Distinct roles for Fgf, Wnt and retinoic acid in posteriorizing the neural ectoderm
|
|
|
- Horatio Parks
- 6 years ago
- Views:
Transcription
1 Development 129, (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV Distinct roles for Fgf, Wnt and retinoic acid in posteriorizing the neural ectoderm Tetsuhiro Kudoh 1,2, *, Stephen W. Wilson 1 and Igor B. Dawid 2 1 Department of Anatomy and Developmental Biology, University College London, Gower St, London WC1E 6BT, UK 2 Laboratory of Molecular Genetics, National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, MD, USA *Author for correspondence ( t.kudoh@ucl.ac.uk) Accepted 25 June 2002 SUMMARY Early neural patterning in vertebrates involves signals that inhibit anterior (A) and promote posterior (P) positional values within the nascent neural plate. In this study, we have investigated the contributions of, and interactions between, retinoic acid (RA), Fgf and Wnt signals in the promotion of posterior fates in the ectoderm. We analyze expression and function of cyp26/p450rai, a gene that encodes retinoic acid 4-hydroxylase, as a tool for investigating these events. Cyp26 is first expressed in the presumptive anterior neural ectoderm and the blastoderm margin at the late blastula. When the posterior neural gene hoxb1b is expressed during gastrulation, it shows a strikingly complementary pattern to cyp26. Using these two genes, as well as otx2 and meis3 as anterior and posterior markers, we show that Fgf and Wnt signals suppress expression of anterior genes, including cyp26. Overexpression of cyp26 suppresses posterior genes, suggesting that the anterior expression of cyp26 is important for restricting the expression of posterior genes. Consistent with this, knock-down of cyp26 by morpholino oligonucleotides leads to the anterior expansion of posterior genes. We further show that Fgf- and Wntdependent activation of posterior genes is mediated by RA, whereas suppression of anterior genes does not depend on RA signaling. Fgf and Wnt signals suppress cyp26 expression, while Cyp26 suppresses the RA signal. Thus, cyp26 has an important role in linking the Fgf, Wnt and RA signals to regulate AP patterning of the neural ectoderm in the late blastula to gastrula embryo in zebrafish. Key words: Fgf, Wnt, Retinoic acid, Posteriorization, Cyp26, Raldh2, Zebrafish INTRODUCTION Studies of axis formation in Xenopus laevis have led to models in which two signaling events that are initiated by localized maternal determinants cause mesendoderm induction in the marginal zone, and organizer formation on its dorsal side at blastula-to-gastrula stages. Mesendoderm induction divides the embryo into three germ layers along the vegetal-to-animal axis, while signals from the organizer induce neural tissue at the dorsal side of the ectoderm. The neural ectoderm is patterned along its anteroposterior (AP) axis, coincident with, and subsequent to, its initial specification; this patterning is initiated by posteriorizing signals derived from prospective or definitive mesendodermal tissues (Nieuwkoop, 1950; Toivonen, 1968). Three candidate posteriorizing signals have been suggested: retinoic acid (RA) (Blumberg et al., 1997; Conlon, 1995; Durston et al., 1989; Sive et al., 1990); fibroblast growth factors (Fgfs) (Cox and Hemmati-Brivanlou, 1995; Kengaku and Okamoto, 1993; Kengaku and Okamoto, 1995; Koshida et al., 1998; Lamb and Harland, 1995) and Wnts (Fekany-Lee et al., 2000; Kazanskaya et al., 2000; Kelly et al., 1995; Kiecker and Niehrs, 2001; McGrew et al., 1995; Yamaguchi, 2001). Although it is likely that factors in all three families participate in this process, the precise role of each in the temporal and spatial aspects of neural patterning as well as the molecular consequences of their action have not been fully clarified. In zebrafish, a region called the yolk syncytial layer (YSL), which is located beneath the blastoderm, may have a role in both mesendoderm and organizer induction (Mizuno et al., 1996). As a consequence of inductive signals that emanate from the YSL, the blastoderm margin forms the mesendoderm, one side of which develops into the organizer (which, in turn, induces neural specification within the dorsal ectoderm). As in Xenopus, there is evidence for signals emanating from the prospective mesendodermal layer that transform early neural ectoderm from an anterior to a posterior fate (Koshida et al., 1998). One feature that adds complexity to the mechanisms of AP patterning is the repeated use of the same type of signal in different stages and regions of the embryo, with contextdependent consequences. Fgfs and Wnts are both expressed in undifferentiated mesendoderm from the blastula stage onwards, and in the area of the presumptive midbrain-
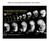
2 4336 T. Kudoh, S. W. Wilson and I. B. Dawid hindbrain region, beginning at the late gastrula stage (Furthauer et al., 1997; Kelly et al., 1995; Phillips et al., 2001). Signals mediated by members of these two major classes of secreted factors are involved in early AP patterning in the neural ectoderm, as well as subsequent regional patterning processes within the developing brain (Houart et al., 2002; Kim et al., 2000; Reifers et al., 1998). To avoid having to consider a large range of these complexities, we have focused on the earliest manifestation of AP specification that is evident from the late blastula through gastrula stages. We have searched for genes that may have an early role in axis formation as part of a random in situ screen for regionally expressed genes in zebrafish embryos (Kudoh et al., 2001). In this screen, we noted the anterior neural ectodermal expression of the gene cyp26/p450rai, which encodes all trans retinoic acid 4-hydroxylase, an enzyme that degrades and inactivates RA. Zebrafish cyp26 has originally been cloned from regenerating fin tissue as an RA-responsive gene (White et al., 1996). We find that cyp26 is specifically expressed in the presumptive anterior neural ectoderm from a surprisingly early stage (from the late blastula onwards). At the early gastrula stage, the earliest known marker of posterior neural ectoderm, hoxb1b (Alexandre et al., 1996), is expressed in a complementary pattern to cyp26. We focused on the regulation of this earliest subdivision along the AP axis in the neural ectoderm, using cyp26 and hoxb1b as our primary tools of analysis. We show that posteriorization of the neural ectoderm has two separable steps: suppression of anterior gene expression and activation of posterior gene expression. Suppression of anterior gene expression in the posterior region is the first step of AP differentiation and is caused by Fgfs/Wnts in an RA-independent pathway. The activation of posterior gene expression is the second step, and this event is RA dependent. These two posteriorizing steps are linked through the regulation and function of the cyp26 gene. MATERIALS AND METHODS Constructs, mrna synthesis and embryo injections The coding region in cyp26 was amplified by PCR and subcloned into the pcs2+ expression vector. Cyp26-GFP fusion construct was made using PCR primers forward (F; ccggatcctcgaggtcgacccacgcg) and reverse (R; ccgaattcggtgtcagagcccaggatgg), which amplifies 82 bp of 5 non-coding region with 375 bp of coding region. This PCR product was inserted to pcs2+ and subsequently EGFP cdna was inserted into the 3 end of cyp26 cdna. mrna was synthesized by mmessage mmachine (Ambion). The following mrnas were injected into zebrafish embryos: cyp26 (500pg), cyp26-egfp (300pg), fgf3 (50pg) (Koshida et al., 2002), XFD (500pg) (Amaya et al., 1991) and dkk1 (50pg) (Hashimoto et al., 2000). Retinoic acid (RA) and LiCl treatments RA was stored at 10 3 M in ethanol and diluted to 10 6 M in fish water before use. Embryos were treated with RA from the 40% epiboly stage onward for 80 minutes, followed by thorough washing. For LiCl treatment, 50% epiboly stage embryos were exposed to 300 mm LiCl for about 8 minutes and washed immediately with fish water several times. These embryos were fixed at late gastrula stage and stained by in situ hybridization. Injection of morpholino oligonucleotides A sequence complementary to the region of cyp26 cdna around the start codon was used to synthesize a morpholino antisense oligonucleotide, mcyp1, by Gene Tools (Philomath, USA). The sequence of mcyp1 is 5 -cgcaactgatcgccaaaacgaaaaa-3. Five nanograms of morpholino were injected at the one- to two-cell stage into the yolk. For comparison, the same amount of standard control morpholino (Gene Tools) was injected. Whole-mount in situ hybridization Embryos were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS) overnight at 4 C, manually dechorionated and stored in methanol up to several weeks. In situ hybridization was performed essentially as described previously (Kudoh and Dawid, 2001). Probes for the following genes were used: cyp26, iro1, meis3, hoxb1b, otx2 and ntl. These probes were obtained from our in situ-based screen (Kudoh et al., 2001). For injected and drug-treated embryos, 15 to 30 embryos were used in each experiment. RESULTS Early expression pattern of cyp26 cyp26 is first expressed at the late blastula stage in two distinct regions, at the animal side of the dorsal ectoderm (Fig. 1A,A, closed arrowhead) and at the blastoderm margin (Fig. 1A,A, open arrowhead). These two domains continue to express cyp26 up to the bud stage at the end of gastrulation (Fig. 1A- D). At later gastrula and somitogenesis stages, ntl expression also marks the notochord (Fig. 1C,F). cyp26 expression in the anterior neural ectoderm decreases during gastrulation so that only weak expression in a region surrounding the neural plate is seen by the three-somite stage (Fig. 1D-F). By contrast, cyp26 expression in the blastoderm margin persists as this domain extends towards the posterior, contributing to the tail. This expression pattern suggests that cyp26 is important for establishing the anterior character of neural ectoderm during gastrula stages, as well as for the formation of the tail during later development. Complementary expression of cyp26 and hoxb1b, an early marker of posterior neural ectoderm The expression of cyp26 was compared with the posterior neural ectoderm expression of hoxb1b (originally named hoxa1) (Alexandre et al., 1996). At the late blastula stage, there is a small gap between the animal and marginal domain of cyp26 expression (Fig. 2A, asterisk); this gap is expanded subsequently through convergence-extension movements during gastrulation (Fig. 2B-E). At about the 60% epiboly stage, hoxb1b expression is initiated within this gap. When cyp26 and hoxb1b are doubly stained by single-color in situ hybridization, they show complementary expression patterns from the onset of hoxb1b expression to the end of gastrulation (Fig. 2K-M); a narrow gap persists between the cyp26 and hoxb1b domains, emphasizing the exclusiveness of these domains. In these double-labeled embryos, cells completely double negative for cyp26/hoxb1b are not observed at high magnification, suggesting that this gap is not a distinct domain but represents the border of the anterior and posterior domains where the expression of the two markers decreases. This complementarity suggests that initial AP patterning in the neural ectoderm is reflected by the anterior expression of cyp26 in the late blastula, while the expression of hoxb1b, the earliest posterior marker, constitutes a subsequent step in the evolving process.
3 Fgf, Wnt and retinoic acid in posteriorization 4337 mesoderm in a pattern complementary to cyp26 along the AP axis, with a small but widening gap between the two expression domains (Fig. 2N,O). The complementarity of RA synthesis (raldh2) and degradation (cyp26) domains supports the idea that refinement of RA-positive and -negative domains of the ectoderm is achieved by the action of these opposing gene products (Chen et al., 2001; Swindell et al., 1999). The expression of cyp26 and of other AP-marker genes is regulated by Fgf signals As cyp26 exhibits extremely early anterior neuroectodermal expression, we tested the regulation of its expression by Fgf, a candidate posteriorizing factor; the behavior of cyp26 was compared with another anterior neural gene, otx2, and to the posterior gene hoxb1b. To generate conditions that correspond to both gain and reduction of function of Fgf signaling, fgf3 and dominant-negative Fgf-receptor (XFD) (Amaya et al., 1991) mrnas were injected into one- to two-cell stage embryos. fgf3 mrna injection led to suppression of anterior expression of cyp26 and otx2 (Fig. 3B,J), whereas expression of the posterior gene hoxb1b was expanded anteriorly (Fig. 3F). XFD mrna injected embryos showed the opposite phenotype in that cyp26 and otx2 domains were expanded posteriorly (Fig. 3D,L) (Koshida et al., 1998) and hoxb1b was suppressed (Fig. 3H arrowhead). These results indicate that Fgf signaling is necessary and sufficient for the suppression of the anterior genes cyp26 and otx2, as well as for the activation of the posterior gene hoxb1b during gastrulation. Fig. 1. Expression of cyp26 at gastrula to early somitogenesis stages. Views of whole embryos (orientation indicated to the left of each row) at the stages indicated on top of each column. cyp26 was stained by in situ hybridization (purple), while ntl (red) was used to mark the blastoderm margin (A, A, open arrowhead) and developing notochord (C,F, asterisk). cyp26 is expressed in the presumptive anterior neural ectoderm (A,A filled arrowhead) and at the blastoderm margin (A,A open arrowhead) throughout gastrulation (B-D). Subsequently, expression in the anterior neural ectoderm decreases rapidly (E, F filled arrowhead), while expression continues in the tail bud. To further resolve and confirm expression domains, cyp26 was also analyzed in combination with iro1, krox20 and raldh2. iro1 is expressed as two pairs of bands at late gastrula stage; the anterior bands contain the presumptive midbrain-hindbrain boundary and possibly rhombomere 1 (Itoh et al., 2002). The anterior domain of iro1 and the cyp26 expression domain share a common posterior border of expression, suggesting that at this stage, the posterior end of cyp26 expression is around prospective rhombomere 1 (Fig. 2P). Double staining of cyp26 with krox20, which marks rhombomeres 3 and 5 at bud stage, revealed a gap between the posterior edge of cyp26 expression and rhombomere 3 (Fig. 2Q). This gap presumably represents the future rhombomere 2. raldh2, which encodes a major RA synthesis enzyme, is expressed in the posterior paraxial RA modulates the expression of AP-marker genes RA has been shown to affect the specification of AP values in the developing nervous system of different vertebrates and is a candidate posteriorizing signal (Blumberg et al., 1997; Conlon, 1995; Durston et al., 1989; Niederreither et al., 1999; Sive et al., 1990). RA is also the substrate of the cyp26 gene product. To examine the role of RA in early AP pattern specification in the neural ectoderm, we examined the expression of the anterior genes cyp26 and otx2 and the posterior genes hoxb1b and meis3 in RA-treated embryos. Consistent with the designation of RA as a posteriorizing factor, the expression of hoxb1b and meis3 was expanded (Fig. 4D,F) and that of otx2 was suppressed (Fig. 4H). However, cyp26 expression was expanded in RA-treated embryos, in spite of the anterior character of its ectodermal expression domain (Fig. 4B). This outcome is most likely to be due to the fact that cyp26 is a direct target of RA and acts as an RA-responsive gene in different stages of embryogenesis such as fin formation (Loudig et al., 2000; White et al., 1996). As it encodes an RAmetabolizing enzyme, cyp26 may be part of a negative feedback loop that limits RA signaling in different biological contexts. Overexpression of cyp26 suppresses posterior neural genes To analyze the function of cyp26 and its substrate, RA, in early AP patterning, cyp26 mrna was injected into one blastomere of two-cell embryos, and the expression of AP marker genes was examined during gastrulation (Fig. 5). Consistent with the activity of its gene product as an RA-degrading enzyme, cyp26 injection showed the opposite effect to application of RA on the expression of the posterior genes hoxb1b and meis3, both
4 4338 T. Kudoh, S. W. Wilson and I. B. Dawid Fig. 2. cyp26 and hoxb1b expression domains define an early AP boundary within the prospective neuroectoderm. Dorsal views of whole embryos at the stages indicated on top of. Genes analyzed are indicated at the left of the column or in the figure, with text color matching the in situ stain. cyp26 expression starts at the 30 to 40% epiboly stage (A) in presumptive anterior neural ectoderm (filled arrowhead) and at the blastoderm margin (open arrowhead), leaving a narrow gap (asterisk). As gastrulation proceeds, hoxb1b expression is initiated within the gap at about the 60% epiboly stage (C,G,K). From this time onwards, cyp26 and hoxb1b are expressed in a complementary manner in the neural ectoderm throughout gastrulation; a narrow domain of reduced expression is observed at the interface between the expression domains of these two genes (K-M, arrow). (N-Q) cyp26 expression (red) in relation to expression domains of other APspecific genes (purple), raldh2 (N,O), iro1 (P) and krox20 (Q). There is a narrow gap between cyp26 and raldh2 expression domains at early gastrula (N, arrow), and this gap becomes wider by late gastrula (O, arrow). At the end of gastrulation, cyp26 and the anterior domain of iro1 expression share a common posterior boundary (P, arrow). There is a gap between cyp26 and rhombomere3 staining of krox20 (Q, arrow); thus the posterior boundary of cyp26 at this stage appears to be positioned around rhombomere 1. Fig. 3. Fgf signaling alters the expression of early AP-specific genes. Dorsal views of whole embryos at late gastrula stage. One- to two-cell embryos were injected with mrnas for fgf3 (B,F,J) or RNA encoding the dominant negative Fgf receptor, XFD (D,H,L). Embryos were stained either with cyp26 (A-D), hoxb1b (E-H) or otx2 (I-L). The anterior expression of cyp26 and otx2 was suppressed by Fgf3 (B,J) and expanded in a posterior direction by XFD (D,L). Expression of the posterior gene hoxb1b was expanded by Fgf3 (F) and suppressed by XFD (H, arrowhead). The entire embryo is affected by injection at the one-cell stage (B), while in some cases only half the embryo is affected when one of two cells is injected (H). of which were suppressed (Fig. 5B,D). We tested an additional gene, iro1, which has two expression domains, an anterior domain that overlaps with cyp26 and a posterior domain that overlaps with hoxb1b (Fig. 2P). Consistent with the results above, only the posterior domain of iro1 was suppressed by cyp26 (Fig. 5F, arrowhead). By contrast, expression of the anterior gene otx2 was not affected by cyp26 (Fig. 5H). To confirm the differential effect of cyp26 on anterior and posterior neural markers, control and injected embryos were double stained with otx2 and hoxb1b, showing that hoxb1b expression was decreased, whereas that of otx2 was neither suppressed nor expanded (Fig. 5J). However, injection of cyp26 could at least partially restore otx2 expression in the presence of RA (Fig. 5L). The effects of cyp26 on gene expression illustrated in Fig. 5 for mid-to-late gastrula stages were also seen at early gastrulation (data not shown). These results suggest that RA is necessary for the induction of posterior gene expression, but not for the suppression of anterior gene expression. This lack of an effect of cyp26 in promoting anterior gene expression in the posterior domain represents a difference between RA and Fgf in their action as posteriorizing agents (compare Fig. 3L with Fig. 5H,J). Distinct roles of Fgf and RA in patterning the neural ectoderm To investigate the relationship of the
5 Fgf, Wnt and retinoic acid in posteriorization 4339 posteriorizing signals delivered by Fgf and RA, we applied agonists and antagonists of these two signals to the same embryos. When fgf3 and cyp26 mrnas were co-injected, fgf3- mediated activation of hoxb1b expression was strongly suppressed (Fig. 6B,C). This result suggests that Fgf3- dependent induction of hoxb1b is mediated by RA. By contrast, Fgf3-mediated suppression of otx2 expression was not affected by co-injection of cyp26 (Fig. 6E,F). Thus, the effect of Fgf in suppressing otx2 does not require RA, and suppression of the RA signal is not sufficient for the induction of otx2. In complementary experiments, embryos injected with XFD mrna were treated with RA. XFD blocks Fgf signaling, and consequently inhibits hoxb1b expression (Fig. 6H). Consistent with the results above, this XFD-induced suppression of hoxb1b was overcome by the addition of RA (Fig. 6I). Furthermore, XFD led to an expansion of otx2 towards the posterior (Fig. 6K), and both the expanded and normal expression of otx2 were suppressed in the presence of RA (Fig. 6L). Thus, RA can repress otx2 expression in either the anterior or posterior compartment, but reduction of RA alone is not sufficient to induce ectopic otx2 expression (Fig. 5H, Fig. 6F). These results support the view that RA acts downstream of Fgf in inducing posterior genes in the neural ectoderm. Distinct roles of Fgfs and Wnts in patterning the neural ectoderm Next, we examined the involvement of Wnt signaling in the regulation of expression of these early marker genes. LiCl inhibits the kinase activity of GSK3 and consequently activates β-catenin, a downstream effector of the canonical Wnt pathway (Klein and Melton, 1996). Activation of the Wnt pathway in the early gastrula by treatment with LiCl at the 50% epiboly stage led to suppression of the anterior markers, cyp26 and otx2(fig. 7A,B,F,G). However, the posterior marker hoxb1b was not induced in the anterior-most area of the embryo, although some anterior expansion was observed (Fig. 7K,L). To reduce canonical Wnt signaling, dkk1 mrna was injected. Dkk1 slightly expanded otx2 and cyp26 expression, and reduced that of hoxb1b, but there was always residual posterior character near the margin (Fig. 7C,M). In order to elucidate the relationship between Wnt and Fgf signals in regulating AP polarity, we concomitantly altered activity of both pathways. XFD injected embryos were Fig. 4. RA alters the expression of early AP-specific genes. Dorsal views of whole embryos at late gastrula stage. Embryos at the 40% epiboly stage were treated with 10 6 M RA for 80 minutes. The embryos were fixed at the 80 to 90% epiboly stage, and stained with cyp26 (A,B), hoxb1b (C,D), meis3 (E,F) or otx2 (G,H). The posterior genes hoxb1b and meis3 were ectopically induced in the anterior region by RA (B,D), while the anterior gene otx2 was suppressed (H). Although cyp26 is expressed in the anterior region, its expression was activated by RA(B). Fig. 5. Cyp26 can suppress posterior genes but does not induce anterior genes in the posterior region. Dorsal views of whole embryos at late gastrula stage. cyp26 mrna was injected at the two-cell stage into one blastomere. Some of the embryos were treated with RA (K,L). Embryos were stained for hoxb1b (A,B), meis3 (C,D), iro1 (E,F), otx2 (G,H,K,L) and otx2/hoxb1b together (I,J). hoxb1b, meis3 and the posterior domain of iro1 were suppressed by cyp26 injection (B,D,F,J, arrowhead), but otx2 and the anterior domain of iro1 expression were neither suppressed nor expanded (F,H,J). The suppression of otx2 by RA was partially rescued by cyp26 (L, arrowhead).
6 4340 T. Kudoh, S. W. Wilson and I. B. Dawid treated with LiCl to suppress Fgf activity, while enhancing Wnt signaling. In these embryos, cyp26 and otx2 as well as hoxb1b were suppressed (Fig. 7D,I,N). This suggests that activation of Wnt signaling can suppress anterior markers in the absence of Fgf signaling but that activation of posterior markers requires Fgf. As a complementary experiment, fgf3 and dkk1 were coexpressed to enhance Fgf while suppressing Wnt signaling. In these embryos, cyp26 and otx2 were suppressed, indicating that Dkk1 is not sufficient to promote anterior markers in the presence of exogenous Fgf signaling (Fig. 7E,J). Dkk1 did, however, inhibit the ability of Fgf to expand hoxb1b expression into anterior regions (Fig. 7O). Fig. 6. Epistatic analysis of the function of Fgf and RA in patterning the neural ectoderm. Dorsal views of whole embryos at late gastrula stage. RNA for fgf3, alone or together with cyp26 RNA, was injected at the one-cell stage. The embryos were stained for hoxb1b (A-C) or otx2 (D-F). XFD mrna was injected at the one-to-two cell stage, and half of the embryos were treated with RA. Embryos were stained for hoxb1b (G-I) or otx2 (J-L). Distinct roles of RA and Wnt in patterning the neural ectoderm In the next set of experiments, we examined the relationship between the RA and Wnt signaling pathways. RA activity was suppressed by overexpression of cyp26, and subsequently LiCl immersion at 50% epiboly stage was used to activate Wnt signaling. In these embryos both otx2 and hoxb1b were suppressed, indicating that Wnt signals inhibit anterior development in the absence of RA (Fig. 8B,E). As a complementary experiment, dkk1 mrna-injected embryos were treated with RA. In these embryos, otx2 was largely suppressed and hoxb1b was ectopically expressed up to the animal pole (Fig. 8C,F). Although there is otx2 expression remaining in the dorsal-most area (Fig. 8C, arrowhead), because these cells are hypoblast they are possibly the expanded axial mesoderm. These results suggest that Wnts and RA both can suppress otx2 in the anterior neural ectoderm but that activation of hoxb1b depends on RA. One possibility is that Wnts promote hoxb1b expression through regulating the production of RA. To assess if this might be the case, expression of the gene encoding an RA synthesis enzyme, raldh2, was examined in conditions in Fig. 7. Epistatic analysis of the function of Wnt and Fgf in patterning the neural ectoderm. Dorsal views of whole embryos at late gastrula stage. Gain and loss of function of Wnt activity was achieved by LiCl treatment (B,G,L) and dkk1 injection (C,H,M), respectively. Epistasis with Fgf was examined by XFD injection followed by LiCl treatment (D,I,N) and by fgf3 +dkk1 co-injection (E,J,O). Embryos were fixed at late gastrula and stained with cyp26 (A-E), otx2 (F-J) and hoxb1b (K- O). Arrowheads in C and M indicate the limit of caudal expansion of cyp26 expression (C) and remaining hoxb1b expression (M). In some cases, only half the embryo is affected when one of two cells is injected (N, arrowhead).
7 Fgf, Wnt and retinoic acid in posteriorization 4341 Fig. 8. Epistatic analysis of the function of Wnt and RA in patterning the neural ectoderm. Dorsal views of whole embryos at late gastrula stage (A-F,I,J), and animal views of 55% epiboly stage embryos (G,H). cyp26-injected embryos were treated with LiCl at the 50% epiboly stage (B,E), and dkk1-injected embryos were treated with RA at 40% epiboly stage (C,F). Embryos were fixed at late gastrula and stained with otx2 (A-C) and hoxb1b (D-F). Regulation of the expression of raldh2 was examined in dkk1-injected embryos (H,J). At 55% epiboly stage, raldh2 expression is not suppressed at the blastoderm margin by dkk1 injection though the raldh2 negative area in the dorsal-most margin is slightly expanded (H). At late gastrula stage, raldh2 expression remains restricted to the blastoderm margin in dkk1-injected embryos (J, arrowhead). which Wnt signaling was increased or decreased. At the 50% epiboly stage, raldh2 is expressed in nascent paraxial mesoderm at the blastoderm margin. In embryos in which the Wnt antagonist dkk1 was injected, raldh2 expression was not suppressed although the dorsal raldh2-negative domain was slightly expanded, most probably because Dkk1 activity is known to enhance axial mesodermal fates (Hashimoto et al., 2000). This suggests that Wnt signaling is not necessary for the initiation of raldh2 expression. However, by late gastrula stage, raldh2 expression remained restricted to the blastoderm margin in dkk1-injected embryos, whereas in control embryos, raldh2 was highly expressed in the involuted mesoderm as it migrated anteriorly (Fig. 8I,J). This limitation of raldh2 expression could account for the similar restriction of hoxb1b expression to the marginal zone of dkk1-injected embryos. raldh2 expression was also examined in LiCl-treated embryos and shown to retain normal expression (data not shown). Together, these results suggest that Wnt signals indirectly influence hoxb1b expression through the modulation of RA activity. Cyp26 morpholino, mcyp1, causes partial posteriorization To examine the physiological role of cyp26 in early AP patterning, morpholino antisense oligonucleotide (mcyp1) was injected into zebrafish embryos. In mcyp1-injected embryos, otx2 expression was decreased (Fig. 9B) and hoxb1b expression was expanded anteriorly (Fig. 9D). Complementing this, the expression domains of meis3 and iro3 were both shifted in an anterior direction (Fig. 9F,H). These results suggest that the activity of endogenous Cyp26 is required to restrict the expression of posterior genes at their anterior border and to protect anterior genes from repression by RA. Cyp26 is likely to carry out these functions by degrading RA molecules that encroach into the anterior ectodermal compartment, thereby limiting their presence to the posterior compartment. The efficacy of mcyp1 was confirmed in experiments using a cyp26-gfp fusion construct. GFP fluorescence was strongly suppressed by the co-injection of mcyp1 but not by the control morpholino (Fig. 9I,J); thus mcyp1 at this concentration efficiently suppressed the translation of Cyp26 fusion protein. In addition to the changes in expression of AP marker genes, morpholino injected embryos were more ovoid in shape by late gastrulation (Fig. 9B,D,F,H). A similar shape change was observed in RA-treated embryos at the end of gastrulation (not shown) supporting the notion that endogenous RA activity is increased by the mcyp1. Summary of experiments The consequences of altering Fgf, RA and Wnt signals upon early AP patterning of the ectoderm are summarized as a cartoon in Fig. 10. In general, all anterior and all posterior markers behaved similarly in each set of experiments, with the exception of the cyp26 gene, which was induced by RA even though it is normally expressed in the anterior region of the neural ectoderm (see Discussion). In the cartoon, we summarize the results obtained from analysis of the expression of other markers, primarily otx2 and hoxb1b. From this, it is evident that anterior gene and posterior gene expression domains never overlap. However, although in some cases, the entire embryo acquired anterior or posterior identity (i.e. fgf3 or XFD injection), in other cases, anterior and posterior genes retained complementarity of expression but with a shifted boundary (i.e. dkk1 injection). Finally, in some situations we observed that anterior or posterior character could be specifically lost in one region (i.e. after cyp26 injection or LiCl treatment) or throughout the entire embryo (i.e. after fgf3+cyp26 injection or cyp26 injection plus LiCl treatment). The latter case is particularly instructive: excessive signaling by either Fgf or Wnt eliminates anterior character, but establishing posterior character depends on RA. Thus, the combination of ectopic Fgf or Wnt signals and suppression of RA leaves the presumptive neuroectoderm without any AP identity. DISCUSSION cyp26 modulates the action of RA in AP patterning of the neural ectoderm Our study has revealed that AP regionalization of the prospective neural ectoderm occurs very early and that the cyp26 gene appears to be involved in this early patterning. We
8 4342 T. Kudoh, S. W. Wilson and I. B. Dawid Fig. 9. Abrogation of Cyp26 activity causes moderate posteriorization. Dorsal views of whole embryos at late gastrula stage (A-H) and lateral views of late blastula stage embryos (I,J). A control morpholino or mcyp1, which is complementary to cyp26 mrna sequence, was injected at 5 ng per embryo. Embryos were stained with otx2 (A,B), hoxb1b (C,D), meis3 (E,F) and iro1 (G,H). In addition, cyp26-gfp fusion construct was injected with either morpholino, and fluorescence was examined. (I,J). (A,C,E,G,I) Control morpholino; (B,D,F,H,J) mcyp1-injected embryos. have analyzed the expression pattern of zebrafish cyp26 and found that its dorsoanterior expression begins around 30-40% epiboly in the late blastula. This observation is of interest as it suggests that AP patterning of the neural ectoderm is initiated well before the onset of gastrulation. Expression in the anterior neural domain persists through gastrulation but disappears at early somitogenesis stages, suggesting that cyp26 affects AP patterning mainly from the blastula through the gastrula stages. cyp26 has been identified in Xenopus and in the mouse, and is also expressed in the anterior neural ectoderm in these species (de Roos et al., 1999; Fujii et al., 1997; Hollemann et al., 1998). As Cyp26 degrades RA it is expected to limit the range of RA-mediated posteriorization in the embryo. Our Cyp26 overexpression and loss-of-function studies support this idea. RA is known to be involved in the expression of posterior genes including Hox genes (Kolm et al., 1997; Kolm and Sive, 1995; Niederreither et al., 1999; Simeone et al., 1990). In our experiments, RA induced posterior genes such as hoxb1b and Fig. 10. Summary of experiments. The experimental results are summarized as a cartoon. otx2 and hob1b are used as representative markers of anterior and posterior neural ectoderm, respectively, as these genes were examined in all of experiments. The top picture is a dorsal view of a late gastrula embryo divided into anterior (purple) and posterior (red) neural ectodermal regions and undifferentiated mesendoderm at the blastoderm margin (green). One half of the dorsal ectoderm is surrounded by yellow line and experimental results are summarized in this area as marker genes for mesoderm and axial area were not analyzed in any detail in this study. meis3, while cyp26 injection suppressed expression of these genes, presumably by reducing the endogenous RA concentration. However, the expression domain of the anterior gene otx2 was not expanded by cyp26 mrna injection, suggesting that RA-independent pathways suppress otx2 in the posterior neural ectoderm. We believe that limiting the range of RA action is the in vivo function of Cyp26 as morpholino antisense oligonucleotide injection caused anterior expansion of posterior genes such as hoxb1b, and a shrinking of the expression domain of the anterior gene, otx2. Cyp26 thus has an important role in defining the border between anterior and posterior neural ectoderm. cyp26 is also expressed at the blastoderm margin with a gap
9 Fgf, Wnt and retinoic acid in posteriorization 4343 between its two domains of expression. The blastoderm margin expression persists into the developing tail region during somitogenesis, suggesting that suppression of RA signals is also required in tail development. Consistent with this view, RA treatment in Xenopus and zebrafish causes truncation of tail as well as head structures (Durston et al., 1989) (data not shown). Recently, mice carrying targeted mutations in cyp26 were generated and these animals showed loss of tail and adjacent posterior structures, stressing the importance of RA signal suppression by cyp26 for tail development (Abu-Abed et al., 2001; Sakai et al., 2001). Overexpression experiments using cyp26 in Xenopus showed otx2 expression was slightly expanded in a posterior direction (Hollemann et al., 1998). This is somewhat different from our observation that otx2 was not expanded by cyp26 overexpression. The differences may be due to the fact that phenotypes in frog were examined at later stages, when many other CNS patterning events are likely to have occurred and modified early effects of exogenous Cyp26 activity. Our observation that RA caused the expansion of hoxb1b expression in anterior neural ectoderm differs from results reported previously by Alexandre et al., who described more subtle phenotypes following RA treatment (Alexandre et al., 1996). This difference can be explained by the different concentration of RA in the two studies. When we applied 10 7 M RA to zebrafish embryos, hoxb1b was induced only in axial mesoderm, as reported by Alexandre et al. (Alexandre et al., 1996). However we used 10 6 M RA because this or higher doses generate anterior truncations in Xenopus (Durston et al., 1989; Sive et al., 1990). Furthermore, the internal concentration of RA under these conditions is not known. Recently, the zebrafish mutants neckless and nofin, which have a decrease in hindbrain size, have been shown to carry mutations in the RA synthesis enzyme, Raldh2 (Begemann et al., 2001; Grandel et al., 2002). Begemann et al. have shown that the mutant phenotype in the hindbrain could be rescued by M or 10 6 M RA, but not by lower concentrations. This suggests that when applied externally, 10 6 M RA may mimic physiological conditions that are achieved by normal RA synthesis in the wild-type embryo (Begemann et al., 2001). Fgf and Wnt signals regulate early AP polarity With the aim of investigating the nature of the signals that initiate differential expression of cyp26 and hoxb1b, we examined the role of Fgfs in this process, as Fgfs have been implicated in posteriorization of the neural ectoderm (Griffin et al., 1995; Koshida et al., 1998). Experiments that involved injection of mrnas for Fgf3 and the truncated receptor XFD into zebrafish embryos showed that Fgf signaling is both necessary and sufficient, directly or indirectly, for the suppression of the anterior genes cyp26 and otx2 and the induction of the posterior gene hoxb1b. Like Fgf signaling, Wnt activity is known to affect the early specification of AP polarity in the neural ectoderm (Fekany- Lee et al., 2000; Kelly et al., 1995). We found that activation of Wnt signaling suppressed otx2 and cyp26, while the expression of hoxb1 was partly expanded by Wnt activation and largely suppressed by inhibition of Wnt signaling. Interactions between the posteriorizing pathways were studied by combined activation of one signal and inhibition of another, as summarized in Fig. 10. These experiments indicate that either an Fgf or a Wnt signal can suppress anterior genes such as cyp26 and otx2 even when the other pathway is inhibited. Therefore, Wnts and Fgfs can act independently of each other in the suppression of anterior genes; this suppression was also independent of the activity of RA, but RA can suppress anterior genes even in the presence of Fgf or Wnt pathway inhibitors. By contrast, activation of posterior gene expression could only be initiated by Fgf or Wnt signals in the presence of an intact RA signaling pathway. It is likely that RA acts directly on the hoxb1b promoter, as RA-responsive elements are present in the mouse Hoxb1 gene and have been shown to have a crucial role in regulating its expression (Marshall et al., 1994; Ogura et al., 1996a; Ogura et al., 1996b). The expansion of hoxb1b expression after ectopic Wnt activation may in part be explained by the suppression of cyp26, which results in expansion of the range of RA activity in an anterior direction. This interpretation is in agreement with the similar expansion of the hoxb1b domain after injection of a cyp26 morpholino antisense oligonucleotide. By contrast, the more extensive expansion of hoxb1b expression by Fgf overactivation is unlikely to simply be due to abrogation of Cyp26 activity. Instead we suggest that other factors must contribute to the ectopic production of an RA signal in anterior regions of Fgf injected embryos. A model for the interactions between Fgf, Wnt and RA in early AP patterning A model for the mechanism of early AP patterning, based on the observations described in this paper, is presented in Fig. 11. A key feature of this model is that promotion of posterior fates and suppression of anterior fates are treated as separable events. Fgfs and Wnts can suppress anterior genes in an RAindependent pathway. The set of genes sensitive to this suppression includes cyp26, which encodes an enzyme that degrades RA. While suppression of RA signaling is not necessary/sufficient for activation of anterior genes, we suggest that it is needed to prevent activation of posterior gene expression within the prospective anterior neural plate. The factors important for activation of anterior gene expression remain unknown, but many studies have suggested that anterior is a default fate for induced neural tissue (Nieuwkoop, 1950; Toivonen, 1968). Within prospective posterior neural tissue, RA is necessary and sufficient for the activation of at least some posterior genes. Furthermore, the ability of Fgfs and Wnts to promote expression of these posterior genes depends upon RA. In part, this is likely to be due to the ability of Fgfs and Wnts to promote RA activity through suppression of cyp26 expression, but is also likely to be due to additional regulatory events, such as promotion of raldh2 expression. In Fig. 11B, a spatial and temporal model underlying these AP patterning events is outlined. At the late blastula stage, genes in the Fgf and Wnt families are expressed in the prospective mesoderm at the blastoderm margin; fgf3, fgf8 and wnt8 are known to be expressed in this pattern (Furthauer et al., 1997; Kelly et al., 1995; Koshida et al., 2002; Phillips et al., 2001). We suggest that an early role for these marginderived signals is to suppress, directly or indirectly, the expression of cyp26 and possibly other anterior genes in dorsal ectoderm adjacent to the margin, thereby initiating the specification of this region as presumptive posterior neural ectoderm at the late blastula stage (Fig. 11B, part i). The initial
10 4344 T. Kudoh, S. W. Wilson and I. B. Dawid suppression of these genes is likely to be achieved through a planar signal because, at the 30-40% epiboly stage when localized cyp26 expression is first seen, the mesendodermal layer has not yet involuted below the ectodermal layer. This view is consistent with the report that posterior neural specification in dorsal ectoderm is observable at the shield stage (50-55% epiboly) but not in the blastula at the 30% epiboly stage (Grinblat et al., 1998). Our model further proposes that the widening Cyp26-free area allows the accumulation of RA and the consequent induction of RAdependent posterior genes, including hoxb1b, at early-to-mid gastrula stages (Fig. 11B, part ii). Subsequently, ongoing convergenceextension movements move lateral posterior ectoderm cells in a dorsal direction, causing further anteroposterior expansion of the Cyp26-negative, RA-positive area, thereby maintaining and enhancing the expression of hoxb1b and other posterior genes. Cyp26 maintains its expression anteriorly, thereby defining the rostral limit of expression of the posterior genes (Fig. 11Biii). The RA-synthesizing enzyme, Raldh2, is likely to be involved in these early patterning events. In several species, including zebrafish, Raldh2 is expressed in the posterior mesoderm (Begemann et al., 2001; Berggren et al., 1999; Chen et al., 2001; Grandel et al., 2002; Niederreither et al., 1997; Swindell et al., 1999). Raldh2 mutations in mouse and zebrafish reduce posterior neural ectoderm with concomitant downregulation of Hox genes (Begemann et al., 2001; Grandel et al., 2002; Niederreither et al., 1999). In a complementary manner, increased Raldh2 levels posteriorize the nervous system of Xenopus embryos (Chen et al., 2001). Therefore, two mechanisms might regulate RA accumulation, one mediated by the synthetic enzyme Raldh2, which augments RA in the posterior neural ectoderm, the other mediated by Cyp26, which degrades RA in the anterior neural ectoderm (Chen et al., 2001; Swindell et al., 1999). Mutations in the Cyp26 gene in mouse do indeed lead to a moderate anterior expansion of the Hoxa1 expression domain (Abu-Abed et al., 2001; Sakai et al., 2001), a result similar to that obtained by cyp26 morpholino injection in fish. As Raldh2 is expressed only in the posterior region, RA may not significantly accumulate in the anterior-most region, even in the absence of Cyp26, possibly explaining the relatively mild phenotypes after abrogation of Cyp26 activity in mouse and zebrafish. Therefore, although cyp26 is widely expressed in the anterior neural ectoderm, its major role may be the definition of the boundary in the presumptive hindbrain beyond which expression of posterior genes such as hoxb1b and meis3 does not expand. Fig. 11. A model for interactions between Fgf, Wnt and RA signaling in the neural ectoderm during gastrulation. (A) Sequence of posteriorization signals. Fgf and/or Wnt signals initiate the first step of posteriorization by suppressing expression of anterior genes, represented here by cyp26 and otx2. This process is not mediated by RA. In the posterior domain, where cyp26 expression is suppressed by Fgfs/Wnts, RA accumulates at least in part due to the activity of Raldh2, and activates posterior genes such as hoxb1b and meis3. (B) At the late blastula stage, Fgfs/Wnts are expressed in the mesoderm at the blastoderm margin. Fgf/Wnt signals from the margin block the expression of cyp26 in the adjacent ectoderm, which will give rise to the posterior neural plate. cyp26 is expressed in the anterior domain, at a distance from the source of the Fgf/Wnt signals; as a consequence, RA is degraded and the expression of posterior genes is prevented. After the beginning of gastrulation, convergence-extension movements lead to a widening of the cyp26-negative area, allowing RA to accumulate to a level where it can activate posterior genes such as hoxb1b. Subsequent cell movements expand the domain that will give rise to the posterior neural ectoderm. In summary, we show in the present paper that AP patterning is initiated in the presumptive neural ectoderm in the late blastula at 30-40% epiboly stage. We can distinguish two posteriorization steps in this process. The earliest step involves the Wnt/Fgfdependent, RA-independent suppression of anterior genes, including cyp26 in the presumptive posterior domain. The next step involves the activation of genes such as hoxb1b and meis3 in the posterior domain; this step is mediated by RA signaling. The antagonism between cyp26 activity and RA signaling links the initial and the subsequent steps of AP patterning, thereby contributing to the establishment of the earliest known border between anterior and posterior neural ectoderm. We thank Elizabeth Laver and the UCL Fish Facility personnel for assistance with zebrafish husbandry; H. Takeda for expression constructs; and A. Chitnis, M. Itoh and M. Tsang for comments on the manuscript. T. K. was supported by JSPS and Fogarty fellowships and is currently supported by Wellcome Trust funding. S. W. is a Wellcome Trust Senior Research Fellow. REFERENCES Abu-Abed, S., Dolle, P., Metzger, D., Beckett, B., Chambon, P. and Petkovich, M. (2001). The retinoic acid-metabolizing enzyme, CYP26A1, is essential for normal hindbrain patterning, vertebral identity, and development of posterior structures. Genes Dev 15,
11 Fgf, Wnt and retinoic acid in posteriorization 4345 Alexandre, D., Clarke, J. D., Oxtoby, E., Yan, Y. L., Jowett, T. and Holder, N. (1996). Ectopic expression of Hoxa-1 in the zebrafish alters the fate of the mandibular arch neural crest and phenocopies a retinoic acid-induced phenotype. Development 122, Amaya, E., Musci, T. J. and Kirschner, M. W. (1991). Expression of a dominant negative mutant of the FGF receptor disrupts mesoderm formation in Xenopus embryos. Cell 66, Begemann, G., Schilling, T. F., Rauch, G. J., Geisler, R. and Ingham, P. W. (2001). The zebrafish neckless mutation reveals a requirement for raldh2 in mesodermal signals that pattern the hindbrain. Development 128, Berggren, K., McCaffery, P., Drager, U. and Forehand, C. J. (1999). Differential distribution of retinoic acid synthesis in the chicken embryo as determined by immunolocalization of the retinoic acid synthetic enzyme, RALDH-2. Dev. Biol. 210, Blumberg, B., Bolado, J., Moreno, T. A., Kintner, C., Evans, R. M. and Papalopulu, N. (1997). An essential role for retinoid signaling in anteroposterior neural patterning. Development 124, Chen, Y., Pollet, N., Niehrs, C. and Pieler, T. (2001). Increased XRALDH2 activity has a posteriorizing effect on the central nervous system of Xenopus embryos. Mech. Dev. 101, Conlon, R. A. (1995). Retinoic acid and pattern formation in vertebrates. Trends Genet. 11, Cox, W. G. and Hemmati-Brivanlou, A. (1995). Caudalization of neural fate by tissue recombination and bfgf. Development 121, de Roos, K., Sonneveld, E., Compaan, B., ten Berge, D., Durston, A. J. and van der Saag, P. T. (1999). Expression of retinoic acid 4-hydroxylase (CYP26) during mouse and Xenopus laevis embryogenesis. Mech. Dev. 82, Durston, A. J., Timmermans, J. P., Hage, W. J., Hendriks, H. F., de Vries, N. J., Heideveld, M. and Nieuwkoop, P. D. (1989). Retinoic acid causes an anteroposterior transformation in the developing central nervous system. Nature 340, Fekany-Lee, K., Gonzalez, E., Miller-Bertoglio, V. and Solnica-Krezel, L. (2000). The homeobox gene bozozok promotes anterior neuroectoderm formation in zebrafish through negative regulation of BMP2/4 and Wnt pathways. Development 127, Fujii, H., Sato, T., Kaneko, S., Gotoh, O., Fujii-Kuriyama, Y., Osawa, K., Kato, S. and Hamada, H. (1997). Metabolic inactivation of retinoic acid by a novel P450 differentially expressed in developing mouse embryos. EMBO J. 16, Furthauer, M., Thisse, C. and Thisse, B. (1997). A role for FGF-8 in the dorsoventral patterning of the zebrafish gastrula. Development 124, Grandel, H., Lun, H., Rauch, G., Rhinn, M., Piotrowski, T., Houart, C., Sordino, P., Ku chler, A., Schulte-Merker, S., Geisler, R. et al. (2002). Retinoic acid signalling in the zebrafish embryo is necessary during presegmentation stages to pattern the anterior-posterior axis of the CNS and to induce a pectoral fin bud. Development 129, Griffin, K., Patient, R. and Holder, N. (1995). Analysis of FGF function in normal and no tail zebrafish embryos reveals separate mechanisms for formation of the trunk and the tail. Development 121, Grinblat, Y., Gamse, J., Patel, M. and Sive, H. (1998). Determination of the zebrafish forebrain: induction and patterning. Development 125, Hashimoto, H., Itoh, M., Yamanaka, Y., Yamashita, S., Shimizu, T., Solnica-Krezel, L., Hibi, M. and Hirano, T. (2000). Zebrafish Dkk1 functions in forebrain specification and axial mesendoderm formation. Dev. Biol. 217, Hollemann, T., Chen, Y., Grunz, H. and Pieler, T. (1998). Regionalized metabolic activity establishes boundaries of retinoic acid signalling. EMBO J. 17, Houart, C., Caneparo, L., Heisenberg, C. P., Barth, K. A., Take-Uchi, M. and Wilson, S. (2002). Establishment of the telencephalon during gastrulation by local suppression of Wnt activity. Nat. Cell Biol. Neuron 35, Itoh, M., Kudoh, T., Dedekian, M., Kim, C. H. and Chitnis, A. B. (2002). A role for iro1 and iro7 in the establishment of an anteroposterior compartment of the ectoderm adjacent to the midbrain-hindbrain boundary. Development 129, Kazanskaya, O., Glinka, A. and Niehrs, C. (2000). The role of Xenopus dickkopf1 in prechordal plate specification and neural patterning. Development 127, Kelly, G. M., Greenstein, P., Erezyilmaz, D. F. and Moon, R. T. (1995). Zebrafish wnt8 and wnt8b share a common activity but are involved in distinct developmental pathways. Development 121, Kengaku, M. and Okamoto, H. (1993). Basic fibroblast growth factor induces differentiation of neural tube and neural crest lineages of cultured ectoderm cells from Xenopus gastrula. Development 119, Kengaku, M. and Okamoto, H. (1995). bfgf as a possible morphogen for the anteroposterior axis of the central nervous system in Xenopus. Development 121, Kiecker, C. and Niehrs, C. (2001). A morphogen gradient of Wnt/betacatenin signalling regulates anteroposterior neural patterning in Xenopus. Development 128, Kim, C. H., Oda, T., Itoh, M., Jiang, D., Artinger, K. B., Chandrasekharappa, S. C., Driever, W. and Chitnis, A. B. (2000). Repressor activity of Headless/Tcf3 is essential for vertebrate head formation. Nature 407, Klein, P. S. and Melton, D. A. (1996). A molecular mechanism for the effect of lithium on development. Proc. Natl. Acad. Sci. USA 93, Kolm, P. J., Apekin, V. and Sive, H. (1997). Xenopus hindbrain patterning requires retinoid signaling. Dev. Biol. 192, Kolm, P. J. and Sive, H. L. (1995). Regulation of the Xenopus labial homeodomain genes, HoxA1 and HoxD1: activation by retinoids and peptide growth factors. Dev. Biol. 167, Koshida, S., Shinya, M., Mizuno, T., Kuroiwa, A. and Takeda, H. (1998). Initial anteroposterior pattern of the zebrafish central nervous system is determined by differential competence of the epiblast. Development 125, Koshida, S., Shinya, M., Nikaido, M., Ueno, N., Schulte-Merker, S., Kuroiwa, A. and Takeda, H. (2002). Inhibition of BMP activity by the FGF signal promotes posterior neural development in zebrafish. Dev. Biol. 244, Kudoh, T. and Dawid, I. B. (2001). Role of the iroquois3 homeobox gene in organizer formation. Proc. Natl. Acad. Sci. USA 98, Kudoh, T., Tsang, M., Hukriede, N. A., Chen, X., Dedekian, M., Clarke, C. J., Kiang, A., Schultz, S., Epstein, J. A., Toyama, R. et al. (2001). A gene expression screen in zebrafish embryogenesis. Genome Res. 11, Lamb, T. M. and Harland, R. M. (1995). Fibroblast growth factor is a direct neural inducer, which combined with noggin generates anterior-posterior neural pattern. Development 121, Loudig, O., Babichuk, C., White, J., Abu-Abed, S., Mueller, C. and Petkovich, M. (2000). Cytochrome P450RAI(CYP26) promoter: a distinct composite retinoic acid response element underlies the complex regulation of retinoic acid metabolism. Mol. Endocrinol. 14, Marshall, H., Studer, M., Popperl, H., Aparicio, S., Kuroiwa, A., Brenner, S. and Krumlauf, R. (1994). A conserved retinoic acid response element required for early expression of the homeobox gene Hoxb-1. Nature 370, McGrew, L. L., Lai, C. J. and Moon, R. T. (1995). Specification of the anteroposterior neural axis through synergistic interaction of the Wnt signaling cascade with noggin and follistatin. Dev. Biol. 172, Mizuno, T., Yamanaka, M., Wakahara, A., Kuroiwa, A. and Takeda, H. (1996). Mesoderm induction in zebrafish. Nature 383, Niederreither, K., McCaffery, P., Drager, U. C., Chambon, P. and Dolle, P. (1997). Restricted expression and retinoic acid-induced downregulation of the retinaldehyde dehydrogenase type 2 (RALDH-2) gene during mouse development. Mech. Dev. 62, Niederreither, K., Subbarayan, V., Dolle, P. and Chambon, P. (1999). Embryonic retinoic acid synthesis is essential for early mouse postimplantation development. Nat. Genet. 21, Nieuwkoop, P. D. (1950). Activation and organization of the central nervous system in amphibians. III. Synthesis of a new working hypothesis. J. Exp. Zool. 120, Ogura, T., Alvarez, I. S., Vogel, A., Rodriguez, C., Evans, R. M. and Izpisua Belmonte, J. C. (1996a). Evidence that Shh cooperates with a retinoic acid inducible co-factor to establish ZPA-like activity. Development 122, Ogura, T., Nakayama, K., Fujisawa, H. and Esumi, H. (1996b). Neuronal nitric oxide synthase expression in neuronal cell differentiation. Neurosci. Lett. 204, Phillips, B. T., Bolding, K. and Riley, B. B. (2001). Zebrafish fgf3 and fgf8 encode redundant functions required for otic placode induction. Dev. Biol. 235, Reifers, F., Bohli, H., Walsh, E. C., Crossley, P. H., Stainier, D. Y. and Brand, M. (1998). Fgf8 is mutated in zebrafish acerebellar (ace) mutants
Supporting Information
 Supporting Information Lu et al. 10.1073/pnas.1106801108 Fig. S1. Analysis of spatial localization of mrnas coding for 23 zebrafish Wnt genes by whole mount in situ hybridization to identify those present
Supporting Information Lu et al. 10.1073/pnas.1106801108 Fig. S1. Analysis of spatial localization of mrnas coding for 23 zebrafish Wnt genes by whole mount in situ hybridization to identify those present
Xenopus gastrulation. Dorsal-Ventral Patterning - The Spemann Organizer. Two mesoderm inducing signals
 Dorsal- atterning - The Spemann Organizer Two mesoderm inducing signals late-blastula early-gastrula nimal Blood Mesothelium Not Vegetal NC Endoderm Hans Spemann (1869-1941) Hilde Mangold (1898-1924) Dr
Dorsal- atterning - The Spemann Organizer Two mesoderm inducing signals late-blastula early-gastrula nimal Blood Mesothelium Not Vegetal NC Endoderm Hans Spemann (1869-1941) Hilde Mangold (1898-1924) Dr
Lecture 3 MOLECULAR REGULATION OF DEVELOPMENT
 Lecture 3 E. M. De Robertis, M.D., Ph.D. August 16, 2016 MOLECULAR REGULATION OF DEVELOPMENT GROWTH FACTOR SIGNALING, HOX GENES, AND THE BODY PLAN Two questions: 1) How is dorsal-ventral (D-V) cell differentiation
Lecture 3 E. M. De Robertis, M.D., Ph.D. August 16, 2016 MOLECULAR REGULATION OF DEVELOPMENT GROWTH FACTOR SIGNALING, HOX GENES, AND THE BODY PLAN Two questions: 1) How is dorsal-ventral (D-V) cell differentiation
Patterning of the Brain
 Patterning of the Brain Clemens.Kiecker@kcl.ac.uk IoPPN 27 Oct 2015 Hundreds of billions of cells, hundreds of cell types Egg = single cell, some 20,000 genes Egg = single cell, some 20,000 genes How is
Patterning of the Brain Clemens.Kiecker@kcl.ac.uk IoPPN 27 Oct 2015 Hundreds of billions of cells, hundreds of cell types Egg = single cell, some 20,000 genes Egg = single cell, some 20,000 genes How is
Readings. Lecture IV. Mechanisms of Neural. Neural Development. September 10, Bio 3411 Lecture IV. Mechanisms of Neural Development
 Readings Lecture IV. Mechanisms of Neural NEUROSCIENCE: References : 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994). EMBO J, 13(21),
Readings Lecture IV. Mechanisms of Neural NEUROSCIENCE: References : 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994). EMBO J, 13(21),
Mesoderm Formation. Fate map of early gastrula. Only two types of mesoderm are induced. Mesoderm induction by the vegetal hemisphere
 Fate map of early gastrula Mesoderm Formation Animal hemisphere forms ectoderm (lacks ) Sperm Entry Point dbl dbl brachyury Vegetal hemisphere forms endoderm (requires ) dbl goosecoid Marginal zone forms
Fate map of early gastrula Mesoderm Formation Animal hemisphere forms ectoderm (lacks ) Sperm Entry Point dbl dbl brachyury Vegetal hemisphere forms endoderm (requires ) dbl goosecoid Marginal zone forms
7.22 Final Exam points
 Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 11 7.22 2004 FINAL FOR STUDY
Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 11 7.22 2004 FINAL FOR STUDY
Fertilization. Animal hemisphere. Sperm entry point
 Fertilization Animal hemisphere Sperm entry point Establishes the dorsal/ventral axis Ventral side sperm entry Dorsal side gray crescent Organized by sperm centriole Cleavage Unequal radial holoblastic
Fertilization Animal hemisphere Sperm entry point Establishes the dorsal/ventral axis Ventral side sperm entry Dorsal side gray crescent Organized by sperm centriole Cleavage Unequal radial holoblastic
Neural Induction. Chapter One
 Neural Induction Chapter One Fertilization Development of the Nervous System Cleavage (Blastula, Gastrula) Neuronal Induction- Neuroblast Formation Cell Migration Mesodermal Induction Lateral Inhibition
Neural Induction Chapter One Fertilization Development of the Nervous System Cleavage (Blastula, Gastrula) Neuronal Induction- Neuroblast Formation Cell Migration Mesodermal Induction Lateral Inhibition
RNAs were transcribed from described expression conhibit a similar capacity to synergize with neural-inducing
 DEVELOPMENTAL BIOLOGY 172, 337 342 (1995) RAPID COMMUNICATION Specification of the Anteroposterior Neural Axis through Synergistic Interaction of the Wnt Signaling Cascade with noggin and follistatin L.
DEVELOPMENTAL BIOLOGY 172, 337 342 (1995) RAPID COMMUNICATION Specification of the Anteroposterior Neural Axis through Synergistic Interaction of the Wnt Signaling Cascade with noggin and follistatin L.
Regulation of Hox gene expression and posterior development by the Xenopus caudal homologue Xcad3
 The EMBO Journal Vol.17 No.12 pp.3413 3427, 1998 Regulation of Hox gene expression and posterior development by the Xenopus caudal homologue Xcad3 Harry V.Isaacs 1, Mary Elizabeth Pownall and Jonathan
The EMBO Journal Vol.17 No.12 pp.3413 3427, 1998 Regulation of Hox gene expression and posterior development by the Xenopus caudal homologue Xcad3 Harry V.Isaacs 1, Mary Elizabeth Pownall and Jonathan
Pregnenolone Stabilizes Microtubules and Promotes Zebrafish Embryonic Cell Movement
 Pregnenolone Stabilizes Microtubules and Promotes Zebrafish Embryonic Cell Movement Hwei-Jan Hsu 1,2, Ming-Ren Liang 3, Chao-Tsen Chen 3, and Bon-Chu Chung 1 * Abstract Embryonic cell movement is essential
Pregnenolone Stabilizes Microtubules and Promotes Zebrafish Embryonic Cell Movement Hwei-Jan Hsu 1,2, Ming-Ren Liang 3, Chao-Tsen Chen 3, and Bon-Chu Chung 1 * Abstract Embryonic cell movement is essential
Neural Development. How does a single cell make a brain??? How are different brain regions specified??? Neural Development
 Neural Development How does a single cell make a brain??? How are different brain regions specified??? 1 Neural Development How do cells become neurons? Environmental factors Positional cues Genetic factors
Neural Development How does a single cell make a brain??? How are different brain regions specified??? 1 Neural Development How do cells become neurons? Environmental factors Positional cues Genetic factors
Understanding embryonic head development. ANAT2341 Tennille Sibbritt Embryology Unit Children s Medical Research Institute
 Understanding embryonic head development ANAT2341 Tennille Sibbritt Embryology Unit Children s Medical Research Institute Head malformations among the most common category of congenital malformations in
Understanding embryonic head development ANAT2341 Tennille Sibbritt Embryology Unit Children s Medical Research Institute Head malformations among the most common category of congenital malformations in
Early Development and Axis Formation in Amphibians
 Biology 4361 Early Development and Axis Formation in Amphibians October 25, 2006 Overview Cortical rotation Cleavage Gastrulation Determination the Organizer mesoderm induction Setting up the axes: dorsal/ventral
Biology 4361 Early Development and Axis Formation in Amphibians October 25, 2006 Overview Cortical rotation Cleavage Gastrulation Determination the Organizer mesoderm induction Setting up the axes: dorsal/ventral
efgf, Xcad3 and Hox genes form a molecular pathway that establishes the
 Development 122, 3881-3892 (1996) Printed in Great Britain The Company of Biologists Limited 1996 DEV3573 3881 efgf, Xcad3 and Hox genes form a molecular pathway that establishes the anteroposterior axis
Development 122, 3881-3892 (1996) Printed in Great Britain The Company of Biologists Limited 1996 DEV3573 3881 efgf, Xcad3 and Hox genes form a molecular pathway that establishes the anteroposterior axis
Neural Induction. Steven McLoon Department of Neuroscience University of Minnesota
 Neural Induction Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour (with Dr. Nakagawa) Friday, Sept 22 8:30-9:30am Surdyks Café in Northrop Auditorium Stop by for
Neural Induction Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour (with Dr. Nakagawa) Friday, Sept 22 8:30-9:30am Surdyks Café in Northrop Auditorium Stop by for
+ + Development and Evolution Dorsoventral axis. Developmental Readout. Foundations. Stem cells. Organ formation.
 Development and Evolution 7.72 9.11.06 Dorsoventral axis Human issues Organ formation Stem cells Developmental Readout Axes Growth control Axon guidance 3D structure Analysis Model + + organisms Foundations
Development and Evolution 7.72 9.11.06 Dorsoventral axis Human issues Organ formation Stem cells Developmental Readout Axes Growth control Axon guidance 3D structure Analysis Model + + organisms Foundations
CHAPTER 2. Stephan A. Wacker*, Hans J. Jansen*, Claire L. McNulty, Erwin Houtzager and Antony J. Durston
 CHAPTER 2 Timed interactions between the Hox expressing nonorganizer mesoderm and the Spemann organizer generate positional information during vertebrate gastrulation Stephan A. Wacker*, Hans J. Jansen*,
CHAPTER 2 Timed interactions between the Hox expressing nonorganizer mesoderm and the Spemann organizer generate positional information during vertebrate gastrulation Stephan A. Wacker*, Hans J. Jansen*,
Induction and patterning of the telencephalon in Xenopus laevis
 Development 129, 5421-5436 (2002) 2002 The Company of Biologists Ltd doi:10.1242/dev.00095 5421 Induction and patterning of the telencephalon in Xenopus laevis Giuseppe Lupo 1,2, William A. Harris 2, Giuseppina
Development 129, 5421-5436 (2002) 2002 The Company of Biologists Ltd doi:10.1242/dev.00095 5421 Induction and patterning of the telencephalon in Xenopus laevis Giuseppe Lupo 1,2, William A. Harris 2, Giuseppina
The competence of Xenopus blastomeres to produce neural and retinal progeny is repressed by two endo-mesoderm promoting pathways
 Developmental Biology 305 (2007) 103 119 www.elsevier.com/locate/ydbio The competence of Xenopus blastomeres to produce neural and retinal progeny is repressed by two endo-mesoderm promoting pathways Bo
Developmental Biology 305 (2007) 103 119 www.elsevier.com/locate/ydbio The competence of Xenopus blastomeres to produce neural and retinal progeny is repressed by two endo-mesoderm promoting pathways Bo
7.22 Example Problems for Exam 1 The exam will be of this format. It will consist of 2-3 sets scenarios.
 Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 10 7.22 Fall 2005 sample exam
Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 10 7.22 Fall 2005 sample exam
Neural crest induction in Xenopus: evidence for a two-signal model
 Development 125, 2403-2414 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV4980 2403 Neural crest induction in Xenopus: evidence for a two-signal model Carole LaBonne* and Marianne
Development 125, 2403-2414 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV4980 2403 Neural crest induction in Xenopus: evidence for a two-signal model Carole LaBonne* and Marianne
The role of the yolk syncytial layer in germ layer patterning in zebrafish
 Development 127, 4681-4689 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV3237 4681 The role of the yolk syncytial layer in germ layer patterning in zebrafish Shaw-Ree Chen and
Development 127, 4681-4689 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV3237 4681 The role of the yolk syncytial layer in germ layer patterning in zebrafish Shaw-Ree Chen and
b-adrenergic signaling promotes posteriorization in Xenopus early development
 The Japanese Society of Developmental Biologists Develop. Growth Differ. (2013) doi: 10.1111/dgd.12046 Original Article b-adrenergic signaling promotes posteriorization in Xenopus early development Shoko
The Japanese Society of Developmental Biologists Develop. Growth Differ. (2013) doi: 10.1111/dgd.12046 Original Article b-adrenergic signaling promotes posteriorization in Xenopus early development Shoko
Regulation of Zebrafish Hindbrain Development by Fibroblast Growth Factor and Retinoic Acid: A Dissertation
 University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 2003-10-01 Regulation of Zebrafish Hindbrain Development by Fibroblast
University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 2003-10-01 Regulation of Zebrafish Hindbrain Development by Fibroblast
Dickkopf-1 regulates gastrulation movements by coordinated modulation of Wnt/
 Dickkopf-1 regulates gastrulation movements by coordinated modulation of Wnt/ catenin and Wnt/PCP activities, through interaction with the Dally-like homolog Knypek Luca Caneparo, 1 Ya-Lin Huang, 3,4 Nicole
Dickkopf-1 regulates gastrulation movements by coordinated modulation of Wnt/ catenin and Wnt/PCP activities, through interaction with the Dally-like homolog Knypek Luca Caneparo, 1 Ya-Lin Huang, 3,4 Nicole
Supplement Figure S1:
 Supplement Figure S1: A, Sequence of Xcadherin-11 Morpholino 1 (Xcad-11MO) and 2 (Xcad-11 MO2) and control morpholino in comparison to the Xcadherin-11 sequence. The Xcad-11MO binding sequence spans the
Supplement Figure S1: A, Sequence of Xcadherin-11 Morpholino 1 (Xcad-11MO) and 2 (Xcad-11 MO2) and control morpholino in comparison to the Xcadherin-11 sequence. The Xcad-11MO binding sequence spans the
A Survey of Genetic Methods
 IBS 8102 Cell, Molecular, and Developmental Biology A Survey of Genetic Methods January 24, 2008 DNA RNA Hybridization ** * radioactive probe reverse transcriptase polymerase chain reaction RT PCR DNA
IBS 8102 Cell, Molecular, and Developmental Biology A Survey of Genetic Methods January 24, 2008 DNA RNA Hybridization ** * radioactive probe reverse transcriptase polymerase chain reaction RT PCR DNA
Retinol dehydrogenase 10 is a feedback regulator of retinoic acid signalling during axis formation and patterning of the central nervous system
 RESEARCH ARTICLE 461 Development 136, 461-472 (2009) doi:10.1242/dev.024901 Retinol dehydrogenase 10 is a feedback regulator of retinoic acid signalling during axis formation and patterning of the central
RESEARCH ARTICLE 461 Development 136, 461-472 (2009) doi:10.1242/dev.024901 Retinol dehydrogenase 10 is a feedback regulator of retinoic acid signalling during axis formation and patterning of the central
MOLECULAR BIOLOGY OF SPEMANN S ORGANIZER AND NEURAL INDUCTION - Lecture 5
 Eddy De Robertis Page 1 MOLECULAR BIOLOGY OF SPEMANN S ORGANIZER AND NEURAL INDUCTION - Lecture 5 Having discussed the early events in mesoderm induction, we now turn to signaling events that take place
Eddy De Robertis Page 1 MOLECULAR BIOLOGY OF SPEMANN S ORGANIZER AND NEURAL INDUCTION - Lecture 5 Having discussed the early events in mesoderm induction, we now turn to signaling events that take place
We are walking and standing with parts of our bodies which could have been used for thinking had they developed in another part of the embryo.
 We are walking and standing with parts of our bodies which could have been used for thinking had they developed in another part of the embryo. Hans Spemann, 1943 Reading from Chapter 3 - types of cell
We are walking and standing with parts of our bodies which could have been used for thinking had they developed in another part of the embryo. Hans Spemann, 1943 Reading from Chapter 3 - types of cell
We are walking and standing with parts of our bodies which could have been used for thinking had they developed in another part of the embryo.
 We are walking and standing with parts of our bodies which could have been used for thinking had they developed in another part of the embryo. Hans Spemann, 1943 Reading from Chapter 3 - types of cell
We are walking and standing with parts of our bodies which could have been used for thinking had they developed in another part of the embryo. Hans Spemann, 1943 Reading from Chapter 3 - types of cell
The Sp1-Related Transcription Factors sp5 and sp5-like Act Downstream of Wnt/ -Catenin Signaling in Mesoderm and Neuroectoderm Patterning
 Current Biology, Vol. 15, 489 500, March 29, 2005, 2005 Elsevier Ltd All rights reserved. DOI 10.1016/j.cub.2005.01.041 The Sp1-Related Transcription Factors sp5 and sp5-like Act Downstream of Wnt/ -Catenin
Current Biology, Vol. 15, 489 500, March 29, 2005, 2005 Elsevier Ltd All rights reserved. DOI 10.1016/j.cub.2005.01.041 The Sp1-Related Transcription Factors sp5 and sp5-like Act Downstream of Wnt/ -Catenin
Patterning of the neural ectoderm of Xenopus laevis by the amino-terminal product of hedgehog autoproteolytic cleavage
 Development 121, 2349-2360 (1995) Printed in Great Britain The Company of Biologists Limited 1995 2349 Patterning of the neural ectoderm of Xenopus laevis by the amino-terminal product of hedgehog autoproteolytic
Development 121, 2349-2360 (1995) Printed in Great Britain The Company of Biologists Limited 1995 2349 Patterning of the neural ectoderm of Xenopus laevis by the amino-terminal product of hedgehog autoproteolytic
The regulation of mesodermal progenitor cell commitment to somitogenesis subdivides the zebrafish body musculature into distinct domains
 The regulation of mesodermal progenitor cell commitment to somitogenesis subdivides the zebrafish body musculature into distinct domains Daniel P. Szeto 1 and David Kimelman 2 Department of Biochemistry,
The regulation of mesodermal progenitor cell commitment to somitogenesis subdivides the zebrafish body musculature into distinct domains Daniel P. Szeto 1 and David Kimelman 2 Department of Biochemistry,
Activity 47.1 What common events occur in the early development of animals? 1. What key events occur at each stage of development?
 Notes to Instructors Chapter 47 Animal Development What is the focus of this activity? Chapter 21 provided a review of how genes act to control development. Chapter 47 reviews some of the major morphological
Notes to Instructors Chapter 47 Animal Development What is the focus of this activity? Chapter 21 provided a review of how genes act to control development. Chapter 47 reviews some of the major morphological
Shawn Burgess 1, *,, Gerlinde Reim 2,, Wenbiao Chen 1,, Nancy Hopkins 1 and Michael Brand 2, SUMMARY
 Development 129, 905-916 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV1707 905 The zebrafish spiel-ohne-grenzen (spg) gene encodes the POU domain protein Pou2 related to mammalian
Development 129, 905-916 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV1707 905 The zebrafish spiel-ohne-grenzen (spg) gene encodes the POU domain protein Pou2 related to mammalian
1. Supplementary Tables
 SUPPLEMENTARY INFORMATION doi:10.1038/nature14215 1. Supplementary Tables Supplementary Table 1 Phenotype of medaka YAP MO-injected embryos Morpholinos Host Amount of MOs Human YAP mrna Total survived
SUPPLEMENTARY INFORMATION doi:10.1038/nature14215 1. Supplementary Tables Supplementary Table 1 Phenotype of medaka YAP MO-injected embryos Morpholinos Host Amount of MOs Human YAP mrna Total survived
Lecture 20: Drosophila melanogaster
 Lecture 20: Drosophila melanogaster Model organisms Polytene chromosome Life cycle P elements and transformation Embryogenesis Read textbook: 732-744; Fig. 20.4; 20.10; 20.15-26 www.mhhe.com/hartwell3
Lecture 20: Drosophila melanogaster Model organisms Polytene chromosome Life cycle P elements and transformation Embryogenesis Read textbook: 732-744; Fig. 20.4; 20.10; 20.15-26 www.mhhe.com/hartwell3
Reading. Lecture III. Nervous System Embryology. Biology. Brain Diseases. September 5, Bio 3411 Lecture III. Nervous System Embryology
 Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 Bio 3411 Wednesday 2 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 Bio 3411 Wednesday 2 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Conserved Patterns of Cell Movements during Vertebrate Gastrulation. Review. Lilianna Solnica-Krezel
 Current Biology, Vol. 15, R213 R228, March 29, 2005, 2005 Elsevier Ltd. All rights reserved. DOI 10.1016/j.cub.2005.03.016 Conserved Patterns of Cell Movements during Vertebrate Gastrulation Review Lilianna
Current Biology, Vol. 15, R213 R228, March 29, 2005, 2005 Elsevier Ltd. All rights reserved. DOI 10.1016/j.cub.2005.03.016 Conserved Patterns of Cell Movements during Vertebrate Gastrulation Review Lilianna
Neural Induction in the Absence of Mesoderm: -Catenin-Dependent Expression of Secreted BMP Antagonists at the Blastula Stage in Xenopus
 Developmental Biology 234, 161 173 (2001) doi:10.1006/dbio.2001.0258, available online at http://www.idealibrary.com on Neural Induction in the Absence of Mesoderm: -Catenin-Dependent Expression of Secreted
Developmental Biology 234, 161 173 (2001) doi:10.1006/dbio.2001.0258, available online at http://www.idealibrary.com on Neural Induction in the Absence of Mesoderm: -Catenin-Dependent Expression of Secreted
Lecture III. Nervous System Embryology
 Bio 3411 Wednesday Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 2 1 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Bio 3411 Wednesday Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 2 1 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Neural induction. Noggin Chordin Follistatin (Xnr3)
 a bird s eye view Since the discovery of the phenomenon of neural induction by Spemann and Mangold in 94, considerable effort has been invested in identifying the signals produced by the organizer that
a bird s eye view Since the discovery of the phenomenon of neural induction by Spemann and Mangold in 94, considerable effort has been invested in identifying the signals produced by the organizer that
ANAT2341 Embryology Introduction: Steve Palmer
 ANAT2341 Embryology Introduction: Steve Palmer Course overview Course lecturers specializations and roles Steve Palmer 9385 2957 Course overview Summary, aims and expected outcomes Course overview Graduate
ANAT2341 Embryology Introduction: Steve Palmer Course overview Course lecturers specializations and roles Steve Palmer 9385 2957 Course overview Summary, aims and expected outcomes Course overview Graduate
Expression Pattern of Xtshz1 and Xtshz3 During Xenopus Early Eye Formation
 SUNY College of Environmental Science and Forestry Digital Commons @ ESF Honors Theses 5-2012 Expression Pattern of Xtshz1 and Xtshz3 During Xenopus Early Eye Formation Katherine L. McKissick Follow this
SUNY College of Environmental Science and Forestry Digital Commons @ ESF Honors Theses 5-2012 Expression Pattern of Xtshz1 and Xtshz3 During Xenopus Early Eye Formation Katherine L. McKissick Follow this
Neural induction in Xenopus requires inhibition of Wnt-β-catenin signaling
 Developmental Biology 298 (2006) 71 86 www.elsevier.com/locate/ydbio Neural induction in Xenopus requires inhibition of Wnt-β-catenin signaling Elizabeth Heeg-Truesdell a, Carole LaBonne a,b, a Department
Developmental Biology 298 (2006) 71 86 www.elsevier.com/locate/ydbio Neural induction in Xenopus requires inhibition of Wnt-β-catenin signaling Elizabeth Heeg-Truesdell a, Carole LaBonne a,b, a Department
Enhancers mutations that make the original mutant phenotype more extreme. Suppressors mutations that make the original mutant phenotype less extreme
 Interactomics and Proteomics 1. Interactomics The field of interactomics is concerned with interactions between genes or proteins. They can be genetic interactions, in which two genes are involved in the
Interactomics and Proteomics 1. Interactomics The field of interactomics is concerned with interactions between genes or proteins. They can be genetic interactions, in which two genes are involved in the
Biphasic wnt8a Expression Is Achieved Through Interactions of Multiple Regulatory Inputs
 a DEVELOPMENTAL DYNAMICS 241:1062 1075, 2012 RESEARCH ARTICLE Biphasic wnt8a Expression Is Achieved Through Interactions of Multiple Regulatory Inputs Anand Narayanan and Arne C. Lekven* Background: Vertebrate
a DEVELOPMENTAL DYNAMICS 241:1062 1075, 2012 RESEARCH ARTICLE Biphasic wnt8a Expression Is Achieved Through Interactions of Multiple Regulatory Inputs Anand Narayanan and Arne C. Lekven* Background: Vertebrate
Combinatorial gene regulation by Bmp and Wnt in zebrafish posterior mesoderm formation
 Corrigendum Combinatorial gene regulation by Bmp and Wnt in zebrafish posterior mesoderm formation Daniel P. Szeto and David Kimelman Development 131, 3751-3760. The labelling in Fig. 6 of this article
Corrigendum Combinatorial gene regulation by Bmp and Wnt in zebrafish posterior mesoderm formation Daniel P. Szeto and David Kimelman Development 131, 3751-3760. The labelling in Fig. 6 of this article
Chapter 3. Two Hoxc6 transcripts are differentially expressed and regulate. primary neurogenesis in Xenopus laevis
 Chapter 3 Two Hoxc6 transcripts are differentially expressed and regulate primary neurogenesis in Xenopus laevis Nabila Bardine 1, Cornelia Donow 1, Brigitte Korte 1, Antony J. Durston 2, Walter Knöchel
Chapter 3 Two Hoxc6 transcripts are differentially expressed and regulate primary neurogenesis in Xenopus laevis Nabila Bardine 1, Cornelia Donow 1, Brigitte Korte 1, Antony J. Durston 2, Walter Knöchel
HOW DOES Wnt SIGNALING POSTERIORIZE THE NEURAL PLATE?
 HOW DOES Wnt SIGNALING POSTERIORIZE THE NEURAL PLATE? An Undergraduate Research Scholars Thesis By JEFFREY JOON TAEK LEE Submitted to Honors and Undergraduate Research Texas A&M University In partial fulfillment
HOW DOES Wnt SIGNALING POSTERIORIZE THE NEURAL PLATE? An Undergraduate Research Scholars Thesis By JEFFREY JOON TAEK LEE Submitted to Honors and Undergraduate Research Texas A&M University In partial fulfillment
ABSTRACT. Transgenic Zebrafish That Carry Inducible zic2a Fusion Proteins
 ABSTRACT Transgenic Zebrafish That Carry Inducible zic2a Fusion Proteins Zic genes encode proteins that contain zinc fingers used to bind DNA and modify transcription of target genes. Several zic genes
ABSTRACT Transgenic Zebrafish That Carry Inducible zic2a Fusion Proteins Zic genes encode proteins that contain zinc fingers used to bind DNA and modify transcription of target genes. Several zic genes
SUPPLEMENTARY INFORMATION
 Normalized dorsal trunk length (µm) Normalized dorsal trunk length (TC1-5) (µm) DOI: 1.138/ncb2467 a b c d btl>src64 Src42, btl>src64 e Src64 f g h DT Src64 DT Midgut Src64 Midgut i Src64 Atpα j k l DT
Normalized dorsal trunk length (µm) Normalized dorsal trunk length (TC1-5) (µm) DOI: 1.138/ncb2467 a b c d btl>src64 Src42, btl>src64 e Src64 f g h DT Src64 DT Midgut Src64 Midgut i Src64 Atpα j k l DT
Concepts and Methods in Developmental Biology
 Biology 4361 Developmental Biology Concepts and Methods in Developmental Biology June 16, 2009 Conceptual and Methodological Tools Concepts Genomic equivalence Differential gene expression Differentiation/de-differentiation
Biology 4361 Developmental Biology Concepts and Methods in Developmental Biology June 16, 2009 Conceptual and Methodological Tools Concepts Genomic equivalence Differential gene expression Differentiation/de-differentiation
Applicazioni biotecnologiche
 Applicazioni biotecnologiche Analisi forense Sintesi di proteine ricombinanti Restriction Fragment Length Polymorphism (RFLP) Polymorphism (more fully genetic polymorphism) refers to the simultaneous occurrence
Applicazioni biotecnologiche Analisi forense Sintesi di proteine ricombinanti Restriction Fragment Length Polymorphism (RFLP) Polymorphism (more fully genetic polymorphism) refers to the simultaneous occurrence
Supplementary Figure 1. Soft fibrin gels promote growth and organized mesodermal differentiation. Representative images of single OGTR1 ESCs cultured
 Supplementary Figure 1. Soft fibrin gels promote growth and organized mesodermal differentiation. Representative images of single OGTR1 ESCs cultured in 90-Pa 3D fibrin gels for 5 days in the presence
Supplementary Figure 1. Soft fibrin gels promote growth and organized mesodermal differentiation. Representative images of single OGTR1 ESCs cultured in 90-Pa 3D fibrin gels for 5 days in the presence
Zebrafish wnt8 Encodes Two Wnt8 Proteins on a Bicistronic Transcript and Is Required for Mesoderm and Neurectoderm Patterning
 Developmental Cell, Vol. 1, 103 114, July, 2001, Copyright 2001 by Cell Press Zebrafish wnt8 Encodes Two Wnt8 Proteins on a Bicistronic Transcript and Is Required for Mesoderm and Neurectoderm Patterning
Developmental Cell, Vol. 1, 103 114, July, 2001, Copyright 2001 by Cell Press Zebrafish wnt8 Encodes Two Wnt8 Proteins on a Bicistronic Transcript and Is Required for Mesoderm and Neurectoderm Patterning
Review Article On becoming neural: what the embryo can tell us about differentiating neural stem cells
 Am J Stem Cells 2013;2(2):74-94 www.ajsc.us /ISSN:2160-4150/AJSC1303003 Review Article : what the embryo can tell us about differentiating neural stem cells Sally A Moody 1,3, Steven L Klein 1, Beverley
Am J Stem Cells 2013;2(2):74-94 www.ajsc.us /ISSN:2160-4150/AJSC1303003 Review Article : what the embryo can tell us about differentiating neural stem cells Sally A Moody 1,3, Steven L Klein 1, Beverley
7.013 Practice Quiz
 MIT Department of Biology 7.013: Introductory Biology - Spring 2005 Instructors: Professor Hazel Sive, Professor Tyler Jacks, Dr. Claudette Gardel 7.013 Practice Quiz 2 2004 1 Question 1 A. The primer
MIT Department of Biology 7.013: Introductory Biology - Spring 2005 Instructors: Professor Hazel Sive, Professor Tyler Jacks, Dr. Claudette Gardel 7.013 Practice Quiz 2 2004 1 Question 1 A. The primer
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2806 Supplemental Material: Figure S1 Wnt4 PCP phenotypes are not mediated through canonical Wnt signaling. (a-a ) Illustration of wg, dwnt4 and dwnt6 expression patterns in wing (a) and
DOI: 10.1038/ncb2806 Supplemental Material: Figure S1 Wnt4 PCP phenotypes are not mediated through canonical Wnt signaling. (a-a ) Illustration of wg, dwnt4 and dwnt6 expression patterns in wing (a) and
Later Development. Caenorhabditis elegans. Later Processes 10/06/12. Cytoplasmic Determinants Fate Mapping & Cell Fate Limb Development
 Later Development Cytoplasmic Determinants Fate Mapping & Cell Fate Limb Development Caenorhabditis elegans Nematoda 10,000 worms/petri dish in cultivation short life cycle (~ 3 days egg to egg) wild-type
Later Development Cytoplasmic Determinants Fate Mapping & Cell Fate Limb Development Caenorhabditis elegans Nematoda 10,000 worms/petri dish in cultivation short life cycle (~ 3 days egg to egg) wild-type
Name AP Biology Mrs. Laux Take home test #11 on Chapters 14, 15, and 17 DUE: MONDAY, DECEMBER 21, 2009
 MULTIPLE CHOICE QUESTIONS 1. Inducible genes are usually actively transcribed when: A. the molecule degraded by the enzyme(s) is present in the cell. B. repressor molecules bind to the promoter. C. lactose
MULTIPLE CHOICE QUESTIONS 1. Inducible genes are usually actively transcribed when: A. the molecule degraded by the enzyme(s) is present in the cell. B. repressor molecules bind to the promoter. C. lactose
Xenopus axis formation: induction of goosecoid by injected Xwnt-8 and activin mrnas
 Development 118, 499-507 (1993) Printed in Great Britain The Company of Biologists Limited 1993 499 Xenopus axis formation: induction of goosecoid by injected Xwnt-8 and activin mrnas Herbert Steinbeisser
Development 118, 499-507 (1993) Printed in Great Britain The Company of Biologists Limited 1993 499 Xenopus axis formation: induction of goosecoid by injected Xwnt-8 and activin mrnas Herbert Steinbeisser
Supplemental Information
 Supplemental Information Itemized List Materials and Methods, Related to Supplemental Figures S5A-C and S6. Supplemental Figure S1, Related to Figures 1 and 2. Supplemental Figure S2, Related to Figure
Supplemental Information Itemized List Materials and Methods, Related to Supplemental Figures S5A-C and S6. Supplemental Figure S1, Related to Figures 1 and 2. Supplemental Figure S2, Related to Figure
1. 40 points Retinoic acid (structure below) is a small hydrophobic molecule that is derived from vitamin A. Figure by MIT OCW.
 Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 9 7.22 Fall 2005 exam 2 practice
Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 9 7.22 Fall 2005 exam 2 practice
Chapter 14 Regulation of Transcription
 Chapter 14 Regulation of Transcription Cis-acting sequences Distance-independent cis-acting elements Dissecting regulatory elements Transcription factors Overview transcriptional regulation Transcription
Chapter 14 Regulation of Transcription Cis-acting sequences Distance-independent cis-acting elements Dissecting regulatory elements Transcription factors Overview transcriptional regulation Transcription
Supplementary Information. Isl2b regulates anterior second heart field development in zebrafish
 Supplementary Information Isl2b regulates anterior second heart field development in zebrafish Hagen R. Witzel 1, Sirisha Cheedipudi 1, Rui Gao 1, Didier Y.R. Stainier 2 and Gergana D. Dobreva 1,3* 1 Origin
Supplementary Information Isl2b regulates anterior second heart field development in zebrafish Hagen R. Witzel 1, Sirisha Cheedipudi 1, Rui Gao 1, Didier Y.R. Stainier 2 and Gergana D. Dobreva 1,3* 1 Origin
Chapter 6 - Molecular Genetic Techniques
 Chapter 6 - Molecular Genetic Techniques Two objects of molecular & genetic technologies For analysis For generation Molecular genetic technologies! For analysis DNA gel electrophoresis Southern blotting
Chapter 6 - Molecular Genetic Techniques Two objects of molecular & genetic technologies For analysis For generation Molecular genetic technologies! For analysis DNA gel electrophoresis Southern blotting
Enzyme that uses RNA as a template to synthesize a complementary DNA
 Biology 105: Introduction to Genetics PRACTICE FINAL EXAM 2006 Part I: Definitions Homology: Comparison of two or more protein or DNA sequence to ascertain similarities in sequences. If two genes have
Biology 105: Introduction to Genetics PRACTICE FINAL EXAM 2006 Part I: Definitions Homology: Comparison of two or more protein or DNA sequence to ascertain similarities in sequences. If two genes have
Biology 4361 Developmental Biology Lecture 4. The Genetic Core of Development
 Biology 4361 Developmental Biology Lecture 4. The Genetic Core of Development The only way to get from genotype to phenotype is through developmental processes. - Remember the analogy that the zygote contains
Biology 4361 Developmental Biology Lecture 4. The Genetic Core of Development The only way to get from genotype to phenotype is through developmental processes. - Remember the analogy that the zygote contains
-RT RT RT -RT XBMPRII
 Base pairs 2000 1650 1000 850 600 500 400 Marker Primer set 1 Primer set 2 -RT RT RT -RT XBMPRII kda 100 75 50 37 25 20 XAC p-xac A 300 B Supplemental figure 1. PCR analysis of BMPRII expression and western
Base pairs 2000 1650 1000 850 600 500 400 Marker Primer set 1 Primer set 2 -RT RT RT -RT XBMPRII kda 100 75 50 37 25 20 XAC p-xac A 300 B Supplemental figure 1. PCR analysis of BMPRII expression and western
Role of Bmi-1 in Epigenetic Regulation. During Early Neural Crest Development
 Role of Bmi-1 in Epigenetic Regulation During Early Neural Crest Development Thesis by Jane I. Khudyakov In Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy California Institute
Role of Bmi-1 in Epigenetic Regulation During Early Neural Crest Development Thesis by Jane I. Khudyakov In Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy California Institute
Optimizing Cas9 and sgrna concentrations for increasing mutation efficiency. Nature Methods: doi: /nmeth.3360
 Supplementary Figure 1 Optimizing Cas9 and sgrna concentrations for increasing mutation efficiency. (a) Wild-type, 2-dpf slc24a5 b1 embryos and a series of slc24a5 CRISPR-injected embryos; anterior is
Supplementary Figure 1 Optimizing Cas9 and sgrna concentrations for increasing mutation efficiency. (a) Wild-type, 2-dpf slc24a5 b1 embryos and a series of slc24a5 CRISPR-injected embryos; anterior is
Animal Development Regulation of gene expression
 Animal Development Regulation of gene expression Prof. Ilan Davis, Department of Biochemistry. Wellcome Trust Research Senior Fellow Senior Research Fellow, Jesus College ilan.davis@bioch.ox.ac.uk http://www.ilandavis.com
Animal Development Regulation of gene expression Prof. Ilan Davis, Department of Biochemistry. Wellcome Trust Research Senior Fellow Senior Research Fellow, Jesus College ilan.davis@bioch.ox.ac.uk http://www.ilandavis.com
Regulation of Acetylcholine Receptor Clustering by ADF/Cofilin- Directed Vesicular Trafficking
 Regulation of Acetylcholine Receptor Clustering by ADF/Cofilin- Directed Vesicular Trafficking Chi Wai Lee, Jianzhong Han, James R. Bamburg, Liang Han, Rachel Lynn, and James Q. Zheng Supplementary Figures
Regulation of Acetylcholine Receptor Clustering by ADF/Cofilin- Directed Vesicular Trafficking Chi Wai Lee, Jianzhong Han, James R. Bamburg, Liang Han, Rachel Lynn, and James Q. Zheng Supplementary Figures
Jak1 kinase is required for cell migrations and anterior specification in zebrafish embryos
 Proc. Natl. Acad. Sci. USA Vol. 94, pp. 3082 3087, April 1997 Developmental Biology Jak1 kinase is required for cell migrations and anterior specification in zebrafish embryos GREG CONWAY*, AYELET MARGOLIATH*,
Proc. Natl. Acad. Sci. USA Vol. 94, pp. 3082 3087, April 1997 Developmental Biology Jak1 kinase is required for cell migrations and anterior specification in zebrafish embryos GREG CONWAY*, AYELET MARGOLIATH*,
Molecular Biology of the Cell ed. By Bruce Albert et al. (free online through ncbi books)
 Lecture 20 continued: Drosophila embryogenesis Embrygenesis Four classes of genes: Maternal genes Gap genes Pair-rule genes Segment polarity genes Homeotic genes Read 826-837Fig. D18- D27; 19.2; 19.16
Lecture 20 continued: Drosophila embryogenesis Embrygenesis Four classes of genes: Maternal genes Gap genes Pair-rule genes Segment polarity genes Homeotic genes Read 826-837Fig. D18- D27; 19.2; 19.16
CHAPTER 21 GENOMES AND THEIR EVOLUTION
 GENETICS DATE CHAPTER 21 GENOMES AND THEIR EVOLUTION COURSE 213 AP BIOLOGY 1 Comparisons of genomes provide information about the evolutionary history of genes and taxonomic groups Genomics - study of
GENETICS DATE CHAPTER 21 GENOMES AND THEIR EVOLUTION COURSE 213 AP BIOLOGY 1 Comparisons of genomes provide information about the evolutionary history of genes and taxonomic groups Genomics - study of
Ethanol induces embryonic malformations by competing for retinaldehyde dehydrogenase activity during vertebrate gastrulation
 Disease Models & Mechanisms 2, 295-35 (29) doi:.242/dmm.42 Ethanol induces embryonic malformations by competing for retinaldehyde dehydrogenase activity during vertebrate gastrulation Hadas Kot-Leibovich
Disease Models & Mechanisms 2, 295-35 (29) doi:.242/dmm.42 Ethanol induces embryonic malformations by competing for retinaldehyde dehydrogenase activity during vertebrate gastrulation Hadas Kot-Leibovich
The Pitx2 Homeobox Protein Is Required Early for Endoderm Formation and Nodal Signaling
 Developmental Biology 229, 287 306 (2001) doi:10.1006/dbio.2000.9950, available online at http://www.idealibrary.com on The Pitx2 Homeobox Protein Is Required Early for Endoderm Formation and Nodal Signaling
Developmental Biology 229, 287 306 (2001) doi:10.1006/dbio.2000.9950, available online at http://www.idealibrary.com on The Pitx2 Homeobox Protein Is Required Early for Endoderm Formation and Nodal Signaling
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/318/5851/794/dc1 Supporting Online Material for A Gene Regulatory Network Subcircuit Drives a Dynamic Pattern of Gene Expression Joel Smith, Christina Theodoris, Eric
www.sciencemag.org/cgi/content/full/318/5851/794/dc1 Supporting Online Material for A Gene Regulatory Network Subcircuit Drives a Dynamic Pattern of Gene Expression Joel Smith, Christina Theodoris, Eric
TRANSGENIC ANIMALS. -transient transfection of cells -stable transfection of cells. - Two methods to produce transgenic animals:
 TRANSGENIC ANIMALS -transient transfection of cells -stable transfection of cells - Two methods to produce transgenic animals: 1- DNA microinjection - random insertion 2- embryonic stem cell-mediated gene
TRANSGENIC ANIMALS -transient transfection of cells -stable transfection of cells - Two methods to produce transgenic animals: 1- DNA microinjection - random insertion 2- embryonic stem cell-mediated gene
The Competence of Marginal Zone Cells to Become Spemann s Organizer Is Controlled by Xcad2
 Developmental Biology 248, 40 51 (2002) doi:10.1006/dbio.2002.0705 The Competence of Marginal Zone Cells to Become Spemann s Organizer Is Controlled by Xcad2 Vered Levy, Karen Marom, Sharon Zins, Natalia
Developmental Biology 248, 40 51 (2002) doi:10.1006/dbio.2002.0705 The Competence of Marginal Zone Cells to Become Spemann s Organizer Is Controlled by Xcad2 Vered Levy, Karen Marom, Sharon Zins, Natalia
BMP-2 and BMP-4 signalling in the developing spinal cord of human and rat embryos
 O R I G I N A L A R T I C L E Folia Morphol. Vol. 74, No. 3, pp. 359 364 DOI: 10.5603/FM.2015.0054 Copyright 2015 Via Medica ISSN 0015 5659 www.fm.viamedica.pl BMP-2 and BMP-4 signalling in the developing
O R I G I N A L A R T I C L E Folia Morphol. Vol. 74, No. 3, pp. 359 364 DOI: 10.5603/FM.2015.0054 Copyright 2015 Via Medica ISSN 0015 5659 www.fm.viamedica.pl BMP-2 and BMP-4 signalling in the developing
Gene Expression: Transcription
 Gene Expression: Transcription The majority of genes are expressed as the proteins they encode. The process occurs in two steps: Transcription = DNA RNA Translation = RNA protein Taken together, they make
Gene Expression: Transcription The majority of genes are expressed as the proteins they encode. The process occurs in two steps: Transcription = DNA RNA Translation = RNA protein Taken together, they make
KEY Reproductive cloning Therapeutic cloning
 1. (20 pts) Define Reproductive and Therapeutic cloning. Make sure your descriptions clearly distinguish the critical differences between them. Describe an example of each. Reproductive cloning refers
1. (20 pts) Define Reproductive and Therapeutic cloning. Make sure your descriptions clearly distinguish the critical differences between them. Describe an example of each. Reproductive cloning refers
CAP BIOINFORMATICS Su-Shing Chen CISE. 10/5/2005 Su-Shing Chen, CISE 1
 CAP 5510-9 BIOINFORMATICS Su-Shing Chen CISE 10/5/2005 Su-Shing Chen, CISE 1 Basic BioTech Processes Hybridization PCR Southern blotting (spot or stain) 10/5/2005 Su-Shing Chen, CISE 2 10/5/2005 Su-Shing
CAP 5510-9 BIOINFORMATICS Su-Shing Chen CISE 10/5/2005 Su-Shing Chen, CISE 1 Basic BioTech Processes Hybridization PCR Southern blotting (spot or stain) 10/5/2005 Su-Shing Chen, CISE 2 10/5/2005 Su-Shing
Expression of Siamois and Twin in the blastula Chordin/Noggin signaling center is required for brain formation in Xenopus laevis embryos
 MECHANISMS OF DEVELOPMENT 125 (2008) 58 66 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/modo Expression of Siamois and Twin in the blastula Chordin/Noggin signaling center
MECHANISMS OF DEVELOPMENT 125 (2008) 58 66 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/modo Expression of Siamois and Twin in the blastula Chordin/Noggin signaling center
OmicsLink shrna Clones guaranteed knockdown even in difficult-to-transfect cells
 OmicsLink shrna Clones guaranteed knockdown even in difficult-to-transfect cells OmicsLink shrna clone collections consist of lentiviral, and other mammalian expression vector based small hairpin RNA (shrna)
OmicsLink shrna Clones guaranteed knockdown even in difficult-to-transfect cells OmicsLink shrna clone collections consist of lentiviral, and other mammalian expression vector based small hairpin RNA (shrna)
HD1 - Introduction to Human Development
 Human Development Ed Laufer, PhD Course Director elaufer@columbia.edu Ping Feng Course Administrator pf2013@columbia.edu Michael Shen, PhD Patricia Ducy, PhD Michael Gershon, MD Cathy Mendelsohn, PhD Richard
Human Development Ed Laufer, PhD Course Director elaufer@columbia.edu Ping Feng Course Administrator pf2013@columbia.edu Michael Shen, PhD Patricia Ducy, PhD Michael Gershon, MD Cathy Mendelsohn, PhD Richard
Activin-mediated mesoderm induction requires FGF
 Development 120, 453-462 (1994) Printed in Great Britain The Company of Biologists Limited 1994 453 Activin-mediated mesoderm induction requires FGF Robert A. Cornell and David Kimelman* Department of
Development 120, 453-462 (1994) Printed in Great Britain The Company of Biologists Limited 1994 453 Activin-mediated mesoderm induction requires FGF Robert A. Cornell and David Kimelman* Department of
Endogenous Cerberus activity is required for anterior head specification in Xenopus
 Research article 4943 Endogenous Cerberus activity is required for anterior head specification in Xenopus Ana Cristina Silva 1,, Mario Filipe 1,, Klaus-Michael Kuerner 2, *, Herbert Steinbeisser 2, *,
Research article 4943 Endogenous Cerberus activity is required for anterior head specification in Xenopus Ana Cristina Silva 1,, Mario Filipe 1,, Klaus-Michael Kuerner 2, *, Herbert Steinbeisser 2, *,
OriGene GFC-Arrays for High-throughput Overexpression Screening of Human Gene Phenotypes
 OriGene GFC-Arrays for High-throughput Overexpression Screening of Human Gene Phenotypes High-throughput Gene Function Validation Tool Introduction sirna screening libraries enable scientists to identify
OriGene GFC-Arrays for High-throughput Overexpression Screening of Human Gene Phenotypes High-throughput Gene Function Validation Tool Introduction sirna screening libraries enable scientists to identify
Supplementary Data. Supplementary Methods Three-step protocol for spontaneous differentiation of mouse induced pluripotent stem (embryonic stem) cells
 Supplementary Data Supplementary Methods Three-step protocol for spontaneous differentiation of mouse induced pluripotent stem (embryonic stem) cells Mouse induced pluripotent stem cells (ipscs) were cultured
Supplementary Data Supplementary Methods Three-step protocol for spontaneous differentiation of mouse induced pluripotent stem (embryonic stem) cells Mouse induced pluripotent stem cells (ipscs) were cultured
Nucleosome regulator Xhmgb3 is required for cell proliferation of the eye and brain as a downstream target of Xenopus rax/rx1
 Developmental Biology 291 (2006) 398 412 www.elsevier.com/locate/ydbio Genomes & Developmental Control Nucleosome regulator Xhmgb3 is required for cell proliferation of the eye and brain as a downstream
Developmental Biology 291 (2006) 398 412 www.elsevier.com/locate/ydbio Genomes & Developmental Control Nucleosome regulator Xhmgb3 is required for cell proliferation of the eye and brain as a downstream
Differential Gene Expression
 Biology 4361 Developmental Biology Differential Gene Expression September 28, 2006 Chromatin Structure ~140 bp ~60 bp Transcriptional Regulation: 1. Packing prevents access CH 3 2. Acetylation ( C O )
Biology 4361 Developmental Biology Differential Gene Expression September 28, 2006 Chromatin Structure ~140 bp ~60 bp Transcriptional Regulation: 1. Packing prevents access CH 3 2. Acetylation ( C O )
The role of erna in chromatin looping
 The role of erna in chromatin looping Introduction Enhancers are cis-acting DNA regulatory elements that activate transcription of target genes 1,2. It is estimated that 400 000 to >1 million alleged enhancers
The role of erna in chromatin looping Introduction Enhancers are cis-acting DNA regulatory elements that activate transcription of target genes 1,2. It is estimated that 400 000 to >1 million alleged enhancers
As a control to demonstrate that we could isolate PCR products that result from ligating
 SUPPLEMENTAL INFORMATION (Mullen and Marzluff) RESULTS Oligo(dA) Clones and Oligo(dT) RT-PCR controls. As a control to demonstrate that we could isolate PCR products that result from ligating the authentic
SUPPLEMENTAL INFORMATION (Mullen and Marzluff) RESULTS Oligo(dA) Clones and Oligo(dT) RT-PCR controls. As a control to demonstrate that we could isolate PCR products that result from ligating the authentic
