The Nutrient Cycle. Atmospheric pool. Organic material. 5 Soil solution storage
|
|
|
- Rebecca Burke
- 6 years ago
- Views:
Transcription
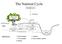
1 The Nutrient Cycle Atmospheric pool Precipitation Canopy, wood, and root Litter fall SOIL 2 Soil and rock minerals cations Groundwater level Organic material 5 Soil solution storage 4 4 Channel BEDROCK 1 Cation exchange 4 Leaching 2 H + and exudates 5 Acids, chelates, nutrients 3 Nutrients
2 Nutrients Distribution in Soils Source: Jobbagy, EG, and RB Jackson The distribution of soil nutrients with depth: Global patterns and the inprint of plants. Biogeochemistry 53:
3 Litter Production Above ground production is easy to measure. Below ground production is very difficult to assess because of the: Short life and rapid root turnover (duration of root growth). Most roots live less than one year. Difficulty in quantifying rates of root exudation.
4 Nutrient Cycling in Soils and Vegetation Biome Total biomass Mineral elements in biomass Net primary production Net mineral uptake Total litter fall Minerals returned in litter (kg ha -1 ) (kg ha -1 y -1 ) Tropical rainforest 517,000 11,081 34,200 2,029 27,500 1,540 (76%) Forest (Central Europe) 370,000 4,196 13, , (71%) Northern taiga 260, , , (85%) Dry savanna 26, , , (98%) Artic tundra 5, , , (97%) Source: Chorley, RJ, SA Schumm, DE Sugden Geomorphology. Methuen
5 Vertical Distribution of Nutrients Source: Jobbagy, EG, and RB Jackson The distribution of soil nutrients with depth: Global patterns and the inprint of plants. Biogeochemistry 53:
6 Figure Distribution of organic carbon in four soil profiles, two well drained and two poorly drained. Poor drainage results in higher organic carbon content, particularly in the surface horizon.
7 Soil Organic Matter in Soil Amount in mineral soil (w/w) range in A horizon % for North Jersey % for South Jersey % subsoils have less: % Organic Soils > 20%
8 Classification of SOM Soil Organic Matter Living Organisms: BIOMASS Indentifiable dead tissue: DETRITUS Non-living, Non-tissue: HUMUS Humic Substances NonHumic substances
9
10 Humus Complex organic substances in soil not identifiable as organic tissue Amorphous, colloidal non-humic compounds identifiable biomolecules humic compounds product of decomposition + synthesis (polymerization) relatively stable, resistant to further breakdown
11 A Model of SOM Source: modified from
12 Functional Groups in SOM Carboxyl groups: -COOH Phenolic groups: -ArOH Proteinaceous material Alcoholic groups: -ROH Saccharides (sugars) Water Source: modified from
13 Humic Substances Random, complex polymers resistant to breakdown Resulting composition C 50-60% N 5% C:N ratio=10:1 P % S 0.5% Solubility classification Humin, Humic Acids, and Fulvic Acids
14 From Schlesinger, W.H Biogeochemistry. Academic Press.
15 Humin and Fulvic and Humic Acids Component Residence time, y C,% N, % Molecular mass Climate Fulvic Acid (FA) 1,800-4, low Cool, temperate Humic Acid (HA) 1,900 5, high warm Humin 2,900 3,500 > high The long residence time of FA and HA contributes to the memory of a soil. Fulvic acids are soluble in water and are transported to deeper horizons along plant roots. Humic acids form strong complexes with Al, Ca, Mg, and Fe. Chatsworth, NJ
16 Simplified Carbon Cycle Photosynthesis: Numbers represent Pg (10 15 g) of carbon stored in the respective pools. Plants + CO 2 = Organic Molecules + O 2 Respiration: Organic Molecules + O 2 = Humus + CO 2
17 Estimates of Active N Pools Medium Air Form Pg N N 2 N 2 O 3,900, Land Plant Animals SOM ,500 Sea Plant Animals Solution or suspension Dissolved N ,200 22,000
18 The Nitrogen Cycle Figure 12.1
19 Carbon and Nitrogen Balances Soil C and N storage (pools) are in general balanced. Exceptions to this are: Peat bogs (accumulation of carbon). Northern peatlands contain ~30% of the global storage of SOC. Extreme deserts (accumulation of nitrogen) The result is that the amounts change rapidly over limited spans of time and then stabilize (steady state) at levels characteristic of climate, topography, etc.
20
21 Soil Carbon vs. Climate Soil C increases with Mean Annual Precipitation (MAP) and decreases with Mean Annual Temperature (MAT). Pattern is due to balance of inputs and losses as influenced by climate. Source: Amundson, R The Carbon Budget of Soils. Annu. Rev. Earth Planet. Sci :535 62
22
23 US Carbon Budget: Land Use Change Annual net sources and sinks of carbon resulting from different types of land use in the United States. Source: Houghton, RA, JL Hackler, and KT Lawrence The U.S. Carbon Budget: Contributions from Land-Use change. Science 285:
24 In the News: April 2008
25 Carbon Losses Carbon was lost from soils across England and Wales over the period at a mean rate of 0.6% yr -1 (relative to the existing soil carbon content), reaching 2% yr -1 in soils with a carbon content greater than 10%. Source: Bellamy, P. H., P. J. Loveland, R. I. Bradley, R. M. Lark, and G. J. D. Kirk Carbon losses from all soils across England and Wales Nature 437: doi: /nature04038.
26 Carbon Content of Different Soil Groups Soil Group Temperate Forest Temperate Grassland Tropical Forest Tropical Grassland Shallow/saline/arid Wetlands/paddy Histosols Andosols TOTAL C mass virgin (Pg C) C mass cultivated (Pg C) Historic loss ( ) Pg C
27 Anthropogenic Alteration to the N Cycle The main source of alteration to the N cycle derives from the application of fertilizers, which shows the most dramatic rate of increase over the last 40 years. Source: Vitousek, PM et al Human alteration of the global nitrogen cycle: Sources and consequences. Ecological Applications 7:
28 Trends in Fertilizer Use Source: Tilman et al., Forecasting agriculturally driven global environmental change. Science 292:
29 Consequences of N Cycle Alteration According to Vitousek et al. (1996) human alteration of the N cycle resulted in: Approximately doubled the rate of input to the terrestrial N cycle, Increased the concentration of N x O gases in the atmosphere, Contributed to the acidification of soils, stream and lakes Increased the quantity of organic C.
30 What is the Effect of Increased N in the Ecosystems? Terrestrial ecosystems: Accumulation in degraded soils. Influences the accumulation (storage) of carbon. Loss in biodiversity In N-saturated systems, N is lost to groundwater (problem in southern NJ), streams, estuaries (eutrophication) and to the atmosphere.
31 Nitrogen Input/Output Relationship in Large Watersheds
32 Organic Carbon in Urban Soils B A A. In the presence of non-native earthworms and higher temperatures, the layer of litter (O horizon) is thinner in urban forest soils, but the amount of C in the soil is greater than in suburban/rural forest soils. Without earthworms the O horizon in urban forest soil is thicker than in suburban/rural soils (lower quality litter in urban stands). B. Well maintained laws (low density residential and institutional land) contained SOC densities similar to forest soils. In the city of Baltimore, Organic matter content in urban soils was negatively correlated with bulk density. Caution: these observations should not be generalized before other similar studies in cities located in different climates. Source: Pouyat, R, P Groffman, I Yesilonisc, and L Hernandez Soil carbon pools and fluxes in urban ecosystems. Environmental Pollution: S107-S118.
33 Decomposition Decomposition and mineralization reactions are microbial enzymatic oxidation and reduction reactions. Enzymes are catalyst that aid decomposition or building of organic material. During decomposition the material is broken down into their organic constituents and finally into CO 2 and H 2 O. Decomposition processes can take place in aerobic or anaerobic conditions.
34 Rate of Decomposition of Organic Materials Organic compound Sugar, starches and single proteins Hemicellulose Rate of decomposition Rapid Cellulose Fats, waxes Lignins and phenolic compounds Slow
35 An Example A mixture of residues from Pinus nigra, P. sylvestry, and Quercus robur % of total %age lost by decomposition by Original litter 1 st year 2 nd year 5 th year 10 th year Sugars Cellulose Hemicellulose Lignins Waxes Phenols
36 Decomposition in Aerobic Soils In the presence of oxygen, the general reaction is: Organic C + O 2 CO 2 + H 2 O + energy First, the cell constituents (aminoacids, proteins, lipids, etc) are released. Then decomposition of the most resistant material occurs in stages. Final products: NH 4+, SO 2-4, NO 3-, and H 2 0.
37 Decomposition in Anaerobic Soils Without oxygen, decomposition proceeds very slowly. Under anaerobic conditions organic matter tends to accumulate. The final products are a variety of partially oxidized compounds: organic acids, alcohol, and methane gas.
38 Sequence of Redox Reactions
39 Factors Controlling Decomposition and Mineralization Environmental conditions promoting mineralization and decompositions are: Temperature: 25-35º C is optimum. Water content: extreme (dry and waterlogged) conditions reduce plant growth and microbial activity (~60% of porosity filled with water is optimum). Soil texture: other conditions being equal, clay tend to retain more humus. Near-neutral ph.
40 The Priming Effect Fresh residue addition fuels microbial activity. Microbial population and metabolic capacity increases Native, stable soil humus is attacked. Slow pool of SOM is depleted, while total SOM has increased.
41 Figure 11.3 Diagram of the general changes that take place when fresh plant residues are added to a soil. The arrows indicate transfers of carbon among compartments. The time required for the process will depend on the nature of the residues and the soil. Most of the carbon released during the initial rapid breakdown of the residues is converted to carbon dioxide, but the smaller amounts of carbon converted into microbially synthesized compounds (biomass) and, eventually, into soil humus should not be overlooked. Although the peak of microbial activity appears to accelerate the decay of the original humus, a phenomenon known as the priming effect, the humus level is increased by the end of the process. Where vegetation, environment, and management remain stable for a long time, the soil humus content will reach an equilibrium level in which, the carbon added to the humus pool through the decomposition of plant residues each year is balanced by carbon lost through the decomposition of existing soil humus.
42 Typical C/N Ratios for Soil-Related Organic Material Young legumes 12-20:1 Young grasses 20-40:1 Manure 20-50:1 Corn stalks 60:1 Oat/wheat straw 80-90:1 Tree leaves :1 Pine needles :1 Woody material :1 A C/N ratio depends on the biochemical composition of a tissue, i.e. relative amounts of protein, cellulose, lignin, etc.
43 Figure 11.4 Changes in microbial activity, in soluble nitrogen level, and in residual C/N ratio following the addition of either high (a) or low (b) C/N ratio organic materials. Where the C/N ratio of added residues is above 25, microbes digesting the residues must supplement the nitrogen contained in the residues with soluble nitrogen from the soil. During the resulting nitrate depression period, competition between higher plants and microbes would be severe enough to cause nitrogen deficiency in the plants. Note that in both cases soluble N in the soil ultimately increases from its original level once the decomposition process has run its course. The trends shown are for soils without growing plants, which, if present, would continually remove a portion of the soluble nitrogen as soon as it is released.
44 Estimates of P Pools Medium Air Land Top 0.5 m Sea Animals Form NOT APPLICABLE Plant SOIL-inorganic P SOIL-organic P Surface (top 300m) Deep Plant (sea and land) Pg P Source: Smil, V Phosphorous in the environment: Natural flows and human interferences. Annu. Rev. Energy Environ. 25:
45 Distinctive Features of the P Cycle Much less attention than the C or N cycles: Between 1979 and 1990 >1,000 papers on the N cycle and <100 on the P cycle. P is not present in the atmosphere: Land- and water-based P cycles are disconnected. It takes longer to notice human interference. There no biotic mechanism to fix the element: Plants rely only on P present in the soil. Litter returns to the soil a large share of the assimilated nutrient.
46 Figure 13.6 The phosphorus cycle in soils. The boxes represent pools of the various forms of phosphorus in the cycle, while the arrows represent translocations and transformations among these pools. The three largest white boxes indicate the principal groups of phosphorus-containing compounds found in soils. Within each of these groups, the less soluble, less available forms tend to dominate.
47 Anthropogenic Alteration of the P Cycle (Smil, 2000) Fluxes Wind Erosion Water Erosion River Transport (particulate) River Transport (dissolved) Biomass combustion Crop Uptake Animal Waste Human Waste Organic Recycling Inorganic Fertilizers 1 Tg: Tera (10 12 ) grams Natural Preindustrial (1800) Recent (2000) Tg 1 P/y <2 <3 >3 >8 >12 >27 >6 >8 >20 >1 <2 >2 <0.1 <0.2 < >1 > <0.5 >6 15
48 Soil P Dynamics The chemical forms of P in soils: organic (20-80%): turned over rapidly (from few days to one year). inorganic: calcium-bound, and iron- or aluminum-bound compounds. The amount of P in solution is very low: Available P 0.01% soil P tot Soil P is immobile: Diffusion to plant root Root extension/mycorrhizae
49 Figure Inorganic fixation of added phosphates at various soil ph values. Average conditions are postulated, and it is not to be inferred that any particular soil would have exactly this distribution. The actual proportion of the phosphorus remaining in an available form will depend upon contact with the soil, time for reaction, and other factors. It should be kept in mind that some of the added phosphorus may be changed to an organic form in which it would be temporarily unavailable.
The Nutrient Cycle. Atmospheric pool. Organic material. 5 Soil solution storage
 The Nutrient Cycle Atmospheric pool Precipitation Canopy, wood, and root Litter fall SOIL 2 Soil and rock minerals cations 1 2 3 Groundwater level Organic material 5 Soil solution storage 4 4 Channel BEDROCK
The Nutrient Cycle Atmospheric pool Precipitation Canopy, wood, and root Litter fall SOIL 2 Soil and rock minerals cations 1 2 3 Groundwater level Organic material 5 Soil solution storage 4 4 Channel BEDROCK
Ecosystems. Trophic relationships determine the routes of energy flow and chemical cycling in ecosystems.
 AP BIOLOGY ECOLOGY ACTIVITY #5 Ecosystems NAME DATE HOUR An ecosystem consists of all the organisms living in a community as well as all the abiotic factors with which they interact. The dynamics of an
AP BIOLOGY ECOLOGY ACTIVITY #5 Ecosystems NAME DATE HOUR An ecosystem consists of all the organisms living in a community as well as all the abiotic factors with which they interact. The dynamics of an
Lab today Finish Inventory work at Rest Area Site
 Lubrecht Forest, Montana NREM 301 Forest Ecology & Soils Day 23 Nov 10, 2009 Nutrient Cycling (Chapters 16-18) Lab today Finish Inventory work at Rest Area Site Quiz on Thursday Also record trees & shrubs
Lubrecht Forest, Montana NREM 301 Forest Ecology & Soils Day 23 Nov 10, 2009 Nutrient Cycling (Chapters 16-18) Lab today Finish Inventory work at Rest Area Site Quiz on Thursday Also record trees & shrubs
Nutrient Cycling & Soils
 Nutrient Cycling & Soils tutorial by Paul Rich Outline 1. Nutrient Cycles What are nutrient cycles? major cycles 2. Water Cycle 3. Carbon Cycle 4. Nitrogen Cycle 5. Phosphorus Cycle 6. Sulfur Cycle 7.
Nutrient Cycling & Soils tutorial by Paul Rich Outline 1. Nutrient Cycles What are nutrient cycles? major cycles 2. Water Cycle 3. Carbon Cycle 4. Nitrogen Cycle 5. Phosphorus Cycle 6. Sulfur Cycle 7.
Ecosystems: Nutrient Cycles
 Ecosystems: Nutrient Cycles Greeks, Native Peoples, Buddhism, Hinduism use(d) Earth, Air, Fire, and Water as the main elements of their faith/culture Cycling in Ecosystems the Hydrologic Cycle What are
Ecosystems: Nutrient Cycles Greeks, Native Peoples, Buddhism, Hinduism use(d) Earth, Air, Fire, and Water as the main elements of their faith/culture Cycling in Ecosystems the Hydrologic Cycle What are
Cycling and Biogeochemical Transformations of N, P, S, and K
 Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 24 September 2013 Reading: Schlesinger & Bernhardt, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification
Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 24 September 2013 Reading: Schlesinger & Bernhardt, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification
Guide 34. Ecosystem Ecology: Energy Flow and Nutrient Cycles. p://www.mordantorange.com/blog/archives/comics_by_mike_bannon/mordant_singles/0511/
 Guide 34 Ecosystem Ecology: Energy Flow and Nutrient Cycles p://www.mordantorange.com/blog/archives/comics_by_mike_bannon/mordant_singles/0511/ Overview: Ecosystems, Energy, and Matter An ecosystem consists
Guide 34 Ecosystem Ecology: Energy Flow and Nutrient Cycles p://www.mordantorange.com/blog/archives/comics_by_mike_bannon/mordant_singles/0511/ Overview: Ecosystems, Energy, and Matter An ecosystem consists
TERRESTRIAL ECOLOGY PART DUEX. Biogeochemical Cycles Biomes Succession
 DO NOW: -GRAB PAPERS FOR TODAY -GET A HIGHLIGHTER -UPDATE HW FOR TONIGHT COMPLETE AQUATIC ECOLOGY PACKET (INCLUDES VIDEO) -BEGIN READING THROUGH THE LECTURE TERRESTRIAL ECOLOGY PART DUEX Biogeochemical
DO NOW: -GRAB PAPERS FOR TODAY -GET A HIGHLIGHTER -UPDATE HW FOR TONIGHT COMPLETE AQUATIC ECOLOGY PACKET (INCLUDES VIDEO) -BEGIN READING THROUGH THE LECTURE TERRESTRIAL ECOLOGY PART DUEX Biogeochemical
Human perturbations to the global Nitrogen cycle
 Human perturbations to the global Nitrogen cycle Lecture for Biogeochemistry and Global Change Edzo Veldkamp The pace of human caused global change has increased in modern history, but none so rapidly
Human perturbations to the global Nitrogen cycle Lecture for Biogeochemistry and Global Change Edzo Veldkamp The pace of human caused global change has increased in modern history, but none so rapidly
Cycling and Biogeochemical Transformations of N, P, S, and K
 Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 18 September 2012 Reading: Schlesinger, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions
Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 18 September 2012 Reading: Schlesinger, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions
Chapter 3 Ecosystem Ecology. Tuesday, September 19, 17
 Chapter 3 Ecosystem Ecology Reversing Deforestation in Haiti Answers the following: Why is deforestation in Haiti so common? What the negative impacts of deforestation? Name three actions intended counteract
Chapter 3 Ecosystem Ecology Reversing Deforestation in Haiti Answers the following: Why is deforestation in Haiti so common? What the negative impacts of deforestation? Name three actions intended counteract
Cycling and Biogeochemical Transformations of N, P and S
 Cycling and Biogeochemical Transformations of N, P and S OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions of N gases from soils
Cycling and Biogeochemical Transformations of N, P and S OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions of N gases from soils
Cycling and Biogeochemical Transformations of N, P, S, and K
 Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 23 September 2014 Reading: Schlesinger & Bernhardt, Chapter 6 2014 Frank Sansone 1. Nitrogen cycle Soil nitrogen
Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 23 September 2014 Reading: Schlesinger & Bernhardt, Chapter 6 2014 Frank Sansone 1. Nitrogen cycle Soil nitrogen
Nutrients elements required for the development, maintenance, and reproduction of organisms.
 Nutrient Cycles Energy flows through ecosystems (one way trip). Unlike energy, however, nutrients (P, N, C, K, S ) cycle within ecosystems. Nutrients are important in controlling NPP in ecosystems. Bottom-up
Nutrient Cycles Energy flows through ecosystems (one way trip). Unlike energy, however, nutrients (P, N, C, K, S ) cycle within ecosystems. Nutrients are important in controlling NPP in ecosystems. Bottom-up
Cycling and Biogeochemical Transformations of N, P and S
 Cycling and Biogeochemical Transformations of N, P and S OCN 401 - Biogeochemical Systems Reading: Schlesinger,, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions of N gases from
Cycling and Biogeochemical Transformations of N, P and S OCN 401 - Biogeochemical Systems Reading: Schlesinger,, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions of N gases from
Ch. 5 - Nutrient Cycles and Soils
 Ch. 5 - Nutrient Cycles and Soils What are Nutrient (biogeochemical) Cycles? a process by which nutrients are recycled between living organisms and nonliving environment. The three general types of nutrient
Ch. 5 - Nutrient Cycles and Soils What are Nutrient (biogeochemical) Cycles? a process by which nutrients are recycled between living organisms and nonliving environment. The three general types of nutrient
We share the Earth. Ecology & Environmental Issues
 We share the Earth Ecology & Environmental Issues 1 with a whole lot of other creatures We don t share very well. 2 Ecology Putting it all together study of interactions between creatures & their environment,
We share the Earth Ecology & Environmental Issues 1 with a whole lot of other creatures We don t share very well. 2 Ecology Putting it all together study of interactions between creatures & their environment,
Ecosystems and Nutrient Cycles Chapters 3
 Ecosystems and Nutrient Cycles Chapters 3 Prokaryotic and Eukaryotic cells Figure 3-2 Prokaryotic cells: Have organelles. Bacteria and Archaea are composed of prokaryotic cells. Eukaryotic cells: cells,
Ecosystems and Nutrient Cycles Chapters 3 Prokaryotic and Eukaryotic cells Figure 3-2 Prokaryotic cells: Have organelles. Bacteria and Archaea are composed of prokaryotic cells. Eukaryotic cells: cells,
How Ecosystems Work Section 2
 Objectives List the three stages of the carbon cycle. Describe where fossil fuels are located. Identify one way that humans are affecting the carbon cycle. List the tree stages of the nitrogen cycle. Describe
Objectives List the three stages of the carbon cycle. Describe where fossil fuels are located. Identify one way that humans are affecting the carbon cycle. List the tree stages of the nitrogen cycle. Describe
Soil Organic Matter (SOM) Important component in soil fertility The higher the SOM soil more fertile
 Soil Organic Matter (SOM) Important component in soil fertility The higher the SOM soil more fertile Advantages of OM Supply nutrients (especially N, P, S) Increase CEC Improve physical properties( soil
Soil Organic Matter (SOM) Important component in soil fertility The higher the SOM soil more fertile Advantages of OM Supply nutrients (especially N, P, S) Increase CEC Improve physical properties( soil
Class XII Chapter 14 Ecosystem Biology
 Question 1: Fill in the blanks. (a) Plants are called as because they fix carbon dioxide. (b) In an ecosystem dominated by trees, the pyramid (of numbers) is type. (c) In aquatic ecosystems, the limiting
Question 1: Fill in the blanks. (a) Plants are called as because they fix carbon dioxide. (b) In an ecosystem dominated by trees, the pyramid (of numbers) is type. (c) In aquatic ecosystems, the limiting
Chapter 4. Ecosystems
 Chapter 4 Ecosystems Chapter 4 Section 1: What Is an Ecosystem Key Vocabulary Terms 7 Adapted from Holt Biology 2008 Community A group of various species that live in the same habitat and interact with
Chapter 4 Ecosystems Chapter 4 Section 1: What Is an Ecosystem Key Vocabulary Terms 7 Adapted from Holt Biology 2008 Community A group of various species that live in the same habitat and interact with
Freshwater Wetlands: Functions & Conservation. ENVIRTHON Workshop 2016 University of Massachusetts Amherst Deborah J.
 Freshwater Wetlands: Functions & Conservation ENVIRTHON Workshop 2016 University of Massachusetts Amherst Deborah J. Henson, PhD, CPSS What is a Wetland? Legal Definition:...those areas that are inundated
Freshwater Wetlands: Functions & Conservation ENVIRTHON Workshop 2016 University of Massachusetts Amherst Deborah J. Henson, PhD, CPSS What is a Wetland? Legal Definition:...those areas that are inundated
The rest of this article describes four biogeochemical cycles: the water cycle, carbon cycle, nitrogen cycle, and phosphorous cycle.
 BIOGEOCHEMICAL CYCLES The chemical elements and water that are needed by living things keep recycling over and over on Earth. These cycles are called biogeochemical cycles. They pass back and forth through
BIOGEOCHEMICAL CYCLES The chemical elements and water that are needed by living things keep recycling over and over on Earth. These cycles are called biogeochemical cycles. They pass back and forth through
ECOSYSTEMS. Follow along in chapter 54. *Means less important
 ECOSYSTEMS Follow along in chapter 54 *Means less important How do ecosystems function? What is an ecosystem? All living things in an area and their abiotic environment Ecosystem function can be easily
ECOSYSTEMS Follow along in chapter 54 *Means less important How do ecosystems function? What is an ecosystem? All living things in an area and their abiotic environment Ecosystem function can be easily
Cycling and Biogeochemical Transformations of N, P, S, and K
 Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 20 September 2016 Reading: Schlesinger & Bernhardt, Chapter 6 2016 Frank Sansone 1. Nitrogen cycle Soil nitrogen
Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 20 September 2016 Reading: Schlesinger & Bernhardt, Chapter 6 2016 Frank Sansone 1. Nitrogen cycle Soil nitrogen
Lesson Overview. Cycles of Matter. Lesson Overview. 3.4 Cycles of Matter
 Lesson Overview 3.4 THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these elements and do not use them up, so where do essential
Lesson Overview 3.4 THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these elements and do not use them up, so where do essential
The water cycle describes the continuous movement of water on, above and below the surface
 Nitrogen and carbon cycles Water cycle The water cycle describes the continuous movement of water on, above and below the surface It is driven by radiation, convection and advection. It includes solid,
Nitrogen and carbon cycles Water cycle The water cycle describes the continuous movement of water on, above and below the surface It is driven by radiation, convection and advection. It includes solid,
Section 2: The Cycling of Materials
 Section 2: The Cycling of Materials Preview Bellringer Objectives The Carbon Cycle How Humans Affect the Carbon Cycle The Nitrogen Cycle Decomposers and the Nitrogen Cycle The Phosphorus Cycle Section
Section 2: The Cycling of Materials Preview Bellringer Objectives The Carbon Cycle How Humans Affect the Carbon Cycle The Nitrogen Cycle Decomposers and the Nitrogen Cycle The Phosphorus Cycle Section
Cycles of Ma,er. Lesson Overview. Lesson Overview. 3.4 Cycles of Matter
 Lesson Overview Cycles of Ma,er Lesson Overview 3.4 Cycles of Matter THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these
Lesson Overview Cycles of Ma,er Lesson Overview 3.4 Cycles of Matter THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these
Managing Soils for Improved Pasture
 Managing Soils for Improved Pasture Jonathan Deenik, PhD Department of Tropical Plant and Soil Sciences University of Hawaii Rota Grazing and Livestock Management Workshop June 10-12, 12, 2010 1 Water
Managing Soils for Improved Pasture Jonathan Deenik, PhD Department of Tropical Plant and Soil Sciences University of Hawaii Rota Grazing and Livestock Management Workshop June 10-12, 12, 2010 1 Water
Do Now. Take out your activity you completed on Friday when I wasn t here!
 Do Now Take out your activity you completed on Friday when I wasn t here! Biogeochemical Cycles 37.18-37.23 Objectives Identify and describe the flow of nutrients in each biogeochemical cycle Explain the
Do Now Take out your activity you completed on Friday when I wasn t here! Biogeochemical Cycles 37.18-37.23 Objectives Identify and describe the flow of nutrients in each biogeochemical cycle Explain the
Biogeochemical Cycles
 Biogeochemical Cycles Biogeochemical Cycles refers to the cycling of materials between living things and the environment. Text Pages 50 51, 62 69 1 The Oxygen Cycle the movement of oxygen between the atmosphere
Biogeochemical Cycles Biogeochemical Cycles refers to the cycling of materials between living things and the environment. Text Pages 50 51, 62 69 1 The Oxygen Cycle the movement of oxygen between the atmosphere
River transport and chemistry
 OCN 401 Biogeochemical Systems (10.15.15) (Schlesinger & Bernhardt: Chapter 8) River transport and chemistry Lecture Outline 1. Introduction - Overview 2. Soil Hydraulics & Stream Hydrology 3. Stream Load
OCN 401 Biogeochemical Systems (10.15.15) (Schlesinger & Bernhardt: Chapter 8) River transport and chemistry Lecture Outline 1. Introduction - Overview 2. Soil Hydraulics & Stream Hydrology 3. Stream Load
2/11/16. Materials in ecosystems are constantly reused Three cycles: The Carbon Cycle The Nitrogen Cycle The Phosphorus Cycle
 Materials in ecosystems are constantly reused Three cycles: The Carbon Cycle The Nitrogen Cycle The Cycle Carbon is essential in proteins, fats, and carbohydrates, which make up all organisms Carbon cycle
Materials in ecosystems are constantly reused Three cycles: The Carbon Cycle The Nitrogen Cycle The Cycle Carbon is essential in proteins, fats, and carbohydrates, which make up all organisms Carbon cycle
10/18/2010 THINK ABOUT IT CHAPTER 3 THE BIOSHPERE RECYCLING IN THE BIOSPHERE RECYCLING IN THE BIOSPHERE
 THINK ABOUT IT CHAPTER 3 THE BIOSHPERE 3.4 Mrs. Michaelsen A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these elements and do not use them
THINK ABOUT IT CHAPTER 3 THE BIOSHPERE 3.4 Mrs. Michaelsen A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these elements and do not use them
River transport and chemistry. Lecture Outline
 OCN 401 Biogeochemical Systems (10.12.17) (Schlesinger & Bernhardt: Chapter 8) River transport and chemistry Lecture Outline 1. Introduction Overview 2. Soil Hydraulics & Stream Hydrology 3. Stream Load
OCN 401 Biogeochemical Systems (10.12.17) (Schlesinger & Bernhardt: Chapter 8) River transport and chemistry Lecture Outline 1. Introduction Overview 2. Soil Hydraulics & Stream Hydrology 3. Stream Load
LABEL AND EXPLAIN THE PROCESSES AT EACH NUMBER IN THE DIAGRAM ABOVE
 HYDROLOGIC CYCLE 3 4 5 2 5 1B 6B 1A 6A 7 6C LABEL AND EXPLAIN THE PROCESSES AT EACH NUMBER IN THE DIAGRAM ABOVE 1A. Evaporation of water from oceans 1B. Evaporation of water from land sources (water and
HYDROLOGIC CYCLE 3 4 5 2 5 1B 6B 1A 6A 7 6C LABEL AND EXPLAIN THE PROCESSES AT EACH NUMBER IN THE DIAGRAM ABOVE 1A. Evaporation of water from oceans 1B. Evaporation of water from land sources (water and
Section 2: The Cycling of Matter
 Section 2: The Cycling of Matter Preview Classroom Catalyst Objectives The Carbon Cycle How Humans Affect the Carbon Cycle The Nitrogen Cycle Decomposers and the Nitrogen Cycle The Phosphorus Cycle Section
Section 2: The Cycling of Matter Preview Classroom Catalyst Objectives The Carbon Cycle How Humans Affect the Carbon Cycle The Nitrogen Cycle Decomposers and the Nitrogen Cycle The Phosphorus Cycle Section
Ch. 4 Ecosystems. Biology I Loulousis
 Ch. 4 Ecosystems Biology I Loulousis Objectives 1.) Define ecology, ecosystem, and succession 2.) Identify and distinguish between the levels of organization in ecology 3.)Distinguish between primary and
Ch. 4 Ecosystems Biology I Loulousis Objectives 1.) Define ecology, ecosystem, and succession 2.) Identify and distinguish between the levels of organization in ecology 3.)Distinguish between primary and
BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through living organisms and the physical environment.
 BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through living organisms and the physical environment. BIOCHEMIST: Scientists who study how LIFE WORKS at a CHEMICAL level. The work of biochemists has
BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through living organisms and the physical environment. BIOCHEMIST: Scientists who study how LIFE WORKS at a CHEMICAL level. The work of biochemists has
Lecture 21: Soil Redox; Hydric Soils; Wetlands
 Lecture 21: Soil Redox; Hydric Soils; Wetlands Soil Oxidation-Reduction (Redox) Many Elements Exist in Multiple Oxidation States in Soils Oxygen: O 2 (0), H 2 O (-2) Carbon: CH 4 (-4), Organic C (-3 to
Lecture 21: Soil Redox; Hydric Soils; Wetlands Soil Oxidation-Reduction (Redox) Many Elements Exist in Multiple Oxidation States in Soils Oxygen: O 2 (0), H 2 O (-2) Carbon: CH 4 (-4), Organic C (-3 to
Building Soil Organic Matter: What, Why, How?
 FarmSmart Conference, Guelph Ontario 20 January 2018 Building Soil Organic Matter: What, Why, How? Ray Weil Biosphere-2. A parable about soil organic matter and the carbon cycle. Biospherians in Biosphere2
FarmSmart Conference, Guelph Ontario 20 January 2018 Building Soil Organic Matter: What, Why, How? Ray Weil Biosphere-2. A parable about soil organic matter and the carbon cycle. Biospherians in Biosphere2
The Enigma of Soil Nitrogen George Rehm, University of Minnesota
 The Enigma of Soil Nitrogen George Rehm, University of Minnesota 1. Introduction Throughout the northern and western Corn Belt, nitrogen (N) is the most dominant nutrient in the world of plant nutrition.
The Enigma of Soil Nitrogen George Rehm, University of Minnesota 1. Introduction Throughout the northern and western Corn Belt, nitrogen (N) is the most dominant nutrient in the world of plant nutrition.
BIOGEOCHEMICAL CYCLES
 BIOGEOCHEMICAL CYCLES BIOGEOCHEMICAL CYCLES A biogeochemical cycle or cycling of substances is a pathway by which a chemical element or molecule moves through both biotic and abiotic compartments of Earth.
BIOGEOCHEMICAL CYCLES BIOGEOCHEMICAL CYCLES A biogeochemical cycle or cycling of substances is a pathway by which a chemical element or molecule moves through both biotic and abiotic compartments of Earth.
Transport & Transformation of chemicals in an ecosystem, involving numerous interrelated physical, chemical, & biological processes
 OPEN Wetland Ecology Lectures 14-15-16 Wetland Biogeochemistry What is biogeochemical cycling? Transport & Transformation of chemicals in an ecosystem, involving numerous interrelated physical, chemical,
OPEN Wetland Ecology Lectures 14-15-16 Wetland Biogeochemistry What is biogeochemical cycling? Transport & Transformation of chemicals in an ecosystem, involving numerous interrelated physical, chemical,
The Biosphere Chapter 3. What Is Ecology? Section 3-1
 The Biosphere Chapter 3 What Is Ecology? Section 3-1 Interactions and Interdependence Ecology is the scientific study of interactions among organisms and between organisms and their environment, or surroundings.
The Biosphere Chapter 3 What Is Ecology? Section 3-1 Interactions and Interdependence Ecology is the scientific study of interactions among organisms and between organisms and their environment, or surroundings.
Nutrient Cycles. Nutrient cycles involve flow of high quality energy from the sun through the environment & of elements.
 Nutrient Cycles Nutrient cycles (= biogeochemical cycles): natural processes that involve the flow of nutrients from the environment (air, water, soil, rock) to living organisms ( ) & back again. Nutrient
Nutrient Cycles Nutrient cycles (= biogeochemical cycles): natural processes that involve the flow of nutrients from the environment (air, water, soil, rock) to living organisms ( ) & back again. Nutrient
3 3 Cycles of Matter
 3 3 Cycles of Matter Recycling in the Biosphere Energy - one way flow matter - recycled within and between ecosystems. biogeochemical cycles matter Elements, chemical compounds, and other forms passed
3 3 Cycles of Matter Recycling in the Biosphere Energy - one way flow matter - recycled within and between ecosystems. biogeochemical cycles matter Elements, chemical compounds, and other forms passed
Biogeochemical Cycles. Nutrient cycling at its finest!
 Biogeochemical Cycles Nutrient cycling at its finest! Four Criteria for Sustainability Sustainable Ecosystems Need: Reliance on Solar Energy High Biodiversity Population Control Nutrient Cycling This note
Biogeochemical Cycles Nutrient cycling at its finest! Four Criteria for Sustainability Sustainable Ecosystems Need: Reliance on Solar Energy High Biodiversity Population Control Nutrient Cycling This note
David Rowlings Institute for Sustainable Resources Queensland University of Technology
 How does carbon influence nitrogen availability and losses? David Rowlings Institute for Sustainable Resources Queensland University of Technology Outline Carbon cycle Global carbon cycle Soil carbon 3
How does carbon influence nitrogen availability and losses? David Rowlings Institute for Sustainable Resources Queensland University of Technology Outline Carbon cycle Global carbon cycle Soil carbon 3
Cycling and Biogeochemical Transformations of N, P, S, and K
 Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 19 September 2016 Reading: Schlesinger & Bernhardt, Chapter 6 2017 Frank Sansone Outline 1. Nitrogen cycle
Cycling and Biogeochemical Transformations of N, P, S, and K OCN 401 - Biogeochemical Systems 19 September 2016 Reading: Schlesinger & Bernhardt, Chapter 6 2017 Frank Sansone Outline 1. Nitrogen cycle
Bio 112 Ecology: Final Practice Exam Multiple Choice
 Final Exam Topics: 1) Basic Ecological Principles a) Biomes, ecosystems, communities and populations i) Biomes: know the major ones and where they occur ii) Ecosystem: communities and physical environment
Final Exam Topics: 1) Basic Ecological Principles a) Biomes, ecosystems, communities and populations i) Biomes: know the major ones and where they occur ii) Ecosystem: communities and physical environment
 CHAPTER 14 ECOSYSTEM POINTS TO REMEMBER Startification : Vertical distribution of different species occupying different levels in an ecosystem. Primary Production : Amount of biomas or organic matter produced
CHAPTER 14 ECOSYSTEM POINTS TO REMEMBER Startification : Vertical distribution of different species occupying different levels in an ecosystem. Primary Production : Amount of biomas or organic matter produced
5/6/2015. Matter is recycled within and between ecosystems.
 Biogeochemical Cycles/ Nutrient Cycles Biogeochemical Cycle Evaporation Water Cycle Transpiration Condensation Precipitation Runoff Vocabulary Seepage Root Uptake Carbon Cycle Phosphorus Cycle Nitrogen
Biogeochemical Cycles/ Nutrient Cycles Biogeochemical Cycle Evaporation Water Cycle Transpiration Condensation Precipitation Runoff Vocabulary Seepage Root Uptake Carbon Cycle Phosphorus Cycle Nitrogen
Ecology, the Environment, and Us
 BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 23 Ecology, the Environment, and Us Lecture Presentation Anne Gasc Hawaii Pacific University and University
BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 23 Ecology, the Environment, and Us Lecture Presentation Anne Gasc Hawaii Pacific University and University
Earth s Life-Support Atmosphere- gases surrounding earth s surface. Troposphere= air we breathe; weather 78% nitrogen, 21% oxygen, 0.
 1 2 3 4 Ecosystems APES CH 3 But First: Cells Complex organic compounds called macromolecules make up the basic molecular units found in cells. Complex carbohydrates Cellulose and starch Proteins- made
1 2 3 4 Ecosystems APES CH 3 But First: Cells Complex organic compounds called macromolecules make up the basic molecular units found in cells. Complex carbohydrates Cellulose and starch Proteins- made
How Ecosystems Work Section 2. Chapter 5 How Ecosystems Work Section 2: Cycling of Materials DAY 1
 Chapter 5 How Ecosystems Work Section 2: Cycling of Materials DAY 1 The Carbon Cycle The carbon cycle is the movement of carbon from the nonliving environment into living things and back Carbon is the
Chapter 5 How Ecosystems Work Section 2: Cycling of Materials DAY 1 The Carbon Cycle The carbon cycle is the movement of carbon from the nonliving environment into living things and back Carbon is the
FACT SHEET. Understanding Soil Microbes and Nutrient Recycling. Agriculture and Natural Resources SAG-16-10
 FACT SHEET Agriculture and Natural Resources Understanding Soil Microbes and Nutrient Recycling James J. Hoorman Cover Crops and Water Quality Extension Educator Ohio State University Extension Rafiq Islam
FACT SHEET Agriculture and Natural Resources Understanding Soil Microbes and Nutrient Recycling James J. Hoorman Cover Crops and Water Quality Extension Educator Ohio State University Extension Rafiq Islam
3 3 Cycles of Matter Slide 1 of 33
 1 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move through the biosphere very differently. Unlike the one-way flow of energy, matter is recycled within and between ecosystems.
1 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move through the biosphere very differently. Unlike the one-way flow of energy, matter is recycled within and between ecosystems.
Nutrient Recycling with Manure and Cover Crops
 Nutrient Recycling with Manure and Cover Crops James J. Hoorman and Dr. Rafiq Islam OSU Extension Center at Lima and OSU Piketon Center Ohio State University Extension Introduction Converting from conventional
Nutrient Recycling with Manure and Cover Crops James J. Hoorman and Dr. Rafiq Islam OSU Extension Center at Lima and OSU Piketon Center Ohio State University Extension Introduction Converting from conventional
2.2 Nutrient Cycles in Ecosystems
 2.2 Nutrient Cycles in Ecosystems are chemicals required for growth and other life processes. Nutrients move through the biosphere in Nutrients often accumulate in areas called Without interference, generally
2.2 Nutrient Cycles in Ecosystems are chemicals required for growth and other life processes. Nutrients move through the biosphere in Nutrients often accumulate in areas called Without interference, generally
The Soil Community: Managing it. Kristy Borrelli REACCH Extension Specialist
 The Soil Community: Managing it for Better Crops Kristy Borrelli REACCH Extension Specialist kborrelli@uidaho.edu Outline Soil Community Soil Quality Soil Organic Matter and Carbon Soil Organic Matter
The Soil Community: Managing it for Better Crops Kristy Borrelli REACCH Extension Specialist kborrelli@uidaho.edu Outline Soil Community Soil Quality Soil Organic Matter and Carbon Soil Organic Matter
Ecosystems and the Biosphere: Energy Flow Through the Ecosystem and the Recycling of Matter
 Name Ecosystems and the Biosphere: Energy Flow Through the Ecosystem and the Recycling of Matter Overview: An ecosystem is: All of the organisms living on Earth need to carry out life processes such as
Name Ecosystems and the Biosphere: Energy Flow Through the Ecosystem and the Recycling of Matter Overview: An ecosystem is: All of the organisms living on Earth need to carry out life processes such as
BC Science Nutrient Cycles in Ecosystems
 BC Science 10 2.2 Nutrient Cycles in Ecosystems Notes Nutrients are chemicals required for growth and other life processes. Nutrients move through the biosphere in nutrient cycles (n.c), or exchanges.
BC Science 10 2.2 Nutrient Cycles in Ecosystems Notes Nutrients are chemicals required for growth and other life processes. Nutrients move through the biosphere in nutrient cycles (n.c), or exchanges.
Warm Up. What process do plants use to make sugar? What is chemosynthesis? What is transpiration?
 Warm Up What process do plants use to make sugar? What is chemosynthesis? What is transpiration? Check your answers: What process do plants use to make sugar? photosynthesis What is chemosynthesis? Organisms
Warm Up What process do plants use to make sugar? What is chemosynthesis? What is transpiration? Check your answers: What process do plants use to make sugar? photosynthesis What is chemosynthesis? Organisms
Monitoring carbon budgets
 Monitoring carbon budgets Suess effect on 14 C/ 12 C in CO 2, from Stuiver and Quay, 1981, EPSL 53:349-362 1 Figure from IPCC 4 th assessment report 2 Biogeochemical cycling (on land) Living tissue C H
Monitoring carbon budgets Suess effect on 14 C/ 12 C in CO 2, from Stuiver and Quay, 1981, EPSL 53:349-362 1 Figure from IPCC 4 th assessment report 2 Biogeochemical cycling (on land) Living tissue C H
Another cause of diversity may be the creation of different habitats within a region by periodic disturbance A community that forms if the land is
 Another cause of diversity may be the creation of different habitats within a region by periodic disturbance A community that forms if the land is undisturbed and that perpetuates itself for as long as
Another cause of diversity may be the creation of different habitats within a region by periodic disturbance A community that forms if the land is undisturbed and that perpetuates itself for as long as
1. Where are nutrients accumulated or stored for short or long periods?
 Use with textbook pages 68 87. Nutrient cycles Answer the questions below. Comprehension 1. Where are nutrients accumulated or stored for short or long periods? 2. Name a biotic process and an abiotic
Use with textbook pages 68 87. Nutrient cycles Answer the questions below. Comprehension 1. Where are nutrients accumulated or stored for short or long periods? 2. Name a biotic process and an abiotic
Biogeochemistry of Wetlands
 Institute of Food and Agricultural Sciences (IFAS) Biogeochemistry of Wetlands Si Science and da Applications Biogeochemical Properties of Wetlands Wetland Biogeochemistry Laboratory Soil and Water Science
Institute of Food and Agricultural Sciences (IFAS) Biogeochemistry of Wetlands Si Science and da Applications Biogeochemical Properties of Wetlands Wetland Biogeochemistry Laboratory Soil and Water Science
10/17/ Cycles of Matter. Recycling in the Biosphere. How does matter move among the living and nonliving parts of an ecosystem?
 2 of 33 3-3 Cycles of Matter How does matter move among the living and nonliving parts of an ecosystem? 3 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move through the
2 of 33 3-3 Cycles of Matter How does matter move among the living and nonliving parts of an ecosystem? 3 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move through the
How Ecosystems Work Section 1. Chapter 5 How Ecosystems Work Section 1: Energy Flow in Ecosystems DAY 1
 Chapter 5 How Ecosystems Work Section 1: Energy Flow in Ecosystems DAY 1 Life Depends on the Sun Energy from the sun enters an ecosystem when plants use sunlight to make sugar molecules. This happens through
Chapter 5 How Ecosystems Work Section 1: Energy Flow in Ecosystems DAY 1 Life Depends on the Sun Energy from the sun enters an ecosystem when plants use sunlight to make sugar molecules. This happens through
7.014 Lecture 20: Biogeochemical Cycles April 1, 2007
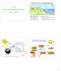 Global Nutrient Cycling - Biogeochemical Cycles 7.14 Lecture 2: Biogeochemical Cycles April 1, 27 Uptake Bioelements in Solution Weathering Precipitation Terrestrial Biomass Decomposition Volatile Elements
Global Nutrient Cycling - Biogeochemical Cycles 7.14 Lecture 2: Biogeochemical Cycles April 1, 27 Uptake Bioelements in Solution Weathering Precipitation Terrestrial Biomass Decomposition Volatile Elements
Bacteria, Fungi, Protozoa and Nematodes: Part of the Carbon to Nitrogen Transformation
 1 Nutrient Recycling with Manure and Cover Crops James J. Hoorman and Dr. Rafiq Islam OSU Extension Center at Lima and OSU Piketon Center Ohio State University Extension Introduction Converting from conventional
1 Nutrient Recycling with Manure and Cover Crops James J. Hoorman and Dr. Rafiq Islam OSU Extension Center at Lima and OSU Piketon Center Ohio State University Extension Introduction Converting from conventional
Lesson 2.4 Biogeochemical Cycles
 Lesson 2.4 Biogeochemical Cycles A carbon atom in your body today may have been part of a blade of grass last year, or a dinosaur bone millions of years ago. Fossilized bones in a Colorado dig. Lesson
Lesson 2.4 Biogeochemical Cycles A carbon atom in your body today may have been part of a blade of grass last year, or a dinosaur bone millions of years ago. Fossilized bones in a Colorado dig. Lesson
Slide 1 / All of Earth's water, land, and atmosphere within which life exists is known as a. Population Community Biome Biosphere
 Slide 1 / 40 1 ll of Earth's water, land, and atmosphere within which life exists is known as a Population ommunity iome iosphere Slide 2 / 40 2 ll the plants, animals, fungi living in a pond make up a
Slide 1 / 40 1 ll of Earth's water, land, and atmosphere within which life exists is known as a Population ommunity iome iosphere Slide 2 / 40 2 ll the plants, animals, fungi living in a pond make up a
WHY DO WE NEED NITROGEN?? Nitrogen is needed to make up DNA and protein!
 Nitrogen Cycle 2.2 WHY DO WE NEED NITROGEN?? Nitrogen is needed to make up DNA and protein! In animals, proteins are vital for muscle function. In plants, nitrogen is important for growth. NITROGEN Nitrogen
Nitrogen Cycle 2.2 WHY DO WE NEED NITROGEN?? Nitrogen is needed to make up DNA and protein! In animals, proteins are vital for muscle function. In plants, nitrogen is important for growth. NITROGEN Nitrogen
Soils and Global Warming. Temperature and Atmosphere. Soils and Water, Spring Lecture 9, Soils and Global Warming 1
 Soils and Global Warming Reading: Lecture Notes Objectives: Introduce climate change Describe measured and expected effects on soil systems Describe prediction of climate change effect on food production.
Soils and Global Warming Reading: Lecture Notes Objectives: Introduce climate change Describe measured and expected effects on soil systems Describe prediction of climate change effect on food production.
Application of Waste to Soils
 Application of Waste to Soils Definition of Waste "Wastes are materials that are not prime products (that is products produced for the market) for which the generator has no further use in terms of his/her
Application of Waste to Soils Definition of Waste "Wastes are materials that are not prime products (that is products produced for the market) for which the generator has no further use in terms of his/her
Climate: describes the average condition, including temperature and precipitation, over long periods in a given area
 Ch. 6 - Biomes Section 6.1: Defining Biomes Biome: a group of ecosystems that share similar biotic and abiotic conditions, large region characterized by a specific type of climate, plants, and animals
Ch. 6 - Biomes Section 6.1: Defining Biomes Biome: a group of ecosystems that share similar biotic and abiotic conditions, large region characterized by a specific type of climate, plants, and animals
OPTION C.6 NITROGEN & PHOSPHORUS CYCLES
 OPTION C.6 NITROGEN & PHOSPHORUS CYCLES C.6 A Cycle INTRO https://www.thewastewaterblog.com/single-post/2017/04/29/-cycle-and-other-graphics IB BIO C.6 3 The nitrogen cycle describes the movement of nitrogen
OPTION C.6 NITROGEN & PHOSPHORUS CYCLES C.6 A Cycle INTRO https://www.thewastewaterblog.com/single-post/2017/04/29/-cycle-and-other-graphics IB BIO C.6 3 The nitrogen cycle describes the movement of nitrogen
DECOMPOSITION OF ORGANIC MATTER IN SOIL
 DECOMPOSITION OF ORGANIC MATTER IN SOIL The rapidity with which a given organic amendment is decomposed depends on temperature, the supply of oxygen, moisture, and available minerals, the C/N ratio of
DECOMPOSITION OF ORGANIC MATTER IN SOIL The rapidity with which a given organic amendment is decomposed depends on temperature, the supply of oxygen, moisture, and available minerals, the C/N ratio of
BASICS OF WASTEWATER TREATMENT
 BASICS OF WASTEWATER TREATMENT Knowing the decisioning criteria relevant to site and drain field suitability, i.e., soil properties, can be enhanced by an understanding of some of the basics of wastewater
BASICS OF WASTEWATER TREATMENT Knowing the decisioning criteria relevant to site and drain field suitability, i.e., soil properties, can be enhanced by an understanding of some of the basics of wastewater
1. Energy to do work 2. Raw material to build/repair things (nutrients)
 1. Energy to do work 2. Raw material to build/repair things (nutrients) Living things are built from water Nutrients: carbon, hydrogen, nitrogen, and oxygen 3. Essential nutrients are cycled through environment
1. Energy to do work 2. Raw material to build/repair things (nutrients) Living things are built from water Nutrients: carbon, hydrogen, nitrogen, and oxygen 3. Essential nutrients are cycled through environment
NREM 301 Forest Ecology & Soils Day 25 November 17, Nutrient Cycling Lab Field Quiz/Exercise Quiz Thursday in class
 NREM 301 Forest Ecology & Soils Day 25 November 17, 2009 Nutrient Cycling Lab Field Quiz/Exercise Quiz Thursday in class Closed Notes - Group Activity On poster paper prepare a Labeled diagram of the N
NREM 301 Forest Ecology & Soils Day 25 November 17, 2009 Nutrient Cycling Lab Field Quiz/Exercise Quiz Thursday in class Closed Notes - Group Activity On poster paper prepare a Labeled diagram of the N
Food web Diagram that shows how food chains are linked together in a complex feeding relationship
 Energy Flow Food web Diagram that shows how food chains are linked together in a complex feeding relationship The food web has a number of advantages over a food chains including: More than one producer
Energy Flow Food web Diagram that shows how food chains are linked together in a complex feeding relationship The food web has a number of advantages over a food chains including: More than one producer
Biogeochemical cycles
 Biogeochemical cycles MATTER CYCLING IN ECOSYSTEMS Nutrient Cycles: Global Recycling Global Cycles recycle nutrients through the earth s air, land, water, and living organisms. Nutrients are the elements
Biogeochemical cycles MATTER CYCLING IN ECOSYSTEMS Nutrient Cycles: Global Recycling Global Cycles recycle nutrients through the earth s air, land, water, and living organisms. Nutrients are the elements
Ecosystems Section 1 What Is an Ecosystem? Objectives Distinguish Describe Sequence Interactions of Organisms and Their Environment Ecology Habitat
 Name Period Ecosystems Section 1 What Is an Ecosystem? Objectives Distinguish an ecosystem from a community. Describe the diversity of a representative ecosystem. Sequence the process of succession. Interactions
Name Period Ecosystems Section 1 What Is an Ecosystem? Objectives Distinguish an ecosystem from a community. Describe the diversity of a representative ecosystem. Sequence the process of succession. Interactions
Biology. Slide 1 of 33. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 33 2 of 33 3-3 Cycles of Matter How does matter move among the living and nonliving parts of an ecosystem? 3 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move
Biology 1 of 33 2 of 33 3-3 Cycles of Matter How does matter move among the living and nonliving parts of an ecosystem? 3 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move
Major changes in forest carbon and nitrogen cycling caused by declining sulphur deposition
 Major changes in forest carbon and nitrogen cycling caused by declining sulphur deposition Filip Oulehle, Chris Evans, Henning Meesenburg, Jakub Hruska, Pavel Kram, Jiri Kopacek, Bridget Emmett, Jack Cosby,
Major changes in forest carbon and nitrogen cycling caused by declining sulphur deposition Filip Oulehle, Chris Evans, Henning Meesenburg, Jakub Hruska, Pavel Kram, Jiri Kopacek, Bridget Emmett, Jack Cosby,
Matter Cycles How are nutrients recycled through ecosystems?
 9. In order to continually use the same area of land for agriculture, some farmers apply fertilizers to improve the level of nitrates in the soil. An alternative to this intensive use of fertilizer is
9. In order to continually use the same area of land for agriculture, some farmers apply fertilizers to improve the level of nitrates in the soil. An alternative to this intensive use of fertilizer is
Ecosystem. Ecosystems. Consumers. Simple Ecosystem Model. Trophic Levels. Food Chain marsh hawk
 Ecosystem Ecosystems Chapter 47 An association of organisms and their physical environment, interconnected by ongoing flow of energy and a cycling of materials Simple Ecosystem Model energy input from
Ecosystem Ecosystems Chapter 47 An association of organisms and their physical environment, interconnected by ongoing flow of energy and a cycling of materials Simple Ecosystem Model energy input from
Lesson Overview. Cycles of Matter. Lesson Overview. 3.4 Cycles of Matter
 Lesson Overview 3.4 THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these elements and do not use them up, so..where do essential
Lesson Overview 3.4 THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these elements and do not use them up, so..where do essential
CHEMICAL COMPOSITION OF NATURAL WATERS
 CHEMICAL COMPOSITION OF NATURAL WATERS DISSOVLED GASES Oxygen (and E h ) Why important? product of photosynthesis needed for aerobic respiration - Much of an aquatic organisms energy budget is devoted
CHEMICAL COMPOSITION OF NATURAL WATERS DISSOVLED GASES Oxygen (and E h ) Why important? product of photosynthesis needed for aerobic respiration - Much of an aquatic organisms energy budget is devoted
Chapter 55: Ecosystems
 Ch. 55 Warm-Up 1. Draw an energy pyramid and label the following trophic levels: Primary producer Primary consumer Secondary consumer Tertiary consumer 2. What is an example of an organism at each level
Ch. 55 Warm-Up 1. Draw an energy pyramid and label the following trophic levels: Primary producer Primary consumer Secondary consumer Tertiary consumer 2. What is an example of an organism at each level
Nutrient Cycles. & how Humans impact nutrient cycling. Accel Bio. Where do energy & nutrients come from?
 Nutrient Cycles & how Humans impact nutrient cycling Accel Bio Where do energy & nutrients come from? What are nutrients? What else do organisms need to survive and grow? Organisms need, Energy, water
Nutrient Cycles & how Humans impact nutrient cycling Accel Bio Where do energy & nutrients come from? What are nutrients? What else do organisms need to survive and grow? Organisms need, Energy, water
Biology. Slide 1 of 33. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 33 2 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move through the biosphere very differently. Unlike the one-way flow of energy, matter is recycled within
Biology 1 of 33 2 of 33 Recycling in the Biosphere Recycling in the Biosphere Energy and matter move through the biosphere very differently. Unlike the one-way flow of energy, matter is recycled within
Chapter Two: Cycles of Matter (pages 32-65)
 Chapter Two: Cycles of Matter (pages 32-65) 2.2 Biogeochemical Cycles (pages 42 52) In order to survive and grow, organisms must obtain nutrients that serve as sources of energy or chemical building blocks,
Chapter Two: Cycles of Matter (pages 32-65) 2.2 Biogeochemical Cycles (pages 42 52) In order to survive and grow, organisms must obtain nutrients that serve as sources of energy or chemical building blocks,
Determining the f ratio 11/16/2010. Incubate seawater in the presence of trace 15
 Plankton production is supported by 2 types of nitrogen: 1) new production supported by external sources of N (e.g. NO 3 and N 2 ), 2) recycled or regenerated production, sustained by recycling of N. Assumptions:
Plankton production is supported by 2 types of nitrogen: 1) new production supported by external sources of N (e.g. NO 3 and N 2 ), 2) recycled or regenerated production, sustained by recycling of N. Assumptions:
2. 2. Nutrient Cycles in Ecosystems. Before You Read. How are nutrients cycled in the biosphere? How does the carbon cycle work?
 Nutrient Cycles in Ecosystems Textbook pages 68 91 Section 2. 2 Summary Before You Read Like other organisms, your body relies on nutrients to stay healthy. Based on your current understanding, create
Nutrient Cycles in Ecosystems Textbook pages 68 91 Section 2. 2 Summary Before You Read Like other organisms, your body relies on nutrients to stay healthy. Based on your current understanding, create
