14.1 Dynamic Equilibrium, Keq, and the Mass Action Expression
|
|
|
- Naomi Thompson
- 6 years ago
- Views:
Transcription
1 14.1 Dynamic Equilibrium, Keq, and the Mass Action Expression The Equilibrium Process Dr. Fred Omega Garces Chemistry 201 Miramar College 1 Dynamic Equilibrium, Keq and the Mass Action Expression
2 Extent of a Reaction Chemical Reaction Most reactions do not occur with 100% conversion to products. At the molecular, when a reaction occurs to form products, some products will back react to form reactants. The extent of the reaction i.e., 20% or 80% can be determine by measuring concentration of each component in solution In general the extent of the reaction is a function of the following: Temperature, concentration and degree of organization, all of which are monitored by some constant value called the The equilibrium constant, K eq 2 Dynamic Equilibrium, Keq and the Mass Action Expression
3 How Equilibrium is achieve Consider the reaction A D B Forward A g B rate f = k f [A] Reverse B f A rate b = k b [B] Overall A D B rate forward k f [A] = rate reverse k b [B] Example: N 2 O 4 D NO 2 Effect of temperature on the NO 2 D N 2 O 4 equilibrium. The tubes contain a mixture of NO 2 and N 2 O 4. As predicted by Le Chatelier s principle, the equilibrium favors colorless N 2 O 4 at lower temperatures. but shifts to the darker brown NO 2 at higher temperature. 3 Dynamic Equilibrium, Keq and the Mass Action Expression
4 Equilibrium: Mass Action Expression When equilibrium is establish, A D B D illustrates that rate forward = rate reverse or K f [A] = K b [B] Rearranging this equation yields K f /K b = [B] / [A] which yields The mass action expression : K eq = [B] / [A] For any generic chemical process at equilibrium aa + bb D pp + qq A mass action expression can be written: [ ] p [ Q] q K eq = P [ A] a B [ ] b This is also referred to as the Law of Mass Action 4 Dynamic Equilibrium, Keq and the Mass Action Expression
5 Haber Process Consider the Haber Process: N 2 (g) + 3H 2 (g) D 2NH 3 (g) (not 100 % process) As soon as NH 3 is form, it back reacts and forms N 2 and H 2. Law of Mass Action Or Mass Action Expression K eq = [ NH 3 ] 2 [ N 2 ] [ H 2 ] =K 3 c 5 Dynamic Equilibrium, Keq and the Mass Action Expression
6 Meaning of K eq : Sidebar Review of Fractions x x y < 1 : If x < y y > 1 : If x > y x y = 1 : If x = y A) For x < y, the denominator dominates. B) For x > y, the numerator dominates. C) For x = y, numerator and denominator are equal. 6 Dynamic Equilibrium, Keq and the Mass Action Expression
7 Which is favored, Reactant or Product? k eq = Meaning of K eq [ P] p [ Q] q = [ A] A [ B] b [ Product] x [ Reactant] y A) For K eq < 1, the ratio [Product] x < [Reactant] y B) For K eq > 1, the ratio [Product] x > [Reactant] y C) For K eq = 1, the ratio [Product] x = [Reactant] y A) For K eq < 1, at equilibrium Reactant is favored. B) For K eq > 1, at equilibrium Product is favored. C) For K eq = 1, at equilibrium Product and Reactant are equal. 7 Dynamic Equilibrium, Keq and the Mass Action Expression
8 Understanding the Concept 1. What is the distinction between rates and the extent of the reaction? 2. What does the term dynamic equilibrium mean physically and at the atomic level? 3. What does the magnitude of K eq mean in terms of the chemical reaction? 4. If there is no change in concentration of reactants and product at equilibrium, why is it considered dynamic? 8 Dynamic Equilibrium, Keq and the Mass Action Expression
9 For Reactions Not at Equilibrium For reactions not yet at equilibrium, the Law of Mass action yield information in terms of the Reaction Quotient. Consider the following chemical process not at equilibrium. aa + bb D rr + pp A reaction quotient expression can be written: Q = [R]r [P] p [A] a [B] b Where the numerical value of Q will determine the direction the reaction will proceed. Q < K eq Reaction shifts to g Right Q > K eq Reaction shifts to f Left 9 Dynamic Equilibrium, Keq and the Mass Action Expression
10 LeChatelier s Principle (Preview) Concentration Effect If a chemical system is at equilibrium and then a substance is added (either a reactant or product), the reaction will shift so as to re-establish equilibrium by subtracting part of the added substance. Conversely, removal of a substance will result in the reaction moving in the direction that forms more of the substance. Consider the Haber reaction that was discussed earlier. N 2 + 3H 2 D 2NH 3 + E If some H 2 is added to the reaction which was at equilibrium, the system self-adjust to remove the excess H 2 by converting it to NH 3 until equilibrium is re-establish; in the process some N 2 is also consumed. 10 Dynamic Equilibrium, Keq and the Mass Action Expression
11 Teeter Totter Pictorial View of LeChatelier At Equilibrium Stress applied Self Adjust Re-establish Equilibrium 11 Dynamic Equilibrium, Keq and the Mass Action Expression
12 Keq and the Q The change in Q during a reaction and its relation to Keq: Consider the reaction: N 2 O 4 D 2 NO 2 The reaction coordinate diagram shows how the concentration of N2O4 and NO2 changes as the reaction approaches equilibrium. This is also reflected in Q. As the reaction proceeds to the right, N2O4 to NO2, the Q value increases, N2O4 becomes smaller and NO2 becomes larger. The reaction finally reaches equilibrium at teq, at which time the concentration of N2O4 and NO2 remains constant. Equilibrium is reached and the reaction quotient becomes equal to the equilibrium constant. Q = K eq. 12 Dynamic Equilibrium, Keq and the Mass Action Expression
13 Heterogeneous Systems The equilibrium of substances with different phases. i.e., solid and aqueous coexisting - Solid and gas coexisting - Which solid is more concentrated? Which solid is more concentrated? Concentration of a solid is always a Constant; Even though the two containers contain different amounts of solid solute, as long as both solids are present at a given temperature, A) the concentration of the solution is the same, B) the partial pressure of the gas is the same. Concentration of solid remains the same (constant)! The solid component is not taken into account in the Mass Action expression or the Reaction Quotient. 13 Dynamic Equilibrium, Keq and the Mass Action Expression
14 Heterogeneous Systems CaCO 3 Consider the following reaction: CaCO 3 (s) D CaO (s) + CO 2 (g) [ ] [ CO 2 ] [ ] [ ] and [ CaCO 3 ] = Constant k eq = CaO CaCO 3 but, CaO [ ] [ CO 2 ] [ ] k eq = Constant 1 Constant 2 k eq = [ CO 2 ] The Mass Action Expression for a heterogenerous system need not take into account the concentration of the solid. Even though the two containers have different amount of solids (CaCO 3 and CaO), as long as both solids are present at the same temperature, the partial pressure of CO 2 is the same at equilibrium. 14 Dynamic Equilibrium, Keq and the Mass Action Expression
15 In Class exercise The reaction is in a 10.0 L vessel. The initial concentration of [H 2 ] is 12.0 molar and that of [N 2 ] is 4.0 molar. i) Sketch the change of the concentration of N 2 from 0 to 3.5 min. T = 200 K ii) At 2 minutes, Q is: a) increasing b) decreasing c) not changing d ) cannot be determine iii) At 5 minutes, Q is: a) increasing b) decreasing c) not changing d ) cannot be determine iv) Calculate the equilibrium constant at the 4.0-minute mark M -2 v) Calculate the equilibrium constant at the 8.0-minute mark M Dynamic Equilibrium, Keq and the Mass Action Expression
16 K eq as a Function of the Written Reaction What is the equilibrium constant if a reaction is written in the reverse direction? Consider, COCl 2 (g) D CO (g) + Cl 2 (g) (i) and the reverse reaction CO (g) + Cl 2 (g) D COCl 2 (g) (ii) What is the relationship of K eq between both reaction (i) & (ii)? K eq (i) = [CO] [Cl 2 ] [COCl 2 ] K eq (ii) = [COCl 2 ] [CO] [Cl 2 ] hence [CO] [Cl 2 ] [COCl 2 ] = " [COCl 2 ] % $ #[CO] [Cl 2 ] ' & 1 Inverse relationship - K eq (i) = K eq (ii) -1 K forward = 1 K reverse 16 Dynamic Equilibrium, Keq and the Mass Action Expression
17 Effects of K eq and Variation of a Chem Equation Consider the variation of a chemical reaction: A D B K eq = [B] / [A] B D A K rev = [A] / [B] K rev = 1 / K eq 1/2 A D 1/2 B K = [B] 1/2 / [A] 1/2 K = (K eq ) 1/2 2B D 2A K = [A] 2 / [B] 2 K = (K eq ) -2 n A D n B K = [B] n / [A] n K = (K eq ) n Note: K rev = 1 / K eq or K eq = 1 / K rev K = (K eq ) 1/2 or K eq = K 2 K = (K eq ) -2 or K eq = (K ) -1/2 17 Dynamic Equilibrium, Keq and the Mass Action Expression
18 Expressing Q and Calculating K The relationship of the Mass Expression, K eq, can be extended to the Reaction Quotient, Q: 18 Dynamic Equilibrium, Keq and the Mass Action Expression
19 Example 2: K eq relationship Consider the following equilibrium, at 480 C: 2Cl 2(g) + 2H 2 O (g) D 4 HCl (g) + O 2 (g) K p = a) What is the value of K p for: 4 HCl (g) + O 2 (g) D 2Cl 2(g) + 2H 2 O (g) b) What is the value of K p : Cl 2(g) + H 2 O (g) D 2HCl (g) + 1/2 O 2 (g) c) What is the value of K c : 2 Cl 2(g) + 2H 2 O (g) D 4HCl (g) + O 2 (g) (To be discussed later) a) This equation is the reverse of the original b) This equation is half the original c) This equation will be solve in the next series of slides. 19 Dynamic Equilibrium, Keq and the Mass Action Expression
20 Relationship Between K c and K p Consider the Reaction: COCl 2 (g) D CO (g) + Cl 2 (g) ( ) * ( p Cl ) 2 ( pcocl ) 2 K P = p CO It can be shown that: or K c = [CO] [Cl 2 ] [COCl 2 ] K c =K ( p RT) Δn K p =K ( c RT) +Δn [RT is positive exponent (+Δn)] where Δn = (gas moles) (gas moles) product reactant 20 Dynamic Equilibrium, Keq and the Mass Action Expression
21 Derivation K c and K p relationship [ ] [ P Cl 2 ] [ ] k p = PCO P COCl 2 but PV = nrt therefore k p = p CO [ ] [ Cl 2 ] [ ] k p = CO COCl 2 P = nrt V [ ] [ p Cl 2 ] [ pcocl 2 ] [ ] [ Cl 2 ] [ ] k p = CO COCl 2 and { = n V = CO [ ] [ Cl 2 ] [ ] k c = CO COCl 2 RT +1 = k c RT +Δn RT = [ conc] RT [ ] RT} {[ Cl 2 ] RT} {[ COCl 2 ] RT} [ = CO ] [ Cl 2 ] RT [ ] 1 RT RT RT COCl 2 k p = k c RT +Δn and k c = k p RT Δn where Δn = (gas moles) (gas moles) product reactant 21 Dynamic Equilibrium, Keq and the Mass Action Expression
22 Summary: Equilibrium constant The mass action expression is equal to the equilibrium constant. The equilibrium constant value is generally written without units. The equilibrium expression for a reaction is the reciprocal of that reaction when it is written in the reverse direction. When the balanced equation for a reaction is multiplied by a factor n, the equilibrium expression for the factored reaction is the original expression raised to the nth power. Equation 1 ref : R D P, Equation 2: nr D np, Thus K2 = (k1 ref ) n The equilibrium constant express in terms of concentration, Kc, is related to the equilibrium constant expressed in terms of partial pressure, Kp, by the amount of gas moles changes during the course of the reaction. 22 Dynamic Equilibrium, Keq and the Mass Action Expression
The Equilibrium Constant (K eq)
 The Constant (K eq) 1. The Law: K eq is called the. Keq = [PRODUCTS] [REACTANTS] eg. aa + bb + xx + yy + where a, b, x, y are of the balanced equation. A,B,X & Y are the and in the equation Keq = [X] x
The Constant (K eq) 1. The Law: K eq is called the. Keq = [PRODUCTS] [REACTANTS] eg. aa + bb + xx + yy + where a, b, x, y are of the balanced equation. A,B,X & Y are the and in the equation Keq = [X] x
Chapter 2 - Practice Test #1
 Chapter 2 - Practice Test #1 6. Chemical systems tend to move toward positions of A. minimum enthalpy and maximum entropy. B. maximum enthalpy and minimum entropy. C. minimum enthalpy and minimum entropy.
Chapter 2 - Practice Test #1 6. Chemical systems tend to move toward positions of A. minimum enthalpy and maximum entropy. B. maximum enthalpy and minimum entropy. C. minimum enthalpy and minimum entropy.
EQUILIBRIUM PRACTICE TEST
 EQUILIBRIUM PRACTICE TEST 1. Consider the following reaction: 2NH 3(g) N 2(g) + 3H 2(g) A flask is initially filled with NH 3. As the system approaches equilibrium, the rate of the forward reaction A.
EQUILIBRIUM PRACTICE TEST 1. Consider the following reaction: 2NH 3(g) N 2(g) + 3H 2(g) A flask is initially filled with NH 3. As the system approaches equilibrium, the rate of the forward reaction A.
Chemistry 12 Tutorial 6-Questions & Self-Test Only
 Chemistry 12 Tutorial 6 Questions and Self-Test Only 1. The equilibrium equation for the formation of ammonia is: N 2(g) + 3H 2(g) 2NH 3(g) In an equilibrium mixture at 200 C, the concentrations were found
Chemistry 12 Tutorial 6 Questions and Self-Test Only 1. The equilibrium equation for the formation of ammonia is: N 2(g) + 3H 2(g) 2NH 3(g) In an equilibrium mixture at 200 C, the concentrations were found
Name Practice Questions Date Equilibrium
 Name Practice Questions Date Equilibrium 1. CaCO3(s) «Ca 2+ (aq) + CO3 2 (aq) Which of the following reagents, when added to the above equilibrium system, would cause more CaCO3 to dissolve? KNO3(s) CaCO3(s)
Name Practice Questions Date Equilibrium 1. CaCO3(s) «Ca 2+ (aq) + CO3 2 (aq) Which of the following reagents, when added to the above equilibrium system, would cause more CaCO3 to dissolve? KNO3(s) CaCO3(s)
Review Package: Equilibrium Constant Keq & Calculations
 3. Consider the following equilibrium system: O2(g) + 2HF(g) OF2(g) H = +318 kj/mol If the temperature of the system is increased, A. Keq will decrease B. H will decrease C. Keq will increase D. H will
3. Consider the following equilibrium system: O2(g) + 2HF(g) OF2(g) H = +318 kj/mol If the temperature of the system is increased, A. Keq will decrease B. H will decrease C. Keq will increase D. H will
Chapter 17 Solubility and Complex Ion Equilibria
 Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
Review of Basic Concepts
 R Review of Basic Concepts Sections R.5 R.7 Copyright 2013, 2009, 2005 Pearson Education. Inc. R Review of Basic Concepts R.5 Rational Expressions R.6 Rational Exponents R.7 Radical Expressions Copyright
R Review of Basic Concepts Sections R.5 R.7 Copyright 2013, 2009, 2005 Pearson Education. Inc. R Review of Basic Concepts R.5 Rational Expressions R.6 Rational Exponents R.7 Radical Expressions Copyright
ENVIRONMENTAL MEASUREMENT. Engr. Yvonne Ligaya F. Musico
 ENVIRONMENTAL MEASUREMENT 1 Concentration in Environment Chemical concentration is one of the most important determinants in almost all aspects of chemical fate, transport, and treatment in both natural
ENVIRONMENTAL MEASUREMENT 1 Concentration in Environment Chemical concentration is one of the most important determinants in almost all aspects of chemical fate, transport, and treatment in both natural
AP*Chemistry Solubility Equilibria
 AP*Chemistry Solubility Equilibria SOLUBILITY EQUILIBRIA (K sp, THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Remember those solubility rules? The fine print
AP*Chemistry Solubility Equilibria SOLUBILITY EQUILIBRIA (K sp, THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Remember those solubility rules? The fine print
 Stoichiometric Calculations 1. The weight of calcium carbonate required to produce carbon-dioxide that is sufficient for conversion of one 0.1 mole sodium carbonate to sodium bicarbonate is 1) 1gm 2) 10gm
Stoichiometric Calculations 1. The weight of calcium carbonate required to produce carbon-dioxide that is sufficient for conversion of one 0.1 mole sodium carbonate to sodium bicarbonate is 1) 1gm 2) 10gm
1601 Redox Titration
 1601 Redox Titration Redox Titration in Analytical Chemistry Dr. Fred Omega Garces Analytical Chemistry 251 Miramar College, SDCCD Recommended: 1 Redox Titration Introduction Oxidation and reduction titrations
1601 Redox Titration Redox Titration in Analytical Chemistry Dr. Fred Omega Garces Analytical Chemistry 251 Miramar College, SDCCD Recommended: 1 Redox Titration Introduction Oxidation and reduction titrations
How Can Chemical Processes Be Designed to Optimise Efficiency?
 How Can Chemical Processes Be Designed to Optimise Efficiency? Area of Study 1: What are the Options for Energy Production? Area of Study 2: How Can the Versatility of Non-Metals Be Explained? What is
How Can Chemical Processes Be Designed to Optimise Efficiency? Area of Study 1: What are the Options for Energy Production? Area of Study 2: How Can the Versatility of Non-Metals Be Explained? What is
Process Design Decisions and Project Economics Dr. V. S. Moholkar Department of Chemical Engineering Indian Institute of Technology, Gawahati
 Process Design Decisions and Project Economics Dr. V. S. Moholkar Department of Chemical Engineering Indian Institute of Technology, Gawahati Module - 3 Reactor Design and Cost Estimation Lecture - 16
Process Design Decisions and Project Economics Dr. V. S. Moholkar Department of Chemical Engineering Indian Institute of Technology, Gawahati Module - 3 Reactor Design and Cost Estimation Lecture - 16
to water. You add 1 drop of phenolphthalein and the solution turns pink. This observation tells you Na 2
 1A End of Lab Questions Notes 9/11/18 Lab 4 1 You add 100 g of to water You add 1 drop of phenolphthalein and the solution turns pink This observation tells you is a base 2 You add vinegar (09 M acetic
1A End of Lab Questions Notes 9/11/18 Lab 4 1 You add 100 g of to water You add 1 drop of phenolphthalein and the solution turns pink This observation tells you is a base 2 You add vinegar (09 M acetic
The ion with Vanadium in its oxidation state of 5 exists as a solid compound in the form of a VO 3
 Chemistry of some Transition elements: V, Mn, Co, Cu, Cr and Ag Vanadium Vanadium has four main oxidation states. Each one has a different colour. VO 2 VO 2 V 3 V 2 O.N. 5 yellow O.N. 4 blue O.N. 3 green
Chemistry of some Transition elements: V, Mn, Co, Cu, Cr and Ag Vanadium Vanadium has four main oxidation states. Each one has a different colour. VO 2 VO 2 V 3 V 2 O.N. 5 yellow O.N. 4 blue O.N. 3 green
2/24/2003 OFB Chapter 9 1
 2/24/2003 OFB Chapter 9 1 Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4
2/24/2003 OFB Chapter 9 1 Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4
01/08/2018. Microbiology Lab. General Information. My Web Page 3 BIO3126
 Microbiology Lab BIO3126 1 General Information 2 Instructor: John Basso Email: jbasso@uottawa.ca Tel. 613-562-5800 Poste 6358 Office: BSC102 My web page: http://mysite.science.uottawa.ca/jbasso/home.htm
Microbiology Lab BIO3126 1 General Information 2 Instructor: John Basso Email: jbasso@uottawa.ca Tel. 613-562-5800 Poste 6358 Office: BSC102 My web page: http://mysite.science.uottawa.ca/jbasso/home.htm
EXPERIMENT 6. Determination of the Ideal Gas Law Constant - R. Magnesium metal reacts with hydrochloric acid according to the following reaction,
 EXPERIMENT 6 Determination of the Ideal Gas Law Constant - R Magnesium metal reacts with hydrochloric acid according to the following reaction, Mg + 2 HCl MgCl 2 + H 2 (g) In this experiment you will use
EXPERIMENT 6 Determination of the Ideal Gas Law Constant - R Magnesium metal reacts with hydrochloric acid according to the following reaction, Mg + 2 HCl MgCl 2 + H 2 (g) In this experiment you will use
concentration of acid in mol / dm 3 temperature / C ti / min
 1 (a A small piece of marble, calcium carbonate, was added to 5 cm 3 of hydrochloric acid at 25 C. The time taken for the reaction to stop was measured. CaCO 3 (s) + 2HCl(aq) CaCl 2 (aq) + CO 2 (g) + H
1 (a A small piece of marble, calcium carbonate, was added to 5 cm 3 of hydrochloric acid at 25 C. The time taken for the reaction to stop was measured. CaCO 3 (s) + 2HCl(aq) CaCl 2 (aq) + CO 2 (g) + H
Operations, vocabulary terms and examples
 Operations, vocabulary terms and examples oh my Fraction- a number that names a part of a whole numerator- the part or top number in a fraction denominator-the bottom or the total number in a fraction
Operations, vocabulary terms and examples oh my Fraction- a number that names a part of a whole numerator- the part or top number in a fraction denominator-the bottom or the total number in a fraction
NANDI NORTH DISTRICT JOINT MOCK EVALUATION TEST 2013
 NAME:. SIGNATURE: INDEX NO:. DATE :.. 233/1 CHEMISTRY PAPER 1 THEORY JULY / AUGUST 2013 TIME: 2 HOURS NANDI NORTH DISTRICT JOINT MOCK EVALUATION TEST 2013 Kenya Certificate of Secondary Education (K.C.S.E.)
NAME:. SIGNATURE: INDEX NO:. DATE :.. 233/1 CHEMISTRY PAPER 1 THEORY JULY / AUGUST 2013 TIME: 2 HOURS NANDI NORTH DISTRICT JOINT MOCK EVALUATION TEST 2013 Kenya Certificate of Secondary Education (K.C.S.E.)
Microbiology lab (BIO 3126) Teaching Staff. My Availability. Lab coordinator: John Basso. Lecturer: Benoît Pagé. By Office hours:
 Microbiology lab (BIO 3126) 1 Teaching Staff Lab coordinator: John Basso Email: jbasso@uottawa.ca Office: Bioscience 102 Tel.: 613-562-5800 Ext. 6358 Lecturer: Benoît Pagé Email: bpage051@uottawa.ca Office:
Microbiology lab (BIO 3126) 1 Teaching Staff Lab coordinator: John Basso Email: jbasso@uottawa.ca Office: Bioscience 102 Tel.: 613-562-5800 Ext. 6358 Lecturer: Benoît Pagé Email: bpage051@uottawa.ca Office:
STANDARD 1 NUMBER and OPERATION
 Goal 1.1: Understand and use numbers. STANDARD 1 NUMBER and OPERATION 6.M.1.1.1 Compare magnitudes and relative magnitudes of positive rational numbers, including whole numbers through billions, fractions,
Goal 1.1: Understand and use numbers. STANDARD 1 NUMBER and OPERATION 6.M.1.1.1 Compare magnitudes and relative magnitudes of positive rational numbers, including whole numbers through billions, fractions,
CHEM 254 EXPERIMENT 8. Phase Diagrams, Solid - Liquid Phase Equilibrium for Two Component System and Missibility Gap
 Temperature, T Temperature, T CHEM 254 EXPERIMENT 8 Phase Diagrams, Solid - Liquid Phase Equilibrium for Two Component System and Missibility Gap Solid and liquid phases may both be present in a system
Temperature, T Temperature, T CHEM 254 EXPERIMENT 8 Phase Diagrams, Solid - Liquid Phase Equilibrium for Two Component System and Missibility Gap Solid and liquid phases may both be present in a system
Rational Numbers in Mathematics. Lecture 2.1: Fractions INTRODUCTION 2.0
 Rational Numbers in Mathematics INTRODUCTION 2.0 In this lesson, we will focus on the exploration of rational numbers in all their forms and operation on rational numbers. We will explore fractions and
Rational Numbers in Mathematics INTRODUCTION 2.0 In this lesson, we will focus on the exploration of rational numbers in all their forms and operation on rational numbers. We will explore fractions and
Unit F FR (part B) Solubility Equilibrium, Ksp (pg 1 of 16)
 Unit F FR (part B) Solubility Equilibrium, Ksp (pg 1 of 16) 1. Answer the following questions about the solubility of some fluoride salts of alkaline earth metals. (a) A student prepares 100. ml of a saturated
Unit F FR (part B) Solubility Equilibrium, Ksp (pg 1 of 16) 1. Answer the following questions about the solubility of some fluoride salts of alkaline earth metals. (a) A student prepares 100. ml of a saturated
Chapter 10 Material Balances for Processes Involving Reaction 10.1 Species Material Balances Processes Involving a Single Reaction
 Chapter 10 Material Balances for Processes Involving Reaction 10.1 Species Material Balances 10.1.1 Processes Involving a Single Reaction The material balance for a species must be augmented to include
Chapter 10 Material Balances for Processes Involving Reaction 10.1 Species Material Balances 10.1.1 Processes Involving a Single Reaction The material balance for a species must be augmented to include
Solubility Equi librium Ksp. Chapter 17 Sections
 Solubility Equi librium Ksp Chapter 17 Sections 17.4 17.6 1 Precipitation Reactions Earlier in this course, we considered precipitation reactions zinc nitrate reacts with sodium hydroxide Zn(NO3)2 + 2NaOH
Solubility Equi librium Ksp Chapter 17 Sections 17.4 17.6 1 Precipitation Reactions Earlier in this course, we considered precipitation reactions zinc nitrate reacts with sodium hydroxide Zn(NO3)2 + 2NaOH
THE UNIVERSITY OF JORDAN
 THE UNIVERSITY OF JORDAN The University of Jordan Faculty of Engineering & Technology Chemical Engineering Department Fuel and Energy Material Balance Part 3: Combustion Reactions Dr.-Ing. Zayed Al-Hamamre
THE UNIVERSITY OF JORDAN The University of Jordan Faculty of Engineering & Technology Chemical Engineering Department Fuel and Energy Material Balance Part 3: Combustion Reactions Dr.-Ing. Zayed Al-Hamamre
Topic Reacting masses Level GCSE Outcomes 1. To calculate reacting masses 2. To set out mole calculations in a grid format
 Topic Reacting masses Level GCSE Outcomes 1. To calculate reacting masses 2. To set out mole calculations in a grid format Problems on Reacting Masses of Solids Section 1 1. What is the mass of magnesium
Topic Reacting masses Level GCSE Outcomes 1. To calculate reacting masses 2. To set out mole calculations in a grid format Problems on Reacting Masses of Solids Section 1 1. What is the mass of magnesium
WJEC England GCSE Chemistry. Topic 11: Production, use and disposal of important chemicals and materials. Notes
 WJEC England GCSE Chemistry Topic 11: Production, use and disposal of important chemicals and materials Notes (Content in bold is for Higher Tier only) The Haber process Used to manufacture ammonia, which
WJEC England GCSE Chemistry Topic 11: Production, use and disposal of important chemicals and materials Notes (Content in bold is for Higher Tier only) The Haber process Used to manufacture ammonia, which
SOFC Modeling Considering Internal Reforming by a Global Kinetics Approach. and My Research in General
 SOFC Modeling Considering Internal Reforming by a Global Kinetics Approach and My Research in General Martin Andersson Division of Heat Transfer, Department of Energy Sciences, Faculty of Engineering (LTH),
SOFC Modeling Considering Internal Reforming by a Global Kinetics Approach and My Research in General Martin Andersson Division of Heat Transfer, Department of Energy Sciences, Faculty of Engineering (LTH),
2. Fractions. 5. Greatest Common Factor (Sometimes called the Greatest Common Divisor)
 2. Fractions 1. What is a Fraction? 2. Improper Fractions 3. Mixed umbers 4. Comparing Fractions 5. Greatest Common Factor (Sometimes called the Greatest Common Divisor) 6. Least Common Multiple (Sometimes
2. Fractions 1. What is a Fraction? 2. Improper Fractions 3. Mixed umbers 4. Comparing Fractions 5. Greatest Common Factor (Sometimes called the Greatest Common Divisor) 6. Least Common Multiple (Sometimes
Labs Covered. Le Chatelier. Calculating Ka. Kitchen Chemistry Project. Calculating Kc. Redox lab
 Labs Covered Le Chatelier Calculating Kc Calculating Ka Redox lab Kitchen Chemistry Project LeChatelier Sample Questions Fe +3 (aq) + SCN -1 (aq) FeSCN +2 (aq) Light orange colourless red a) To create
Labs Covered Le Chatelier Calculating Kc Calculating Ka Redox lab Kitchen Chemistry Project LeChatelier Sample Questions Fe +3 (aq) + SCN -1 (aq) FeSCN +2 (aq) Light orange colourless red a) To create
Solubility Equilibrium
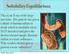 Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Carbon dioxide is formed during the complete combustion of octane. + 12½O 2 8CO 2... [1] Why does incomplete combustion sometimes take place?
![Carbon dioxide is formed during the complete combustion of octane. + 12½O 2 8CO 2... [1] Why does incomplete combustion sometimes take place? Carbon dioxide is formed during the complete combustion of octane. + 12½O 2 8CO 2... [1] Why does incomplete combustion sometimes take place?](/thumbs/81/82825046.jpg) 1 Crude oil is a source of hydrocarbons which can be used as fuels or for processing into petrochemicals. Octane, C 8 18, is one of the alkanes present in petrol. Carbon dioxide is formed during the complete
1 Crude oil is a source of hydrocarbons which can be used as fuels or for processing into petrochemicals. Octane, C 8 18, is one of the alkanes present in petrol. Carbon dioxide is formed during the complete
Lecture 3: Solutions: Activities and. Phase Diagrams
 Lecture 3: Solutions: Activities and Lecture plan: Phase Diagrams 21-09-2010 Gibbs phase rule vapour composition two-component phase diagrams phase diagrams in material science: microstructures in isomorphous
Lecture 3: Solutions: Activities and Lecture plan: Phase Diagrams 21-09-2010 Gibbs phase rule vapour composition two-component phase diagrams phase diagrams in material science: microstructures in isomorphous
Experiment 3: Determination of an Empirical Formula
 Background Information The composition of a compound is defined by its chemical formula, which gives the number ratio of the different elements in the compound. For example, water has a fixed composition
Background Information The composition of a compound is defined by its chemical formula, which gives the number ratio of the different elements in the compound. For example, water has a fixed composition
Determine whether the metal is magnesium, iron, or zinc based on the value of the calculated molar mass.
 Gases Part A: A student working at METAL Company found an unlabelled bottle of a metal in the lab. The metal could be magnesium, iron, or zinc. Each of these metals react with dilute hydrochloric acid
Gases Part A: A student working at METAL Company found an unlabelled bottle of a metal in the lab. The metal could be magnesium, iron, or zinc. Each of these metals react with dilute hydrochloric acid
CARBONATION OF WOLLASTONITE USING SUPERCRITICAL CARBON DIOXIDE
 CARBONATION OF WOLLASTONITE USING SUPERCRITICAL CARBON DIOXIDE Clifford Y. Tai*, W.-R. Chen and Shin-Min Shih Department of Chemical Engineering National Taiwan University Email: cytai@ntu.edu.tw The feasibility
CARBONATION OF WOLLASTONITE USING SUPERCRITICAL CARBON DIOXIDE Clifford Y. Tai*, W.-R. Chen and Shin-Min Shih Department of Chemical Engineering National Taiwan University Email: cytai@ntu.edu.tw The feasibility
GraspIT AQA GCSE Chemical changes
 A. Reactivity of metals The reactivity series, metal oxides and extractions 1. Three metals, X, Y and Z were put into water. The reactions are shown below: a) Use the diagrams to put metals X, Y and Z
A. Reactivity of metals The reactivity series, metal oxides and extractions 1. Three metals, X, Y and Z were put into water. The reactions are shown below: a) Use the diagrams to put metals X, Y and Z
Total Grade /150 Checked by
 FIRST LETTER OF YOUR LAST NAME CHEMISTRY 1127 EXAM I NAME (PRINT) SECTION SIGNATURE TA PLEASE READ THE FOLLOWING INSTRUCTIONS Do NOT begin the exam until asked to do so. There are 8 numbered pages, a useful
FIRST LETTER OF YOUR LAST NAME CHEMISTRY 1127 EXAM I NAME (PRINT) SECTION SIGNATURE TA PLEASE READ THE FOLLOWING INSTRUCTIONS Do NOT begin the exam until asked to do so. There are 8 numbered pages, a useful
Use equivalent fractions as a strategy to add and subtract fractions.
 Standard 8 Benchmark 1 3 4 5 6 Numbers and Operations - Fractions Develop understanding of fractions as numbers. Extend understanding of fraction equivalence and ordering. Use equivalent fractions as a
Standard 8 Benchmark 1 3 4 5 6 Numbers and Operations - Fractions Develop understanding of fractions as numbers. Extend understanding of fraction equivalence and ordering. Use equivalent fractions as a
Example 1: Cost of Operation. Example 2: Education Pays Off. 3.1: Algebraic Translations of English Phrases. 3.2: Strategy for Solving Word Problems
 3.1: Algebraic Translations of English Phrases 3.2: Strategy for Solving Word Problems Addition Subtraction Multiplication Division sum more than increased by plus added to Total of sum of 6 and 7 3 more
3.1: Algebraic Translations of English Phrases 3.2: Strategy for Solving Word Problems Addition Subtraction Multiplication Division sum more than increased by plus added to Total of sum of 6 and 7 3 more
Point out that when you compare two sets by using fractions, the objects in the sets do not need to be the same size or even the same kind.
 Fractions What are some of the ways we use fractions every day? Fractions represent a part of a whole object or unit. (Provide some sets of objects for students to work with to help them understand the
Fractions What are some of the ways we use fractions every day? Fractions represent a part of a whole object or unit. (Provide some sets of objects for students to work with to help them understand the
Quantitive Chemistry Question paper
 Quantitive Chemistry Question paper Level Subject Exam Board Topic Sub-Topic Booklet GCSE Chemistry CCEA Quantitative Chemistry Quantitive Chemistry Question paper Time Allowed: 93 minutes Score: /77 Percentage:
Quantitive Chemistry Question paper Level Subject Exam Board Topic Sub-Topic Booklet GCSE Chemistry CCEA Quantitative Chemistry Quantitive Chemistry Question paper Time Allowed: 93 minutes Score: /77 Percentage:
Unit 3 - Solubility Practice Test # 2
 1. onsider the following experiment: Unit 3 Solubility Practice Test # 2 1.0 ml 0.20 M g + + an unknown solution precipitate 1.0 ml 020 M Sr 2+ + an unknown solution no precipitate The unknown solution
1. onsider the following experiment: Unit 3 Solubility Practice Test # 2 1.0 ml 0.20 M g + + an unknown solution precipitate 1.0 ml 020 M Sr 2+ + an unknown solution no precipitate The unknown solution
3. [7 points] How many significant figures should there be in the answer to the following problem?
![3. [7 points] How many significant figures should there be in the answer to the following problem? 3. [7 points] How many significant figures should there be in the answer to the following problem?](/thumbs/83/87492512.jpg) 1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
Some Basic Concepts of Chemistry
 Q 1. Some Basic Concepts of Chemistry What weight of AgCI will be precipitated when a solution containing 4.77 g of NaCI is added to a solution of 5.77 g of AgNO 3? (IIT JEE 1978 3 Marks) Q 2. One gram
Q 1. Some Basic Concepts of Chemistry What weight of AgCI will be precipitated when a solution containing 4.77 g of NaCI is added to a solution of 5.77 g of AgNO 3? (IIT JEE 1978 3 Marks) Q 2. One gram
Chemical reactions and electrolysis
 Chemical reactions and electrolysis Higher Revision Questions Name: Class: Date: Time: 95 minutes Marks: 95 marks Comments: Page of 29 (a) Magnesium metal is shaped to make magnesium ribbon. Explain why
Chemical reactions and electrolysis Higher Revision Questions Name: Class: Date: Time: 95 minutes Marks: 95 marks Comments: Page of 29 (a) Magnesium metal is shaped to make magnesium ribbon. Explain why
REPORTING CATEGORY I NUMBER, OPERATION, QUANTITATIVE REASONING
 Texas 6 th Grade STAAR (TEKS)/ Correlation Skills (w/ STAAR updates) Stretch REPORTING CATEGORY I NUMBER, OPERATION, QUANTITATIVE REASONING (6.1) Number, operation, and quantitative reasoning. The student
Texas 6 th Grade STAAR (TEKS)/ Correlation Skills (w/ STAAR updates) Stretch REPORTING CATEGORY I NUMBER, OPERATION, QUANTITATIVE REASONING (6.1) Number, operation, and quantitative reasoning. The student
Thermodynamics and Electrode Potential ME Dr. Zuhair M. Gasem
 Thermodynamics and Electrode Potential ME 472-062 Copyright Dr. Zuhair M. Gasem Corrosion Science and Engineering 2 Corrosion Science Engineering: corrosion forms, and controlling methods Chpater2 Thermodynamics
Thermodynamics and Electrode Potential ME 472-062 Copyright Dr. Zuhair M. Gasem Corrosion Science and Engineering 2 Corrosion Science Engineering: corrosion forms, and controlling methods Chpater2 Thermodynamics
+ 10Cl 2Mn H 2 O + 5Cl 2. Deduce the half-equation for the oxidation of chloride ions to chlorine.
 Q1.Chlorine is an important industrial chemical. (a) Chlorine is formed when KMnO 4 reacts with hydrochloric acid. The ionic equation for this redox reaction is 16H + + 2MnO 4 + 10Cl 2Mn 2+ + 8H 2 O +
Q1.Chlorine is an important industrial chemical. (a) Chlorine is formed when KMnO 4 reacts with hydrochloric acid. The ionic equation for this redox reaction is 16H + + 2MnO 4 + 10Cl 2Mn 2+ + 8H 2 O +
Differences in Free Energy of Solvation in a Two-Layer System
 Differences in Free Energy of Solvation in a Two-Layer System (Original exercise designed by William Steel and modified by James Foresman) Purpose: By observing the changes in the concentration of a solute
Differences in Free Energy of Solvation in a Two-Layer System (Original exercise designed by William Steel and modified by James Foresman) Purpose: By observing the changes in the concentration of a solute
NATIONAL BUSINESS AND TECHNICAL EXAMINATIONS BOARD (GENERAL EDUCATION EXAMINATION) MAY/JUNE 2007 SECTION B CHEMISTRY (ESSAY) TIME: 1 HOUR 40 MINUTES
 NATIONAL BUSINESS AND TECHNICAL EXAMINATIONS BOARD (GENERAL EDUCATION EXAMINATION) MAY/JUNE 2007 SECTION B CHEMISTRY (ESSAY) TIME: 1 HOUR 40 MINUTES 1. (a) Give THREE differences between a physical and
NATIONAL BUSINESS AND TECHNICAL EXAMINATIONS BOARD (GENERAL EDUCATION EXAMINATION) MAY/JUNE 2007 SECTION B CHEMISTRY (ESSAY) TIME: 1 HOUR 40 MINUTES 1. (a) Give THREE differences between a physical and
MSE 352 Engineering Ceramics II
 Kwame Nkrumah University of Science & Technology, Kumasi, Ghana MSE 352 Engineering Ceramics II Ing. Anthony Andrews (PhD) Department of Materials Engineering Faculty of Mechanical and Chemical Engineering
Kwame Nkrumah University of Science & Technology, Kumasi, Ghana MSE 352 Engineering Ceramics II Ing. Anthony Andrews (PhD) Department of Materials Engineering Faculty of Mechanical and Chemical Engineering
The Predom module -O 2. at different temperatures) One-metal
 The Predom module Use Predom to calculate and plot isothermal 1-, 2- and 3-metal predominance area diagrams. Predom makes exclusively use of compound databases. Table of Contents Section 1 Section 2 Section
The Predom module Use Predom to calculate and plot isothermal 1-, 2- and 3-metal predominance area diagrams. Predom makes exclusively use of compound databases. Table of Contents Section 1 Section 2 Section
ACTIVITY: Dividing by a Fraction. Work with a partner. Write the division problem and solve it using a model. a. How many two-thirds are in three?
 . Dividing Fractions How can you divide by a fraction? ACTIVITY: Dividing by a Fraction Work with a partner. Write the division problem and solve it using a model. a. How many two-thirds are in three?
. Dividing Fractions How can you divide by a fraction? ACTIVITY: Dividing by a Fraction Work with a partner. Write the division problem and solve it using a model. a. How many two-thirds are in three?
Experiment 1 Hard-Soft Acids and Bases: Altering the Cu + /Cu 2+ Equilibrium with Nitrogen, Oxygen, or Halide Ligands
 Experiment 1 Hard-Soft Acids and Bases: Altering the Cu + /Cu 2+ Equilibrium with Nitrogen, Oxygen, or Halide Ligands *Parts of the experiment are based on C. E. Ophardt & G. Wollaston, J. Chem. Ed. 1991,
Experiment 1 Hard-Soft Acids and Bases: Altering the Cu + /Cu 2+ Equilibrium with Nitrogen, Oxygen, or Halide Ligands *Parts of the experiment are based on C. E. Ophardt & G. Wollaston, J. Chem. Ed. 1991,
EMP I RICAL FORMULA OF MAGNESI U M OXIDE
 Experiment 6 Name: 53 EMP I RICAL FORMULA OF MAGNESI U M OXIDE In this experiment, you will synthesize oxide via the reaction pathways summarized in Figure 1. Note that [1] is the main reaction and [2]
Experiment 6 Name: 53 EMP I RICAL FORMULA OF MAGNESI U M OXIDE In this experiment, you will synthesize oxide via the reaction pathways summarized in Figure 1. Note that [1] is the main reaction and [2]
Fugacity From Wikipedia, the free encyclopedia
 頁 1 / 5 Fugacity From Wikipedia, the free encyclopedia In chemical thermodynamics, the fugacity (f) of a real gas is an effective pressure which replaces the true mechanical pressure in accurate chemical
頁 1 / 5 Fugacity From Wikipedia, the free encyclopedia In chemical thermodynamics, the fugacity (f) of a real gas is an effective pressure which replaces the true mechanical pressure in accurate chemical
14.4 (Chp16) Solubility and Solubility Product
 14.4 (Chp16) Solubility and Solubility Product What drives substances to dissolve and others to remain as a precipitate? Dr. Fred Omega Garces Chemistry 201 Miramar College 1 Solubility and Solubility
14.4 (Chp16) Solubility and Solubility Product What drives substances to dissolve and others to remain as a precipitate? Dr. Fred Omega Garces Chemistry 201 Miramar College 1 Solubility and Solubility
The project goal is to design a process capable of converting methane, obtained from remote
 Introduction The project goal is to design a process capable of converting methane, obtained from remote sites, to a more easily transportable fuel, methanol. The design should focus on minimizing the
Introduction The project goal is to design a process capable of converting methane, obtained from remote sites, to a more easily transportable fuel, methanol. The design should focus on minimizing the
Date: October 11, 2012 Section Number: Chem
 Experiment 1: Oxidation of an Alcohol Report Sheet Last Name: Siva First Name: Krishnaa Date: October 11, 2012 Section Number: Chem 222-05 Objective: 1 mark (What is the purpose of this experiment?) The
Experiment 1: Oxidation of an Alcohol Report Sheet Last Name: Siva First Name: Krishnaa Date: October 11, 2012 Section Number: Chem 222-05 Objective: 1 mark (What is the purpose of this experiment?) The
CCE PR KARNATAKA SECONDARY EDUCATION EXAMINATION BOARD, MALLESWARAM, BANGALORE S. S. L. C. EXAMINATION, JUNE, 2017 MODEL ANSWERS
 CCE PR 560 003 KARNATAKA SECONDARY EDUCATION EXAMINATION BOARD, MALLESWARAM, BANGALORE 560 003 07 S. S. L. C. EXAMINATION, JUNE, 07 MODEL ANSWERS :. 06. 07 ] : 83-E (Chem.) Date :. 06. 07 ] CODE NO. :
CCE PR 560 003 KARNATAKA SECONDARY EDUCATION EXAMINATION BOARD, MALLESWARAM, BANGALORE 560 003 07 S. S. L. C. EXAMINATION, JUNE, 07 MODEL ANSWERS :. 06. 07 ] : 83-E (Chem.) Date :. 06. 07 ] CODE NO. :
EXERGY ANALYSIS P.V. Aravind Assistant Professor, Delft University of Technology Visiting Lecturer, TU Munich
 EXERGY ANALYSIS P.V. Aravind Assistant Professor, Delft University of Technology Visiting Lecturer, TU Munich EXERGY definition calculation of exergy values (incl. chemical exergy) calculation of exergy
EXERGY ANALYSIS P.V. Aravind Assistant Professor, Delft University of Technology Visiting Lecturer, TU Munich EXERGY definition calculation of exergy values (incl. chemical exergy) calculation of exergy
, and note the. Student Outcomes. Classwork. Discussion (8 minutes) Example 1 (5 minutes) Students write numerical expressions in two forms, and
 Student Outcomes Students write numerical expressions in two forms, and, and note the relationship between the two. Classwork Discussion (8 minutes) The discussion will serve as a chance for students to
Student Outcomes Students write numerical expressions in two forms, and, and note the relationship between the two. Classwork Discussion (8 minutes) The discussion will serve as a chance for students to
Chapter 9. Dissolution and Precipitation Equilibria
 Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4 The Effects of ph on Solubility
Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4 The Effects of ph on Solubility
Phase diagrams are diagrammatic representations of the phases present in a
 Chapter 4 What is a binary phase diagram? Phase diagrams are diagrammatic representations of the phases present in a system under specified equilibrium conditions, most often composition, temperature and
Chapter 4 What is a binary phase diagram? Phase diagrams are diagrammatic representations of the phases present in a system under specified equilibrium conditions, most often composition, temperature and
Monroe County School District Elementary Pacing Guide
 Grade 5 Unit 1: Place Value/Operations/Algebraic Thinking Timeline: August 3-September 21 CMA: Week of September 24 th I can recite multiplication facts (1-12) Fluency Tests 5.NBT.5 Multi-Digit multiplication
Grade 5 Unit 1: Place Value/Operations/Algebraic Thinking Timeline: August 3-September 21 CMA: Week of September 24 th I can recite multiplication facts (1-12) Fluency Tests 5.NBT.5 Multi-Digit multiplication
Equilibria of Slightly Soluble Ionic Compounds: We lied to you in chem 115
 Precipitation Reactions Predicting Whether a Precipitate Will Form Note the ions present in the reactants Consider all possible cation-anion combinations Use the solubility rules to decide whether any
Precipitation Reactions Predicting Whether a Precipitate Will Form Note the ions present in the reactants Consider all possible cation-anion combinations Use the solubility rules to decide whether any
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
1. Which of the given statements about the reaction below are incorrect?
 1. Which of the given statements about the reaction below are incorrect? 2PbO(s) + C(s) 2Pb(s) + CO 2 (g) a. Lead is getting reduced b. Carbon dioxide is getting oxidised c. Carbon is getting oxidised
1. Which of the given statements about the reaction below are incorrect? 2PbO(s) + C(s) 2Pb(s) + CO 2 (g) a. Lead is getting reduced b. Carbon dioxide is getting oxidised c. Carbon is getting oxidised
SUCCEEDING IN THE VCE 2017 UNIT 3 CHEMISTRY STUDENT SOLUTIONS
 SUCCEEDING IN THE VCE 017 UNIT CHEMISTRY STUDENT SOLUTIONS FOR ERRORS AND UPDATES, PLEASE VISIT WWW.TSFX.COM.AU/VCE-UPDATES QUESTION 1 The word "released" takes the negative sign into consideration. Answer
SUCCEEDING IN THE VCE 017 UNIT CHEMISTRY STUDENT SOLUTIONS FOR ERRORS AND UPDATES, PLEASE VISIT WWW.TSFX.COM.AU/VCE-UPDATES QUESTION 1 The word "released" takes the negative sign into consideration. Answer
Lafayette College Department of Civil and Environmental Engineering
 Lafayette College Department of Civil and Environmental Engineering CE 321: Environmental Engineering and Science Fall 2017 Homework #12 Due Tuesday: 12/12/17 SOLUTIONS The town of Easton is considering
Lafayette College Department of Civil and Environmental Engineering CE 321: Environmental Engineering and Science Fall 2017 Homework #12 Due Tuesday: 12/12/17 SOLUTIONS The town of Easton is considering
Chapter 12: Intermolecular Forces and Liquids and Solids
 1. Which one of the following substances is expected to have the highest boiling point? A) Br 2 B) Cl 2 C) F 2 D) I 2 3. Which one of the following substances is expected to have the highest boiling point?
1. Which one of the following substances is expected to have the highest boiling point? A) Br 2 B) Cl 2 C) F 2 D) I 2 3. Which one of the following substances is expected to have the highest boiling point?
CO 2 Emission. (2) Scientific processes. The student uses scientific methods during field and laboratory investigations. The student is expected to:
 CO 2 Emission Subject: Chemistry Grade Level: 11 th Rational or Purpose: This activity is intended to strengthen students concepts on scientific notation, unit conversion, and moles. The content will help
CO 2 Emission Subject: Chemistry Grade Level: 11 th Rational or Purpose: This activity is intended to strengthen students concepts on scientific notation, unit conversion, and moles. The content will help
Determination of the Empirical Formula of Magnesium Oxide
 Determination of the Empirical Formula of Magnesium Oxide The quantitative stoichiometric relationships governing mass and amount will be studied using the combustion reaction of magnesium metal. Magnesium
Determination of the Empirical Formula of Magnesium Oxide The quantitative stoichiometric relationships governing mass and amount will be studied using the combustion reaction of magnesium metal. Magnesium
Chapter 18 Homework Answers
 Chapter 18 Homework Answers 18.19 (a) AgI(s) Ag + + I K sp = [Ag + ][I ] (b) Ag PO (s) Ag + + PO K sp = [Ag + ] [PO ] (c) PbCrO (s) Pb + + CrO K sp = [Pb + ][CrO ] (d) Al(OH) (s) Al + + OH K sp = [Al +
Chapter 18 Homework Answers 18.19 (a) AgI(s) Ag + + I K sp = [Ag + ][I ] (b) Ag PO (s) Ag + + PO K sp = [Ag + ] [PO ] (c) PbCrO (s) Pb + + CrO K sp = [Pb + ][CrO ] (d) Al(OH) (s) Al + + OH K sp = [Al +
Solubility Equi librium Ksp. Chapter 17 Sections
 Solubility Equi librium Ksp Chapter 17 Sections 17.4 17.6 Precipitation Reactions Earlier in this course, we considered precipitation reactions zinc nitrate reacts with sodium hydroxide Zn(NO3)2 + 2NaOH
Solubility Equi librium Ksp Chapter 17 Sections 17.4 17.6 Precipitation Reactions Earlier in this course, we considered precipitation reactions zinc nitrate reacts with sodium hydroxide Zn(NO3)2 + 2NaOH
Solidification & Binary Phase Diagrams II. Solidification & Binary Phase Diagrams II
 Module 19 Solidification & inary Phase Diagrams II ecture 19 Solidification & inary Phase Diagrams II 1 NPTE Phase II : IIT Kharagpur : Prof. R. N. hosh, Dept of Metallurgical and Materials Engineering
Module 19 Solidification & inary Phase Diagrams II ecture 19 Solidification & inary Phase Diagrams II 1 NPTE Phase II : IIT Kharagpur : Prof. R. N. hosh, Dept of Metallurgical and Materials Engineering
Lecture 11. Solubility of Ionic Compounds in Water
 Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
Numerical investigation of separation of metals by non-steady-state cyclic counter-current liquid-liquid extraction.
 Research Article http://www.alliedacademies.org/journal-industrial-environmental-chemistry/ Numerical investigation of separation of metals by non-steady-state cyclic counter-current liquid-liquid extraction.
Research Article http://www.alliedacademies.org/journal-industrial-environmental-chemistry/ Numerical investigation of separation of metals by non-steady-state cyclic counter-current liquid-liquid extraction.
ICSE-Science 2 (Chemistry) 2004
 ICSE-Science 2 (Chemistry) 2004 Answers to this Paper must be written on the paper provided separately. You will not be allowed to write during the first 15 minutes. This time is to be spent in reading
ICSE-Science 2 (Chemistry) 2004 Answers to this Paper must be written on the paper provided separately. You will not be allowed to write during the first 15 minutes. This time is to be spent in reading
Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below :
 Questions:- Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below : Ammonium chloride, ammonium nitrate, chlorine, dilute hydrochloric
Questions:- Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below : Ammonium chloride, ammonium nitrate, chlorine, dilute hydrochloric
5. Supplemental Materials
 . Supplemental Materials a. Fractions Tiles b. Fraction Circles c. Fraction Addition Flash Cards d. Fraction Subtraction Flash Cards e. Fraction Cards f. Multiplication Chart g. Improper Fraction Cards
. Supplemental Materials a. Fractions Tiles b. Fraction Circles c. Fraction Addition Flash Cards d. Fraction Subtraction Flash Cards e. Fraction Cards f. Multiplication Chart g. Improper Fraction Cards
SSRG International Journal of Chemical Engineering Research ( SSRG IJCER ) Volume 5 Issue 2 May to Aug 2018
 Simulation and Energy Optimization of Ammonia Synthesis Loop Ashutosh Desai#1, Shreyans Shah#2, Sanchit Goyal#3 Under the guidance of, Prof. Arvind Prasad Department of chemical engineering, Dwarkadas.J.Sanghavi
Simulation and Energy Optimization of Ammonia Synthesis Loop Ashutosh Desai#1, Shreyans Shah#2, Sanchit Goyal#3 Under the guidance of, Prof. Arvind Prasad Department of chemical engineering, Dwarkadas.J.Sanghavi
ADVANCES in NATURAL and APPLIED SCIENCES
 ADVANCES in NATURAL and APPLIED SCIENCES ISSN: 1995-772 Published BY AENSI Publication EISSN: 1998-19 http://www.aensiweb.com/anas 216 April 1(4): pages 472-477 Open Access Journal Kinetic Modeling of
ADVANCES in NATURAL and APPLIED SCIENCES ISSN: 1995-772 Published BY AENSI Publication EISSN: 1998-19 http://www.aensiweb.com/anas 216 April 1(4): pages 472-477 Open Access Journal Kinetic Modeling of
Kenya Certificate of Secondary Education (K.C.S.E.)
 Name: Index No. School:. Candidate s Sign.... Date:... 233/2 CHEMISTRY PAPER 2 JULY/AUGUST 2011 TIME: 2 HOURS Kenya Certificate of Secondary Education (K.C.S.E.) Chemistry Paper 2 INSTRUCTIONS TO CANDIDATES:
Name: Index No. School:. Candidate s Sign.... Date:... 233/2 CHEMISTRY PAPER 2 JULY/AUGUST 2011 TIME: 2 HOURS Kenya Certificate of Secondary Education (K.C.S.E.) Chemistry Paper 2 INSTRUCTIONS TO CANDIDATES:
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.1476 Ammonia synthesis using a stable electride as an electron donor and reversible hydrogen store Masaaki Kitano, 1 Yasunori Inoue, 1 Youhei Yamazaki, 1 Fumitaka Hayashi, 2 Shinji Kanbara,
DOI: 10.1038/NCHEM.1476 Ammonia synthesis using a stable electride as an electron donor and reversible hydrogen store Masaaki Kitano, 1 Yasunori Inoue, 1 Youhei Yamazaki, 1 Fumitaka Hayashi, 2 Shinji Kanbara,
Department of Civil Engineering, IIT Delhi (India) CVL722 (Sem1, ) (Dr. Arun Kumar;
 Department of Civil Engineering, IIT Delhi (India) CVL722 (Sem1, 2015-16) (Dr. Arun Kumar; Email: arunku@civil.iitd.ac.in) Set 1 Do not submit. Please mention your assumptions explicitly. Q1. Say a raw
Department of Civil Engineering, IIT Delhi (India) CVL722 (Sem1, 2015-16) (Dr. Arun Kumar; Email: arunku@civil.iitd.ac.in) Set 1 Do not submit. Please mention your assumptions explicitly. Q1. Say a raw
Fee versus royalty licensing in a Cournot competition model: An extension to the case of J firms
 Care, Micro 1 Fee versus royalty licensing in a Cournot competition model: An extension to the case of J firms 1. Introduction When looking at situations involving the Cournot duopoly, we generally structure
Care, Micro 1 Fee versus royalty licensing in a Cournot competition model: An extension to the case of J firms 1. Introduction When looking at situations involving the Cournot duopoly, we generally structure
الجامعة التكنولوجية قسم الهندسة الكيمياوية هندسة التاكل بشير ا حمد
 الجامعة التكنولوجية قسم الهندسة الكيمياوية الرابعة المرحلة هندسة التاكل م. بشير ا حمد Save from: http://www.uotechnology.edu.iq/dep-chem-eng/index.htm TYPES OF CORROSION CELLS There are several types of
الجامعة التكنولوجية قسم الهندسة الكيمياوية الرابعة المرحلة هندسة التاكل م. بشير ا حمد Save from: http://www.uotechnology.edu.iq/dep-chem-eng/index.htm TYPES OF CORROSION CELLS There are several types of
1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is.. The precipitate which forms first is
 CHEMISTRY 12 UNIT 3 - REVIEW Ksp Part A - Multiple Choice 1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is A. Fe 2 (SO 4 ) 3 (s) 3 Fe 2+ (aq) + 2 SO 4 3 (aq)
CHEMISTRY 12 UNIT 3 - REVIEW Ksp Part A - Multiple Choice 1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is A. Fe 2 (SO 4 ) 3 (s) 3 Fe 2+ (aq) + 2 SO 4 3 (aq)
OXIDATION-REDUCTION EXPERIMENT
 Chem 112 OXIDATION-REDUCTION EXPERIMENT INTRODUCTION An oxidation-reduction (redox) reaction involves the movement of electrons from one reactant to another. Many reactions that you have already studied
Chem 112 OXIDATION-REDUCTION EXPERIMENT INTRODUCTION An oxidation-reduction (redox) reaction involves the movement of electrons from one reactant to another. Many reactions that you have already studied
Biofixation of high-concentration carbon dioxide using a. deep-sea bacterium: Sulfurovum lithotrophicum 42BKT T
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supplementary information Biofixation of high-concentration carbon dioxide using a deep-sea
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supplementary information Biofixation of high-concentration carbon dioxide using a deep-sea
Example: Compute the wavelength of a 1 [kg] block moving at 1000 [m/s].
![Example: Compute the wavelength of a 1 [kg] block moving at 1000 [m/s]. Example: Compute the wavelength of a 1 [kg] block moving at 1000 [m/s].](/thumbs/95/123245062.jpg) Example: Calculate the energy required to excite the hydrogen electron from level n = 1 to level n = 2. Also calculate the wavelength of light that must be absorbed by a hydrogen atom in its ground state
Example: Calculate the energy required to excite the hydrogen electron from level n = 1 to level n = 2. Also calculate the wavelength of light that must be absorbed by a hydrogen atom in its ground state
Chapter 5: Extent of Reaction
 Chapter 5: Extent of Reaction Accumulation = input - output + generation - consumption Steady state system: 0 = input - output + generation - consumption output = input + generation - consumption Input
Chapter 5: Extent of Reaction Accumulation = input - output + generation - consumption Steady state system: 0 = input - output + generation - consumption output = input + generation - consumption Input
