Quality Systems Compliance LLC is your Compliance Partner
|
|
|
- Merry Thornton
- 5 years ago
- Views:
Transcription
1 Mark Durivage, Managing Principal Consultant and Member Quality Systems Compliance LLC 3118 Chanson Valley Road Lambertville, MI This table maps the clauses of ISO 9001:2015 to ISO 9001:2008 and is intended to help in the transition to the updated standard. It should be obvious there are some new and old requirements that do not fully map to the standards. The ISO 9001:2015 standard incorporates seven Quality Management Principles including; customer focus, leadership, engagement of people, process approach, improvement, evidence-based decision making, and relationship management all in the context of Risk-Based thinking. Before making the transition, it is strongly recommended to purchase and familiarize yourself with the following documents; ISO 9001: Quality management systems - Requirements, ISO 9000: Quality management systems -Fundamentals and vocabulary, ISO 9004: Managing for the sustained success of an organization - A quality management approach, and ISO 19011: Guidelines for auditing management systems. Let Quality Systems Compliance LLC help your company make a successful transition. Contact us today for a free consultation and quote. Quality Systems Compliance LLC is your Compliance Partner 1 Scope 1 Scope 2 Normative Reference 2 Normative Reference 3 Terms and Definitions 3 Terms and Definitions 4 Context of the Organization n/a 4.1 Understanding the organization and its context 4.2 Understanding the needs and expectations of interested parties
2 4.3 Determining the scope of the quality management system 4.4 Quality management system and its 4.1 General requirements processes 5 Leadership n/a 5.1 Leadership and commitment 5.1 Management commitment 5.2 Customer focus 5.2 Policy 5.3 Quality policy 5.3 Organizational roles, responsibilities Responsibility and authority and authorities 6 Planning n/a 6.1 Actions to address risks and opportunities 6.2 Quality objectives and planning to Quality objectives achieve them 6.3 Planning of changes Quality management system planning 7 Support n/a 7.1 Resources 6.1 Provision of resources General 6.1 Provision of resources People 6.2 Human resources Infrastructure 6.3 Infrastructure Environment for the operation of 6.4 Work environment processes Monitoring and measuring resources 7.6 Control of monitoring and measuring equipment Organizational knowledge 7.2 Competence 6.2 Competence, training and awareness 7.3 Awareness 6.2 Competence, training and awareness 7.4 Communication Internal communication Page 2 of 5
3 7.5 Documented Information Control of documents General Control of documents Creating and Updating Control of documents Control of Documented Information Control of documents 8 Operation n/a 8.1 Operational planning and control 7.1 Planning of product realization 8.2 Requirements for products and 7.2 Customer-related processes services Customer communication Customer communication Determination of requirements Determination of requirements related to related to the product Review of requirements related to Changes to requirements for Review of requirements related to the product Review of requirements related to the product 8.3 Design and development of 7.3 Design and development General Design and development planning Design and development planning Design and development inputs Design and development inputs Design and development controls Design and development review Design and development verification Design and development validation Design and development outputs Design and development outputs Page 3 of 5
4 8.3.6 Design and development changes Control of design and development changes 8.4 Control of externally provided processes, 7.4 Purchasing General Purchasing process Type and extent of control Purchasing process Verification of purchased product Information for external providers Purchasing information 8.5 Production and service 7.5 Production and service Control of production and service Control of production and service Validation of processes for production and service Identification and traceability Identification and traceability Property belonging to customers or external providers Customer property Preservation Preservation of product Post-delivery activities Control of production and service Determination of requirements related to the product Control of changes Control of documents Quality management system planning Control of design and development changes 8.6 Release of Monitoring and measurement of product 8.7 Control of nonconforming outputs 8.3 Control of nonconforming product 9 Performance evaluation n/a Page 4 of 5
5 9.1 Monitoring, measurement, analysis and evaluation 8.1 General 8.2 Monitoring and measurement General 8.1 General 8.2 Monitoring and measurement Customer satisfaction Customer satisfaction Analysis and evaluation 8.4 Analysis of data 9.2 Internal audit Internal audit 9.3 Management review 5.6 Management review General General Management review inputs Review input Management review outputs Review output 10 Improvement n/a 10.1 General 8.3 Control of nonconforming product 8.5 Improvement 10.2 Nonconformity and Corrective Action 8.3 Control of nonconforming product Corrective action 10.3 Continual Improvement Continual improvement Key Definitions context of the organization - is a process that determines factors which influence the organization s purpose, objectives and sustainability. It considers internal factors such as values, culture, knowledge and performance of the organization. It also considers external factors such as legal, technological, competitive, market, cultural, social and economic environments interested parties - are those that provide significant risk to organizational sustainability if their needs and expectations are not met Plan-Do-Check-Act (PDCA) is a four-step process for the control and continuous improvement of processes and products process approach - the systematic definition and management of processes, and their interactions, to achieve the intended results in accordance with the quality policy and strategic direction of the organization risk-based thinking addresses both risks and opportunities and establishes a basis for increasing the effectiveness of the quality management system, achieving improved results and preventing negative effects. Page 5 of 5
Correlation matrices between ISO 9001:2008 and ISO 9001:2015
 Correlation matrices between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 ISO 9001:2008 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system 4.1 Understanding the organization
Correlation matrices between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 ISO 9001:2008 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system 4.1 Understanding the organization
4. Quality management system 4. Context of the organization. 5 Management responsibility 5 Leadership
 AS 9100:2009 (Rev.C) AS 9100:2016 (Rev.D) 4. Quality management system 4. Context of the organization 4.1 General requirements 4.4 Quality management system and its processes products and 4.2 Documentation
AS 9100:2009 (Rev.C) AS 9100:2016 (Rev.D) 4. Quality management system 4. Context of the organization 4.1 General requirements 4.4 Quality management system and its processes products and 4.2 Documentation
ISO/TC 176/SC 2 Document N1224, July 2014
 ISO/TC 176/SC 2 Document N1224, July 2014 Correlation matrices between ISO 9001:2008 and ISO/DIS 9001 This document gives correlation matrices from ISO 9001:2008 to the current Draft International Standard
ISO/TC 176/SC 2 Document N1224, July 2014 Correlation matrices between ISO 9001:2008 and ISO/DIS 9001 This document gives correlation matrices from ISO 9001:2008 to the current Draft International Standard
ISO 13485: :2015 CLIENT TRANSITION CHECKLIST
 - 9001:2015 CLIENT TRANSITION CHECKLIST Audit Conclusions: All requirements have been addressed. The organization is recommended for ISO 13485:2016 certification. Recommendation for registration is dependent
- 9001:2015 CLIENT TRANSITION CHECKLIST Audit Conclusions: All requirements have been addressed. The organization is recommended for ISO 13485:2016 certification. Recommendation for registration is dependent
INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT
 INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT M a r ch 2015 OBJECTIVE ISO and Project Quality Management Process Are they different or the same? ISO 9000 QMS FAMILY ISO 9000:2005 Vocabulary
INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT M a r ch 2015 OBJECTIVE ISO and Project Quality Management Process Are they different or the same? ISO 9000 QMS FAMILY ISO 9000:2005 Vocabulary
9110 Correlation matrices
 9110 Correlation matrices 9110:2016 to 9110:2012 9110:2012 to 9110:2016 This document provides correlation matrices from 9110:2016 to 9110:2012 and 9110:2012 to 9110:2016. This document can be used to
9110 Correlation matrices 9110:2016 to 9110:2012 9110:2012 to 9110:2016 This document provides correlation matrices from 9110:2016 to 9110:2012 and 9110:2012 to 9110:2016. This document can be used to
Correlation Matrix & Change Summary
 The correlation matrix compares the new requirements of ISO 9001:2015 to the requirements of ISO 9001:2008, and provides a summary of the changes. Correlation Matrix & Change Summary Introduction Correlation
The correlation matrix compares the new requirements of ISO 9001:2015 to the requirements of ISO 9001:2008, and provides a summary of the changes. Correlation Matrix & Change Summary Introduction Correlation
ISO 9001:2015. Presented By: ASEAN Eng. DEXTER T. CHUA, PIE. Conference Room, University of Mindanao March 17, 2017
 ISO 9001:2015 Presented By: ASEAN Eng. DEXTER T. CHUA, PIE Conference Room, University of Mindanao March 17, 2017 @CQL Business Systems Consulting (11-2015) ISO The International Organization for Standardization
ISO 9001:2015 Presented By: ASEAN Eng. DEXTER T. CHUA, PIE Conference Room, University of Mindanao March 17, 2017 @CQL Business Systems Consulting (11-2015) ISO The International Organization for Standardization
ISO 9001:2015. Presented By: ASEAN Eng. DEXTER T. CHUA, PIE. November 25, Business Systems Consulting ( )
 ISO 9001:2015 Presented By: ASEAN Eng. DEXTER T. CHUA, PIE November 25, 2016 @CQL Business Systems Consulting (11-2015) ISO The International Organization for Standardization based in Geneva, Switzerland
ISO 9001:2015 Presented By: ASEAN Eng. DEXTER T. CHUA, PIE November 25, 2016 @CQL Business Systems Consulting (11-2015) ISO The International Organization for Standardization based in Geneva, Switzerland
Correlation Matrix EN 9100:2016 EN 9100:2009
 Correlation Matrix EN 9100:2016 EN 9100:2009 EN 9100:2016 EN 9100:2009 4 Context of the organization 4 Quality management system 4.2 Understanding the organization and its context Understanding the needs
Correlation Matrix EN 9100:2016 EN 9100:2009 EN 9100:2016 EN 9100:2009 4 Context of the organization 4 Quality management system 4.2 Understanding the organization and its context Understanding the needs
Clause Map IATF 16949:2016 to ISO/TS 16949:2009
 Table of Contents Table of Contents Foreword Foreword + Foreword Automotive QMS Standard + History + Goal + 0.5 Goal of this Technical Specification + Remarks for Certification + Remarks for Certification
Table of Contents Table of Contents Foreword Foreword + Foreword Automotive QMS Standard + History + Goal + 0.5 Goal of this Technical Specification + Remarks for Certification + Remarks for Certification
UNIVERSITY OF NAIROBI
 Q# UNIVERSITY OF NAIROBI 4 CONTEXTS OF THE ORGANIZATION Yes No Remarks 4.1 UNDERSTANDING THE ORGANIZATION AND ITS CONTEXT 1 Have we determined the external and internal issues that are relevant to our
Q# UNIVERSITY OF NAIROBI 4 CONTEXTS OF THE ORGANIZATION Yes No Remarks 4.1 UNDERSTANDING THE ORGANIZATION AND ITS CONTEXT 1 Have we determined the external and internal issues that are relevant to our
Business Management System: Requirements 00-BMS-002
 Business Management System: Requirements 00-BMS-002 Preface This document defines the applicability of International s requirements to the Business Management System (BMS) processes implemented at GM Nameplate
Business Management System: Requirements 00-BMS-002 Preface This document defines the applicability of International s requirements to the Business Management System (BMS) processes implemented at GM Nameplate
ISO 9001: Understanding the changes
 ISO 9001: 2015 Understanding the changes Tel: 01952 288325 Email: info@interface-nrm.co.uk Why has ISO 9001 been Revised? ISO 9001, the world's leading Quality Management System has been revised to a new
ISO 9001: 2015 Understanding the changes Tel: 01952 288325 Email: info@interface-nrm.co.uk Why has ISO 9001 been Revised? ISO 9001, the world's leading Quality Management System has been revised to a new
Executive Overview. Transitioning to ISO 9001:2015 Quality Management System. Biafore Associates Inc. Overview Objectives
 Executive Transitioning to ISO 9001:2015 Quality Management System Biafore Associates Inc. This guideline is for training purposes only; Not ISO controlled Objectives The overview objectives are as follows:
Executive Transitioning to ISO 9001:2015 Quality Management System Biafore Associates Inc. This guideline is for training purposes only; Not ISO controlled Objectives The overview objectives are as follows:
ISO 9001: 2015 Quality Management System Certification. Awareness Training
 ISO 9001: 2015 Quality Management System Certification Awareness Training ISO 9001: 2015 STRUCTURE The new standard is modeled around the ISO Directive Annex SL, a high level structure (HSL) based on the
ISO 9001: 2015 Quality Management System Certification Awareness Training ISO 9001: 2015 STRUCTURE The new standard is modeled around the ISO Directive Annex SL, a high level structure (HSL) based on the
QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES
 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract: This handbook documents
QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract: This handbook documents
This is a preview - click here to buy the full publication. IEC Quality Assessment System for Electronic Components (IECQ System)
 QC 080000 Edition 4.0 2017-05 IECQ PUBLICATION IEC Quality Assessment System for Electronic Components (IECQ System) Hazardous Substance Process Management (HSPM) System Requirements INTERNATIONAL ELECTROTECHNICAL
QC 080000 Edition 4.0 2017-05 IECQ PUBLICATION IEC Quality Assessment System for Electronic Components (IECQ System) Hazardous Substance Process Management (HSPM) System Requirements INTERNATIONAL ELECTROTECHNICAL
BUSINESS ASSURANCE GUIDANCE FOR THE TRANSITIONING FROM ISO 9001:2008 TO ISO 9001:2015 SAFER, SMARTER, GREENER
 BUSINESS ASSURANCE GUIDANCE FOR THE TRANSITIONING FROM ISO 9001:2008 TO ISO 9001:2015 SAFER, SMARTER, GREENER 2 ISO 9001 ABOUT THIS DOCUMENT The purpose of this document is to provide you with some useful
BUSINESS ASSURANCE GUIDANCE FOR THE TRANSITIONING FROM ISO 9001:2008 TO ISO 9001:2015 SAFER, SMARTER, GREENER 2 ISO 9001 ABOUT THIS DOCUMENT The purpose of this document is to provide you with some useful
ISO 9001:2015: Change or Not to Change or How to Transition from ISO9001:2008 and ISO9001:2015. Joan Richter ASQ Baltimore (Section 502) June 2016
 ISO 9001:2015: Change or Not to Change or How to Transition from ISO9001:2008 and ISO9001:2015 Joan Richter ASQ Baltimore (Section 502) June 2016 HISTORY OF ISO 9001 Standard (28 Years of ISO) 1987 Procedures
ISO 9001:2015: Change or Not to Change or How to Transition from ISO9001:2008 and ISO9001:2015 Joan Richter ASQ Baltimore (Section 502) June 2016 HISTORY OF ISO 9001 Standard (28 Years of ISO) 1987 Procedures
Correlation Matrix. ISO 9001:2008 and ISO 9001: INTRODUCTION CORRELATION MATRIX ISO 9001:2008 AND ISO 9001:2015
 Correlation Matrix ISO 9001:2008 and ISO 9001:2015 INTRODUCTION This document provides a correlation between the requirements of ISO 9001:2008 and the ISO 9001:2015 standard. The document will provide
Correlation Matrix ISO 9001:2008 and ISO 9001:2015 INTRODUCTION This document provides a correlation between the requirements of ISO 9001:2008 and the ISO 9001:2015 standard. The document will provide
Summary of changes between ISO 9001:2008 and ISO/CD 9001
 Summary of changes between ISO 9001:2008 and ISO/CD 9001 Introduction This document is intended to summarise the proposed changes to ISO 9001:2008 as detailed in the Committee Draft (CD). It does not contain
Summary of changes between ISO 9001:2008 and ISO/CD 9001 Introduction This document is intended to summarise the proposed changes to ISO 9001:2008 as detailed in the Committee Draft (CD). It does not contain
AESOP 15604; ISSUE 2; STATUS PENDING APPROVAL; AUTHORITY CARL BLAZIK This document is the property of NSF ISR. Page 1 of 9
 This document is the property of NSF ISR. Page 1 of 9 ISO 9001 Quality Management Systems registration provides a set of uniform requirements for a quality management system. A number of quality management
This document is the property of NSF ISR. Page 1 of 9 ISO 9001 Quality Management Systems registration provides a set of uniform requirements for a quality management system. A number of quality management
ISO 9001:2015. Quality Management System. Manual
 ISO 9001:2015 Quality Management System Manual Introduction Company has made the Strategic Business Decision to develop and implement an effective Quality Management Systems (QMS) across all areas of the
ISO 9001:2015 Quality Management System Manual Introduction Company has made the Strategic Business Decision to develop and implement an effective Quality Management Systems (QMS) across all areas of the
ISO 9001:2015 Readiness Review
 ISO 9001:2015 Readiness Review Company Name Address Certification No. Contact Name Job Title Telephone Email BSI is committed to ensuring a smooth assessment for all clients wishing to certify to ISO 9001:2015,
ISO 9001:2015 Readiness Review Company Name Address Certification No. Contact Name Job Title Telephone Email BSI is committed to ensuring a smooth assessment for all clients wishing to certify to ISO 9001:2015,
QUALITY POLICIES HANDBOOK
 QUALITY POLICIES HANDBOOK Origination Date: (mo/yr) Document Identifier: Date: Document Status: Document Link: Latest Revision Date Draft, Redline, Released, Obsolete Location on Server (if used) Abstract:
QUALITY POLICIES HANDBOOK Origination Date: (mo/yr) Document Identifier: Date: Document Status: Document Link: Latest Revision Date Draft, Redline, Released, Obsolete Location on Server (if used) Abstract:
Sections of the Standard. Evidence / Comments. (Y) / Nonconforming (NC)
 Date: Sections of the Standard 4 Context of the Organization 4.1 Understanding the organization and its context 4.2 Understanding the needs and expectations of interested parties 4.3 Determining the scope
Date: Sections of the Standard 4 Context of the Organization 4.1 Understanding the organization and its context 4.2 Understanding the needs and expectations of interested parties 4.3 Determining the scope
ISO 9001:2015 TRANSITION INFORMATION
 ISO 9001:2015 TRANSITION INFORMATION ISO 9001:2015 Transition It Doesn t Need to be Difficult Whether your organization is transitioning from ISO 9001:2008 to the new ISO 9001:2015, or implementing an
ISO 9001:2015 TRANSITION INFORMATION ISO 9001:2015 Transition It Doesn t Need to be Difficult Whether your organization is transitioning from ISO 9001:2008 to the new ISO 9001:2015, or implementing an
Quality system implementation from a manufacturers viewpoint
 OD005 IECEx Quality System Requirements for Manufacturers Quality system implementation from a manufacturers viewpoint Evans Massy Rockwell Automation October 3, 2005 31/10/2005 1 1515 1545 Manufacturer
OD005 IECEx Quality System Requirements for Manufacturers Quality system implementation from a manufacturers viewpoint Evans Massy Rockwell Automation October 3, 2005 31/10/2005 1 1515 1545 Manufacturer
Patsy Brown Brown & Associates Quality Consulting, Inc. Pine Bluff, AR Documents. Standards ISO 9001:2008 ISO 22000:2005 PAS 220:2008
 Patsy Brown Brown & Associates Quality Consulting, Inc. Pine Bluff, AR www.brownqc.com Documents Standards ISO 9001:2008 PAS 220:2008 Supporting Documentation ISO 9000:2005 ISO 22004:2005 ISO 19011:2002
Patsy Brown Brown & Associates Quality Consulting, Inc. Pine Bluff, AR www.brownqc.com Documents Standards ISO 9001:2008 PAS 220:2008 Supporting Documentation ISO 9000:2005 ISO 22004:2005 ISO 19011:2002
CO-ORDINATION OF NOTIFIED BODIES PPE Regulation 2016/425 RECOMMENDATION FOR USE
 CO-ORDINATION OF NOTIFIED BODIES PPE Regulation 2016/425 PPE-R/00.018 Version 1 RECOMMENDATION FOR USE Number of pages: 5 Approval stage : Approved on : Origin : Horizontal Committee, C2D Ad hoc group
CO-ORDINATION OF NOTIFIED BODIES PPE Regulation 2016/425 PPE-R/00.018 Version 1 RECOMMENDATION FOR USE Number of pages: 5 Approval stage : Approved on : Origin : Horizontal Committee, C2D Ad hoc group
Summary of ISO 9001:2015 New and Changed Requirements
 This is a summary of the new and changed ISO 9001:2015 requirements compared to ISO 9001:2008. 4. Context of the Organization 4.1 Changes Understanding the Organization and its Context New requirement
This is a summary of the new and changed ISO 9001:2015 requirements compared to ISO 9001:2008. 4. Context of the Organization 4.1 Changes Understanding the Organization and its Context New requirement
Continual Improvement
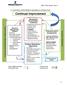 15. CONTINUAL IMPROVEMENT SEQUENCE & INTERACTION Continual Customer and Stakeholders Requirements Resource / Support Finance Document Control Information System Human Resources Competence Training Performance
15. CONTINUAL IMPROVEMENT SEQUENCE & INTERACTION Continual Customer and Stakeholders Requirements Resource / Support Finance Document Control Information System Human Resources Competence Training Performance
ASQ Madison, WI Section Using your ISO 9001:2008 QMS to Plan for the 2015 Changes
 ASQ Madison, WI Section 1217 Using your ISO 9001:2008 QMS to Plan for the 2015 Changes SESSION AGENDA High Level Review of Requirements Historical Perspective Basic Requirements Change It s a Process Inputs,
ASQ Madison, WI Section 1217 Using your ISO 9001:2008 QMS to Plan for the 2015 Changes SESSION AGENDA High Level Review of Requirements Historical Perspective Basic Requirements Change It s a Process Inputs,
ISO/DIS 9001: 2014 comparison with ISO 9001:2008. ISO 9001:2015 Updates. (Based on Draft International Standard, DIS) ISO/DIS 9001 ISO 9001:2008
 ISO 9001:2015 Updates (Based ondraft International Standard, DIS) August 2014 Page 1 ISO 9001:2015 Updates (Based on Draft International Standard, DIS) ISO/DIS 9001: 2014 comparison with ISO 9001:2008
ISO 9001:2015 Updates (Based ondraft International Standard, DIS) August 2014 Page 1 ISO 9001:2015 Updates (Based on Draft International Standard, DIS) ISO/DIS 9001: 2014 comparison with ISO 9001:2008
Quality management systems Requirements
 INTERNATIONAL STANDARD ISO 9001 Fifth edition 2015-09-15 Quality management systems Requirements Systèmes de management de la qualité Exigences Reference number ISO 9001:2015(E) ISO 2015 ISO 9001:2015(E)
INTERNATIONAL STANDARD ISO 9001 Fifth edition 2015-09-15 Quality management systems Requirements Systèmes de management de la qualité Exigences Reference number ISO 9001:2015(E) ISO 2015 ISO 9001:2015(E)
Quality management systems Requirements
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 9001 Fifth edition 2015-09-15 Quality management systems Requirements Systèmes de management de la qualité Exigences Reference number ISO 2015
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 9001 Fifth edition 2015-09-15 Quality management systems Requirements Systèmes de management de la qualité Exigences Reference number ISO 2015
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 15378 Fourth edition 2017-09 Primary packaging materials for medicinal products Particular requirements for the application of ISO 9001:2015, with reference to good manufacturing
INTERNATIONAL STANDARD ISO 15378 Fourth edition 2017-09 Primary packaging materials for medicinal products Particular requirements for the application of ISO 9001:2015, with reference to good manufacturing
Differences between ISO 9001:2008 and ISO 9001:2015
 Differences between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 HAS TEN CLAUSES INSTEAD OF EIGHT ISO 9001:2015 has ten clauses instead of eight. The following table shows the relationship of the ISO
Differences between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 HAS TEN CLAUSES INSTEAD OF EIGHT ISO 9001:2015 has ten clauses instead of eight. The following table shows the relationship of the ISO
Summary of changes between ISO 9001:2008 and ISO/CD 9001
 Summary of changes between ISO 9001:2008 and ISO/CD 9001 Introduction This document is intended to summarise the proposed changes to ISO 9001:2008 as detailed in the Committee Draft (CD). It does not contain
Summary of changes between ISO 9001:2008 and ISO/CD 9001 Introduction This document is intended to summarise the proposed changes to ISO 9001:2008 as detailed in the Committee Draft (CD). It does not contain
ISO 9001:2015 Gap Analysis Check Sheet
 CONTEXT OF THE ORGANIZATION 4.1 Understanding the organization and its context Organization shall determine external and internal issues that are relevant to its purpose and strategic direction and that
CONTEXT OF THE ORGANIZATION 4.1 Understanding the organization and its context Organization shall determine external and internal issues that are relevant to its purpose and strategic direction and that
INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 9001 Third edition 2000-12-15 Quality management systems Requirements Systèmes de management de la qualité Exigences Reference number ISO 9001:2000(E) ISO 2000 Contents Page
INTERNATIONAL STANDARD ISO 9001 Third edition 2000-12-15 Quality management systems Requirements Systèmes de management de la qualité Exigences Reference number ISO 9001:2000(E) ISO 2000 Contents Page
Supplier Auditing Beyond the Standards
 Supplier Auditing Beyond the Standards Mark Durivage, ASQ Fellow Managing Principal Consultant Quality Systems Compliance LLC 2018 Quality Systems Compliance LLC 1 Supplier Auditing Beyond the Standards
Supplier Auditing Beyond the Standards Mark Durivage, ASQ Fellow Managing Principal Consultant Quality Systems Compliance LLC 2018 Quality Systems Compliance LLC 1 Supplier Auditing Beyond the Standards
ISO 9001:2015 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES
 ISO 9001:2015 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 Policies and Procedures Latest Revision Date Abstract: This handbook
ISO 9001:2015 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 Policies and Procedures Latest Revision Date Abstract: This handbook
WHITE PAPER ISO 14001:2015. Environment Management System. The impact in international standard for environment management system
 WHITE PAPER Ultimate transition guide ISO 14001:2015 Environment Management System The impact in international standard for environment management system Success through management excellence Global Standards
WHITE PAPER Ultimate transition guide ISO 14001:2015 Environment Management System The impact in international standard for environment management system Success through management excellence Global Standards
ISO 9001:2015. October 5 th, Brad Fischer.
 ISO 9001:2015 October 5 th, 2017 Brad Fischer www.sdmanufacturing.com Purpose of presentation Provide a summary of notable changes from ISO 9001:2008 to ISO 9001:2015 Key perspectives ISO 9001 needs to
ISO 9001:2015 October 5 th, 2017 Brad Fischer www.sdmanufacturing.com Purpose of presentation Provide a summary of notable changes from ISO 9001:2008 to ISO 9001:2015 Key perspectives ISO 9001 needs to
ISO9001:2015 and ISO14001:2014 Transition Update March 2017
 ISO9001:2015 and ISO14001:2014 Transition Update March 2017 TIMESCALES The ISO 9001:2015 and ISO14001:2015 standards were issued on the 15 th September 2015 and they replace ISO 9001:2008 and ISO14001:2004.
ISO9001:2015 and ISO14001:2014 Transition Update March 2017 TIMESCALES The ISO 9001:2015 and ISO14001:2015 standards were issued on the 15 th September 2015 and they replace ISO 9001:2008 and ISO14001:2004.
ISO Food Safety Management System Compliance Summary
 ISO 22000 Clause ISO 9001 Clause FSMS Manual Reference Policy / Procedure Title 4. Quality Management System (ISO 9001) 4. Food Safety Quality Management System (ISO 22000) 4.1 General Requirements 4.1
ISO 22000 Clause ISO 9001 Clause FSMS Manual Reference Policy / Procedure Title 4. Quality Management System (ISO 9001) 4. Food Safety Quality Management System (ISO 22000) 4.1 General Requirements 4.1
ISO 9001:2015 AWARENESS
 ISO 9001:2015 AWARENESS All rights reserved. 2017 SIRIM STS Sdn Bhd COURSE AGENDA 0900-1030 am : Introduction to ISO 9001:2015 1030-1045 am : Tea Break 1045 0100 pm : Understanding of ISO 9001:2015 requirements
ISO 9001:2015 AWARENESS All rights reserved. 2017 SIRIM STS Sdn Bhd COURSE AGENDA 0900-1030 am : Introduction to ISO 9001:2015 1030-1045 am : Tea Break 1045 0100 pm : Understanding of ISO 9001:2015 requirements
QUALITY MANUAL. Revision K. JADE PRECISION MEDICAL COMPONENTS, LLC 3063 B Philmont Avenue Huntingdon Valley, PA 19006
 QUALITY MANUAL Revision K JADE PRECISION MEDICAL COMPONENTS, LLC 3063 B Philmont Avenue Huntingdon Valley, PA 19006 Page: 1 of 14 Table of Contents 1. Purpose & Scope... 2 2. Applicable Standards... 2
QUALITY MANUAL Revision K JADE PRECISION MEDICAL COMPONENTS, LLC 3063 B Philmont Avenue Huntingdon Valley, PA 19006 Page: 1 of 14 Table of Contents 1. Purpose & Scope... 2 2. Applicable Standards... 2
ISO 9001:2015 GAP ANALYSIS CHECKLIST
 Context of the Organization 4.1 Understanding the organization and its context Organization shall determine external and internal issues that are relevant to its purpose and strategic direction and that
Context of the Organization 4.1 Understanding the organization and its context Organization shall determine external and internal issues that are relevant to its purpose and strategic direction and that
ISO 31000:2009(E):Risk Management Principles and Guidelines
 International Organization for Standardization ISO 31000:2009(E):Risk Management Principles and Guidelines Sections 4 through Section 5 First Edition: November 15, 2009 2 International Organization for
International Organization for Standardization ISO 31000:2009(E):Risk Management Principles and Guidelines Sections 4 through Section 5 First Edition: November 15, 2009 2 International Organization for
WHATS NEW IN ISO 9001:2015
 WHATS NEW IN ISO 9001:2015 Introduction This presentation will cover the following topics: The ISO 9001 Revision Process Key Inputs to ISO 9001:2015 The High Level Structure Key Changes in ISO 9001:2015
WHATS NEW IN ISO 9001:2015 Introduction This presentation will cover the following topics: The ISO 9001 Revision Process Key Inputs to ISO 9001:2015 The High Level Structure Key Changes in ISO 9001:2015
P. 1. Identify the Differences between ISO9001:2000 與 ISO9001:2008 ISO9001:2008 ISO9001:2000 版本的異同. 5 January 2009 ISO 9000 SERIES
 Identify the Differences between ISO9001:2000 and ISO 9001:2008 審視 ISO9001:2000 與 ISO9001:2008 版本的異同 ISO 9000 SERIES ISO 19011 ISO9000 5 January 2009 ISO9001 ISO9004 2 ISO 9000 SERIES ISO 9001 ISO 9000
Identify the Differences between ISO9001:2000 and ISO 9001:2008 審視 ISO9001:2000 與 ISO9001:2008 版本的異同 ISO 9000 SERIES ISO 19011 ISO9000 5 January 2009 ISO9001 ISO9004 2 ISO 9000 SERIES ISO 9001 ISO 9000
AS/NZS ISO 9001:2016. Quality management systems Requirements AS/NZS ISO 9001:2016. Australian/New Zealand Standard. Superseding AS/NZS ISO 9001:2008
 AS/NZS ISO 9001:2016 (ISO 9001:2015, IDT) Australian/New Zealand Standard Quality management systems Requirements Superseding AS/NZS ISO 9001:2008 AS/NZS ISO 9001:2016 AS/NZS ISO 9001:2016 This joint Australian/New
AS/NZS ISO 9001:2016 (ISO 9001:2015, IDT) Australian/New Zealand Standard Quality management systems Requirements Superseding AS/NZS ISO 9001:2008 AS/NZS ISO 9001:2016 AS/NZS ISO 9001:2016 This joint Australian/New
This document is a preview generated by EVS
 2015 by SNV / Foqus Quality AG / MSB Bartels & Partner Consulting page: 1 von 77 0. Introduction On the occasion of the complete revision of the Standard ISO 9001 the Audit-Checklist (edition 2008) has
2015 by SNV / Foqus Quality AG / MSB Bartels & Partner Consulting page: 1 von 77 0. Introduction On the occasion of the complete revision of the Standard ISO 9001 the Audit-Checklist (edition 2008) has
Transistion Planning Guidance
 www.iso-9001-checklist.co.uk We re committed to helping you and your organization understand the updated requirements. This guidance document identifies the steps you should take to achieve compliance
www.iso-9001-checklist.co.uk We re committed to helping you and your organization understand the updated requirements. This guidance document identifies the steps you should take to achieve compliance
Specification for Quality Programs for the Petroleum, Petrochemical and Natural Gas Industry
 Addendum 1 June 2010 Effective Date: December 1, 2010 Specification for Quality Programs for the Petroleum, Petrochemical and Natural Gas Industry ANSI/API SPECIFICATION Q1 EIGHTH EDITION, DECEMBER 2007
Addendum 1 June 2010 Effective Date: December 1, 2010 Specification for Quality Programs for the Petroleum, Petrochemical and Natural Gas Industry ANSI/API SPECIFICATION Q1 EIGHTH EDITION, DECEMBER 2007
QMS Team: MR and all HODs (Internal Auditors) MR March 10. Quality policy Define quality policy The Steering committee Objectives and targets
 QMS Roles, Responsibility and Authority Process Clause Activities Records Required Responsibility Authority Deadline Clause 4: Process Development 4.1 Develop processes and sequence, operation controls
QMS Roles, Responsibility and Authority Process Clause Activities Records Required Responsibility Authority Deadline Clause 4: Process Development 4.1 Develop processes and sequence, operation controls
IMPORTANCE OF AUDITING ISO 9001: 2015
 IMPORTANCE OF AUDITING ISO 9001: 2015 PMI, PMP, PMBOK and the PMI Registered Education Provider logo are registered marks of the Project Management Institute, Inc. At the end of the class, please fill
IMPORTANCE OF AUDITING ISO 9001: 2015 PMI, PMP, PMBOK and the PMI Registered Education Provider logo are registered marks of the Project Management Institute, Inc. At the end of the class, please fill
ISO9001:2008 SYSTEM KARAN ADVISER & INFORMATION CENTER QUALITY MANAGEMENT SYSTEM SYSTEM KARAN ADVISER & INFORMATION CENTER
 SYSTEM KARAN ADVISER & INFORMATION CENTER QUALITY MANAGEMENT SYSTEM WWW.SYSTEMKARAN.COM 1 www.systemkaran.org Foreword... 5 Introduction... 6 0.1 General... 6 0.2 Process approach... 6 0.3 Relationship
SYSTEM KARAN ADVISER & INFORMATION CENTER QUALITY MANAGEMENT SYSTEM WWW.SYSTEMKARAN.COM 1 www.systemkaran.org Foreword... 5 Introduction... 6 0.1 General... 6 0.2 Process approach... 6 0.3 Relationship
25 D.L. Martin Drive Mercersburg, PA (717)
 QUALITY MANUAL D. L. MARTIN CO. 25 D.L. Martin Drive Mercersburg, PA 17236 (717) 328-2141 Revision 14 August 2012 Michael A. White Manager, QA & Engineering D.L. Martin Co. Quality Manual UNCONTROLLED
QUALITY MANUAL D. L. MARTIN CO. 25 D.L. Martin Drive Mercersburg, PA 17236 (717) 328-2141 Revision 14 August 2012 Michael A. White Manager, QA & Engineering D.L. Martin Co. Quality Manual UNCONTROLLED
ISO 9001:2000 Making the Transition
 ISO 9001:2000 Making the Transition ISO 9001:2000 Making the Transition Annette Dennis McCully Debra L. Reese Excerpts from interviews conducted by the author with Garnett Davis and William Poliseo published
ISO 9001:2000 Making the Transition ISO 9001:2000 Making the Transition Annette Dennis McCully Debra L. Reese Excerpts from interviews conducted by the author with Garnett Davis and William Poliseo published
Provläsningsexemplar / Preview. Third edition
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 15378 Third edition 2015-10-01 Primary packaging materials for medicinal products Particular requirements for the application of ISO 9001:2008,
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 15378 Third edition 2015-10-01 Primary packaging materials for medicinal products Particular requirements for the application of ISO 9001:2008,
ISO 13485:2016 Mandatory documentation requirements & MyEasyISO
 ts & Mandatory documents All below mandatory documents can be created using Documented information module of. An organization can use their own procedures but if required can provide these procedures to
ts & Mandatory documents All below mandatory documents can be created using Documented information module of. An organization can use their own procedures but if required can provide these procedures to
AP Quality Consulting works with you to provide a custom solution
 AP Quality Consulting works with you to provide a custom solution ISO 9001, ISO/IEC 17025, AS6081, AS5553, AS6171 (in development) Quality auditing and management of internal audit program Corporate training
AP Quality Consulting works with you to provide a custom solution ISO 9001, ISO/IEC 17025, AS6081, AS5553, AS6171 (in development) Quality auditing and management of internal audit program Corporate training
EHQMS Manual & Policy Document
 Quality management input comprises the standard requirements from ISO 9001:2015 which are strategically deployed by our organization to achieve customer satisfaction through process control. Environmental
Quality management input comprises the standard requirements from ISO 9001:2015 which are strategically deployed by our organization to achieve customer satisfaction through process control. Environmental
QUALITY MANUAL Revision H
 QUALITY MANUAL Revision H JADE PRECISION MEDICAL COMPONENTS, LLC 3063 B Philmont Avenue Huntingdon Valley, PA 19006 Page: 1 of 15 Table of Contents 1. Purpose & Scope... 2 2. Applicable Standards... 2
QUALITY MANUAL Revision H JADE PRECISION MEDICAL COMPONENTS, LLC 3063 B Philmont Avenue Huntingdon Valley, PA 19006 Page: 1 of 15 Table of Contents 1. Purpose & Scope... 2 2. Applicable Standards... 2
Compliance Solutions (Life Sciences) Ltd
 Welcome to the Workshop Fire precautions & exit routes Please take valuables with you at break Keep cell phones switched off Any problems, please let us know Above all, relax & enjoy the course Learning
Welcome to the Workshop Fire precautions & exit routes Please take valuables with you at break Keep cell phones switched off Any problems, please let us know Above all, relax & enjoy the course Learning
LMS Certification Ltd. ISO 9001 and ISO Transition
 LMS Certification Ltd. ISO 9001 and ISO 14001 Transition Objective Section 1: presentation of LMS s interpretation of the high level structure of Annex SL; Section 2: To provide an overview of the changes
LMS Certification Ltd. ISO 9001 and ISO 14001 Transition Objective Section 1: presentation of LMS s interpretation of the high level structure of Annex SL; Section 2: To provide an overview of the changes
LMS Certifications Pvt. Ltd. ISO 9001 and ISO Transition
 LMS Certifications Pvt. Ltd. ISO 9001 and ISO 14001 Transition Objective Section 1: presentation of LMS s interpretation of the high level structure of Annex SL; Section 2: To provide an overview of the
LMS Certifications Pvt. Ltd. ISO 9001 and ISO 14001 Transition Objective Section 1: presentation of LMS s interpretation of the high level structure of Annex SL; Section 2: To provide an overview of the
FDA 21 CFR Part 820 vs. ISO 13485:2016 Comparison Table created by greenlight.guru
 FDA 21 CFR Part 820 vs. ISO 13485:2016 Comparison Table created by greenlight.guru FDA QSR (21 CFR Part 820) ISO 13485:2016 820.1 Scope 1 Scope 2 Normative References 820.3 Definitions 3 Terms and Definitions
FDA 21 CFR Part 820 vs. ISO 13485:2016 Comparison Table created by greenlight.guru FDA QSR (21 CFR Part 820) ISO 13485:2016 820.1 Scope 1 Scope 2 Normative References 820.3 Definitions 3 Terms and Definitions
9100 Team July, IAQG is a trademark the International Aerospace Quality Group. Copyright 2014 IAQG. All rights reserved.
 9100 Series 2016 Revision Overview 9100 Team July, 2014 1 9100 Revision The Plan 9100 Series Revision High Level Plan The 9100 is based on ISO 9001 and is thus affected by the ISO TC176 revision activity
9100 Series 2016 Revision Overview 9100 Team July, 2014 1 9100 Revision The Plan 9100 Series Revision High Level Plan The 9100 is based on ISO 9001 and is thus affected by the ISO TC176 revision activity
Gap Analysis Checklist ISO 9001:2015 Self-assessment
 www.iso-9001-checklist.co.uk Gap Analysis Checklist Contents Guidance 2 About this Checklist 2 Summary of Key Changes 2 Process Approach 2 Context of the Organization 2 Scope of the Quality Management
www.iso-9001-checklist.co.uk Gap Analysis Checklist Contents Guidance 2 About this Checklist 2 Summary of Key Changes 2 Process Approach 2 Context of the Organization 2 Scope of the Quality Management
ICM CERTIFICATION (P) LIMITED
 ICM CERTIFICATION (P) LIMITED Transition to ISO 22000:2018 Food safety management System ASUS Quality HACCP ICMC ISO 22000 Food safety management system The 2018 version of ISO 22000 was published on June
ICM CERTIFICATION (P) LIMITED Transition to ISO 22000:2018 Food safety management System ASUS Quality HACCP ICMC ISO 22000 Food safety management system The 2018 version of ISO 22000 was published on June
Educational organizations Management systems for educational organizations Requirements with guidance for use
 INTERNATIONAL STANDARD ISO 21001 First edition 2018-05 Educational organizations Management systems for educational organizations Requirements with guidance for use Organismes d'éducation/formation Systèmes
INTERNATIONAL STANDARD ISO 21001 First edition 2018-05 Educational organizations Management systems for educational organizations Requirements with guidance for use Organismes d'éducation/formation Systèmes
Conducting Quality Audit based on ISO 9001:2015
 Conducting Quality Audit based on ISO 9001:2015 FREE Professional Development Seminar Series At the end of the class, please fill the training feedback and referral forms! Return the form at the reception!
Conducting Quality Audit based on ISO 9001:2015 FREE Professional Development Seminar Series At the end of the class, please fill the training feedback and referral forms! Return the form at the reception!
ISO 9001:2000 Revision G
 ISO 9001:2000 Revision G 1531 W. 17 th Street, Tempe AZ 85281 Table of contents Section Title Page Index 1 Revision History and Distribution 2 Introduction 3 Quality Policy / Mission Statement 4 Organizational
ISO 9001:2000 Revision G 1531 W. 17 th Street, Tempe AZ 85281 Table of contents Section Title Page Index 1 Revision History and Distribution 2 Introduction 3 Quality Policy / Mission Statement 4 Organizational
SUPPLIER SURVEY FORM Instructions
 SUPPLIER SURVEY FORM Instructions 1. The following Supplier Survey was developed by Vishay Measurements Group, Inc. to assess and document the capability of its supplier base. 2. The Supplier Survey is
SUPPLIER SURVEY FORM Instructions 1. The following Supplier Survey was developed by Vishay Measurements Group, Inc. to assess and document the capability of its supplier base. 2. The Supplier Survey is
Quality Systems Manual Rev. NC Issued July 9 / 2018
 NMT Specialized Machining Inc 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Quality Systems Manual Rev. NC Issued July 9 / 2018 Conforms to AS9100 Rev D and ISO 9001:2015 Table of Contents Introduction
NMT Specialized Machining Inc 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Quality Systems Manual Rev. NC Issued July 9 / 2018 Conforms to AS9100 Rev D and ISO 9001:2015 Table of Contents Introduction
SYSTEMKARAN ADVISER & INFORMATION CENTER QUALITY MANAGEMENT SYSTEM ISO9001:
 SYSTEM KARAN ADVISER & INFORMATION CENTER QUALITY MANAGEMENT SYSTEM ISO9001:2015 WWW.SYSTEMKARAN.ORG 1 WWW.SYSTEMKARAN.ORG Foreword... 5 Introduction... 6 0.1 General... 6 0.2 Quality management principles...
SYSTEM KARAN ADVISER & INFORMATION CENTER QUALITY MANAGEMENT SYSTEM ISO9001:2015 WWW.SYSTEMKARAN.ORG 1 WWW.SYSTEMKARAN.ORG Foreword... 5 Introduction... 6 0.1 General... 6 0.2 Quality management principles...
How ISO 9001:2015 can improve your food safety management. Dr. David Rosenblatt Sher Consulting and Training
 How ISO 9001:2015 can improve your food safety management Dr. David Rosenblatt Sher Consulting and Training ISO management systems in the food industry A little bit of history 2000 ISO publishes the brand
How ISO 9001:2015 can improve your food safety management Dr. David Rosenblatt Sher Consulting and Training ISO management systems in the food industry A little bit of history 2000 ISO publishes the brand
QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES
 Your Company Name QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract:
Your Company Name QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract:
ISO/TS TECHNICAL SPECIFICATION
 TECHNICAL SPECIFICATION ISO/TS 29001 Third edition 2010-06-01 Petroleum, petrochemical and natural gas industries Sector-specific quality management systems Requirements for product and service supply
TECHNICAL SPECIFICATION ISO/TS 29001 Third edition 2010-06-01 Petroleum, petrochemical and natural gas industries Sector-specific quality management systems Requirements for product and service supply
ISO 9001:2015 what will change, and why? Dr Nigel H Croft
 ISO 9001:2015 what will change, and why? Dr Nigel H Croft June 2014 (c) TCA Global 2014 1 Disclaimer! Any opinions expressed by the presenter are personal viewpoints, and no formal endorsement of ISO,
ISO 9001:2015 what will change, and why? Dr Nigel H Croft June 2014 (c) TCA Global 2014 1 Disclaimer! Any opinions expressed by the presenter are personal viewpoints, and no formal endorsement of ISO,
Tools to Assist Risk Based Thinking PMPA Quality Technical Committee Presentation 2018 National Technical Conference Program
 Tools to Assist Risk Based Thinking PMPA Quality Technical Committee Presentation 2018 National Technical Conference Program Foundation Tools to Assist Risked Based Thinking (RBT) Why and Where the ISO9001:2015
Tools to Assist Risk Based Thinking PMPA Quality Technical Committee Presentation 2018 National Technical Conference Program Foundation Tools to Assist Risked Based Thinking (RBT) Why and Where the ISO9001:2015
Continental Steel & Tube Co. Quality Manual
 312 SE 22 nd Street Fort Lauderdale, FL 33316 Mailing address P.O. Box 030040 Fort Lauderdale, FL 33303-0040 954-332-2290 Fax 954-332-2296 info@continentalsteel.com www.continentalsteel.com Introduction
312 SE 22 nd Street Fort Lauderdale, FL 33316 Mailing address P.O. Box 030040 Fort Lauderdale, FL 33303-0040 954-332-2290 Fax 954-332-2296 info@continentalsteel.com www.continentalsteel.com Introduction
ISO 9001: 2000 (December 13, 2000) QUALITY MANAGEMENT SYSTEM DOCUMENTATION OVERVIEW MATRIX
 In completing your Documented Quality Management System Review, it is important that the following matrix be completed and returned to us as soon as possible. This will save time during the review and
In completing your Documented Quality Management System Review, it is important that the following matrix be completed and returned to us as soon as possible. This will save time during the review and
84 Ways To Go Beyond ISO Rick Hill 13 October 2017
 84 Ways To Go Beyond ISO 9001 Rick Hill 13 October 2017 ISO 9001 is a great QMS standard but All the requirements of this International Standard are generic and are intended to be applicable to any organization,
84 Ways To Go Beyond ISO 9001 Rick Hill 13 October 2017 ISO 9001 is a great QMS standard but All the requirements of this International Standard are generic and are intended to be applicable to any organization,
Electronics. Document: QUALITY MANUAL. Revision: W. Date: 08/22/2016
 Document: QUALITY MANUAL Revision: W Date: 08/22/2016 Electronics Gowanda Electronics One Magnetics Parkway Gowanda, NY 14070 Ph: 716-532-2234 Fax: 716-532-2702 www.gowanda.com Table of Contents Table
Document: QUALITY MANUAL Revision: W Date: 08/22/2016 Electronics Gowanda Electronics One Magnetics Parkway Gowanda, NY 14070 Ph: 716-532-2234 Fax: 716-532-2702 www.gowanda.com Table of Contents Table
Brief Introduction to the ISO 9001:2000 and Quality Principles
 Vilmos Berényi H2509 H2509 Esztergom-Kertváros, Hőtáv Hőtávu. u. 59 59 Hungary Hungary +36703806768, fax: fax: +3633319117 berenyi@gmail.com Brief Introduction to the ISO 9001:2000 and Quality Principles
Vilmos Berényi H2509 H2509 Esztergom-Kertváros, Hőtáv Hőtávu. u. 59 59 Hungary Hungary +36703806768, fax: fax: +3633319117 berenyi@gmail.com Brief Introduction to the ISO 9001:2000 and Quality Principles
Stewart Industries Inc.
 A QUALITY MANAGEMENT SYSTEM MANUAL Release Date: 5/16/2017 Revision: A Stewart Industries Inc. 150 McQuiston Drive, Battle Creek MI 49037 Telephone: 1 (866) 660-9290 Fax: 1 (866) 660-0421 WEB Site http://stewartindustriesusa.com/
A QUALITY MANAGEMENT SYSTEM MANUAL Release Date: 5/16/2017 Revision: A Stewart Industries Inc. 150 McQuiston Drive, Battle Creek MI 49037 Telephone: 1 (866) 660-9290 Fax: 1 (866) 660-0421 WEB Site http://stewartindustriesusa.com/
QM-1 Supplement QUALITY MANAGEMENT SYSTEMS MANUAL SUPPLEMENT. Revision 7. Quality Director / Management Representative n. B.
 QUALITY MANAGEMENT SYSTEMS MANUAL SUPPLEMENT Revision 7 APPROVED BY: TITLE DATE B. Olevson Quality Director / Management Representative 07 3-0 n DOCUMENT REVISION HISTORY Paragraph Revisions: or Rev: Date:
QUALITY MANAGEMENT SYSTEMS MANUAL SUPPLEMENT Revision 7 APPROVED BY: TITLE DATE B. Olevson Quality Director / Management Representative 07 3-0 n DOCUMENT REVISION HISTORY Paragraph Revisions: or Rev: Date:
AS 9100 Rev C Quality Systems Manual AS-050C-QM
 AS 9100 Rev C Quality Systems Manual AS-050C-QM Innovative Control Systems, Inc. 10801 N. 24 th Ave. Suite 101-103 Phoenix, AZ 85029 U.S.A. www.icsaero.com +01-602-861-6984 VOICE +01-602-588-9440 FAX Table
AS 9100 Rev C Quality Systems Manual AS-050C-QM Innovative Control Systems, Inc. 10801 N. 24 th Ave. Suite 101-103 Phoenix, AZ 85029 U.S.A. www.icsaero.com +01-602-861-6984 VOICE +01-602-588-9440 FAX Table
NMT Specialized Machining Inc & NMT General Machining Inc AS 9100 Rev C Quality Systems Manual
 NMT Specialized Machining Inc & NMT General Machining Inc AS 9100 Rev C Quality Systems Manual 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Table of Contents Introduction 5 Documentation Scheme..
NMT Specialized Machining Inc & NMT General Machining Inc AS 9100 Rev C Quality Systems Manual 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Table of Contents Introduction 5 Documentation Scheme..
con cen tric (ken-sĕn trĭk) adjective - Having a common center. con cen tric (ken-sĕn trĭk) adjective - Having a common center. acommoncenter.
 Changes to ISO 9001:2015 Presented by Jim Thompson 266 Meeting Street - Suite B Charleston, SC 29401 843.469.8279 acommoncenter.com January 26 th, 2016 ASQ Charleston con cen tric (ken-sĕn trĭk) adjective
Changes to ISO 9001:2015 Presented by Jim Thompson 266 Meeting Street - Suite B Charleston, SC 29401 843.469.8279 acommoncenter.com January 26 th, 2016 ASQ Charleston con cen tric (ken-sĕn trĭk) adjective
QUALITY MANUAL. Number: M-001 Revision: C Page 1 of 18 THIS DOCUMENT IS CONSIDERED UNCONTROLLED UNLESS ISSUED IDENTIFIED AS CONTROLLED
 Page 1 of 18 THIS DOCUMENT IS CONSIDERED UNCONTROLLED UNLESS ISSUED IDENTIFIED AS CONTROLLED Page 2 of 18 REVISION HISTORY DATE CHANGE DESCRIPTION 10/11/06 Original release 10/21/09 Revised to ISO9001:2008
Page 1 of 18 THIS DOCUMENT IS CONSIDERED UNCONTROLLED UNLESS ISSUED IDENTIFIED AS CONTROLLED Page 2 of 18 REVISION HISTORY DATE CHANGE DESCRIPTION 10/11/06 Original release 10/21/09 Revised to ISO9001:2008
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 30301 First edition 2011-11-15 Information and documentation records Requirements Information et documentation Systèmes de gestion des documents d'activité Exigences Reference
INTERNATIONAL STANDARD ISO 30301 First edition 2011-11-15 Information and documentation records Requirements Information et documentation Systèmes de gestion des documents d'activité Exigences Reference
Supplier Quality System Survey
 Supplier Division Address Date: Quality Contact: Reports to: Title: Title: Phone Fax e-mail Plant size (ft 2 ): Union: Contract expires: Total Employment: Direct Labor: Shifts: QA employees: This report
Supplier Division Address Date: Quality Contact: Reports to: Title: Title: Phone Fax e-mail Plant size (ft 2 ): Union: Contract expires: Total Employment: Direct Labor: Shifts: QA employees: This report
ISO High Level Standards (HSL)
 ISO High Level Standards (HSL) 4. Context of the organization Understanding the organization and its context Understanding the needs and expectations of interested parties Determining the scope of the
ISO High Level Standards (HSL) 4. Context of the organization Understanding the organization and its context Understanding the needs and expectations of interested parties Determining the scope of the
