Focus on biological and biosimilar drugs in Europe: pricing, reimbursement and access issues
|
|
|
- Christiana Webb
- 6 years ago
- Views:
Transcription
1 Corvinus Health Policy and Health Economics Conference Series 2015/5 Organiser: Department of Health Economics, Corvinus University of Budapest Co-organisers: Health Economics Study Circle, Corvinus University of Budapest Health and Health Care Economics Section of the Hungarian Economic Association Focus on biological and biosimilar drugs in Europe: pricing, reimbursement and access issues INVITATION
2 Corvinus Health Policy and Health Economics Conference Series 2015/5 organized by Department of Health Economics, Corvinus University of Budapest co-organisers Health Economics Study Circle, Corvinus University of Budapest Health and Health Care Economics Section of the Hungarian Economic Association Focus on biological and biosimilar drugs in Europe: pricing, reimbursement and access issues Corvinus University of Budapest 20 October, 2015 Room 3001, 15:30-17:45 Budapest, Fővám tér 8 1 Background Biological drugs revolutionized the treatment of chronic inflammatory diseases in rheumatology, gastroenterology and dermatology, as well as of oncology diseases. This conference aims to provide an update on the main clinical and health economic issues related to biological drugs with special focus on biosimilars. 1 Map: hl=hu&ie=utf8&sll= , &sspn= , &oq=f%C5%91v%C3%A1m+t%C3%A9r+8&hnear=Budapest,+IX.+ker %C3%BClet,+F%C5%91v%C3%A1m+t%C3%A9r+8,+1093+Magyarorsz%C3%A1g&t=m&z=17
3 Conference chairperson: Prof. Márta Péntek, Corvinus University of Budapest, Department of Health Economics; President of the Health and Health Care Economics Section of the Hungarian Economic Association Scientific programme committee: Prof. László Gulácsi, head, Department of Health Economics Corvinus University of Budapest Prof. Márta Péntek, Department of Health Economics Corvinus University of Budapest; President of the Health and Health Care Economics Section of the Hungarian Economic Association Programme 15:30 15:35 Opening Prof. Márta Péntek 15:35 16:15 NICE 2 approach to biological and biosimilar drugs Paul Cornes Clinical Outcomes Group, Bristol, United Kingdom Abstract: Introduction to biological biosimilar drugs and QALYs NICE decision making Transferability of NICE decision to other countries Practicing physicians perspective 16:15 16:40 Biosimilars at the interface of science, medicine and economics Uwe Gudat Head of Safety Biosimilars, Merck Serono, Aubonne, Switzerland Abstract: Benefitting the patient by enhancing access to medicines A long history of comparable biologics Biologic product identity: a heterogeneous blend of molecular species Totality of evidence: minimizing residual uncertainty, maximizing safety Benefit vs. risk at the population and personal level Understanding is the key to acceptance 16:40 17:05 Biotechnology revolution: The industry perspective. Mike Muenzberg Global Medical Director of Biosimilar Unit, Merck Serono, Aubonne, Switzerland Abstract: Manufacturer s challenges and manufacturing costs, Why expensive diversity in approach in different countries Challenges in the 2 nd wave of biosimilars Access issues & Eligibility 2 National Institute for Health and Care Excellence 3
4 17:05 17:15 Issues of biological therapies in Central and Eastern Europe Zsombor Zrubka Ph.D. student, Corvinus University of Budapest Head of Specialty Business Unit, Sandoz 17:15 17:45 Discussion Invited discutant: Ágnes Mészáros Pharm.D., Ph.D., habil associate professor University Pharmacy Department of Pharmacy Administration Faculty of Pharmacy Semmelweis University; honorary professor Department of Health Economics, Faculty of Economics, Corvinus University of Budapest 17:45 Closing Relevant recent publications, Department of Health Economics, Corvinus University Brodszky V, Rencz F, Péntek, Baji P, Lakatos PL, Gulácsi L, A budget impact model for biosimilar infliximab in Crohn's disease in Bulgaria, the Czech Republic, Hungary, Poland, Romania and Slovakia. Expert Rev Pharmacoecon Outcomes Res Jul 10:1-7. IF: 1,669. Brodszky V, Baji P, Balogh O, Pentek M: Budget impact analysis of biosimilar infliximab (CT-P13) for the treatment of rheumatoid arthritis in six Central and Eastern European countries. European Journal Of Health Economics 15:(1) pp IF: 1,774 Gulácsi L, Brodszky V, Baji P, HoUng Kim, Su Yeon Kim, Yu Young Cho, PéntekM, Biosimilars for the management of rheumatoid arthritis: economic considerations. Expert Rev. Clin. Immunol (S1), S43 S52 (2015) IF: 2,484 Gulácsi, L., Rencz, F., Péntek, M., Brodszky, V., Lopert, R., Hever, N. V., Baji, P. Transferability of results of cost utility analyses for biologicals in inflammatory conditions for Central and Eastern European countries. Eur J Health Econ. 2014, 15 Suppl 1, S IF: 1,913 Gulácsi, L., Rotar, A. M., Niewada, M., Loblova, O., Rencz, F., Petrova, G., Boncz, I., Klazinga, N. S. Health technology assessment in Poland, the Czech Republic, Hungary, Romania and Bulgaria. Eur J Health Econ. 2014, 15 Suppl 1, IF: 1,913 Baji P, Gulácsi L, Lovász BD, Golovics PA., Brodszky V, Péntek M, Rencz F, Lakatos P. Treatment preferences of originator versus biosimilar drugs in Crohn s disease; discrete choice experiment among gastroenterologists. Scandinavian Journal of Gastroenterology June 10: 1-6. (doi: / ) Early Online. IF: 2,361 Baji P, Gulácsi L, Golovics PA., Lovász BD, Péntek M, Brodszky V, Rencz F, Lakatos P. Perceived risks contra benefits of using biosimilar drugs in ulcerative colitis; discrete choice experiment among gastroenterologists. Submitted to Digestive and Liver disease (IF=2,963), October Baji, P., Péntek, M., Czirják, L., Szeé8kanecz, Z., Nagy, G., Gulácsi, L., Brodszky, V. Efficacy and safety of infliximab-biosimilar compared to other biological drugs in rheumatoid arthritis: a mixed treatment comparison. Eur J Health Econ. 2014, 15 Suppl 1, IF: 1,774 Baji, P., Péntek, M., Szanto, S., Geher, P., Gulacsi, L., Balogh, O., Brodszky, V. Comparative efficacy and safety of biosimilar infliximab and other biological treatments in ankylosing spondylitis: systematic literature review and meta-analysis. Eur J Health Econ. 2014, 15 Suppl 1, IF: 1,774 4
5 Paul Cornes Clinical Outcomes Group 9 Royal Victoria Park, Bristol, BS10 6TD, UK paul.cornes@yahoo.co.uk Phone: CV Paul Cornes is an Oncologist from Bristol, UK, where biosimilars and generics are routine practice. He is part of the steering group for the European School of Oncology Working Party on the Access to Innovation in Cancer Treatment. He encourages physicians that health economics is to be embraced as part of routine medical practice. We live in an era of both unprecedented medical advances in targeted treatments and international economic troubles. Physicians, hospitals and nations now have to make difficult choices to offer the best care to patients when budgets are under such strain. Paul teaches that we have to move forward from Evidence Based Medicine EBM to adopt Value Based Medicine VBM if innovation is to be affordable. Education is the single most important weapon against cancer. Paul has taught cost effective care in Countries as diverse as the USA, Russia, Saudi Arabia, China and Japan and has been the American Society of Hematology Annual Spotlight Lecturer for cost control. He gives the health economics lectures for his EORTC group. He organised the first ever cost-effectiveness conference in Turkey, and at the first teaching programme on cost-control for Oncologists in Japan. He gave 2 key invited lectures on the importance of VBM at the recent 18th ECCO 40th ESMO European Cancer Congress in Vienna, 2015 He has a special interest in delivering innovation in cancer treatment in middle-income countries. With the Malaysian Ministry of Health he has run workshops on cost-effective care to redesign their National Cancer Reimbursement Programme. You can also see him as a key speaker in the special day on cost control at the 18th ECCO 40th ESMO European Cancer Congress in Vienna,
6 Uwe Gudat Merck Serono Head of Safety Biosimilars Zone Industrielle de l Ouriettaz Aubonne. CH Switzerland uwe.gudat@merckgroup.com Dr. Uwe Gudat is Head of Safety, Biosimilars at the Merck Serono Biosimilar Unit. Dr. Gudat is licensed in internal medicine with diabetology as a sub specialty. He studied in Marburg, Germany, and received his post-graduate training under Michael Berger in Düsseldorf, Germany. He joined the pharmaceutical industry in 1995 and has held positions at Eli Lilly & Company, Actelion, and Novartis as medical director, leading global clinical development and in-licensing teams, as well as guiding clinical trial methodology and clinical safety evaluations. Dr Gudat joined Merck Serono in 2009 and has been Head of Safety Biosimilars since Oct
7 Mike Muenzberg Ares Trading S.A. Aubonne Business Center-Building Z Z.I. de l'ouriettaz CH-1170 Aubonne mike.muenzberg@merckgroup.com Dr. Mike Muenzberg is Global Medical Director at Merck Seronos Biosimilar Unit. He was born in Austria and educated in Austria, Canada and Sweden. Dr. Muenzberg is licensed as Doctor of Nuclear Medicine and has more than 15 years experience in Pharmaceutical Industry, working as local as well as global Medical Manager/Director for Serono, Novartis, Amgen, Roche and Sandoz International Biopharmaceuticals. Since 2014 Dr. Muenzberg holds his position as Global Medical Director Biosimilars at Merck Seronos Biosimilar Unit, responsible for Merck Seronos pipeline Biosimilars. 7
8 Zsombor Zrubka M.D., MBA Contact: cell phone: ; to PROFESSIONAL EXPERIENCE Resident in psychiatry National Institute of Psychiatry and Neurology Child psychiatrist Vadaskert Clinic of Child and Adolescent Psychiatry Sales representative Janssen-Cilag, division of Johnson & Johnson Ltd Product Manager Pfizer Hungary Brand Manager, Zeldox European Brand Team (EBT) Pfizer Germany Senior Brand Manager Zeldox, Neuroscience European Brand Team Pfizer UK Medical Team Leader Psychiatry / Zeldox - Specialty Care Business Unit Europe / Australia New Zealand Employer: Pfizer Hungary, team location: Paris, France 2010 Head of Marketing Business Unit 1 Pfizer Hungary Business Development Manager Egis Pharmaceuticals Plc International Biotechnology Business Unit Manager Autoimmune Portfolio Egis Pharmaceuticals Plc International Biotechnology and Oncology Business Unit Egis Manager Pharmaceuticals Plc International Biotechnology and Oncology Marketing Team Leader Egis Pharmaceuticals Plc Head of Specialty Business Unit Sandoz Hungary Plc. EDUCATION AND TRAINING 1997 Medical Doctor at Semmelweis University, Budapest Two semesters at Faculty in Biomedical Engineering, Budapest Technical University 2005 MBA at Oxford Brookes University Omegaglen 2009 Pfizer Advanced Leadership Program (Pfizer UK) 2015 PhD Student at Corvinus University SCIENTIFIC WORKS, PUBLICATIONS 2003 Diploma in Management Studies - Project Report: Effective sales force management (FF optimization techniques) 8
9 2005 MBA Diploma: Improving promotional effectiveness of a premium pharmaceutical brand (promotional ROI, brand equity management) 2005 Invited speaker & workshop leader on pharmaceutical brand building - Pirula Katedra Workshop, Hungary 2006 Speaker at Jacob-Fleming VI. International Pharmaceutical Branding Conference in Barcelona Neuroscience of Branding 2014 Speaker at Informa Life Sciences Biosimilars Congress in Berlin Experiences 2015 Scientific poster accepted for the European Workshop for Rheumatology Research (EWRR) in Budapest: Authorized Manufacturing Changes of Therapeutic Monoclonal Antibodies in EPAR Documents 9
10 Ágnes Mészáros Ph.D., habil Contact Department of Pharmacy Administration Faculty of Pharmacy Semmelweis University H-1092, Hőgyes Endre u Dr Agnes Meszaros qualified as a pharmacist in 1997 at the Semmelweis Medical University; since then she was working and teaching at the Faculty of Pharmacy. As Chevening scholar she was studying at the University of York and obtained an MSc degree in Health Economics. Her research interest focuses on the field of pharmacoeconomics, quality of life studies, adherence evaluation of chronic patients and clinical pharmacy/pharmaceutical care. She is the founder member of the Hungarian Health Economics Association, and member of several national and international organisations. 10
11 Márta Péntek M.D., Ph.D., habil CONTACT Corvinus University of Budapest, Department of Health Economics Fővám tér 8, 1093 Budapest, Hungary QUALIFICATIONS Medical Doctor, Semmelweis University Medical School, 1989 Rheumatology and Physiatry, specialisation - Semmelweis University, 1997 Ph.D. - Semmelweis University 2008, thesis: Health status and disease burden of rheumatoid arthritis patients in Hungary (Supervisor: Prof. László Gulácsi) Habilitation University of Pécs, 2013 WORKPLACE 1989 to present: senior consultant rheumatologist, Flór Ferenc County Hospital, Department of Rheumatology (part-time since 2008); Address: Semmelweis tér 1, Kistracsa, H-2143 Hungary and 2009 to present: associate professor, Corvinus University of Budapest, Department of Health Economics; Address: Fővám tér 8, Budapest, H-2143 Hungary LANGUAGE English, Portuguese, Hungarian PUBLICATIONS Scientific papers: 77 (IF: 68.7) Independent citations: 266 Hirsch index: 13 LIST OF PUBLICATIONS s_on=&sp_on=&st_on=&url_on=1&cite_type=2&orderby=3d1a&scientific=1&independent =&top10=&lang=1&location=mtmt&debug=&stn=1&authorid= &documentid=& tipus=&besorolas=&jelleg= 11
Department of Health Economics, Corvinus University of Budapest; 8 Fővám tér, 1092 Budapest, HUNGARY
 PETRA BAJI Address E-mail Department of Health Economics, Corvinus University of Budapest; 8 Fővám tér, 1092 Budapest, HUNGARY petra.baji@uni-corvinus.hu; pbaji.bce@gmail.com WORK EXPERIENCE 2016 Associate
PETRA BAJI Address E-mail Department of Health Economics, Corvinus University of Budapest; 8 Fővám tér, 1092 Budapest, HUNGARY petra.baji@uni-corvinus.hu; pbaji.bce@gmail.com WORK EXPERIENCE 2016 Associate
Biosimilars 101: How similar are they?
 Biosimilars 101: How similar are they? Matthew Marston, Pharm.D., BCPS, BCOP Senior Manager of Clinical Pharmacy Services Miller Drug, LLC Outline Describe the approval and manufacturing process of biologics
Biosimilars 101: How similar are they? Matthew Marston, Pharm.D., BCPS, BCOP Senior Manager of Clinical Pharmacy Services Miller Drug, LLC Outline Describe the approval and manufacturing process of biologics
what you need to know
 ESMO 2017 INDUSTRY SATELLITE SYMPOSIUM A sector group Biosimilars for Oncologists FRIDAY 8 SEPTEMBER 2017 18:00-20:00 PAMPLONA AUDITORIUM, HALL 4 sss IFEMA, FERIA DE MADRID, MADRID, SPAIN The Biosimilar
ESMO 2017 INDUSTRY SATELLITE SYMPOSIUM A sector group Biosimilars for Oncologists FRIDAY 8 SEPTEMBER 2017 18:00-20:00 PAMPLONA AUDITORIUM, HALL 4 sss IFEMA, FERIA DE MADRID, MADRID, SPAIN The Biosimilar
A Physician s consideration towards Biosimilars. João Eurico Fonseca
 A Physician s consideration towards Biosimilars João Eurico Fonseca Disclosure I received unrestricted research grants or acted as a speaker for Abbvie, Amgen, BMS, Celtrion, Celgene, Janssen, MSD, Novartis,
A Physician s consideration towards Biosimilars João Eurico Fonseca Disclosure I received unrestricted research grants or acted as a speaker for Abbvie, Amgen, BMS, Celtrion, Celgene, Janssen, MSD, Novartis,
Biosimilars Market Update
 Biosimilars Market Update Panel: Matthew Brougham Consultant Economist, Brougham Consulting Inc Mark Jackson Consultant Pharmacist, TELUS Health Dr. Ed Keystone Professor of Medicine, University of Toronto
Biosimilars Market Update Panel: Matthew Brougham Consultant Economist, Brougham Consulting Inc Mark Jackson Consultant Pharmacist, TELUS Health Dr. Ed Keystone Professor of Medicine, University of Toronto
Workshop on Access to and Uptake of Biosimilar Medicinal Products
 Workshop on Access to and Uptake of Biosimilar Medicinal Products A follow-up event to Process on Corporate Responsibility in the Pharma Sector Brussels, 6 October 2015, 10.30am Auditorium, Directorate
Workshop on Access to and Uptake of Biosimilar Medicinal Products A follow-up event to Process on Corporate Responsibility in the Pharma Sector Brussels, 6 October 2015, 10.30am Auditorium, Directorate
This presentation was developed by the Government & Industry Affairs Committee of the Crohn s & Colitis Foundation of America
 This presentation was developed by the Government & Industry Affairs Committee of the Crohn s & Colitis Foundation of America The development of this presentation was supported in 2016 by a sponsorship
This presentation was developed by the Government & Industry Affairs Committee of the Crohn s & Colitis Foundation of America The development of this presentation was supported in 2016 by a sponsorship
Hospira at a 5/20/2015. The World s Leading Provider of Injectable Drugs and Infusion Technologies
 Sumant Ramachandra, MD, PhD SVP and Chief Scientific Officer Alliance for Health Reform May 20 th, 2015 Hospira at a glance The World s Leading Provider of Injectable Drugs and Infusion Technologies Market
Sumant Ramachandra, MD, PhD SVP and Chief Scientific Officer Alliance for Health Reform May 20 th, 2015 Hospira at a glance The World s Leading Provider of Injectable Drugs and Infusion Technologies Market
The benefits of biosimilar medicines
 Chapter 5 The benefits of biosimilar medicines Biosimilar medicines have demonstrated similarity with reference biologicals in terms of structure, function, safety and efficacy, but what are their benefits?
Chapter 5 The benefits of biosimilar medicines Biosimilar medicines have demonstrated similarity with reference biologicals in terms of structure, function, safety and efficacy, but what are their benefits?
The importance of interchangeability in the procurement of medications: Biosimilar case
 The importance of interchangeability in the procurement of medications: Biosimilar case ALIMS Congres, Kragujevac, November 6, 2015 Prof. Borut Štrukelj, PhD Brief CV Current position: professor, Pharmaceutical
The importance of interchangeability in the procurement of medications: Biosimilar case ALIMS Congres, Kragujevac, November 6, 2015 Prof. Borut Štrukelj, PhD Brief CV Current position: professor, Pharmaceutical
RE: Considerations in Demonstrating Interchangeability With a Reference Product; Draft Guidance for Industry; Availability (FDA 2017 D )
 May 19, 2017 Scott Gottlieb, MD Commissioner U.S. Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 RE: Considerations in Demonstrating Interchangeability With a Reference
May 19, 2017 Scott Gottlieb, MD Commissioner U.S. Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 RE: Considerations in Demonstrating Interchangeability With a Reference
CLINICAL TRIALS IN RUSSIA
 RESEARCH REPORT CLINICAL TRIALS IN RUSSIA SUMMER 2018 RESEARCH GROUP WWW.SRGCRO.COM FOREWORD The Orange Paper is a free publication produced by Synergy Research Group for the pharmaceutical industry since
RESEARCH REPORT CLINICAL TRIALS IN RUSSIA SUMMER 2018 RESEARCH GROUP WWW.SRGCRO.COM FOREWORD The Orange Paper is a free publication produced by Synergy Research Group for the pharmaceutical industry since
Publications for payors: what evidence do they really need? Ian Pickles, Strategy Consultant, Complete Clarity
 Publications for payors: what evidence do they really need? Ian Pickles, Strategy Consultant, Complete Clarity OBJECTIVES FOR THE SESSION To be able to build payer evidence requirements into publication
Publications for payors: what evidence do they really need? Ian Pickles, Strategy Consultant, Complete Clarity OBJECTIVES FOR THE SESSION To be able to build payer evidence requirements into publication
The Evolving Role Of Prescription Benefit Managers
 The Evolving Role Of Prescription Benefit Managers Steve Miller, MD Senior Vice President & Chief Medical Officer November 1, 2016 1 Prescription drugs: A growing share of U.S. healthcare spend Net spending
The Evolving Role Of Prescription Benefit Managers Steve Miller, MD Senior Vice President & Chief Medical Officer November 1, 2016 1 Prescription drugs: A growing share of U.S. healthcare spend Net spending
Naming, tracing, switching and other safety issues after 10 years learning
 Naming, tracing, switching and other safety issues after 10 years learning Prof. Dr. Irene Krämer, Department of Pharmacy Johannes Gutenberg-University Medical Center, Mainz (Germany) Disclosures Speaker
Naming, tracing, switching and other safety issues after 10 years learning Prof. Dr. Irene Krämer, Department of Pharmacy Johannes Gutenberg-University Medical Center, Mainz (Germany) Disclosures Speaker
Professor Kimme Hyrich, MD, PhD, FRCPC, UK
 GaBI Scientific Meetings 26 January 2017, Pullman London St Pancras, London, UK ROUNDTABLE ON REGISTRIES Practical Considerations for Registries making them work Professor Kimme Hyrich, MD, PhD, FRCPC,
GaBI Scientific Meetings 26 January 2017, Pullman London St Pancras, London, UK ROUNDTABLE ON REGISTRIES Practical Considerations for Registries making them work Professor Kimme Hyrich, MD, PhD, FRCPC,
THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 118 PATIENT GROUPS with an interest in RESPIRATORY CONDTIONS
 THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 118 PATIENT GROUPS with an interest in RESPIRATORY CONDTIONS PUBLISHED OCTOBER 2016 [Question to respondents: Which companies have the best record
THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 118 PATIENT GROUPS with an interest in RESPIRATORY CONDTIONS PUBLISHED OCTOBER 2016 [Question to respondents: Which companies have the best record
LSE Health Market Access Academy April 2018, London September 2018, London
 LSE Health Market Access Academy 9 13 April 2018, London 10 14 September 2018, London In cooperation with the Boston Consulting Group Market Access Roundtable LSE Health Market Access Academy 2 The London
LSE Health Market Access Academy 9 13 April 2018, London 10 14 September 2018, London In cooperation with the Boston Consulting Group Market Access Roundtable LSE Health Market Access Academy 2 The London
The Future Role of Biosimilars: An Unknown Frontier in IBD Treatment
 The Future Role of Biosimilars: An Unknown Frontier in IBD Treatment Miguel Regueiro, MD, FACG Professor of Medicine Medical Director, IBD Center Senior Medical Lead, Specialty Homes University of Pittsburgh
The Future Role of Biosimilars: An Unknown Frontier in IBD Treatment Miguel Regueiro, MD, FACG Professor of Medicine Medical Director, IBD Center Senior Medical Lead, Specialty Homes University of Pittsburgh
Speakers Title & Biography
 Speakers Title & Biography Dr. Janice M. Soreth Deputy Director, FDA Europe Office/CDER, Liaison to EMA Dr. Soreth 20-year agency veteran with unparalleled experience, including serving as CDER's director
Speakers Title & Biography Dr. Janice M. Soreth Deputy Director, FDA Europe Office/CDER, Liaison to EMA Dr. Soreth 20-year agency veteran with unparalleled experience, including serving as CDER's director
Professor Ahmed H Al-jedai, PharmD, MBA, BCPS, FCCP, FAST, Saudi Arabia
 GaBI Scientific Meetings First GCC Stakeholder Meeting on Approval Process, Interchangeability/Substitution and Safety of Biosimilars 20 November 2017, Holiday Inn Izdihar Riyadh, Saudi Arabia Professor
GaBI Scientific Meetings First GCC Stakeholder Meeting on Approval Process, Interchangeability/Substitution and Safety of Biosimilars 20 November 2017, Holiday Inn Izdihar Riyadh, Saudi Arabia Professor
Roche in Australia Innovation Leader
 Roche in Australia Innovation Leader About Roche Innovation: it s in our DNA 7,000,874 hours of work 6,587 experiments 423 researchers 1 drug For nearly 120 years, Roche has been committed to improving
Roche in Australia Innovation Leader About Roche Innovation: it s in our DNA 7,000,874 hours of work 6,587 experiments 423 researchers 1 drug For nearly 120 years, Roche has been committed to improving
Publication Planning in Emerging Markets: Scientific, Cultural, and Practical Issues
 Publication Planning in Emerging Markets: Scientific, Cultural, and Practical Issues Leslie Citrome, MD, MPH Associate Editor for Psychiatry, International Journal of Clinical Practice, Wiley-Blackwell
Publication Planning in Emerging Markets: Scientific, Cultural, and Practical Issues Leslie Citrome, MD, MPH Associate Editor for Psychiatry, International Journal of Clinical Practice, Wiley-Blackwell
Workshop on Access to and Uptake of Biosimilar Medicinal Products
 EUROPEAN COMMISSION Directorate-General for Internal Market, Industry, Entrepreneurship and SMEs Consumer, Environmental and Health Technologies Biotechnology and Food Supply Chain Workshop on Access to
EUROPEAN COMMISSION Directorate-General for Internal Market, Industry, Entrepreneurship and SMEs Consumer, Environmental and Health Technologies Biotechnology and Food Supply Chain Workshop on Access to
Why even have this talk? Disclosures. What will biosimilars mean for us? Annual Revenue Due to Adalimumab. Biosimilars 2017: What We Need to Know
 Biosimilars 2017: What We Need to Know Why even have this talk? Miguel Regueiro, M.D. Professor of Medicine IBD Clinical Medical Director Senior Medical Lead, Specialty Medical Homes University of Pittsburgh
Biosimilars 2017: What We Need to Know Why even have this talk? Miguel Regueiro, M.D. Professor of Medicine IBD Clinical Medical Director Senior Medical Lead, Specialty Medical Homes University of Pittsburgh
Outsourcing in Clinical Trials Europe 2014
 TIME TO BUST E-RECRUITMENT MYTHS! Outsourcing in Clinical Trials Europe 2014 21-22 May 2014, Brussels, Belgium you not only saved us a couple of months time, but definitively disproved two myths that have
TIME TO BUST E-RECRUITMENT MYTHS! Outsourcing in Clinical Trials Europe 2014 21-22 May 2014, Brussels, Belgium you not only saved us a couple of months time, but definitively disproved two myths that have
Re: Therapeutic Goods Administration, Consultation: Nomenclature of Biological Medicines
 September 7, 2017 Biological Science Section Therapeutic Goods Administration (TGA) PO Box 100 Woden ACT 2606 Re: Therapeutic Goods Administration, Consultation: Nomenclature of Biological Medicines Dear
September 7, 2017 Biological Science Section Therapeutic Goods Administration (TGA) PO Box 100 Woden ACT 2606 Re: Therapeutic Goods Administration, Consultation: Nomenclature of Biological Medicines Dear
Sandoz Biopharmaceuticals. Sandoz Biosimilars. More than a decade of experience
 Sandoz Biopharmaceuticals Sandoz Biosimilars More than a decade of experience 2 SANDOZ BIOPHARMACEUTICALS The pioneer and global leader in biosimilars Repeatedly innovating to break new ground 2006 Launched
Sandoz Biopharmaceuticals Sandoz Biosimilars More than a decade of experience 2 SANDOZ BIOPHARMACEUTICALS The pioneer and global leader in biosimilars Repeatedly innovating to break new ground 2006 Launched
BIOSIMILARS OF PROTEIN BASED DRUGS: CURRENT STATUS AND FUTURE DIRECTIONS
 BIOSIMILARS OF PROTEIN BASED DRUGS: CURRENT STATUS AND FUTURE DIRECTIONS George Dranitsaris B.Pharm. PhD Consultant in Health Economics and Biostatistics What is a Biosimilar? Protein based drugs are made
BIOSIMILARS OF PROTEIN BASED DRUGS: CURRENT STATUS AND FUTURE DIRECTIONS George Dranitsaris B.Pharm. PhD Consultant in Health Economics and Biostatistics What is a Biosimilar? Protein based drugs are made
Interdisciplinary Perspectives on Development of Naming Standards for Biosimilar Medicines
 Interdisciplinary Perspectives on Development of Naming Standards for Biosimilar Medicines Richard Dolinar, MD Chairman, Alliance for Safe Biologic Medicines Disclaimer The views and opinions expressed
Interdisciplinary Perspectives on Development of Naming Standards for Biosimilar Medicines Richard Dolinar, MD Chairman, Alliance for Safe Biologic Medicines Disclaimer The views and opinions expressed
Message from Genentech s Executive Sponsor
 Message from Genentech s Executive Sponsor Dear Prospective Fellow: Genentech is excited to be a part of the University of the Pacific s Fellowship in Industry Program (FIP) for Doctor of Pharmacy graduates.
Message from Genentech s Executive Sponsor Dear Prospective Fellow: Genentech is excited to be a part of the University of the Pacific s Fellowship in Industry Program (FIP) for Doctor of Pharmacy graduates.
How to maximize patient access Perspectives from The Netherlands
 Budapest, June 13, 2018 How to maximize patient access Perspectives from The Netherlands Vs18f13 Arnold G. Vulto PharmD PhD FCP Professor Em. of Hospital Pharmacy & Practical Therapeutics Erasmus University
Budapest, June 13, 2018 How to maximize patient access Perspectives from The Netherlands Vs18f13 Arnold G. Vulto PharmD PhD FCP Professor Em. of Hospital Pharmacy & Practical Therapeutics Erasmus University
SUCCESSFUL BRANDS: LATVIA ESTONIA CROATIA GERMANY UNITED KINGDOM BOSNIA-HERZEGOVINA BULGARIA MACEDONIA SERBIA CZECH REPUBLIC SLOVAKIA FRANCE AUSTRIA
 ACCESS TO SUCCESS SUCCESSFUL BRANDS: CROATIA ESTONIA LATVIA BOSNIA-HERZEGOVINA BULGARIA MACEDONIA SERBIA GERMANY UNITED KINGDOM SLOVAKIA CZECH REPUBLIC FRANCE HUNGARY AUSTRIA ITALY ACCESS TO OUR MARKETS
ACCESS TO SUCCESS SUCCESSFUL BRANDS: CROATIA ESTONIA LATVIA BOSNIA-HERZEGOVINA BULGARIA MACEDONIA SERBIA GERMANY UNITED KINGDOM SLOVAKIA CZECH REPUBLIC FRANCE HUNGARY AUSTRIA ITALY ACCESS TO OUR MARKETS
The introduction of biosimilars into clinical practice
 Commissioning Chemotherapy Services The introduction of biosimilars into clinical practice Jatinder Harchowal Chief Pharmacist / Clinical Director Royal Marsden NHS Foundation Trust Medicines lead for
Commissioning Chemotherapy Services The introduction of biosimilars into clinical practice Jatinder Harchowal Chief Pharmacist / Clinical Director Royal Marsden NHS Foundation Trust Medicines lead for
New Ways to Source Innovation in the Healthcare Industry In-licensing needs Oncology of Janssen/Johnson & Johnson Innovation
 New Ways to Source Innovation in the Healthcare Industry In-licensing needs Oncology of Janssen/Johnson & Johnson Innovation Dr. Klaus Suwelack Johnson & Johnson Innovation, Janssen-Cilag Germany, Neuss
New Ways to Source Innovation in the Healthcare Industry In-licensing needs Oncology of Janssen/Johnson & Johnson Innovation Dr. Klaus Suwelack Johnson & Johnson Innovation, Janssen-Cilag Germany, Neuss
Your Perfect Match Innovative labels. Smart solutions.
 Your Perfect Match Innovative labels. Smart solutions. Solutions Partner for the Pharmaceutical and Medical Device Industry Your Partner for Innovative Label Solutions Welcomes You! 1,000 employees generate
Your Perfect Match Innovative labels. Smart solutions. Solutions Partner for the Pharmaceutical and Medical Device Industry Your Partner for Innovative Label Solutions Welcomes You! 1,000 employees generate
NHS BioValue: The Potential for a Landmark UK NHS Real World Evidence Study on Biosimilars
 NHS BioValue: The Potential for a Landmark UK NHS Real World Evidence Study on Biosimilars Angela McFarlane Market Development Director, QuintilesIMS Angela.McFarlane@QuintilesIMS.com Copyright 2016 QuintilesIMS.
NHS BioValue: The Potential for a Landmark UK NHS Real World Evidence Study on Biosimilars Angela McFarlane Market Development Director, QuintilesIMS Angela.McFarlane@QuintilesIMS.com Copyright 2016 QuintilesIMS.
Innovations in Drug Pricing and Reimbursement:
 ADVISORY REPORT AM PL E PA G ES S A S G ES A FirstWord Dossier Advisory report and Reimbursement Published Copyright 2016 Doctor s Guide Publishing Limited Part of the FirstWord Dossier family of reports
ADVISORY REPORT AM PL E PA G ES S A S G ES A FirstWord Dossier Advisory report and Reimbursement Published Copyright 2016 Doctor s Guide Publishing Limited Part of the FirstWord Dossier family of reports
BIOSIMILARS: ON THE REALM OF REALITY DETERMINANTS OF SUCCESS
 1 Originally presented at the 2013 AAPS National Biotechnology Conference - BIOTEC Open Forum Update (see slide #2) 7/14/2013 BIOSIMILARS: ON THE REALM OF REALITY DETERMINANTS OF SUCCESS Ajaz Insight 2
1 Originally presented at the 2013 AAPS National Biotechnology Conference - BIOTEC Open Forum Update (see slide #2) 7/14/2013 BIOSIMILARS: ON THE REALM OF REALITY DETERMINANTS OF SUCCESS Ajaz Insight 2
Happy holidays from ACE!
 Arthritis research, education and advocacy news : December 2016 Happy holidays from ACE! A time to give thanks, reflect on the past and look forward to the future This is one of our favorite times of the
Arthritis research, education and advocacy news : December 2016 Happy holidays from ACE! A time to give thanks, reflect on the past and look forward to the future This is one of our favorite times of the
INDUSTRY OVERVIEW BIOLOGICS MARKET GLOBALLY AND IN CHINA. Definition of Biologics
 Certain information and statistics set out in this section and elsewhere in this Document relating to the industry in which we operate are derived from the industry report prepared by Frost & Sullivan,
Certain information and statistics set out in this section and elsewhere in this Document relating to the industry in which we operate are derived from the industry report prepared by Frost & Sullivan,
Sandoz Biopharmaceuticals. Sandoz Biosimilars. From concept to reality
 Sandoz Biopharmaceuticals Sandoz Biosimilars From concept to reality 2 SANDOZ BIOPHARMACEUTICALS The basics of biosimilars Biosimilars explained Biologic medicines are produced by a complex process involving
Sandoz Biopharmaceuticals Sandoz Biosimilars From concept to reality 2 SANDOZ BIOPHARMACEUTICALS The basics of biosimilars Biosimilars explained Biologic medicines are produced by a complex process involving
Biomanufacturing Capacity for Biosimilars: Is there enough?
 Biomanufacturing Capacity for Biosimilars: Is there enough? Cambridge Healthtech Institute s Third Annual BIOAnalytical Summit 2012 Mar 19-22 Baltimore, MD BioProcess Technology Consultants www.bptc.com
Biomanufacturing Capacity for Biosimilars: Is there enough? Cambridge Healthtech Institute s Third Annual BIOAnalytical Summit 2012 Mar 19-22 Baltimore, MD BioProcess Technology Consultants www.bptc.com
FORWARD THINKING HEALTHCARE
 AT A 2018 GLANCE 02 FORWARD THINKING HEALTHCARE Fresenius is a global healthcare group offering high-quality products and services for dialysis, hospitals, and outpatient treatment. With over 270,000 employees
AT A 2018 GLANCE 02 FORWARD THINKING HEALTHCARE Fresenius is a global healthcare group offering high-quality products and services for dialysis, hospitals, and outpatient treatment. With over 270,000 employees
Thijs J Giezen, PharmD, MSc, PhD The Netherlands
 Thijs J Giezen, PharmD, MSc, PhD The Netherlands Hospital Pharmacist, Foundation Pharmacy for Hospitals in Haarlem, The Netherlands Member of the Biosimilar Medicinal Product Working Party of European
Thijs J Giezen, PharmD, MSc, PhD The Netherlands Hospital Pharmacist, Foundation Pharmacy for Hospitals in Haarlem, The Netherlands Member of the Biosimilar Medicinal Product Working Party of European
Biosimilars are interchangeable with their reference products under the supervision of a health care person.
 EXTENDING THE USE OF BIOSIMILAR DRUGS: ARE WE WILLING TO ACCEPT THE UNCERTAINTY RELATED TO SWITCHING IN ORDER TO IMPROVE PATIENT ACCESS TO MODERN MEDICINES? Tomas Tesar, PharmD, PhD, MBA, MSc Member of
EXTENDING THE USE OF BIOSIMILAR DRUGS: ARE WE WILLING TO ACCEPT THE UNCERTAINTY RELATED TO SWITCHING IN ORDER TO IMPROVE PATIENT ACCESS TO MODERN MEDICINES? Tomas Tesar, PharmD, PhD, MBA, MSc Member of
TRIANNI Media Kit MEDIA CONTACT CORPORATE HEADQUARTERS. Trianni, Inc. San Francisco, CA
 TRIANNI Media Kit CORPORATE HEADQUARTERS Trianni, Inc. San Francisco, CA Telephone: 1.866.374.9314 MEDIA CONTACT Mandy Boyd Director of Marketing @Trianni_mAb Website: Trianni.com TECHNOLOGY LEADERSHIP
TRIANNI Media Kit CORPORATE HEADQUARTERS Trianni, Inc. San Francisco, CA Telephone: 1.866.374.9314 MEDIA CONTACT Mandy Boyd Director of Marketing @Trianni_mAb Website: Trianni.com TECHNOLOGY LEADERSHIP
Uniform licensing by EMA followed by patchwork implementation in the European Union when it comes to implementation
 Vs15i17 2nd MENA Regulatory conference on Bioequivalence, Biowaivers, Bioanalysis, Dissolution and Biosimilars Amman, 17 September 2015 Uniform licensing by EMA followed by patchwork implementation in
Vs15i17 2nd MENA Regulatory conference on Bioequivalence, Biowaivers, Bioanalysis, Dissolution and Biosimilars Amman, 17 September 2015 Uniform licensing by EMA followed by patchwork implementation in
Thema: Maatschappelijke verantwoordelijkheid van de farmaceutische industrie: beschikbaarheid en betaalbaarheid van de toegang tot zorg
 Thema: Maatschappelijke verantwoordelijkheid van de farmaceutische industrie: beschikbaarheid en betaalbaarheid van de toegang tot zorg Farma uitgedaagd. Kunnen wij spreken van fair medicine? Carin Uyl-de
Thema: Maatschappelijke verantwoordelijkheid van de farmaceutische industrie: beschikbaarheid en betaalbaarheid van de toegang tot zorg Farma uitgedaagd. Kunnen wij spreken van fair medicine? Carin Uyl-de
Trends in Medication Management. Jayson Gallant, B.Pharm, M.Sc Pharmacist, Health Benefit Management
 Trends in Medication Management Jayson Gallant, B.Pharm, M.Sc Pharmacist, Health Benefit Management Agenda Drug Claims Trends Legislative / Public Plan Initiatives Biosimilars Pipeline Focus on Oncology
Trends in Medication Management Jayson Gallant, B.Pharm, M.Sc Pharmacist, Health Benefit Management Agenda Drug Claims Trends Legislative / Public Plan Initiatives Biosimilars Pipeline Focus on Oncology
Biosimilars 2016 Balancing Fast Uptake with Industry Sustainability. Dan Ionescu Head of Pricing and Market Access Sandoz Biopharmaceuticals
 Biosimilars 2016 Balancing Fast Uptake with Industry Sustainability Dan Ionescu Head of Pricing and Market Access Sandoz Biopharmaceuticals 10 years on, biosimilars have had a strong impact on global healthcare
Biosimilars 2016 Balancing Fast Uptake with Industry Sustainability Dan Ionescu Head of Pricing and Market Access Sandoz Biopharmaceuticals 10 years on, biosimilars have had a strong impact on global healthcare
IYNC - International Youth Nuclear Congress
 IYNC - International Youth Nuclear Congress Atoms for the Future, Paris, France, October 22 2013 Nicolas ANCIAUX, Westinghouse IYNC President Ekaterina RYABIKOVSKAYA, Rosatom Country Newspaper IYNC Grants
IYNC - International Youth Nuclear Congress Atoms for the Future, Paris, France, October 22 2013 Nicolas ANCIAUX, Westinghouse IYNC President Ekaterina RYABIKOVSKAYA, Rosatom Country Newspaper IYNC Grants
3rd Global Congress On Medical & Clinical Case Reports Theme: Medical & Clinical Exploration By Global Experts About the Conference: Aim
 3rd Global Congress On Medical & Clinical Case Reports Theme: Medical & Clinical Exploration By Global Experts About the Conference: Gavin Conferences invites participants from all over the world to attend
3rd Global Congress On Medical & Clinical Case Reports Theme: Medical & Clinical Exploration By Global Experts About the Conference: Gavin Conferences invites participants from all over the world to attend
Specialty Pharmacy 101
 Specialty Pharmacy 101 The Landscape of Specialty Pharmacy Services DATE: October 27, 2011 NAME: TITLE: Clinical Pharmacist, Serve You Agenda Define specialty pharmacy Examine market trends and current
Specialty Pharmacy 101 The Landscape of Specialty Pharmacy Services DATE: October 27, 2011 NAME: TITLE: Clinical Pharmacist, Serve You Agenda Define specialty pharmacy Examine market trends and current
Europe s Number One Medical Diagnostics Provider
 Europe s Number One Medical Diagnostics Provider English www.synlab.com A network of more than 1.000 medical experts Present in more than 30 countries on 4 continents Areas of expertise: human, veterinary,
Europe s Number One Medical Diagnostics Provider English www.synlab.com A network of more than 1.000 medical experts Present in more than 30 countries on 4 continents Areas of expertise: human, veterinary,
March 17, 2018 Ranking the Experience of European Physicians Interactions With Pharma. by Tim van Tongeren with Dennis van Rooij and Carlos Capella
 March 17, 2018 Ranking the Experience of European Physicians Interactions With Pharma by Tim van Tongeren with Dennis van Rooij and Carlos Capella Executive Summary Each year, the pharmaceutical industry
March 17, 2018 Ranking the Experience of European Physicians Interactions With Pharma by Tim van Tongeren with Dennis van Rooij and Carlos Capella Executive Summary Each year, the pharmaceutical industry
TOP PAPERS FROM 2016: IBD & BIOSIMILARS CYNTHIA SEOW MBBS(HONS), MSC, FRACP UNIVERSITY OF CALGARY CDDW, BANFF, AB MARCH 5, 2017
 TOP PAPERS FROM 2016: IBD & BIOSIMILARS CYNTHIA SEOW MBBS(HONS), MSC, FRACP UNIVERSITY OF CALGARY CDDW, BANFF, AB MARCH 5, 2017 FINANCIAL INTEREST DISCLOSURE (OVER THE PAST 24 MONTHS) Speaker: Janssen,
TOP PAPERS FROM 2016: IBD & BIOSIMILARS CYNTHIA SEOW MBBS(HONS), MSC, FRACP UNIVERSITY OF CALGARY CDDW, BANFF, AB MARCH 5, 2017 FINANCIAL INTEREST DISCLOSURE (OVER THE PAST 24 MONTHS) Speaker: Janssen,
Promise and Pitfalls of MicrobiomeModulating Methods for Inflammatory
 Promise and Pitfalls of MicrobiomeModulating Methods for Inflammatory and Autoimmune Diseases Arpita Maiti, PhD Senior Director, External Science & Innovation Pfizer Inc. June 29, 2017 Boston Forward-looking
Promise and Pitfalls of MicrobiomeModulating Methods for Inflammatory and Autoimmune Diseases Arpita Maiti, PhD Senior Director, External Science & Innovation Pfizer Inc. June 29, 2017 Boston Forward-looking
Patents & Health. Dr. Brian W Tempest.
 Patents & Health Dr. Brian W Tempest www.briantempest.com Seminar on the Relationship between Patent Systems and the Availability of Medicines in Developing Countries and Least Developed Countries World
Patents & Health Dr. Brian W Tempest www.briantempest.com Seminar on the Relationship between Patent Systems and the Availability of Medicines in Developing Countries and Least Developed Countries World
The role of the Hospital Pharmacist: Availability of medicines above price-only procurement
 The role of the Hospital Pharmacist: Availability of medicines above price-only procurement Thijs J. Giezen, PhD, PharmD, MSc Hospital Pharmacist, Foundation Pharmacy for Hospitals in Haarlem, Haarlem,
The role of the Hospital Pharmacist: Availability of medicines above price-only procurement Thijs J. Giezen, PhD, PharmD, MSc Hospital Pharmacist, Foundation Pharmacy for Hospitals in Haarlem, Haarlem,
SafeSciMET. European Modular Education and Training Programme in Safety Sciences for Medicines
 SafeSciMET European Modular Education and Training Programme in Safety Sciences for Medicines What patients need What Europe needs Rapid exploitation of insights in disease mechanisms for introduction
SafeSciMET European Modular Education and Training Programme in Safety Sciences for Medicines What patients need What Europe needs Rapid exploitation of insights in disease mechanisms for introduction
intelligent use of health information to individualize and integrate health care Hopkins inhealth
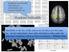 intelligent use of health information to individualize and integrate health care Hopkins inhealth Variability is the law of life, and as no two faces are the same, so no two individuals react alike and
intelligent use of health information to individualize and integrate health care Hopkins inhealth Variability is the law of life, and as no two faces are the same, so no two individuals react alike and
Competition in the off-patent biological market:
 November 14th, 2018 ISPOR Europe, Barcelona Competition in the off-patent biological market: Policies for biosimilars in Europe Evelien Moorkens PhD researcher Market access biosimilars Promotors: prof.
November 14th, 2018 ISPOR Europe, Barcelona Competition in the off-patent biological market: Policies for biosimilars in Europe Evelien Moorkens PhD researcher Market access biosimilars Promotors: prof.
Professor Tore Kristian Kvien, MD, PhD, Norway
 GaBI Scientific Meetings 10 October 2018, Le Meridien Dubai, United Arab Emirates 2nd MENA Stakeholder Meeting on Regulatory Approval, Clinical Settings, Interchangeability and Pharmacovigilance of Biosimilars
GaBI Scientific Meetings 10 October 2018, Le Meridien Dubai, United Arab Emirates 2nd MENA Stakeholder Meeting on Regulatory Approval, Clinical Settings, Interchangeability and Pharmacovigilance of Biosimilars
Datamonitor Healthcare Pharma intelligence informa Datamonitor Healthcare Content Calendar
 Datamonitor Healthcare 2017 Datamonitor Healthcare Content Calendar 2017 Content Calendar Published February 2017 Datamonitor Healthcare provides up-to-date insights and analysis that you can trust. Disease
Datamonitor Healthcare 2017 Datamonitor Healthcare Content Calendar 2017 Content Calendar Published February 2017 Datamonitor Healthcare provides up-to-date insights and analysis that you can trust. Disease
Immunogenicity: Impact on the Design of Clinical Trials for Biosimilars
 Immunogenicity: Impact on the Design of Clinical Trials for Biosimilars Alexander Berghout, M.D., Ph.D. Head Global Clinical Research and Development Sandoz Biopharmaceuticals BMWP/BWP Workshop on Immunogenicity
Immunogenicity: Impact on the Design of Clinical Trials for Biosimilars Alexander Berghout, M.D., Ph.D. Head Global Clinical Research and Development Sandoz Biopharmaceuticals BMWP/BWP Workshop on Immunogenicity
Wassenaar Arrangement. What is it all about? India s entry: What it means for us? What critics have to say?
 Wassenaar Arrangement What is it all about? India s entry: What it means for us? What critics have to say? Roots Cold War-era Coordinating Committee for Multilateral Export Controls (COCOM) COCOM existed
Wassenaar Arrangement What is it all about? India s entry: What it means for us? What critics have to say? Roots Cold War-era Coordinating Committee for Multilateral Export Controls (COCOM) COCOM existed
Moving from volume to value in the generic business model
 Moving from volume to value in the generic business model CPhI Pre-connect conference Frankfurt 23rd October 2017 Copyright 2016 QuintilesIMS. All rights reserved. Key drivers to 2021 Global growth moderating
Moving from volume to value in the generic business model CPhI Pre-connect conference Frankfurt 23rd October 2017 Copyright 2016 QuintilesIMS. All rights reserved. Key drivers to 2021 Global growth moderating
Inflation Near 10% Drug Price Growth Nearly Flat at 0.2%
 Drug Trend Report 2017 Despite AWP Inflation Near 10% CVS Health Kept Drug Price Growth Nearly Flat at 0.2% and Adherence Improved Drug Trend Report 2017 In 2017, despite AWP inflation ~10% our strategies
Drug Trend Report 2017 Despite AWP Inflation Near 10% CVS Health Kept Drug Price Growth Nearly Flat at 0.2% and Adherence Improved Drug Trend Report 2017 In 2017, despite AWP inflation ~10% our strategies
THE CORPORATE REPUTATION OF PHARMA THE PATIENT PERSPECTIVE IN 2015 (NORDIC EDITION)
 THE CORPORATE REPUTATION OF PHARMA THE PATIENT PERSPECTIVE IN 2015 (NORDIC EDITION) Feedback from 147 Nordic patient groups PUBLISHED JUNE 2016 Syöpälääkkeiden kehitys on ollut hyvä, ja yhä useammat paranevat.
THE CORPORATE REPUTATION OF PHARMA THE PATIENT PERSPECTIVE IN 2015 (NORDIC EDITION) Feedback from 147 Nordic patient groups PUBLISHED JUNE 2016 Syöpälääkkeiden kehitys on ollut hyvä, ja yhä useammat paranevat.
Biosimilar Medicines: Opportunities for Sustainable Healthcare. Opening Address Greg Perry Director General EGA London 3-4 May 2007
 Biosimilar Medicines: Opportunities for Sustainable Healthcare Opening Address Greg Perry Director General EGA London 3-4 May 2007 Building on The Generic Medicines Legacy 1. Generic medicines account
Biosimilar Medicines: Opportunities for Sustainable Healthcare Opening Address Greg Perry Director General EGA London 3-4 May 2007 Building on The Generic Medicines Legacy 1. Generic medicines account
5 key characteristics
 Biosimilars 5 key characteristics Biologics are medicinal products derived from living organisms. 1 Their use has impacted the treatment of a variety of diseases. 1 Patents for many biologics will soon
Biosimilars 5 key characteristics Biologics are medicinal products derived from living organisms. 1 Their use has impacted the treatment of a variety of diseases. 1 Patents for many biologics will soon
Solution Partner Program Global Perspective
 Solution Partner Program Global Perspective SPACe 2012 Siemens Process Automation Conference Business Development Solution Partner Program Copyright Siemens AG 2012. All rights reserved Solution Partner
Solution Partner Program Global Perspective SPACe 2012 Siemens Process Automation Conference Business Development Solution Partner Program Copyright Siemens AG 2012. All rights reserved Solution Partner
An update on development strategies of recently approved biosimilars in Europe. Johanna Mielke
 An update on development strategies of recently approved biosimilars in Europe Johanna Mielke Acknowledgement & disclaimer This project was supported by the Swiss State Secretariat for Education, Research
An update on development strategies of recently approved biosimilars in Europe Johanna Mielke Acknowledgement & disclaimer This project was supported by the Swiss State Secretariat for Education, Research
What is a biosimilar and how does sound biosimilar use look like?
 Vs14f17 LBP Opleidingsinstituut voor Care & Cure: Biosimilars in the Market; what does it mean for us? Rotterdam, June 18, 2014. What is a biosimilar and how does sound biosimilar use look like? Arnold
Vs14f17 LBP Opleidingsinstituut voor Care & Cure: Biosimilars in the Market; what does it mean for us? Rotterdam, June 18, 2014. What is a biosimilar and how does sound biosimilar use look like? Arnold
CXP GROUP. SITSI - your source for exceptional analyst insight
 SITSI - your source for exceptional analyst insight SITSI 2018 The most comprehensive research platform on software and IT services since 1992. active users annual downloads SITSI statistics 3k 65k 7k
SITSI - your source for exceptional analyst insight SITSI 2018 The most comprehensive research platform on software and IT services since 1992. active users annual downloads SITSI statistics 3k 65k 7k
Pharmaceutical Pricing Regulation and Central Health System Structures
 Pharmaceutical Pricing Regulation and Central Health System Structures Panos Kanavos London School of Economics NPPA Seminar, New Delhi, 12 April 2008 Pricing of Medicines: The Context Supply-side Regulatory
Pharmaceutical Pricing Regulation and Central Health System Structures Panos Kanavos London School of Economics NPPA Seminar, New Delhi, 12 April 2008 Pricing of Medicines: The Context Supply-side Regulatory
Innovation and Sustainability: An integrated approach
 Innovation and Sustainability: An integrated approach Karl Mahler Head of Investor Relations Dianne Young Sustainability Communications Swiss Sustainability Leaders SRI Conference Zurich, 4 October 2013
Innovation and Sustainability: An integrated approach Karl Mahler Head of Investor Relations Dianne Young Sustainability Communications Swiss Sustainability Leaders SRI Conference Zurich, 4 October 2013
HTAs and EMA working together: 23 parallel scientific advice procedures later - what have we learned?
 HTAs and EMA working together: 23 parallel scientific advice procedures later - what have we learned? DIA 26th Annual EuroMeeting, Vienna 2014 Jan Regnstrom, MD, PhD Senior Scientific Officer An agency
HTAs and EMA working together: 23 parallel scientific advice procedures later - what have we learned? DIA 26th Annual EuroMeeting, Vienna 2014 Jan Regnstrom, MD, PhD Senior Scientific Officer An agency
SSPPS STRATEGIC PLAN. Pharmacy Education, Pharmaceutical Sciences Research, Clinical Pharmacy Practice. April 30, Overview
 SSPPS STRATEGIC PLAN Pharmacy Education, Pharmaceutical Sciences Research, Clinical Pharmacy Practice April 30, 2012 Overview Here we present the 2012-2017 strategic plan for the Skaggs School of Pharmacy
SSPPS STRATEGIC PLAN Pharmacy Education, Pharmaceutical Sciences Research, Clinical Pharmacy Practice April 30, 2012 Overview Here we present the 2012-2017 strategic plan for the Skaggs School of Pharmacy
Biosimilar mabs Clinical issues Regulatory perspective
 Biosimilar mabs Clinical issues Regulatory perspective EMEA Workshop on Biosimilar Monoclonal Antibodies, MD BMWP Chairman European Medicines Agency (EMEA), UK Paul-Ehrlich-Institut, Germany Mechanisms
Biosimilar mabs Clinical issues Regulatory perspective EMEA Workshop on Biosimilar Monoclonal Antibodies, MD BMWP Chairman European Medicines Agency (EMEA), UK Paul-Ehrlich-Institut, Germany Mechanisms
INVESTOR PRESENTATION 2018
 INVESTOR PRESENTATION 2018 IMMUNOPRECISE ANTIBODIES, LTD. UNIT 3204-4464 MARKHAM STREET VICTORIA, BRITISH COLUMBIA V8Z 7X8, CANADA IMMUNOPRECISE.COM 1 Disclosures DISCLAIMER This presentation is not, and
INVESTOR PRESENTATION 2018 IMMUNOPRECISE ANTIBODIES, LTD. UNIT 3204-4464 MARKHAM STREET VICTORIA, BRITISH COLUMBIA V8Z 7X8, CANADA IMMUNOPRECISE.COM 1 Disclosures DISCLAIMER This presentation is not, and
Inflammatory Bowel Disease
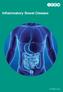 Inflammatory Bowel Disease ICONplc.com Inflammatory bowel disease (IBD) is a chronic, relapsing, inflammatory disorder of the gastrointestinal tract and includes Ulcerative Colitis (UC) and Crohn s Disease
Inflammatory Bowel Disease ICONplc.com Inflammatory bowel disease (IBD) is a chronic, relapsing, inflammatory disorder of the gastrointestinal tract and includes Ulcerative Colitis (UC) and Crohn s Disease
The impact of Quality by Design on Modern Manufacturing. Dr Tom Sam Secretary FIP s Industrial Pharmacy Section
 The impact of Quality by Design on Modern Manufacturing Dr Tom Sam Secretary FIP s Industrial Pharmacy Section FIP - Who we are The International Pharmaceutical Federation (FIP) is the global Federation
The impact of Quality by Design on Modern Manufacturing Dr Tom Sam Secretary FIP s Industrial Pharmacy Section FIP - Who we are The International Pharmaceutical Federation (FIP) is the global Federation
THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 199 PATIENT GROUPS with an interest in RARE DISEASES
 THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 199 PATIENT GROUPS with an interest in RARE DISEASES PUBLISHED MAY 2016 Dans notre domaine de maladies rares où des efforts ont été faits pour
THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 199 PATIENT GROUPS with an interest in RARE DISEASES PUBLISHED MAY 2016 Dans notre domaine de maladies rares où des efforts ont été faits pour
Strategic vision of Pharma Market
 Strategic vision of Pharma Market Evolution 2012-2016 ACIP Presentation, Paris January 30, 2013 Introduction This presentation draws strategic orientations for pharma companies considering the expected
Strategic vision of Pharma Market Evolution 2012-2016 ACIP Presentation, Paris January 30, 2013 Introduction This presentation draws strategic orientations for pharma companies considering the expected
Medical Pharmacy Trend Report: Managing the Trends and Complexity of Provider- Administered Drugs
 Medical Pharmacy Trend Report: Managing the Trends and Complexity of Provider- Administered Drugs Thursday, March 10, 2016 1:00 2:30 EST 12:00 1:30 CST 11:00 12:30 MST 10:00 11:30 PST Casandra Stockman,
Medical Pharmacy Trend Report: Managing the Trends and Complexity of Provider- Administered Drugs Thursday, March 10, 2016 1:00 2:30 EST 12:00 1:30 CST 11:00 12:30 MST 10:00 11:30 PST Casandra Stockman,
Adis Journals and Newsletters The premier collection of drug-focused medical journals
 adis.com Adis Journals and Newsletters The premier collection of drug-focused medical journals An invaluable resource for all involved in medical research, practice or teaching, drug regulation or reimbursement,
adis.com Adis Journals and Newsletters The premier collection of drug-focused medical journals An invaluable resource for all involved in medical research, practice or teaching, drug regulation or reimbursement,
Cancer survival trends and inequalities: what is the role for Europe?
 Cancer survival trends and inequalities: what is the role for Europe? European Joint Action on Comprehensive Cancer Control 2014-2017 Brussels, Belgium, 13 May 2015 Measures of cancer burden definition
Cancer survival trends and inequalities: what is the role for Europe? European Joint Action on Comprehensive Cancer Control 2014-2017 Brussels, Belgium, 13 May 2015 Measures of cancer burden definition
Market Analysis Pharmacies in Europe & Worldwide
 Interconnection Vienna I Bratislava I Lviv I Mexico City I Oberstdorf www.interconnectionconsulting.com we show you the way Market Analysis Pharmacies in Europe & Worldwide Offer 2016 Benefits Your Benefits
Interconnection Vienna I Bratislava I Lviv I Mexico City I Oberstdorf www.interconnectionconsulting.com we show you the way Market Analysis Pharmacies in Europe & Worldwide Offer 2016 Benefits Your Benefits
Anti-TNF s: Still the Best First Line Therapy for IBD Treatment?
 Anti-TNF s: Still the Best First Line Therapy for IBD Treatment? Pfizer Symposium - Reaching New Horizons with Biosimilars in IBD Karen I. Kroeker May 26, 2017 Disclosure of Commercial Support This program
Anti-TNF s: Still the Best First Line Therapy for IBD Treatment? Pfizer Symposium - Reaching New Horizons with Biosimilars in IBD Karen I. Kroeker May 26, 2017 Disclosure of Commercial Support This program
Biosimilars Scientific and Regulatory Considerations
 Biosimilars Scientific and Regulatory Considerations Gustavo Grampp Regulatory Policy Director, Amgen Presented at Maryland Pharmacists Association meeting January 31, 2016 Program objectives 1. Review
Biosimilars Scientific and Regulatory Considerations Gustavo Grampp Regulatory Policy Director, Amgen Presented at Maryland Pharmacists Association meeting January 31, 2016 Program objectives 1. Review
Genentech Contacts: Media: Tara Cooper (650) Investor: Kathee Littrell (650)
 NEWS RELEASE Genentech Contacts: Media: Tara Cooper (650) 225-5505 Investor: Kathee Littrell (650) 225-1034 XOMA Media/Investor Contacts: Laura Zobkiw (510) 204-7273 Peter Davis (510) 204-7231 FDA APPROVES
NEWS RELEASE Genentech Contacts: Media: Tara Cooper (650) 225-5505 Investor: Kathee Littrell (650) 225-1034 XOMA Media/Investor Contacts: Laura Zobkiw (510) 204-7273 Peter Davis (510) 204-7231 FDA APPROVES
BIOPHARMACEUTICAL BUSINESSES
 BIOPHARMACEUTICAL BUSINESSES Our Biopharmaceuticals businesses consist of four patientcentric, customer-facing business units. The business units are responsible for further clinical development and lifecycle
BIOPHARMACEUTICAL BUSINESSES Our Biopharmaceuticals businesses consist of four patientcentric, customer-facing business units. The business units are responsible for further clinical development and lifecycle
News For Immediate Release
 News For Immediate Release Contact: Gary Gatyas IMS Institute for Healthcare Informatics (610) 244-2600 ggatyas@us.imshealth.com IMS Study Forecasts Rebound in Global Spending on Medicines, Reaching Nearly
News For Immediate Release Contact: Gary Gatyas IMS Institute for Healthcare Informatics (610) 244-2600 ggatyas@us.imshealth.com IMS Study Forecasts Rebound in Global Spending on Medicines, Reaching Nearly
BIOSIMILARS OVERVIEW..
 Overview .... BIOSIMILARS OVERVIEW.. 7#%,8.895.3$%&$'$()*&.:%*,'... 2.. 3$%&$'$()*&.;
Overview .... BIOSIMILARS OVERVIEW.. 7#%,8.895.3$%&$'$()*&.:%*,'... 2.. 3$%&$'$()*&.;
Technology appraisal guidance Published: 28 September 2016 nice.org.uk/guidance/ta407
 Secukinumab for active ankylosing spondylitis after treatment with non- steroidal anti-inflammatory drugs or TNF-alpha inhibitors Technology appraisal guidance Published: 28 September 16 nice.org.uk/guidance/ta407
Secukinumab for active ankylosing spondylitis after treatment with non- steroidal anti-inflammatory drugs or TNF-alpha inhibitors Technology appraisal guidance Published: 28 September 16 nice.org.uk/guidance/ta407
From Bench To Clinic : a personal journey Career Research Advancement Focused Training
 From Bench To Clinic : a personal journey Career Research Advancement Focused Training February 13, 2018 Irina Tcherepanova Ph.D. Vice President of Translational Medicine Argos Therapeutics Inc. FDA view
From Bench To Clinic : a personal journey Career Research Advancement Focused Training February 13, 2018 Irina Tcherepanova Ph.D. Vice President of Translational Medicine Argos Therapeutics Inc. FDA view
Medical Affairs Not a Sunshine Day Analyzing the Impact of the Sunshine Act on Medical Affairs: Challenges for 2014 and Beyond
 Medical Affairs Not a Sunshine Day Analyzing the Impact of the Sunshine Act on Medical Affairs: Challenges for 2014 and Beyond Jodi Smith, PhD and Peg Crowley Nowick, PhD, MBA Medical Affairs Not a Sunshine
Medical Affairs Not a Sunshine Day Analyzing the Impact of the Sunshine Act on Medical Affairs: Challenges for 2014 and Beyond Jodi Smith, PhD and Peg Crowley Nowick, PhD, MBA Medical Affairs Not a Sunshine
Walgreens Rx Supply Chain Transforming to an Outsource Model
 Walgreens Rx Supply Chain Transforming to an Outsource Model Michael Bleser Divisional Vice President, Rx Supply Chain & Analytics April 2, 2014 2013 Walgreen Co. All rights reserved. Background BS-Finance
Walgreens Rx Supply Chain Transforming to an Outsource Model Michael Bleser Divisional Vice President, Rx Supply Chain & Analytics April 2, 2014 2013 Walgreen Co. All rights reserved. Background BS-Finance
