By: ALVARO OROZCO JARAMILLO CONSULTANT ENGINEER Professor (retired) Universidad de los Andes
|
|
|
- Loraine Kennedy
- 6 years ago
- Views:
Transcription
1 By: ALVARO OROZCO JARAMILLO CONSULTANT ENGINEER Professor (retired) Universidad de los Andes
2 CONTENIDO: 1. INTRODUCTION 2. GLOBAL WARMING AND GREENHOUSE GASES (GHG) 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS 4. IMPACT OF WWTP ON GLOBAL WARMING 5. CONCLUSIONS 6. BIBLIOGRAPHY
3 1. INTRODUCTION Global warming is the increase in the average temperature of the Earth's nearsurface air and oceans since the mid 20th century and its projected continuation. Global warming is very likely due to the observed increase in anthropogenic GHG concentrations (mostly CO 2 ) generated by fossil fuels burning (carbon, oil, natural gas, etc.). Other GHG (CH4, N2O, SF 6 ) emissions also contributes to global warming. Water vapor is also a GHG but its concentration depends on Earth s temperature and is considered in mathematically based Global Climate Models (GCM) as the water vapor feedback. The major non gas contributor to the Earth's greenhouse effect, clouds, also absorb and emit infrared radiation and thus have an effect on radiative properties of the greenhouse gases. Earth's most abundant greenhouse gases are: (i) water vapor, which contributes 36 70%; (ii) carbon dioxide, which contributes 9 26%; (iii) methane, which contributes 4 9%; and (iv) ozone, which contributes 3 7%.
4 1. INTRODUCTION The Intergovernmental Panel on Climate Change (IPCC) is a scientific intergovernmental body tasked to evaluate the risk of climate change caused by human activity. The panel was established in 1988 by the World Meteorological Organization(WMO) and the United Nations Environmental Programme (UNEP), two organizations of the United Nations. The IPCC shared the 2007 Nobel Peace Prize with Al Gore. The Kyoto Protocol is a protocol to the United Nations Framework Convention on Climate Change (UNFCCC or FCCC) produced at the United Nations Conference on Environment and Development (UNCED), (also known as the Earth Summit), held in Rio de Janeiro (Brazil) in June The treaty is intended to achieve "stabilization of green house gas (GHG) concentrations in the atmosphere at a level that would prevent dangerous anthropogenic interference with the climate system
5 1. INTRODUCTION Wastewater Treatment Plant (WWTP) practice only considers as GHG the CH 4 (produced by sludge and wastewater anaerobic treatment) and N 2 O (produced by nitrification, denitrification and, in a minor scale, activated sludge treatment). The 100 years global warming potential (GWP) of CH4 is 23 times higher than that of CO 2 (1 t CH4 = 23t equivalent CO 2 or CO 2 e) and is the GHG more important in WWTP. The 100 years global warming potential (GWP) of N 2 O is 320 times higher than that of CO 2 (1 t N 2 O = 296 t CO 2 e). Nitrous oxide (N 2 O) may be generated by nitrification and denitrification processes during biological treatment of the wastewater. Direct discharges of wastewater into water masses will produce N2O anyway and should be account for as indirect emissions. CO 2 emission in wastewater treatment processes was found to be neutral in terms of global warming due to its biogenic origin, and is not considered by IPCC as a global warming contributor by WWTP. WWTP accounts for less than 0.5% of the total GHG emissions worldwide. In any case, WWTP practices should incorporate methods to control GHG.
6 2. GLOBAL WARMING Sun is the Earth s main source of energy. There are others (e.g. geothermic sources) but they are minute in comparison with solar radiation. Earth s temperature is the resultant of: (i) incoming solar radiation (S) mostly irradiated in the range of visible light of the electromagnetic spectrum, minus the fraction (α) which is reflected by polar caps and clouds. This fraction is called the terrestrial albedo; (ii) the emission of terrestrial radiation, which depend on mean Earth s temperature. GHG are transparent to short wave radiation (99% emitted by the Sun) and opaque to long wave radiation (99% emitted by Earth)
7 2. GLOBAL WARMING How much is the solar incoming radiation?: On the average the solar incoming radiation is 1370 W/m 2. This radiation coming through the Earth s projected circle is distributed on the whole area of Earth s sphere which is four times greater. In other words, the average radiation arriving to Earth surface is 1370/4 = 342 W/m 2. However, of the total solar incoming radiation 30% is reflected by the polar caps, clouds, etc. This percentage in decimal form (0.3) is the terrestrial albedo (α).
8 2. GLOBAL WARMING How much heat is re radiated by Earth?: Assuming that Earth is in thermal equilibrium, then the incoming Sun radiation (heat) should be equal to Earth s radiation as a black body (Stefan Boltzmann s Law). Solving the equilibrium equation for a terrestrial albedo α = 0.3 obtain an Earth s average temperature of 18 o C. It seems a bit cold! (real Average surface temperature is 15 C). Is the physics wrong? No, it s just incomplete. To obtain the right answer it is necessary to have into account the Greenhouse Effect.
9 2. GLOBAL WARMING What happens then? Because of its temperature, the Earth s surface emits radiation in the 4.0 to μm region. Most of this is absorbed by greenhouse gases (GHG). But the atmosphere is at a similar temperature, so these gases will re emit much of this radiation, some to space, but more back to the surface, making the surface warmer. This is known as the Greenhouse Effect. Taking into account the Greenhouse Effect, the GHG reflection from the atmosphere back to Earth is 155 W/m 2 and solving again the thermal balance equation we found that the mean temperature is 15ºC, in agreement with reality. The Greenhouse Effect is a natural phenomenon and without it life on Earth would be inexistent! The GHG have existed for millions years.
10 2. GLOBAL WARMING
11 2. GLOBAL WARMING In the long range, solar radiation is also affected by Milankovitch cycles, due to position changes of the planet respect to the Sun.
12 2. GLOBAL WARMING
13 2. GLOBAL WARMING There are two ways of measuring the greenhouse effect: (i) The first is the 33 C difference between the effective temp ( 18 C) and the actual (average) surface temp (15 C). (ii) The second is the difference between the 390 Wm 2 surface emission and the 237 Wm 2 emission to space: 153 Wm 2 (iii) about 50, Of this 153 Wm 2, H 2 O vapor accounts for about 95, CO 2 for and N 2 O, CH 4, O 3 and CFCs about 2 each. The interesting question which now confronts us is: How are these numbers changing, as a result of our actions?
14 2. GLOBAL WARMING
15 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS Wastewater is a source of methane (CH 4 ) when treated or disposed of anaerobically. It can also be a source of nitrous oxide (N 2 O) emissions. Carbon dioxide (CO 2 ) emissions from wastewater are not considered in the IPCC Guidelines because these are of biogenic origin and should not be included in inventories of emissions. Wastewater originates from a variety of domestic, commercial and industrial sources and may be treated: (i) on site (uncollected), (ii) sewered to a centralized plant (collected) or (iii) disposed untreated nearby or via an outfall. Domestic wastewater is defined as wastewater from household water use, while industrial wastewater is from industrial practices only. The most common wastewater treatment methods in developed countries are centralized aerobic wastewater treatment plants and lagoons for both domestic and industrial wastewater. The degree of wastewater treatment varies in most developing countries. Domestic wastewater is treated in centralized plants, pit latrines, septic systems or disposed of in unmanaged lagoons or waterways, via open or closed sewers. Presently, anaerobic treatment in warm climate is increasingly used.
16 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS Centralized wastewater treatment methods can be classified as primary, secondary, and tertiary treatment. In primary treatment, physical barriers remove larger solids from the wastewater. Remaining particulates are then allowed to settle. Secondary treatment consists of a combination of biological processes that promote biodegradation by microorganisms. These may include aerobic stabilization ponds, trickling filters, and activated sludge processes, as well as anaerobic reactors and lagoons. Tertiary treatment processes are used to further purify the wastewater of pathogens, contaminants, and remaining nutrients such as nitrogen and phosphorus compounds
17 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS
18 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS 3.1 N 2 O EMISSION FACTOR (EF) Nitrous oxide (N 2 O) is associated with the degradation of nitrogen components in the wastewater, e.g., urea, nitrate and protein. Direct emissions of N 2 O may be generated during both nitrification and denitrification of the nitrogen present. Both processes can occur in the plant and in the effluent receiving water body. Nitrification is an aerobic process converting ammonia and other nitrogen compounds into nitrate (NO 3 ) Denitrification occurs under anoxic conditions (without free oxygen), and involves the biological conversion of nitrate into nitrogen gas (N 2 ). Nitrous oxide can be an intermediate product of both processes, but is more often associated with denitrification. N 2 O emissions should be converted into CO 2 equivalent. (1 t N 2 O = 296 t CO 2 e)
19 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS
20 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS Emissions from advanced centralized wastewater treatment plants are typically much smaller than those from effluent and may only be of interest for centralized wastewater treatment plants with controlled nitrification and denitrification steps. The overall emission factor (EF) to estimate N 2 O emissions from such plants is 3.2 g N 2 O/person/year. If there are commercial and industrial discharges a default factor (F ind/com ) of 1,25, should be applied: (kg N 2 O /yr) WWTP =P x 3,2 kg N 2 O /person.yr x F ind/com = P x 4 kg N 2 O /person.yr where P is the population equivalent treated at the WWTP
21 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS
22 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS 3.2 CH 4 EMISSION FACTOR (EF) Wastewater as well as its sludge components can produce CH 4 if it degrades anaerobically. The extent of CH 4 production depends primarily on the quantity of degradable organic material (DOM) in the wastewater. The temperature, and the type of treatment system. With increases in temperature, the rate of CH 4 production increases. This is especially important in uncontrolled systems and in warm climates. Below 15 C, significant CH 4 production is unlikely because methanogens are not active and the lagoon will serve principally as a sedimentation tank. The BOD concentration indicates only the amount of carbon that is aerobically biodegradable. The standard measurement for BOD is a 5 day test, denoted as BOD5. CH 4 production should be converted into CO 2 e (1 t CH 4 = 23 t CO 2 e)
23 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS The IPCC general equation to estimate CH 4 emissions from domestic wastewater is as follows: CH 4 Emissions =[ (U i T i,j EF j )] (TOW S ) R CH 4 Emissions = CH 4 emissions in inventory year, kg CH 4 /yr TOW= total organics in wastewater in inventory year, kg BOD/yr S = organic component removed as sludge in inventory year, kg BOD/yr U i = fraction of population in income group i in inventory year. degree of utilization of treatment/discharge pathway or system, j, for each income group T i,j = fraction i in inventory year. i= income group: rural, urban high income and urban low income j= each treatment/discharge pathway or system R = amount of CH 4 recovered in inventory year, kg CH4/yr EF j = emission factor, kg CH 4 / kg BOD
24 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS The IPCC proposes the following equation for calculating the CH 4 Emission Factor of anaerobic WWTP: kg CH 4 = P * DOM * (1 % DOM disposed of as sludge) * (CH 4 ) max * MCF kg (CH 4 ) burned To calculate the EF from sludge treatment, the IPCC proposed the following equation: kg CH 4 = P * DOM* % DOM disposed of as sludge * (CH 4 ) max * MCF kg (CH 4 ) burned In both equations, DOM is the degradable organic matter per capita (as BOD). A default value of DOM= 0.05 kg DBO5/person.d, is suggested unless the country real value is known. The good practice IPCC guide proposes a maximum production of 0.6 kg CH4/kgDBO. The correction factor for methane production depends on treatment type and affect the proposed maximum production (see table below)
25 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS
26 3. WASTEWATER TREATMENT PLANT EMISSION FACTORS 3.3 INDIRECT EMISSIONS For the fully aerobic process, up to 1.4 kg CO 2 /kg COD removed originates from power generation needed for aeration. This means that considerably more CO 2 is produced in power generation needed for aeration than in the actual treatment process, and all of this is typically from fossil fuels, whereas the energy from the wastewater pollutants comes primarily from renewable energy sources (food, i.e. biogenic origin ). A change from anaerobic to aerobic sludge treatment processes (for the same aerobic main process) has a massive impact on the CO 2 production from fossil fuels: an additional 0.8 kg CO 2 /kg COD removed is produced by changing to aerobic sludge digestion. In terms of GHG production, the total output (in CO 2 equivalents) can be reduced from 2.4 kg CO 2 /kg COD removed for fully aerobic treatment to 1.0 kg CO 2 /kg COD removed for primarily anaerobic processes. Major advantages can be gained by using primarily anaerobic processes as it is possible to largely eliminate any net energy input to the process, and therefore the production of GHG from fossil fuels.
27 4. IMPACT OF WWTP ON GLOBAL WARMING
28 4. IMPACT OF WWTP ON GLOBAL WARMING
29 4. IMPACT OF WWTP ON GLOBAL WARMING
30 4. CONCLUSIONS Wastewater Treatment Plants (WWTP) contributes 21% of total CO 2 e from sector Waste, which is the 2.3 % of total CO 2 e USA GHG emissions. This account for less than 0.5% of USA greenhouse gas emissions. Wastewater as well as its sludge components can produce CH 4 if it degrades anaerobically. Nitrous oxide (N 2 O) is associated with the degradation of nitrogen components in the wastewater, e.g., urea, nitrate and protein. Direct emissions of N 2 O may be generated during both nitrification and denitrification of the nitrogen present. Both processes can occur in the plant and in the water body that is receiving the effluent. Carbon dioxide (CO 2 ) emissions from wastewater are not considered in the IPCC Guidelines because these are of biogenic origin and should not be included in inventories of emissions.
31 4. CONCLUSIONS More CO 2 is produced in power generation needed for aeration than in the actual treatment process, and all of this is typically from fossil fuels, whereas the energy from the wastewater pollutants comes primarily from renewable energy sources (food, i.e. biogenic origin ). Major advantages can be gained by using primarily anaerobic processes as it is possible to largely eliminate any net energy input to the process, and therefore the production of GHG from fossil fuels. Burning produced CH 4 in WWTP additionally reduces de CO 2 e emissions due that the Potential Global Warming of 1 t CH 4 = 23 t CO 2 e.
32 7. BIBLIOGRAFÍA Orozco, A. (2007) La lucha contra el calentamiento global: un conflicto de intereses, Corporación Otraparte, Medellín. Orozco, Álvaro (2005), Bioingeniería de aguas residuales: teoría y diseño, ACODAL, Bogotá, Colombia. METCALF & EDDY (2003), Wastewater engineering: treatment and reuse, 4th Edition, McGraw Hill Co. Yochanan Kushnir, The Green House Effect and the two dimensional pattern of Earth s Radiation Budget, Greehouse pdf Thomsen Marianne y Erik Lyck (2005). Emission of CH 4 and N 2 O from Wastewater Treatment Plants (6B). IPCC, WMO, UNEP (2006), 2006 IPCC guidelines for national greenhouse gas inventories, Volume 5. Dautremont Smith, J (2002); en Guidelines for College Level Greenhouse Gas Emissions Inventories UNFCCC/CCNUCC (1996), Approved baseline and monitoring methodology AM0039: Methane emissions reduction from organic waste water and bioorganic solid waste using composting. FAO (2006), Food Security Statistics Colombia Keller J. and Hartley K. (2002), Greenhouse gas production in wastewater treatment: process selection is the major factor, Water science and technology ISSN CODEN WSTED4, London, UK. EPA (2008), Inventory of U.S. Greenhouse Gas Emissions and Sinks: , USA. Daiger, G, R. Peterson, J. Whiterspoon and E. Allen (2000), Impact of global warming concerns on wastewater treatment plant design and operation, ASCE, USA.
Discussion Paper for a Wastewater Treatment Plant Sector Greenhouse Gas Emissions Reporting Protocol
 F inal Report Discussion Paper for a Wastewater Treatment Plant Sector Greenhouse Gas Emissions Reporting Protocol Prepared for California Wastewater Climate Change Group July 2008 (Revision 3) Prepared
F inal Report Discussion Paper for a Wastewater Treatment Plant Sector Greenhouse Gas Emissions Reporting Protocol Prepared for California Wastewater Climate Change Group July 2008 (Revision 3) Prepared
2006 IPCC Guidelines for National Greenhouse Gas Inventories: Waste Sector
 Task Force on National Greenhouse Gas Inventories 2006 IPCC Guidelines for National Greenhouse Gas Inventories: Waste Sector African Regional Workshop: Technical Training on National GHG Inventories 24-27
Task Force on National Greenhouse Gas Inventories 2006 IPCC Guidelines for National Greenhouse Gas Inventories: Waste Sector African Regional Workshop: Technical Training on National GHG Inventories 24-27
The Energy and Carbon Footprint of Water Reclamation and Water Management in Greater Chicago
 METROPOLITAN WATER RECLAMATION DISTRICT OF GREATER CHICAGO The Energy and Carbon Footprint of Water Reclamation and Water Management in Greater Chicago Joseph Kozak, PhD, PE Catherine O Connor, O PhD,
METROPOLITAN WATER RECLAMATION DISTRICT OF GREATER CHICAGO The Energy and Carbon Footprint of Water Reclamation and Water Management in Greater Chicago Joseph Kozak, PhD, PE Catherine O Connor, O PhD,
Climate MRV for Africa Phase 2 Development of National GHG Inventory Domestic Wastewater Treatment
 Climate MRV for Africa Phase 2 Development of National GHG Inventory Domestic Wastewater Treatment Lead partner Project of the European Commission DG Clima Action EuropeAid/136245/DH/SER/MULTI Amr Osama
Climate MRV for Africa Phase 2 Development of National GHG Inventory Domestic Wastewater Treatment Lead partner Project of the European Commission DG Clima Action EuropeAid/136245/DH/SER/MULTI Amr Osama
Introduction to the Energy performance and Carbon emissions Assessment and Monitoring Tool (ECAM-Tool)
 Introduction to the Energy performance and Carbon emissions Assessment and Monitoring Tool (ECAM-Tool) Beta Version WaCCliM June 2015 Table of Contents Preamble... 3 The Objective of the ECAM-Tool... 3
Introduction to the Energy performance and Carbon emissions Assessment and Monitoring Tool (ECAM-Tool) Beta Version WaCCliM June 2015 Table of Contents Preamble... 3 The Objective of the ECAM-Tool... 3
Greenhouse Gas Emissions From Wastewater Treatment A Comprehensive Review
 Greenhouse Gas Emissions From Wastewater Treatment A Comprehensive Review S. Rao Chitikela,* and W.F. Ritter** *Johnson Controls, Inc. **University of Delaware Objectives Municipal wastewater and residuals
Greenhouse Gas Emissions From Wastewater Treatment A Comprehensive Review S. Rao Chitikela,* and W.F. Ritter** *Johnson Controls, Inc. **University of Delaware Objectives Municipal wastewater and residuals
Chapter 19 Global Change. Wednesday, April 18, 18
 Chapter 19 Global Change Module 62 Global Climate Change and the Greenhouse Effect After reading this module you should be able to distinguish among global change, global climate change, and global warming.
Chapter 19 Global Change Module 62 Global Climate Change and the Greenhouse Effect After reading this module you should be able to distinguish among global change, global climate change, and global warming.
Global Climate Change. The sky is falling! The sky is falling!
 Global Climate Change The sky is falling! The sky is falling! 1 Global Climate Change Radiative Equilibrium, Solar and Earth Radiation Atmospheric Greenhouse Effect Greenhouse Gases Global Climate Change
Global Climate Change The sky is falling! The sky is falling! 1 Global Climate Change Radiative Equilibrium, Solar and Earth Radiation Atmospheric Greenhouse Effect Greenhouse Gases Global Climate Change
Some resources (more websites later)
 Some resources (more websites later) Intergovernmental Panel Climate Change 2001: The Scientific Basis at http://www.ipcc.ch/pub/reports.htm John Houghton Global Warming - the complete briefing Cambridge
Some resources (more websites later) Intergovernmental Panel Climate Change 2001: The Scientific Basis at http://www.ipcc.ch/pub/reports.htm John Houghton Global Warming - the complete briefing Cambridge
Climate: Earth s Dynamic Equilibrium
 Climate: Earth s Dynamic Equilibrium review session CCIU April 30, 2016 High-school standard HS-ESS2-4 focuses on the role energy flows play in Earth s climate HS-ESS2-4 Use a model to describe how variations
Climate: Earth s Dynamic Equilibrium review session CCIU April 30, 2016 High-school standard HS-ESS2-4 focuses on the role energy flows play in Earth s climate HS-ESS2-4 Use a model to describe how variations
Chapter 19 Global Change
 Chapter 19 Global Change Global Change Global change- any chemical, biological or physical property change of the planet. Examples include cold temperatures causing ice ages. Global climate change-changes
Chapter 19 Global Change Global Change Global change- any chemical, biological or physical property change of the planet. Examples include cold temperatures causing ice ages. Global climate change-changes
Chapter 19 Global Change
 Chapter 19 Global Change Global Change change - any chemical, biological or physical property change of the planet. Examples include cold temperatures causing ice ages. Global change - changes in the climate
Chapter 19 Global Change Global Change change - any chemical, biological or physical property change of the planet. Examples include cold temperatures causing ice ages. Global change - changes in the climate
Compost and the Clean Development Mechanism (CDM)
 Compost and the Clean Development Mechanism (CDM) ISWA Beacon Conference May 22, 2008 Presentation Topics Solid waste and greenhouse gas emissions CDM and the Kyoto Protocol CDM methodologies Application
Compost and the Clean Development Mechanism (CDM) ISWA Beacon Conference May 22, 2008 Presentation Topics Solid waste and greenhouse gas emissions CDM and the Kyoto Protocol CDM methodologies Application
Introduction. Introduction. Introduction. Outline Last IPCC report : 2001 Last IPCC report :
 Introduction Greenhouse Gases & Climate Change Laurent Bopp LSCE, Paris When did the story start? ¾1827 Fourier hypothesizes greenhouse effect ¾1860 Tyndal identifies CO2 and water vapor as heat trapping
Introduction Greenhouse Gases & Climate Change Laurent Bopp LSCE, Paris When did the story start? ¾1827 Fourier hypothesizes greenhouse effect ¾1860 Tyndal identifies CO2 and water vapor as heat trapping
Chapter 19: Global Change
 1 Summary Of the Case Study Polar Bear population in the Antarctic going down because temperatures are going up and melting the caps. Polar bears are losing their habitat, they also can t get their food
1 Summary Of the Case Study Polar Bear population in the Antarctic going down because temperatures are going up and melting the caps. Polar bears are losing their habitat, they also can t get their food
Greenhouse Gas Protocol Accounting Notes No. 1
 Greenhouse Gas Protocol Accounting Notes No. 1 Accounting and Reporting Standard Amendment February, 2012 Required greenhouse gases for inclusion in corporate and product life cycle This Accounting Note
Greenhouse Gas Protocol Accounting Notes No. 1 Accounting and Reporting Standard Amendment February, 2012 Required greenhouse gases for inclusion in corporate and product life cycle This Accounting Note
Evaluation of Green House Gas Emissions from Proposed Sewerage Treatment Plant in Satna City
 IOSR Journal of Engineering (IOSRJEN) ISSN (e): 2250-3021, ISSN (p): 2278-8719 Vol. 08, Issue 01 (January. 2018), V1 PP 43-50 www.iosrjen.org Evaluation of Green House Gas Emissions from Proposed Sewerage
IOSR Journal of Engineering (IOSRJEN) ISSN (e): 2250-3021, ISSN (p): 2278-8719 Vol. 08, Issue 01 (January. 2018), V1 PP 43-50 www.iosrjen.org Evaluation of Green House Gas Emissions from Proposed Sewerage
Effects of Greenhouse Gas Emission
 Effects of Greenhouse Gas Emission Reshmi Banerjee Assistant Professor, Dept. of EE, Guru Nanak Institute of Technology, Kolkata, W.B., India ABSTRACT: Gases that trap heat in the atmosphere are called
Effects of Greenhouse Gas Emission Reshmi Banerjee Assistant Professor, Dept. of EE, Guru Nanak Institute of Technology, Kolkata, W.B., India ABSTRACT: Gases that trap heat in the atmosphere are called
Carbon Footprint Analyses of Wastewater Treatment Systems in Puducherry
 Computational Water, Energy, and Environmental Engineering, 2017, 6, 281-303 http://www.scirp.org/journal/cweee ISSN Online: 2168-1570 ISSN Print: 2168-1562 Carbon Footprint Analyses of Wastewater Treatment
Computational Water, Energy, and Environmental Engineering, 2017, 6, 281-303 http://www.scirp.org/journal/cweee ISSN Online: 2168-1570 ISSN Print: 2168-1562 Carbon Footprint Analyses of Wastewater Treatment
Global Warming Science Solar Radiation
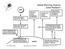 SUN Ozone and Oxygen absorb 190-290 nm. Latent heat from the surface (evaporation/ condensation) Global Warming Science Solar Radiation Turbulent heat from the surface (convection) Some infrared radiation
SUN Ozone and Oxygen absorb 190-290 nm. Latent heat from the surface (evaporation/ condensation) Global Warming Science Solar Radiation Turbulent heat from the surface (convection) Some infrared radiation
Climate Change Potentials of Sanitation options: Nepal Prospective
 Climate Change Potentials of Sanitation options: Nepal Prospective Nam Raj Khatri 1, 1.WHO Nepal ABSTRACT: In Sanitation is very essential need and part of life for human beings. Sanitation and hygiene
Climate Change Potentials of Sanitation options: Nepal Prospective Nam Raj Khatri 1, 1.WHO Nepal ABSTRACT: In Sanitation is very essential need and part of life for human beings. Sanitation and hygiene
Human Activity and Climate Change
 Human Activity and Climate Change Textbook pages 482 501 Section 11.1 11.2 Summary Before You Read How might climate change affect the region where you live? Record your thoughts in the lines below. What
Human Activity and Climate Change Textbook pages 482 501 Section 11.1 11.2 Summary Before You Read How might climate change affect the region where you live? Record your thoughts in the lines below. What
The first Internet portal dedicated to the carbon footprint of water activities
 PRESS FILE www.lifecarbontool.com The first Internet portal dedicated to the carbon footprint of water activities 1. Greenhouse gas (GHG) emissions: situation and challenges 2. Application to the water
PRESS FILE www.lifecarbontool.com The first Internet portal dedicated to the carbon footprint of water activities 1. Greenhouse gas (GHG) emissions: situation and challenges 2. Application to the water
Main Natural Sources of Greenhouse Gases
 Main Natural Sources of Greenhouse Gases Content Atmospheric Composition Composition of the Earth s Atmosphere Greenhouse Gases The Radiative Forcing bar chart: AR5 version Natural Greenhouse Gases Water
Main Natural Sources of Greenhouse Gases Content Atmospheric Composition Composition of the Earth s Atmosphere Greenhouse Gases The Radiative Forcing bar chart: AR5 version Natural Greenhouse Gases Water
Wastewater Pollutants & Treatment Processes. Dr. Deniz AKGÜL Marmara University Department of Environmental Engineering
 Wastewater Pollutants & Treatment Processes Dr. Deniz AKGÜL Marmara University Department of Environmental Engineering Wastewater combination of the liquid or water carried wastes removed from residences,
Wastewater Pollutants & Treatment Processes Dr. Deniz AKGÜL Marmara University Department of Environmental Engineering Wastewater combination of the liquid or water carried wastes removed from residences,
Winter 2009: ATMS/OCN/ESS 588 The Global Carbon Cycle and Greenhouse Gases. Course Goals
 PCC 588 - January 6 and 8 2009 Winter 2009: ATMS/OCN/ESS 588 The Global Carbon Cycle and Greenhouse Gases T,Th 12:00-1:20 pm OSB 25 Course Goals The course focuses on factors controlling the global cycle
PCC 588 - January 6 and 8 2009 Winter 2009: ATMS/OCN/ESS 588 The Global Carbon Cycle and Greenhouse Gases T,Th 12:00-1:20 pm OSB 25 Course Goals The course focuses on factors controlling the global cycle
Climate Change. Climate Change. Global past, present and future. Past-Present-Future
 Climate Change Global past, present and future Climate Change Past-Present-Future Definition Climate change refers to a statistically significant variation in either the mean state of the climate or in
Climate Change Global past, present and future Climate Change Past-Present-Future Definition Climate change refers to a statistically significant variation in either the mean state of the climate or in
WASTEWATER TREATMENT
 WASTEWATER TREATMENT Every community produces both liquid and solid wastes. The liquid portion-wastewater-is essentially the water supply of the community after it has been fouled by a variety of uses.
WASTEWATER TREATMENT Every community produces both liquid and solid wastes. The liquid portion-wastewater-is essentially the water supply of the community after it has been fouled by a variety of uses.
Greenhouse Effect. The Greenhouse Effect
 Greenhouse Effect The Greenhouse Effect Greenhouse gases let short-wavelength radiation come into the Earth s atmosphere from the sun. However, they absorb and re-radiate Earth s long-wavelength radiation
Greenhouse Effect The Greenhouse Effect Greenhouse gases let short-wavelength radiation come into the Earth s atmosphere from the sun. However, they absorb and re-radiate Earth s long-wavelength radiation
Estimation of Carbon dioxide and Methane Emissions Generated from industrial (WWT) plants
 Estimation of Carbon dioxide and Methane Emissions Generated from industrial (WWT) plants Article Info Received: 15 th September 2012 Accepted: 21 th September 2012 Published online: 1st October 2012 Ahmmed
Estimation of Carbon dioxide and Methane Emissions Generated from industrial (WWT) plants Article Info Received: 15 th September 2012 Accepted: 21 th September 2012 Published online: 1st October 2012 Ahmmed
PHY392S Physics of Climate. Lecture 1. Introduction
 PHY392S Physics of Climate Lecture 1 Introduction Slides based on material from Prof. K. Strong PHY392S - Physics of Climate Lecture 1, Page 1 Some Definitions Weather the fluctuating state of the atmosphere
PHY392S Physics of Climate Lecture 1 Introduction Slides based on material from Prof. K. Strong PHY392S - Physics of Climate Lecture 1, Page 1 Some Definitions Weather the fluctuating state of the atmosphere
Selecting municipal wastewater treatment technologies for greenhouse gas emissions reduction: the case of Mexico for year 2030
 Selecting municipal wastewater treatment technologies for greenhouse gas emissions reduction: the case of Mexico for year 2030 Adalberto Noyola Instituto de Ingeniería, Universidad Nacional Autónoma de
Selecting municipal wastewater treatment technologies for greenhouse gas emissions reduction: the case of Mexico for year 2030 Adalberto Noyola Instituto de Ingeniería, Universidad Nacional Autónoma de
Climate Change, Greenhouse Gases and Aerosols
 Climate Change, Greenhouse Gases and Aerosols J Srinivasan J Srinivasan is a Professor at the Centre for Atmospheric and Oceanic Sciences at Indian Institute of Science, Bangalore. He was a lead author
Climate Change, Greenhouse Gases and Aerosols J Srinivasan J Srinivasan is a Professor at the Centre for Atmospheric and Oceanic Sciences at Indian Institute of Science, Bangalore. He was a lead author
ENVIS- IITM NEWSLETTER The Air Quality: A Global Challenge
 ENVIS- IITM NEWSLETTER The Air Quality: A Global Challenge GLOBAL WARMING Editorial Prof. B.N. Goswami (Director, IITM, Pune) Dr. G. Beig (ENVIS Co-ordinetor) Ms. Neha S. Parkhi (Program Officer) Mr. Rajnikant
ENVIS- IITM NEWSLETTER The Air Quality: A Global Challenge GLOBAL WARMING Editorial Prof. B.N. Goswami (Director, IITM, Pune) Dr. G. Beig (ENVIS Co-ordinetor) Ms. Neha S. Parkhi (Program Officer) Mr. Rajnikant
Climate Change and Air Quality
 Climate Change and Air Quality SW PA Air Quality Action June 6, 2007 Peter J. Adams Associate Professor Civil and Environmental Engineering Engineering and Public Policy Outline Climate Change Primer What
Climate Change and Air Quality SW PA Air Quality Action June 6, 2007 Peter J. Adams Associate Professor Civil and Environmental Engineering Engineering and Public Policy Outline Climate Change Primer What
UNFCCC/CCNUCC. CDM Executive Board III.H./Version 4 Scope 13, 15 Page 1 EB 28
 Page 1 TYPE III - OTHER PROJECT ACTIVITIES All the approved small-scale methodologies, general guidance to the methodologies, information on additionality and abbreviations can be found at: http://cdm.unfccc.int/methodologies/sscmethodologies/approved.html
Page 1 TYPE III - OTHER PROJECT ACTIVITIES All the approved small-scale methodologies, general guidance to the methodologies, information on additionality and abbreviations can be found at: http://cdm.unfccc.int/methodologies/sscmethodologies/approved.html
Environmental Engineering Atmosphere & pollution 2
 Environmental Engineering Atmosphere & pollution 2 Global radiation Greenhouse effect Kyoto protocol David Zumr Dpt. of Drainage, Irrigation and Landscape Eng. 1/ insolation from the Sun Electromagnetic
Environmental Engineering Atmosphere & pollution 2 Global radiation Greenhouse effect Kyoto protocol David Zumr Dpt. of Drainage, Irrigation and Landscape Eng. 1/ insolation from the Sun Electromagnetic
UNFCCC/CCNUCC. CDM Executive Board III.H./Version 05.1 Sectoral Scope: 13 EB 32
 TYPE III - OTHER PROJECT ACTIVITIES Project participants shall take into account the general guidance to the methodologies, information on additionality, abbreviations and general guidance on leakage provided
TYPE III - OTHER PROJECT ACTIVITIES Project participants shall take into account the general guidance to the methodologies, information on additionality, abbreviations and general guidance on leakage provided
Global Warming & Climate Change Review Learning Target 1
 Learning Target 1 Click and drag the word to fit the proper description. Word Ice Off Dates Ice Core Samples Description 1.Scientist can check the gases in the bubbles and provide a good estimate of the
Learning Target 1 Click and drag the word to fit the proper description. Word Ice Off Dates Ice Core Samples Description 1.Scientist can check the gases in the bubbles and provide a good estimate of the
UNFCCC/CCNUCC. CDM Executive Board III.H./Version 4 Scope 13, 15 Page 1 EB 28
 Page 1 TYPE III - OTHER PROJECT ACTIVITIES All the approved small-scale methodologies, general guidance to the methodologies, information on additionality and abbreviations can be found at: http://cdm.unfccc.int/methodologies/sscmethodologies/approved.html
Page 1 TYPE III - OTHER PROJECT ACTIVITIES All the approved small-scale methodologies, general guidance to the methodologies, information on additionality and abbreviations can be found at: http://cdm.unfccc.int/methodologies/sscmethodologies/approved.html
Industrial Waste Water Treatment. Unit 5
 Industrial Waste Water Treatment Unit 5 Outline Levels of treatment methods 1 Biological wastewater treatment Caste study Heavy metals Biological wastewater treatment Treatment Methods employed Biological
Industrial Waste Water Treatment Unit 5 Outline Levels of treatment methods 1 Biological wastewater treatment Caste study Heavy metals Biological wastewater treatment Treatment Methods employed Biological
How Can Thermal Effects Be Explained?
 How Can Thermal Effects Be Explained? Lesson 6, Part 3: Climate Science The Enhanced Greenhouse Effect The Earth will maintain equilibrium (constant stable temperature level) if the energy coming in is.
How Can Thermal Effects Be Explained? Lesson 6, Part 3: Climate Science The Enhanced Greenhouse Effect The Earth will maintain equilibrium (constant stable temperature level) if the energy coming in is.
Global warming. Models for global warming Sand analogy
 8.10 Global warming Assessment statements 8.6.1 Describe some possible models of global warming. 8.6. State what is meant by the enhanced greenhouse effect. 8.6.3 Identify the increased combustion of fossil
8.10 Global warming Assessment statements 8.6.1 Describe some possible models of global warming. 8.6. State what is meant by the enhanced greenhouse effect. 8.6.3 Identify the increased combustion of fossil
Tananyag fejlesztés idegen nyelven
 Tananyag fejlesztés idegen nyelven Prevention of the atmosphere KÖRNYEZETGAZDÁLKODÁSI AGRÁRMÉRNÖKI MSC (MSc IN AGRO-ENVIRONMENTAL STUDIES) Calculation of greenhouse effect. The carbon cycle Lecture 11
Tananyag fejlesztés idegen nyelven Prevention of the atmosphere KÖRNYEZETGAZDÁLKODÁSI AGRÁRMÉRNÖKI MSC (MSc IN AGRO-ENVIRONMENTAL STUDIES) Calculation of greenhouse effect. The carbon cycle Lecture 11
Discussion about Cause of Nitrous Oxide (N 2 O) Emission in Wastewater Treatment Plant, Based on Long-Term Continuous Measurement
 Discussion about Cause of Nitrous Oxide (N 2 O) Emission in Wastewater Treatment Plant, Based on Long-Term Continuous Measurement Tokyo Metropolitan Government, the Bureau of Sewerage Kiyoaki Kitamura
Discussion about Cause of Nitrous Oxide (N 2 O) Emission in Wastewater Treatment Plant, Based on Long-Term Continuous Measurement Tokyo Metropolitan Government, the Bureau of Sewerage Kiyoaki Kitamura
Carbon Heat Energy Assessment and. (CHEApet) Tutorials: Carbon Footprint Primer
 Carbon Heat Energy Assessment and Plant Evaluation Tool (CHEApet) Tutorials: Carbon Footprint Primer Tutorials Sources of Greenhouse Gases (GHG) at WWTPs How does CHEApet measure Carbon Footprint? Comparing
Carbon Heat Energy Assessment and Plant Evaluation Tool (CHEApet) Tutorials: Carbon Footprint Primer Tutorials Sources of Greenhouse Gases (GHG) at WWTPs How does CHEApet measure Carbon Footprint? Comparing
GEOG 401: Week 2 Earth s Energy Balance
 GEOG 401: Week 2 Earth s Energy Balance Dr. John Abatzoglou Spring 2011 Lecture Goals (1) Understand the global balance of energy and associated fluxes of energy and energy types (2) Qualita@ve and quan@ta@ve
GEOG 401: Week 2 Earth s Energy Balance Dr. John Abatzoglou Spring 2011 Lecture Goals (1) Understand the global balance of energy and associated fluxes of energy and energy types (2) Qualita@ve and quan@ta@ve
Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32)
 Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32) METR 113/ENVS 113 Spring Semester 2011 February 15, 2011 Suggested Reading
Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32) METR 113/ENVS 113 Spring Semester 2011 February 15, 2011 Suggested Reading
T8-1 [166 marks] Which energy resource is renewable? A. Natural gas B. Uranium C. Biogas D. Coal
![T8-1 [166 marks] Which energy resource is renewable? A. Natural gas B. Uranium C. Biogas D. Coal T8-1 [166 marks] Which energy resource is renewable? A. Natural gas B. Uranium C. Biogas D. Coal](/thumbs/78/78599564.jpg) T8-1 [166 marks] 1. Which energy resource is renewable? A. Natural gas B. Uranium C. Biogas D. Coal 2. For a black-body at absolute temperature T the power emitted per unit area is P. What is the power
T8-1 [166 marks] 1. Which energy resource is renewable? A. Natural gas B. Uranium C. Biogas D. Coal 2. For a black-body at absolute temperature T the power emitted per unit area is P. What is the power
GLOBAL Energy Flow Thru Atmosphere
 GLOBAL Energy Flow Thru Atmosphere Global Atmo Energy Balance In a stable climate, Solar Energy IN = IR Energy OUT IR Out Ahrens, Fig. 2.14 Solar in The Natural Greenhouse Effect: clear sky O 3 8% CH 4
GLOBAL Energy Flow Thru Atmosphere Global Atmo Energy Balance In a stable climate, Solar Energy IN = IR Energy OUT IR Out Ahrens, Fig. 2.14 Solar in The Natural Greenhouse Effect: clear sky O 3 8% CH 4
Philippines GHG Emissions : Waste Sector. Teresita R. Perez, Ph.D. Ateneo de Manila University
 Philippines GHG Emissions : Waste Sector Teresita R. Perez, Ph.D. Ateneo de Manila University Rapid urbanization is positively correlated with the increase in the consumption and production of goods Leads
Philippines GHG Emissions : Waste Sector Teresita R. Perez, Ph.D. Ateneo de Manila University Rapid urbanization is positively correlated with the increase in the consumption and production of goods Leads
2. Climate Change: Projections of Climate Change: 2100 and beyond
 Global Warming: Science, Projections and Uncertainties Global Warming: Science, Projections and Uncertainties An overview of the basic science An overview of the basic science 1. A Brief History of Global
Global Warming: Science, Projections and Uncertainties Global Warming: Science, Projections and Uncertainties An overview of the basic science An overview of the basic science 1. A Brief History of Global
CHAPTER 16 Oceans & Climate Change Chapter Overview Earth s Climate System Earth s Climate System Earth s Climate System Earth s Climate System
 1 CHAPTER 16 2 3 4 5 6 7 8 9 10 11 12 Oceans & Climate Change Chapter Overview Humans are adding greenhouse gases to Earth s atmosphere. Climate change will cause many severe problems in the ocean environment.
1 CHAPTER 16 2 3 4 5 6 7 8 9 10 11 12 Oceans & Climate Change Chapter Overview Humans are adding greenhouse gases to Earth s atmosphere. Climate change will cause many severe problems in the ocean environment.
Feedback loops modify atmospheric processes
 Chapter Overview CHAPTER 16 Oceans & Climate Change Humans are adding greenhouse gases to Earth s atmosphere. Climate change will cause many severe problems in the ocean environment. It is necessary to
Chapter Overview CHAPTER 16 Oceans & Climate Change Humans are adding greenhouse gases to Earth s atmosphere. Climate change will cause many severe problems in the ocean environment. It is necessary to
Radiative forcing of gases, aerosols and, clouds.
 Lecture 23. Radiative forcing of gases, aerosols and, clouds. 1. Concepts of radiative forcing, climate sensitivity, and radiation feedbacks. 2. Radiative forcing of anthropogenic greenhouse gases. 3.
Lecture 23. Radiative forcing of gases, aerosols and, clouds. 1. Concepts of radiative forcing, climate sensitivity, and radiation feedbacks. 2. Radiative forcing of anthropogenic greenhouse gases. 3.
TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition
 TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition There s one more thing to correct in our the depiction of incoming Solar....... the atmosphere is NOT totally TRANSPARENT to INCOMING Solar
TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition There s one more thing to correct in our the depiction of incoming Solar....... the atmosphere is NOT totally TRANSPARENT to INCOMING Solar
G R E E N H O U S E G A S M I T I G A T I O N W A S T E SECTOR
 Overview of Sector Vietnam s urban population is growing 4.4 percent annually, industry is growing 17 percent annually, and the agriculture sector continues its steady growth. As a result, Vietnam produces
Overview of Sector Vietnam s urban population is growing 4.4 percent annually, industry is growing 17 percent annually, and the agriculture sector continues its steady growth. As a result, Vietnam produces
Area-wide Emissions. Briefing Note 1: Introduction to Greenhouse Gas Estimation May In partnership with
 May 2014 In partnership with Guidance Guidance Note 1: Introduction to GHG Estimation Sustainable Scotland Network (SSN) supports public sector action on sustainable development. This includes programmes
May 2014 In partnership with Guidance Guidance Note 1: Introduction to GHG Estimation Sustainable Scotland Network (SSN) supports public sector action on sustainable development. This includes programmes
Necessity of Global Warming Projection Information
 TCC Training Seminar on Global Warming Projection Information 11:00 12:45 on 26 January 2015 Necessity of Global Warming Projection Information Koji Ishihara Hirokazu Murai Nubuyuki Kayaba Souichirou Yasui
TCC Training Seminar on Global Warming Projection Information 11:00 12:45 on 26 January 2015 Necessity of Global Warming Projection Information Koji Ishihara Hirokazu Murai Nubuyuki Kayaba Souichirou Yasui
KEY WORDS Anaerobic lagoons ; reaction constant ; operation ; per capita sludge ; loading rate.
 The Operation of Anaerobic Lagoons A. Gheisari : Lecturer Mohajer Technical School, and senior Scientist, Water & Wastewater Consulting Engineers, Isfahan, Iran M.A. Kazemi: Executive Manager, Water &
The Operation of Anaerobic Lagoons A. Gheisari : Lecturer Mohajer Technical School, and senior Scientist, Water & Wastewater Consulting Engineers, Isfahan, Iran M.A. Kazemi: Executive Manager, Water &
Chapter 2. Climate Change: Scientific Basis
 a. The Greenhouse Effect Chapter 2 Climate Change: Scientific Basis Climate scientists have clearly established that: The Earth s atmosphere is like a greenhouse, reflecting some of the sun s harmful rays
a. The Greenhouse Effect Chapter 2 Climate Change: Scientific Basis Climate scientists have clearly established that: The Earth s atmosphere is like a greenhouse, reflecting some of the sun s harmful rays
LIVING IN THE ENVIRONMENT, 18e G. TYLER MILLER SCOTT E. SPOOLMAN. Climate Disruption. Cengage Learning 2015
 LIVING IN THE ENVIRONMENT, 18e G. TYLER MILLER SCOTT E. SPOOLMAN 19 Climate Disruption 19-1 How Is the Earth s Climate Changing? Considerable scientific evidence indicates that the earth s atmosphere is
LIVING IN THE ENVIRONMENT, 18e G. TYLER MILLER SCOTT E. SPOOLMAN 19 Climate Disruption 19-1 How Is the Earth s Climate Changing? Considerable scientific evidence indicates that the earth s atmosphere is
Wastewater in Ontario: The ECO's perspective on onsite and decentralized wastewater management
 Wastewater in Ontario: The ECO's perspective on onsite and decentralized wastewater management Michelle Kassel Senior Manager, Legislative Analysis Environmental Commissioner of Ontario April 17, 2018
Wastewater in Ontario: The ECO's perspective on onsite and decentralized wastewater management Michelle Kassel Senior Manager, Legislative Analysis Environmental Commissioner of Ontario April 17, 2018
Chapter 4: The Global Energy System
 Discovering Physical Geography Third Edition by Alan Arbogast Chapter 4: The Global Energy System The Electromagnetic Spectrum and Solar Energy Solar Energy as Radiation Electromagnetic energy transmitted
Discovering Physical Geography Third Edition by Alan Arbogast Chapter 4: The Global Energy System The Electromagnetic Spectrum and Solar Energy Solar Energy as Radiation Electromagnetic energy transmitted
Essentials of Oceanography Eleventh Edition
 Chapter Chapter 1 16 Clickers Lecture Essentials of Oceanography Eleventh Edition The Oceans and Climate Change Alan P. Trujillo Harold V. Thurman Chapter Overview Humans are adding greenhouse gases to
Chapter Chapter 1 16 Clickers Lecture Essentials of Oceanography Eleventh Edition The Oceans and Climate Change Alan P. Trujillo Harold V. Thurman Chapter Overview Humans are adding greenhouse gases to
Global Climate Change
 Global Climate Change MODULE 11: GLOBAL CLIMATE CHANGE UNIT 1: BIODIVERSITY Objectives Define terms. Understand global climate change. Describe the basic predictions of the global climate models. Understand
Global Climate Change MODULE 11: GLOBAL CLIMATE CHANGE UNIT 1: BIODIVERSITY Objectives Define terms. Understand global climate change. Describe the basic predictions of the global climate models. Understand
Water Challenges - Past, Present, and Future. Perry L. McCarty Department of Civil and Environmental Engineering Stanford University
 Water Challenges - Past, Present, and Future Perry L. McCarty Department of Civil and Environmental Engineering Stanford University Water Supply and Distribution Listed by the National Academy of Engineering
Water Challenges - Past, Present, and Future Perry L. McCarty Department of Civil and Environmental Engineering Stanford University Water Supply and Distribution Listed by the National Academy of Engineering
Climate Change: The Debate
 Climate Change: The Debate Key Concepts: Greenhouse Gas Carbon dioxide Fossil fuels Greenhouse effect Greenhouse gases Methane Nitrous oxides Radiative forcing WHAT YOU WILL LEARN 1. You will learn about
Climate Change: The Debate Key Concepts: Greenhouse Gas Carbon dioxide Fossil fuels Greenhouse effect Greenhouse gases Methane Nitrous oxides Radiative forcing WHAT YOU WILL LEARN 1. You will learn about
Radiative forcing of climate change
 Radiative forcing of climate change Joanna D. Haigh Imperial College of Science, Technology and Medicine, London Radiative forcing concept, definition and applications On a global and annual average, and
Radiative forcing of climate change Joanna D. Haigh Imperial College of Science, Technology and Medicine, London Radiative forcing concept, definition and applications On a global and annual average, and
CHAPTER 19. Global Change
 CHAPTER 19 Global Change WALKING ON THIN ICE Polar bears play important role in North Pole ecosystem Food seals Important as food and fur for clothing source for indigenous people Problem temperatures
CHAPTER 19 Global Change WALKING ON THIN ICE Polar bears play important role in North Pole ecosystem Food seals Important as food and fur for clothing source for indigenous people Problem temperatures
The Greenhouse Effect
 Name: Date: The Greenhouse Effect This document provides an overview of the earth's atmospheric "greenhouse effect" by briefly exploring the atmospheres of nearby planets and discussing our atmosphere's
Name: Date: The Greenhouse Effect This document provides an overview of the earth's atmospheric "greenhouse effect" by briefly exploring the atmospheres of nearby planets and discussing our atmosphere's
Draft Methodological Tool. Project and leakage emissions from anaerobic digesters. (Version )
 Page 1 Draft Methodological Tool Project and leakage emissions from anaerobic digesters (Version 01.0.0) I. DEFINITIONS, SCOPE, APPLICABILITY AND PARAMETERS Definitions For the purpose of this tool, the
Page 1 Draft Methodological Tool Project and leakage emissions from anaerobic digesters (Version 01.0.0) I. DEFINITIONS, SCOPE, APPLICABILITY AND PARAMETERS Definitions For the purpose of this tool, the
Climate Change. Black-Body Radiation. Factors that affect how an object absorbs, emits (radiates), and reflects EM radiation incident on them:
 Climate Change Black-Body Radiation Factors that affect how an object absorbs, emits (radiates), and reflects EM radiation incident on them: 1) Nature of the surface: material, shape, texture, etc. 2)
Climate Change Black-Body Radiation Factors that affect how an object absorbs, emits (radiates), and reflects EM radiation incident on them: 1) Nature of the surface: material, shape, texture, etc. 2)
Wastewater treatment objecives
 Wastewater treatment objecives Removal of suspended and floatable materials Degradation of biodegradable organics Removal of nutrients Elimination of priority pollutants Elimination of pathogenic organisms
Wastewater treatment objecives Removal of suspended and floatable materials Degradation of biodegradable organics Removal of nutrients Elimination of priority pollutants Elimination of pathogenic organisms
Radiative Forcing Components
 Radiative Forcing Components Content Definition of Radiative Forcing Radiation Balance Climate sensitivity Solar forcing Forcing due to atmospheric gas Definition of Radiative Forcing In climate science,
Radiative Forcing Components Content Definition of Radiative Forcing Radiation Balance Climate sensitivity Solar forcing Forcing due to atmospheric gas Definition of Radiative Forcing In climate science,
Greenhouse Gas Emissions and Reduction Stratagems from Waste Sector in India
 Greenhouse Gas Emissions and Reduction Stratagems from Waste Sector in India Sourabh Manuja 1*, Atul Kumar 2, Suneel Pandey 3 1 Associate fellow and Area Convener, Center for waste management, The Energy
Greenhouse Gas Emissions and Reduction Stratagems from Waste Sector in India Sourabh Manuja 1*, Atul Kumar 2, Suneel Pandey 3 1 Associate fellow and Area Convener, Center for waste management, The Energy
Greenhouse gases. A snow-covered surface refl ects massive amounts of sunlight and therefore has a cooling effect on the climate.
 A k t u e l N a t u r v i d e n s k a b 2 0 0 9 G R E E N H O U S E G A S E S 13 Greenhouse gases - and their impact on the climate The greenhouse effect is the best understood and well mapped of the mechanisms
A k t u e l N a t u r v i d e n s k a b 2 0 0 9 G R E E N H O U S E G A S E S 13 Greenhouse gases - and their impact on the climate The greenhouse effect is the best understood and well mapped of the mechanisms
Welcome to ATMS 111 Global Warming.
 Welcome to ATMS 111 Global Warming http://www.atmos.washington.edu/2010q1/111 Censored Spotted by Jennifer Le Today Review and Finish up The Greenhouse Effect - RG p 21-30 A rogues gallery of greenhouse
Welcome to ATMS 111 Global Warming http://www.atmos.washington.edu/2010q1/111 Censored Spotted by Jennifer Le Today Review and Finish up The Greenhouse Effect - RG p 21-30 A rogues gallery of greenhouse
Point source pollution and climate change impact from sequential batch reactor wastewater treatment plant
 Global NEST Journal, Vol 20, No 1, pp 33-41 Copyright 2018 Global NEST Printed in Greece. All rights reserved Point source pollution and climate change impact from sequential batch reactor wastewater treatment
Global NEST Journal, Vol 20, No 1, pp 33-41 Copyright 2018 Global NEST Printed in Greece. All rights reserved Point source pollution and climate change impact from sequential batch reactor wastewater treatment
Fact sheet 18 June Selecting and Using GWP values for Refrigerants
 Selecting and Using GWP values for Refrigerants Summary and Recommendations Refrigerant GWP values have been revised with each new IPCC Assessment Report. Regulations and guidance for the use of GWP values
Selecting and Using GWP values for Refrigerants Summary and Recommendations Refrigerant GWP values have been revised with each new IPCC Assessment Report. Regulations and guidance for the use of GWP values
UN Climate Council Words in red are defined in vocabulary section (pg. 9)
 UN Climate Council Words in red are defined in vocabulary section (pg. 9) To minimize the negative effects of global climate change, scientists have advocated for action to limit global warming to no more
UN Climate Council Words in red are defined in vocabulary section (pg. 9) To minimize the negative effects of global climate change, scientists have advocated for action to limit global warming to no more
(First Earth Summit-1992)
 Summits related to Climate Change India launched an international solar alliance of over 120 countries (Completely or partially between tropic of cancer and Capricorn) at the Paris COP21 climate summit.
Summits related to Climate Change India launched an international solar alliance of over 120 countries (Completely or partially between tropic of cancer and Capricorn) at the Paris COP21 climate summit.
CANADA-WIDE APPROACH FOR THE MANAGEMENT OF WASTEWATER BIOSOLIDS. October 11, 2012 PN 1477
 CANADA-WIDE APPROACH FOR THE MANAGEMENT OF WASTEWATER BIOSOLIDS October 11, 2012 PN 1477 Canadian Council of Ministers of the Environment, 2012 Table of Contents 1. Context... 1 2. Policy Statement and
CANADA-WIDE APPROACH FOR THE MANAGEMENT OF WASTEWATER BIOSOLIDS October 11, 2012 PN 1477 Canadian Council of Ministers of the Environment, 2012 Table of Contents 1. Context... 1 2. Policy Statement and
Chapter 43 Ecosystems & Human Interferences
 How do organisms obtain energy from their environment? Autotrophs produce their own food Heterotrophs feed on other organisms 1. Herbivores Consume plants 2. Carnivores Consume other animals 3. Omnivores
How do organisms obtain energy from their environment? Autotrophs produce their own food Heterotrophs feed on other organisms 1. Herbivores Consume plants 2. Carnivores Consume other animals 3. Omnivores
Wastewater Treatment clarifier
 Wastewater Treatment Pretreatment During pretreatment, items would normally be removed that would hinder the further processes of treatment. Items commonly removed include roots, rags, cans, or other large
Wastewater Treatment Pretreatment During pretreatment, items would normally be removed that would hinder the further processes of treatment. Items commonly removed include roots, rags, cans, or other large
1. The diagram below shows a greenhouse.
 1. The diagram below shows a greenhouse. 5. A gradual increase in atmospheric carbon dioxide would warm Earth s because carbon dioxide is a A) poor reflector of ultraviolet radiation B) good reflector
1. The diagram below shows a greenhouse. 5. A gradual increase in atmospheric carbon dioxide would warm Earth s because carbon dioxide is a A) poor reflector of ultraviolet radiation B) good reflector
Why is carbon dioxide so important? Examining the evidence
 Why is carbon dioxide so important? Examining the evidence In the light of new evidence and taking into account the remaining uncertainties, most of the observed warming over the last 50 years is likely
Why is carbon dioxide so important? Examining the evidence In the light of new evidence and taking into account the remaining uncertainties, most of the observed warming over the last 50 years is likely
Municipal wastewater treatment in Latin-America:
 Municipal wastewater treatment in Latin-America: A zoom to Mexico with emphasis in GHG emissions from municipal wastewater treatment centro mario molina Adalberto Noyola Instituto de Ingeniería Universidad
Municipal wastewater treatment in Latin-America: A zoom to Mexico with emphasis in GHG emissions from municipal wastewater treatment centro mario molina Adalberto Noyola Instituto de Ingeniería Universidad
19 March Reyer Gerlagh, Tilburg University Professor of Economics
 19 March 2013 Reyer Gerlagh, Tilburg University Professor of Economics Overview History The discovery of global warming The greenhouse effect: basics All greenhouse gases Emissions: population, income,
19 March 2013 Reyer Gerlagh, Tilburg University Professor of Economics Overview History The discovery of global warming The greenhouse effect: basics All greenhouse gases Emissions: population, income,
3/5/2012. Study Questions. Global Warming. Weather: daily temperature and moisture conditions Climate: long-term weather patterns
 Study Questions Global Warming 1. Explain what is meant by the term Greenhouse effect. 2. List 3 indications that global climate is currently increasing. 3. Describe 3 possible consequences of global warming.
Study Questions Global Warming 1. Explain what is meant by the term Greenhouse effect. 2. List 3 indications that global climate is currently increasing. 3. Describe 3 possible consequences of global warming.
Climate Dynamics (PCC 587): Climate Forcings
 Climate Dynamics (PCC 587): Climate Forcings DARGAN M. W. FRIERSON UNIVERSITY OF WASHINGTON, DEPARTMENT OF ATMOSPHERIC SCIENCES DAY 7: 10-16-13 Outline of This Topic Climate forcings Things that directly
Climate Dynamics (PCC 587): Climate Forcings DARGAN M. W. FRIERSON UNIVERSITY OF WASHINGTON, DEPARTMENT OF ATMOSPHERIC SCIENCES DAY 7: 10-16-13 Outline of This Topic Climate forcings Things that directly
What is the Greenhouse Gas Contribution from Agriculture in Alberta?
 What is the Greenhouse Gas Contribution from Agriculture in Alberta? The most recent GHG inventory estimated that in 2002 nationwide, agricultural related GHG emissions contributed about 59,000 kt (kilotonnnes)
What is the Greenhouse Gas Contribution from Agriculture in Alberta? The most recent GHG inventory estimated that in 2002 nationwide, agricultural related GHG emissions contributed about 59,000 kt (kilotonnnes)
08 Energy, Power and climate change review answers
 08 Energy, Power and climate change review answers Power generation 1. Copy and complete: Thermal energy may be completely converted into work in a single process such as the adiabatic expansion of a gas
08 Energy, Power and climate change review answers Power generation 1. Copy and complete: Thermal energy may be completely converted into work in a single process such as the adiabatic expansion of a gas
GLOBAL WARMING COMPUTER LAB
 GLOBAL WARMING COMPUTER LAB A COMPUTER SIMULATION PROGRAM ON TEMPERATURE CHANGE AND SEA LEVEL RISING After performing this computer simulation lab you will be able to: 1) understand the greenhouse effect
GLOBAL WARMING COMPUTER LAB A COMPUTER SIMULATION PROGRAM ON TEMPERATURE CHANGE AND SEA LEVEL RISING After performing this computer simulation lab you will be able to: 1) understand the greenhouse effect
Water Pollution. Objective: Name, describe, and cite examples of the eight major types of water pollution.
 Water Pollution Objective: Name, describe, and cite examples of the eight major types of water pollution. Types of Water Pollution Water pollutants are divided into eight categories: 1. Sediment pollution
Water Pollution Objective: Name, describe, and cite examples of the eight major types of water pollution. Types of Water Pollution Water pollutants are divided into eight categories: 1. Sediment pollution
Lecture 22: Atmospheric Chemistry and Climate
 Lecture 22: Atmospheric Chemistry and Climate Required Reading: FP Chapter 14 (only sections that I cover) Suggested Introductory Reading: Jacob Chapter 7 Atmospheric Chemistry CHEM-5151 / ATOC-5151 Spring
Lecture 22: Atmospheric Chemistry and Climate Required Reading: FP Chapter 14 (only sections that I cover) Suggested Introductory Reading: Jacob Chapter 7 Atmospheric Chemistry CHEM-5151 / ATOC-5151 Spring
OPPORTUNITIES FOR MITIGATION OF METHANE EMISSIONS IN CHINA
 PRESENTATION FOR THE STEP SEMINAR SERIES FEBRUARY 11, 2013 PRINCETON, NJ OPPORTUNITIES FOR MITIGATION OF METHANE EMISSIONS IN CHINA Presenters: Shannon Brink, Harrison Godfrey, Mary Kang, Shelly Lyser,
PRESENTATION FOR THE STEP SEMINAR SERIES FEBRUARY 11, 2013 PRINCETON, NJ OPPORTUNITIES FOR MITIGATION OF METHANE EMISSIONS IN CHINA Presenters: Shannon Brink, Harrison Godfrey, Mary Kang, Shelly Lyser,
Sanitary Sewer Systems. Sewage Collection System. Types of Sewage 10/12/2016. General Overview
 Sanitary Sewer Systems General Overview Sewage Collection System Pipes Pumping stations Maintenance entry points manholes Types of Sewage Sanitary Domestic sewage: human wastes and washwater from public
Sanitary Sewer Systems General Overview Sewage Collection System Pipes Pumping stations Maintenance entry points manholes Types of Sewage Sanitary Domestic sewage: human wastes and washwater from public
The Chemistry of Climate Change. Reading: Chapter 8 Environmental Chemistry, G. W. vanloon. S. J. Duffy
 The Chemistry of Climate Change Reading: Chapter 8 Environmental Chemistry, G. W. vanloon. S. J. Duffy The Science of Global Climate There's a lot of differing data, but as far as I can gather, over the
The Chemistry of Climate Change Reading: Chapter 8 Environmental Chemistry, G. W. vanloon. S. J. Duffy The Science of Global Climate There's a lot of differing data, but as far as I can gather, over the
Chapter Overview. Earth s Climate System. Earth s Climate System. Earth s Climate System. CHAPTER 16 The Oceans and Climate Change
 Chapter Overview CHAPTER 16 The Oceans and Climate Humans are adding greenhouse gases to Earth s atmosphere. Climate change will cause many severe problems in the ocean environment. It is necessary to
Chapter Overview CHAPTER 16 The Oceans and Climate Humans are adding greenhouse gases to Earth s atmosphere. Climate change will cause many severe problems in the ocean environment. It is necessary to
