Transcription
1 362 Manchester Public Health Laboratory, Withington Hospital, Manchester M20 2LR, UK M Guiver K Levi B A Oppenheim Correspondence to: Dr Guiver mguiver@nw.phls.nhs.uk Accepted for publication 22 October 2000 Rapid identification of candida species by TaqMan PCR M Guiver, K Levi, B A Oppenheim Abstract Aim To develop and evaluate a Taq- Man polymerase chain reaction (PCR) for the rapid identification and speciation of candida species. Methods Species specific primer and probe sets were designed for the identification of Candida albicans, C parapsilosis, C tropicalis, C krusei, C kefyr, and C glabrata from clinical isolates in a 5' exonuclease (TaqMan ) assay. The probes were labelled with three fluorescent dyes to enable diverentiation between species when three primer and probe sets were combined in two multiplexes. The specificity of these assays was evaluated against a range of National Collection of Pathogenic Fungi strains, clinical isolates of yeast, bacterial and viral pathogens. Results The primer and probe sets have been shown to be 100% specific for their respective species; there was no crossreaction between any set and human DNA, or extracts from other candida species, fungal, bacterial, or viral pathogens tested. Extracts from two clinical isolates, originally identified as C albicans on the basis of germ tube formation, were not amplified by any of the primer and probe sets. These isolates have been putatively re-identified as C dubliniensis after sequencing of the variable internal transcribed spacer region ITS2 and lack of growth at 45 C. Conclusion This TaqMan assay provides a rapid alternative to conventional culture based techniques for the identification and speciation of the most frequently isolated candida species. The simple extraction method followed by TaqMan PCR can identify the six species mentioned in four hours. (J Clin Pathol 2001;54: ) Keywords: candida identification; TaqMan ; polymerase chain reaction The rise in incidence of opportunistic mucosal and systemic candida infections has been attributed to the increasing population of immunocompromised patients, intensive immunosuppressive treatment regimens, organ transplantation, and the use of broad spectrum antibiotics. 1 3 Long term treatment with amphotericin and fluconazole has led to antifungal drug resistance in Candida albicans and an increase in non-albicans candida infections. Certain yeast species, such as C krusei, are intrinsically resistant to fluconazole, and isolates of C glabrata and C tropicalis may become J Clin Pathol 2001;54: resistant. 4 Thus, rapid speciation of candida isolates allows appropriate clinical decisions to be made concerning targeted antifungal treatment and its dosage and duration. The identification of candida isolates to the species level using carbohydrate assimilation protocols 5 can be problematic, although primary isolation using CHROMagar can be used with reasonable specificity for rapid identification. Pan-fungal PCR assays have been evaluated for the detection of a broad spectrum of species, 6 7 as have candida specific primers 8 and C albicans specific and pan-fungal primers in conjunction with species specific probes In this approach, species specific primers and fluorescent probes have been designed for a 5' exonuclease (TaqMan ) assay for the six most clinically important candida species, namely: C albicans, C parapsilosis, C tropicalis, C krusei, C kefyr, and C glabrata. 12 The primer/ probe sets can be used individually or combined in two multiplex sets. Species can be differentiated within the multiplex by the use of three spectrally distinct fluorescent reporter dyes (FAM (6-carboxy-fluorescein), TET (tetrachloro-6-carboxy-fluorescein), or VIC ) attached to the 5' end of the probes. The TaqMan 5' exonuclease assay is a homogenous system using a fluorescent labelled probe for the detection of PCR product in real time. This provides a rapid automated combined PCR amplification and detection system with no postamplification manipulation of amplicons, thereby considerably reducing the risk of contamination. Coupled with a quick, simple DNA extraction method, this protocol allows rapid speciation of clinical isolates. Testing time is reduced from a mean of 3.5 days after the isolation of a yeast isolate 10 to four hours by the TaqMan procedure. Material and methods BACTERIAL AND FUNGAL STRAINS Fungal type strains were obtained from the National Collection of Pathogenic Fungi (NCPF, Public Health Laboratory Service (PHLS), Bristol) (tables 1 and 2). Fungal and bacterial clinical isolates used were obtained from the bacteriology and mycology departments of the PHLS, Manchester. Clinical isolates were identified by morphological and biochemical tests. Yeast species were also identified by means of the carbohydrate assimilation test API 20 Candida (BioMerieux, Lyon, France). DNA EXTRACTION Yeast and Gram positive bacterial strains DNA was extracted from overnight yeast colonies grown on Sabouraud medium at 30 C.
2 Rapid identification of candida species by TaqMan PCR 363 Table 1 Species specific TaqMan primer and probe sequences and fluorescent labels Table 2 Species tested against all primer/probe sets Species 5' to 3' primer and probe sequences and probe label Multiplex A Candida albicans F GGG TTT GCT TGA AAG ACG GTA R TTG AAG ATA TAC GTG GTG GAC GTT A P FAM-ACC TAA GCC ATT GTC AAA GCG ATC CCG C kefyr F GGC TGC GTG TCG AGT CTA TG R TGA CCC AAG CTT ACC ACG AAT P TET-ACT CTT GCA CAT CTA CGT CTT AGG TTT GCG C C glabrata F TTT CTC CTG CCT GCG CTT AA R ACG CAC ACT CCC AGG TCT TT P VIC-AGA ACA CCC ACC AAC CGC GCA Multiplex B C parapsilosis F GGG TTT GGT GTT GAG CGA TAC R GGA GTT TGT ACC AAT GAG TGG AAA P FAM-CTC CGC CTT TCT TTC AAG CAA ACC CAG C tropicalis F CGT GGT AAC TTA TTT TAA GCG R GCT TAA GTT CAG CGG GTA GTC CTA P TET-TGG CCA CCA TTT ATT TCA TAA CTT TGA CC C krusei F GCT GCG ACT CGC CTG AA R TTG TCT CGC AAC ACT CGC TCT P VIC-CTA GTT CGC TCG GCC AGC TTC GCT Probe labels are highlighted in bold. F, forward primer; R, reverse primer; P probe (phosphorylated at the 3' end). Single colonies were suspended in 1 ml sterile water and adjusted photometrically to an optical density of 0.1 at A 650. Gram positive bacterial cultures were suspended in sterile water and adjusted to approximately colony forming units (CFU)/ml by carrying out Miles and Misra viable counts for each of the bacterial isolates, and the concentrations adjusted accordingly. DNA was extracted by adding 100 µl of these dilutions to 1 ml DNAzol solution (Life Technologies, Glasgow, UK), vortexing, and incubation at room temperature for 10 minutes. DNA was precipitated by adding 1 ml ethanol and centrifuging at g for 10 minutes. The pellet was washed with 1 ml ethanol and resuspended in 50 µl sterile water. Gram negative bacterial strains Five colonies from an overnight culture were suspended in 500 µl sterile water and heated at 99 C for 10 minutes. The samples were transferred to 20 C for one minute to denature the DNA and were then centrifuged at g for 10 minutes. The supernatant was used directly as a PCR template. Aspergillus strains Aspergillus strains were incubated at 30 C on Sabouraud medium for 72 hours. Conidia were harvested into 0.01% Tween 20. DNA extraction was carried out according to Einsele et al, with minor modifications. 6 A 1 ml sample, containing CFU, was incubated with 5 ml white cell lysis buver (10 mm Tris/HCl (ph 7.4), 10 mm EDTA (ph 8.0), 50 mm NaCl, 0.2% lauryl sulphate, 200 µg proteinase K/ml (Sigma, Poole, UK)) at 65 C for two hours. After centrifugation for 15 minutes at 1500 g, 5 ml of supernatant was removed and discarded. The remaining 1 ml was transferred to a microcentrifuge tube and spun at g for 10 minutes. The pellet was resuspended in 50 mm NaOH and incubated at 95 C for 10 minutes. After centrifugation at 5000 g for 10 minutes the pellet was treated with 500 µl zymolase buver (50 mm Tris/HCl (ph 7.4), 10 mm EDTA, 28 mm β-mercaptoethanol, 300 µg zymolase/ml (ICN Species TaqMan result Candida albicans NCPF 3153 C albicans + C albicans NCPF 3281 C albicans + C albicans NCPF 8154 C albicans + C albicans NCPF 8154 C albicans + C tropicalis NCPF 3111 C tropicalis + C tropicalis NCPF 8113 C tropicalis + C tropicalis NCPF 8156 C tropicalis + C tropicalis NCPF 3980 C tropicalis + C tropicalis NCPF 8157 C tropicalis + C parapsilosis NCPF 3104 C parapsilosis + C parapsilosis NCPF 3938 C parapsilosis + C krusei NCPF 3100 C krusei + C krusei NCPF 3930 C krusei + C krusei NCPF 3953 C krusei + C kefyr NCPF 3106 C kefyr + C glabrata NCPF 3309 C glabrata + C glabrata NCPF 8018 C glabrata + C glabrata NCPF 3943 C glabrata + C glabrata NCPF 8155 C glabrata + C lusitaniae NCPF 3833 C pelliculosa NCPF 3356 C zeylanoides NCPF 3871 C viswanathiae NCPF 3151 C dubliniensis NCPF 3949 Saccharomyces cerevisiae NCPF 3178 Aspergillus fumigatus NCPF 2109 A flavus NCPF 2008 A terreus NCPF 2026 A niger NCPF 2275 A nidulans NCPF 2691 A versicolor NCPF 7246 Neisseria meningitidis (n = 2) N lactamica (n = 1) N gonorrhoeae (n = 1) Methicillin resistant Staphylococcus aureus (n=3) Coagulase negative staphylococci (n = 3) Group B streptococci (n = 3) Streptococcus pneumoniae (n = 3) Escherichia coli (n = 3) Moraxella cattharralis (n = 3) Pseudomonas aeruginosa (n = 3) Diptheroids (n = 3) Enterococcus faecalis (n = 5) Acinetobacter (n = 3) Klebsiella aerogenes (n = 3) Haemophilus influenzae (n = 3) Proteus mirabilis (n = 3) Cytomegalovirus (n = 3) Varicella zoster virus (n = 1) Herpes simplex virus (n = 3) Biomedicals, Ohio, USA)) at 37 C for 45 minutes. The spheroplast solution was pelleted at 5000 g for 10 minutes. The extraction procedure was completed according to the manufacturer s instructions for the tissue protocol included in the QIAamp DNA mini kit (Qiagen, Crawley, UK) and was eluted from the spin column using the supplied AE elution buver. Human genomic DNA extraction DNA was extracted from whole blood from a healthy volunteer with the Generation Capture Column kit (Gentra Systems, Minneapolis, USA), according to the manufacturer s instructions. Viral extracts DNA was extracted from 100 µl of hepatitis B positive serum and herpes simplex virus and varicella zoster virus tissue culture fluid by the addition of 1 ml DNAzol solution (Life Technologies), vortexing, and incubation at room temperature for 10 minutes. DNA was precipitated by adding 1 ml ethanol and centrifuging at g for 10 minutes. The pellet was washed with 1 ml ethanol and resuspended in 50 µl sterile water.
3 364 Guiver, Levi, Oppenheim DNA was extracted from blood samples from cytomegalovirus positive patients according to the instructions of the Generation Capture Column kit (Gentra Systems). SPECIFICITY A range of fungal bacterial and viral pathogens was evaluated in specificity tests with all candida primer and probe sets (table 2). In addition, the amplification of human genomic DNA, extracted from whole blood, was assessed. PRIMERS AND PROBES Oligonucleotide primer and probe sets were designed from the variable internal transcribed spacer region ITS for the specific amplification of C albicans, C tropicalis, C parapsilosis, C krusei, C kefyr, and C glabrata (table 1) using the Primer Express program (PE-ABI, Foster City, California, USA). Primers and probes were synthesised by PE-ABI (Warrington, UK); TaqMan probes were synthesised with one of three reporter dyes (FAM, TET, or VIC) (table 1) covalently linked to the 5' end and the quencher dye TAMRA (6- carboxytetremethylrhodamine) at the 3' ends. The 3' ends were phosphorylated to prevent extension of the probe. AMPLIFICATION USING THE PERKIN-ELMER APPLIED BIOSYSTEMS (PE-ABI) 7700 SEQUENCE DETECTION SYSTEM The 25 µl reaction contained 2 µl extracted sample with 12.5 µl TaqMan PCR Universal Master Mix (PE-ABI), 0.2 µm each primer, and 0.1 µm probe. In multiplex, the FAM and TET labelled probe concentrations were reduced to 50 nm. DNA amplification was carried out in 96 well microtitre plates sealed with MicroAmp optical caps (PE-ABI). The cycling programme on the PE-ABI 7700 sequence detection system consisted of heating for two minutes at 50 C and 95 C for 10 minutes, which preceded a two stage temperature profile of 95 C for 15 seconds and 60 C for one minute, repeated for 45 cycles. Data points collected at the end of each extension stage were analysed at the end of thermal cycling. During the first 15 cycles, data are collected from all wells and the background fluorescence is determined. A threshold value is calculated as 10 SD above the mean background fluorescence. A sample is considered positive if the fluorescence signal crosses this threshold within 45 cycles; the cycle number at which this occurs is recorded as the C T value. Table 3 Sample DNA SEQUENCING OF THE ITS2 REGION The 100 µl reaction was composed of 1.5 mm MgCl 2, 200 µm dntps, 500 nm each primer (ITS3 and ITS4), 9 and 2.5 U Taq DNA polymerase (Life Technologies) with the manufacturer s PCR buver (20 mm Tris/HCl (ph 8.4), 50 mm KCl) and 0.05% (vol/vol) of detergent W-1. The cycling programme was carried out in an automated thermal cycler (PTC 200 DNA Engine; MJ Research, Massachusetts, USA) and comprised denaturation at 94 C for one minute, followed by 35 cycles of 94 C for 30 seconds, 55 C for 30 seconds, and 72 C for 30 seconds. The final extension step was five minutes at 72 C. Amplicons were visualised on a 2% TBE gel, stained with ethidium bromide. PCR products were purified with GFX PCR DNA and Gel Band purification kit (Amersham Pharmacia Biotech, St Albans, UK), according to the manufacturer s instructions. Cycling sequencing was performed for 25 cycles of 95 C for 30 seconds, 50 C for 15 seconds, and 60 C for four minutes using Big Dye reaction mix (PE- ABI). After ethanol precipitation, automated sequencing was performed on an ABI 310 Genetic Analyser (PE-ABI) using the ITS3 and ITS4 primers. Results SPECIFICITY OF TaqMan PRIMER AND PROBE SETS Six primer/probe sets were designed for the specific identification of C albicans, C tropicalis, C parapsilosis, C krusei, C kefyr, and C glabrata (table 1). They were tested in a TaqMan assay against a range of NCPF candida strains and other fungal, bacterial, and viral pathogens (table 2). The primer/probe sets correctly identified 100% of NCPF strains without crossreaction with DNA from heterologous species (table 3). None of the six sets amplified DNA isolated from human whole blood. SPECIFICITY OF TaqMan PRIMER AND PROBE SETS FOR CLINICAL ISOLATES DNA extracts from a total of 80 clinical isolates were analysed with the TaqMan candida primer and probe sets (table 4). The C tropicalis, C parapsilosis, C krusei, C kefyr and C glabrata sets correctly identified all the isolates previously identified as these species. There was no crossreaction of any of the candida sets with C lusitaniae, C famata, or Cryptococcus neoformans isolates. One Saccharomyces cerevisiae isolate was identified as C parapsilosis with the TaqMan assay. Subsequent testing of this isolate revealed that it was a mixed culture of Specificity of candida primer and probe sets tested against NCPF strains and clinical isolates Mean C T value (SE) Candida albicans C parapsilosis C tropicalis C krusei C kefyr C glabrata C albicans (n = 11) (0.86) C parapsilosis (n = 13) (1.57) C tropicalis (n = 14) (0.63) C krusei (n = 12) (0.43) C kefyr (n = 4) (2.47) 45 C glabrata (n = 16) (0.55) NCPF, National Collection of Pathogenic Fungi.
4 Rapid identification of candida species by TaqMan PCR 365 Table 4 Identification of clinical isolates by candida TaqMan assay Mycology identification TaqMan result Final identification Candida albicans (n = 16) C albicans 14/16 C albicans 14/16 2/16 C dubliniensis* 2/16 C tropicalis (n = 12) C tropicalis 12/12 C tropicalis 12/12 C parapsilosis (n = 13) C parapsilosis 13/13 C parapsilosis 13/13 C krusei (n = 10) C krusei 10/10 C krusei 10/10 C kefyr (n=3) C kefyr 3/3 C kefyr 3/3 C glabrata (n = 12) C glabrata 12/12 C glabrata 12/12 Saccharomyces cerevisiae (n = 10) 9/10 S cerevisiae 9/10 C parapsilosis 1/10 S cerevisiae + C parapsilosis 1/10 Cryptococcus neoformans (n = 1) 1/1 Cryptococcus neoformans 1/1 C lusitaniae (n = 1) 1/1 C lusitaniae 1/1 C famata (n = 2) 2/2 C famata 2/2 Mycology identification: identification was based on API 20C and germ tube formation for C albicans. *Putative identification based on sequencing of the internal transcribed spacer region ITS2 region, germ tube formation, chlamydospore production, and lack of growth at 45 C. S cerevisiae and C parapsilosis. This result demonstrates that the specificity of the assay is not compromised by mixed culture isolates. Two isolates identified as C albicans as a result of germ tube formation and chlamydospore production were negative with all the specific primer and probe sets. Nucleotide sequence analysis of the ITS2 region was undertaken and comparison of this sequence with the ITS2 region from C dubliniensis (accession number U96719) showed 100% homology. The combination of these data, the lack of growth at 45 C of both isolates, and presence of chlamydospores and germ tubes enabled re-identification of both isolates as C dubliniensis. Discussion The prevalence of candida infection and the rise in species resistant to polyene and azole drugs means that rapid speciation of isolates has become increasingly important for targeted treatment. This is the first report of the use of species specific primers and probes in a rapid 5' exonuclease format for the detection of the six most clinically important candida species from clinical isolates. The primer and probe sets have been shown to have 100% specificity for their respective species, with no crossreaction with DNA extracts from other candida species, whole blood, or other fungal, bacterial, and viral species tested. This assay is currently being evaluated in our laboratory for the detection of candida species directly from clinical samples and, therefore, it was important to ensure during this evaluation that there was no crossreaction with other bacteria and viruses likely to be present in such samples. The assay combines a simple, rapid, and non-toxic (no phenol/chloroform) DNA extraction with Taq- Man amplification. The latter is a real time PCR format; therefore, no postamplification detection is necessary. The six primer and probe sets are combined into two multiplex reactions with each of the species specific probes labelled with a spectrally distinct fluorescent dye. The amplified species from the multiplex reaction can therefore be identified without further testing. In our study, species specific primer pairs have been used in conjunction with species specific probes. Previously described methods have combined pan-fungal primers with species specific probes for the detection of candida species The pan-fungal approach has led to specificity problems in some cases: Einsele and colleagues 6 noted crossreactions between the probe for C glabrata and C tropicalis DNA and between the C krusei probe and C parapsilosis extracts. A matrix system of probe hybridisation was used to diverentiate between crossreacting species. Non-specific hybridisation of C glabrata probes with S cerevisiae DNA has also been noted In addition, Elie and colleagues 10 observed crossreaction between their C albicans probe and DNA from C stellatoidea, although the separate status of type I C stellatoidea from C albicans has been questioned. 15 A candida specific probe was shown to hybridise with ITS3 and ITS4 amplified products from S cerevisiae, Aspergillus fumigatus, and A flavus, necessitating further analysis with aspergillus and S cerevisiae probes. 11 The presence of two putative C dubliniensis clinical isolates, originally identified as C albicans on the basis of germ tube formation and chlamydospore production, was highlighted by the absence of amplification with the C albicans primer and probe set. Germ tube formation, abundant chlamydospore production, lack of growth at 45 C, and sequence analysis of the ITS2 region amplified from these samples resulted in their suggested identification. Germ tube positive isolates are assumed to be C albicans in our laboratory; however, it might be useful to distinguish C dubliniensis species, which may rapidly develop resistance to fluconazole. 18 We are currently evaluating a C dubliniensis specific primer and probe set. This assay allows rapid speciation of isolates of C albicans, C parapsilosis, C tropicalis, Ckru- sei, C kefyr, and C glabrata to be undertaken in four hours, compared with a mean of 3.5 days reported for conventional phenotypic tests. 10 The estimated consumable cost for this assay is about 2.00, compared with 1.55 for the cost of a CHROMagar plate, although considerable capital investment is required for the PE-ABI 7700 Sequence Detection system. In a routine laboratory, this approach could be focused on the rapid identification of candida isolates from high risk patient groups for example, bone marrow transplant recipients, other patients on intensive immunosuppressive regimens, and patients in intensive treatment units where this information may guide prompt and appropriate treatment. The evectiveness of this assay for the direct identification of candida species from blood culture or clinical samples is currently being evaluated. 1 Jarvis WR. Epidemiology of nosocomial fungal infections, with emphasis on candida species. Clin Infect Dis 1995;20: Fridkin SK, Jarvis WR. Epidemiology of nosocomial fungal infections. Clin Microbiol Rev 1996;9: White TC, Marr KA, Bowden RA. Clinical, cellular and molecular factors that contribute to antifungal drug resistance. Clin Microbiol Rev 1998;11: Vanden Bossche H, Dromer F, Improvis I, et al. Antifungal drug resistance in pathogenic fungi. Med Mycol 1998;36:
5 366 Guiver, Levi, Oppenheim 5 Ainscough S, Kibbler CC. An evaluation of the costevectiveness of using CHROMagar for yeast identification in a routine microbiology laboratory. J Med Microbiol 1998; 47: Einsele H, Hebart H, Roller G, et al. Detection and identification of fungal pathogens in blood by using molecular probes. J Clin Microbiol 1997;35: van Burik J-A, Myerson D, Schreckhise RW, et al. Panfungal PCR assay for detection of fungal infection in human blood samples. J Clin Microbiol 1998;36: van Deventer AJM, Goessens WHF, van Belkum A, et al. Improved detection of Candida albicans by PCR in blood of neutropenic mice with systemic candidiasis J Clin Microbiol 1995;33: Fujita S-I, Lasker BA, Lott TJ, et al. Microtitration plate enzyme immunoassay to detect PCR-amplified DNA from candida species in blood. J Clin Microbiol 1995;33: Elie CM, Lott TJ, Reiss E, et al. Rapid identification of candida species with species-specific DNA probes. J Clin Microbiol 1998;36: Shin JH, Nolte FS, Holloway BP, et al. Rapid identification of up to three candida species in a single reaction tube by a 5' exonuclease assay using fluorescent DNA probes. J Clin Microbiol 1999;37: Readers' favourite Top 10 Click on the ''Top 10 '' button on the homepage to see which are the best read articles each month 12 Murray PR, Rosenthal KS, Kobayashi GS, et al. Opportunistic mycoses. In: Medical microbiology. St Louis: Mosby- Year Book Inc, Lott TJ, Kuykendall RJ, Reiss E. Nucleotide sequence analysis of the 5.8S rdna and adjacent ITS2 region of Candida albicans and relates species. Yeast 1993;9: Lott TJ, Burns BM, Zancope-Oliveira R, et al. Sequence analysis of the internal transcribed spacer 2 (ITS2) from yeast species within the genus candida. Curr Microbiol 1998;36: McCullough MJ, Clemons KV, Stevens DA. Molecular and phenotypic characterization of genotypic Candida albicans subgroups and comparison with Candida dubliniensis and Candida stellatoidea. J Clin Microbiol 1999;37: Sullivan D, Coleman D. Candida dubliniensis: characteristics and identification. J Clin Microbiol 1998;36: Odds FC, van NuVel L, Dams G. Prevalence of Candida dubliniensis isolates in a yeast stock collection. J Clin Microbiol 1998;36: Jabra-Rizk MA, Baqui AAMA, Kelley JI, et al. Identification of Candida dublinienisis in a prospective study of patients in the United States. J Clin Microbiol 1999;37: J Clin Pathol: first published as /jcp on 1 May Downloaded from on 22 August 2018 by guest. Protected by copyright.
Y-chromosomal haplogroup typing Using SBE reaction
 Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC
 Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR
 Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH).
 Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis
 1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
Supplemental Data Supplemental Figure 1.
 Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
MacBlunt PCR Cloning Kit Manual
 MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
Cat. # Product Size DS130 DynaExpress TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1
 Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Supplementary Figure 1A A404 Cells +/- Retinoic Acid
 Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
Supplemental material
 Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supporting Information
 Supporting Information Transfection of DNA Cages into Mammalian Cells Email: a.turberfield@physics.ox.ac.uk Table of Contents Supporting Figure 1 DNA tetrahedra used in transfection experiments 2 Supporting
Supporting Information Transfection of DNA Cages into Mammalian Cells Email: a.turberfield@physics.ox.ac.uk Table of Contents Supporting Figure 1 DNA tetrahedra used in transfection experiments 2 Supporting
Hes6. PPARα. PPARγ HNF4 CD36
 SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
Supplementary. Table 1: Oligonucleotides and Plasmids. complementary to positions from 77 of the SRα '- GCT CTA GAG AAC TTG AAG TAC AGA CTG C
 Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
Arabidopsis actin depolymerizing factor AtADF4 mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB
 Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells
 Supplementary Information for: PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells Ju Hye Jang 1, Hyun Kim 2, Mi Jung Jang 2, Ju Hyun Cho 1,2,* 1 Research Institute
Supplementary Information for: PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells Ju Hye Jang 1, Hyun Kim 2, Mi Jung Jang 2, Ju Hyun Cho 1,2,* 1 Research Institute
Legends for supplementary figures 1-3
 High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular
![strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular](/thumbs/72/66361212.jpg) Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Supplementary Information
 Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Supporting information for Biochemistry, 1995, 34(34), , DOI: /bi00034a013
 Supporting information for Biochemistry, 1995, 34(34), 10807 10815, DOI: 10.1021/bi00034a013 LESNIK 10807-1081 Terms & Conditions Electronic Supporting Information files are available without a subscription
Supporting information for Biochemistry, 1995, 34(34), 10807 10815, DOI: 10.1021/bi00034a013 LESNIK 10807-1081 Terms & Conditions Electronic Supporting Information files are available without a subscription
PCR analysis was performed to show the presence and the integrity of the var1csa and var-
 Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Supplemental Information. Human Senataxin Resolves RNA/DNA Hybrids. Formed at Transcriptional Pause Sites. to Promote Xrn2-Dependent Termination
 Supplemental Information Molecular Cell, Volume 42 Human Senataxin Resolves RNA/DNA Hybrids Formed at Transcriptional Pause Sites to Promote Xrn2-Dependent Termination Konstantina Skourti-Stathaki, Nicholas
Supplemental Information Molecular Cell, Volume 42 Human Senataxin Resolves RNA/DNA Hybrids Formed at Transcriptional Pause Sites to Promote Xrn2-Dependent Termination Konstantina Skourti-Stathaki, Nicholas
Supplemental Data. mir156-regulated SPL Transcription. Factors Define an Endogenous Flowering. Pathway in Arabidopsis thaliana
 Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
Gene synthesis by circular assembly amplification
 Gene synthesis by circular assembly amplification Duhee Bang & George M Church Supplementary figures and text: Supplementary Figure 1. Dpo4 gene (1.05kb) construction by various methods. Supplementary
Gene synthesis by circular assembly amplification Duhee Bang & George M Church Supplementary figures and text: Supplementary Figure 1. Dpo4 gene (1.05kb) construction by various methods. Supplementary
Disease and selection in the human genome 3
 Disease and selection in the human genome 3 Ka/Ks revisited Please sit in row K or forward RBFD: human populations, adaptation and immunity Neandertal Museum, Mettman Germany Sequence genome Measure expression
Disease and selection in the human genome 3 Ka/Ks revisited Please sit in row K or forward RBFD: human populations, adaptation and immunity Neandertal Museum, Mettman Germany Sequence genome Measure expression
Dierks Supplementary Fig. S1
 Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
ΔPDD1 x ΔPDD1. ΔPDD1 x wild type. 70 kd Pdd1. Pdd3
 Supplemental Fig. S1 ΔPDD1 x wild type ΔPDD1 x ΔPDD1 70 kd Pdd1 50 kd 37 kd Pdd3 Supplemental Fig. S1. ΔPDD1 strains express no detectable Pdd1 protein. Western blot analysis of whole-protein extracts
Supplemental Fig. S1 ΔPDD1 x wild type ΔPDD1 x ΔPDD1 70 kd Pdd1 50 kd 37 kd Pdd3 Supplemental Fig. S1. ΔPDD1 strains express no detectable Pdd1 protein. Western blot analysis of whole-protein extracts
I. Things to keep in mind before you start this protocol
 Inverse PCR and Sequencing Protocol on 5 Fly Preps For recovery of sequences flanking piggybac elements This protocol is an adaptation of "Inverse PCR and Cycle Sequencing Protocols" by E. Jay Rehm Berkeley
Inverse PCR and Sequencing Protocol on 5 Fly Preps For recovery of sequences flanking piggybac elements This protocol is an adaptation of "Inverse PCR and Cycle Sequencing Protocols" by E. Jay Rehm Berkeley
Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were
 1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
Supporting Online Information
 Supporting Online Information Isolation of Human Genomic DNA Sequences with Expanded Nucleobase Selectivity Preeti Rathi, Sara Maurer, Grzegorz Kubik and Daniel Summerer* Department of Chemistry and Chemical
Supporting Online Information Isolation of Human Genomic DNA Sequences with Expanded Nucleobase Selectivity Preeti Rathi, Sara Maurer, Grzegorz Kubik and Daniel Summerer* Department of Chemistry and Chemical
RPA-AB RPA-C Supplemental Figure S1: SDS-PAGE stained with Coomassie Blue after protein purification.
 RPA-AB RPA-C (a) (b) (c) (d) (e) (f) Supplemental Figure S: SDS-PAGE stained with Coomassie Blue after protein purification. (a) RPA; (b) RPA-AB; (c) RPA-CDE; (d) RPA-CDE core; (e) RPA-DE; and (f) RPA-C
RPA-AB RPA-C (a) (b) (c) (d) (e) (f) Supplemental Figure S: SDS-PAGE stained with Coomassie Blue after protein purification. (a) RPA; (b) RPA-AB; (c) RPA-CDE; (d) RPA-CDE core; (e) RPA-DE; and (f) RPA-C
ORFs and genes. Please sit in row K or forward
 ORFs and genes Please sit in row K or forward https://www.flickr.com/photos/teseum/3231682806/in/photostream/ Question: why do some strains of Vibrio cause cholera and others don t? Methods Mechanisms
ORFs and genes Please sit in row K or forward https://www.flickr.com/photos/teseum/3231682806/in/photostream/ Question: why do some strains of Vibrio cause cholera and others don t? Methods Mechanisms
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1 Characterization of GSCs. a. Immunostaining of primary GSC spheres from GSC lines. Nestin (neural progenitor marker, red), TLX (green). Merged images of nestin,
Supplementary Figures Supplementary Fig. 1 Characterization of GSCs. a. Immunostaining of primary GSC spheres from GSC lines. Nestin (neural progenitor marker, red), TLX (green). Merged images of nestin,
Multiplexing Genome-scale Engineering
 Multiplexing Genome-scale Engineering Harris Wang, Ph.D. Department of Systems Biology Department of Pathology & Cell Biology http://wanglab.c2b2.columbia.edu Rise of Genomics An Expanding Toolbox Esvelt
Multiplexing Genome-scale Engineering Harris Wang, Ph.D. Department of Systems Biology Department of Pathology & Cell Biology http://wanglab.c2b2.columbia.edu Rise of Genomics An Expanding Toolbox Esvelt
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/10/494/eaan6284/dc1 Supplementary Materials for Activation of master virulence regulator PhoP in acidic ph requires the Salmonella-specific protein UgtL Jeongjoon
www.sciencesignaling.org/cgi/content/full/10/494/eaan6284/dc1 Supplementary Materials for Activation of master virulence regulator PhoP in acidic ph requires the Salmonella-specific protein UgtL Jeongjoon
NESTED Sequence-based Typing (SBT) protocol for epidemiological typing of Legionella pneumophila directly from clinical samples
 NESTED Sequence-based Typing (SBT) protocol for epidemiological typing of Legionella pneumophila directly from clinical samples VERSION 2.0 SUMMARY This procedure describes the use of nested Sequence-Based
NESTED Sequence-based Typing (SBT) protocol for epidemiological typing of Legionella pneumophila directly from clinical samples VERSION 2.0 SUMMARY This procedure describes the use of nested Sequence-Based
Annex 6.5 CDC real-time rubella RT-PCR protocol targeting rubella p150 gene (154 nt region)
 Annex 6.5 CDC real-time rubella RT-PCR protocol targeting rubella p150 gene (154 nt region) NOTE: This document is intended to provide basic test method details and is not an SOP. Laboratories need to
Annex 6.5 CDC real-time rubella RT-PCR protocol targeting rubella p150 gene (154 nt region) NOTE: This document is intended to provide basic test method details and is not an SOP. Laboratories need to
Annex 6.2 CDC real-time measles RT-PCR protocol targeting measles N gene (75 nt region)
 Annex 6.2 CDC real-time measles RT-PCR protocol targeting measles N gene (75 nt region) NOTE: This document is intended to provide basic test method details and is not an SOP. Laboratories need to develop
Annex 6.2 CDC real-time measles RT-PCR protocol targeting measles N gene (75 nt region) NOTE: This document is intended to provide basic test method details and is not an SOP. Laboratories need to develop
Table S1. Bacterial strains (Related to Results and Experimental Procedures)
 Table S1. Bacterial strains (Related to Results and Experimental Procedures) Strain number Relevant genotype Source or reference 1045 AB1157 Graham Walker (Donnelly and Walker, 1989) 2458 3084 (MG1655)
Table S1. Bacterial strains (Related to Results and Experimental Procedures) Strain number Relevant genotype Source or reference 1045 AB1157 Graham Walker (Donnelly and Walker, 1989) 2458 3084 (MG1655)
An engineered tryptophan zipper-type peptide as a molecular recognition scaffold
 SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
Materials Protein synthesis kit. This kit consists of 24 amino acids, 24 transfer RNAs, four messenger RNAs and one ribosome (see below).
 Protein Synthesis Instructions The purpose of today s lab is to: Understand how a cell manufactures proteins from amino acids, using information stored in the genetic code. Assemble models of four very
Protein Synthesis Instructions The purpose of today s lab is to: Understand how a cell manufactures proteins from amino acids, using information stored in the genetic code. Assemble models of four very
Supporting Information
 Supporting Information Table S1. Oligonucleotide sequences used in this work Oligo DNA A B C D CpG-A CpG-B CpG-C CpG-D Sequence 5 ACA TTC CTA AGT CTG AAA CAT TAC AGC TTG CTA CAC GAG AAG AGC CGC CAT AGT
Supporting Information Table S1. Oligonucleotide sequences used in this work Oligo DNA A B C D CpG-A CpG-B CpG-C CpG-D Sequence 5 ACA TTC CTA AGT CTG AAA CAT TAC AGC TTG CTA CAC GAG AAG AGC CGC CAT AGT
Event-specific Method for the Quantification of Soybean SYHT0H2 by Real-time PCR. Validated Method
 EUROPEAN COMMISSION JOINT RESEARCH CENTRE Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification of Soybean SYHT0H2 by Real-time PCR
EUROPEAN COMMISSION JOINT RESEARCH CENTRE Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification of Soybean SYHT0H2 by Real-time PCR
Event-specific Method for the Quantification of Maize Line MIR604 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Maize Line MIR604 Using Real-time PCR Protocol 03 April 2007 Directorate General-Joint Research Centre Institute for Health and Consumer Protection Biotechnology
Event-specific Method for the Quantification of Maize Line MIR604 Using Real-time PCR Protocol 03 April 2007 Directorate General-Joint Research Centre Institute for Health and Consumer Protection Biotechnology
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Investigation of the Biosynthesis of the Lasso Peptide Chaxapeptin Using an E. coli-based Production System Helena Martin-Gómez, Uwe Linne, Fernando Albericio, Judit Tulla-Puche,*
SUPPORTING INFORMATION Investigation of the Biosynthesis of the Lasso Peptide Chaxapeptin Using an E. coli-based Production System Helena Martin-Gómez, Uwe Linne, Fernando Albericio, Judit Tulla-Puche,*
Event-specific Method for the Quantification of Maize Line GA21 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Maize Line GA21 Using Real-time PCR Protocol 30 March 2010 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Event-specific Method for the Quantification of Maize Line GA21 Using Real-time PCR Protocol 30 March 2010 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
PCR-based Markers and Cut Flower Longevity in Carnation
 PCRbased Markers and Cut Flower Longevity in Carnation Laura De Benedetti, Luca Braglia, Simona Bruna, Gianluca Burchi *, Antonio Mercuri and Tito Schiva Istituto Sperimentale per la Floricoltura, Corso
PCRbased Markers and Cut Flower Longevity in Carnation Laura De Benedetti, Luca Braglia, Simona Bruna, Gianluca Burchi *, Antonio Mercuri and Tito Schiva Istituto Sperimentale per la Floricoltura, Corso
Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR
 Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
Supplemental Data. Bennett et al. (2010). Plant Cell /tpc
 BRN1 ---------MSSSNGGVPPGFRFHPTDEELLHYYLKKKISYEKFEMEVIKEVDLNKIEPWDLQDRCKIGSTPQNEWYFFSHKDRKYPTGS 81 BRN2 --------MGSSSNGGVPPGFRFHPTDEELLHYYLKKKISYQKFEMEVIREVDLNKLEPWDLQERCKIGSTPQNEWYFFSHKDRKYPTGS 82 SMB
BRN1 ---------MSSSNGGVPPGFRFHPTDEELLHYYLKKKISYEKFEMEVIKEVDLNKIEPWDLQDRCKIGSTPQNEWYFFSHKDRKYPTGS 81 BRN2 --------MGSSSNGGVPPGFRFHPTDEELLHYYLKKKISYQKFEMEVIREVDLNKLEPWDLQERCKIGSTPQNEWYFFSHKDRKYPTGS 82 SMB
Supplementary Methods Quantitative RT-PCR. For mrna, total RNA was prepared using TRIzol reagent (Invitrogen) and genomic DNA was eliminated with TURB
 Supplementary Methods Quantitative RT-PCR. For mrna, total RNA was prepared using TRIzol reagent (Invitrogen) and genomic DNA was eliminated with TURBO DNA-free Kit (Ambion). One µg of total RNA was reverse
Supplementary Methods Quantitative RT-PCR. For mrna, total RNA was prepared using TRIzol reagent (Invitrogen) and genomic DNA was eliminated with TURBO DNA-free Kit (Ambion). One µg of total RNA was reverse
NAME:... MODEL ANSWER... STUDENT NUMBER:... Maximum marks: 50. Internal Examiner: Hugh Murrell, Computer Science, UKZN
 COMP710, Bioinformatics with Julia, Test One, Thursday the 20 th of April, 2017, 09h30-11h30 1 NAME:...... MODEL ANSWER... STUDENT NUMBER:...... Maximum marks: 50 Internal Examiner: Hugh Murrell, Computer
COMP710, Bioinformatics with Julia, Test One, Thursday the 20 th of April, 2017, 09h30-11h30 1 NAME:...... MODEL ANSWER... STUDENT NUMBER:...... Maximum marks: 50 Internal Examiner: Hugh Murrell, Computer
Anti-Pim-1 (Cat#3247), anti-met (Cat#3127), anti-ron (Cat#2654), Anti-EGFR
 Supplementary Methods Antibodies Anti-Pim-1 (Cat#3247), anti-met (Cat#3127), anti-ron (Cat#2654), Anti-EGFR (Cat#2646), anti-igf1r (Cat#3018), anti-insr (Cat#3020), anti-akt (pan, Cat#4691), anti-phospho-akt
Supplementary Methods Antibodies Anti-Pim-1 (Cat#3247), anti-met (Cat#3127), anti-ron (Cat#2654), Anti-EGFR (Cat#2646), anti-igf1r (Cat#3018), anti-insr (Cat#3020), anti-akt (pan, Cat#4691), anti-phospho-akt
Search for and Analysis of Single Nucleotide Polymorphisms (SNPs) in Rice (Oryza sativa, Oryza rufipogon) and Establishment of SNP Markers
 DNA Research 9, 163 171 (2002) Search for and Analysis of Single Nucleotide Polymorphisms (SNPs) in Rice (Oryza sativa, Oryza rufipogon) and Establishment of SNP Markers Shinobu Nasu, Junko Suzuki, Rieko
DNA Research 9, 163 171 (2002) Search for and Analysis of Single Nucleotide Polymorphisms (SNPs) in Rice (Oryza sativa, Oryza rufipogon) and Establishment of SNP Markers Shinobu Nasu, Junko Suzuki, Rieko
Supplementary Information. Construction of Lasso Peptide Fusion Proteins
 Supplementary Information Construction of Lasso Peptide Fusion Proteins Chuhan Zong 1, Mikhail O. Maksimov 2, A. James Link 2,3 * Departments of 1 Chemistry, 2 Chemical and Biological Engineering, and
Supplementary Information Construction of Lasso Peptide Fusion Proteins Chuhan Zong 1, Mikhail O. Maksimov 2, A. James Link 2,3 * Departments of 1 Chemistry, 2 Chemical and Biological Engineering, and
hcd1tg/hj1tg/ ApoE-/- hcd1tg/hj1tg/ ApoE+/+
 ApoE+/+ ApoE-/- ApoE-/- H&E (1x) Supplementary Figure 1. No obvious pathology is observed in the colon of diseased ApoE-/me. Colon samples were fixed in 1% formalin and laid out in Swiss rolls for paraffin
ApoE+/+ ApoE-/- ApoE-/- H&E (1x) Supplementary Figure 1. No obvious pathology is observed in the colon of diseased ApoE-/me. Colon samples were fixed in 1% formalin and laid out in Swiss rolls for paraffin
SUPPLEMENTARY MATERIALS
 SUPPLEMENTARY MATERIALS Supplementary Table S1: Water sampling sites in Brisbane River and their characteristics Sampling sites GPS coordinates Site characteristics Suspected source of fecal pollution
SUPPLEMENTARY MATERIALS Supplementary Table S1: Water sampling sites in Brisbane River and their characteristics Sampling sites GPS coordinates Site characteristics Suspected source of fecal pollution
evaluated with UAS CLB eliciting UAS CIT -N Libraries increase in the
 Supplementary Figures Supplementary Figure 1: Promoter scaffold library assemblies. Many ensembless of libraries were evaluated in this work. As a legend, the box outline color in top half of the figure
Supplementary Figures Supplementary Figure 1: Promoter scaffold library assemblies. Many ensembless of libraries were evaluated in this work. As a legend, the box outline color in top half of the figure
II 0.95 DM2 (RPP1) DM3 (At3g61540) b
 Table S2. F 2 Segregation Ratios at 16 C, Related to Figure 2 Cross n c Phenotype Model e 2 Locus A Locus B Normal F 1 -like Enhanced d Uk-1/Uk-3 149 64 36 49 DM2 (RPP1) DM1 (SSI4) a Bla-1/Hh-0 F 3 111
Table S2. F 2 Segregation Ratios at 16 C, Related to Figure 2 Cross n c Phenotype Model e 2 Locus A Locus B Normal F 1 -like Enhanced d Uk-1/Uk-3 149 64 36 49 DM2 (RPP1) DM1 (SSI4) a Bla-1/Hh-0 F 3 111
11th Meeting of the Science Working Group. Lima, Peru, October 2012 SWG-11-JM-11
 11th Meeting of the Science Working Group Lima, Peru, 15-19 October 2012 Russian population genetics studies of jack mackerel in the South Pacific P.K.Afanasiev M.A.Rabchun A.I.Glubokov Introduction. In
11th Meeting of the Science Working Group Lima, Peru, 15-19 October 2012 Russian population genetics studies of jack mackerel in the South Pacific P.K.Afanasiev M.A.Rabchun A.I.Glubokov Introduction. In
Protocol Rf3 Community Reference Laboratory for GM Food and Feed
 EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Event-specific Method for the Quantification
EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Event-specific Method for the Quantification
SUPPLEMENTARY MATERIALS AND METHODS. E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1)
 SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
Lecture 11: Gene Prediction
 Lecture 11: Gene Prediction Study Chapter 6.11-6.14 1 Gene: A sequence of nucleotides coding for protein Gene Prediction Problem: Determine the beginning and end positions of genes in a genome Where are
Lecture 11: Gene Prediction Study Chapter 6.11-6.14 1 Gene: A sequence of nucleotides coding for protein Gene Prediction Problem: Determine the beginning and end positions of genes in a genome Where are
Overexpression Normal expression Overexpression Normal expression. 26 (21.1%) N (%) P-value a N (%)
 SUPPLEMENTARY TABLES Table S1. Alteration of ZNF322A protein expression levels in relation to clinicopathological parameters in 123 Asian and 74 Caucasian lung cancer patients. Asian patients Caucasian
SUPPLEMENTARY TABLES Table S1. Alteration of ZNF322A protein expression levels in relation to clinicopathological parameters in 123 Asian and 74 Caucasian lung cancer patients. Asian patients Caucasian
Supplemental Table 1. Primers used for PCR.
 Supplemental Table 1. Primers used for PCR. Gene Type Primer Sequence Genotyping and semi-quantitative RT-PCR F 5 -TTG CCC GAT CAC CAT CTG TA-3 rwa1-1 R 5 -TGT AGC GAT CAA GGC CTG ATC TAA-3 LB 5 -TAG CAT
Supplemental Table 1. Primers used for PCR. Gene Type Primer Sequence Genotyping and semi-quantitative RT-PCR F 5 -TTG CCC GAT CAC CAT CTG TA-3 rwa1-1 R 5 -TGT AGC GAT CAA GGC CTG ATC TAA-3 LB 5 -TAG CAT
2
 1 2 3 4 5 6 7 Supplemental Table 1. Magnaporthe oryzae strains generated in this study. Strain background Genotype Strain name Description Guy-11 H1:RFP H1:RFP Strain expressing Histone H1- encoding gene
1 2 3 4 5 6 7 Supplemental Table 1. Magnaporthe oryzae strains generated in this study. Strain background Genotype Strain name Description Guy-11 H1:RFP H1:RFP Strain expressing Histone H1- encoding gene
S4B fluorescence (AU)
 A S4B fluorescence (AU) S4B fluorescence (AU) dsbb csgba csgd dsbb csgba bcsa 5000 * NS NS 4000 * 3000 2000 1000 0 ΔcsgBAΔbcsA ΔcsgDΔdsbBΔbcsA ΔcsgBA ΔdsbBΔcsgBA ΔcsgDΔdsbB B -1000 4000 * * NS 3500 * 3000
A S4B fluorescence (AU) S4B fluorescence (AU) dsbb csgba csgd dsbb csgba bcsa 5000 * NS NS 4000 * 3000 2000 1000 0 ΔcsgBAΔbcsA ΔcsgDΔdsbBΔbcsA ΔcsgBA ΔdsbBΔcsgBA ΔcsgDΔdsbB B -1000 4000 * * NS 3500 * 3000
Event-specific Method for the Quantification of Soybean Event DP Using Real-time PCR. Validated Method
 Event-specific Method for the Quantification of Soybean Event DP-356043-5 Using Real-time PCR Validated Method 29 March 2010 Joint Research Centre Institute for Health and Consumer Protection Molecular
Event-specific Method for the Quantification of Soybean Event DP-356043-5 Using Real-time PCR Validated Method 29 March 2010 Joint Research Centre Institute for Health and Consumer Protection Molecular
Method of Identifying Genetically Modified Papaya (PRSV-YK-resistant Strains) for Seeds and Leaves
 Method of Identifying Genetically Modified Papaya (PRSV-YK-resistant Strains) for Seeds and Leaves I. Points to Note All testing steps, including sample preparation, shall be performed in an area with
Method of Identifying Genetically Modified Papaya (PRSV-YK-resistant Strains) for Seeds and Leaves I. Points to Note All testing steps, including sample preparation, shall be performed in an area with
SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer
 TEACHER S GUIDE SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer SYNOPSIS This activity uses the metaphor of decoding a secret message for the Protein Synthesis process. Students teach themselves
TEACHER S GUIDE SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer SYNOPSIS This activity uses the metaphor of decoding a secret message for the Protein Synthesis process. Students teach themselves
Supporting Information. Trifluoroacetophenone-Linked Nucleotides and DNA for Studying of DNA-protein Interactions by 19 F NMR Spectroscopy
 Supporting Information Trifluoroacetophenone-Linked Nucleotides and DNA for Studying of DNA-protein Interactions by 19 F NMR Spectroscopy Agata Olszewska, Radek Pohl and Michal Hocek # * Institute of Organic
Supporting Information Trifluoroacetophenone-Linked Nucleotides and DNA for Studying of DNA-protein Interactions by 19 F NMR Spectroscopy Agata Olszewska, Radek Pohl and Michal Hocek # * Institute of Organic
Event-specific Method for the Quantification of Maize Line LY038 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Maize Line LY038 Using Real-time PCR Protocol 6 October 2008 Joint Research Centre Institute for Health and Consumer Protection Biotechnology & GMOs Unit
Event-specific Method for the Quantification of Maize Line LY038 Using Real-time PCR Protocol 6 October 2008 Joint Research Centre Institute for Health and Consumer Protection Biotechnology & GMOs Unit
G+C content. 1 Introduction. 2 Chromosomes Topology & Counts. 3 Genome size. 4 Replichores and gene orientation. 5 Chirochores.
 1 Introduction 2 Chromosomes Topology & Counts 3 Genome size 4 Replichores and gene orientation 5 Chirochores 6 7 Codon usage 121 marc.bailly-bechet@univ-lyon1.fr Bacterial genome structures Introduction
1 Introduction 2 Chromosomes Topology & Counts 3 Genome size 4 Replichores and gene orientation 5 Chirochores 6 7 Codon usage 121 marc.bailly-bechet@univ-lyon1.fr Bacterial genome structures Introduction
Event-specific Method for the Quantification of Oilseed Rape RF1 using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Oilseed Rape RF1 using Real-time PCR Protocol 7 July 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Event-specific Method for the Quantification of Oilseed Rape RF1 using Real-time PCR Protocol 7 July 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system
 Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system Dr. Tim Welsink Molecular Biology Transient Gene Expression OUTLINE Short
Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system Dr. Tim Welsink Molecular Biology Transient Gene Expression OUTLINE Short
Detection and quantification of bovine polyomavirus by real-time PCR
 Page 1 of 5 EU FP VII PROJECT VITAL STANDARD OPERATING CREATED: REVISED: APPROVED: David Rodríguez Lázaro: 18-02-2010 FERA: 28-03-2010 Wim Van der Poel: 31-03-2010 Page 2 of 5 WARNING All samples and controls
Page 1 of 5 EU FP VII PROJECT VITAL STANDARD OPERATING CREATED: REVISED: APPROVED: David Rodríguez Lázaro: 18-02-2010 FERA: 28-03-2010 Wim Van der Poel: 31-03-2010 Page 2 of 5 WARNING All samples and controls
Engineering D66N mutant using quick change site directed mutagenesis. Harkewal Singh 09/01/2010
 Engineering D66N mutant using quick change site directed mutagenesis Harkewal Singh 09/01/2010 1 1- What is quick change site directed mutagenesis? 2- An overview of the kit contents. 3- A brief information
Engineering D66N mutant using quick change site directed mutagenesis Harkewal Singh 09/01/2010 1 1- What is quick change site directed mutagenesis? 2- An overview of the kit contents. 3- A brief information
Sequencing of DNA lesions facilitated by site-specific excision via base. excision repair DNA glycosylases yielding ligatable gaps
 Supporting information Sequencing of DNA lesions facilitated by site-specific excision via base excision repair DNA glycosylases yielding ligatable gaps Jan Riedl, Aaron M. Fleming, and Cynthia J. Burrows*
Supporting information Sequencing of DNA lesions facilitated by site-specific excision via base excision repair DNA glycosylases yielding ligatable gaps Jan Riedl, Aaron M. Fleming, and Cynthia J. Burrows*
Protocol Ms8 Community Reference Laboratory for GM Food and Feed CRLVL06/04VP corrected version 1
 EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Event-specific Method for the Quantification
EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Event-specific Method for the Quantification
SUPPLEMENTAL MATERIAL GENOTYPING WITH MULTIPLEXING TARGETED RESEQUENCING
 SUPPLEMENTAL MATERIAL GENOTYPING WITH MULTIPLEXING TARGETED RESEQUENCING All of the patients and control subjects were sequenced and genotyped in the same way. Shotgun libraries of approximately 250 bp
SUPPLEMENTAL MATERIAL GENOTYPING WITH MULTIPLEXING TARGETED RESEQUENCING All of the patients and control subjects were sequenced and genotyped in the same way. Shotgun libraries of approximately 250 bp
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature07182 SUPPLEMENTAL FIGURES AND TABLES Fig. S1. myf5-expressing cells give rise to brown fat depots and skeletal muscle (a) Perirenal BAT from control (cre negative) and myf5-cre:r26r3-yfp
doi: 10.1038/nature07182 SUPPLEMENTAL FIGURES AND TABLES Fig. S1. myf5-expressing cells give rise to brown fat depots and skeletal muscle (a) Perirenal BAT from control (cre negative) and myf5-cre:r26r3-yfp
Wet Lab Tutorial: Genelet Circuits
 Wet Lab Tutorial: Genelet Circuits DNA 17 This tutorial will introduce the in vitro transcriptional circuits toolkit. The tutorial will focus on the design, synthesis, and experimental testing of a synthetic
Wet Lab Tutorial: Genelet Circuits DNA 17 This tutorial will introduce the in vitro transcriptional circuits toolkit. The tutorial will focus on the design, synthesis, and experimental testing of a synthetic
Event-specific Method for the Quantification of Cotton Line GHB614 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Cotton Line GHB614 Using Real-time PCR Protocol 5 September 2008 Joint Research Centre Institute for Health and Consumer Protection Biotechnology & GMOs
Event-specific Method for the Quantification of Cotton Line GHB614 Using Real-time PCR Protocol 5 September 2008 Joint Research Centre Institute for Health and Consumer Protection Biotechnology & GMOs
Project 07/111 Final Report October 31, Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines
 Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
Primer Design Workshop. École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria
 Primer Design Workshop École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria Scenario You have discovered the presence of a novel endophy5c organism living inside the cells
Primer Design Workshop École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria Scenario You have discovered the presence of a novel endophy5c organism living inside the cells
SUPPORTING INFORMATION FILE
 Intrinsic and extrinsic connections of Tet3 dioxygenase with CXXC zinc finger modules Nan Liu, Mengxi Wang, Wen Deng, Christine S. Schmidt, Weihua Qin, Heinrich Leonhardt and Fabio Spada Department of
Intrinsic and extrinsic connections of Tet3 dioxygenase with CXXC zinc finger modules Nan Liu, Mengxi Wang, Wen Deng, Christine S. Schmidt, Weihua Qin, Heinrich Leonhardt and Fabio Spada Department of
Event-specific Method for the Quantification of Cotton Event MON Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Cotton Event MON 88913 Using Real-time PCR Protocol 5 May 2009 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Event-specific Method for the Quantification of Cotton Event MON 88913 Using Real-time PCR Protocol 5 May 2009 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of
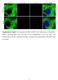 Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of the cell line) were immunostained for HA, acetylated
Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of the cell line) were immunostained for HA, acetylated
Event-specific Method for the Quantification of. Oilseed Rape Line RT73 Using Real-time PCR. Protocol. 07 February 2007
 EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Event-specific Method for the Quantification
EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Event-specific Method for the Quantification
Event-specific Method for the Quantification of Oilseed Rape Topas 19/2 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Oilseed Rape Topas 19/2 Using Real-time PCR Protocol 7 July 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and
Event-specific Method for the Quantification of Oilseed Rape Topas 19/2 Using Real-time PCR Protocol 7 July 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and
BioInformatics and Computational Molecular Biology. Course Website
 BioInformatics and Computational Molecular Biology Course Website http://bioinformatics.uchc.edu What is Bioinformatics Bioinformatics upgrades the information content of biological measurements. Discovery
BioInformatics and Computational Molecular Biology Course Website http://bioinformatics.uchc.edu What is Bioinformatics Bioinformatics upgrades the information content of biological measurements. Discovery
Event-specific Method for the Quantification of Soybean CV127 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Soybean CV127 Using Real-time PCR Protocol 20 September 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Event-specific Method for the Quantification of Soybean CV127 Using Real-time PCR Protocol 20 September 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Copy number estimation of P. falciparum pfmdr1 v1.1
 Copy number estimation of P. falciparum pfmdr1 v1.1 Procedure Molecular Module WorldWide Antimalarial Resistance Network (WWARN) Suggested citation: Molecular Module. 2011. Copy number estimation of P.
Copy number estimation of P. falciparum pfmdr1 v1.1 Procedure Molecular Module WorldWide Antimalarial Resistance Network (WWARN) Suggested citation: Molecular Module. 2011. Copy number estimation of P.
Yeast BioBrick Assembly (YBA)
 Yeast BioBrick Assembly (YBA) Standardized method for vector assembly of BioBrick devices via homologous recombination in Saccharomyces cerevisiae Martin Schneider, Leonard Fresenborg, Virginia Schadeweg
Yeast BioBrick Assembly (YBA) Standardized method for vector assembly of BioBrick devices via homologous recombination in Saccharomyces cerevisiae Martin Schneider, Leonard Fresenborg, Virginia Schadeweg
Event-specific Method for the Quantification of Cotton Line LLCotton25 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Cotton Line LLCotton25 Using Real-time PCR Protocol 14 March 2007 Directorate General Joint Research Centre Institute for Health and Consumer Protection
Event-specific Method for the Quantification of Cotton Line LLCotton25 Using Real-time PCR Protocol 14 March 2007 Directorate General Joint Research Centre Institute for Health and Consumer Protection
Event-specific Method for the Quantification of Soybean MON87708 Using Real-time PCR. Validated Method
 EUROPEAN COMMISSION JOINT RESEARCH CENTRE Ref. Ares(2013)1103954-15/05/2013 Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification
EUROPEAN COMMISSION JOINT RESEARCH CENTRE Ref. Ares(2013)1103954-15/05/2013 Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification
Supporting Information. Table of Contents
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2015 Supporting Information Spatial Regulation of a Common Precursor from Two Distinct Genes
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2015 Supporting Information Spatial Regulation of a Common Precursor from Two Distinct Genes
Amplified Analysis of DNA by the Autonomous Assembly of Polymers Consisting of DNAzyme Wires
 Supporting information for the paper: Amplified Analysis of DNA by the Autonomous Assembly of Polymers Consisting of DNAzyme Wires Fuan Wang, Johann Elbaz, Ron Orbach, Nimrod Magen and Itamar Willner*
Supporting information for the paper: Amplified Analysis of DNA by the Autonomous Assembly of Polymers Consisting of DNAzyme Wires Fuan Wang, Johann Elbaz, Ron Orbach, Nimrod Magen and Itamar Willner*
Electronic Supplementary Information - ESI
 Electronic Supplementary Information - ESI Centrifugal gas-phase transition magnetophoresis GTM a generic method for automation of magnetic bead based assays on the centrifugal microfluidic platform and
Electronic Supplementary Information - ESI Centrifugal gas-phase transition magnetophoresis GTM a generic method for automation of magnetic bead based assays on the centrifugal microfluidic platform and
Event-specific Method for the Quantification of Soybean DAS by Real-time PCR. Validated Method
 EUROPEAN COMMISSION JOINT RESEARCH CENTRE Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification of Soybean DAS-44406-6 by Real-time
EUROPEAN COMMISSION JOINT RESEARCH CENTRE Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification of Soybean DAS-44406-6 by Real-time
