Introduction RESEARCH ARTICLE. Michael E. Woodman, Anne E. Cooley & Brian Stevenson
|
|
|
- Adrian Wiggins
- 6 years ago
- Views:
Transcription
1 RESEARCH ARTICLE Production of outer surface protein A by Borrelia burgdorferi during transmission from infected mammals to feeding ticks is insu cient to trigger OspA seroconversion Michael E. Woodman, Anne E. Cooley & Brian Stevenson Department of Microbiology, Immunology, and Molecular Genetics, University of Kentucky College of Medicine, Lexington, KY, USA Correspondence: Brian Stevenson, Department of Microbiology, Immunology, and Molecular Genetics, University of Kentucky College of Medicine, MS421 W.R. Willard Medical Education Building, Lexington, KY , USA. Tel.: ; fax: ; brian.stevenson@uky.edu Received 23 April 2008; revised 31 July 2008; accepted 7 August First published online 11 September DOI: /j X x Editor: Patrick Brennan Keywords Lyme disease; OspA; tick. Abstract The Lyme disease spirochete, Borrelia burgdorferi, produces two outer surface lipoproteins, OspA and OspB, that are essential for colonization of tick vectors. Both proteins are highly expressed during transmission from infected mammals to feeding ticks and during colonization of tick midguts, but are repressed when bacteria are transmitted from ticks to mammals. Humans and other infected mammals generally do not produce antibodies against either protein, although some Lyme disease patients do seroconvert and produce antibodies against OspA for unknown reasons. We hypothesized that, if such patients had been fed upon by additional ticks, bacteria moving from the patients bodies to the feeding ticks would have produced OspA and OspB proteins, which then led to immune system recognition and antibody production. This hypothesis was tested by analyzing immune responses of infected mice following feedings by additional Ixodes scapularis ticks. However, results of the present studies demonstrate that expression of OspA and OspB by B. burgdorferi during transmission from infected mammals to feeding ticks does not trigger seroconversion. Introduction The causative agent of Lyme disease, Borrelia burgdorferi, is maintained in nature through an infectious cycle between vertebrate reservoir hosts and infected Ixodes spp. ticks. Borrelia burgdorferi regulates the expression of a number of proteins important for each stage of the life cycle in order to persist in these drastically different environments. Two of the most intensively studied proteins of B. burgdorferi are outer surface proteins A and B (OspA and OspB), encoded by the ospab operon (Howe et al., 1986). These surfaceexposed lipoproteins are abundantly expressed by bacteria in the tick midgut. However, production is repressed during tick feeding as bacteria migrate to the ticks salivary glands and are transmitted to the vertebrate host, and very few bacteria produce either protein at the time of deposition in the host (Schwan et al., 1995; de Silva et al., 1996; Schwan & Piesman, 2000). Early inhibition of OspA and OspB is essential for vertebrate infection, as B. burgdorferi mutants that constitutively express OspA and Osp B are rapidly killed by host immune responses (Strother et al., 2007). In contrast, bacteria deficient in OspA or OspB appear to be fully infectious to mice (Yang et al., 2004). Both OspA and OspB are necessary for colonization of the tick midgut (Pal et al., 2000, 2004). OspA can bind a glycoprotein named TROSPA (tick receptor for OspA) found on the lining of the tick midgut (Pal et al., 2000, 2004). Together, these data suggest roles for OspA and OspB only during the tick portion of the B. burgdorferi infection cycle. Consistent with this hypothesis, laboratory animals experimentally infected via tick bite do not produce antibodies against either OspA or OspB (Roehrig et al., 1992; Golde et al., 1994; Brunet et al., 1995; Nowalk et al., 2006 and this work). Wild mice also do not appear to produce OspA- or OspB-directed antibodies (Bunikis et al., 2004). Similarly, the majority of human Lyme disease patients do not produce OspA- and OspB-directed antibodies. However, some humans with prolonged B. burgdorferi infections do seroconvert to OspA and/or OspB (Kalish et al., 1993; Batsford et al., 1998; Akin et al., 1999; Nowalk et al., 2006). Development of OspA-directed antibodies has been associated with severe arthritis and neurological manifestations,
2 278 M.E. Woodman et al. possibly due to autoimmune reactions (Gross et al., 1998; Akin et al., 1999; Alaedini & Latov, 2005). The reason some patients produce OspA- or OspB-directed antibodies during chronic Lyme disease is unknown. The present study was designed to address this apparent contradiction: if OspA and OspB are tick-specific proteins, how is it possible for infected patients to seroconvert? A possible explanation for OspA and OspB seroconversion was hinted at by studies that investigated acquisition of B. burgdorferi by ticks feeding on infected mammals. Such bacteria produce high levels of both OspA and OspB during the time they leave mammalian bodies and enter feeding ticks (Schwan et al., 1995; de Silva et al., 1996; Schwan & Piesman, 2000). We hypothesized that if infected humans or other mammals were fed upon by additional ticks, B. burgdorferi that produce OspA and OspB as the bacteria move from the host into the feeding ticks could be detected by the patient s immune system, thereby triggering seroconversion. To address this hypothesis, mice were infected by tick bite, and then 1 month later, were fed upon a second time by uninfected ticks. One month after that reinfestation, the mice were examined for changes in serum antibody content, including antibodies recognizing OspA and OspB. Materials and methods Bacteria and cultivation The infectious, clonal B. burgdorferi strain B31-MI-16 (Miller et al., 2003) was utilized for all infection and serological studies. Bacteria were cultured in Barbour Stoenner Kelly II (BSK-II) medium (Barbour, 1984) at 34 1C. Tick and mouse infections Ixodes scapularis egg masses were obtained from the Department of Entomology at Oklahoma State University (Stillwater, OK), and held in a humidified chamber. After hatching, cohorts of 200 larval ticks were each fed on female BALB/c mice (Harlan Sprague Dawley, Indianapolis, IN) that had been previously infected with B. burgdorferi strain B31-MI-16 (Miller et al., 2003). Larval ticks were allowed to feed to repletion and naturally detach from the mice. Fed larvae were returned to the humidified chamber to molt into nymphs. After ecdysis, 20 nymphs each were fed to repletion on 12 naïve BALB/c mice. Over 50% of each mouse s nymphs completed blood feeding and dropped off naturally. Seven days later, a 1 mm 2 ear biopsy from each mouse was individually inoculated into BSK-II medium. Antibiotics that do not affect B. burgdorferi (phosphomycin and rifampicin) and antifungal agents (amphotericin B) (Sigma, St Louis, MO) were included to reduce contamination. Mouse infections were confirmed 10 days later by examination of each biopsy culture using dark-field microscopy. One month postinfection, six of the nymph-infected mice were reinfested with uninfected larval ticks (200 ticks per mouse). This number of animals per group is sufficient for statistically significant analysis of infection parameters (Richardson & Overbaugh, 2005). Greater than 50% of the larvae placed on each mouse fed to repletion and detached naturally. Cohorts of engorged ticks from each mouse were dissected immediately after detachment for immunofluorescence analysis. Serum samples were collected from all 12 BALB/c mice before the initial nymph feeding. Samples were also collected from all 12 mice 1 month postinfection (immediately before the feeding of larvae on half the mice), and then again after 2 months of infection. Tick dissection and indirect immunofluorescence analysis Following reinfestations, midguts from two engorged ticks per mouse were dissected into 10 ml phosphate-buffered saline (PBS) on glass slides and allowed to air dry overnight. Slides were then fixed and permeabilized in acetone for 15 min and allowed to air dry. Slides were blocked overnight in PBS containing 0.2% bovine serum albumin (BSA) at 4 1C. Slides were then incubated with the monoclonal antibodies H5332, reactive with OspA (Barbour et al., 1983), or H4610, reactive with OspB (Rosa et al., 1992), for 1 h at room temperature. Slides were washed in PBS 0.2% BSA and incubated for 1 h at room temperature with rabbit polyclonal antiserum that was raised against B. burgdorferi total membrane proteins (Miller et al., 2003). Slides were washed and incubated simultaneously with 1 : 1000 dilutions of Alexa-Fluor 488-labeled goat anti-mouse IgG and Alexa-Fluor 594-labeled goat anti-rabbit IgG (Molecular Probes, Eugene, OR) for 45 min at room temperature. Slides were then washed, dried, and mounted in ProLong Anti- Fade mounting medium (Molecular Probes). Slides were analyzed with a 40 objective lens using a BX51 epifluorescence microscope (Olympus, Melville, NY) and a Retiga 200R Fast 1394 imaging system (Q-Imaging, Burnaby, BC, Canada). At least 25 fields were examined per dissected tick, with at least one spirochete visible per field. Recombinant OspA and OspB Borrelia burgdorferi strain B31-MI-16 purified DNA was used as the template for PCR amplification of ospa and ospb. The oligonucleotide primers MW-OspA F (5 0 -CAC CAA GCA AAA TGT TAG CAG CCT TGA CGA-3 0 ) and MW-OspA R (5 0 -TTA TTA TTT TAA AGC GTT TTT AAT TTC ATC AAG-3 0 ) were used for amplification of ospa, and MW-OspB F (5 0 -CAC CGC ACA AAA AGG TGC TGA G-3 0 ) and MW-OspB R (5 0 -TTA TTA AAG CGT TTT TAA GCT CTG-3 0 ) were used to amplify ospb. Each PCR product was separately cloned into pet200 Champion TOPO
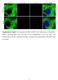
3 Borrelia burgdorferi OspA seroconversion 279 (Invitrogen, Carlsbad, CA). The insert of one clone of each was completely sequenced to confirm that errors had not been introduced during the cloning process. Recombinant proteins were produced in Escherichia coli Rosetta 2(DE3)- plyss (Novagen, San Diego, CA) cultivated in super broth medium (32 g L 1 tryptone, 20 g L 1 yeast extract, and 5gL 1 sodium chloride). Bacteria were lysed by sonication, and recombinant OspA and OspB were purified using MagneHis Ni-particles (Promega, Madison, WI). Recombinant protein quality and purity were assessed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS- PAGE) with staining with Coomassie brilliant blue, and by immunoblotting with anti-ospa (H5332) or anti-ospb (H4610) monoclonal antibodies followed by sheep antimouse immunoglobulin horseradish peroxidase conjugate (Amersham, Piscataway, NJ) and chemiluminescence (Pierce Rockford, IL). Enzyme-linked immunosorbent analyses (ELISA) Wells of 96-well Maxisorp Nunc-Immuno plates (Nalge Nunc International, Naperville, IL) were coated overnight at 4 1C with 10mgmL 1 of either recombinant OspA or recombinant OspB in 100 ml of 50 mm carbonate buffer (1.60 g L 1 Na 2 CO 3, 2.93 g L 1 NaHCO 3 ), ph 9.6. Plates were washed in PBS 0.05% Tween 20 (PBS T) and blocked for 2 h at room temperature in PBS T containing 2% BSA. After three washes, plates were incubated for 2 h at room temperature with 100 ml of 1 : 100-diluted mouse sera in duplicate. Following three PBS T washes, wells were incubated with a 1 : 5000 dilution of sheep anti-mouse immunoglobulin horseradish peroxidase conjugate (Amersham) for 1 h at room temperature. Plates were washed again with PBS T, 100 ml of 3,3 0,5,5 0 -tetramethylbenzidine (Pierce) was then added to each well, and color was allowed to develop for 30 min at room temperature. The reaction was stopped by adding 50 ml 2NH 2 SO 4 per well. A 450 nm was then read with a VersaMax tunable microplate reader and SOFTMAX PRO software (Molecular Devices, Sunnyvale, CA). OspA and OspB ELISAs were each performed twice with each serum sample being analyzed in duplicate in each ELISA. Wells were coated with BSA to serve as a negative control. Anti-OspA (H5332) or anti-ospb (H4610) monoclonal antibodies were used as positive controls to ensure detection of recombinant OspA or OspB-coated wells, respectively. Immunoblot analysis Borrelia burgdorferi strain B31-MI-16 whole-cell lysate or purified recombinant OspA or OspB were separated by SDS- PAGE, electrotransferred to nitrocellulose membranes, and blocked with 5% nonfat dry milk in Tris-buffered saline Tween 20 (TBS T) [20 mm Tris (ph 7.5), 150 mm NaCl, 0.05% (v/v) Tween 20] overnight at 4 1C. Membranes were washed after blocking, then simultaneously incubated for 1 h at room temperature with each mouse serum sample, diluted 1 : 100 in TBS T, using a MiniProtean II Multiscreen apparatus (Bio-Rad, Hercules, CA). Membranes were also incubated with monoclonal antibodies against OspA and OspB, as controls to identify where those proteins migrated in each gel. Membranes were then washed and incubated with sheep anti-mouse immunoglobulin horseradish peroxidase conjugate (Amersham) and visualized by enhanced chemiluminescence (Pierce). Statistical analysis To determine statistical significance, mouse serum antibody titers were compared using a repeated measures model. A P-value of 0.05 was considered significant. Results Borrelia burgdorferi expressed OspA and OspB as they were acquired by larval ticks feeding on infected mice Previous studies demonstrated that B. burgdorferi produce OspA and OspB as they migrate from infected mammalian hosts into feeding ticks (Schwan et al., 1995; de Silva et al., 1996; Schwan & Piesman, 2000; Neelakanta et al., 2007). To confirm that the bacteria used in the present study also followed that pattern, mice previously infected by tick bite were reinfested with naïve, larval ticks. Prior cultivation of ear punch biopsies had demonstrated that all tested mice were infected with B. burgdorferi. Fed larvae were dissected, and their midguts were examined by immunofluorescence for the presence of B. burgdorferi and bacterial expression of both OspA and OspB. Borrelia burgdorferi were detected in the midguts of ticks that fed on all six of the reinfested, infected mice, further confirming that all the animals were indeed infected. Greater than 90% of B. burgdorferi detected in tick midguts demonstrated detectable production of OspA and OspB (Fig. 1 and data not shown), as described previously (Schwan et al., 1995; de Silva et al., 1996; Schwan & Piesman, 2000; Neelakanta et al., 2007). Serum analyses of OspA- and OspB- directed antibodies For each reinfested mouse, serum samples were collected before infection, 1 month after infection (but before the second tick feeding), and 2 months after infection (1 month after the second tick feeding). ELISA demonstrated that none of the six reinfested mice developed significantly increased levels of antibodies that recognized OspA or OspB after the second tick feeding (Fig. 2). Immunoblot analyses
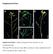
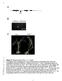
4 280 M.E. Woodman et al. Fig. 1. Representative immunofluorescence images of Borrelia burgdorferi expressing (a) OspA and (b) OspB following acquisition by larval ticks feeding on infected mice. Monoclonal antibodies against the lipoproteins labeled those spirochetes producing OspA (a-ospa) or OspB (a-ospb) (left), while rabbit polyclonal antiserum raised against B. burgdorferi total membrane proteins labeled all spirochetes in the same field (a-bb) (center). Merged images are shown on the right. The a-ospa antibody recognized slightly different regions of some spirochetes than the a-bb antibody, possibly due to antigen distribution in such bacteria. These results are comparable to previous reports (Schwan et al., 1995; de Silva et al., 1996; Schwan & Piesman, 2000). using recombinant proteins with the same serum samples similarly demonstrated no increases in antibodies recognizing either OspA or OspB (data not shown). Additionally, there were not any significant differences in antibody levels when ELISA results of mice that were reinfested with ticks were compared with animals that were not reinfested with larvae (Fig. 2). Positive controls using anti-ospa or anti- OspB monoclonal antibodies produced significant signals, with A 450 nm values between 0.8 and 1.0 (data not shown), confirming that we had used the appropriate recombinant proteins and borrelial lysates. One mouse had above-background levels of antibodies that bound OspB at all points, including before the time of B. burgdorferi infection (Fig. 2). These antibodies were not of a level sufficient for detection by immunoblot (data not shown and Fig. 3). This observation indicates that natural antibodies produced by that particular mouse coincidentally recognized OspB. Such natural antibodies that can bind OspA and/or OspB have been reported previously (Dorward et al., 1992; Belperron & Bockenstedt, 2001). Importantly, this animal did not develop increased levels of antibodies that recognized OspB at any time during infection. Also, of note, this animal never produced antibodies that bound OspA (Fig. 2). Antibody profile against B. burgdorferi of infected mice after second tick feeding In addition to examining the antibody responses to OspA and OspB, possible changes in antibody levels against other B. burgdorferi proteins were examined by the use of immunoblot analyses with B. burgdorferi whole-cell lysates. As noted above, all animals were confirmed to be infected with B. burgdorferi by outgrowth of B. burgdorferi from skin biopsies of each mouse. Robust humoral responses were evident in both sets of infected mice, demonstrated by prominent immunoblot bands that migrate at the same size as the well-characterized B. burgdorferi antigens flagellin at c. 41 kda and BmpA (P39) at c. 39 kda (Fig. 3). One animal of each set exhibited only mild antibody responses, the reasons for which are unknown, as infection of those animals was demonstrated by both biopsy cultivation and tick xenodiagnosis. Profiles of B. burgdorferi-directed serum antibodies were indistinguishable between mice that had been reinfested with tick larvae and those that had not. This suggests that other B. burgdorferi proteins produced during mammal-to-tick transmission likewise do not provoke production of host serum antibodies. Discussion We hypothesized that OspA and OspB seroconversion might be caused by production of the outer surface proteins by B. burgdorferi during transmission from infected mammals to feeding ticks. However, our testing of this hypothesis indicated that the OspA or OspB seroconversion must be due to another cause. What, then, is the trigger for OspAB seroconversion during some cases of Lyme disease? There is some evidence that OspA and OspB may be expressed at some locations in the host body, independent of tick feeding, but the reasons for that are unknown (Coyle et al., 1995; Schutzer et al., 1997; Liang et al., 2004). Host inflammatory processes during mammalian infection have been proposed to induce the production of OspA by B. burgdorferi (Crowley & Huber, 2003; Scheckelhoff et al., 2007), although the observation that most infected humans do not seroconvert indicates that the trigger(s) for OspA production must be more complex than just inflammation. We also note that severe inflammation is observed in many animal models of Lyme disease, but OspA or OspB seroconversion has not been reported in such animals. Another, not necessarily exclusionary, explanation is that human antibodies that recognize OspA or OspB are natural antibodies unrelated to infection, as was seen with one mouse in the present studies. In conclusion, even though B. burgdorferi produces both OspA and OspB during transmission from infected mammals to feeding ticks, such protein expression is not sufficient to prompt seroconversion. Continued research on the OspA and OspB proteins, and host immune system
5 Borrelia burgdorferi OspA seroconversion 281 Fig. 2. (a) OspA- and (b) OspB-directed antibody levels in mice as determined by ELISA, before infection (white bars), 1 month after infection by tick (black bars), and 1 month after reinfestation or 2 months after infection (gray bars). The mouse number is indicated along the bottom. Mice 1 6 were reinfested with naïve ticks and mice 7 12 were not reinfested. The A 450 nm values of the reinfested mice were not significantly different compared with the not reinfested mice. Mouse 9 demonstrated high levels of anti-ospb antibodies at all time points including before infection, indicative of natural antibody. None of the animals exhibited statistically significant changes in levels of serum antibodies recognizing either OspA or OspB. BSA served as a negative control to determine background detection levels. Positive controls using anti-ospa or anti-ospb monoclonal antibodies produced significant signals, with A 450 nm values of between 0.8 and 1.0 (data not shown). responses, will lead to a more complete understanding of B. burgdorferi infection processes as well as the treatment of late-stage disease manifestations of Lyme disease. Acknowledgements We thank Kathryn Tucker for her help with the statistical analysis and Tomasz Bykowski, Sean Riley, Catherine Brissette, Ashutosh Verma, and Logan Burns for their technical assistance and helpful comments on this work and manuscript. M.E.W. was supported in part by the NIH Training Grant in Microbial Pathogenesis T32-AI Fig. 3. Immunoblot analyses of individual mouse sera using Borrelia burgdorferi whole-cell lysate. The mouse number is indicated along the top, with mice 1 6 having been reinfested with naïve ticks and mice 7 12 not having been reinfested. All mice were infected with B. burgdorferi, as demonstrated by the outgrowth of bacteria from skin biopsies and by tick xenodiagnosis (see text). Although mice numbers 2 and 9 demonstrated weak antibody responses, both were confirmed as being infected, and ticks that fed on mouse 9 acquired the same number of B. burgdorferi as did ticks that fed on the other mice. The positions of molecular mass markers (in kda) are indicated on the left. References Akin E, McHugh GL, Flavell RA, Fikrig E & Steere AC (1999) The immunoglobulin (IgG) antibody response to OspA and OspB correlates with severe and prolonged Lyme arthritis and the IgG response to P35 correlates with mild and brief arthritis. Infect Immun 67: Alaedini A & Latov N (2005) Antibodies against OspA epitopes of Borrelia burgdorferi cross-react with neural tissue. J Neuroimmunol 159: Barbour AG (1984) Isolation and cultivation of Lyme disease spirochetes. Yale J Biol Med 57: Barbour AG, Tessier SL & Todd WJ (1983) Lyme disease spirochetes and ixodid tick spirochetes share a common
6 282 M.E. Woodman et al. surface antigenic determinant defined by a monoclonal antibody. Infect Immun 41: Batsford S, Rust C & Neubert U (1998) Analysis of antibody response to the outer surface protein family in Lyme borreliosis patients. J Infect Dis 178: Belperron AA & Bockenstedt LK (2001) Natural antibody affects survival of the spirochete Borrelia burgdorferi within feeding ticks. Infect Immun 69: Brunet LR, Sellitto C, Spielman A & Telford SR (1995) Antibody response of the mouse reservoir of Borrelia burgdorferi in nature. Infect Immun 63: Bunikis J, Tsao J, Luke CJ, Luna MG, Fish D & Barbour AG (2004) Borrelia burgdorferi infection in a natural population of Peromyscus leucopus mice: a longitudinal study in an area where Lyme borreliosis is highly endemic. J Infect Dis 189: Coyle PK, Schutzer SE, Deng Z, Krupp LB, Belman AL, Benach JL & Luft BJ (1995) Detection of Borrelia burgdorferi-specific antigen in antibody-negative cerebrospinal fluid in neurologic Lyme disease. Neurology 45: Crowley H & Huber BT (2003) Host-adapted Borrelia burgdorferi in mice expresses OspA during inflammation. Infect Immun 71: de Silva AM, Telford SR, Brunet LR, Barthold SW & Fikrig E (1996) Borrelia burgdorferi OspA is an arthropod-specific transmission-blocking Lyme disease vaccine. J Exp Med 183: Dorward DW, Huguenel ED, Davis G & Garon CF (1992) Interactions between extracellular Borrelia burgdorferi proteins and non-borrelia-directed immunoglobulin M antibodies. Infect Immun 60: Golde WT, Kappel KJ, Dequesne G, Feron C, Plainchamp D, Capiau C & Lobet Y (1994) Tick transmission of Borrelia burgdorferi to inbred strains of mice induces an antibody response to P39 but not to outer surface protein A. Infect Immun 62: Gross DM, Forsthuber T, Tary-Lehmann M, Etling C, Ito K, Nagy ZA, Field JA, Steere AC & Huber BT (1998) Identification of LFA-1 as a candidate autoantigen in treatment-resistant Lyme arthritis. Science 281: Howe TR, LaQuier FW & Barbour AG (1986) Organization of genes encoding two outer membrane proteins of the Lyme disease agent Borrelia burgdorferi within a single transcriptional unit. Infect Immun 54: Kalish RA, Leong JM & Steere AC (1993) Association of treatment-resistant chronic Lyme arthritis with HLA-DR4 and antibody reactivity to OspA and OspB of Borrelia burgdorferi. Infect Immun 61: Liang FT, Caimano MJ, Radolf JD & Fikrig E (2004) B. burgdorferi outer surface protein (osp) B expression independent of ospa. Microb Pathog 37: Miller JC, von Lackum K, Babb K, McAlister JD & Stevenson B (2003) Temporal analysis of Borrelia burgdorferi Erp protein expression throughout the mammal tick infectious cycle. Infect Immun 71: Neelakanta G, Li X, Pal U, Liu X, Beck DS, DePonte K, Fish D, Kantor FS & Fikrig E (2007) Outer surface protein B is critical for B. burgdorferi adherence and survival within Ixodes ticks. PLoS Pathog 3: Nowalk AJ, Gilmore RD Jr & Carroll JA (2006) Serologic proteome analysis of Borrelia burgdorferi membraneassociated proteins. Infect Immun 74: Pal U, desilva AM, Montgomery RR, Fish D, Anguita J, Anderson JF, Lobet Y & Fikrig E (2000) Attachment of Borrelia burgdorferi within Ixodes scapularis mediated by outer surface protein A. J Clin Invest 106: Pal U, Li X, Wang T et al. (2004) TROSPA, an Ixodes scapularis receptor for Borrelia burgdorferi. Cell 119: Richardson BA & Overbaugh J (2005) Basic statistical considerations in virological experiments. J Virol 79: Roehrig JT, Piesman J, Hunt AR, Keen MG, Happ CM & Johnson BJB (1992) The hamster immune response to tick-transmitted Borrelia burgdorferi differs from the response to needle-inoculated, cultured organisms. J Immunol 149: Rosa PA, Schwan T & Hogan D (1992) Recombination between genes encoding major outer surface proteins A and B of Borrelia burgdorferi. Mol Microbiol 6: Scheckelhoff MR, Telford SR, Wesley M & Hu LT (2007) Borrelia burgdorferi intercepts host hormonal signals to regulate expression of outer surface protein A. Proc Natl Acad Sci USA 104: Schutzer SE, Coyle PK, Krupp LB, Deng Z, Belman AL, Dattwyler R & Luft BJ (1997) Simultaneous expression of Borrelia OspA and OspC and IgM response in cerebrospinal fluid in early neurologic Lyme disease. J Clin Invest 100: Schwan TG & Piesman J (2000) Temporal changes in outer surface proteins A and C of the Lyme disease-associated spirochete, Borrelia burgdorferi, during the chain of infection in ticks and mice. J Clin Microbiol 38: Schwan TG, Piesman J, Golde WT, Dolan MC & Rosa PA (1995) Induction of an outer surface protein on Borrelia burgdorferi during tick feeding. Proc Natl Acad Sci USA 92: Strother KO, Hodzic E, Barthold SW & de Silva AM (2007) Infection of mice with Lyme disease spirochetes constitutively producing outer surface proteins A and B. Infect Immun 75: Yang XF, Pal U, Alani SM, Fikrig E & Norgard MV (2004) Essential role for OspA/B in the life cycle of the Lyme disease spirochete. J Exp Med 199:
Influence of Cultivation Media on Genetic Regulatory Patterns in Borrelia burgdorferi
 INFECTION AND IMMUNITY, June 2001, p. 4159 4163 Vol. 69, No. 6 0019-9567/01/$04.00 0 DOI: 10.1128/IAI.69.6.4159 4163.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Influence
INFECTION AND IMMUNITY, June 2001, p. 4159 4163 Vol. 69, No. 6 0019-9567/01/$04.00 0 DOI: 10.1128/IAI.69.6.4159 4163.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Influence
OspA Antibodies Inhibit the Acquisition of Borrelia burgdorferi by Ixodes Ticks
 INFECTION AND IMMUNITY, Aug. 1997, p. 3146 3150 Vol. 65, No. 8 0019-9567/97/$04.00 0 Copyright 1997, American Society for Microbiology OspA Antibodies Inhibit the Acquisition of Borrelia burgdorferi by
INFECTION AND IMMUNITY, Aug. 1997, p. 3146 3150 Vol. 65, No. 8 0019-9567/97/$04.00 0 Copyright 1997, American Society for Microbiology OspA Antibodies Inhibit the Acquisition of Borrelia burgdorferi by
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
1. Immunization. What is Immunization? 12/9/2016. Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Infection of Mice with Lyme Disease Spirochetes Constitutively Producing Outer Surface Proteins A and B
 INFECTION AND IMMUNITY, June 2007, p. 2786 2794 Vol. 75, No. 6 0019-9567/07/$08.00 0 doi:10.1128/iai.01307-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Infection of Mice with
INFECTION AND IMMUNITY, June 2007, p. 2786 2794 Vol. 75, No. 6 0019-9567/07/$08.00 0 doi:10.1128/iai.01307-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Infection of Mice with
PCR analysis was performed to show the presence and the integrity of the var1csa and var-
 Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits
 WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits Code N221-KIT N220-KIT Description WesternMAX Chemiluminescent AP Kit, Anti-Mouse Includes: Alkaline Phosphatase (AP) Conjugated Anti-Mouse
WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits Code N221-KIT N220-KIT Description WesternMAX Chemiluminescent AP Kit, Anti-Mouse Includes: Alkaline Phosphatase (AP) Conjugated Anti-Mouse
Lyme disease, caused by Borrelia burgdorferi, is the most
 Outer-surface protein C of the Lyme disease spirochete: A protein induced in ticks for infection of mammals Dorothee Grimm*, Kit Tilly*, Rebecca Byram*, Philip E. Stewart*, Jonathan G. Krum*, Dawn M. Bueschel*,
Outer-surface protein C of the Lyme disease spirochete: A protein induced in ticks for infection of mammals Dorothee Grimm*, Kit Tilly*, Rebecca Byram*, Philip E. Stewart*, Jonathan G. Krum*, Dawn M. Bueschel*,
Outer Surface Protein A Protects Lyme Disease Spirochetes from Acquired Host Immunity in the Tick Vector
 INFECTION AND IMMUNITY, Nov. 2008, p. 5228 5237 Vol. 76, No. 11 0019-9567/08/$08.00 0 doi:10.1128/iai.00410-08 Outer Surface Protein A Protects Lyme Disease Spirochetes from Acquired Host Immunity in the
INFECTION AND IMMUNITY, Nov. 2008, p. 5228 5237 Vol. 76, No. 11 0019-9567/08/$08.00 0 doi:10.1128/iai.00410-08 Outer Surface Protein A Protects Lyme Disease Spirochetes from Acquired Host Immunity in the
Anti-Asian Sea bass (Lates calcarifer) IgM monoclonal antibody labelled with horseradish peroxidase. Product no: C2-HRP
 Anti-Asian Sea bass (Lates calcarifer) IgM monoclonal antibody labelled with horseradish peroxidase Product no: C2-HRP Product Description This monoclonal antibody (Mab) reacts with Asian Sea bass (Lates
Anti-Asian Sea bass (Lates calcarifer) IgM monoclonal antibody labelled with horseradish peroxidase Product no: C2-HRP Product Description This monoclonal antibody (Mab) reacts with Asian Sea bass (Lates
Anti-Cobia (Rachycentron canadum) monoclonal antibody. Product no: F18
 Anti-Cobia (Rachycentron canadum) monoclonal antibody Product no: F18 Product Description This monoclonal antibody (Mab) reacts with Cobia (Rachycentron canadum) immunoglobulin M (IgM). The Mab is of an
Anti-Cobia (Rachycentron canadum) monoclonal antibody Product no: F18 Product Description This monoclonal antibody (Mab) reacts with Cobia (Rachycentron canadum) immunoglobulin M (IgM). The Mab is of an
Anti-Bluefin Tuna (Thunnus thynnus) monoclonal antibody. Product no: F19
 Anti-Bluefin Tuna (Thunnus thynnus) monoclonal antibody Product no: F19 Product Description This monoclonal antibody (Mab) reacts with Bluefin Tuna (Thunnus thynnus) immunoglobulin M (IgM). The Mab is
Anti-Bluefin Tuna (Thunnus thynnus) monoclonal antibody Product no: F19 Product Description This monoclonal antibody (Mab) reacts with Bluefin Tuna (Thunnus thynnus) immunoglobulin M (IgM). The Mab is
Evasion of protective immunity by Borrelia burgdorferi by truncation of outer surface protein B
 Proc. Natl. Acad. Sci. USA Vol. 90, pp. 4092-4096, May 1993 Immunology Evasion of protective immunity by Borrelia burgdorferi by truncation of outer surface protein B EROL FIKRIG*t, HONG TAO*, FRED S.
Proc. Natl. Acad. Sci. USA Vol. 90, pp. 4092-4096, May 1993 Immunology Evasion of protective immunity by Borrelia burgdorferi by truncation of outer surface protein B EROL FIKRIG*t, HONG TAO*, FRED S.
Supplemental Information
 Supplemental Information Intrinsic protein-protein interaction mediated and chaperonin assisted sequential assembly of a stable Bardet Biedl syndome protein complex, the BBSome * Qihong Zhang 1#, Dahai
Supplemental Information Intrinsic protein-protein interaction mediated and chaperonin assisted sequential assembly of a stable Bardet Biedl syndome protein complex, the BBSome * Qihong Zhang 1#, Dahai
Practical Applications of Immunology (Chapter 18) Lecture Materials for Amy Warenda Czura, Ph.D. Suffolk County Community College Eastern Campus
 Practical Applications of Immunology (Chapter 18) Lecture Materials for Amy Warenda Czura, Ph.D. Suffolk County Community College Eastern Campus Primary Source for figures and content: Tortora, G.J. Microbiology
Practical Applications of Immunology (Chapter 18) Lecture Materials for Amy Warenda Czura, Ph.D. Suffolk County Community College Eastern Campus Primary Source for figures and content: Tortora, G.J. Microbiology
Segments of the obstructed intestinal loops were fixed in 4% paraformaldehyde
 Supplementary text Supplementary materials and methods Histopathological examination Segments of the obstructed intestinal loops were fixed in 4% paraformaldehyde (PFA) and embedded in paraffin wax with
Supplementary text Supplementary materials and methods Histopathological examination Segments of the obstructed intestinal loops were fixed in 4% paraformaldehyde (PFA) and embedded in paraffin wax with
Anti-Tilapia (Oreochromis niloticus) IgM monoclonal antibody. Product no: F04
 Anti-Tilapia (Oreochromis niloticus) IgM monoclonal antibody Product no: F04 Product Description This monoclonal antibody (Mab) reacts with Anti-Tilapia (Oreochromis niloticus) immunoglobulin M (IgM).
Anti-Tilapia (Oreochromis niloticus) IgM monoclonal antibody Product no: F04 Product Description This monoclonal antibody (Mab) reacts with Anti-Tilapia (Oreochromis niloticus) immunoglobulin M (IgM).
Received 11 January 1996/Returned for modification 7 March 1996/Accepted 3 April 1996
 INFECTION AND IMMUNITY, June 1996, p. 2234 2239 Vol. 64, No. 6 0019-9567/96/$04.00 0 Copyright 1996, American Society for Microbiology Outer Surface Protein C (OspC), but Not P39, Is a Protective Immunogen
INFECTION AND IMMUNITY, June 1996, p. 2234 2239 Vol. 64, No. 6 0019-9567/96/$04.00 0 Copyright 1996, American Society for Microbiology Outer Surface Protein C (OspC), but Not P39, Is a Protective Immunogen
The Expression of Recombinant Sheep Prion Protein (RecShPrPC) and its Detection Using Western Blot and Immuno-PCR
 The Expression of Recombinant Sheep Prion Protein (RecShPrPC) and its Detection Using Western Blot and Immuno-PCR S. Thomas, C. S. Fernando, J. Roach, U. DeSilva and C. A. Mireles DeWitt The objective
The Expression of Recombinant Sheep Prion Protein (RecShPrPC) and its Detection Using Western Blot and Immuno-PCR S. Thomas, C. S. Fernando, J. Roach, U. DeSilva and C. A. Mireles DeWitt The objective
A guide to selecting control, diluent and blocking reagents
 Specializing in Secondary Antibodies and Conjugates A guide to selecting control, diluent and blocking reagents Optimize your experimental protocols with Jackson ImmunoResearch Secondary antibodies and
Specializing in Secondary Antibodies and Conjugates A guide to selecting control, diluent and blocking reagents Optimize your experimental protocols with Jackson ImmunoResearch Secondary antibodies and
A guide to selecting control, diluent and blocking reagents
 Specializing in Secondary Antibodies and Conjugates A guide to selecting control, diluent and blocking reagents Optimize your experimental protocols with Jackson ImmunoResearch Secondary antibodies and
Specializing in Secondary Antibodies and Conjugates A guide to selecting control, diluent and blocking reagents Optimize your experimental protocols with Jackson ImmunoResearch Secondary antibodies and
TGFβ/TGFβR3 Signaling Pathways in Autoimmune Disease
 Holifield 1 TGFβ/TGFβR3 Signaling Pathways in Autoimmune Disease Reggie Holifield Faculty mentor: Dr. Christine Sestero The University of Montevallo Dept. of Biology, Chemistry, and Mathematics Abstract
Holifield 1 TGFβ/TGFβR3 Signaling Pathways in Autoimmune Disease Reggie Holifield Faculty mentor: Dr. Christine Sestero The University of Montevallo Dept. of Biology, Chemistry, and Mathematics Abstract
Immunofluorescence microscopy for Lyme borreliosis
 Vector-Borne-Infection Research, Analysis, Strategy Immunofluorescence microscopy for Lyme borreliosis At present, UK NHS testing fails to detect Lyme in an unknown number of patients which could amount
Vector-Borne-Infection Research, Analysis, Strategy Immunofluorescence microscopy for Lyme borreliosis At present, UK NHS testing fails to detect Lyme in an unknown number of patients which could amount
Mouse Monoclonal Antibody Isotyping Reagents
 Mouse Monoclonal Antibody Isotyping Reagents Catalog Number: SEK003 Storage Temperature: 2-8 C Fax : +86-10-58628220 Tel : +86-400-890-9989 http://www.sinobiological.com Description Mouse Monoclonal Antibody
Mouse Monoclonal Antibody Isotyping Reagents Catalog Number: SEK003 Storage Temperature: 2-8 C Fax : +86-10-58628220 Tel : +86-400-890-9989 http://www.sinobiological.com Description Mouse Monoclonal Antibody
Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR
 Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
Supplemental material
 Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Temporal Analysis of Borrelia burgdorferi Erp Protein Expression throughout the Mammal-Tick Infectious Cycle
 INFECTION AND IMMUNITY, Dec. 2003, p. 6943 6952 Vol. 71, No. 12 0019-9567/03/$08.00 0 DOI: 10.1128/IAI.71.12.6943 6952.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Temporal
INFECTION AND IMMUNITY, Dec. 2003, p. 6943 6952 Vol. 71, No. 12 0019-9567/03/$08.00 0 DOI: 10.1128/IAI.71.12.6943 6952.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Temporal
Temporal Analysis of Borrelia burgdorferi Erp Protein Expression throughout the Mammal-Tick Infectious Cycle
 INFECTION AND IMMUNITY, Dec. 2003, p. 6943 6952 Vol. 71, No. 12 0019-9567/03/$08.00 0 DOI: 10.1128/IAI.71.12.6943 6952.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Temporal
INFECTION AND IMMUNITY, Dec. 2003, p. 6943 6952 Vol. 71, No. 12 0019-9567/03/$08.00 0 DOI: 10.1128/IAI.71.12.6943 6952.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Temporal
Effect of Immunization with Recombinant OspA on Serologic Tests for Lyme Borreliosis
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Jan. 2001, p. 79 84 Vol. 8, No. 1 1071-412X/01/$04.00 0 DOI: 10.1128/CDLI.8.1.79 84.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved.
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Jan. 2001, p. 79 84 Vol. 8, No. 1 1071-412X/01/$04.00 0 DOI: 10.1128/CDLI.8.1.79 84.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved.
Supplementary. Table 1: Oligonucleotides and Plasmids. complementary to positions from 77 of the SRα '- GCT CTA GAG AAC TTG AAG TAC AGA CTG C
 Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
Tumor tissues or cells were homogenized and proteins were extracted using
 SUPPLEMENTAL MATERIALS AND METHODS Western Blotting Tumor tissues or cells were homogenized and proteins were extracted using T-PER tissue protein extraction buffer. Protein concentrations were determined
SUPPLEMENTAL MATERIALS AND METHODS Western Blotting Tumor tissues or cells were homogenized and proteins were extracted using T-PER tissue protein extraction buffer. Protein concentrations were determined
Immunological Applications. Chapter 8: Background
 Immunological Applications Chapter 8: Background The Immune System Types of Immunity Innate The natural immunity present at birth Acquired A specific response to foreign substances. Some cells remember
Immunological Applications Chapter 8: Background The Immune System Types of Immunity Innate The natural immunity present at birth Acquired A specific response to foreign substances. Some cells remember
Electrophoresis and transfer
 Electrophoresis and transfer Electrophoresis Cation = positively charged ion, it moves toward the cathode (-) Anion = negatively charged ion, it moves toward the anode (+) Amphoteric substance = can have
Electrophoresis and transfer Electrophoresis Cation = positively charged ion, it moves toward the cathode (-) Anion = negatively charged ion, it moves toward the anode (+) Amphoteric substance = can have
Inactivation of Genes for Antigenic Variation in the Relapsing Fever Spirochete Borrelia hermsii Reduces Infectivity in Mice and Transmission by Ticks
 University of Montana ScholarWorks at University of Montana Biological Sciences Faculty Publications Biological Sciences 4-3-2014 Inactivation of Genes for Antigenic Variation in the Relapsing Fever Spirochete
University of Montana ScholarWorks at University of Montana Biological Sciences Faculty Publications Biological Sciences 4-3-2014 Inactivation of Genes for Antigenic Variation in the Relapsing Fever Spirochete
Borrelia burgdorferi OspC Protein Required Exclusively in a Crucial Early Stage of Mammalian Infection
 INFECTION AND IMMUNITY, June 2006, p. 3554 3564 Vol. 74, No. 6 0019-9567/06/$08.00 0 doi:10.1128/iai.01950-05 Borrelia burgdorferi OspC Protein Required Exclusively in a Crucial Early Stage of Mammalian
INFECTION AND IMMUNITY, June 2006, p. 3554 3564 Vol. 74, No. 6 0019-9567/06/$08.00 0 doi:10.1128/iai.01950-05 Borrelia burgdorferi OspC Protein Required Exclusively in a Crucial Early Stage of Mammalian
MIC1-MAG1-SAG1 chimeric protein, a most effective antigen for detection of human
 CVI Accepts, published online ahead of print on 3 October 2012 Clin. Vaccine Immunol. doi:10.1128/cvi.00452-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 MIC1-MAG1-SAG1
CVI Accepts, published online ahead of print on 3 October 2012 Clin. Vaccine Immunol. doi:10.1128/cvi.00452-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 MIC1-MAG1-SAG1
Supplemental Data Supplemental Figure 1.
 Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Mouse ICAM-1 / CD54 ELISA Pair Set
 Mouse ICAM-1 / CD54 ELISA Pair Set Catalog Number : SEK50440 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General
Mouse ICAM-1 / CD54 ELISA Pair Set Catalog Number : SEK50440 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General
Supplementary Material - Methods
 Novel Protein-Protein Interactions in the Schizophrenia interactome Supplementary Material - Methods Experimental validations of predicted interactions Table S1-1: Protein pairs that were validated by
Novel Protein-Protein Interactions in the Schizophrenia interactome Supplementary Material - Methods Experimental validations of predicted interactions Table S1-1: Protein pairs that were validated by
Product Datasheet. ATF3 Antibody NBP Unit Size: 0.1 ml. Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles.
 Product Datasheet ATF3 Antibody NBP1-02935 Unit Size: 0.1 ml Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles. Publications: 2 Protocols, Publications, Related Products,
Product Datasheet ATF3 Antibody NBP1-02935 Unit Size: 0.1 ml Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles. Publications: 2 Protocols, Publications, Related Products,
Transcriptional Regulation of the Borrelia burgdorferi Antigenically Variable VlsE Surface Protein
 JOURNAL OF BACTERIOLOGY, July 2006, p. 4879 4889 Vol. 188, No. 13 0021-9193/06/$08.00 0 doi:10.1128/jb.00229-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Transcriptional Regulation
JOURNAL OF BACTERIOLOGY, July 2006, p. 4879 4889 Vol. 188, No. 13 0021-9193/06/$08.00 0 doi:10.1128/jb.00229-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Transcriptional Regulation
PRODUCT INFORMATION. Composition of SOC medium supplied :
 Product Name : Competent Cell BL21(DE3)pLysS Code No. : DS260 Size : 100 μl 10 Competency : > 5 10 7 cfu/μg (puc19) Supplied product : SOC medium, 1 ml 10 This product is for research use only Description
Product Name : Competent Cell BL21(DE3)pLysS Code No. : DS260 Size : 100 μl 10 Competency : > 5 10 7 cfu/μg (puc19) Supplied product : SOC medium, 1 ml 10 This product is for research use only Description
Anti-HB-EGF (Human) mab
 Page 1 For Research Use Only. Not for use in diagnostic procedures. CODE No. D308-3 Anti-HB-EGF (Human) mab CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 3H4 Mouse IgG1
Page 1 For Research Use Only. Not for use in diagnostic procedures. CODE No. D308-3 Anti-HB-EGF (Human) mab CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 3H4 Mouse IgG1
Apoptosis And Anti-tumor Effect Induced By Mtor Inhibitor And Autophagy Inhibitor In Human Osteosarcoma Cells
 Apoptosis And Anti-tumor Effect Induced By Mtor Inhibitor And Autophagy Inhibitor In Human Osteosarcoma Cells Ryosuke Horie. Kagawa University of medecine, Kita-gun, Japan. Disclosures: R. Horie: None.
Apoptosis And Anti-tumor Effect Induced By Mtor Inhibitor And Autophagy Inhibitor In Human Osteosarcoma Cells Ryosuke Horie. Kagawa University of medecine, Kita-gun, Japan. Disclosures: R. Horie: None.
Chapter 3. Clonal selection
 Chapter 3. Clonal selection I have called this principle, by which each slight variation, if useful, is preserved, by the term of Natural Selection -Charles Darwin, On the Origin of Species, 1859 4 The
Chapter 3. Clonal selection I have called this principle, by which each slight variation, if useful, is preserved, by the term of Natural Selection -Charles Darwin, On the Origin of Species, 1859 4 The
RheB Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based RheB Activation Assay Kit Catalog Number: 81201 20 assays NewEast Biosciences 1 FAX: 610-945-2008 Table
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based RheB Activation Assay Kit Catalog Number: 81201 20 assays NewEast Biosciences 1 FAX: 610-945-2008 Table
Mouse IFNAR1 ELISA Pair Set
 Mouse IFNAR1 ELISA Pair Set Catalog Number : SEK50469 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General ELISA
Mouse IFNAR1 ELISA Pair Set Catalog Number : SEK50469 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General ELISA
T-cell response. Taken from NIAID: s.aspx
 T-cell receptor T-cell response 1. Macrophage or dendritic cell digest antigen bacteria, virus 2. Fragments of Ag bind to major histo-compatiblity (MHC) proteins in macrophage. 3. MHC I-Ag fragment expressed
T-cell receptor T-cell response 1. Macrophage or dendritic cell digest antigen bacteria, virus 2. Fragments of Ag bind to major histo-compatiblity (MHC) proteins in macrophage. 3. MHC I-Ag fragment expressed
EAR BIOPSY LOCATION INFLUENCES DETECTION OF BORRELIA BURGDORFERI BY PCR, BUT NOT BY CULTURE IN NATURALLY INFECTED PEROMYSCUS LEUCOPUS
 EAR BIOPSY LOCATION INFLUENCES DETECTION OF BORRELIA BURGDORFERI BY PCR, BUT NOT BY CULTURE IN NATURALLY INFECTED PEROMYSCUS LEUCOPUS Authors: Erik K. Hofmeister, and James E. Childs Source: Journal of
EAR BIOPSY LOCATION INFLUENCES DETECTION OF BORRELIA BURGDORFERI BY PCR, BUT NOT BY CULTURE IN NATURALLY INFECTED PEROMYSCUS LEUCOPUS Authors: Erik K. Hofmeister, and James E. Childs Source: Journal of
Nongenetic Reprogramming of the Ligand Specificity. of Growth Factor Receptors by Bispecific DNA Aptamers
 Supporting Information For Nongenetic Reprogramming of the Ligand Specificity of Growth Factor Receptors by Bispecific DNA Aptamers Ryosuke Ueki,* Saki Atsuta, Ayaka Ueki and Shinsuke Sando* Department
Supporting Information For Nongenetic Reprogramming of the Ligand Specificity of Growth Factor Receptors by Bispecific DNA Aptamers Ryosuke Ueki,* Saki Atsuta, Ayaka Ueki and Shinsuke Sando* Department
Cdc42 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
Quantifying small numbers of antibodies with a near-universal protein-dna chimera
 Quantifying small numbers of antibodies with a near-universal protein-dna chimera Ian Burbulis, Kumiko Yamaguchi, Richard Yu, Orna Resnekov & Roger Brent Supplementary figures and text: Supplementary figure
Quantifying small numbers of antibodies with a near-universal protein-dna chimera Ian Burbulis, Kumiko Yamaguchi, Richard Yu, Orna Resnekov & Roger Brent Supplementary figures and text: Supplementary figure
Supplementary Information
 Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Electronic Supplementary Information Sensitive detection of polynucleotide kinase using rolling circle amplification-induced chemiluminescence
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Sensitive detection of polynucleotide kinase
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Sensitive detection of polynucleotide kinase
Dierks Supplementary Fig. S1
 Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were
 1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
STANDARD OPERATIONS PROCEDURES FOR THE COMMON FUND: PROTEIN CAPTURE REAGENTS PROGRAM (ELISA)
 STANDARD OPERATIONS PROCEDURES FOR THE COMMON FUND: PROTEIN CAPTURE REAGENTS PROGRAM (ELISA) 1. PURPOSE This procedure is to be used for the characterization of purified monoclonal antibody. 2. SCOPE This
STANDARD OPERATIONS PROCEDURES FOR THE COMMON FUND: PROTEIN CAPTURE REAGENTS PROGRAM (ELISA) 1. PURPOSE This procedure is to be used for the characterization of purified monoclonal antibody. 2. SCOPE This
strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular
![strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular](/thumbs/72/66361212.jpg) Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
SCIENTIFIC REPORT. regarding the implementation of the project between December December 2014
 SCIENTIFIC REPORT regarding the implementation of the project between December 013 - December 014 Title of the project: DETECTION AND IDENTIFICATION OF BIOMOLECULES OF MEDICAL INTEREST BY USING MAGNETIC
SCIENTIFIC REPORT regarding the implementation of the project between December 013 - December 014 Title of the project: DETECTION AND IDENTIFICATION OF BIOMOLECULES OF MEDICAL INTEREST BY USING MAGNETIC
Rat IgG ELISA Catalog #:
 INTENDED USE The IMMUNO-TEK Rat IgG ELISA* Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA) designed for the measurement of rat IgG in rat serum, plasma, saliva, mucosa, cell culture
INTENDED USE The IMMUNO-TEK Rat IgG ELISA* Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA) designed for the measurement of rat IgG in rat serum, plasma, saliva, mucosa, cell culture
Antibody to a 39-Kilodalton Borrelia burgdorferi Antigen (P39)
 JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 1991, p. 236-243 Vol. 29, No. 2 0095-1137/91/020236-08$02.00/0 Copyright C) 1991, American Society for Microbiology Antibody to a 39-Kilodalton Borrelia burgdorferi
JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 1991, p. 236-243 Vol. 29, No. 2 0095-1137/91/020236-08$02.00/0 Copyright C) 1991, American Society for Microbiology Antibody to a 39-Kilodalton Borrelia burgdorferi
SUPPLEMENTAL MATERIAL. Supplemental Methods:
 SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
Attachment of Borrelia burgdorferi within Ixodes scapularis mediated by outer surface protein A
 Attachment of Borrelia burgdorferi within Ixodes scapularis mediated by outer surface protein A Utpal Pal, 1 Aravinda M. de Silva, 2 Ruth R. Montgomery, 1 Durland Fish, 3 Juan Anguita, 1 John F. Anderson,
Attachment of Borrelia burgdorferi within Ixodes scapularis mediated by outer surface protein A Utpal Pal, 1 Aravinda M. de Silva, 2 Ruth R. Montgomery, 1 Durland Fish, 3 Juan Anguita, 1 John F. Anderson,
Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of
 Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of the cell line) were immunostained for HA, acetylated
Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of the cell line) were immunostained for HA, acetylated
Arf6 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Arf6 Activation Assay Kit Catalog Number: 82401 20 assays NewEast Biosciences 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Arf6 Activation Assay Kit Catalog Number: 82401 20 assays NewEast Biosciences 1 Table of Content Product
Product Datasheet. Perilipin Antibody NB Unit Size: 0.1 ml
 Product Datasheet Perilipin Antibody NB110-40760 Unit Size: 0.1 ml Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles. Publications: 6 Protocols, Publications, Related
Product Datasheet Perilipin Antibody NB110-40760 Unit Size: 0.1 ml Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles. Publications: 6 Protocols, Publications, Related
SANTA CRUZ BIOTECHNOLOGY, INC.
 TECHNICAL SERVICE GUIDE: Western Blotting 2. What size bands were expected and what size bands were detected? 3. Was the blot blank or was a dark background or non-specific bands seen? 4. Did this same
TECHNICAL SERVICE GUIDE: Western Blotting 2. What size bands were expected and what size bands were detected? 3. Was the blot blank or was a dark background or non-specific bands seen? 4. Did this same
Human ICAM-2 / CD102 ELISA Pair Set
 Human ICAM-2 / CD102 ELISA Pair Set Catalog Number : SEK10332 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General
Human ICAM-2 / CD102 ELISA Pair Set Catalog Number : SEK10332 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General
Induction of an outer surface protein on Borrelia burgdorferi during tick feeding
 Proc. Natl. Acad. Sci. USA Vol. 92, pp. 299-2913, March 1995 Microbiology Induction of an outer surface protein on Borrelia burgdorferi during tick feeding (Ixodes scapularis/lyme disease/blood meal/outer
Proc. Natl. Acad. Sci. USA Vol. 92, pp. 299-2913, March 1995 Microbiology Induction of an outer surface protein on Borrelia burgdorferi during tick feeding (Ixodes scapularis/lyme disease/blood meal/outer
Human IL-10 ELISA MAX Set Deluxe
 Human IL-10 ELISA MAX Set Deluxe Cat. No. 430604 (5 plates) 430605 (10 plates) 430606 (20 plates) ELISA Set for Accurate Cytokine Quantification from Cell Culture Supernatant, Serum, Plasma or Other Body
Human IL-10 ELISA MAX Set Deluxe Cat. No. 430604 (5 plates) 430605 (10 plates) 430606 (20 plates) ELISA Set for Accurate Cytokine Quantification from Cell Culture Supernatant, Serum, Plasma or Other Body
IR-Blot Secondary antibodies Rev01
 0 About us Cyanagen is a biotech company located in Bologna, dedicated to research, development and production of reagents for molecular diagnostic since 2003 and one of the leading companies in the field
0 About us Cyanagen is a biotech company located in Bologna, dedicated to research, development and production of reagents for molecular diagnostic since 2003 and one of the leading companies in the field
Strep-tag detection in Western blots
 Strep-tag detection in Western blots General protocol for the detection of Strep-tag fusion proteins Last date of revision April 2012 Version PR07-0010 www.strep-tag.com For research use only Important
Strep-tag detection in Western blots General protocol for the detection of Strep-tag fusion proteins Last date of revision April 2012 Version PR07-0010 www.strep-tag.com For research use only Important
Mouse CD30L ELISA Kit
 Product Information Sheet Catalog No. BO0150 Size 96T Range 15.6pg/ml-1000pg/ml Sensitivity < 1pg/ml Specificity No detectable cross-reactivity with any other cytokine. Storage Store at 4 for frequent
Product Information Sheet Catalog No. BO0150 Size 96T Range 15.6pg/ml-1000pg/ml Sensitivity < 1pg/ml Specificity No detectable cross-reactivity with any other cytokine. Storage Store at 4 for frequent
Project 07/111 Final Report October 31, Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines
 Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
HELICA BIOSYSTEMS, INC. HIGH SENSITIVITY HUMAN C-REACTIVE PROTEIN FOR RESEARCH USE ONLY (Not for in vitro diagnostic use)
 INTENDED USE HELICA BIOSYSTEMS, INC. HIGH SENSITIVITY HUMAN C-REACTIVE PROTEIN FOR RESEARCH USE ONLY (Not for in vitro diagnostic use) The Helica C-reactive protein assay is intended for the detection
INTENDED USE HELICA BIOSYSTEMS, INC. HIGH SENSITIVITY HUMAN C-REACTIVE PROTEIN FOR RESEARCH USE ONLY (Not for in vitro diagnostic use) The Helica C-reactive protein assay is intended for the detection
Received 13 November 2001/Returned for modification 12 February 2002/Accepted 27 March 2002
 INFECTION AND IMMUNITY, July 2002, p. 3468 3478 Vol. 70, No. 7 0019-9567/02/$04.00 0 DOI: 10.1128/IAI.70.7.3468 3478.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Changes
INFECTION AND IMMUNITY, July 2002, p. 3468 3478 Vol. 70, No. 7 0019-9567/02/$04.00 0 DOI: 10.1128/IAI.70.7.3468 3478.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Changes
HELICA BIOSYSTEMS, INC. MOUSE C-REACTIVE PROTEIN QUANTITATION BY ELISA FOR RESEARCH USE ONLY
 HELICA BIOSYSTEMS, INC. MOUSE C-REACTIVE PROTEIN QUANTITATION BY ELISA FOR RESEARCH USE ONLY INTENDED USE The Helica C-reactive protein assay is intended for the detection and quantification of mouse C-reactive
HELICA BIOSYSTEMS, INC. MOUSE C-REACTIVE PROTEIN QUANTITATION BY ELISA FOR RESEARCH USE ONLY INTENDED USE The Helica C-reactive protein assay is intended for the detection and quantification of mouse C-reactive
IR-Blot Secondary antibodies Rev00
 0 About us Cyanagen is a biotech company located in Bologna, dedicated to research, development and production of reagents for molecular diagnostic since 2003 and one of the leading companies in the field
0 About us Cyanagen is a biotech company located in Bologna, dedicated to research, development and production of reagents for molecular diagnostic since 2003 and one of the leading companies in the field
Mouse IgM ELISpot BASIC
 Mouse IgM ELISpot BASIC Product Code: 3885-2H CONTENTS: Vial 1 (green top) Monoclonal antibody MT6A3 (1.2 ml) Concentration: 0.5 mg/ml Vial 2 (yellow top) Biotinylated monoclonal antibody MT9A2 (100 μl)
Mouse IgM ELISpot BASIC Product Code: 3885-2H CONTENTS: Vial 1 (green top) Monoclonal antibody MT6A3 (1.2 ml) Concentration: 0.5 mg/ml Vial 2 (yellow top) Biotinylated monoclonal antibody MT9A2 (100 μl)
Supplementary Figure 1A A404 Cells +/- Retinoic Acid
 Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
Human C-Reactive Protein / CRP ELISA Pair Set
 Human C-Reactive Protein / CRP ELISA Pair Set Catalog Number : SEK11250 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Human C-Reactive Protein / CRP ELISA Pair Set Catalog Number : SEK11250 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Human IgG ELISpot BASIC
 Human IgG ELISpot BASIC Product Code: 3850-2H CONTENTS: Vial 1 (yellow top) Monoclonal antibodies MT91/145 (1.2 ml) Concentration: 0.5 mg/ml Vial 2 (green top) Biotinylated monoclonal antibodies MT78/145
Human IgG ELISpot BASIC Product Code: 3850-2H CONTENTS: Vial 1 (yellow top) Monoclonal antibodies MT91/145 (1.2 ml) Concentration: 0.5 mg/ml Vial 2 (green top) Biotinylated monoclonal antibodies MT78/145
Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis
 1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
BL21(DE3) expression competent cell pack DS265. Component Code No. Contents Competent cell BL21(DE3) DS250 5 tubes (100 μl/tube)
 Product Name : Code No. : Kit Component : BL21(DE3) expression competent cell pack DS265 Component Code No. Contents Competent cell BL21(DE3) DS250 5 tubes (100 μl/tube) transfromation efficiency: 5 10
Product Name : Code No. : Kit Component : BL21(DE3) expression competent cell pack DS265 Component Code No. Contents Competent cell BL21(DE3) DS250 5 tubes (100 μl/tube) transfromation efficiency: 5 10
ELISA MAX Set Deluxe. Human IL-32α
 ELISA MAX Set Deluxe Human IL-32α Cat. No. 433504 (5 plates) 433505 (10 plates) ELISA Set for Accurate Cytokine Quantitation from Cell Culture Supernatant, Serum, Plasma or Other Body Fluids BioLegend,
ELISA MAX Set Deluxe Human IL-32α Cat. No. 433504 (5 plates) 433505 (10 plates) ELISA Set for Accurate Cytokine Quantitation from Cell Culture Supernatant, Serum, Plasma or Other Body Fluids BioLegend,
Western blotting technique: principle, procedure and application
 Western blotting technique: principle, procedure and application The term blotting refers to the transfer of biological samples from a gel to a membrane and their subsequent detection on the surface of
Western blotting technique: principle, procedure and application The term blotting refers to the transfer of biological samples from a gel to a membrane and their subsequent detection on the surface of
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Svegliati Baroni S, Santillo M, Bevilacqua F, et al. Stimulatory
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Svegliati Baroni S, Santillo M, Bevilacqua F, et al. Stimulatory
BIOMAN 2015 IMMUNOASSAYS. Barbara Bielska Northampton Community College Tannersville, PA
 BIOMAN 2015 IMMUNOASSAYS Barbara Bielska Northampton Community College Tannersville, PA 1 Immunoassays An immunoassay is a biochemical test that measures the presence or concentration of a molecules (typically
BIOMAN 2015 IMMUNOASSAYS Barbara Bielska Northampton Community College Tannersville, PA 1 Immunoassays An immunoassay is a biochemical test that measures the presence or concentration of a molecules (typically
Borrelia burgdorferi Gene Expression In Vivo and Spirochete Pathogenicity
 INFECTION AND IMMUNITY, Mar. 2000, p. 1222 1230 Vol. 68, No. 3 0019-9567/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Borrelia burgdorferi Gene Expression In Vivo
INFECTION AND IMMUNITY, Mar. 2000, p. 1222 1230 Vol. 68, No. 3 0019-9567/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Borrelia burgdorferi Gene Expression In Vivo
Lab. 7: Serological Tests ELISA. 320 MIC Microbial Diagnosis 320 MBIO PRACTICAL. Amal Alghamdi 2018
 Lab. 7: 320 MIC Microbial Diagnosis Serological Tests ELISA. 320 MBIO PRACTICAL Amal Alghamdi 2018 1 Infection and Immunity Serology is the study of immune bodies in human blood. These are products of
Lab. 7: 320 MIC Microbial Diagnosis Serological Tests ELISA. 320 MBIO PRACTICAL Amal Alghamdi 2018 1 Infection and Immunity Serology is the study of immune bodies in human blood. These are products of
Ab SpinTrap Ab Buffer Kit
 GE Healthcare Data File 28-9020-30 AB Protein Sample Preparation Ab SpinTrap Ab Buffer Kit The Ab SpinTrap and the Protein G HP SpinTrap are identical columns. The difference between the article numbers
GE Healthcare Data File 28-9020-30 AB Protein Sample Preparation Ab SpinTrap Ab Buffer Kit The Ab SpinTrap and the Protein G HP SpinTrap are identical columns. The difference between the article numbers
CD93 and dystroglycan cooperation in human endothelial cell adhesion and migration
 /, Supplementary Advance Publications Materials 2016 CD93 and dystroglycan cooperation in human endothelial cell adhesion and migration Supplementary Materials Supplementary Figure S1: In ECs CD93 silencing
/, Supplementary Advance Publications Materials 2016 CD93 and dystroglycan cooperation in human endothelial cell adhesion and migration Supplementary Materials Supplementary Figure S1: In ECs CD93 silencing
INVESTIGATION OF THE BINDING SPECIFICITY OF IGF-IR USING MONOCLONAL ANTIBODIES
 INVESTIGATION OF THE BINDING SPECIFICITY OF IGF-IR USING MONOCLONAL ANTIBODIES By Mehrnaz Keyhanfar, Pharm.D. A thesis submitted to the University of Adelaide, South Australia in fulfilment of the requirements
INVESTIGATION OF THE BINDING SPECIFICITY OF IGF-IR USING MONOCLONAL ANTIBODIES By Mehrnaz Keyhanfar, Pharm.D. A thesis submitted to the University of Adelaide, South Australia in fulfilment of the requirements
Rhesus CD16 / FCGR3 ELISA Pair Set
 Rhesus CD16 / FCGR3 ELISA Pair Set Catalog Number : SEK90013 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General
Rhesus CD16 / FCGR3 ELISA Pair Set Catalog Number : SEK90013 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in the General
An engineered tryptophan zipper-type peptide as a molecular recognition scaffold
 SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
Hes6. PPARα. PPARγ HNF4 CD36
 SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
Immunohistochemistry guide
 Immunohistochemistry guide overview immunohistochemistry Overview Immunohistochemistry is a laboratory technique utilized for the visual detection of antigens in tissue. When working with cells this technique
Immunohistochemistry guide overview immunohistochemistry Overview Immunohistochemistry is a laboratory technique utilized for the visual detection of antigens in tissue. When working with cells this technique
Borrelia burgdorferi Escape Mutants That Survive in the Presence of Antiserum to the OspA Vaccine Are Killed When Complement Is Also Present
 INFECTION AND IMMUNITY, June 1998, p. 2540 2546 Vol. 66, No. 6 0019-9567/98/$04.00 0 Copyright 1998, American Society for Microbiology Borrelia burgdorferi Escape Mutants That Survive in the Presence of
INFECTION AND IMMUNITY, June 1998, p. 2540 2546 Vol. 66, No. 6 0019-9567/98/$04.00 0 Copyright 1998, American Society for Microbiology Borrelia burgdorferi Escape Mutants That Survive in the Presence of
