Regulated Medical Waste Management and Proper Waste Segregation. State-Specific Information California
|
|
|
- Cori Anthony
- 6 years ago
- Views:
Transcription
1 Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information California
2 Learning Topics Objective: Understand how to properly segregate waste streams and properly handle the most common regulated waste. I. Waste Stream Overview II. Identifying & Segregating Waste III. Managing RMW Collection and Storage IV. Proper Packaging of RMW V. Labeling & Documentation VI. DOT Requirements 2 VII. Summary
3 Waste Stream Overview Improper Segregation Practices General waste commingled with RMW General waste commingled with RMW and loose sharps
4 Waste Stream Overview Your Legal Obligation OSHA mandates special waste handling for Regulated Medical Waste to protect staff members and waste haulers Signed shipping document indicate your responsibility for the contained wastes and proper segregation 4
5 Identifying & Segregating Waste Regulated Medical Waste Cal-OSHA s Definition of Regulated Medical Waste Liquid or semi liquid blood or other potentially infectious material (OPIM) Contaminated items that contain liquid or semi-liquid blood or are caked with dried blood or OPIM Are capable of releasing these materials when handled or compressed Contaminated sharps Pathological and microbiological wastes containing blood or OPIM Medical waste regulated by California Health and Safety Code 5
6 Medical Waste Management Act California Health and Safety Code Sections
7 Biohazardous Waste California Health and Safety Code: Laboratory waste cultures and stocks Sharps waste Contaminated waste Pathological Chemotherapeutic Pharmaceutical Human surgery specimens/tissues suspected of being contaminated with infectious agents known to be contagious to humans Animal parts/tissues/carcasses suspected by veterinarian of being contaminated with infectious agents known to be contagious to humans 7 Excretions, exudates, secretions from humans/animals that are required to be isolated by the infection control staff, attending physician, veterinarian, or local health officer, to protect others from highly communicable diseases or diseases of animals that are highly communicable to humans
8 Registration Registration of Medical Waste Generators (Shippers) Generators (shippers) of medical waste, both SQ and LQ, are required to register with either: Local Enforcement Agency (LEA) Counties with LEAs are listed on next slide California Department of Public Health (CDPH) (if there is no LEA and State acts as LEA)
9 Local Enforcement Agencies Alameda County of Solid/Medical Waste Mgmt. Colusa County Environmental Health Contra Costa County Environmental Health Del Norte County DPH Env. Mgmt. Dept. Kings County Division of Environmental Health Lassen County Health and Human Services Dept. City of Long Beach Health Department City of Vernon Health & Environmental Control Madera County Environmental Health Marin County Environmental Health Services Merced County Dept. Public Health, Div. Env. Hlth Modoc County Environmental Health Monterey County Health Department Orange County Environmental Health Riverside County Environmental Health Services Sacramento County Environmental Health San Bernardino County Environmental Health San Diego County Dept. of Environmental Health San Francisco City & County DPH/ Hazardous Materials Program San Joaquin County Environmental Health Services San Mateo County Environmental Health Division Santa Clara County Department of Environmental Health Santa Cruz County Environmental Health Services Shasta County Environmental Health Division Siskiyou County Public Health Sonoma County Public Health Department Stanislaus County Department of Environmental Resources Tehama County Environmental Health Tuolumne County Environmental Health Ventura County Environmental Health Division Yuba County Environmental Health
10 Medical Waste Management Plan MWMP is a document completed by generators (shippers) of medical waste Plan is filed with the enforcement agency, either the State or, where applicable, the Local Enforcement Agency State filers can obtain a checklist CDPH Form 8661 for assistance in creating their plans 10 LEA filers can check for specific forms from the LEA
11 Identifying & Segregating Waste What Goes in the RMW Box? Closed disposable sharps containers Blood and OPIM Soaked, dripping bloody dressings Intact glass or plastic bottles with bloody fluid or OPIM (Some states require them to be placed in a sharps container) All disposable items soaked or dripping with blood or OPIM 11
12 Identifying & Segregating Waste Items NOT in the RMW Category NOT RMW: Chemicals RCRA Hazardous waste Radioactive waste Complete human remains Bulk Chemotherapy waste Compressed gas cylinders, canisters, inhalers, and aerosol cans Any devices or solutions containing mercury or other heavy metals Regular office garbage or solid waste RMW But REQUIRES SEGREGATION/INCINERATION Pathological waste Trace chemotherapeutic waste 12
13 Managing RMW Collection and Storage OSHA Packaging Requirements Cal-OSHA 5193 d E 3 Regulated waste shall be disposed of in containers which are closable, labeled, and color-coded Proper packaging requires 3 things: 1. Lid, or other means of closure 2. Red Bag 3. Biohazard labeling on 4 sides and top Applies to containers used for office storage and final transport containers 13
14 14 Managing RMW Collection and Storage Sharps Requirements Generators (shippers) must properly select sharps containers in order to protect employees during transport Definition of Sharps Needles and syringes Scalpel blades and lancets Glass pipettes, slides, and tubes Broken, contaminated glass Staples and wires (cardio-catheter wires) Orthodontic wires Disposable suture sets and biopsy forceps Electrocautery tips (no cauterizers or batteries)
15 Managing RMW Collection and Storage Sharps Container Placement Healthcare workers should be able to comfortably view the entire inlet opening of the disposal container Containers should be located within arm s reach Place containers slightly below eye level or on a counter top 15
16 Managing RMW Collection and Storage Improper Usage of Sharps Containers Direct visibility of the inlet opening is necessary Containers should be promptly replaced when contents reach manufacturer s fill line Overfilled containers increase the risk of needlestick injuries and violate proper product usage requirements 16
17 Managing RMW Collection and Storage Sharps Container Requirements Sharps containers must be placed in a secondary container for transport 17
18 Proper Packaging of RMW Preparing Reusable Containers Tie off red bag Container with red bag 18
19 Proper Packaging of RMW Preparing Reusable Containers Tie off red bag 19
20 Chemo and Path Waste Required to be Segregated CHEMOTHERAPEUTIC WASTE Biohazardous waste that is contaminated through contact with, or having previously contained chemotherapeutic agents Must be segregated for storage and placed in a secondary container that is labeled Chemotherapy Waste, CHEMO, etc. Stericycle can only accept trace chemo, i.e., less than 3% of original contents PATHOLOGICAL WASTE Human surgery specimens, or tissues which have been fixed with formaldehyde or other fixatives Must be segregated for storage and placed in a secondary container that is labeled Pathology Waste, or Path, etc. LABELING 20 Labeling shall be provided on the lid and on the sides and be visible from any lateral direction
21 Proper Packaging of RMW Weight Limitation The packaged waste must be within the required weight limits
22 Proper Packaging of RMW Improper Packaging A non-acceptable container may include any of the following: Red Bag not tied Leaking container Objects protruding Improperly closed (includes sharps containers) Overfilled Not properly labeled/marked 22 An improperly packaged container should NEVER be offered for transportation
23 Managing RMW Collection and Storage Proper Storage of RMW Containers Storage must be in containers that are leak resistant, have tight fitting covers and are kept clean and in good repair Secondary containers must be clearly labeled with appropriate symbols on the sides so as to be visible from any lateral direction Warning signs English CAUTION BIOHAZARDOUS WASTE STORAGE AREA UNAUTHORIZED PERSONS KEEP OUT Spanish CUIDADO ZONA DE RESIDUOS BIOLOGICOS PELIGROSOS PROHIBIDA LA ENTRADA A PERSONAS NO AUTORIZADAS 23
24 24
25 Labeling & Documentation Understanding the Shipping Document Typical shipping document varies by state Shipper s Certification: Properly described Properly packaged Properly marked and labeled per US DOT Agreement as to volumes transported RMW cannot be transported without a properly completed and signed shipping document by the generator (shipper). The waste hauler is not authorized to sign the shipping document for the generator (shipper). 25
26 Labeling & Documentation California State Requirement All boxes, reusable containers, and/or secondary containers prepared for shipment shall be marked with a water-resistant tag containing: Generator s (shipper) name and address Transporter s name, address, and permit number Date the transporter accepted the RMW
27 Labeling & Documentation Requirements for Transport DOT Shipping Papers (49 CFR ) Legible, in English DOT Proper shipping description: Waste identification number UN 3291 Name - Regulated Medical Waste, n.o.s. Hazard class and division 6.2 Packaging Group PGII Total Quantity cubic feet or weight Requires that all hazardous materials be accompanied by shipping documents Provide clear communication in event of emergency when hazardous material is in transit Critical for documents to be complete, correct. and to have emergency response number (manned 24/7) Shipper s certification (Generator s Signature) CDPH 27 SQ Generators (shippers) maintain documents for 2 years LQ Generators (shippers) maintain documents for 3 years
28 Emergencies Emergency spill procedures (may include spill kits): Make sure procedures are consistent with Medical Waste Management Plan Post a current list of emergency numbers Be sure to review and update the list annually 28
29 DOT Requirements RMW Container Markings 29
30 Training Requirements Since Regulated Medical Waste contains sharps and potentially infectious substances, all affected employees are required to have initial and annual OSHA Bloodborne Pathogens training The transport of Regulated Medical Waste is regulated by the United States Department of Transportation as a hazardous material while it is in transport. All affected employees (those who perform the functions of either packaging or signing the shipping papers) must complete DOT hazardous materials training initially and every three years thereafter Stericycle can provide the necessary DOT training for your employees. Please contact your sales or customer service representative for details 30
31 Thank you for your interest in the specific laws governing Regulated Medical Waste in California Be sure to also check actual State website for further details and any possible changes to State regulations If you have additional specific questions relating to waste segregation please feel free to Stericycle at 31
Regulated Medical Waste Management and Proper Waste Segregation. Regulated Medical Waste Management and Proper Waste Segregation.
 Regulated Medical Waste Management and Proper Waste Segregation New Jersey New Arkansas Jersey Regulated Medical Waste Management and Proper Waste Segregation The regulations in this presentation are specific
Regulated Medical Waste Management and Proper Waste Segregation New Jersey New Arkansas Jersey Regulated Medical Waste Management and Proper Waste Segregation The regulations in this presentation are specific
Regulated Medical Waste Management and Proper Waste Segregation. State-Specific Information New Jersey
 Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information New Jersey Identifying & Segregating Regulated Medical Waste RMW in New Jersey Six Categories of Regulated Medical
Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information New Jersey Identifying & Segregating Regulated Medical Waste RMW in New Jersey Six Categories of Regulated Medical
Regulated Medical Waste Management and Proper Waste Segregation. Regulated Medical Waste Management and Proper Waste Segregation.
 Regulated Medical Waste Management and Proper Waste Segregation Maine Maine Arkansas Regulated Medical Waste Management and Proper Waste Segregation The regulations in this presentation are specific to
Regulated Medical Waste Management and Proper Waste Segregation Maine Maine Arkansas Regulated Medical Waste Management and Proper Waste Segregation The regulations in this presentation are specific to
Regulated Medical Waste Management and Proper Waste Segregation. State-Specific Information Washington
 Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Washington Biomedical Waste Regulation In the State of Washington biomedical waste, where regulated, is regulated
Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Washington Biomedical Waste Regulation In the State of Washington biomedical waste, where regulated, is regulated
RMW Segregation Training H2E Fall Extravaganza October 2, 2009
 RMW Segregation Training H2E Fall Extravaganza October 2, 2009 WASTE STREAMS Regulated Medical Waste Recyclable Waste Solid Waste (Trash) Medical Waste Pick-up by Stericycle AUTOCLAVABLE WASTE: Red Bag
RMW Segregation Training H2E Fall Extravaganza October 2, 2009 WASTE STREAMS Regulated Medical Waste Recyclable Waste Solid Waste (Trash) Medical Waste Pick-up by Stericycle AUTOCLAVABLE WASTE: Red Bag
Regulated Medical Waste Management and Proper Waste Segregation
 Regulated Medical Waste Management and Proper Waste Segregation Connecticut Connecticut Regulated Medical Waste Management and Proper Waste Segregation The regulations in this presentation are specific
Regulated Medical Waste Management and Proper Waste Segregation Connecticut Connecticut Regulated Medical Waste Management and Proper Waste Segregation The regulations in this presentation are specific
Regulated Medical Waste Management and Proper Waste Segregation. State-Specific Information Pennsylvania
 Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Pennsylvania Identifying & Segregating Regulated Medical Waste RMW in Pennsylvania Six Categories of Regulated
Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Pennsylvania Identifying & Segregating Regulated Medical Waste RMW in Pennsylvania Six Categories of Regulated
DOT Hazardous Materials and Regulated Medical Waste Prepared By: George Weishoff
 DOT Hazardous Materials and Regulated Medical Waste Prepared By: George Weishoff Medical Waste Removal & Disposal Introduction Training will include the following: Code of Federal Regulations: Title 49
DOT Hazardous Materials and Regulated Medical Waste Prepared By: George Weishoff Medical Waste Removal & Disposal Introduction Training will include the following: Code of Federal Regulations: Title 49
Regulated Medical Waste Management and Proper Waste Segregation. State-Specific Information Florida
 Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Florida Biomedical Waste in Florida Regulated by Florida DOH Florida Administrative Code (FAC) 64E-16 Contains
Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Florida Biomedical Waste in Florida Regulated by Florida DOH Florida Administrative Code (FAC) 64E-16 Contains
Infectious Waste Disposal Procedure 25 PA Code 284 Ursinus College Collegeville, PA 19426
 The purpose of the Infectious Waste Disposal procedure is to establish procedures for the safe handling and proper disposal of infectious waste generated at in accordance with regulation 25 PA Code Chapter
The purpose of the Infectious Waste Disposal procedure is to establish procedures for the safe handling and proper disposal of infectious waste generated at in accordance with regulation 25 PA Code Chapter
General Decision Number: CA /02/2015 CA3. Superseded General Decision Number: CA State: California
 General Decision Number: CA150003 01/02/2015 CA3 Superseded General Decision Number: CA20140003 State: California Construction Type: Heavy Water Well Drilling Counties: California Statewide. WATER WELL
General Decision Number: CA150003 01/02/2015 CA3 Superseded General Decision Number: CA20140003 State: California Construction Type: Heavy Water Well Drilling Counties: California Statewide. WATER WELL
Policy & Procedure Manual
 Environmental Health & Safety Policy & Procedure Manual Title: 1. Purpose: To establish policies, work practices, and systematic procedures for the handling, packaging, collection, treatment, and disposal
Environmental Health & Safety Policy & Procedure Manual Title: 1. Purpose: To establish policies, work practices, and systematic procedures for the handling, packaging, collection, treatment, and disposal
LABORATORY WASTE DISPOSAL GUIDE
 LABORATORY WASTE DISPOSAL GUIDE Purpose This guide will help lab members properly identify and manage hazardous waste in their laboratory. Identifying the types of waste generated is crucial to handling
LABORATORY WASTE DISPOSAL GUIDE Purpose This guide will help lab members properly identify and manage hazardous waste in their laboratory. Identifying the types of waste generated is crucial to handling
MEDICAL WASTE MANAGEMENT ACT ATTENTION ALL GENERATORS OF MEDICAL WASTE
 El Dorado County Environmental Management Division Placerville Office: (530) 621-5300 South Lake Tahoe Office: (530) 573-3450 MEDICAL WASTE MANAGEMENT ACT ATTENTION ALL GENERATORS OF MEDICAL WASTE The
El Dorado County Environmental Management Division Placerville Office: (530) 621-5300 South Lake Tahoe Office: (530) 573-3450 MEDICAL WASTE MANAGEMENT ACT ATTENTION ALL GENERATORS OF MEDICAL WASTE The
Biological and Regulated Medical Waste Plan
 Biological and Regulated Medical Waste Plan TUFTS ENVIRONMENTAL HEALTH AND SAFETY OCTOBER 2016 Table of Contents I. Purpose... 3 II. Responsibilities... 3 III. Sink and sewer discharge of specific medical
Biological and Regulated Medical Waste Plan TUFTS ENVIRONMENTAL HEALTH AND SAFETY OCTOBER 2016 Table of Contents I. Purpose... 3 II. Responsibilities... 3 III. Sink and sewer discharge of specific medical
Biomedical Waste Management Guide
 Biomedical Waste Management Guide Yale University Office of Environmental Health & Safety 135 College Street, Suite 100, New Haven, CT 06510 Updated April 2017 Telephone: 203-785-3550 / Fax: 203-785-7588
Biomedical Waste Management Guide Yale University Office of Environmental Health & Safety 135 College Street, Suite 100, New Haven, CT 06510 Updated April 2017 Telephone: 203-785-3550 / Fax: 203-785-7588
NOTE: Do NOT overfill sharps containers; there should be NO more than 50ml of residual liquid in the mailback kit when returned
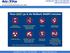 These DO go in the Mailback SHARPS Containers Sharps Waste Such As: Needles & syringes Razor blades Orthodon?c wires Scalpel blades & lancets Glass pipebes, slides & tubes Broken, contaminated glass Staples
These DO go in the Mailback SHARPS Containers Sharps Waste Such As: Needles & syringes Razor blades Orthodon?c wires Scalpel blades & lancets Glass pipebes, slides & tubes Broken, contaminated glass Staples
California Health Benefit Exchange QDP Certification Application for Plan Year 2018 Attachment C1 Current & Projected Enrollment
 Attachment C1 Current & Projected Enrollment Please provide the following for each product (DHMO/DPPO) in the individual market: 1 Effectuated Enrollment as of April 1, 2017. Effectuated means binder payment
Attachment C1 Current & Projected Enrollment Please provide the following for each product (DHMO/DPPO) in the individual market: 1 Effectuated Enrollment as of April 1, 2017. Effectuated means binder payment
Healthcare Waste Regulatory Challenges. Selin Hoboy November 17, 2011
 Healthcare Waste Regulatory Challenges Selin Hoboy November 17, 2011 Overview What are healthcare wastes? What regulations and agencies affect healthcare waste? How to cope with it all! BMPs What are upcoming
Healthcare Waste Regulatory Challenges Selin Hoboy November 17, 2011 Overview What are healthcare wastes? What regulations and agencies affect healthcare waste? How to cope with it all! BMPs What are upcoming
WHO NEEDS TO KNOW THIS PROGRAM All NYU employees that generate, handle or transport RMW should be familiar with this written program.
 Title: NYU Regulated Medical Waste Safety Written Program Effective Date: October 2002 Revision Date: April 14, 2017 Issuing Authority: Responsible Officer: VP, Facilities and Construction Management Director
Title: NYU Regulated Medical Waste Safety Written Program Effective Date: October 2002 Revision Date: April 14, 2017 Issuing Authority: Responsible Officer: VP, Facilities and Construction Management Director
Slide 1. Welcome to the on-line training course for Waste Management!
 Slide 1 Welcome to the on-line training course for Waste Management! Slide 2 Course Information Course Title: Waste Management Non-Clinical Regulations/Standards: PA Department of Health, PA Department
Slide 1 Welcome to the on-line training course for Waste Management! Slide 2 Course Information Course Title: Waste Management Non-Clinical Regulations/Standards: PA Department of Health, PA Department
You are required to complete this course with a passing score of 85% or higher if you:
 OHS Biosafety BIO301L Medical Waste Management for Labs Training You are required to complete this course with a passing score of 85% or higher if you: Offer medical waste for transport from a UAB campus
OHS Biosafety BIO301L Medical Waste Management for Labs Training You are required to complete this course with a passing score of 85% or higher if you: Offer medical waste for transport from a UAB campus
BIOMEDICAL WASTE MANAGEMENT POLICY
 BIOMEDICAL WASTE MANAGEMENT POLICY Managing biomedical waste in a safe and environmentally responsible manner is a key focus of the Environmental Compliance and Sustainability office. 1.0 PURPOSE In an
BIOMEDICAL WASTE MANAGEMENT POLICY Managing biomedical waste in a safe and environmentally responsible manner is a key focus of the Environmental Compliance and Sustainability office. 1.0 PURPOSE In an
INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL
 INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL UNIVERSITY RISK MANAGEMENT Occupational Safety and Health Programs 19 Hagood Avenue, Suite 908 Charleston, SC 29425 843-792-3604 Revised: November 2016 TABLE
INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL UNIVERSITY RISK MANAGEMENT Occupational Safety and Health Programs 19 Hagood Avenue, Suite 908 Charleston, SC 29425 843-792-3604 Revised: November 2016 TABLE
User Guide for the Chief Probation Officers of California Data Dashboard: Juvenile Probation
 User Guide for the Chief Probation Officers of California Data Dashboard: Juvenile Probation I. Introduction This guide will help users understand and customize the data presented in the Chief Probation
User Guide for the Chief Probation Officers of California Data Dashboard: Juvenile Probation I. Introduction This guide will help users understand and customize the data presented in the Chief Probation
CALIFORNIA CODES HEALTH AND SAFETY CODE SECTION
 CALIFORNIA CODES HEALTH AND SAFETY CODE SECTION 117625-117780 117625. Unless the context requires otherwise, the definitions in this article govern the construction of this part. 117630. "Biohazard bag"
CALIFORNIA CODES HEALTH AND SAFETY CODE SECTION 117625-117780 117625. Unless the context requires otherwise, the definitions in this article govern the construction of this part. 117630. "Biohazard bag"
Property Assessed Clean Energy (PACE) Survey
 Final Report Property Assessed Clean Energy (PACE) Survey Understanding Homeowners Motivation for Electing PACE Financing: Discretionary or Non-Discretionary Reasons May 31, 2017 Final Report Property
Final Report Property Assessed Clean Energy (PACE) Survey Understanding Homeowners Motivation for Electing PACE Financing: Discretionary or Non-Discretionary Reasons May 31, 2017 Final Report Property
CAIR Before and After: The Evolution of Implementing a New State Immunization System
 CAIR Before and After: The Evolution of Implementing a New State Immunization System Background Project goal to consolidate CAIR from a seven region registry system into a single statewide immunization
CAIR Before and After: The Evolution of Implementing a New State Immunization System Background Project goal to consolidate CAIR from a seven region registry system into a single statewide immunization
DEPARTMENT OF BIOLOGICAL SCIENCES MEDICAL/BIOHAZARDOUS WASTE DISPOSAL AUTOCLAVE TRAINING
 DEPARTMENT OF BIOLOGICAL SCIENCES MEDICAL/BIOHAZARDOUS WASTE DISPOSAL AUTOCLAVE TRAINING INTRODUCTION In accordance with the California Medical Waste Management Act, Health and Safety Code Stanford University
DEPARTMENT OF BIOLOGICAL SCIENCES MEDICAL/BIOHAZARDOUS WASTE DISPOSAL AUTOCLAVE TRAINING INTRODUCTION In accordance with the California Medical Waste Management Act, Health and Safety Code Stanford University
University Health Services Health and Safety
 ADVISORY NO. 10.2: MANAGEMENT OF BIOLOGICAL AND INFECTIOUS WASTES The following recommendations are based on the Ohio Revised Code (ORC), Ohio Administrative Code (OAC) and EPA guidelines regarding biological
ADVISORY NO. 10.2: MANAGEMENT OF BIOLOGICAL AND INFECTIOUS WASTES The following recommendations are based on the Ohio Revised Code (ORC), Ohio Administrative Code (OAC) and EPA guidelines regarding biological
Title V Changes Regulatory Overview. New EPA Air Quality Standards
 Title V Changes Regulatory Overview New EPA Overview History of the industry standards. What the heightened standards require? What are the goals of these new requirements? What is the timeline for implementation?
Title V Changes Regulatory Overview New EPA Overview History of the industry standards. What the heightened standards require? What are the goals of these new requirements? What is the timeline for implementation?
GUIDELINES FOR TRANSPORTING ANTHRAX AND ANTHRAX-CONTAMINATED OBJECTS AND MATERIALS
 RESEARCH AND SPECIAL PROGRAMS ADMINISTRATION GUIDELINES FOR TRANSPORTING ANTHRAX AND ANTHRAX-CONTAMINATED OBJECTS AND MATERIALS What regulations apply to the transportation of anthrax and other infectious
RESEARCH AND SPECIAL PROGRAMS ADMINISTRATION GUIDELINES FOR TRANSPORTING ANTHRAX AND ANTHRAX-CONTAMINATED OBJECTS AND MATERIALS What regulations apply to the transportation of anthrax and other infectious
LABORATORY WASTE MANAGEMENT PROGRAMMES
 LABORATORY WASTE MANAGEMENT PROGRAMMES Sylvia Wanjiru Kamau INTERNATIONAL LIVESTOCK RESEARCH INSTITUTE LABORATORY MANAGEMENT AND EQUIPMENT OPERATIONS WORKSHOP. 17 TH -21 ST JUNE 2012 TRAINING OBJECTIVES
LABORATORY WASTE MANAGEMENT PROGRAMMES Sylvia Wanjiru Kamau INTERNATIONAL LIVESTOCK RESEARCH INSTITUTE LABORATORY MANAGEMENT AND EQUIPMENT OPERATIONS WORKSHOP. 17 TH -21 ST JUNE 2012 TRAINING OBJECTIVES
Regulated Medical Waste Generator Inspections An Inspector s View Point
 Regulated Medical Waste Generator Inspections An Inspector s View Point Amy Scaffidi, Investigator I Bur. of Hazardous Waste & UST Compliance & Enforcement NJ Dept. of Environmental Protection Phone (609)
Regulated Medical Waste Generator Inspections An Inspector s View Point Amy Scaffidi, Investigator I Bur. of Hazardous Waste & UST Compliance & Enforcement NJ Dept. of Environmental Protection Phone (609)
THE SAFE DISPOSAL OF CLINICAL/DOMESTIC WASTE
 Section V THE SAFE DISPOSAL OF CLINICAL/DOMESTIC WASTE The Trust is currently reviewing the requirements of the recent guidelines Health Technical Memorandum Safe Management of Healthcare Waste (HTML 07-01).
Section V THE SAFE DISPOSAL OF CLINICAL/DOMESTIC WASTE The Trust is currently reviewing the requirements of the recent guidelines Health Technical Memorandum Safe Management of Healthcare Waste (HTML 07-01).
Employee Environmental, Health & Safety Manual. INFECTIOUS WASTE MANAGEMENT PROGRAM Latest Revision: 05/27/08
 Note: Print document as needed for MATC Departmental purposes; it is user responsibility to ensure use of latest revision by CONTENT checking online at: http://matcmadison.edu/ad/facilities/ehs/ under
Note: Print document as needed for MATC Departmental purposes; it is user responsibility to ensure use of latest revision by CONTENT checking online at: http://matcmadison.edu/ad/facilities/ehs/ under
UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES
 UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES October 2016 Table of Contents Section Page I. Introduction.... 1 II. Characteristics of Regulated Medical Waste 1-2 III. Exclusions...2-3
UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES October 2016 Table of Contents Section Page I. Introduction.... 1 II. Characteristics of Regulated Medical Waste 1-2 III. Exclusions...2-3
Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP)
 Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP) NOTE: This SOP for biological waste management does not supersede the requirements for radioactive and/or hazardous
Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP) NOTE: This SOP for biological waste management does not supersede the requirements for radioactive and/or hazardous
Appendix J. University of Alabama at Birmingham Medical Waste Management Plan
 University of Alabama at Birmingham Medical Waste Management Plan I. Definitions per the Alabama Department of Environmental Management Land Division 13-Solid Waste Program, Chapter 335-13-1, Medical Waste
University of Alabama at Birmingham Medical Waste Management Plan I. Definitions per the Alabama Department of Environmental Management Land Division 13-Solid Waste Program, Chapter 335-13-1, Medical Waste
Dartmouth College. Institutional Biosafety Committee. Biohazardous Waste Disposal Guide IBC Approved: 10/3/18
 Biohazardous Waste Disposal IBC Approved: 10/3/18 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical laboratories that may contain infectious
Biohazardous Waste Disposal IBC Approved: 10/3/18 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical laboratories that may contain infectious
Dartmouth College. Institutional Biosafety Committee. Biohazardous Waste Disposal Guide IBC Approved: 4/5/17
 Institutional Biosafety Committee Biohazardous Waste Disposal IBC Approved: 4/5/17 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical
Institutional Biosafety Committee Biohazardous Waste Disposal IBC Approved: 4/5/17 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical
Contents. Introduction
 Introduction Contents Medi-Waste is a specialist company that evolved in 1987 providing the collection, transport and disposal of biomedical waste, (including off spec and out of date pharmaceuticals,
Introduction Contents Medi-Waste is a specialist company that evolved in 1987 providing the collection, transport and disposal of biomedical waste, (including off spec and out of date pharmaceuticals,
SPECIAL WASTE CONTAINERS
 Department of Transportation (DOT) regulations require special packaging, marking and labeling, shipping documentation, emergency response, and employee training when managing regulated waste material.
Department of Transportation (DOT) regulations require special packaging, marking and labeling, shipping documentation, emergency response, and employee training when managing regulated waste material.
ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES
 United States Office of the EPA 233-B-00-001 Environmental Protection Administrator May 2000 Agency (2131) ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES 3.5 Biologically Active Substances and Wastes
United States Office of the EPA 233-B-00-001 Environmental Protection Administrator May 2000 Agency (2131) ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES 3.5 Biologically Active Substances and Wastes
Regulated Medical Waste (RMW) Rules, Registration & Reports
 Regulated Medical Waste (RMW) Rules, Registration & Reports 1 Prepared by: Bret Reburn Envi. Specialist 3 Hazardous Waste/UST Compliance & Enforcement Central Regional Office - Trenton, NJ (609) 292-3949
Regulated Medical Waste (RMW) Rules, Registration & Reports 1 Prepared by: Bret Reburn Envi. Specialist 3 Hazardous Waste/UST Compliance & Enforcement Central Regional Office - Trenton, NJ (609) 292-3949
What Non-Riders Want. Presented by: Joey M. Goldman, Principal CTA Fall Conference, Oakland. November 17, 2016
 What Non-Riders Want Presented by: Joey M. Goldman, Principal CTA Fall Conference, Oakland November 17, 2016 I thank God every day that my children don t have to ride the bus. Parent of a high school student
What Non-Riders Want Presented by: Joey M. Goldman, Principal CTA Fall Conference, Oakland November 17, 2016 I thank God every day that my children don t have to ride the bus. Parent of a high school student
Policy Name: Effective Date: 10/1/2013 Department/ Area Policy #:
 Purpose: An autoclave is a commonly used piece of equipment in biomedical laboratories. Autoclaves pose many hazards including physical hazards (e.g. heat, steam and pressure) and biological hazards. These
Purpose: An autoclave is a commonly used piece of equipment in biomedical laboratories. Autoclaves pose many hazards including physical hazards (e.g. heat, steam and pressure) and biological hazards. These
Protocol for Cytotoxic Spills and Waste Management
 Occupational Health and Safety (OHS) Policy Document Number # QH-PTL-275-3-2:2012 Protocol for Cytotoxic Spills and Waste Management Custodian/Review O fficer: Graham Easterby, a/director, Safety and Wellbeing.
Occupational Health and Safety (OHS) Policy Document Number # QH-PTL-275-3-2:2012 Protocol for Cytotoxic Spills and Waste Management Custodian/Review O fficer: Graham Easterby, a/director, Safety and Wellbeing.
Bloodborne Pathogens Exposure Control Plan
 Bloodborne Pathogens Exposure Control Plan The Texas Department of Insurance, Division of Workers Compensation (TDI-DWC) Provided by Workplace Safety HS96-089C (10/09) Bloodborne Pathogens Exposure Control
Bloodborne Pathogens Exposure Control Plan The Texas Department of Insurance, Division of Workers Compensation (TDI-DWC) Provided by Workplace Safety HS96-089C (10/09) Bloodborne Pathogens Exposure Control
Pharmaceutical Waste Compliance Program Clinician Educators and Managers In-Service
 Pharmaceutical Waste Compliance Program Clinician Educators and Managers In-Service Pharmaceutical Waste Services Stericycle Pharmaceutical Waste Compliance Program Here s our Solution 1. Identification
Pharmaceutical Waste Compliance Program Clinician Educators and Managers In-Service Pharmaceutical Waste Services Stericycle Pharmaceutical Waste Compliance Program Here s our Solution 1. Identification
Zebra and Quagga Mussel Veliger Sampling Protocol Vertical Tow * California Department of Fish and Game
 Zebra and Quagga Mussel Veliger Sampling Protocol Vertical Tow * California Department of Fish and Game *This protocol was adapted from the California Department of Water Resources Zebra and Quagga Mussel
Zebra and Quagga Mussel Veliger Sampling Protocol Vertical Tow * California Department of Fish and Game *This protocol was adapted from the California Department of Water Resources Zebra and Quagga Mussel
Procedure for Health Care Risk Waste Management. Procedure No. 403
 Procedure for Health Care Risk Waste Management Procedure No. 403 Print Name Title Date Prepared by J.G. MacNamara T.S.O. 01/11/04 Reviewed by J. Hoare CATSO 01/11/04 Corporate Authorisation J.G. MacNamara
Procedure for Health Care Risk Waste Management Procedure No. 403 Print Name Title Date Prepared by J.G. MacNamara T.S.O. 01/11/04 Reviewed by J. Hoare CATSO 01/11/04 Corporate Authorisation J.G. MacNamara
Northeastern University Procedure for Disposal of Medical or Biological Waste
 Biohazardous Waste Disposal Fact Sheet Northeastern University Procedure for Disposal of Medical or Biological Waste Fact Sheet #14 August 2016 Revision: Seventh Definition: The State of Massachusetts
Biohazardous Waste Disposal Fact Sheet Northeastern University Procedure for Disposal of Medical or Biological Waste Fact Sheet #14 August 2016 Revision: Seventh Definition: The State of Massachusetts
This policy applies to employees, students and volunteers at all OHSU facilities who work with, generate or dispose of hazardous materials and waste.
 Hazardous Materials and Waste Policy Reviewed: April 2011 Applicability This policy applies to employees, students and volunteers at all OHSU facilities who work with, generate or dispose of hazardous
Hazardous Materials and Waste Policy Reviewed: April 2011 Applicability This policy applies to employees, students and volunteers at all OHSU facilities who work with, generate or dispose of hazardous
KEEPING SHARP Volume 1, Issue 2
 KEEPING SHARP Volume 1, Issue 2 Medical Waste Update HCA Published by Environmental Health Medical Waste, County of Orange Health Care Agency Save & Comply Medical waste treatment and disposal methods
KEEPING SHARP Volume 1, Issue 2 Medical Waste Update HCA Published by Environmental Health Medical Waste, County of Orange Health Care Agency Save & Comply Medical waste treatment and disposal methods
University of Tennessee Health Science Center. Skills Transfer Center. Exposure Control Plan. Hamilton Eye Institute. 930 Madison Avenue, Third Floor
 University of Tennessee Health Science Center Skills Transfer Center Exposure Control Plan Hamilton Eye Institute 930 Madison Avenue, Third Floor Memphis, Tennessee Skills Transfer Center Hamilton Eye
University of Tennessee Health Science Center Skills Transfer Center Exposure Control Plan Hamilton Eye Institute 930 Madison Avenue, Third Floor Memphis, Tennessee Skills Transfer Center Hamilton Eye
FIELD RESEARCH CORPORATION
 FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Registered Voters About
FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Registered Voters About
Environment, Health and Safety
 IlJ!UNC FINANCE AND OPERATIONS Environment, Health and Safety Chapter 15: Safe Handling of Biological Hazards This chapter is an overview of the requirements for working with biological hazards. You can
IlJ!UNC FINANCE AND OPERATIONS Environment, Health and Safety Chapter 15: Safe Handling of Biological Hazards This chapter is an overview of the requirements for working with biological hazards. You can
OFFICE OF CONTRACT ADMINISTRATION PURCHASING DIVISION Bid Addendum No. 2
 1. Summary of Service Requirements: SCOPE OF SERVICES AND SPECIAL CONTRACT REQUIREMENTS MEDICAL WASTE MANAGEMENT SERVICES CITY AND COUNTY OF SAN FRANCISCO 1.1. The City and County of San Francisco (CCSF)
1. Summary of Service Requirements: SCOPE OF SERVICES AND SPECIAL CONTRACT REQUIREMENTS MEDICAL WASTE MANAGEMENT SERVICES CITY AND COUNTY OF SAN FRANCISCO 1.1. The City and County of San Francisco (CCSF)
Biological Materials and Biohazardous/Medical Waste Disposal Program
 Biological Materials and Biohazardous/Medical Waste Disposal Program Document Number: EHS.BIOW.08.02 Effective Date: 7/18/08 Revision Date: 7/15/10 1.0 Purpose and Applicability 1.1. Biological waste is
Biological Materials and Biohazardous/Medical Waste Disposal Program Document Number: EHS.BIOW.08.02 Effective Date: 7/18/08 Revision Date: 7/15/10 1.0 Purpose and Applicability 1.1. Biological waste is
FIELD RESEARCH CORPORATION
 FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Voters About Voting Preferences
FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Voters About Voting Preferences
HackensackUMC Sustainability Program. Presented by Kyle Tafuri Sustainability Advisor
 HackensackUMC Sustainability Program Presented by Kyle Tafuri Sustainability Advisor About HackensackUHN HackensackUMC HackensackUMC Mountainside HackensackUMC at Pascack Valley Palisades Healthcare System
HackensackUMC Sustainability Program Presented by Kyle Tafuri Sustainability Advisor About HackensackUHN HackensackUMC HackensackUMC Mountainside HackensackUMC at Pascack Valley Palisades Healthcare System
Medical Waste Information Bulletin and Disposal Guide
 Public Health Department Cathy A. Raevsky, Director Andy Miller, M.D., Health Officer Environmental Health 202 Mira Loma Drive T: 530.538.7281 Oroville, California 95965 F: 530.538.5339 buttecounty.net/publichealth
Public Health Department Cathy A. Raevsky, Director Andy Miller, M.D., Health Officer Environmental Health 202 Mira Loma Drive T: 530.538.7281 Oroville, California 95965 F: 530.538.5339 buttecounty.net/publichealth
BIOMEDICAL WASTE PLAN Health Sciences Campus / Biological Sciences Titusville Campus
 Courtesy of: BIOMEDICAL WASTE PLAN Health Sciences Campus / Biological Sciences Brevard Community College Environmental Health Services 2725 Judge Fran Jamieson Way, Bldg. A Viera, FL 32940 (321) 633-2053
Courtesy of: BIOMEDICAL WASTE PLAN Health Sciences Campus / Biological Sciences Brevard Community College Environmental Health Services 2725 Judge Fran Jamieson Way, Bldg. A Viera, FL 32940 (321) 633-2053
UN 3291 Clinical and Related Wastes Services Specification
 SSS013 UN 3291 Clinical and Related Wastes Services Specification 1. Scope This specification describes the acceptable waste materials and packaging requirements for Daniels Health services for a subset
SSS013 UN 3291 Clinical and Related Wastes Services Specification 1. Scope This specification describes the acceptable waste materials and packaging requirements for Daniels Health services for a subset
Inspection Directions
 Inspection Directions To ensure compliance with OSHA regulations, annual training is a must. Here s what to cover and how to tackle it. by Leslie Canham, CDA, RDA Leslie Canham, CDA, RDA, is an international
Inspection Directions To ensure compliance with OSHA regulations, annual training is a must. Here s what to cover and how to tackle it. by Leslie Canham, CDA, RDA Leslie Canham, CDA, RDA, is an international
A Better Way: Product Stewardship
 A Better Way: Product Stewardship Mission To shift California s i product waste management system from one focused on government tfunded dand ratepayer financed waste diversion to one that relies on producer
A Better Way: Product Stewardship Mission To shift California s i product waste management system from one focused on government tfunded dand ratepayer financed waste diversion to one that relies on producer
BIO-MEDICAL WASTE MANAGEMENT (AMENDMENT) RULES -2018
 1 BIO-MEDICAL WASTE MANAGEMENT (AMENDMENT) RULES -2018 BIOMEDICAL WASTE RULES APPLY TO:- All who generate, collect, receive, store, transport, treat, dispose, or handle bio medical waste :- Hospitals,
1 BIO-MEDICAL WASTE MANAGEMENT (AMENDMENT) RULES -2018 BIOMEDICAL WASTE RULES APPLY TO:- All who generate, collect, receive, store, transport, treat, dispose, or handle bio medical waste :- Hospitals,
Impacts of MTBE on California Groundwater
 Impacts of MTBE on California Groundwater Graham E. Fogg, Mary E. Meays, James C. Trask, Christopher T. Green, Eric M. LaBolle, Timothy W. Shenk, and Dennis E. Rolston Hydrologic Sciences, Department of
Impacts of MTBE on California Groundwater Graham E. Fogg, Mary E. Meays, James C. Trask, Christopher T. Green, Eric M. LaBolle, Timothy W. Shenk, and Dennis E. Rolston Hydrologic Sciences, Department of
Access to Paid Sick Days in California
 IWPR #B342 August 2014 Access to Paid Sick Days in California An analysis by the Institute for Women s Policy Research (IWPR) finds that approximately 44 percent of workers living in California lack even
IWPR #B342 August 2014 Access to Paid Sick Days in California An analysis by the Institute for Women s Policy Research (IWPR) finds that approximately 44 percent of workers living in California lack even
SECTION.1200 MEDICAL WASTE MANAGEMENT 15A NCAC 13B.1201 DEFINITIONS
 SECTION.1200 MEDICAL WASTE MANAGEMENT 15A NCAC 13B.1201 DEFINITIONS (1) "Blood and body fluids" means liquid blood, serum, plasma, other blood products, emulsified human tissue, spinal fluids, and pleural
SECTION.1200 MEDICAL WASTE MANAGEMENT 15A NCAC 13B.1201 DEFINITIONS (1) "Blood and body fluids" means liquid blood, serum, plasma, other blood products, emulsified human tissue, spinal fluids, and pleural
SOP BIO-006 USE OF AUTOCLAVE FOR STERILIZATION OF MATERIALS AND BIOLOGICAL WASTE
 ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-006 USE OF AUTOCLAVE FOR STERILIZATION OF MATERIALS
ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-006 USE OF AUTOCLAVE FOR STERILIZATION OF MATERIALS
WASTE MANAGEMENT GUIDE. Creighton University Environmental Health and Safety
 WASTE MANAGEMENT GUIDE Creighton University Environmental Health and Safety www.creighton.edu/ehs 402 546 6400 Overview With over 150 teaching and research labs, as well as other areas such as fine arts
WASTE MANAGEMENT GUIDE Creighton University Environmental Health and Safety www.creighton.edu/ehs 402 546 6400 Overview With over 150 teaching and research labs, as well as other areas such as fine arts
RECOMMENDED AREA DESIGNATIONS FOR THE PPM FEDERAL 8-HOUR OZONE STANDARD STAFF REPORT
 California Air Resources Board Air Quality Planning and Science Division Air Quality Analysis Section RECOMMENDED AREA DESIGNATIONS FOR THE 0.070 PPM FEDERAL 8-HOUR OZONE STANDARD STAFF REPORT September
California Air Resources Board Air Quality Planning and Science Division Air Quality Analysis Section RECOMMENDED AREA DESIGNATIONS FOR THE 0.070 PPM FEDERAL 8-HOUR OZONE STANDARD STAFF REPORT September
FIELD RESEARCH CORPORATION
 FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Registered Voters about
FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Registered Voters about
Three Federal Pesticide Injunctions for Protection of Endangered Species
 Three Federal Pesticide Injunctions for Protection of Endangered Species Sustainable Ag Expo, Monterey November 15, 2010 Patricia Matteson Pest Mgmt. & Licensing Branch Dept. of Pesticide Regulation DPR
Three Federal Pesticide Injunctions for Protection of Endangered Species Sustainable Ag Expo, Monterey November 15, 2010 Patricia Matteson Pest Mgmt. & Licensing Branch Dept. of Pesticide Regulation DPR
2/07. PROCEDURE for DISPOSAL of RADIOACTIVE MATERIALS
 2/07 CORNELL UNIVERSITY PROCEDURE for DISPOSAL of RADIOACTIVE MATERIALS This procedure has been developed to ensure the safety of those individuals who handle radioactive waste and to comply with University,
2/07 CORNELL UNIVERSITY PROCEDURE for DISPOSAL of RADIOACTIVE MATERIALS This procedure has been developed to ensure the safety of those individuals who handle radioactive waste and to comply with University,
MEDICAL WASTE MANAGEMENT PLAN
 MEDICAL WASTE MANAGEMENT PLAN California Polytechnic State University San Luis Obispo, CA 93407 Environmental Health & Safety (805) 756-6662 July 2017 Type of Facility California Polytechnic State University
MEDICAL WASTE MANAGEMENT PLAN California Polytechnic State University San Luis Obispo, CA 93407 Environmental Health & Safety (805) 756-6662 July 2017 Type of Facility California Polytechnic State University
Biomedical Waste Management Plan
 Biomedical Waste Management Plan USF Biomedical Waste Management Plan for: Facility Address: Facility Phone Number: Facility Contact: Division of Environmental Health and Safety 4202 E. Fowler Ave, OPM
Biomedical Waste Management Plan USF Biomedical Waste Management Plan for: Facility Address: Facility Phone Number: Facility Contact: Division of Environmental Health and Safety 4202 E. Fowler Ave, OPM
Clinical Waste. Policy. Responsibilities
 Clinical Waste Policy Clinical waste is defined as hazardous or offensive waste arising from: - any dental, medical, nursing or veterinary practice, or any other practice or establishment providing medical
Clinical Waste Policy Clinical waste is defined as hazardous or offensive waste arising from: - any dental, medical, nursing or veterinary practice, or any other practice or establishment providing medical
Biosafety Checklist. 07-BiosafetyChecklist-LTC-SOP-v2.0-17Feb of 6
 Title: Biosafety Checklist Origination Date: 31 Jan 1997 Total Pages: 5 Effective Date: 17 Feb 2012 SOP Number LTC-SOP-07 v2.0 Supersedes SOP Written By: ACTG/IMPAACT Lab Tech Committee Dated: 01 Jun 2004
Title: Biosafety Checklist Origination Date: 31 Jan 1997 Total Pages: 5 Effective Date: 17 Feb 2012 SOP Number LTC-SOP-07 v2.0 Supersedes SOP Written By: ACTG/IMPAACT Lab Tech Committee Dated: 01 Jun 2004
FIELD RESEARCH CORPORATION
 FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Registered Voters Regarding
FIELD RESEARCH CORPORATION FOUNDED IN 1945 BY MERVIN FIELD 601 California Street, Suite 900 San Francisco, California 94108 415-392-5763 Tabulations From a Survey of California Registered Voters Regarding
UNIVERSITY OF TOLEDO
 UNIVERSITY OF TOLEDO SUBJECT: INFECTIOUS WASTE DISPOSAL Procedure No: HM-08-019 PROCEDURE STATEMENT Departments or laboratories generating infectious waste shall develop procedures for identification,
UNIVERSITY OF TOLEDO SUBJECT: INFECTIOUS WASTE DISPOSAL Procedure No: HM-08-019 PROCEDURE STATEMENT Departments or laboratories generating infectious waste shall develop procedures for identification,
Regulated Waste Management Plan
 Regulated Waste Management Plan November 1, 2016 (First approved: June 1, 2006) Maintained online by the Environmental Health & Safety Committee Regulated Waste Management Plan -1- Section 1: Introduction
Regulated Waste Management Plan November 1, 2016 (First approved: June 1, 2006) Maintained online by the Environmental Health & Safety Committee Regulated Waste Management Plan -1- Section 1: Introduction
MEDICAL WASTE MANAGEMENT STUDY NOTES
 MEDICAL WASTE MANAGEMENT STUDY NOTES UNIT 1 INTRODUCTION Hospital and other health care establishment has a duty of care for the environment and for public health and have particular responsibility in
MEDICAL WASTE MANAGEMENT STUDY NOTES UNIT 1 INTRODUCTION Hospital and other health care establishment has a duty of care for the environment and for public health and have particular responsibility in
Standard Operating Procedure Title: Good laboratory practices (GLP) for microbiology and chemistry laboratories
 Standard Operating Procedure Title: Good laboratory practices (GLP) for microbiology and chemistry laboratories Department Micro Laboratory Document no MICLAB 155 Title Good laboratory practices (GLP)
Standard Operating Procedure Title: Good laboratory practices (GLP) for microbiology and chemistry laboratories Department Micro Laboratory Document no MICLAB 155 Title Good laboratory practices (GLP)
OCCUPATIONAL EXPOSURE TO BLOODBORNE PATHOGENS
 University Policies and Procedures 06-11.01 OCCUPATIONAL EXPOSURE TO BLOODBORNE PATHOGENS I. Policy Statement: It is recognized that certain job activities at Towson University ( University ) may result
University Policies and Procedures 06-11.01 OCCUPATIONAL EXPOSURE TO BLOODBORNE PATHOGENS I. Policy Statement: It is recognized that certain job activities at Towson University ( University ) may result
Biosafety Level 1 ( BSL1 ) and Biosafety Level 2 ( BSL2 ) Biological Waste Management Guideline
 Implementation Page #: Page 1 of 14 Last Reviewed/Update November 2015 Biosafety Level 1 ( BSL1 ) and Biosafety Level 2 ( BSL2 ) Biological Waste Guideline 1.0 Purpose and Scope The purpose of this Guideline
Implementation Page #: Page 1 of 14 Last Reviewed/Update November 2015 Biosafety Level 1 ( BSL1 ) and Biosafety Level 2 ( BSL2 ) Biological Waste Guideline 1.0 Purpose and Scope The purpose of this Guideline
New Jersey Department of Environmental Protection. Transportation Oversight Unit
 New Jersey Department of Environmental Protection Transportation Oversight Unit Bob Gomez, Supervisor 9 Ewing St., Mail Code 09-01 P.O. Box 420 Trenton, New Jersey 08625-0420 (609) 292-6305 DOT Requirements
New Jersey Department of Environmental Protection Transportation Oversight Unit Bob Gomez, Supervisor 9 Ewing St., Mail Code 09-01 P.O. Box 420 Trenton, New Jersey 08625-0420 (609) 292-6305 DOT Requirements
UNIVERSITY OF TOLEDO HEALTH SCIENCE CAMPUS
 UNIVERSITY OF TOLEDO HEALTH SCIENCE CAMPUS SUBJECT: SPECIMEN TRANSPORT IN Procedure No: S-08-011 COMPUTERIZED TUBE SYSTEM PROCEDURE STATEMENT All laboratory specimens will be transported in a manner consistent
UNIVERSITY OF TOLEDO HEALTH SCIENCE CAMPUS SUBJECT: SPECIMEN TRANSPORT IN Procedure No: S-08-011 COMPUTERIZED TUBE SYSTEM PROCEDURE STATEMENT All laboratory specimens will be transported in a manner consistent
Section: Administration Date of Issue: 05/09/2011 Issued By: Administration Part: Pneumatic Tube System Revision #: Revision Date:
 STANDARD OPERATING PROCEDURE VETERINARY HEALTH COMPLEX Section: Administration Date of Issue: 05/09/2011 Issued By: Administration Part: Pneumatic Tube System Revision #: Revision Date: Pages: Board Approval
STANDARD OPERATING PROCEDURE VETERINARY HEALTH COMPLEX Section: Administration Date of Issue: 05/09/2011 Issued By: Administration Part: Pneumatic Tube System Revision #: Revision Date: Pages: Board Approval
Radioactive Waste Radiation Safety Orientation Open Source Booklet 7 (June 1, 2018)
 Radioactive Waste Radiation Safety Orientation Open Source Booklet 7 (June 1, 2018) For more information, refer to the Radiation Safety Manual, 2017 RSP-3 and the Quick Step for waste in the radiation
Radioactive Waste Radiation Safety Orientation Open Source Booklet 7 (June 1, 2018) For more information, refer to the Radiation Safety Manual, 2017 RSP-3 and the Quick Step for waste in the radiation
AREAS OF RESPONSIBILITY
 Applies To: UNM Hospitals Responsible Department: Infection Prevention & Control Revised: 2/2017 Procedure Patient Age Group: (X ) N/A ( ) All Ages ( ) Newborns ( ) Pediatric ( ) Adult DESCRIPTION/OVERVIEW
Applies To: UNM Hospitals Responsible Department: Infection Prevention & Control Revised: 2/2017 Procedure Patient Age Group: (X ) N/A ( ) All Ages ( ) Newborns ( ) Pediatric ( ) Adult DESCRIPTION/OVERVIEW
Pointers on Shipping: Clinical Samples, Biological Substance Category B and Environmental Test Samples
 Pointers on Shipping: Clinical Samples, Biological Substance Category B and Environmental Test Samples At FedEx, we understand the importance of ensuring the safe shipping of clinical samples such as human
Pointers on Shipping: Clinical Samples, Biological Substance Category B and Environmental Test Samples At FedEx, we understand the importance of ensuring the safe shipping of clinical samples such as human
INDEX. Laboratory Safety Guide
 INDEX A accident reporting, 1, 70, 125 acids, 154 neutralization, 164-168 strength table, 165 acrylamide, 154 acute toxicity, 31, 313, 314 acute toxicity classes, 32 administrative controls (fume hoods),
INDEX A accident reporting, 1, 70, 125 acids, 154 neutralization, 164-168 strength table, 165 acrylamide, 154 acute toxicity, 31, 313, 314 acute toxicity classes, 32 administrative controls (fume hoods),
PI s Name Date Bldg./Rm#
 PI s Name Date Bldg./Rm# Animal Biosafety Level 3 (ABSL-3) Yes No 1. Is access to the animal facility limited or restricted only to those persons authorized for program or support purposes? Yes No 2. Does
PI s Name Date Bldg./Rm# Animal Biosafety Level 3 (ABSL-3) Yes No 1. Is access to the animal facility limited or restricted only to those persons authorized for program or support purposes? Yes No 2. Does
ACP.HLB PROGRAM UPDATE
 ACP.HLB PROGRAM UPDATE California Department of Food and Agriculture Protecting California s agricultural and natural resources against damage caused by exotic plant pests and diseases 1 BACKGROUND.OVERVIEW
ACP.HLB PROGRAM UPDATE California Department of Food and Agriculture Protecting California s agricultural and natural resources against damage caused by exotic plant pests and diseases 1 BACKGROUND.OVERVIEW
r l i u r a Farming is Everywhere
 C A g r a i i c n u r l o t f i u l re a Farming is Everywhere Hello! Think about your clothes, paper, shoes, toothpaste, and basketball. Do you ever wonder what these products have in common? They all
C A g r a i i c n u r l o t f i u l re a Farming is Everywhere Hello! Think about your clothes, paper, shoes, toothpaste, and basketball. Do you ever wonder what these products have in common? They all
Animal cell and tissue culture. Lab 1
 Animal cell and tissue culture Lab 1 Tissue culture Laboratory Safety Outline Lab Safety Biohazards Biosafety Levels Biosafety Cabinets Decontamination Biological Waste Introduction A cell culture laboratory
Animal cell and tissue culture Lab 1 Tissue culture Laboratory Safety Outline Lab Safety Biohazards Biosafety Levels Biosafety Cabinets Decontamination Biological Waste Introduction A cell culture laboratory
Amherst College. Health and Safety Manual for the Use of Recombinant DNA and Biological Agents Current as of May 14, 2010
 Amherst College Health and Safety Manual for the Use of Recombinant DNA and Biological Agents Current as of May 14, 2010 Introduction This manual contains the basic procedures to be followed for all work
Amherst College Health and Safety Manual for the Use of Recombinant DNA and Biological Agents Current as of May 14, 2010 Introduction This manual contains the basic procedures to be followed for all work
