Support Services STERILE PROCESSING SERVICES
|
|
|
- Chad Beasley
- 6 years ago
- Views:
Transcription
1 Section 2 linical Services.1 SERVIE DESRIPTION.1.1 Scope of linical Services This section sets out the requirements for the centralized facilities for the Facility's Sterile Processing Services Department (SPD) to be achieved or accommodated by Project o in providing the Works and the Services. The range of services to be provided within this component includes: Decontamination and disassembling/processing ing/sanitizing Pasteurizing of respiratory equipment leaning/assembly Sterilizing (steam only) Sterile storage for case cart items and items destined for care units SPD administration ase cart picking from sterile storage Sterile Processing Services for the Abbotsford Hospital & ancer entre will include the processing of most supplies and equipment requiring on-site preparation, including the controlled sterilization of most goods and equipment in support of the surgical case cart system, cleaning and control of certain equipment required by selected care units, and preparation and control of special trays for selected areas (e.g., Medical Imaging, outpatient clinics, Emergency, General Day are Unit, respiratory therapy, Laboratory (glassware), and the ancer entre). All items requiring ethylene oxide (ETO) sterilization will be transferred off-site to one of FHA s other facilities and then returned to the sterile storage area ready for distribution. The ancer entre s requirements will comprise 1-day sterilization turn around of re-usable supplies and equipment used by ambulatory care, chemotherapy/medical day care, dentistry and radiation therapy, plus daily top-up of the sterilized equipment inventory (workload details/weekly). Items such as regular patient beds, stretchers, and wheelchairs will not be handled in this area. These items will be washed on the inpatient units and will be sent to Plant Services for repair when necessary. Stretchers and wheelchairs will be cleaned by Housekeeping staff. Patient equipment such as infusion pumps, and suction devices, all respiratory equipment, ventilators, special beds, etc. will be routed through this area, with patient contact surfaces and components changed, washed, or sterilized as required urrent Trends In providing the Works and Services, Project o shall take into account the following trends: Increased requirements and changing standards resulting from new infectious diseases (e.g., SARS, JD). Increasing use of disposables, including wrappers for bundles and custom procedure packs. 399
2 Section 2 linical Services.1.2 Scope of Education Services None.1.3 Scope of Research Services None.1.4 Specific Exclusions This specification excludes sterile processing services/ requirements provided elsewhere, including: Materiel Services (see section E7 Materiel Services) Surgical Services (see section 7 Surgical Services) General Day are Unit (endoscopy) (see section A3 General Day are Unit).2 OPERATIONAL DESRIPTION.2.1 Minimum Hours of Operation Hours of operation for the component are 0600h to 2400h, 7 days/week..2.2 Patient Management Processes Not applicable.2.3 Patient Information Management SPD will be connected to the OR booking management system, receive slates and the pick lists for all OR cases. Also refer to Output Specifications, Section 3: Non-linical Services, subsection D1 Information Management; Section 5: Design and Technical, subsection Technology and ommunication Systems; and Section 6: IT/Tel Services. 400
3 Section 2 linical Services.2.4 Staff Work Processes Disassembly/Decontamination/Processing The decontamination area provides a centralized area for the cleaning of all items (including all surgical instruments) following use in any functional area of the facilities. AH Return This function is organized internally into three major areas corresponding to the sequential flow of soiled items through the decontamination process, as follows: Receiving, sorting, washing, sanitizing/sterilizing art wash Equipment wash Receiving, sorting, washing, sanitizing/sterilizing provides an initial entry point for soiled materials. These may be on carts, on manual conveyances or hand-carried. All items are separated initially by type, processing method and size. Large items such as equipment are routed directly to the equipment wash area. Respiratory equipment will be processed through liquid sterilizing (pasteurmatic) machines and dryers. Delicate equipment will be processed through ultrasonic machines. Patient contact surfaces and components are removed from the equipment for disposal or sterilization. The remainder of the equipment is decontaminated normally by washing and/or disinfection, and then transferred to the clean equipment assembly/storage area for reassembly and holding. Dedicated Dedicated lean Decontamination Assembly/ Sterilization Sterile Storage art Assembly AH lean Delivery Process Flow Diagram Sorted items are moved to the appropriate decontamination equipment and automatically processed. linen and trash, if received, will be collected in hampers and later transferred to their respective collection destinations. Following decontamination operations, all items are unloaded into the packaging and preparation area for subsequent processing. lean racks and similar containers are returned to decontamination for re-use following the removal of the clean materials. All soiled carts used to transfer soiled materials are collected following unloading and routed to the cart wash facilities for decontamination. There, the carts/conveyances are automatically cleaned and dried. They are transferred after decontamination into the cart queuing area for re-use or holding. The placement of the cart wash facility should be contiguous to the final cart unloading area to eliminate needless circulation. 401
4 Section 2 linical Services lean Supplies Packaging/Assembly/Sterile Storage/ lean and sanitized/sterilized items arrive in the packaging and assembly area following the decontamination process. Small items are removed from the racks, trays or other containers and placed on sorting tables. Equipment is reassembled and either stored in the equipment stores/assembly room or returned via dispatch to the department to which it belongs. arts are routed either to cart assembly and holding, case cart marshalling or to the department to which they belong. Small items are sorted, instruments placed in the instrument library and items transferred to the setup/packaging tables for processing. The primary function of packaging and assembly is to prepare various instruments, utensils and supplies in packs, trays and packages for issue. Packaging and assembly staff will select items needed, inspect for quality and assemble them into standard and special order packs, trays or packages. Disposable supply goods necessary for the assembly of packs, trays or packages such as needles, soft goods, etc. are supplied by sterile stores. Linens, wrappers, towels, etc. are supplied as part of the E6 Service ategory. All items requiring sterilization are loaded into sterilizer carts upon completion. Any items not requiring sterilization are transferred to clean storage. Linen bundles and processed goods loaded on autoclave cart and awaiting sterilization are held in a waiting area near the sterilizers. The steam sterilizers are loaded and when the cycle is completed goods are removed and held in a cooling area. After goods have been cooled they are placed on mobile shelving in the sterile storage area. ase cart goods are stored together in one area of sterile storage on carousels. All other sterile goods are stored on wire carts allowing medical/surgical supply carts to be wheeled through the storage area for cart replenishment. After case carts have been assembled, they are held in the cart marshalling area for dispatch via a dedicated lift to the sterile core areas of the Surgical Suite and Maternal are Unit. Picking items for case carts will utilize automated software supply management systems integral to the carousels. ase carts for Medical Imaging will be delivered by Materiel Services portering personnel. Equipment and other large apparatus which are not easily stored in the processed stores mobile carts or shelves are kept and maintained in this space. Similar to processed stores, items of equipment are required as common issue on a regular basis as supplements to shelf top-up supplies and equipment continuously stored in user components..2.5 Materiel Services Refer to Output Specifications, Section 4: Facility Management Services, subsection E7 Materiel Services..2.6 Linen/Housekeeping Services Refer to Output Specifications, Section 4: Facility Management Services, subsections E5 Housekeeping Services and E6 Laundry/Linen Services. 402
5 Section 2 linical Services.2.7 Equipment Asset Management Refer to Output Specifications, Section 4: Facility Management Services, subsection E2 Biomedical Engineering; and Section 7: Equipment..3 ATIVITY INDIATORS The table below summarized the projected activity for Sterile Processing Services which must be addressed by Project o in performing the Works and the Services..3.1 Hospital Activity Unit Minimum Projected Yearly Activity Sterile Processing Services # Beds 300 # Surgical ases 14,000 # Surgery Hours 18,200 # Births 2,200 # ystoscopies 3,749 # Endoscopies 6,748 # Bronchoscopies 350 # Minor OR ases 7, ancer entre Activity (Incl. in Hospital Activity above).4 PEOPLE REQUIREMENTS This component will have a total staff complement in the range of 21 FTE, consisting of 1 coordinator, 1 supervisor, and 19 SPD aides. It is anticipated that the key functional areas in the component will need to accommodate the following maximum number of people. Functional Areas Patients Staff Visitors Others Total Administrative Area Decontamination Area lean/assembly Area Sterilization Area Sterile Storage/lean Supplies/Assembly Area
6 Section 2 linical Services.5 DESIGN RITERIA.5.1 Key External Relationships The following key relationships will be achieved in the priority order as numbered for the purposes stated: STERILE PROESSING SERVIES 1 Materiel Services 1 Provide direct access by internal circulation to Materiel Services for the movement of supplies, staff and waste. 2 3 Surgical Services 2 Provide direct access by internal circulation (if on a different level, by dedicated clean and soiled elevators) to Surgical Services (the Surgical Suite) for the movement of case carts and other supplies. 4 Maternal hild Program Intensive/Stepdown are Units 3 Provide direct access by internal circulation (if on a different level, by dedicated clean and soiled elevators) to the Maternal hild Program (LBRP area) for the movement of case carts and other supplies. 5 Service s 4 Provide direct access by general circulation to Intensive/Stepdown are Units (respiratory therapy area) for the movement of equipment. 5 Provide direct access by general circulation to the service elevators for the movement of supplies and equipment..5.2 KEY INTERNAL RELATIONSHIPS/ ENVIRONMENTAL ONSIDERATIONS The following will be achieved: Zoning Physical zones are to be established to achieve separation of flows for clean and soiled materials within the component. The SPD staff working within the assembly and sterile supplies areas of the SPD should be segregated from staff working within the soiled areas of the SPD. Provide controlled access to the decontamination and sterile processing areas of the component Sterile Supplies Flow In the Facility sterile supplies should not cross paths with soiled materials or the public, and those destined for the Surgical Suite and Maternal hild Program should leave and return the component via a dedicated elevator. 404
7 Section 2 linical Services Lighting Provide glare-free full-spectrum, artificial or natural, lighting and special task types for technical work areas. The concept of "indirect" lighting should be used if reasonably possible. Interior design should ensure simple unobtrusive colour schemes that minimize interference with the concentration of the SPD staff working on fine detail. Also refer to Output Specifications, Section 5: Design and Technical, subsection Lighting ontrol Ergonomics onsiderations Refer to Output Specifications, Section 1: Key Site and Building Design riteria, subsection Ergonomics. 405
8 Section 2 linical Services omponent Functional Diagrams The spatial organization of this component will be generally as shown in the diagrams below. Macro Relationship Diagram Dedicated Dedicated lean Service s In lean Out Supplies Out Service orridor Receive/ Hold/ Sort ase arts from O.R. / LBRP Elev. lean Elev. Equip. lean ase arts to O.R. / LBRP ase art Assembly Holding/lean Equipment Hold Loading Dock Waste Holding Supplies/ Returns In/Out Waste Out returns pass-thru art Sterile/lean Stores Bulk Storage instruments pass-thru Auto Legend Immediately Adjacent Direct Access lean Assembly Sterilizers Reasonably lose Access Direct Visual Supervision lean Supplies Entry Linen Pack Entry E7 Materiel Services w Ster ers Sterilizers 406
9 Section 2 linical Services Micro Relationship Diagram ase arts out to other areas (e.g., Medical Imaging) Decontamination Area Holding Returns lean Hskp Unloading/ Sorting Equip. Resp. 1 lean Equip. lean Resp. ase art Hold Small Instr. Auto ers W W W W art Staff Gown Empty arts lean Assembly Area Instr. Pass-thru Returns Pass-thru Assembly Workstations Sterile Stores lean Supplies Entry Office oord. Workstn., Supervisor Ster Ster Ster ool Administrative Area Sterilization Area Sterile/Storage/Distribution Service Access Legend Immediately Adjacent Direct Access Reasonably lose Access Direct Visual Supervision w Ster ers Sterilizers Note: 1 Part (or all) of respiratory equipment may be routed through the assembly area instead of directly through to the distribution area. 2 Ethylene oxide (ETO) is not part of the plan for this site. ETO requirements will be met by accessing FHA services available at another site. 407
10 Section 2 linical Services.5.3 Schedule of Accommodation (Note: Spaces listed in parentheses ( ) are spaces supporting services provided by Project o and are included in the total net square metres.) Area Requirements Ref Space units nsm/unit nsm Administrative Area 01 Office, oordinator Office, Supervisor Break Room, Staff Staff hange/shower/room, Female room, Staff, Female Staff hange/shower/room, Male room, Staff, Male Subtotal 68.0 Decontamination Area 08 ase art Holding Area Storage, Detergent Receiving/ Returns Holding Area Unloading/Sorting/ing Area Holding Room Automatic ing/sanitizing Area art Area Equipment Area Respiratory Equipment Area Housekeeping loset () 1 (5.0) Subtotal
11 Section 2 linical Services Area Requirements Ref Space units nsm/unit nsm lean/assembly/prep Area 18 lean Equipment/ase art (35) Holding Area art Wipedown/Drying Area Packaging and Assembly Area lean Linen Pack Holding Area 1 (5.0) Subtotal Sterilization Area 22 Sterilizing Area, Steam Holding Area, Transfer arts/ ooling Area Quarantine Holding Area Subtotal 39.0 Sterile Storage/lean Supplies/Picking Area 25 Sterile Stores Pneumatic Tube Station ase art (Empty) Holding Area ase art/sterile Supply art Holding Area Orthopedic Supply Room Housekeeping loset (lean) 1 (5.0) Subtotal Total
12 Section 2 linical Services.6 DESIGN GUIDANE Project o shall comply with: anadian Standards Association (Z314.1, Z314.2, Z314.8-M88, Z , Z , Z314.3-M91. Z ).7 OTHER SPEIFIATIONS Sterile processing services are primarily based in the Sterile Processing Services component, however, other specifications that will be consulted are: E7 Materiel Services 7 Surgical Serves 410
35.0 Sterile Supply Unit (SSU)
 35.0 Sterile Supply Unit (SSU) 35.1 Introduction 35.1.1 General A Hospital must provide adequate facilities for cleaning, sterilisation and storage of equipment and instruments to ensure the care and safety
35.0 Sterile Supply Unit (SSU) 35.1 Introduction 35.1.1 General A Hospital must provide adequate facilities for cleaning, sterilisation and storage of equipment and instruments to ensure the care and safety
QUALITY ASSURANCE IN AN MDRD
 QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
HEALTHCARE SOLUTIONS TRANSCAR AUTOMATED GUIDED VEHICLE
 HEALTHCARE SOLUTIONS TRANSCAR AUTOMATED GUIDED VEHICLE 2 TRANSCAR GENERATION 3 SWISSLOG IS A LEADING PROVIDER OF INNOVATIVE, INTEGRATED LOGISTICS SOLUTIONS FOR HOSPITALS Answering tomorrow s hospital logistics
HEALTHCARE SOLUTIONS TRANSCAR AUTOMATED GUIDED VEHICLE 2 TRANSCAR GENERATION 3 SWISSLOG IS A LEADING PROVIDER OF INNOVATIVE, INTEGRATED LOGISTICS SOLUTIONS FOR HOSPITALS Answering tomorrow s hospital logistics
Volume 13, Issue 5/ Medtech
 Volume 13, Issue 5/2011 - Medtech The New Central Sterile Supply Department of University Hospital Authority St. Orsola- Malpighi Polyclinic The new Central Sterile Supply Department (CSSD) of the University
Volume 13, Issue 5/2011 - Medtech The New Central Sterile Supply Department of University Hospital Authority St. Orsola- Malpighi Polyclinic The new Central Sterile Supply Department (CSSD) of the University
GETINGE 633HC STEAM STERILIZERS. Always with you
 GETINGE 633HC STEAM STERILIZERS Always with you GETINGE 633HC Steam Sterilizers 3 MAKING EVERY SQUARE FOOT COUNT With the ever increasing demands being placed on today s surgical workplaces, space limitations
GETINGE 633HC STEAM STERILIZERS Always with you GETINGE 633HC Steam Sterilizers 3 MAKING EVERY SQUARE FOOT COUNT With the ever increasing demands being placed on today s surgical workplaces, space limitations
Are you on track for success?
 Transitioning from a manual process to automated instrument management Are you on track for success? an ebook from Censis Technologies, Inc. Introduction It is well known that the HITECH Act (Health Information
Transitioning from a manual process to automated instrument management Are you on track for success? an ebook from Censis Technologies, Inc. Introduction It is well known that the HITECH Act (Health Information
01.48 Receiving and Processing Vendor Owned Surgical Instrumentation in the Sterile Processing Department
 01.48 Receiving and Processing Vendor Owned Surgical Instrumentation in the Purpose To provide effective management of and ensure standardization of processing for all reusable surgical instruments that
01.48 Receiving and Processing Vendor Owned Surgical Instrumentation in the Purpose To provide effective management of and ensure standardization of processing for all reusable surgical instruments that
8/13/2013. Keys to Facilities Planning. 3M Sterile U Network 3M Sterile U Web Meeting August 15th, 2013
 3M Sterile U Network 3M Sterile U Web Meeting August 15th, 2013 Keys to Facilities Planning Today s meeting times: 9:00 a.m., 11:00 a.m. and 1:00 p.m. CST To hear audio, call 800-937-0042 and enter access
3M Sterile U Network 3M Sterile U Web Meeting August 15th, 2013 Keys to Facilities Planning Today s meeting times: 9:00 a.m., 11:00 a.m. and 1:00 p.m. CST To hear audio, call 800-937-0042 and enter access
The Reconditioned Option.
 The Reconditioned Option. Thank you for the opportunity to further introduce the products and services of Monet Medical. With cuts in healthcare reimbursements and a host of other factors pressuring the
The Reconditioned Option. Thank you for the opportunity to further introduce the products and services of Monet Medical. With cuts in healthcare reimbursements and a host of other factors pressuring the
Material Handling. Chapter 5
 Material Handling Chapter 5 Designing material handling systems Overview of material handling equipment Unit load design Material handling equipment selection Material Handling Definitions Material handling
Material Handling Chapter 5 Designing material handling systems Overview of material handling equipment Unit load design Material handling equipment selection Material Handling Definitions Material handling
HEALTHCARE SOLUTIONS UNIPICK 2 AUTOMATED PACK DISPENSING SYSTEM
 HEALTHCARE SOLUTIONS AUTOMATED PACK DISPENSING SYSTEM 1 «SWISSLOG, AN ESTABLISHED WORLDWIDE PRESENCE» SWISSLOG HEALTHCARE SOLUTIONS LEADING CHANGE THROUGH HOSPITAL AUTOMATION. AUTOMATED PACK DISPENSING
HEALTHCARE SOLUTIONS AUTOMATED PACK DISPENSING SYSTEM 1 «SWISSLOG, AN ESTABLISHED WORLDWIDE PRESENCE» SWISSLOG HEALTHCARE SOLUTIONS LEADING CHANGE THROUGH HOSPITAL AUTOMATION. AUTOMATED PACK DISPENSING
IUSS HEALTH FACILITY GUIDES
 IUSS HEALTH FACILITY GUIDES Central Sterile Service Department Gazetted 30 June 2014 Task Team: B:08 Supported by: Document tracking Version Date Name Edited 13 th March 2014 Beverley van Reenen Technical
IUSS HEALTH FACILITY GUIDES Central Sterile Service Department Gazetted 30 June 2014 Task Team: B:08 Supported by: Document tracking Version Date Name Edited 13 th March 2014 Beverley van Reenen Technical
STERILIZATION ON MSF EMERGENGY SETTING. Pascale CHOQUENET Médecins Sans Frontières Paris France Milan 5th June 2008
 STERILIZATION ON MSF EMERGENGY SETTING Pascale CHOQUENET Médecins Sans Frontières Paris France Milan 5th June 2008 MSF = Doctors Without Borders International Humanitarian Organisation Medical NGO (Non
STERILIZATION ON MSF EMERGENGY SETTING Pascale CHOQUENET Médecins Sans Frontières Paris France Milan 5th June 2008 MSF = Doctors Without Borders International Humanitarian Organisation Medical NGO (Non
Unloading, Storage, Distribution, Transportation and Aseptic Presentation of Sterile Items
 Unloading, Storage, Distribution, Transportation and Aseptic Presentation of Sterile Items Comprehensive guide to steam sterilization and sterility assurance in health care facilities (ANSI/AAMI ST79:2006)
Unloading, Storage, Distribution, Transportation and Aseptic Presentation of Sterile Items Comprehensive guide to steam sterilization and sterility assurance in health care facilities (ANSI/AAMI ST79:2006)
BIOMEDICAL WASTE MANAGEMENT POLICY
 BIOMEDICAL WASTE MANAGEMENT POLICY Managing biomedical waste in a safe and environmentally responsible manner is a key focus of the Environmental Compliance and Sustainability office. 1.0 PURPOSE In an
BIOMEDICAL WASTE MANAGEMENT POLICY Managing biomedical waste in a safe and environmentally responsible manner is a key focus of the Environmental Compliance and Sustainability office. 1.0 PURPOSE In an
Operating Room Manual
 CHAPTER 6 SUPPLY CHAIN Lead Author: Tangwan Azefor, MD; Clinical Associate, Department of Anesthesiology & Critical Care Medicine; Johns Hopkins Bayview Medical Center Baltimore, MD Checklist 1. How many
CHAPTER 6 SUPPLY CHAIN Lead Author: Tangwan Azefor, MD; Clinical Associate, Department of Anesthesiology & Critical Care Medicine; Johns Hopkins Bayview Medical Center Baltimore, MD Checklist 1. How many
LEAN WAREHOUSE OPERATIONS.
 LEAN WAREHOUSE OPERATIONS www.fourprinciples.com BACKGROUND Warehousing s historical core responsibility has been the storage of goods. However, the scope and core responsibilities of warehouse management
LEAN WAREHOUSE OPERATIONS www.fourprinciples.com BACKGROUND Warehousing s historical core responsibility has been the storage of goods. However, the scope and core responsibilities of warehouse management
CERTIFIED WAREHOUSING AND STOREKEEPING COURSE
 CERTIFIED WAREHOUSING AND STOREKEEPING COURSE Unic Foundation Entrepreneurship Training Manual Page 1 1.0.WAREHOUSING Warehousing refers to the activities involving storage of goods on a large-scale in
CERTIFIED WAREHOUSING AND STOREKEEPING COURSE Unic Foundation Entrepreneurship Training Manual Page 1 1.0.WAREHOUSING Warehousing refers to the activities involving storage of goods on a large-scale in
System Catalogue. Medical Waste and Linen Transfer System Living a Safe Future Living the Dream. System Catalogue
 Medical Waste and Linen Transfer System Living a Safe Future Living the Dream System Catalogue Page 1 of 20 Safe Transfer Summary 1. Concept 2. System components 3. Algorithm 4. Mechanical System Features
Medical Waste and Linen Transfer System Living a Safe Future Living the Dream System Catalogue Page 1 of 20 Safe Transfer Summary 1. Concept 2. System components 3. Algorithm 4. Mechanical System Features
Biomedical Waste Handling and Disposal
 Approved by: Biomedical Waste Handling and Disposal Corporate Director, Environmental Supports Environmental Services Operating Standards Manual Number: Date Approved Next Review May 3, 2018 Purpose Applicability
Approved by: Biomedical Waste Handling and Disposal Corporate Director, Environmental Supports Environmental Services Operating Standards Manual Number: Date Approved Next Review May 3, 2018 Purpose Applicability
MODULE 14: Off-site Transport and Storage of Healthcare Waste
 MODULE 14: Off-site Transport and Storage of Healthcare Waste Module Overview Define external transport of healthcare waste Describe requirements for off-site transport of healthcare wastes including training
MODULE 14: Off-site Transport and Storage of Healthcare Waste Module Overview Define external transport of healthcare waste Describe requirements for off-site transport of healthcare wastes including training
HEALTHCARE SOLUTIONS AUTOMATED SOLUTIONS FOR CONSOLIDATED SERVICE CENTERS
 HEALTHCARE SOLUTIONS AUTOMATED SOLUTIONS FOR CONSOLIDATED SERVICE CENTERS CHANGE THE WAY YOU WORK THROUGH INTEGRATED AUTOMATION SOLUTIONS The AVEN warehouse is the largest, advanced healthcare logistics
HEALTHCARE SOLUTIONS AUTOMATED SOLUTIONS FOR CONSOLIDATED SERVICE CENTERS CHANGE THE WAY YOU WORK THROUGH INTEGRATED AUTOMATION SOLUTIONS The AVEN warehouse is the largest, advanced healthcare logistics
Getinge da Vinci Solution
 Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
Best Practices for Reprocessing Endoscopes
 Best Practices for Reprocessing Endoscopes Reprocessing of Endoscopes Reprocessing Room Standards The resources referenced include: Canadian Standards Association Provincial Infectious Disease Advisory
Best Practices for Reprocessing Endoscopes Reprocessing of Endoscopes Reprocessing Room Standards The resources referenced include: Canadian Standards Association Provincial Infectious Disease Advisory
Testing of routinely sterilized products should
 Product Quality Assurance Testing: One Hospital s Experience Testing of routinely sterilized products should be part of a healthcare facility s quality control program, and ANSI/AAMI ST79:2006, Comprehensive
Product Quality Assurance Testing: One Hospital s Experience Testing of routinely sterilized products should be part of a healthcare facility s quality control program, and ANSI/AAMI ST79:2006, Comprehensive
Total Engineering Solution
 Total Engineering Solution Hyundai Elevator is a total moving solution provider that offers comprehensive engineering solutions for optimal supply. These range from providing consultations, designs as
Total Engineering Solution Hyundai Elevator is a total moving solution provider that offers comprehensive engineering solutions for optimal supply. These range from providing consultations, designs as
Medical, Pathological and Low-level Radioactive Waste Management Plan for Incinerator Revised 3-08
 Background Medical, Pathological and Low-level Radioactive Waste Management Plan for Incinerator Revised 3-08 Many WSU laboratories and departments generate medical waste (contaminated solids), pathological
Background Medical, Pathological and Low-level Radioactive Waste Management Plan for Incinerator Revised 3-08 Many WSU laboratories and departments generate medical waste (contaminated solids), pathological
DOCUMENT DESPATCH ADVICE DRAFT IN WIDE CIRCULATION MHR 06 /T- 50/51/52/53/ TECHNICAL COMMITTEE: MHR 06
 DRAFT IN WIDE CIRCULATION TECHNICAL COMMITTEE: MHR 06 DOCUMENT DESPATCH ADVICE Ref Date MHR 06 /T- 50/51/52/53/54 09-05-2008 DOC NO: MHR 06 (0114)/ISO 7199:1996 TITLE Cardiovascular implants and artificial
DRAFT IN WIDE CIRCULATION TECHNICAL COMMITTEE: MHR 06 DOCUMENT DESPATCH ADVICE Ref Date MHR 06 /T- 50/51/52/53/54 09-05-2008 DOC NO: MHR 06 (0114)/ISO 7199:1996 TITLE Cardiovascular implants and artificial
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants
 Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
OFFICE RELOCATION ORIENTATION
 OFFICE RELOCATION ORIENTATION Thank you for selecting Ray Hamilton Company and for your support in continuing our tradition. Establishing an effective internal organization to manage and coordinate your
OFFICE RELOCATION ORIENTATION Thank you for selecting Ray Hamilton Company and for your support in continuing our tradition. Establishing an effective internal organization to manage and coordinate your
RFID Revolutionizing Healthcare with Smart Surgical Tools
 RFID Revolutionizing Healthcare with Smart Surgical Tools RFID Moves Beyond Asset Tracking to Optimize Clinical Operations Hospitals spend a significant amount of time and money searching for equipment,
RFID Revolutionizing Healthcare with Smart Surgical Tools RFID Moves Beyond Asset Tracking to Optimize Clinical Operations Hospitals spend a significant amount of time and money searching for equipment,
STANDARD OPERATING PROCEDURES FOR ANIMAL QUARANTINE & CERTIFICATION SERVICES
 STANDARD OPERATING PROCEDURES FOR ANIMAL QUARANTINE & CERTIFICATION SERVICES GOVERNMENT OF INDIA MINISTRY OF AGRICULTURE DEPARTMENT OF ANIMAL HUSBANDRY, DAIRYING & FISHERIES Entry protocol for vehicle
STANDARD OPERATING PROCEDURES FOR ANIMAL QUARANTINE & CERTIFICATION SERVICES GOVERNMENT OF INDIA MINISTRY OF AGRICULTURE DEPARTMENT OF ANIMAL HUSBANDRY, DAIRYING & FISHERIES Entry protocol for vehicle
FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE
 FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE What is an Automated Endoscope Reprocessor? An Automated Endoscopes Reprocessor (AER) performs all the cycle steps (fresh water preflush, rinsing, disinfection,
FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE What is an Automated Endoscope Reprocessor? An Automated Endoscopes Reprocessor (AER) performs all the cycle steps (fresh water preflush, rinsing, disinfection,
Logistics I. Collection, transport and disposal of non-hazardous waste and recyclables
 Logistics I Collection, transport and disposal of non-hazardous waste and recyclables ETLog GmbH Brunnenstraße 164 10119 Berlin Germany www.etlog.com info@etlog.com Contains of the lecture The collection
Logistics I Collection, transport and disposal of non-hazardous waste and recyclables ETLog GmbH Brunnenstraße 164 10119 Berlin Germany www.etlog.com info@etlog.com Contains of the lecture The collection
BITO LEO LOCATIVE READY, STEADY, GO! This driverless transport system is immediately.
 solutions BITO LEO LOCATIVE This driverless transport system is immediately READY, STEADY, GO! www.leo-locative.com » BITO LEO LOCATIVE There is no easier way to move your bins from one workstation to
solutions BITO LEO LOCATIVE This driverless transport system is immediately READY, STEADY, GO! www.leo-locative.com » BITO LEO LOCATIVE There is no easier way to move your bins from one workstation to
UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES
 UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES October 2016 Table of Contents Section Page I. Introduction.... 1 II. Characteristics of Regulated Medical Waste 1-2 III. Exclusions...2-3
UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES October 2016 Table of Contents Section Page I. Introduction.... 1 II. Characteristics of Regulated Medical Waste 1-2 III. Exclusions...2-3
What Dentistry Can Learn from AAMI ST79. Best Practices for device and instrument reprocessing
 What Dentistry Can Learn from AAMI ST79 Best Practices for device and instrument reprocessing Learning goals: Purpose and goals of AAMI organization. Overview of ST79. Review selected topics: IFUs, tools
What Dentistry Can Learn from AAMI ST79 Best Practices for device and instrument reprocessing Learning goals: Purpose and goals of AAMI organization. Overview of ST79. Review selected topics: IFUs, tools
2018 Resources for Health Care Delivery Organizations
 AMA HEALTH SOLUTIONS 2018 Resources for Health Care Delivery Organizations Health Care Delivery Organizations Toolkit for Counting Users & FAQs September 2017 User Proxy Model Calculator for data file
AMA HEALTH SOLUTIONS 2018 Resources for Health Care Delivery Organizations Health Care Delivery Organizations Toolkit for Counting Users & FAQs September 2017 User Proxy Model Calculator for data file
We engineer your success. All over the world. Mushrooms
 We engineer your success. All over the world. Mushrooms Introducing Mushrooms Mushrooms are delicate products. Therefore, mushrooms require extra care to prevent damaging and to preserve high quality
We engineer your success. All over the world. Mushrooms Introducing Mushrooms Mushrooms are delicate products. Therefore, mushrooms require extra care to prevent damaging and to preserve high quality
P11 Handling and Storage E212 - Facilities Planning and Design
 P11 Handling and Storage E212 - Facilities Planning and Design Material Handling Is the art and science of moving, storing, protecting and controlling material. Means providing the: Right amount of Material
P11 Handling and Storage E212 - Facilities Planning and Design Material Handling Is the art and science of moving, storing, protecting and controlling material. Means providing the: Right amount of Material
TRACECARE Monitoring, Reporting & Traceability Solutions
 TRACECARE Monitoring, Reporting & Traceability Solutions THE CISA GROUP COMPANY COMPANY PROFILE CISA has been manufacturing and selling sterilization systems for over 60 years for both hospitals and industrial
TRACECARE Monitoring, Reporting & Traceability Solutions THE CISA GROUP COMPANY COMPANY PROFILE CISA has been manufacturing and selling sterilization systems for over 60 years for both hospitals and industrial
Use of Queuing Models in Health Care - PPT
 University of Wisconsin-Madison From the SelectedWorks of Vikas Singh December, 2006 Use of Queuing Models in Health Care - PPT Vikas Singh, University of Arkansas for Medical Sciences Available at: https://works.bepress.com/vikas_singh/13/
University of Wisconsin-Madison From the SelectedWorks of Vikas Singh December, 2006 Use of Queuing Models in Health Care - PPT Vikas Singh, University of Arkansas for Medical Sciences Available at: https://works.bepress.com/vikas_singh/13/
ELECTRONETICS. Scope: Prep Area
 ELECTRONETICS 5S TRAINING Scope: Prep Area February 19, 2010 Sponsor Michael Quinn, VP Manufacturing Production & Assembly, Northwest Center Scott Patterson, Plant Manager, Electronetics What is 5S? It
ELECTRONETICS 5S TRAINING Scope: Prep Area February 19, 2010 Sponsor Michael Quinn, VP Manufacturing Production & Assembly, Northwest Center Scott Patterson, Plant Manager, Electronetics What is 5S? It
Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP)
 Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP) NOTE: This SOP for biological waste management does not supersede the requirements for radioactive and/or hazardous
Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP) NOTE: This SOP for biological waste management does not supersede the requirements for radioactive and/or hazardous
NORTH CAROLINA VETERINARY MEDICAL BOARD PRACTICE INSPECTION 1611 Jones Franklin Road, Suite 106, Raleigh NC (919) FAX (919)
 NORTH CAROLINA VETERINARY MEDICAL BOARD PRACTICE INSPECTION 1611 Jones Franklin Road, Suite 106, Raleigh NC 27606 (919) 854-5601 FAX (919) 854-5606 FOR OFFICE STAFF ONLY Photo(s): $125.00 Inspection Fee
NORTH CAROLINA VETERINARY MEDICAL BOARD PRACTICE INSPECTION 1611 Jones Franklin Road, Suite 106, Raleigh NC 27606 (919) 854-5601 FAX (919) 854-5606 FOR OFFICE STAFF ONLY Photo(s): $125.00 Inspection Fee
Optimising Inbound, Warehousing and Outbound Strategies
 Warehouse Management EXECUTIVE SUMMARY Optimising Inbound, Warehousing and Outbound Strategies Control the movement of stock within a warehouse or across multiple facilities. Process associated transactions
Warehouse Management EXECUTIVE SUMMARY Optimising Inbound, Warehousing and Outbound Strategies Control the movement of stock within a warehouse or across multiple facilities. Process associated transactions
Partnership. Medical Waste Autoclaves. Medical Waste Autoclaves
 Partnership Medical Waste Autoclaves Medical Waste Autoclaves Safe, Cost-Effective, Medical Waste Sterilization Keep your environment safe from Medical Waste with high performance Tuttnauer Medical Waste
Partnership Medical Waste Autoclaves Medical Waste Autoclaves Safe, Cost-Effective, Medical Waste Sterilization Keep your environment safe from Medical Waste with high performance Tuttnauer Medical Waste
INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL
 INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL UNIVERSITY RISK MANAGEMENT Occupational Safety and Health Programs 19 Hagood Avenue, Suite 908 Charleston, SC 29425 843-792-3604 Revised: November 2016 TABLE
INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL UNIVERSITY RISK MANAGEMENT Occupational Safety and Health Programs 19 Hagood Avenue, Suite 908 Charleston, SC 29425 843-792-3604 Revised: November 2016 TABLE
Building a Omni- Channel Distribution Center A Case Study
 Building a Omni- Channel Distribution Center A Case Study Sponsored by: Presented by: Chris Castaldi Allen Sisk Marc Teychene 2015 MHI Copyright claimed for audiovisual works and sound recordings of seminar
Building a Omni- Channel Distribution Center A Case Study Sponsored by: Presented by: Chris Castaldi Allen Sisk Marc Teychene 2015 MHI Copyright claimed for audiovisual works and sound recordings of seminar
Implementing VMI to Reduce Direct and Indirect Cost
 Implementing VMI to Reduce Direct and Indirect Cost James M. Truog, Manager Strategic Purchasing Wacker Corp. 262-250-3518; james.truog@am.wackergroup.com 89 th Annual International Supply Management Conference,
Implementing VMI to Reduce Direct and Indirect Cost James M. Truog, Manager Strategic Purchasing Wacker Corp. 262-250-3518; james.truog@am.wackergroup.com 89 th Annual International Supply Management Conference,
Competency Guide: Care and Handling of Rigid Endoscopes
 Competency Guide: Care and Handling of Rigid Endoscopes This document details the basic steps to correctly process and maintain a rigid endoscope. This document can be used to perform a competency review
Competency Guide: Care and Handling of Rigid Endoscopes This document details the basic steps to correctly process and maintain a rigid endoscope. This document can be used to perform a competency review
Getinge Steam Sterilizers From cycle to cycle through lifecycle. Reducing risk in Central Sterile.
 Getinge Steam Sterilizers From cycle to cycle through lifecycle. Reducing risk in Central Sterile. Because uptime, over time, is the only time that counts. Central Sterile (CS) technicians and managers,
Getinge Steam Sterilizers From cycle to cycle through lifecycle. Reducing risk in Central Sterile. Because uptime, over time, is the only time that counts. Central Sterile (CS) technicians and managers,
NSK Steering Systems America
 PAGE NO: 1 of 13 NSK Steering Systems America Supplier Logistics and Packaging Manual PAGE NO: 2 of 13 Table of Contents 1 Scope... 3 1.1 General... 3 2 Communication... 3 2.1 Supplier contacts... 3 2.2
PAGE NO: 1 of 13 NSK Steering Systems America Supplier Logistics and Packaging Manual PAGE NO: 2 of 13 Table of Contents 1 Scope... 3 1.1 General... 3 2 Communication... 3 2.1 Supplier contacts... 3 2.2
Plant Layout and Material Handling
 Plant Layout and Material Handling IV year-ii semester COURSE CODE A80365 Prepared by G S Vivek, Assistant Professor A.Venu Prasad, Assistant Professor MECHANICAL ENGINEERING INSTITUTE OF AERONAUTICAL
Plant Layout and Material Handling IV year-ii semester COURSE CODE A80365 Prepared by G S Vivek, Assistant Professor A.Venu Prasad, Assistant Professor MECHANICAL ENGINEERING INSTITUTE OF AERONAUTICAL
Chapter 9 Storage & Warehouse Systems
 FACILITIES PLANNING & DESIGN Alberto Garcia-Diaz J. MacGregor Smith Chapter 9 Storage & Warehouse Systems 1. Warehouse Layout Model 2. Storage Equipment 3. Automated Storage & Retrieval System 1 Dedicated
FACILITIES PLANNING & DESIGN Alberto Garcia-Diaz J. MacGregor Smith Chapter 9 Storage & Warehouse Systems 1. Warehouse Layout Model 2. Storage Equipment 3. Automated Storage & Retrieval System 1 Dedicated
Term used to describe concept for storing bags that are not immediately due for Aircraft Loading. Also referred to as Cold Bag Storage (CBS)
 January 12 th 2017 Term used to describe concept for storing bags that are not immediately due for Aircraft Loading. Also referred to as Cold Bag Storage (CBS) Bags are accumulated at Check-In and manually
January 12 th 2017 Term used to describe concept for storing bags that are not immediately due for Aircraft Loading. Also referred to as Cold Bag Storage (CBS) Bags are accumulated at Check-In and manually
32.0 Public & Staff Amenities Unit
 32.0 Public & Staff Amenities Unit 32.1 Introduction 32.1.1 Description The Amenities Unit shall provide facilities for the convenience and comfort of staff and visitors to the hospital. Some of the amenities
32.0 Public & Staff Amenities Unit 32.1 Introduction 32.1.1 Description The Amenities Unit shall provide facilities for the convenience and comfort of staff and visitors to the hospital. Some of the amenities
Sterilization Service Revolution Service Enhancement in HA Hospitals
 Sterilization Service Revolution Service Enhancement in HA Hospitals Hong Kong Hospital Authority Convention 2014 7 May 2014 LAW Tat Hong Samuel Cluster Operations Manager Central Sterile Supplies Department,
Sterilization Service Revolution Service Enhancement in HA Hospitals Hong Kong Hospital Authority Convention 2014 7 May 2014 LAW Tat Hong Samuel Cluster Operations Manager Central Sterile Supplies Department,
Optimizing and Tracking - RFID in Medical & Health. Richard Aufreiter, PM, HID Global
 Optimizing and Tracking - RFID in Medical & Health Richard Aufreiter, PM, HID Global An ASSA ABLOY Company The global leader in door opening solutions Operations in more than 50 countries $ 5.8 billion
Optimizing and Tracking - RFID in Medical & Health Richard Aufreiter, PM, HID Global An ASSA ABLOY Company The global leader in door opening solutions Operations in more than 50 countries $ 5.8 billion
Insulin Syringes ACCU-SHOT TM ACCU-SHOT U40 & U100. (with Ultra Thin & Short Needle) Sizes. Colour Code of Top Cap ml U G 6 mm Red
 Be aware be safe... SPM Medicare Pvt. Ltd. (An EN ISO 13485:2012 Certified Company) Corp. & Mfd. Address: B-40, Phase-II, NOIDA, U.P., INDIA Customer Care: +91-120-4202413 Insulin Syringes ACCU-SHOT U40
Be aware be safe... SPM Medicare Pvt. Ltd. (An EN ISO 13485:2012 Certified Company) Corp. & Mfd. Address: B-40, Phase-II, NOIDA, U.P., INDIA Customer Care: +91-120-4202413 Insulin Syringes ACCU-SHOT U40
SUPPLEMENTARY ACCESSIBILITY REQUIREMENTS
 SUPPLEMENTARY ACCESSIBILITY REQUIREMENTS (Not adopted by the State of Oregon) The provisions contained in this appendix are not mandatory unless specifically referenced in the adopting ordinance. SECTION
SUPPLEMENTARY ACCESSIBILITY REQUIREMENTS (Not adopted by the State of Oregon) The provisions contained in this appendix are not mandatory unless specifically referenced in the adopting ordinance. SECTION
Heini Simsek UTILIZING RFID-TECHNOLOGY IN IN-HOUSE LOGISTICS
 Heini Simsek UTILIZING RFID-TECHNOLOGY IN IN-HOUSE LOGISTICS UTILIZING RFID-TECHNOLOGY IN IN-HOUSE LOGISTICS Heini Simsek Bachelor s Thesis Autumn 2016 Degree Programme in Mechanical and Production Engineering
Heini Simsek UTILIZING RFID-TECHNOLOGY IN IN-HOUSE LOGISTICS UTILIZING RFID-TECHNOLOGY IN IN-HOUSE LOGISTICS Heini Simsek Bachelor s Thesis Autumn 2016 Degree Programme in Mechanical and Production Engineering
STERILE PROCESSING CHALLENGES
 STERILE PROCESSING CHALLENGES And How to Overcome Them Lucas Higman, BA, MBA Introduction Lucas Higman, BA, MBA Vice President at Soyring Consulting Worked with more than 65 healthcare facilities and Surgical
STERILE PROCESSING CHALLENGES And How to Overcome Them Lucas Higman, BA, MBA Introduction Lucas Higman, BA, MBA Vice President at Soyring Consulting Worked with more than 65 healthcare facilities and Surgical
Infection control doctor. Mrs Muneera Al. Training supervisor. Abdulsalam. Dr Naser Yahya. Dr Haifa Al Mousa
 1 State of KUWAIT Ministry of health Infection control directorate Section location Central sterile department ( C.S.S.D ) Effective Date May 2015 Applied to All workers in C.S.S.D Prepared by : Dr. Mohammed
1 State of KUWAIT Ministry of health Infection control directorate Section location Central sterile department ( C.S.S.D ) Effective Date May 2015 Applied to All workers in C.S.S.D Prepared by : Dr. Mohammed
SALES EVENT MANAGEMENT FOOD STOREROOM
 CURRENT JOB OPENINGS To apply on-line go to: www.kcmarriott.com SALES Business Transient Sales Manager The Kansas City Marriott Downtown has an opening for a Business Transient Sales Manager on our Sales
CURRENT JOB OPENINGS To apply on-line go to: www.kcmarriott.com SALES Business Transient Sales Manager The Kansas City Marriott Downtown has an opening for a Business Transient Sales Manager on our Sales
Introduction This article investigates the challenges
 Public Safety Comparison of Dedicated and Swing Public/Service Elevators in Hospitals by Samson Rajan Babu photos courtesy of Hydraulik Liftsysteme Walter Mayer GmbH, InterMetro, Cambro, Schindler, Lobby
Public Safety Comparison of Dedicated and Swing Public/Service Elevators in Hospitals by Samson Rajan Babu photos courtesy of Hydraulik Liftsysteme Walter Mayer GmbH, InterMetro, Cambro, Schindler, Lobby
BIOMEDICAL EQUIPMENT CUSTOMER DEPARTMENT REFERENCE MANUAL
 BIOMEDICAL EQUIPMENT CUSTOMER DEPARTMENT REFERENCE MANUAL Biomedical Engineering Department 747 52 nd Street, Oakland CA 94609 510-428-3610 (After Hours pager # 7566) THE BIOMEDICAL EQUIPMENT REFERENCE
BIOMEDICAL EQUIPMENT CUSTOMER DEPARTMENT REFERENCE MANUAL Biomedical Engineering Department 747 52 nd Street, Oakland CA 94609 510-428-3610 (After Hours pager # 7566) THE BIOMEDICAL EQUIPMENT REFERENCE
General Operating Fund Budget Reduction Scenario Narrative Business & Finance
 Division UNIT or Department 0 0 FTE MPP -1.0 TOTAL FTE -1.0 Cut Priority Number 1 Total Reduction Amount $93,584 Operation or Strategic Impact of reduction: Eliminate Landscape, Transportation and Custodial
Division UNIT or Department 0 0 FTE MPP -1.0 TOTAL FTE -1.0 Cut Priority Number 1 Total Reduction Amount $93,584 Operation or Strategic Impact of reduction: Eliminate Landscape, Transportation and Custodial
Subject: Apparel Production Management. Unit 3: Manufacturing Systems. Quadrant 1 e-text. Describe the various types of production systems.
 Subject: Apparel Production Management Unit 3: Manufacturing Systems Quadrant 1 e-text Learning Objectives The learning objectives of this unit are: Describe the various types of production systems. Identify
Subject: Apparel Production Management Unit 3: Manufacturing Systems Quadrant 1 e-text Learning Objectives The learning objectives of this unit are: Describe the various types of production systems. Identify
BGSU s Biosafety and Infectious Waste Safety Procedures. Environmental Health & Safety 1851 N. Research Drive Bowling Green, OH
 BGSU s Biosafety and Infectious Waste Safety Procedures Environmental Health & Safety 1851 N. Research Drive Bowling Green, OH 43403 419-372-2171 Revised: January 2018 Table of Contents Foreword... 1 Biosafety
BGSU s Biosafety and Infectious Waste Safety Procedures Environmental Health & Safety 1851 N. Research Drive Bowling Green, OH 43403 419-372-2171 Revised: January 2018 Table of Contents Foreword... 1 Biosafety
Steam Sterilizer. SA Series. for hospitals, CSSD and medical centers
 Steam Sterilizer SA Series for hospitals, CSSD and medical centers Driven by customer needs Colussi is a leading infection control solution provider, supplying the healthcare, laboratory research and pharma
Steam Sterilizer SA Series for hospitals, CSSD and medical centers Driven by customer needs Colussi is a leading infection control solution provider, supplying the healthcare, laboratory research and pharma
3M Intelligent Return Plus and Sorter System SL. Architect/Contractor's Package
 3M Intelligent Return Plus and Sorter System SL Architect/Contractor's Package 3M, 2015. All rights reserved. 3M Intelligent Return Plus and Sorter System SL Architect/Contractor's Package, 78-8129-4937-4B
3M Intelligent Return Plus and Sorter System SL Architect/Contractor's Package 3M, 2015. All rights reserved. 3M Intelligent Return Plus and Sorter System SL Architect/Contractor's Package, 78-8129-4937-4B
Case study: Granada La Palma Granada La Palma integrates two new large capacity warehouses in their production centre
 Case study: Granada La Palma Granada La Palma integrates two new large capacity warehouses in their production centre Location: Spain The fruit and veg cooperative Granada La Palma, the largest producer
Case study: Granada La Palma Granada La Palma integrates two new large capacity warehouses in their production centre Location: Spain The fruit and veg cooperative Granada La Palma, the largest producer
Method for a manufacturing WIP cart for integrated factory automation systems
 Method for a manufacturing WIP cart for integrated factory automation systems Disclosed is a method for a manufacturing work-in-progress (WIP) cart for integrated factory automation systems. Benefits include
Method for a manufacturing WIP cart for integrated factory automation systems Disclosed is a method for a manufacturing work-in-progress (WIP) cart for integrated factory automation systems. Benefits include
Medi-Therm Precise. Easy. Versatile.
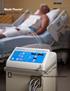 Medi-Therm Precise. Easy. Versatile. Medi-Therm Hyper/Hypothermia System Patient warming and cooling are often critical factors in achieving positive outcomes. The Stryker Medi-Therm system can be used
Medi-Therm Precise. Easy. Versatile. Medi-Therm Hyper/Hypothermia System Patient warming and cooling are often critical factors in achieving positive outcomes. The Stryker Medi-Therm system can be used
Vertical Transportation Planning Retail/Mixed-Use
 Fortune Consultants LTD Elevator Consulting Vertical Transportation Planning Retail/Mixed-Use This paper is to provide design guidance for the initial planning of vertical transportation in either a standalone
Fortune Consultants LTD Elevator Consulting Vertical Transportation Planning Retail/Mixed-Use This paper is to provide design guidance for the initial planning of vertical transportation in either a standalone
PROGRAM FOR THE MANAGEMENT AND DISPOSAL OF RADIOACTIVE WASTE
 RADIATION SAFETY DEPARTMENT West Virginia University Health Sciences Center WVU Hospitals Jefferson Memorial Hospital G-139 Health Sciences Center PO Box 9006 Morgantown, WV 26506-9006 Phone: 304-293-3413
RADIATION SAFETY DEPARTMENT West Virginia University Health Sciences Center WVU Hospitals Jefferson Memorial Hospital G-139 Health Sciences Center PO Box 9006 Morgantown, WV 26506-9006 Phone: 304-293-3413
ASEAN Guidelines on GMP for Traditional Medicines / Health Supplement
 ASEAN Guidelines on GMP for Traditional Medicines / Health Supplement Prepared by: PHILIPPINES Approved by: ASEAN TMHS GMP Task Force Endorsed by: ASEAN TMHS Product Working Group OUTLINE 1. Principle
ASEAN Guidelines on GMP for Traditional Medicines / Health Supplement Prepared by: PHILIPPINES Approved by: ASEAN TMHS GMP Task Force Endorsed by: ASEAN TMHS Product Working Group OUTLINE 1. Principle
AS/NZS 4187 RELEVANT STANDARDS. Robyn Williams NUM CSSD LBHHS & Standards Australia Chair HE-023
 AS/NZS 4187 RELEVANT STANDARDS Robyn Williams NUM CSSD LBHHS & Standards Australia Chair HE-023 Relevant Standards AS 2514 AS 2774 AS 2773 AS 2773.1 Drying cabinets for medical equipment Drying cabinets
AS/NZS 4187 RELEVANT STANDARDS Robyn Williams NUM CSSD LBHHS & Standards Australia Chair HE-023 Relevant Standards AS 2514 AS 2774 AS 2773 AS 2773.1 Drying cabinets for medical equipment Drying cabinets
Complete system to track the flow of flat and packed linen with RFID UHF tags for Industrial Laundries
 Complete system to track the flow of flat and packed linen with RFID UHF tags for Industrial Laundries Folded Garment Distribution System Automatic Management for Uniforms Conveyor Belts Management System
Complete system to track the flow of flat and packed linen with RFID UHF tags for Industrial Laundries Folded Garment Distribution System Automatic Management for Uniforms Conveyor Belts Management System
You are required to complete this course with a passing score of 85% or higher if you:
 OHS Biosafety BIO301L Medical Waste Management for Labs Training You are required to complete this course with a passing score of 85% or higher if you: Offer medical waste for transport from a UAB campus
OHS Biosafety BIO301L Medical Waste Management for Labs Training You are required to complete this course with a passing score of 85% or higher if you: Offer medical waste for transport from a UAB campus
MUST 1 & 2 Mobile Unit Surgical Trailer
 MUST 1 & 2 Mobile Unit Surgical Trailer Arnaud Badinier ( PC MUST 1 ) Clement Chauvel ( Log Cell 3- log tech MUST 1 ) Hathi Anne Khoudiacoff ( tactical Referent MUST 1 & 2) Federico Ubaldi ( PM - Log tech
MUST 1 & 2 Mobile Unit Surgical Trailer Arnaud Badinier ( PC MUST 1 ) Clement Chauvel ( Log Cell 3- log tech MUST 1 ) Hathi Anne Khoudiacoff ( tactical Referent MUST 1 & 2) Federico Ubaldi ( PM - Log tech
SAFETY MANUAL OF GOOD LABORATORY
 SAFETY MANUAL OF GOOD LABORATORY PRACTICES 1 THE LABORATORY The biotechnology laboratory platform is an area where many risks are present. In it we use different products and agents that may be harmful
SAFETY MANUAL OF GOOD LABORATORY PRACTICES 1 THE LABORATORY The biotechnology laboratory platform is an area where many risks are present. In it we use different products and agents that may be harmful
AUTOMOTIVE. How is it possible to extend production facilities and save money? Simple ask the experts.
 AUTOMOTIVE How is it possible to extend production facilities and save money? Simple ask the experts. AT A GLANCE. OPTIMUM FLOW OF GOODS IN EVERY DIRECTION Today s automotive industry expects maximum speed,
AUTOMOTIVE How is it possible to extend production facilities and save money? Simple ask the experts. AT A GLANCE. OPTIMUM FLOW OF GOODS IN EVERY DIRECTION Today s automotive industry expects maximum speed,
ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing.
 11157 ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing. ETD DOUBLE: INTRODUCING THE FIRST OLYMPUS PASS- THROUGH WASHER-DISINFECTOR With the ETD Double, Olympus Has Advanced Its
11157 ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing. ETD DOUBLE: INTRODUCING THE FIRST OLYMPUS PASS- THROUGH WASHER-DISINFECTOR With the ETD Double, Olympus Has Advanced Its
ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES
 United States Office of the EPA 233-B-00-001 Environmental Protection Administrator May 2000 Agency (2131) ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES 3.5 Biologically Active Substances and Wastes
United States Office of the EPA 233-B-00-001 Environmental Protection Administrator May 2000 Agency (2131) ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES 3.5 Biologically Active Substances and Wastes
BSL3 and BSL4 Autoclaves. Laboratory Line
 BSL3 and BSL4 Autoclaves Laboratory Line BSL3 and BSL4 Autoclaves In today s society the need for safe bio-containment systems is increasingly important. This is especially true in the sterilization process
BSL3 and BSL4 Autoclaves Laboratory Line BSL3 and BSL4 Autoclaves In today s society the need for safe bio-containment systems is increasingly important. This is especially true in the sterilization process
UNIVERSITY OF TOLEDO HEALTH SCIENCE CAMPUS
 UNIVERSITY OF TOLEDO HEALTH SCIENCE CAMPUS SUBJECT: SPECIMEN TRANSPORT IN Procedure No: S-08-011 COMPUTERIZED TUBE SYSTEM PROCEDURE STATEMENT All laboratory specimens will be transported in a manner consistent
UNIVERSITY OF TOLEDO HEALTH SCIENCE CAMPUS SUBJECT: SPECIMEN TRANSPORT IN Procedure No: S-08-011 COMPUTERIZED TUBE SYSTEM PROCEDURE STATEMENT All laboratory specimens will be transported in a manner consistent
EH&S. Sheet. Fact. Safe and Effective Use of Autoclaves. What are autoclaves? Factors for effective sterilization. Dry heat cycle - when to use
 Please post or circulate Fact heet nvironment, ealth and afety Information for the Berkeley Campus No. 33 Revised 04/04/11 afe and ffective Use of Autoclaves Autoclaves are easy to use but can pose a safety
Please post or circulate Fact heet nvironment, ealth and afety Information for the Berkeley Campus No. 33 Revised 04/04/11 afe and ffective Use of Autoclaves Autoclaves are easy to use but can pose a safety
Products made in Switzerland
 Products made in Switzerland HYGIENIC AND MEDICALLY CLEAR DIFFERENT PRODUCTS BUT THE SAME QUALITY LABEL HYGIENIC AND MEDICALLY CLEAR 2 MDD TECHNOLOGIES FOR: 1. Cleaning 2. Disinfection 3. High Level Disinfection
Products made in Switzerland HYGIENIC AND MEDICALLY CLEAR DIFFERENT PRODUCTS BUT THE SAME QUALITY LABEL HYGIENIC AND MEDICALLY CLEAR 2 MDD TECHNOLOGIES FOR: 1. Cleaning 2. Disinfection 3. High Level Disinfection
One such problem is blood residual inside the channels of a robotic arm.
 SUBJECT: Robotic Arm Testing for residue blood DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue inside and outside of a robotic arm and
SUBJECT: Robotic Arm Testing for residue blood DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue inside and outside of a robotic arm and
Process Mapping Tools. Business Process Kaizen
 Process Mapping Tools Business Process Kaizen Module Objectives At the end of this module you will be able to Construct a spaghetti map of a process Construct a business process flow map Describe the difference
Process Mapping Tools Business Process Kaizen Module Objectives At the end of this module you will be able to Construct a spaghetti map of a process Construct a business process flow map Describe the difference
THE SAFE DISPOSAL OF CLINICAL/DOMESTIC WASTE
 Section V THE SAFE DISPOSAL OF CLINICAL/DOMESTIC WASTE The Trust is currently reviewing the requirements of the recent guidelines Health Technical Memorandum Safe Management of Healthcare Waste (HTML 07-01).
Section V THE SAFE DISPOSAL OF CLINICAL/DOMESTIC WASTE The Trust is currently reviewing the requirements of the recent guidelines Health Technical Memorandum Safe Management of Healthcare Waste (HTML 07-01).
Review Validation of aseptic processes for pharmaceuticals
 OPEM www.opem.org Oriental Pharmacy and Experimental Medicine 2010 10(4), 231-238 DOI 10.3742/OPEM.2010.10.4.231 Review Validation of aseptic processes for pharmaceuticals Lincy Joseph*, Mathew George
OPEM www.opem.org Oriental Pharmacy and Experimental Medicine 2010 10(4), 231-238 DOI 10.3742/OPEM.2010.10.4.231 Review Validation of aseptic processes for pharmaceuticals Lincy Joseph*, Mathew George
Ergonomic Risk Assessment Naval Facilities Engineering Command (NAVFAC) East Division
 Ergonomic Risk Assessment Naval Facilities Engineering Command (NAVFAC) East Division An ergonomic risk assessment was conducted on November 30th, 2000. The mail processing facility was observed in order
Ergonomic Risk Assessment Naval Facilities Engineering Command (NAVFAC) East Division An ergonomic risk assessment was conducted on November 30th, 2000. The mail processing facility was observed in order
Instructions for Use Reprocessed Balloon Inflation Devices
 Reprocessed by Instructions for Use Reprocessed Balloon Inflation Devices Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Reprocessed by Instructions for Use Reprocessed Balloon Inflation Devices Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
