SYMBIOTIC HEALTH. Targeted GI Delivery of Orally Administered Cells and Proteins for Microbiome-Based Therapeutics
|
|
|
- Angelica Cox
- 6 years ago
- Views:
Transcription
1 SYMBIOTIC HEALTH Targeted GI Delivery of Orally Administered Cells and Proteins for Microbiome-Based Therapeutics Contact: Gerard Honig PhD, Director
2 Overview Biotechnology startup developing drug delivery solutions to realize the therapeutic potential of the microbiome Drug delivery & formulation technology for controlled release of orally administered therapeutics for local action in the GI tract SHP-01 for C.difficile: Towards an ultra-targeted microbiomesparing antimicrobial for gastrointestinal infection New funding opportunity for IBD product development Capsule
3 Drug delivery solutions to realize the therapeutic potential of the microbiome Novel microbederived proteins Novel microbiome organisms Validated biologic therapeutics Targeted oral drug delivery
4 Formulation platform for targeted oral delivery of proteins & cells Small molecules Probiotic bacteria Standard enteric formulations Anaerobic bacteria Recombinant proteins Proprietary oral biologics delivery platform Standard capsule Loss of therapeutic activity
5 Formulation platform for targeted oral delivery of proteins & cells Microencapsulated hydrogel phase retains proteins & cells in native hydrated state Stabilized macroemulsions confer stability & controlled release Scalable, cost-effective manufacturing Preserves activity of sensitive biologics Stable product formulation GRAS composition with option to integrate addt l validated chemistries
6 Multi-stage delivery system for delivery of proteins & cells to the lower GI tract
7 Compatibility with large molecule & cell-based therapeutics 1μm
8 Targeted & tunable delivery of protein in simulated GI transit Stomach Small Intestine Colon Formulation A Formulation B Formulation C Time (hours)
9 Non-invasive in vivo imaging of oral formulations in GI tract by microct Noninvasive tracking of capsule location in GI tract in vivo using sphere tracer Visualization of capsule matrix integrity Capsuledue to differential density
10 Imaging delivery of fluorescently labeled protein therapeutic to the colon Delivery is colon-specific; protein not observed in small intestine Labelled protein (SHP-01, yellow) accumulates specifically in the cecum and proximal colon Capsule Protein cargo labelled with long-wavelength fluorescent dyes Combined with quantitative PK Label any protein or bacterial strain with fluorophores
11 Tunable controlled release Oral drug delivery for proteins & cells: Competitive landscape Compatibility with diverse cargo
12 C.difficile infection: Disease of the microbiome Antibiotics kill microbiome bacteria, leading to C.difficile 400% increase since 1993; $4B excess US medical cost Unmet medical need: Protracted & recurrent infections
13 SHP-01: Ultra-targeted lysin enzyme antimicrobial for C.difficile infection C.difficile Log CFU/mL Phage-derived lysin engineered to cleave cell wall Bacterial Cell Wall Lysin rapidly kills C.difficile in infected GI tract 6 * Cytoplasm 5 Undetectable acquired resistance Clear regulatory precedents Licensed from Rockefeller University 4 3 * Significant (400-fold) decrease Antimicrob Agents & Chemother. 59(12):7447 (2015)
14 SHP-01 kills diverse C.difficile strains while sparing microbiome bacteria *Predicted Antimicrob Agents & Chemother. 59(12):7447 (2015)
15 Symbiotic Health Team Management, Staff & Advisors Founder & President: Gerard Honig PhD Chief Technology Officer: Noel Goddard PhD Biomaterials & genomics expert & serial entrepreneur Founder & VP Clinical: Bruce Hirsch MD Infectious disease medicine innovator Selected Advisors: Vincent Fischetti PhD: Professor, Rockefeller University; leading antimicrobial inventor Larry Miller MD: Chief of Gastroenterology, Northwell Health; functional GI innovator Directors Steve Winick & Leo Guthart, Topspin Partners Leading biotechnology portfolio & exits (Immune Design, NGM, Aragon, Seragon) Katy Stein PhD: Frmr FDA Division Chief & SVP, Macrogenics; bacterial products expert Funding & Partners
16 Crohn s & Colitis Foundation Entrepreneurial Investing Initiative Contact: Gerard Honig PhD, Manager entrepreneurship@crohnscolitisfoundation.org
17 Up to $500K per project per year for product discovery & development Accelerator resources Must address unmet need in IBD All technologies considered Companies preferred Apply by July 21! Contact: Gerard Honig PhD, Manager
18 Appendix Capsule
19 Oral delivery of biologics: Challenges & opportunities Since 2011, biologic therapeutics are 30% of new FDA approvals. In 2015, 1,367 biologic therapeutic INDs were filed Of the >240 biologics approved, only 5 are oral formulations The majority of commercial delivery technologies are devoted to protecting the cargo as it passages through the stomach or improving drug absorption in the small intestine. Our delivery technology is tunable, allowing release in the small intestine or lower GI tract, making it uniquely suited for delivery of peptides, proteins, and organisms into the colonic microbiome.
20 Symbiotic Health Product Development Pipeline Discovery Preclinical Clinical 1 st gen delivery platform* 2 nd gen delivery platform** SHP-01 for acute & recurrent Cdiff Monoclonal Antibodies for IBD *Hirsch et al, BMC ID Superseded by 2 nd gen platform. **Integrated into current therapeutic product candidates.
21 Current & Future C.difficile Therapy: SHP-01 Competitive Differentiation Acute or recurrent label Microbiome sparing Undetectable resistance Synergistic w/ standard-of-care Stable oral formulation
22 SHP-01: Potential Clinical Benefits & Synergies for Recurrent Infection Microbiome Transplantation Defined Probiotics Recurrent C.difficile infection Suppression of acute infection >60% risk of relapse Reduced risk of relapse Potential to accelerate resolution & reduce relapse
Assembly Biosciences Jefferies 2015 Microbiome Summit. December 16, 2015
 Assembly Biosciences Jefferies 2015 Microbiome Summit December 16, 2015 Forward-Looking Statements This presentation contains forward-looking statements regarding future events. Forward-looking statements
Assembly Biosciences Jefferies 2015 Microbiome Summit December 16, 2015 Forward-Looking Statements This presentation contains forward-looking statements regarding future events. Forward-looking statements
May Microbiome Modulation. Assaf Oron, CBO Biomed Conference Tel Aviv, May 2017
 May 2017 Microbiome Modulation Assaf Oron, CBO Biomed Conference Tel Aviv, May 2017 Microbiome A revolutionary cure Linda, lives in California 2010 Was treated with antibiotics due to a severe urinary
May 2017 Microbiome Modulation Assaf Oron, CBO Biomed Conference Tel Aviv, May 2017 Microbiome A revolutionary cure Linda, lives in California 2010 Was treated with antibiotics due to a severe urinary
Assembly Biosciences, Inc.
 Assembly Biosciences, Inc. Jefferies Global Healthcare Conference June 2017 Nasdaq: ASMB Cautionary note regarding forward-looking statements The information in this presentation contains estimates and
Assembly Biosciences, Inc. Jefferies Global Healthcare Conference June 2017 Nasdaq: ASMB Cautionary note regarding forward-looking statements The information in this presentation contains estimates and
Customized Phage Therapies To Eradicate Harmful Bacteria In Chronic Diseases. Europe Microbiome Congress London, 14 Nov., 2018
 Customized Phage Therapies To Eradicate Harmful Bacteria In Chronic Diseases Europe Microbiome Congress London, 14 Nov., 2018 Biomx At A Glance We are a microbiome drug discovery company developing customized
Customized Phage Therapies To Eradicate Harmful Bacteria In Chronic Diseases Europe Microbiome Congress London, 14 Nov., 2018 Biomx At A Glance We are a microbiome drug discovery company developing customized
March Company Introduction. Microbiome R&D and Business Collaboration Forum: Europe Myriam Golembo, VP Development
 March 2018 Company Introduction Microbiome R&D and Business Collaboration Forum: Europe Myriam Golembo, VP Development Biomx At A Glance We are a microbiome drug discovery company developing customized
March 2018 Company Introduction Microbiome R&D and Business Collaboration Forum: Europe Myriam Golembo, VP Development Biomx At A Glance We are a microbiome drug discovery company developing customized
SYNTHETIC BIOLOGICS, INC.
 SYNTHETIC BIOLOGICS, INC. FORM DEFA14A (Additional Proxy Soliciting Materials (definitive)) Filed 05/12/14 Address 155 GIBBS STREET SUITE 412 ROCKVILLE, MD 20850 Telephone (734) 332-7800 CIK 0000894158
SYNTHETIC BIOLOGICS, INC. FORM DEFA14A (Additional Proxy Soliciting Materials (definitive)) Filed 05/12/14 Address 155 GIBBS STREET SUITE 412 ROCKVILLE, MD 20850 Telephone (734) 332-7800 CIK 0000894158
Jan Company Introduction. Microbiome Drug Development Europe Assaf Oron, CBO
 Jan. 2018 Company Introduction Microbiome Drug Development Europe Assaf Oron, CBO Biomx At A Glance We are a microbiome drug discovery company developing customized phage therapies that target and destroy
Jan. 2018 Company Introduction Microbiome Drug Development Europe Assaf Oron, CBO Biomx At A Glance We are a microbiome drug discovery company developing customized phage therapies that target and destroy
THE HUMAN MICROBIOME: RECENT DISCOVERIES AND APPLICATIONS TO MEDICINE
 THE HUMAN MICROBIOME: RECENT DISCOVERIES AND APPLICATIONS TO MEDICINE American Society for Clinical Laboratory Science April 21, 2017 Richard A. Van Enk, Ph.D., CIC FSHEA Director, Infection Prevention
THE HUMAN MICROBIOME: RECENT DISCOVERIES AND APPLICATIONS TO MEDICINE American Society for Clinical Laboratory Science April 21, 2017 Richard A. Van Enk, Ph.D., CIC FSHEA Director, Infection Prevention
Oncology Product and Platform Partnering Opportunity
 Protein Medicinal Chemistry with an Expanded Genetic Code Oncology Product and Platform Partnering Opportunity April 2017 Ambrx has Made Advances in Proprietary Platform and Titer while Achieving Clinical
Protein Medicinal Chemistry with an Expanded Genetic Code Oncology Product and Platform Partnering Opportunity April 2017 Ambrx has Made Advances in Proprietary Platform and Titer while Achieving Clinical
Synthetic Biologics Reports Year End 2012 Financial Results
 April 16, 2013 Synthetic Biologics Reports Year End 2012 Financial Results -- Strengthening Infectious Disease Portfolio to Include C. difficile, Pertussis and Acinetobacter Targets -- ROCKVILLE, Md.,
April 16, 2013 Synthetic Biologics Reports Year End 2012 Financial Results -- Strengthening Infectious Disease Portfolio to Include C. difficile, Pertussis and Acinetobacter Targets -- ROCKVILLE, Md.,
Elena BM Breidenstein, PhD 21 April 2018
 Discovery of a Novel Oral Antibiotic, DDS-01 (SMT-571), to Treat Multi-Drug Resistant Neisseria gonorrhoeae Enabled by a Proprietary Transposon Technology Elena BM Breidenstein, PhD 21 April 2018 Forward-Looking
Discovery of a Novel Oral Antibiotic, DDS-01 (SMT-571), to Treat Multi-Drug Resistant Neisseria gonorrhoeae Enabled by a Proprietary Transposon Technology Elena BM Breidenstein, PhD 21 April 2018 Forward-Looking
New Hope For Serious Infections
 New Hope For Serious Infections Forward-Looking Statements Statements contained in this press release regarding matters that are not historical facts are "forward-looking statements" within the meaning
New Hope For Serious Infections Forward-Looking Statements Statements contained in this press release regarding matters that are not historical facts are "forward-looking statements" within the meaning
Have you wondered why it is called: a gut feeling or a gut instinct?
 Have you wondered why it is called: a gut feeling or a gut instinct? 2500 years ago, Hippocrates said: death sits in the bowels microgut: engage your microbiome! SCIENTIFIC ADVISORY BOARD Luxembourg Centre
Have you wondered why it is called: a gut feeling or a gut instinct? 2500 years ago, Hippocrates said: death sits in the bowels microgut: engage your microbiome! SCIENTIFIC ADVISORY BOARD Luxembourg Centre
PlantForm Corporation
 PlantForm Corporation Annual General Meeting September 21, 2010 Dr. Don Stewart President and CEO +1 (416) 452 7242 don.stewart@plantformcorp.com PlantForm Corporation 2010 www.plantformcorp.com The Opportunity
PlantForm Corporation Annual General Meeting September 21, 2010 Dr. Don Stewart President and CEO +1 (416) 452 7242 don.stewart@plantformcorp.com PlantForm Corporation 2010 www.plantformcorp.com The Opportunity
NIH-RAID: A ROADMAP Program
 NIH-RAID: A ROADMAP Program (Rapid Access to Interventional Development} A Program designed to facilitate the development of new therapeutics The NIH-RAID Pilot Program is intended to reduce some of the
NIH-RAID: A ROADMAP Program (Rapid Access to Interventional Development} A Program designed to facilitate the development of new therapeutics The NIH-RAID Pilot Program is intended to reduce some of the
Precigen Company Update
 Precigen Company Update Helen Sabzevari, PhD President, Precigen 9 January 2019 JP Morgan 37 th Annual Healthcare Conference Forward-looking statements Precigen, Inc. is a subsidiary of Intrexon Corporation
Precigen Company Update Helen Sabzevari, PhD President, Precigen 9 January 2019 JP Morgan 37 th Annual Healthcare Conference Forward-looking statements Precigen, Inc. is a subsidiary of Intrexon Corporation
Astellas to Acquire Agensys, Inc.
 November 27, 2007 to Acquire, Inc. Japan, November 27, 2007 - Pharma Inc. ( ; headquarters: Tokyo; President and CEO: Masafumi Nogimori) today announced that its US subsidiary, US Holding, Inc. (headquarters:
November 27, 2007 to Acquire, Inc. Japan, November 27, 2007 - Pharma Inc. ( ; headquarters: Tokyo; President and CEO: Masafumi Nogimori) today announced that its US subsidiary, US Holding, Inc. (headquarters:
Oral vaccines to protect patients against Clostridium difficile infection
 Oral vaccines to protect patients against Clostridium difficile infection Jonathan Kearsey: Leads To Development Antibiotics and their alternatives-fixing and feeding the pipeline Project number: 601810
Oral vaccines to protect patients against Clostridium difficile infection Jonathan Kearsey: Leads To Development Antibiotics and their alternatives-fixing and feeding the pipeline Project number: 601810
The current options to control infectious diseases in livestock and the stimulation of innovation to develop new treatments
 The current options to control infectious diseases in livestock and the stimulation of innovation to develop new treatments Scott A Brown, DVM, PhD Executive Director, Global Therapeutics Research 1 A
The current options to control infectious diseases in livestock and the stimulation of innovation to develop new treatments Scott A Brown, DVM, PhD Executive Director, Global Therapeutics Research 1 A
TRIANNI Media Kit MEDIA CONTACT CORPORATE HEADQUARTERS. Trianni, Inc. San Francisco, CA
 TRIANNI Media Kit CORPORATE HEADQUARTERS Trianni, Inc. San Francisco, CA Telephone: 1.866.374.9314 MEDIA CONTACT Mandy Boyd Director of Marketing @Trianni_mAb Website: Trianni.com TECHNOLOGY LEADERSHIP
TRIANNI Media Kit CORPORATE HEADQUARTERS Trianni, Inc. San Francisco, CA Telephone: 1.866.374.9314 MEDIA CONTACT Mandy Boyd Director of Marketing @Trianni_mAb Website: Trianni.com TECHNOLOGY LEADERSHIP
Antibody Discovery at Evotec
 Antibody Discovery at Evotec - Overview - Evotec Antibodies Adding value to our partners research Innovative and flexible solutions from target ID to pre-clinical candidate The people A wide therapeutic
Antibody Discovery at Evotec - Overview - Evotec Antibodies Adding value to our partners research Innovative and flexible solutions from target ID to pre-clinical candidate The people A wide therapeutic
Course Agenda. Day One
 Course Agenda BioImmersion: Biotech for the Non-Scientist A three-day, in-depth course that provides the background required for understanding today s fast-paced biotech marketplace. Beginning with an
Course Agenda BioImmersion: Biotech for the Non-Scientist A three-day, in-depth course that provides the background required for understanding today s fast-paced biotech marketplace. Beginning with an
Career Growth Areas in Physiology / Pharmacology
 Career Growth Areas in Physiology / Pharmacology Magdalena Alonso-Galicia, PhD Pharmacology Department Forest Research Institute, Inc. Jersey City, NJ 1 Career Growth Areas in BioPharma n Pharmaceutical
Career Growth Areas in Physiology / Pharmacology Magdalena Alonso-Galicia, PhD Pharmacology Department Forest Research Institute, Inc. Jersey City, NJ 1 Career Growth Areas in BioPharma n Pharmaceutical
Microfluidic Devices that Capture Bacteria for Growth and Kill Analysis
 Microfluidic Devices that Capture Bacteria for Growth and Kill Analysis Michael J. Lochhead, PhD. Accelr8 Technology Corporation Denver, CO AVS Symposium Biomaterial Interfaces Microbe-Surface Interactions
Microfluidic Devices that Capture Bacteria for Growth and Kill Analysis Michael J. Lochhead, PhD. Accelr8 Technology Corporation Denver, CO AVS Symposium Biomaterial Interfaces Microbe-Surface Interactions
The Human Microbiome Project : Implications for Probiotics
 The Human Microbiome Project : Implications for Probiotics James Versalovic, M.D., Ph.D. Associate Professor Departments of Pathology Baylor College of Medicine and Texas Children s Hospital Michael Hsieh,
The Human Microbiome Project : Implications for Probiotics James Versalovic, M.D., Ph.D. Associate Professor Departments of Pathology Baylor College of Medicine and Texas Children s Hospital Michael Hsieh,
Moderna Therapeutics Announces Transition to a Clinical Stage Company, Provides Business Update and Outlines 2016 Strategic Priorities
 Moderna Therapeutics Announces Transition to a Clinical Stage Company, Provides Business Update and Outlines 26 Strategic Priorities First Phase I study underway in Europe for mrna 44; IND for second program,
Moderna Therapeutics Announces Transition to a Clinical Stage Company, Provides Business Update and Outlines 26 Strategic Priorities First Phase I study underway in Europe for mrna 44; IND for second program,
LTX 109. BIO International Philadelphia 2015
 LTX 109 BIO International Philadelphia 2015 Lytix Biopharma company background Established in 2003 to develop and commercialize medical applications of lytic peptides originating from the University of
LTX 109 BIO International Philadelphia 2015 Lytix Biopharma company background Established in 2003 to develop and commercialize medical applications of lytic peptides originating from the University of
Advancing Manufacturing for Advanced Therapies
 Advancing Manufacturing for Advanced Therapies Peter Marks, MD, PhD Center For Biologics Evaluation and Research, FDA CASSS Cell & Gene Therapy Symposium July 10, 2018 Overview Cell and gene therapy products
Advancing Manufacturing for Advanced Therapies Peter Marks, MD, PhD Center For Biologics Evaluation and Research, FDA CASSS Cell & Gene Therapy Symposium July 10, 2018 Overview Cell and gene therapy products
Discovery of a First-In-Class Topically Bioavailable Kit Inhibitor With Clinical Activity Using Computational Chemogenomics Technology
 20131009-03 Discovery of a First-In-Class Topically Bioavailable Kit Inhibitor With Clinical Activity Using Computational Chemogenomics Technology James Hendrix, Ph.D. President - Technology Med Chem &
20131009-03 Discovery of a First-In-Class Topically Bioavailable Kit Inhibitor With Clinical Activity Using Computational Chemogenomics Technology James Hendrix, Ph.D. President - Technology Med Chem &
Introduction of Development Center for Biotechnology TAIWAN
 Introduction of Development Center for Biotechnology TAIWAN DCB Nonprofit Organization Founded in 1984 Funded Mainly by Ministry of Economic Affairs (MOEA), National Science Council and the Industry 394
Introduction of Development Center for Biotechnology TAIWAN DCB Nonprofit Organization Founded in 1984 Funded Mainly by Ministry of Economic Affairs (MOEA), National Science Council and the Industry 394
Global Allied Pharmaceuticals, LLC. Innovation, Research, and Technology. Breaking the bounders, first in its class, thinking outside the box.
 Global Allied Pharmaceuticals, LLC. Innovation, Research, and Technology. Breaking the bounders, first in its class, thinking outside the box. The purpose of this presentation Looking for Joint Venture
Global Allied Pharmaceuticals, LLC. Innovation, Research, and Technology. Breaking the bounders, first in its class, thinking outside the box. The purpose of this presentation Looking for Joint Venture
NIAID and Global Health Research Resources for Tuberculosis
 NIAID and Global Health Research Resources for Tuberculosis Maria Y. Giovanni, Ph.D. Director, Office of Genomics and Advanced Technologies National Institute of Allergy & Infectious Diseases NIH/DHHS
NIAID and Global Health Research Resources for Tuberculosis Maria Y. Giovanni, Ph.D. Director, Office of Genomics and Advanced Technologies National Institute of Allergy & Infectious Diseases NIH/DHHS
Phylogica. Harnessing Biodiversity for Peptide Therapies. Life Sciences Showcase, 23 August 2011 Nick Woolf: CFO & VP Corporate Development
 Phylogica Harnessing Biodiversity for Peptide Therapies Life Sciences Showcase, 23 August 2011 Nick Woolf: CFO & VP Corporate Development Introduction Biotech company offering leading peptide drug discovery
Phylogica Harnessing Biodiversity for Peptide Therapies Life Sciences Showcase, 23 August 2011 Nick Woolf: CFO & VP Corporate Development Introduction Biotech company offering leading peptide drug discovery
Mustang Bio Reports Third Quarter 2018 Financial Results and Recent Corporate Highlights
 Mustang Bio Reports Third Quarter 2018 Financial Results and Recent Corporate Highlights New York, NY November 13, 2018 Mustang Bio, Inc. ( Mustang ) (NASDAQ: MBIO), a company focused on the development
Mustang Bio Reports Third Quarter 2018 Financial Results and Recent Corporate Highlights New York, NY November 13, 2018 Mustang Bio, Inc. ( Mustang ) (NASDAQ: MBIO), a company focused on the development
Fecal transplant: Con Position
 Fecal transplant: Con Position Elaine O. Petrof, MD, MSc Dept.Medicine Division of Infectious Diseases Gastrointestinal Diseases Research Unit Queen s University ISAPP Meeting Cork, Ireland Oct.1 st, 2012
Fecal transplant: Con Position Elaine O. Petrof, MD, MSc Dept.Medicine Division of Infectious Diseases Gastrointestinal Diseases Research Unit Queen s University ISAPP Meeting Cork, Ireland Oct.1 st, 2012
Medicinal Chemistry of Modern Antibiotics
 Chemistry 259 Medicinal Chemistry of Modern Antibiotics Spring 2012 Lecture 5: Modern Target Discovery & MOA Thomas Hermann Department of Chemistry & Biochemistry University of California, San Diego Drug
Chemistry 259 Medicinal Chemistry of Modern Antibiotics Spring 2012 Lecture 5: Modern Target Discovery & MOA Thomas Hermann Department of Chemistry & Biochemistry University of California, San Diego Drug
Medicinal Chemistry of Modern Antibiotics
 Chemistry 259 Medicinal Chemistry of Modern Antibiotics Spring 2008 Lecture 5: Modern Target Discovery & MOA Thomas Hermann Department of Chemistry & Biochemistry University of California, San Diego Drug
Chemistry 259 Medicinal Chemistry of Modern Antibiotics Spring 2008 Lecture 5: Modern Target Discovery & MOA Thomas Hermann Department of Chemistry & Biochemistry University of California, San Diego Drug
ADVANCING SCIENTIFIC INNOVATION Together, the possibilities are endless.
 ADVANCING SCIENTIFIC INNOVATION Together, the possibilities are endless. COMMUNICATE INNOVATE EXPLORE COLLABORATE PARTNERSHIPS THAT BENEFIT YOU, US, AND ALL OF MANKIND Since Eli Lilly and Company was founded
ADVANCING SCIENTIFIC INNOVATION Together, the possibilities are endless. COMMUNICATE INNOVATE EXPLORE COLLABORATE PARTNERSHIPS THAT BENEFIT YOU, US, AND ALL OF MANKIND Since Eli Lilly and Company was founded
Chimerix Announces First Quarter 2017 Financial Results
 May 9, 2017 Chimerix Announces First Quarter 2017 Financial Results - Conference Call at 8:30 a.m. ET Today - DURHAM, N.C., May 09, 2017 (GLOBE NEWSWIRE) -- Chimerix (NASDAQ:CMRX), a biopharmaceutical
May 9, 2017 Chimerix Announces First Quarter 2017 Financial Results - Conference Call at 8:30 a.m. ET Today - DURHAM, N.C., May 09, 2017 (GLOBE NEWSWIRE) -- Chimerix (NASDAQ:CMRX), a biopharmaceutical
Global Leader in Bacteriophage Development to Combat Infectious Diseases
 Global Leader in Bacteriophage Development to Combat Infectious Diseases September 13, 2016 Safe Harbor Statement This presentation contains "forward-looking" statements that involve risks, uncertainties
Global Leader in Bacteriophage Development to Combat Infectious Diseases September 13, 2016 Safe Harbor Statement This presentation contains "forward-looking" statements that involve risks, uncertainties
Drug Development: Why Does it Cost so Much? Lewis J. Smith, MD Professor of Medicine Director, Center for Clinical Research Associate VP for Research
 Drug Development: Why Does it Cost so Much? Lewis J. Smith, MD Professor of Medicine Director, Center for Clinical Research Associate VP for Research Drug Development Process by which new chemical entities
Drug Development: Why Does it Cost so Much? Lewis J. Smith, MD Professor of Medicine Director, Center for Clinical Research Associate VP for Research Drug Development Process by which new chemical entities
PROFESSIONAL ETHICS, LEADERSHIP AND INTEGRITY: WHAT DOES IT MATTER HOW OUR RESEARCH IS USED? Suzanne Hendrich, PhD University Professor FSHN
 PROFESSIONAL ETHICS, LEADERSHIP AND INTEGRITY: WHAT DOES IT MATTER HOW OUR RESEARCH IS USED? Suzanne Hendrich, PhD University Professor FSHN shendric@ia state.edu LEARNING OUTCOMES Examine ethical implications
PROFESSIONAL ETHICS, LEADERSHIP AND INTEGRITY: WHAT DOES IT MATTER HOW OUR RESEARCH IS USED? Suzanne Hendrich, PhD University Professor FSHN shendric@ia state.edu LEARNING OUTCOMES Examine ethical implications
NIAID Resources to Facilitate Medical Countermeasure Development
 NIAID Resources to Facilitate Medical Countermeasure Development Paula Bryant, Ph.D. Senior Scientific Officer Concept Acceleration Program Biodefense, Research Resources, and Translational Research (OBRRTR)
NIAID Resources to Facilitate Medical Countermeasure Development Paula Bryant, Ph.D. Senior Scientific Officer Concept Acceleration Program Biodefense, Research Resources, and Translational Research (OBRRTR)
Micar Innovation. Drug Discovery Factory for novel drug molecules
 Problem There are so many incurable diseases around the world that need adequate novel compounds for the treatment, like: - Chronic pain still a pandemic in the 21 st century and affecting 1.5bn people
Problem There are so many incurable diseases around the world that need adequate novel compounds for the treatment, like: - Chronic pain still a pandemic in the 21 st century and affecting 1.5bn people
The Emerging Technology Program: FDA s Perspective
 The Emerging Technology Program: FDA s Perspective Mohan Sapru, M.S., Ph.D. Member Emerging Technology Team (ETT) CMC Lead Application Technical Lead Office of New Drug Products Office of Pharmaceutical
The Emerging Technology Program: FDA s Perspective Mohan Sapru, M.S., Ph.D. Member Emerging Technology Team (ETT) CMC Lead Application Technical Lead Office of New Drug Products Office of Pharmaceutical
Erika Smith - Director of the Blavatnik Fund for Innovation at Yale
 Erika Smith - Director of the Blavatnik Fund for Innovation at Yale erika.smith@yale.edu 1 GOAL: Advance Yale s biomedical technologies to the stage where they are ready for partnership (i.e. bridge the
Erika Smith - Director of the Blavatnik Fund for Innovation at Yale erika.smith@yale.edu 1 GOAL: Advance Yale s biomedical technologies to the stage where they are ready for partnership (i.e. bridge the
This presentation was developed by the Government & Industry Affairs Committee of the Crohn s & Colitis Foundation of America
 This presentation was developed by the Government & Industry Affairs Committee of the Crohn s & Colitis Foundation of America The development of this presentation was supported in 2016 by a sponsorship
This presentation was developed by the Government & Industry Affairs Committee of the Crohn s & Colitis Foundation of America The development of this presentation was supported in 2016 by a sponsorship
I. Company Information
 Please fill out this form as completely as possible. Note: There is no fee to apply. Please send additional materials such as your pitch deck and executive summary to resi@lifesciencenation.com for us
Please fill out this form as completely as possible. Note: There is no fee to apply. Please send additional materials such as your pitch deck and executive summary to resi@lifesciencenation.com for us
Supplementing the Immune System with Plant-Produced Antibodies
 Supplementing the Immune System with Plant-Produced Antibodies MICH B. HEIN 1 Epicyte Pharmaceutical, Inc. San Diego, CA Antibodies are inherently stable proteins found in all mammals and in fish. They
Supplementing the Immune System with Plant-Produced Antibodies MICH B. HEIN 1 Epicyte Pharmaceutical, Inc. San Diego, CA Antibodies are inherently stable proteins found in all mammals and in fish. They
FACTSHEET. Introduction. Antibodies ALTERNATIVES TO ANTIBIOTICS
 FACTSHEET ALTERNATIVES TO ANTIBIOTICS Introduction Due to antibiotic resistance and the long-term lack of antibiotic discovery, alternative treatments and effective prevention strategies for bacterial
FACTSHEET ALTERNATIVES TO ANTIBIOTICS Introduction Due to antibiotic resistance and the long-term lack of antibiotic discovery, alternative treatments and effective prevention strategies for bacterial
Horizon 2020 Health Summit-IIS La Fe. Valencia, 12 de diciembre de 2017
 Horizon 2020 Health Summit-IIS La Fe Valencia, 12 de diciembre de 2017 2 History 3 AsphaTeam +75 members Consultants with Life Science Background 20 team members with PhD High knowhow in drug development
Horizon 2020 Health Summit-IIS La Fe Valencia, 12 de diciembre de 2017 2 History 3 AsphaTeam +75 members Consultants with Life Science Background 20 team members with PhD High knowhow in drug development
UNLEASH THE POWER OF PRECISION MEDICINE
 FROM BENCH TO BEDSIDE UNLEASH THE POWER OF PRECISION MEDICINE DCB, pioneer in biotech. Your partner in drug discovery. www.dcb.org.tw The Center for Biotechnology With over 400 dedicated researchers and
FROM BENCH TO BEDSIDE UNLEASH THE POWER OF PRECISION MEDICINE DCB, pioneer in biotech. Your partner in drug discovery. www.dcb.org.tw The Center for Biotechnology With over 400 dedicated researchers and
ECO-PHARMACO- STEWARDSHIP (EPS) PILLAR 1 - RESEARCH & DEVELOPMENT: INTELLIGENCE-LED ASSESSMENT OF PHARMACEUTICALS IN THE ENVIRONMENT (ipie)
 ECO-PHARMACO- STEWARDSHIP (EPS) PILLAR 1 - RESEARCH & DEVELOPMENT: INTELLIGENCE-LED ASSESSMENT OF PHARMACEUTICALS IN THE ENVIRONMENT (ipie) PILLAR 1 - RESEARCH & DEVELOPMENT: INTELLIGENCE-LED ASSESSMENT
ECO-PHARMACO- STEWARDSHIP (EPS) PILLAR 1 - RESEARCH & DEVELOPMENT: INTELLIGENCE-LED ASSESSMENT OF PHARMACEUTICALS IN THE ENVIRONMENT (ipie) PILLAR 1 - RESEARCH & DEVELOPMENT: INTELLIGENCE-LED ASSESSMENT
I. Company Information
 Please fill out this form as completely as possible. Note: There is no fee to apply. Please send additional materials such as your pitch deck and executive summary to resi@lifesciencenation.com for us
Please fill out this form as completely as possible. Note: There is no fee to apply. Please send additional materials such as your pitch deck and executive summary to resi@lifesciencenation.com for us
University of California Center for Accelerated Innovation
 University of California Center for Accelerated Innovation MICHAEL PALAZZOLO April 6, 2015 Outline CAI overview and introduction RFA and important dates Selection process Pre-application Finding help 2
University of California Center for Accelerated Innovation MICHAEL PALAZZOLO April 6, 2015 Outline CAI overview and introduction RFA and important dates Selection process Pre-application Finding help 2
I. Company Information
 If your company won (1st-3rd place) an Innovation Challenge since RESI JPM 2018, or were a finalist at RESI NYC 2018, you do not qualify to participate in the Innovation Challenge at RESI JPM 2019. Please
If your company won (1st-3rd place) an Innovation Challenge since RESI JPM 2018, or were a finalist at RESI NYC 2018, you do not qualify to participate in the Innovation Challenge at RESI JPM 2019. Please
Vertex and Moderna Establish Exclusive Collaboration to Discover and Develop mrna Therapeutics for Cystic Fibrosis
 Vertex and Moderna Establish Exclusive Collaboration to Discover and Develop mrna Therapeutics for Cystic Fibrosis -Collaboration to explore use of mrna Therapeutics to treat the underlying cause of CF
Vertex and Moderna Establish Exclusive Collaboration to Discover and Develop mrna Therapeutics for Cystic Fibrosis -Collaboration to explore use of mrna Therapeutics to treat the underlying cause of CF
The Right Molecules. Designed. Delivered.
 The Right Molecules. Designed. Delivered. 400+ Worldwide Clients 80+ Medicinal Chemistry Programs 20+ Preclinical Candidates Delivered Since 2008 10+ In IND Stage 5 Entered Phase I Clinical Trials 2 Entered
The Right Molecules. Designed. Delivered. 400+ Worldwide Clients 80+ Medicinal Chemistry Programs 20+ Preclinical Candidates Delivered Since 2008 10+ In IND Stage 5 Entered Phase I Clinical Trials 2 Entered
Roche. New York City 7 December 2016
 Roche Andrew C. Chan, M.D. Ph.D. Senior Vice President, Research Biology of Genentech Research and Early Development (gred) New York City 7 December 2016 This presentation contains certain forward-looking
Roche Andrew C. Chan, M.D. Ph.D. Senior Vice President, Research Biology of Genentech Research and Early Development (gred) New York City 7 December 2016 This presentation contains certain forward-looking
EMA Workshop Non-Clinical Models to Identify PK/PD Indices and PD Targets In Vitro
 EMA Workshop Non-Clinical Models to Identify PK/PD Indices and PD Targets In Vitro G.L. Drusano, M.D. Professor and Director Institute for Therapeutic Innovation University of Florida In Vitro There are
EMA Workshop Non-Clinical Models to Identify PK/PD Indices and PD Targets In Vitro G.L. Drusano, M.D. Professor and Director Institute for Therapeutic Innovation University of Florida In Vitro There are
NSE Grantees Meeting December 2015
 NSE Grantees Meeting December 2015 The Spherical Nucleic Acid (SNA) Nanoparticle Changes the Paradigm for Oligo Therapeutics Linear DNA SNA Each company limited by chemistry, modality, and tissue of interest
NSE Grantees Meeting December 2015 The Spherical Nucleic Acid (SNA) Nanoparticle Changes the Paradigm for Oligo Therapeutics Linear DNA SNA Each company limited by chemistry, modality, and tissue of interest
Bioprotectants for control of the emerging food-borne pathogen Mycobacterium avium subspecies paratuberculosis (MAP)
 Snapshots of Doctoral Research at University College Cork 2010 Bioprotectants for control of the emerging food-borne pathogen Mycobacterium avium subspecies paratuberculosis (MAP) Brian Healy Department
Snapshots of Doctoral Research at University College Cork 2010 Bioprotectants for control of the emerging food-borne pathogen Mycobacterium avium subspecies paratuberculosis (MAP) Brian Healy Department
Melinta Therapeutics The Antibiotics Company
 Melinta Therapeutics The Antibiotics Company January 10, 2018 Dan Wechsler, CEO Cautionary Note Regarding Forward-looking Statements This presentation contains forward-looking statements that involve a
Melinta Therapeutics The Antibiotics Company January 10, 2018 Dan Wechsler, CEO Cautionary Note Regarding Forward-looking Statements This presentation contains forward-looking statements that involve a
Precigen Company Update
 Precigen Company Update Helen Sabzevari, PhD President, Precigen 7 January 2019 Forward-looking statements Precigen, Inc. is a subsidiary of Intrexon Corporation (Nasdaq: XON). Some of the statements made
Precigen Company Update Helen Sabzevari, PhD President, Precigen 7 January 2019 Forward-looking statements Precigen, Inc. is a subsidiary of Intrexon Corporation (Nasdaq: XON). Some of the statements made
UBS 2016 Health Care Conference May 24, 2016
 Leading the Microbiome Revolution UBS 2016 Health Care Conference May 24, 2016 Forward Looking Statements Some of the statements in this presentation constitute forward looking statements under the Private
Leading the Microbiome Revolution UBS 2016 Health Care Conference May 24, 2016 Forward Looking Statements Some of the statements in this presentation constitute forward looking statements under the Private
I. Company Information
 Please fill out this form as completely as possible. Note: There is no fee to apply. Please send additional materials such as your pitch deck and executive summary to resi@lifesciencenation.com for us
Please fill out this form as completely as possible. Note: There is no fee to apply. Please send additional materials such as your pitch deck and executive summary to resi@lifesciencenation.com for us
Advancing New Treatments for DMD and C. difficile Infection
 Advancing New Treatments for DMD and C. difficile Infection 34 th Annual Canaccord Genuity Growth Conference August 13, 2014 Legal Disclaimer FORWARD-LOOKING STATEMENTS This Document contains forward-looking
Advancing New Treatments for DMD and C. difficile Infection 34 th Annual Canaccord Genuity Growth Conference August 13, 2014 Legal Disclaimer FORWARD-LOOKING STATEMENTS This Document contains forward-looking
Chemoinformatic Tools for the Hit Discovery Process
 Chemoinformatic Tools for the Hit Discovery Process GmbH Björn Windshügel May 29 2013 ESP in a Nutshell Established in 2007 Service provider for academics High-throughput screening High-content screening
Chemoinformatic Tools for the Hit Discovery Process GmbH Björn Windshügel May 29 2013 ESP in a Nutshell Established in 2007 Service provider for academics High-throughput screening High-content screening
Corporate Presentation OCTOBER 2018
 Corporate Presentation OCTOBER 2018 This presentation contains forward-looking statements. All statements other than statements of historical fact are forward-looking statements, which are often indicated
Corporate Presentation OCTOBER 2018 This presentation contains forward-looking statements. All statements other than statements of historical fact are forward-looking statements, which are often indicated
ibio, Inc. Holds Annual Meeting in College Station, Texas
 April 7, 2016 ibio, Inc. Holds Annual Meeting in College Station, Texas NEW YORK, NY -- (Marketwired) -- 04/07/16 -- ibio, Inc. (NYSE MKT: IBIO) - Speaking to shareholders at the ibio, Inc. (NYSE MKT:
April 7, 2016 ibio, Inc. Holds Annual Meeting in College Station, Texas NEW YORK, NY -- (Marketwired) -- 04/07/16 -- ibio, Inc. (NYSE MKT: IBIO) - Speaking to shareholders at the ibio, Inc. (NYSE MKT:
Microbial Biotechnology agustin krisna wardani
 Microbial Biotechnology agustin krisna wardani 1. The Structure of Microbes Microbes (microorganisms) are tiny organisms that are too small to be seen individually by the naked eye and must be viewed with
Microbial Biotechnology agustin krisna wardani 1. The Structure of Microbes Microbes (microorganisms) are tiny organisms that are too small to be seen individually by the naked eye and must be viewed with
leading the way in research & development
 leading the way in research & development people. passion. possibilities. ABBVIE 2 immunology AbbVie Immunology has a demonstrated record of success in identifying and developing both small molecule and
leading the way in research & development people. passion. possibilities. ABBVIE 2 immunology AbbVie Immunology has a demonstrated record of success in identifying and developing both small molecule and
Trust Yours. Protect Theirs.
 Trust Yours. Protect Theirs. kemin.com/guthealth Ruminant Gut Health Due to customer demands, producers have been asked to change the way they raise production animals. In order to maintain feed efficiency,
Trust Yours. Protect Theirs. kemin.com/guthealth Ruminant Gut Health Due to customer demands, producers have been asked to change the way they raise production animals. In order to maintain feed efficiency,
From Discovery to Development of new Drugs. and pitfalls along the way. by Kim Dekermendjian, PhD in Medicine BD & Key Account manager
 From Discovery to Development of new Drugs. and pitfalls along the way by Kim Dekermendjian, PhD in Medicine BD & Key Account manager The roots of Drug Discovery Before 20 th century the term didn't exists,
From Discovery to Development of new Drugs. and pitfalls along the way by Kim Dekermendjian, PhD in Medicine BD & Key Account manager The roots of Drug Discovery Before 20 th century the term didn't exists,
-- Sarepta strengthens position as a leader in gene therapy; expands rare disease franchise --
 Sarepta Therapeutics Announces Partnership with Myonexus Therapeutics for the Advancement of Multiple Gene Therapy Programs Aimed at Treating Distinct Forms of Limb-Girdle Muscular Dystrophies -- Sarepta
Sarepta Therapeutics Announces Partnership with Myonexus Therapeutics for the Advancement of Multiple Gene Therapy Programs Aimed at Treating Distinct Forms of Limb-Girdle Muscular Dystrophies -- Sarepta
Microbiota: Agents for Health and Disease Dr. B. Brett Finlay
 Microbiota: Agents for Health and Disease, OC, OBC Michael Smith Laboratory University of British Columbia Vancouver, Canada 1 Talk outline A general overview: Several aspects of microbiota Various contribution
Microbiota: Agents for Health and Disease, OC, OBC Michael Smith Laboratory University of British Columbia Vancouver, Canada 1 Talk outline A general overview: Several aspects of microbiota Various contribution
CHAPTER INTRODUCTION 1.1 ORAL DRUG DELIVERY: SCOPE AND ADVANTAGES
 1.0 INTRODUCTION 1.1 ORAL DRUG DELIVERY: SCOPE AND ADVANTAGES Oral ingestion is the preferred route for administration of therapeutic agents, providing a convenient method of effectively achieving both
1.0 INTRODUCTION 1.1 ORAL DRUG DELIVERY: SCOPE AND ADVANTAGES Oral ingestion is the preferred route for administration of therapeutic agents, providing a convenient method of effectively achieving both
Inflammatory Bowel Disease
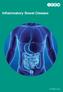 Inflammatory Bowel Disease ICONplc.com Inflammatory bowel disease (IBD) is a chronic, relapsing, inflammatory disorder of the gastrointestinal tract and includes Ulcerative Colitis (UC) and Crohn s Disease
Inflammatory Bowel Disease ICONplc.com Inflammatory bowel disease (IBD) is a chronic, relapsing, inflammatory disorder of the gastrointestinal tract and includes Ulcerative Colitis (UC) and Crohn s Disease
Patentability/Literature Research
 Patentability/Literature Research The probiotic-based solution to combat cholera as presented here comprises of two major innovative components: (1) the metabolite-dependent pathogen inhibition by a probiotic
Patentability/Literature Research The probiotic-based solution to combat cholera as presented here comprises of two major innovative components: (1) the metabolite-dependent pathogen inhibition by a probiotic
SECURITIES AND EXCHANGE COMMISSION Washington, D.C FORM 8-K CURRENT REPORT
 SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event reported):
SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event reported):
A D V A N C I N G I N N O V A T I O N I N H E A L T H C A R E Partners Innovation Fund
 A D V A N C I N G I N N O V A T I O N I N H E A L T H C A R E Partners Innovation Fund Partners HealthCare July 2017 P A R T N E R S I N N O V A T I O N F U N D In Brief Capitalization $165M fund $100
A D V A N C I N G I N N O V A T I O N I N H E A L T H C A R E Partners Innovation Fund Partners HealthCare July 2017 P A R T N E R S I N N O V A T I O N F U N D In Brief Capitalization $165M fund $100
REIMAGINING DRUG DEVELOPMENT:
 Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
The Leader in AAV Gene Therapy. A Guide to AAV Gene Therapy for MPS I and II
 The Leader in AAV Gene Therapy A Guide to AAV Gene Therapy for MPS I and II REGENXBIO seeks to understand the diverse perspectives of patients, caregivers and families, and to learn from their experiences
The Leader in AAV Gene Therapy A Guide to AAV Gene Therapy for MPS I and II REGENXBIO seeks to understand the diverse perspectives of patients, caregivers and families, and to learn from their experiences
Q Earnings and Corporate Developments. October 31, 2018
 Q3 2018 Earnings and Corporate Developments October 31, 2018 1 Intellia Therapeutics Legal Disclaimers This presentation contains forward-looking statements of Intellia Therapeutics, Inc. ( Intellia )
Q3 2018 Earnings and Corporate Developments October 31, 2018 1 Intellia Therapeutics Legal Disclaimers This presentation contains forward-looking statements of Intellia Therapeutics, Inc. ( Intellia )
Advanced Microbial Protein Expression
 Advanced Microbial Protein Expression Partners for Life Advancing tomorrow s medicines w A Biologics and Vaccines CDMO partner for your complete clinical journey Good science, experience and a quality
Advanced Microbial Protein Expression Partners for Life Advancing tomorrow s medicines w A Biologics and Vaccines CDMO partner for your complete clinical journey Good science, experience and a quality
Risk-based testing for anti-drug neutralizing antibodies during development of biological therapeutics
 Risk-based testing for anti-drug neutralizing antibodies during development of biological therapeutics April 18 th, 2016 AAPS National Biotechnology Conference, Boston, MA Shalini Gupta, PhD Amgen Inc.
Risk-based testing for anti-drug neutralizing antibodies during development of biological therapeutics April 18 th, 2016 AAPS National Biotechnology Conference, Boston, MA Shalini Gupta, PhD Amgen Inc.
IDSA is pleased to offer the following comments on specific priority areas identified by FDA:
 October 31, 2018 Shashi Malhotra Office of Acquisitions & Grants Services (OAGS) Food and Drug Administration Telephone: 240-402-7592 Email: Shashi.Malhotra@fda.hhs.gov SUBJECT: NOT-FD-18-16: Development
October 31, 2018 Shashi Malhotra Office of Acquisitions & Grants Services (OAGS) Food and Drug Administration Telephone: 240-402-7592 Email: Shashi.Malhotra@fda.hhs.gov SUBJECT: NOT-FD-18-16: Development
Lux Biosciences-New Jersey Center for Biomaterials/Rutgers University Collaboration. Eddy Anglade, MD Chief Medical Officer
 Lux Biosciences-New Jersey Center for Biomaterials/Rutgers University Collaboration Eddy Anglade, MD Chief Medical fficer Company verview Development and commercialization of unique and targeted products
Lux Biosciences-New Jersey Center for Biomaterials/Rutgers University Collaboration Eddy Anglade, MD Chief Medical fficer Company verview Development and commercialization of unique and targeted products
PATHWAY TO VALUE CREATION NASDAQ: BNTC ASX: BLT
 PATHWAY TO VALUE CREATION NASDAQ: BNTC ASX: BLT Annual General Meeting Presentation 8 November 2018 Jerel A. Banks, M.D., Ph.D. Chief Executive Officer and Executive Chairman SAFE HARBOR STATEMENT This
PATHWAY TO VALUE CREATION NASDAQ: BNTC ASX: BLT Annual General Meeting Presentation 8 November 2018 Jerel A. Banks, M.D., Ph.D. Chief Executive Officer and Executive Chairman SAFE HARBOR STATEMENT This
PATHWAY TO VALUE CREATION NASDAQ: BNTC ASX: BLT. For personal use only
 PATHWAY TO VALUE CREATION NASDAQ: BNTC ASX: BLT Annual General Meeting Presentation 8 November 2018 Jerel A. Banks, M.D., Ph.D. Chief Executive Officer and Executive Chairman SAFE HARBOR STATEMENT This
PATHWAY TO VALUE CREATION NASDAQ: BNTC ASX: BLT Annual General Meeting Presentation 8 November 2018 Jerel A. Banks, M.D., Ph.D. Chief Executive Officer and Executive Chairman SAFE HARBOR STATEMENT This
Basic Information. I.1 If your company performs biotechnology research and development, uses a. The Categories of Biotechnology Applications.
 APPENDIX 2 SURVEY QUESTIONNAIRE Part I. Basic Information I.1 If your company performs biotechnology research and development, uses a biotechnology process in manufacturing, or produces research tools,
APPENDIX 2 SURVEY QUESTIONNAIRE Part I. Basic Information I.1 If your company performs biotechnology research and development, uses a biotechnology process in manufacturing, or produces research tools,
Аquaphotomics in biotechnology - new tools and opportunities
 Аquaphotomics in biotechnology - new tools and opportunities Prof. Albert Krastanov UNIVERSITY OF FOOD TECHNOLOGIES AQUAPHOTOMICS time-efficient allows rapid, chemical-free, noninvasive in vivo assessment
Аquaphotomics in biotechnology - new tools and opportunities Prof. Albert Krastanov UNIVERSITY OF FOOD TECHNOLOGIES AQUAPHOTOMICS time-efficient allows rapid, chemical-free, noninvasive in vivo assessment
Antibody therapeutic approaches for cancer
 Antibody therapeutic approaches for cancer Date2innovate Utrecht, March 21, 2014 Jan van de Winkel Forward Looking Statement This presentation contains forward looking statements. The words believe, expect,
Antibody therapeutic approaches for cancer Date2innovate Utrecht, March 21, 2014 Jan van de Winkel Forward Looking Statement This presentation contains forward looking statements. The words believe, expect,
MASTER IN PHARMACEUTICAL SCIENCES (Thesis Track)
 MASTER IN PHARMACEUTICAL SCIENCES (Thesis Track) I. GENERAL RULES CONDITIONS: Plan Number 1 \ 12 70 2005 T 1. This plan conforms to the valid regulations of the programs of graduate studies. 2. Areas of
MASTER IN PHARMACEUTICAL SCIENCES (Thesis Track) I. GENERAL RULES CONDITIONS: Plan Number 1 \ 12 70 2005 T 1. This plan conforms to the valid regulations of the programs of graduate studies. 2. Areas of
EOS: a Focussed Oncology Company Overview Forum della Ricerca e dell Innovazione Padova, May 16, 2008
 Growing and Nurturing Right Ideas EOS: a Focussed Oncology Company Overview Forum della Ricerca e dell Innovazione Padova, May 16, 2008 EOS S.p.A. (Ethical Oncology Science) Via Monte di Pietà 1/A, Milan,
Growing and Nurturing Right Ideas EOS: a Focussed Oncology Company Overview Forum della Ricerca e dell Innovazione Padova, May 16, 2008 EOS S.p.A. (Ethical Oncology Science) Via Monte di Pietà 1/A, Milan,
L Intérêt du Projet CTI
 L Intérêt du Projet CTI Prof. Yogeshvar N. Kalia Molecular Therapeutics Delivery Group Section des sciences pharmaceutiques CMU, 1 Rue Michel Servet, Genève Email: yogi.kalia@unige.ch REUSSIR VOS PROJETS
L Intérêt du Projet CTI Prof. Yogeshvar N. Kalia Molecular Therapeutics Delivery Group Section des sciences pharmaceutiques CMU, 1 Rue Michel Servet, Genève Email: yogi.kalia@unige.ch REUSSIR VOS PROJETS
From Resilience To Growth:
 From Resilience To Growth: Mapping a New Direction for Life Sciences & Medical Devices Attorney Advertising Prior results do not guarantee a similar outcome Models used are not Mapping the Way: Opportunities
From Resilience To Growth: Mapping a New Direction for Life Sciences & Medical Devices Attorney Advertising Prior results do not guarantee a similar outcome Models used are not Mapping the Way: Opportunities
Nanotechnology: A Brief History and Its Convergence with Medicine. Weston Daniel, PhD Director of Program Management
 Nanotechnology: A Brief History and Its Convergence with Medicine Weston Daniel, PhD Director of Program Management Outline Introduction The Nanoscale Applications Realization of a Vision There s Plenty
Nanotechnology: A Brief History and Its Convergence with Medicine Weston Daniel, PhD Director of Program Management Outline Introduction The Nanoscale Applications Realization of a Vision There s Plenty
Phase 1 Clinical Studies First-In-Human (FIH) <Chapter 31> Pharmacologically-Guided Dose Escalation
 Phase 1 Clinical Studies First-In-Human (FIH) Pharmacologically-Guided Dose Escalation Jerry M. Collins, Ph.D. Developmental Therapeutics Program Division of Cancer Treatment and Diagnosis,
Phase 1 Clinical Studies First-In-Human (FIH) Pharmacologically-Guided Dose Escalation Jerry M. Collins, Ph.D. Developmental Therapeutics Program Division of Cancer Treatment and Diagnosis,
Adoptive cellular therapies are based on the administration of live cells into a patient in order for them to serve a therapeutic purpose.
 Adoptive cellular therapies are based on the administration of live cells into a patient in order for them to serve a therapeutic purpose. Pharmaceutical products have been historically made of purified
Adoptive cellular therapies are based on the administration of live cells into a patient in order for them to serve a therapeutic purpose. Pharmaceutical products have been historically made of purified
