with extensive sequence homology to Escherichia coli SSB (DNA and protein sequences/functional domains/secondary structure/evolution)
|
|
|
- Dortha Charles
- 6 years ago
- Views:
Transcription
1 Proc. Nati. Acad. Sci. USA Vol. 80, pp , September 1983 Biochemistry F sex factor encodes a single-stranded DNA binding protein (SSB) with extensive sequence homology to Escherichia coli SSB (DNA and protein sequences/functional domains/secondary structure/evolution) JOHN W. CHASE*, BARBARA M. MERRILLt, AND KENNETH R. WILLIAMSt *Department of Molecular Biology, Albert Einstein College of Medicine, Bronx, NY 10461; and tdepartment of Molecular Biophysics and Biochemistry, Yale University School of Medicine, New Haven, CT Communicated by Evelyn M. Witkin, May 23, 1983 ABSTRACT We have determined the sequence of the gene encoding a single-stranded DNA (ss DNA) binding protein (SSB) from the Escherichia coli F sex factor and the amino acid sequence of the protein it encodes. The protein has extensive homology with E. coli SSB, particularly within its NH2-terminal region, where 87 of the first 115 amino acid residues are identical to those of the E. coli protein. We have previously shown that this portion of E. coli SSB contains the DNA binding region. The sequences diverge extensively in their COOH-terminal regions, although small areas of homology exist in several places. Six of the last seven amino acid residues of the two proteins are identical, which may have implications in terms of the direct interactions of these proteins with other proteins required for DNA replication, recombination, and repair. The coding region of the F plasmid ssfgene is 537 base pairs. The protein encoded by the gene contains 178 amino acids (one more than E. coli SSB) and has a calculated molecular weight of 19,505. Other than the presumptive Shine-Dalgarno sequence, the promoter and terminator regions of both genes are not similar. The most significant feature in this regard may be the lack of a region of dyad symmetry within the presumptive promoter of the F plasmid ssf gene as is found in the region of the presumptive E. coli ssb promoter. In this report the predicted secondary structures of both the F plasmid and E. coli SSB proteins are compared and the evolutionary significance of their sequence and structural similarities to the functional domains of the proteins are discussed. It is now well established that the Escherichia coli single-stranded DNA (ss DNA) binding protein (SSB) plays a necessary role in DNA replication (1), recombination (2, 3), and repair (4, 5). Corresponding proteins from several other systems, including eukaryotic organisms, have also been described and similarly implicated in DNA metabolic processes (6, 7). Recently it has been shown that the E. coli F sex factor (F plasmid) encodes a protein that binds strongly to ss DNA and partially complements defects caused by a chromosomal ssb-l mutation (8). We have determined the amino acid sequence of this protein and the nucleotide sequence of the gene that encodes it and have found it to contain regions of extensive homology to E. coli SSB. Although evidence has been presented for corresponding functional domains of the E. coli and phage T4 DNA binding proteins (9), obvious sequence homology to a previously characterized SSB has not been described for a protein of this type. Our results are discussed in terms of the functional domains of E. coli and F plasmid SSB and the possible interaction of these proteins with other replication, recombination, and repair proteins. The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" in accordance with 18 U.S.C solely to indicate this fact MATERIALS AND METHODS Bacterial Strains and Plasmids. The E. coli K-12 strains used were HMS91 (W3110 rha-, thy-) and KLC850 (ssb-1 derivative of HMS91). pkac51 is an ampicillin-resistant tetracyclinesensitive derivative of pbr322 that was constructed by isolating the Pvu Il/Cia I DNA fragment from pbr322 encoding /3-lactamase and the origin of replication before nuclease SI treatment and subsequent ligation and transformation. pkac50 was constructed by ligating the Pvu Il/CMa I DNA fragment from pbr322 described above for pkac51 to the Cla I/Sma I DNA fragment containing the BamHI cleavage site from EcoRI fragment 3 from F plasmid. This DNA fragment was isolated from pkl1 (see diagrams of pkl1 and pkac50 in ref. 8). peg310 was derived from pkac50 by cleavage with BamHI, filling in the resulting termini with deoxynucleoside triphosphates in the presence of the large subunit of E. coli DNA polymerase I, and ligating (8). Media and Reagents. All restriction enzymes were from New England BioLabs. Polynucleotide kinase was from P-L Biochemicals. All radioactively labeled nucleoside triphosphates were from Amersham. F plasmid SSB for tryptic peptide analysis was purified from pkac50 by a modification of the previously described procedure (10). Sequence Determination of the F Plasmid sf Gene. All DNA fragments were purified from 5% or 8% (wt/vol) polyacrylamide gels. Termini were labeled and the sequence was determined as described (11). Protein Chemistry of F Plasmid SSB. F plasmid SSB was purified by a procedure to be described later. The protein was denatured by adding trichloroacetic acid (10%, vol/vol). The acid-precipitated SSB was redissolved in 8 M urea and then diluted to a final concentration of 2.2 M urea, 0.07 M NH4HCO3. After trypsin digestion (protein-to-enzyme weight ratio of 25, 24 hr at 370C) the resulting peptide mixture was fractionated by HPLC and then characterized as described (12-14). The sequence of amino acid residues 2-14 in F plasmid SSB was determined with 1.5 nmol of SSB that had been coupled to diisothiocyanate glass (15). Some SSB tryptic peptides were also sequenced after they were coupled to aminopolystyrene by using a water-soluble carbodiimide (15). The secondary structure of F plasmid SSB was predicted from a computer program (16) based on the method of Chou and Fasman (17). Abbreviations: ss DNA, single-stranded DNA; SSB, the Escherichia coli or F plasmid ss DNA binding protein [the E. coli SSB has also been referred to as a DNA helix-destabilizing protein (EcoHD-protein I)]; ssf, structural gene for F plasmid-encoded SSB as previously suggested by Kolodkin et al. (8); EOP, efficiency of plating.
2 RESULTS F Plasmid-Encoded SSB Complements the ss DNA Binding Deficiency and Temperature-Sensitive Conditional Lethality of an E. coli ssb-1 Mutant Strain. Studies by Kolodkin et al. (8) showed that a high copy number plasmid containing EcoRI DNA fragment 3 from F plasmid complemented the conditional lethality of an ssb-1 strain and that mutation of the unique BamHI cleavage site in this DNA fragment prevented this effect. We inserted the Sma I to Cla I DNA fragment that includes the BamHI cleavage site of EcoRI DNA fragment 3 into the Pvu II to Cla I portion of pbr322 encoding 13-lactamase and the origin of replication. This plasmid (pkac50) encoded two proteins as visualized by maxicells, one of which bound tightly to ss DNA and appeared to have a subunit molecular weight of 22,000. This latter protein was not made by a derivative of pkac50 that was altered at the BamHI cleavage site (peg310) (8). Measurements of the binding activity of extracts of ssb-1 mutant strains containing these plasmids are shown in Table 1. We have previously shown that extracts of ssb-1 strains do not contain measurable amounts of ss DNA binding activity as determined under the conditions of our filter binding assay (18). Nearly 80% of wild-type binding activity is, however, restored in an ssb-1 strain containing pkac50, but none is detected in the same strain carrying a derivative of pkac50 with a mutation at the BamHI cleavage site (peg310). This binding activity also directly correlates with the EOP of these strains measured at 430C. We conclude, in agreement with the previous studies of Kolodkin et al. (8), that these complementation effects of the ssb-1 mutation are due to a ss DNA binding protein encoded by F plasmid. Nucleotide Sequence of the F Plasmid-Encoded ssf Gene. From the analyses of pkac50 performed in our laboratory and in Low's laboratory (8), it was known that the BamHI cleavage site was within either the coding sequence or a control region of the F plasmid ssf gene. We subsequently determined that a cleavage site for Bal I was very close to the BamHI site, and Table 1. Biochemistrv: Chase et al. Complementation of ss DNA binding activity by F plasmid-encoded SSB in extracts of ssb-1 mutant cells Binding activity,t Genotype,ug SSBW nmol DNA Chromo- per mg bound per EOP Strain* some Plasmid protein mg protein (43 C) HMS91 ssb KLC850 ssb < pkac51 ssb < pkac50 ssb-1 F ssb peg310 ssb-1 F ssb < * pkac51 is the vector portion of pkac50 and peg310. peg310 was derived from pkac50 by mutation of the BamHI cleavage site. tdetermined by solid-phase radioimmunoassay of crude extracts using antibody to wild-type SSB. Although precise measurements of the quantity of F plasmid SSB produced by pkac50 have not been made, qualitative estimates have been made on the basis of sodium dodecyl sulfate/polyacrylamide gels of fractions analyzed during purification; it is interesting that more SSB was not detected. Despite extensive homology between F plasmid and E. coli SSB it is possible that antibody to E. coli SSB does not react well with the F plasmid protein; however, this point has not been directly examined. t ss DNA binding activity determined in crude extracts by method of Whittier and Chase (18). Efficiency of plating (EOP) at 43 C is the ratio of viable cells at 43 C to those at 30 C determined on LB agar plates without NaCl. Proc. Natl. Acad. Sci. USA 80 (1983) 5481 AvaIl Socl BomHI Bo/I AccI IT T I ATG SOf TGA Avall FIG. 1. Diagram of the portion of pkac50 sequenced in this study. The ATG start and TGA termination codons of the F plasmid ssf gene are indicated. Also indicated is the sequencing strategy (see Results). The arrows below the map indicate the direction ofsequencing and length of sequences determined in individual experiments. we also determined the location of Ava II cleavage sites in pkac50. Cleavage sites for other restriction enzymes utilized in the sequencing of the gene were found as sequences were determined, proceeding from the 5' termini of the Bal I site and the 3' termini of the BamHI site. The region of pkac50 whose sequence was determined and the sequencing strategy are diagrammed in Fig. 1. It was apparent when the first sequences were determined that extensive homology existed between the E. coli ssb (13) and F plasmid ssf genes, at least within the regions of the genes encoding the NH2 termini of the proteins. Further consideration of the implications of the similarities in sequence and structure of these ss DNA binding proteins are discussed below. The DNA and protein sequences of the F plasmid ssf and E. coli ssb genes and proteins are both presented in Fig. 2 for comparison.t The coding region of the F plasmid ssfgene is 537 base pairs. However, because the methionine of the initiation codon is not present in the protein, F plasmid SSB contains 178 amino acid residues-one more than E. coli SSB. Both SSBs utilize a UGA termination codon. The presumptive promoter sequences of the E. coli ssb and F plasmid ssf genes are not similar beyond their presumptive Shine-Dalgarno sequences (Fig. 3A). The coding region of the F plasmid ssf gene is followed by a dyad symmetry of 19 base pairs compared to 27 base pairs for the E. coli ssb gene (13) (Fig. 3B). The sequences of these regions are not similar. Protein Chemistry of F Plasmid SSB. Solid-phase sequence analysis of F plasmid SSB resulted in the identification of residues 2-6 and 8-14, all of which were in complete agreement with those predicted on the basis of the DNA sequence given in Fig. 2. Dansylation of 0.3 nmol of the protein revealed alanine as the NH2-terminal amino acid, also in agreement with the DNA sequence. Carboxypeptidase A digestion of 1 nmol of F plasmid SSB released 0.92 nmol of phenylalanine and no other amino acids, again in accord with the DNA sequence. The remainder of the amino acid sequence was confirmed by determining amino acid compositions for both the intact F plasmid SSB and its tryptic peptides. In addition, the sequences of three of the tryptic peptides were determined. The total amino acid composition of F plasmid SSB agrees well with the predicted composition (data not shown). Hydrolysis and analysis of performic acid-oxidized SSB and of tryptic peptide T-8 (Table 2) confirmed the finding that (in contrast to the E. coli SSB) the F plasmid protein does contain a single cysteine. A better confirmation of the DNA sequence for F plasmid SSB was obtained by isolating 14 of its tryptic peptides, which together account for 161 out of the 178 amino acids in this protein (Table 2). The amino acid compositions of these purified peptides as well as the sequences determined for T-2, T-7, and T-8 are all in ext Two errors in the E. coli ssb sequence (13) have now been corrected. The codon for serine-39 was reported to be TCA and has now been shown to be TCC. It should be noted that this change introduces a BstNI recognition site. Amino acid residue 133 was reported to be serine (codon AGC). This residue has now been correctly shown to be glycine (codon GGC). These corrections have been confirmed by both DNA and protein sequencing (unpublished data).
3 5482 Biochemistry: Chase et al. Proc. Natl. Acad. Sci. USA 80 (1983) F plasmid E. coli Ala Val Arg GCA GTT CGT C AGC A A Ser 10 Gly Ile Asn Lys GGC ATT AAC AAG Val Ile GTC ATC Lou CTC G A T T Va Gly Gly Ala Val Ala Asn Leu Gln Val Ala Thr Ser Glu Ser Trp GGG GGC GCA GTG GCA AAC CTG CAG GTG GCC ACG TCA GAA AGC TGG T T C A T AC C T T C TC Ile Thr Leu 60 Arg Val Val Len Phe Gly Lys CGC GTG GTG CTG TTC GGC AAG T A 90 Arg Ser Trp Glu Asp Asn Gly Ile Thr CGT AGC TGG GAG GAT **S AAC GGT ATC ACC AA ACC CAA TC CAG GAT Lys Thr Gln Ser Gin Asp 120 Ala Gly Ala Gln Thr Gln Pro GCA GGT GCT CAG ACT CAG CCG CAG G GGC G ** Gln Gly Gly Ala 70 Len Ala Glu Val Ala Gly Glu Cys CTC GCG GAA GTG GCA GGT GAA TGT G A G A C A Ser Tyr Arg Tyr Val Thr Glu CGT TAC GTC ACC GAA C AC A Thr Glu Glu Gly Gln Gln Phe Ser Gly GAA GAG GGG CAA CAG TTC AGC GGT C GT C A T ATC GGT G T Ala Gly Asn Ile Gly Gly Val Gly ArS Len Gly Lys Asp GTA GGA CGC CTG GGA AAG GAT T T AAT T C C Asn Gin Arg Asp Lys Gin Thr Gly Glu CGT GAC AAA CAG ACG GGG GAG T GC C C Ala Len Arg Lys Gly Ala Gln Val CTG CGC AAG GGC GCG CAG GTC T A T T T T Ser 100 Ile Leu ATT CTT G C G G Val Va1 130 Gln Pro CAG CCT G 1S0 160 Gly Ala Lys Thr Lys Gly Arg Gly Ar; Lys Ala Ala Gln Pro Gln Pro Gin Pro Gln Pro Pro Gin GGC GCA AAA ACG AAA GGC CGT GGA CGT AAG GCC GCG CAG CCG GAG CCT CAG CCG CAA CCG CCG GAG MT CAG TTC GC GGC GCG CAG TC CGC C G CA T C CT G GCA G CG T TAAC Asn Gln Ph. Ser Gly Gly Ala Gln Ser Ar; Pro Gln Ser Ala Ala Ala Pro Ser Asn 178 Asp Ile Pro Phe TERM GAT ATC CCG TTC TGA C T 20 Pro Giu Val Arg Tyr Ile Pro Asn CCG GAA GTC CGT TAC ATC CCC AAC A C G A T Net SO Met Arg Giu Gln Thr Gin Trp His ATG CGG GAG CAG ACA GAA TGG CAT AAA A T C Lys 80 Tyr Ile Glu Gly Gln Len Arg Thr TAC ATC GAA GGT CAG CTT CGC ACC T G T 110 Val Lys Thr Thr Gly Thr Met Gln Met Leu Val ArS Ala GTT AAG ACC ACG GGC ACC ATG CAG ATG CTG GTA CGT GCC G CGTT GGC GTG CGT Asn Val Gly Gly Gly Arg 140 Gin Pro Glu Pro Gln Ala Glu Ala Gly Thr Lys Lys Gly CAG CCG GAA CCA CAG GCG GAG GCC GGT ACG AAA AAA GGT GGC GT TGG GGT Gly Gly Trp Gly CAG CCT Gin Pro CAG CAG C Gln Gin Pro C G GGT Gin Gly C 170 Gly Asp Asp Tyr Gly Phe Ser Asp GGT GAC GAT TAC GGG TTT TCA GAC CCG CCG ATG G ** GAT T Pro Pro Met Asp Asp FIG. 2. Nucleotide and amino acid sequences of the F plasmid ssfand E. coli ssb genes and proteins. The complete sequences of F plasmid ssf are given and differences that exist in the E. coli sequences are listed below. At one place in the F plasmid ssf sequence and at two places in the E. coli ssb sequence a space equivalent to one codon has been introduced in order to maximize homology between the amino acid sequences. These regions are indicated by asterisks. F plasmid SSB contains 178 amino acid residues and E. coli SSB contains 177. TERM, termination codon. cellent agreement with the compositions expected from the DNA sequence. Carboxypeptidase A digestion of T-14 released 0.8 nmol of phenylalanine per mol of peptide and no other amino acids, thus confirming that this peptide is at the COOH terminus of the F plasmid SSB. A secondary structure for F plasmid SSB was predicted by an empirical method based on its amino acid sequence (Fig. 4). The first two-thirds of the protein (approximately residues 1-115) is highly ordered, with 70% of the amino acids predicted to occur in either a-helical or,3-pleated structures. In contrast, the COOH-terminal third of SSB has no predicted 13-pleated sheets and only three short stretches of a-helix, which together account for less than 30% of this region. While charged amino acids are distributed uniformly throughout the first two-thirds of the F plasmid SSB, the COOH-terminal third of the molecule contains both an unusually basic region (residues have a net charge of +7) and an unusually acidic region (residues have a net charge of -5). Overall, the F plasmid com- SSB should have a total net charge at ph 7 of only + 1 pared to -2 for E. coli SSB. DISCUSSION We determined the sequence of a gene from F plasmid that encodes a ss DNA binding protein and confirmed 90% of the predicted amino acid sequence by direct sequence determination or by amino acid analysis of tryptic peptides. The protein encoded by this gene has extensive homology to E. coli SSB. This is most evident in the NH2-terminal two-thirds of the protein (residues 1-115), where nearly 80% of the sequence exactly corresponds to the E. coli protein. Analysis of the secondary structure by the method of Chou and Fasman (17) predicts this region to be very similar to E. coli SSB (Fig. 4). Our previous studies utilizing proteolytic products of E. coli SSB have shown that this portion of the protein contains the DNA binding region (9). We have also shown that the ssb-1 mutation of A F plasmid GGTCCCCGCAAAACGTCAAAACC6C.UCTCCCGAAACTCAAAAIATIAT.AGGGGCTGAAAACATCAACAGAAMGA1ACACATCIO E. coli ACAACATAGTAAAAGCGCTATTfSTAATGGTACAATCGCGCGTTTACACTTATTCAGAACGATTTTTTTCA66AOACACGAACjj B F plasaid 5&CGACTGACTGTGACAACCGCCCC6TCCTGTGC6C CATCACCGGAGAGATGAGGATGAGCGAATATTTCAGAATACTTCAG E. col i STGTTGTCATTAAACAATAGGTTATATTGTTTTAAGGTGGATGATTAAAGCATCTGCCAGCCATAAAAAAGAAGCCTCCGTTAT FIG. 3. Comparison of the presumptive F plasmid ssfand E. coli ssb promoter (A) and terminator (B) sequences. The presumptive RNA polymerase recognition sequences, Pribnow sequences, and Shine-Dalgarno sequences are underlined. Those indicated for the E. coli ssb gene are as suggested previously (13). The dyad symmetry following the coding region of each gene is underlined. The ATG initiation codon and the termination codon of each gene are enclosed in boxes.
4 142 ~~~ , ' ~ _ ~~~~~~~~~~~~~ _ + C, F PLASMID SSB 140; 75 tt~ ' e ~~--- Q Ma le ;' L_ E. COLI SSB Biochemistry: Chase et al. Proc. Natl. Acad. Sci. USA 80 (1983) Table 2. Amino acid compositions* of F plasmid SSB tryptic peptides Amino acid T-1 T-2t T-3 T-4 T-5 T-6 T-7V T-8t T-9 T-10 T-11 T-12 T-13 T-14 Cys (1) Asx (1) 2.0 (2) 0.8 (1) (2) (4) Thr (1) 1.1 (1) 0.9 (1) (1) 1.1 (1) 2.8 (3) 2.1 (2) 0.3 Ser (2) (1) (1) 1.2 (1) Glx (1) 2.2 (2) 2.2 (2) 3.0 (3) (2) 2.9 (3) 1.4 (1) 1.2 (1) 1.2 (1) 10.5 (11) 4.8 (5) Pro (1) 1.1 (1) (4) 6.3(6) Gly (1) 1.0 (1) 2.7 (2) 1.1 (1) (1) 1.0 (1) 2.1 (2) 1.2 (1) (1) 4.1 (4) 2.4 (2) Ala 1.0 (1) (3) (2) 1.0 (1) (5) 2.1 (2) Val 1.1 (1) 1.7 (2) 0.9 (1) 1.8 (2) (2) 0.7 (1) 1.0 (1) (2) 1.2 (1) Met (1) (2) Ile (1) (1) (1) 0.8 (1) 0.9 (1) (1) Leu (1) 0.9 (1) 1.1 (1) (1) 1.8 (2) 1.1 (1) (1) 1.1 (1) Tyr (1) (1) (1) (1) Phe (1) (1) 1.6 (2) His (1) Lys (1) (1) (1) (1) (1) (1) 1.0 (1) Arg 1.0 (1) 1.3 (1) 1.1 (1) 1.1 (1) 1.2 (1) 1.2 (1) (1) 1.2 (1) 1.3 (1) (1) Trp - + (1) - + (1) - - _ +(1) Yield, % Residue numbers * Presented as residues per peptide molecule. Numbers in parentheses are calculated from the DNA sequence. t Identity confirmed by solid-phase sequence analysis of the peptide. tthese two peptides were isolated as a 60:40 ratio of T-7 to T-8. The sequence of the mixture was then determined, to confirm the identification. An additional 3.7 nmol (30%) of this peptide was recovered as T-14', residues E. coli SSB results in the substitution of tyrosine for histidine at position 55, resulting in the production of a protein defective in DNA binding (refs. 12 and 18 and unpublished results). This alteration should interrupt the extensive a-helical region predicted to occur between residues 45 and 67 of both proteins. These correlations are striking and further suggest the importance of this predicted a-helical region in DNA binding. Our previous characterization of proteolytic products of E. coli SSB produced by digestion with trypsin and chymotrypsin has led to the conclusion that regions of the protein in the area i ' QQ 16<l FIG. 4. Secondary structure of F plasmid SSB.9.Q,V1\, and denote a-helical, 3-pleated-sheet, and random-coil residues, respectively;,- structure turns are indicated by chain reversals. The + and - signs refer to charged residues (ph 7), and the numbers refer to residues at conformational boundaries.
5 5484 Biochemistry: Chase et A of residues are important in maintaining the integrity of its tetrameric structure and may also participate in interactions between tetramers that result in cooperative binding to ss DNA (9). Although only 7 of the 19 amino acids in this region (residues ) are identical in both the E. coh and F plasmid SSB, there is one stretch of 4 amino acids (residues ) that are homologous and that are predicted to form a 3- bend in both proteins. There is very little homology between the two proteins within their COOH-terminal regions. Between residues 136 and 170 only 4 residues of the F plasmid protein directly correspond to E. coli SSB. In addition, the F plasmid SSB contains 12 charged amino acids in this region compared to only 3 for the E. coli SSB. Even so, the difference in net charge is.only 3 because the region containing residues should have a net charge of +2 for the F plasmid SSB compared to -1 for the E. coli SSB. Although this entire region is devoid of extensive secondary structure;, a-helical regions are predicted to occur in the F plasmid SSB between residues and This differs from E. coli SSB, which is predicted to be completely random coil in this region except for several R-turns. It also differs in that the a-helical region predicted to occur in the E. coli protein between residues 166 and 173 is not predicted to occur in the F plasmid-encoded protein. It is striking, however, that 6 of the last 7 amino acids (residues ) of the F plasmid protein exactly correspond to the COOH-terminal sequence of E. coli SSB. It is important to note that the ssb-113 mutation of E. coli SSB results in the substitution of serine for proline- 176 (the next-to-the-last amino acid residue in the E. coli protein). This alteration does not greatly affect DNA binding as does the ssb-1 mutation (unpublished results) but does affect DNA replication, repair, and recombination in much the same manner as ssb-1, although with a number of subtle differences (4). The known effects of this alteration and the fact that the COOH-terminal amino acid sequences of both proteins are nearly identical strongly suggests that the integrity of this region is essential for the functioning of both proteins. On the basis of genetic and biochemical arguments, we have previously suggested that the pleiotropic effects exhibited by a deficiency in SSB could best be explained if the protein exerts its effects not only through binding-ss DNA but also by directly interacting with proteins intimately involved in DNA replication, recombination, and repair (4). We have suggested that the COOH terminus of the protein is the most likely region for such interactions (19), and we believe that the observations we have made on F plasmid SSB strengthen these arguments. The sequences immediately preceding the initiation codon of each gene are very similar; however, the sequences in the presumptive promoter and terminator regions of these genes are generally not homologous (Fig. 3) and they show minimal homology to the consensus sequence (20). The dyad symmetry that exists in the E. coli SSB promoter (13) is not present in the F plasmid gene. The significance of this dyad symmetry, if any, is still unknown. We speculated that it may be a recognition site for a regulator that affects transcription of ssb (13). In vivo studies in our laboratory (unpublished results) and recent studies of Salles et al. (21) have failed to produce any evidence for either positive or negative regulation of ssb. The question of ssb regulation in E. coli is particularly important due to the known involvement of SSB in induction of the SOS repair process and the proximity of the ssb and uvra promoters, the latter gene being a known damage inducible (din) locus (22). In vitro transcription or gene fusion studies of both genes are needed in order to evaluate their regulatory mechanisms. Because the F plasmid and E. coli ss DNA binding proteins almost certainly did not evolve independently, it is interesting that their control Proc. Natl. Acad. Sci. USA 80 (1983) regions are quite different, at least in sequence. It may be of basic importance that we understand their regulation in order to arrive at a complete understanding of their participation in biochemical reactions. It is important to note the absence.of the region of DNA from F plasmid encoding its ss DNA binding protein from mini-f plasmids capable of autonomous replication (23, 24). This observation suggests an additional role for F plasmid SSB apart from a role in replication that may. be compensated for by, the corresponding host protein. It has been noted by Kolodkin et al. (8) that certain other transmissible plasmids also produce an activity that suppresses the phenotype of ssb-1. Analysis of the. ss DNA binding proteins from these systems may lead to a better understanding of the interactions of various DNA metabolic proteins that is crucial to a complete understanding of DNA replication, recombination, and repair, as well as DNA transfer at the molecular level. It is a pleasure to acknowledge the free exchange of ideas between our laboratory and that of Dr. Brooks Low throughout this study. We are also grateful to Dr. William Konigsberg for his advice and encouragement during this work and to Janet Murphy and Elke Lorenson for their expert technical assistance. We especially thank Laura Dalheim for operating the Sequemat Mini-15 sequencer. This investigation was supported by Public Health Service Grants GM 11301, GM23451, and CA13330 (to J.W.C.), Public Health Service Grant GM12607 (to William H. Konigsberg), and National Science Foundation Grant PCM (to K.R.W.). J.W.C. is an established- investigator of the American Heart Association, Grant B. M. M. is a Predoctoral Fellow of the National Science Foundation. 1. Meyer, R. R., Glassberg, J. & Kornberg, A. (1979) Proc. Natl. Acad. Sci. USA 76, Glassberg, J., Meyer, R. & Kornberg, A. (1979)J. Bacteriol. 140, Golub, E. I. & Low, K. B. (1983) Proc. Natl. Acad. Sci. USA 80, Whittier, R. F. & Chase, J. W. (1983) Mol. Gen. Genet. 190, Lieberman, H. B. & Witkin, E. M. (1983) Mol. Gen. Genet. 190, Coleman, J. & Oakley, J. (1979) Crit. Rev. Riochem. 7, Falaschi, A., Cobianchi, F. & Riva, S. (1980) Trends Biochem. Sci. 5, Kolodkin, A. L., Capage, M. A., Golub, E. I. & Low, K. B. (1983) Proc. Natl. Acad. Sci. USA 80, Williams, K. R., Spicer, E. K., LoPresti, M. B., Guggenheimer, R. A. & Chase, J. W. (1983)J. Biol. Chem. 258, Chase, J., Whittier, R., Auerbach, J., Sancar, A. & Rupp, W. D. (1980) Nucleic Acids Res. 8, Maxam, A. & Gilbert, W. (1980) Methods Enzymol. 65, Williams, K. R., L'Italien, J. J., Guggenheimer, R. A., Sillerud, L., Spicer, E., Chase, J. W. & Konigsberg, W. (1982) in Methods in Protein Sequence Analysis, ed. Elzinga, M. (Humana, Clifton, NJ), pp Sancar, A., Williams, K. R., Chase, J. W. & Rupp, W. D. (1981) Proc. Natl. Acad. Sci. USA 78, Williams, K. R., LoPresti, M. B., Setoguchi, M. & Konigsberg, W. H. (1980) Proc. Natl. Acad. Sci. USA 77, L'Italien, J. R. & Laursen, R. A. (1982) in Methods in Protein Sequence Analysis, ed. Elzinga, M. (Humana, Clifton, NJ), pp Cohen, F. (1979) Dissertation (Yale Univ., New Haven, CT). 17. Chou, P. Y. & Fasman, G. D. (1978) Adv. Enzymol. 47, Whittier, R. F. & Chase, J. W. (1980) Anal. Biochern. 106, Chase, J. W., Murphy, J. B., Whittier, R. F., Lorensen, E. & Sninsky, J. J. (1983)J. Mol. Biol. 164, Rosenberg, M. & Court, D. (1980) Annu. Rev. Genet. 13, Salles, B., Paoletti, C. & Villani, G. (1983) Mol. Gen. Genet. 189, Kenyon, C. J. & Walker, G. C. (1981) Nature (London) 289, Timmis, K., Cabello, F. & Cohen, S. N. (1975) Proc. Nati. Acad. Sci. USA 72, Lovett, M. A. & Helinski, D. R. (1976)J. Bacteriol. 127,
Materials Protein synthesis kit. This kit consists of 24 amino acids, 24 transfer RNAs, four messenger RNAs and one ribosome (see below).
 Protein Synthesis Instructions The purpose of today s lab is to: Understand how a cell manufactures proteins from amino acids, using information stored in the genetic code. Assemble models of four very
Protein Synthesis Instructions The purpose of today s lab is to: Understand how a cell manufactures proteins from amino acids, using information stored in the genetic code. Assemble models of four very
Lecture 19A. DNA computing
 Lecture 19A. DNA computing What exactly is DNA (deoxyribonucleic acid)? DNA is the material that contains codes for the many physical characteristics of every living creature. Your cells use different
Lecture 19A. DNA computing What exactly is DNA (deoxyribonucleic acid)? DNA is the material that contains codes for the many physical characteristics of every living creature. Your cells use different
Disease and selection in the human genome 3
 Disease and selection in the human genome 3 Ka/Ks revisited Please sit in row K or forward RBFD: human populations, adaptation and immunity Neandertal Museum, Mettman Germany Sequence genome Measure expression
Disease and selection in the human genome 3 Ka/Ks revisited Please sit in row K or forward RBFD: human populations, adaptation and immunity Neandertal Museum, Mettman Germany Sequence genome Measure expression
Project 07/111 Final Report October 31, Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines
 Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
ORFs and genes. Please sit in row K or forward
 ORFs and genes Please sit in row K or forward https://www.flickr.com/photos/teseum/3231682806/in/photostream/ Question: why do some strains of Vibrio cause cholera and others don t? Methods Mechanisms
ORFs and genes Please sit in row K or forward https://www.flickr.com/photos/teseum/3231682806/in/photostream/ Question: why do some strains of Vibrio cause cholera and others don t? Methods Mechanisms
G+C content. 1 Introduction. 2 Chromosomes Topology & Counts. 3 Genome size. 4 Replichores and gene orientation. 5 Chirochores.
 1 Introduction 2 Chromosomes Topology & Counts 3 Genome size 4 Replichores and gene orientation 5 Chirochores 6 7 Codon usage 121 marc.bailly-bechet@univ-lyon1.fr Bacterial genome structures Introduction
1 Introduction 2 Chromosomes Topology & Counts 3 Genome size 4 Replichores and gene orientation 5 Chirochores 6 7 Codon usage 121 marc.bailly-bechet@univ-lyon1.fr Bacterial genome structures Introduction
NAME:... MODEL ANSWER... STUDENT NUMBER:... Maximum marks: 50. Internal Examiner: Hugh Murrell, Computer Science, UKZN
 COMP710, Bioinformatics with Julia, Test One, Thursday the 20 th of April, 2017, 09h30-11h30 1 NAME:...... MODEL ANSWER... STUDENT NUMBER:...... Maximum marks: 50 Internal Examiner: Hugh Murrell, Computer
COMP710, Bioinformatics with Julia, Test One, Thursday the 20 th of April, 2017, 09h30-11h30 1 NAME:...... MODEL ANSWER... STUDENT NUMBER:...... Maximum marks: 50 Internal Examiner: Hugh Murrell, Computer
Det matematisk-naturvitenskapelige fakultet
 UNIVERSITETET I OSLO Det matematisk-naturvitenskapelige fakultet Exam in: MBV4010 Arbeidsmetoder i molekylærbiologi og biokjemi I MBV4010 Methods in molecular biology and biochemistry I Day of exam: Friday
UNIVERSITETET I OSLO Det matematisk-naturvitenskapelige fakultet Exam in: MBV4010 Arbeidsmetoder i molekylærbiologi og biokjemi I MBV4010 Methods in molecular biology and biochemistry I Day of exam: Friday
Homework. A bit about the nature of the atoms of interest. Project. The role of electronega<vity
 Homework Why cited articles are especially useful. citeulike science citation index When cutting and pasting less is more. Project Your protein: I will mail these out this weekend If you haven t gotten
Homework Why cited articles are especially useful. citeulike science citation index When cutting and pasting less is more. Project Your protein: I will mail these out this weekend If you haven t gotten
Lecture 11: Gene Prediction
 Lecture 11: Gene Prediction Study Chapter 6.11-6.14 1 Gene: A sequence of nucleotides coding for protein Gene Prediction Problem: Determine the beginning and end positions of genes in a genome Where are
Lecture 11: Gene Prediction Study Chapter 6.11-6.14 1 Gene: A sequence of nucleotides coding for protein Gene Prediction Problem: Determine the beginning and end positions of genes in a genome Where are
 www.lessonplansinc.com Topic: Gene Mutations WS Summary: Students will learn about frame shift mutations and base substitution mutations. Goals & Objectives: Students will be able to demonstrate how mutations
www.lessonplansinc.com Topic: Gene Mutations WS Summary: Students will learn about frame shift mutations and base substitution mutations. Goals & Objectives: Students will be able to demonstrate how mutations
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR
 Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
National PHL TB DST Reference Center PSQ Reporting Language Table of Contents
 PSQ Reporting Language Table of Contents Document Page Number PSQ for Rifampin 2-6 Comparison table for rpob Codon Numbering 2 rpob mutation list (new numbering system) 3-5 rpob interpretations 6 PSQ for
PSQ Reporting Language Table of Contents Document Page Number PSQ for Rifampin 2-6 Comparison table for rpob Codon Numbering 2 rpob mutation list (new numbering system) 3-5 rpob interpretations 6 PSQ for
Codon Bias with PRISM. 2IM24/25, Fall 2007
 Codon Bias with PRISM 2IM24/25, Fall 2007 from RNA to protein mrna vs. trna aminoacid trna anticodon mrna codon codon-anticodon matching Watson-Crick base pairing A U and C G binding first two nucleotide
Codon Bias with PRISM 2IM24/25, Fall 2007 from RNA to protein mrna vs. trna aminoacid trna anticodon mrna codon codon-anticodon matching Watson-Crick base pairing A U and C G binding first two nucleotide
Protein Structure Analysis
 BINF 731 Protein Structure Analysis http://binf.gmu.edu/vaisman/binf731/ Iosif Vaisman COMPUTATIONAL BIOLOGY COMPUTATIONAL STRUCTURAL BIOLOGY COMPUTATIONAL MOLECULAR BIOLOGY BIOINFORMATICS STRUCTURAL BIOINFORMATICS
BINF 731 Protein Structure Analysis http://binf.gmu.edu/vaisman/binf731/ Iosif Vaisman COMPUTATIONAL BIOLOGY COMPUTATIONAL STRUCTURAL BIOLOGY COMPUTATIONAL MOLECULAR BIOLOGY BIOINFORMATICS STRUCTURAL BIOINFORMATICS
Expression of Recombinant Proteins
 Expression of Recombinant Proteins Uses of Cloned Genes sequencing reagents (eg, probes) protein production insufficient natural quantities modify/mutagenesis library screening Expression Vector Features
Expression of Recombinant Proteins Uses of Cloned Genes sequencing reagents (eg, probes) protein production insufficient natural quantities modify/mutagenesis library screening Expression Vector Features
BIOSTAT516 Statistical Methods in Genetic Epidemiology Autumn 2005 Handout1, prepared by Kathleen Kerr and Stephanie Monks
 Rationale of Genetic Studies Some goals of genetic studies include: to identify the genetic causes of phenotypic variation develop genetic tests o benefits to individuals and to society are still uncertain
Rationale of Genetic Studies Some goals of genetic studies include: to identify the genetic causes of phenotypic variation develop genetic tests o benefits to individuals and to society are still uncertain
Supplementary. Table 1: Oligonucleotides and Plasmids. complementary to positions from 77 of the SRα '- GCT CTA GAG AAC TTG AAG TAC AGA CTG C
 Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
Supplemental Data Supplemental Figure 1.
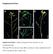 Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Lezione 10. Bioinformatica. Mauro Ceccanti e Alberto Paoluzzi
 Lezione 10 Bioinformatica Mauro Ceccanti e Alberto Paoluzzi Dip. Informatica e Automazione Università Roma Tre Dip. Medicina Clinica Università La Sapienza Lezione 10: Sintesi proteica Synthesis of proteins
Lezione 10 Bioinformatica Mauro Ceccanti e Alberto Paoluzzi Dip. Informatica e Automazione Università Roma Tre Dip. Medicina Clinica Università La Sapienza Lezione 10: Sintesi proteica Synthesis of proteins
RPA-AB RPA-C Supplemental Figure S1: SDS-PAGE stained with Coomassie Blue after protein purification.
 RPA-AB RPA-C (a) (b) (c) (d) (e) (f) Supplemental Figure S: SDS-PAGE stained with Coomassie Blue after protein purification. (a) RPA; (b) RPA-AB; (c) RPA-CDE; (d) RPA-CDE core; (e) RPA-DE; and (f) RPA-C
RPA-AB RPA-C (a) (b) (c) (d) (e) (f) Supplemental Figure S: SDS-PAGE stained with Coomassie Blue after protein purification. (a) RPA; (b) RPA-AB; (c) RPA-CDE; (d) RPA-CDE core; (e) RPA-DE; and (f) RPA-C
Protein Synthesis. Application Based Questions
 Protein Synthesis Application Based Questions MRNA Triplet Codons Note: Logic behind the single letter abbreviations can be found at: http://www.biology.arizona.edu/biochemistry/problem_sets/aa/dayhoff.html
Protein Synthesis Application Based Questions MRNA Triplet Codons Note: Logic behind the single letter abbreviations can be found at: http://www.biology.arizona.edu/biochemistry/problem_sets/aa/dayhoff.html
Primer Design Workshop. École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria
 Primer Design Workshop École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria Scenario You have discovered the presence of a novel endophy5c organism living inside the cells
Primer Design Workshop École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria Scenario You have discovered the presence of a novel endophy5c organism living inside the cells
for Programmed Chemo-enzymatic Synthesis of Antigenic Oligosaccharides
 Supporting Information Design of α-transglucosidases of Controlled Specificity for Programmed Chemo-enzymatic Synthesis of Antigenic Oligosaccharides Elise Champion ±,,,, Isabelle André ±,,, Claire Moulis
Supporting Information Design of α-transglucosidases of Controlled Specificity for Programmed Chemo-enzymatic Synthesis of Antigenic Oligosaccharides Elise Champion ±,,,, Isabelle André ±,,, Claire Moulis
Table S1. Bacterial strains (Related to Results and Experimental Procedures)
 Table S1. Bacterial strains (Related to Results and Experimental Procedures) Strain number Relevant genotype Source or reference 1045 AB1157 Graham Walker (Donnelly and Walker, 1989) 2458 3084 (MG1655)
Table S1. Bacterial strains (Related to Results and Experimental Procedures) Strain number Relevant genotype Source or reference 1045 AB1157 Graham Walker (Donnelly and Walker, 1989) 2458 3084 (MG1655)
Supporting information for Biochemistry, 1995, 34(34), , DOI: /bi00034a013
 Supporting information for Biochemistry, 1995, 34(34), 10807 10815, DOI: 10.1021/bi00034a013 LESNIK 10807-1081 Terms & Conditions Electronic Supporting Information files are available without a subscription
Supporting information for Biochemistry, 1995, 34(34), 10807 10815, DOI: 10.1021/bi00034a013 LESNIK 10807-1081 Terms & Conditions Electronic Supporting Information files are available without a subscription
Supplementary Information. Construction of Lasso Peptide Fusion Proteins
 Supplementary Information Construction of Lasso Peptide Fusion Proteins Chuhan Zong 1, Mikhail O. Maksimov 2, A. James Link 2,3 * Departments of 1 Chemistry, 2 Chemical and Biological Engineering, and
Supplementary Information Construction of Lasso Peptide Fusion Proteins Chuhan Zong 1, Mikhail O. Maksimov 2, A. James Link 2,3 * Departments of 1 Chemistry, 2 Chemical and Biological Engineering, and
FROM DNA TO GENETIC GENEALOGY Stephen P. Morse
 1. GENES, CHROMOSOMES, AND DNA Chromosomes FROM DNA TO GENETIC GENEALOGY Stephen P. Morse (steve@stevemorse.org) Every human cell = 46 chromosomes (1 to 22 in pairs, 2 sex chromosomes) Male: sex chromosomes
1. GENES, CHROMOSOMES, AND DNA Chromosomes FROM DNA TO GENETIC GENEALOGY Stephen P. Morse (steve@stevemorse.org) Every human cell = 46 chromosomes (1 to 22 in pairs, 2 sex chromosomes) Male: sex chromosomes
Supplemental material
 Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC
 Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
1. DNA, RNA structure. 2. DNA replication. 3. Transcription, translation
 1. DNA, RNA structure 2. DNA replication 3. Transcription, translation DNA and RNA are polymers of nucleotides DNA is a nucleic acid, made of long chains of nucleotides Nucleotide Phosphate group Nitrogenous
1. DNA, RNA structure 2. DNA replication 3. Transcription, translation DNA and RNA are polymers of nucleotides DNA is a nucleic acid, made of long chains of nucleotides Nucleotide Phosphate group Nitrogenous
Supporting Information
 Supporting Information Barderas et al. 10.1073/pnas.0801221105 SI Text: Docking of gastrin to Constructed scfv Models Interactive predocking of the 4-WL-5 motif into the central pocket observed in the
Supporting Information Barderas et al. 10.1073/pnas.0801221105 SI Text: Docking of gastrin to Constructed scfv Models Interactive predocking of the 4-WL-5 motif into the central pocket observed in the
Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis
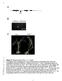 1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
Multiplexing Genome-scale Engineering
 Multiplexing Genome-scale Engineering Harris Wang, Ph.D. Department of Systems Biology Department of Pathology & Cell Biology http://wanglab.c2b2.columbia.edu Rise of Genomics An Expanding Toolbox Esvelt
Multiplexing Genome-scale Engineering Harris Wang, Ph.D. Department of Systems Biology Department of Pathology & Cell Biology http://wanglab.c2b2.columbia.edu Rise of Genomics An Expanding Toolbox Esvelt
Supplemental Data. mir156-regulated SPL Transcription. Factors Define an Endogenous Flowering. Pathway in Arabidopsis thaliana
 Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
Hes6. PPARα. PPARγ HNF4 CD36
 SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
An engineered tryptophan zipper-type peptide as a molecular recognition scaffold
 SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/10/494/eaan6284/dc1 Supplementary Materials for Activation of master virulence regulator PhoP in acidic ph requires the Salmonella-specific protein UgtL Jeongjoon
www.sciencesignaling.org/cgi/content/full/10/494/eaan6284/dc1 Supplementary Materials for Activation of master virulence regulator PhoP in acidic ph requires the Salmonella-specific protein UgtL Jeongjoon
PROTEIN SYNTHESIS Study Guide
 PART A. Read the following: PROTEIN SYNTHESIS Study Guide Protein synthesis is the process used by the body to make proteins. The first step of protein synthesis is called Transcription. It occurs in the
PART A. Read the following: PROTEIN SYNTHESIS Study Guide Protein synthesis is the process used by the body to make proteins. The first step of protein synthesis is called Transcription. It occurs in the
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells
 Supplementary Information for: PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells Ju Hye Jang 1, Hyun Kim 2, Mi Jung Jang 2, Ju Hyun Cho 1,2,* 1 Research Institute
Supplementary Information for: PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells Ju Hye Jang 1, Hyun Kim 2, Mi Jung Jang 2, Ju Hyun Cho 1,2,* 1 Research Institute
Cat. # Product Size DS130 DynaExpress TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1
 Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Genomics and Gene Recognition Genes and Blue Genes
 Genomics and Gene Recognition Genes and Blue Genes November 1, 2004 Prokaryotic Gene Structure prokaryotes are simplest free-living organisms studying prokaryotes can give us a sense what is the minimum
Genomics and Gene Recognition Genes and Blue Genes November 1, 2004 Prokaryotic Gene Structure prokaryotes are simplest free-living organisms studying prokaryotes can give us a sense what is the minimum
INTRODUCTION TO THE MOLECULAR GENETICS OF THE COLOR MUTATIONS IN ROCK POCKET MICE
 The Making of the The Fittest: Making of the Fittest Natural Selection Natural and Adaptation Selection and Adaptation Educator Materials TEACHER MATERIALS INTRODUCTION TO THE MOLECULAR GENETICS OF THE
The Making of the The Fittest: Making of the Fittest Natural Selection Natural and Adaptation Selection and Adaptation Educator Materials TEACHER MATERIALS INTRODUCTION TO THE MOLECULAR GENETICS OF THE
Arabidopsis actin depolymerizing factor AtADF4 mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB
 Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
Basic concepts of molecular biology
 Basic concepts of molecular biology Gabriella Trucco Email: gabriella.trucco@unimi.it Life The main actors in the chemistry of life are molecules called proteins nucleic acids Proteins: many different
Basic concepts of molecular biology Gabriella Trucco Email: gabriella.trucco@unimi.it Life The main actors in the chemistry of life are molecules called proteins nucleic acids Proteins: many different
SUPPLEMENTARY MATERIALS AND METHODS. E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1)
 SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
(a) Which enzyme(s) make 5' - 3' phosphodiester bonds? (c) Which enzyme(s) make single-strand breaks in DNA backbones?
 EXAMPLE QUESTIONS AND ANSWERS 1. Topoisomerase does which one of the following? (a) Makes new DNA strands. (b) Unties knots in DNA molecules. (c) Joins the ends of double-stranded DNA molecules. (d) Is
EXAMPLE QUESTIONS AND ANSWERS 1. Topoisomerase does which one of the following? (a) Makes new DNA strands. (b) Unties knots in DNA molecules. (c) Joins the ends of double-stranded DNA molecules. (d) Is
Level 2 Biology, 2017
 91159 911590 2SUPERVISOR S Level 2 Biology, 2017 91159 Demonstrate understanding of gene expression 2.00 p.m. Wednesday 22 November 2017 Credits: Four Achievement Achievement with Merit Achievement with
91159 911590 2SUPERVISOR S Level 2 Biology, 2017 91159 Demonstrate understanding of gene expression 2.00 p.m. Wednesday 22 November 2017 Credits: Four Achievement Achievement with Merit Achievement with
Biomolecules: lecture 6
 Biomolecules: lecture 6 - to learn the basics on how DNA serves to make RNA = transcription - to learn how the genetic code instructs protein synthesis - to learn the basics on how proteins are synthesized
Biomolecules: lecture 6 - to learn the basics on how DNA serves to make RNA = transcription - to learn how the genetic code instructs protein synthesis - to learn the basics on how proteins are synthesized
Degenerate Code. Translation. trna. The Code is Degenerate trna / Proofreading Ribosomes Translation Mechanism
 Translation The Code is Degenerate trna / Proofreading Ribosomes Translation Mechanism Degenerate Code There are 64 possible codon triplets There are 20 naturally-encoding amino acids Several codons specify
Translation The Code is Degenerate trna / Proofreading Ribosomes Translation Mechanism Degenerate Code There are 64 possible codon triplets There are 20 naturally-encoding amino acids Several codons specify
Dierks Supplementary Fig. S1
 Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
Genomic Sequence Analysis using Electron-Ion Interaction
 University of Aizu, Graduation Thesis. March, 25 s1985 1 Genomic Sequence Analysis using Electron-Ion Interaction Potential Masumi Kobayashi s1985 Supervised by Hiroshi Toyoizumi Abstract This paper proposes
University of Aizu, Graduation Thesis. March, 25 s1985 1 Genomic Sequence Analysis using Electron-Ion Interaction Potential Masumi Kobayashi s1985 Supervised by Hiroshi Toyoizumi Abstract This paper proposes
iclicker Question #28B - after lecture Shown below is a diagram of a typical eukaryotic gene which encodes a protein: start codon stop codon 2 3
 Bio 111 Handout for Molecular Biology 4 This handout contains: Today s iclicker Questions Information on Exam 3 Solutions Fall 2008 Exam 3 iclicker Question #28A - before lecture Which of the following
Bio 111 Handout for Molecular Biology 4 This handout contains: Today s iclicker Questions Information on Exam 3 Solutions Fall 2008 Exam 3 iclicker Question #28A - before lecture Which of the following
strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular
![strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular](/thumbs/72/66361212.jpg) Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Sequences of the ssb gene and protein (DNA sequence/helix-destabilizing protein/tryptic peptides/secondary structure/domains)
 Proc. NatL Acad. Sci. USA Vol. 78, No. 7, pp. 4274-4278, July 1981 Biochemistry Sequences of the ssb gene and protein (DNA sequence/helix-destabilizing protein/tryptic peptides/secondary structure/domains)
Proc. NatL Acad. Sci. USA Vol. 78, No. 7, pp. 4274-4278, July 1981 Biochemistry Sequences of the ssb gene and protein (DNA sequence/helix-destabilizing protein/tryptic peptides/secondary structure/domains)
How life. constructs itself.
 How life constructs itself Life constructs itself using few simple rules of information processing. On the one hand, there is a set of rules determining how such basic chemical reactions as transcription,
How life constructs itself Life constructs itself using few simple rules of information processing. On the one hand, there is a set of rules determining how such basic chemical reactions as transcription,
ΔPDD1 x ΔPDD1. ΔPDD1 x wild type. 70 kd Pdd1. Pdd3
 Supplemental Fig. S1 ΔPDD1 x wild type ΔPDD1 x ΔPDD1 70 kd Pdd1 50 kd 37 kd Pdd3 Supplemental Fig. S1. ΔPDD1 strains express no detectable Pdd1 protein. Western blot analysis of whole-protein extracts
Supplemental Fig. S1 ΔPDD1 x wild type ΔPDD1 x ΔPDD1 70 kd Pdd1 50 kd 37 kd Pdd3 Supplemental Fig. S1. ΔPDD1 strains express no detectable Pdd1 protein. Western blot analysis of whole-protein extracts
7.016 Problem Set 3. 1 st Pedigree
 7.016 Problem Set 3 Question 1 The following human pedigree shows the inheritance pattern of a specific disease within a family. Assume that the individuals marrying into the family for all generations
7.016 Problem Set 3 Question 1 The following human pedigree shows the inheritance pattern of a specific disease within a family. Assume that the individuals marrying into the family for all generations
Genes and Proteins. Objectives
 Genes and Proteins Lecture 15 Objectives At the end of this series of lectures, you should be able to: Define terms. Explain the central dogma of molecular biology. Describe the locations, reactants, and
Genes and Proteins Lecture 15 Objectives At the end of this series of lectures, you should be able to: Define terms. Explain the central dogma of molecular biology. Describe the locations, reactants, and
Supplementary Figure 1A A404 Cells +/- Retinoic Acid
 Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Chapter 17 Genes to Proteins Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. The following questions refer to Figure 17.1, a simple metabolic
Exam Chapter 17 Genes to Proteins Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. The following questions refer to Figure 17.1, a simple metabolic
The combination of a phosphate, sugar and a base forms a compound called a nucleotide.
 History Rosalin Franklin: Female scientist (x-ray crystallographer) who took the picture of DNA James Watson and Francis Crick: Solved the structure of DNA from information obtained by other scientist.
History Rosalin Franklin: Female scientist (x-ray crystallographer) who took the picture of DNA James Watson and Francis Crick: Solved the structure of DNA from information obtained by other scientist.
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Ch 17 Practice Questions MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Garrod hypothesized that "inborn errors of metabolism" such as alkaptonuria
Ch 17 Practice Questions MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Garrod hypothesized that "inborn errors of metabolism" such as alkaptonuria
Chemistry 121 Winter 17
 Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
PCR analysis was performed to show the presence and the integrity of the var1csa and var-
 Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Y-chromosomal haplogroup typing Using SBE reaction
 Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
CHE-3H84: Protein Engineering Past Exam Papers
 CHE-3H84: Protein Engineering Past Exam Papers Sorted by Topic then Year Knowledge-Based Engineering of Proteins, Large Scale Production of Recombinant Proteins, and Protein Purification Dr. Hemmings 2006/7
CHE-3H84: Protein Engineering Past Exam Papers Sorted by Topic then Year Knowledge-Based Engineering of Proteins, Large Scale Production of Recombinant Proteins, and Protein Purification Dr. Hemmings 2006/7
ANCIENT BACTERIA? 250 million years later, scientists revive life forms
 ANCIENT BACTERIA? 250 million years later, scientists revive life forms Thursday, October 19, 2000 U.S. researchers say they have revived bacteria that have been dormant for more then 250 million years,
ANCIENT BACTERIA? 250 million years later, scientists revive life forms Thursday, October 19, 2000 U.S. researchers say they have revived bacteria that have been dormant for more then 250 million years,
Overexpression Normal expression Overexpression Normal expression. 26 (21.1%) N (%) P-value a N (%)
 SUPPLEMENTARY TABLES Table S1. Alteration of ZNF322A protein expression levels in relation to clinicopathological parameters in 123 Asian and 74 Caucasian lung cancer patients. Asian patients Caucasian
SUPPLEMENTARY TABLES Table S1. Alteration of ZNF322A protein expression levels in relation to clinicopathological parameters in 123 Asian and 74 Caucasian lung cancer patients. Asian patients Caucasian
Creation of A Caspese-3 Sensing System Using A Combination of Split- GFP and Split-Intein
 Supplementary Information Creation of A Caspese-3 Sensing System Using A Combination of Split- GFP and Split-Intein Seiji Sakamoto,* Mika Terauchi, Anna Hugo, Tanner Kim, Yasuyuki Araki and Takehiko Wada*
Supplementary Information Creation of A Caspese-3 Sensing System Using A Combination of Split- GFP and Split-Intein Seiji Sakamoto,* Mika Terauchi, Anna Hugo, Tanner Kim, Yasuyuki Araki and Takehiko Wada*
A Zero-Knowledge Based Introduction to Biology
 A Zero-Knowledge Based Introduction to Biology Konstantinos (Gus) Katsiapis 25 Sep 2009 Thanks to Cory McLean and George Asimenos Cells: Building Blocks of Life cell, membrane, cytoplasm, nucleus, mitochondrion
A Zero-Knowledge Based Introduction to Biology Konstantinos (Gus) Katsiapis 25 Sep 2009 Thanks to Cory McLean and George Asimenos Cells: Building Blocks of Life cell, membrane, cytoplasm, nucleus, mitochondrion
CONVERGENT EVOLUTION. Def n acquisition of some biological trait but different lineages
 CONVERGENT EVOLUTION Def n acquisition of some biological trait but different lineages Living Rock cactus Baseball plant THE QUESTION From common ancestor or independent acquisition? By Lineage By Convergence
CONVERGENT EVOLUTION Def n acquisition of some biological trait but different lineages Living Rock cactus Baseball plant THE QUESTION From common ancestor or independent acquisition? By Lineage By Convergence
MacBlunt PCR Cloning Kit Manual
 MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
MCB421 FALL2005 EXAM#3 ANSWERS Page 1 of 12. ANSWER: Both transposon types form small duplications of adjacent host DNA sequences.
 Page 1 of 12 (10pts) 1. There are two mechanisms for transposition used by bacterial transposable elements: replicative (Tn3) and non-replicative (Tn5 and Tn10). Compare and contrast the two mechanisms
Page 1 of 12 (10pts) 1. There are two mechanisms for transposition used by bacterial transposable elements: replicative (Tn3) and non-replicative (Tn5 and Tn10). Compare and contrast the two mechanisms
Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were
 1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
Supporting Online Information
 Supporting Online Information Isolation of Human Genomic DNA Sequences with Expanded Nucleobase Selectivity Preeti Rathi, Sara Maurer, Grzegorz Kubik and Daniel Summerer* Department of Chemistry and Chemical
Supporting Online Information Isolation of Human Genomic DNA Sequences with Expanded Nucleobase Selectivity Preeti Rathi, Sara Maurer, Grzegorz Kubik and Daniel Summerer* Department of Chemistry and Chemical
Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR
 Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer
 TEACHER S GUIDE SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer SYNOPSIS This activity uses the metaphor of decoding a secret message for the Protein Synthesis process. Students teach themselves
TEACHER S GUIDE SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer SYNOPSIS This activity uses the metaphor of decoding a secret message for the Protein Synthesis process. Students teach themselves
2
 1 2 3 4 5 6 7 Supplemental Table 1. Magnaporthe oryzae strains generated in this study. Strain background Genotype Strain name Description Guy-11 H1:RFP H1:RFP Strain expressing Histone H1- encoding gene
1 2 3 4 5 6 7 Supplemental Table 1. Magnaporthe oryzae strains generated in this study. Strain background Genotype Strain name Description Guy-11 H1:RFP H1:RFP Strain expressing Histone H1- encoding gene
Important points from last time
 Important points from last time Subst. rates differ site by site Fit a Γ dist. to variation in rates Γ generally has two parameters but in biology we fix one to ensure a mean equal to 1 and the other parameter
Important points from last time Subst. rates differ site by site Fit a Γ dist. to variation in rates Γ generally has two parameters but in biology we fix one to ensure a mean equal to 1 and the other parameter
Describe the features of a gene which enable it to code for a particular protein.
 1. Answers should be written in continuous prose. Credit will be given for biological accuracy, the organisation and presentation of the information and the way in which the answer is expressed. Cancer
1. Answers should be written in continuous prose. Credit will be given for biological accuracy, the organisation and presentation of the information and the way in which the answer is expressed. Cancer
Thr Gly Tyr. Gly Lys Asn
 Your unique body characteristics (traits), such as hair color or blood type, are determined by the proteins your body produces. Proteins are the building blocks of life - in fact, about 45% of the human
Your unique body characteristics (traits), such as hair color or blood type, are determined by the proteins your body produces. Proteins are the building blocks of life - in fact, about 45% of the human
A Circular Code in the Protein Coding Genes of Mitochondria
 J. theor. Biol. (1997) 189, 273 290 A Circular Code in the Protein Coding Genes of Mitochondria DIDIER G. ARQUE` S* AND CHRISTIAN J. MICHEL *Equipe de Biologie The orique, Universite de Marne la Valle
J. theor. Biol. (1997) 189, 273 290 A Circular Code in the Protein Coding Genes of Mitochondria DIDIER G. ARQUE` S* AND CHRISTIAN J. MICHEL *Equipe de Biologie The orique, Universite de Marne la Valle
Basic concepts of molecular biology
 Basic concepts of molecular biology Gabriella Trucco Email: gabriella.trucco@unimi.it What is life made of? 1665: Robert Hooke discovered that organisms are composed of individual compartments called cells
Basic concepts of molecular biology Gabriella Trucco Email: gabriella.trucco@unimi.it What is life made of? 1665: Robert Hooke discovered that organisms are composed of individual compartments called cells
Supporting Information. Electrochemiluminescence Peptide-Based Biosensor with Hetero-
 Supporting Information Electrochemiluminescence Peptide-Based Biosensor with Hetero- Nanostructures as Co-reaction Accelerator for the Ultra-sensitive Determination of Tryptase Fang-Fang Wu, Ying Zhou,
Supporting Information Electrochemiluminescence Peptide-Based Biosensor with Hetero- Nanostructures as Co-reaction Accelerator for the Ultra-sensitive Determination of Tryptase Fang-Fang Wu, Ying Zhou,
Biomolecules: lecture 6
 Biomolecules: lecture 6 - to learn the basics on how DNA serves to make RNA = transcription - to learn how the genetic code instructs protein synthesis - to learn the basics on how proteins are synthesized
Biomolecules: lecture 6 - to learn the basics on how DNA serves to make RNA = transcription - to learn how the genetic code instructs protein synthesis - to learn the basics on how proteins are synthesized
DNA Begins the Process
 Biology I D N A DNA contains genes, sequences of nucleotide bases These Genes code for polypeptides (proteins) Proteins are used to build cells and do much of the work inside cells DNA Begins the Process
Biology I D N A DNA contains genes, sequences of nucleotide bases These Genes code for polypeptides (proteins) Proteins are used to build cells and do much of the work inside cells DNA Begins the Process
Table S1. Sequences of mutagenesis primers used to create altered rdpa- and sdpa genes
 Supplementary Table and Figures for Structural Basis for the Enantiospecificities of R- and S-Specific Phenoxypropionate/α-Ketoglutarate Dioxygenases by Tina A. Müller, Maria I. Zavodszky, Michael Feig,
Supplementary Table and Figures for Structural Basis for the Enantiospecificities of R- and S-Specific Phenoxypropionate/α-Ketoglutarate Dioxygenases by Tina A. Müller, Maria I. Zavodszky, Michael Feig,
Sampling Random Bioinformatics Puzzles using Adaptive Probability Distributions
 Sampling Random Bioinformatics Puzzles using Adaptive Probability Distributions Christian Theil Have 1, Emil Vincent Appel 1, Jette Bork-Jensen 1, and Ole Torp Lassen 2 1 Novo Nordisk Foundation Center
Sampling Random Bioinformatics Puzzles using Adaptive Probability Distributions Christian Theil Have 1, Emil Vincent Appel 1, Jette Bork-Jensen 1, and Ole Torp Lassen 2 1 Novo Nordisk Foundation Center
Supplemental Table 1. Primers used for PCR.
 Supplemental Table 1. Primers used for PCR. Gene Type Primer Sequence Genotyping and semi-quantitative RT-PCR F 5 -TTG CCC GAT CAC CAT CTG TA-3 rwa1-1 R 5 -TGT AGC GAT CAA GGC CTG ATC TAA-3 LB 5 -TAG CAT
Supplemental Table 1. Primers used for PCR. Gene Type Primer Sequence Genotyping and semi-quantitative RT-PCR F 5 -TTG CCC GAT CAC CAT CTG TA-3 rwa1-1 R 5 -TGT AGC GAT CAA GGC CTG ATC TAA-3 LB 5 -TAG CAT
Supplemental Data. Bennett et al. (2010). Plant Cell /tpc
 BRN1 ---------MSSSNGGVPPGFRFHPTDEELLHYYLKKKISYEKFEMEVIKEVDLNKIEPWDLQDRCKIGSTPQNEWYFFSHKDRKYPTGS 81 BRN2 --------MGSSSNGGVPPGFRFHPTDEELLHYYLKKKISYQKFEMEVIREVDLNKLEPWDLQERCKIGSTPQNEWYFFSHKDRKYPTGS 82 SMB
BRN1 ---------MSSSNGGVPPGFRFHPTDEELLHYYLKKKISYEKFEMEVIKEVDLNKIEPWDLQDRCKIGSTPQNEWYFFSHKDRKYPTGS 81 BRN2 --------MGSSSNGGVPPGFRFHPTDEELLHYYLKKKISYQKFEMEVIREVDLNKLEPWDLQERCKIGSTPQNEWYFFSHKDRKYPTGS 82 SMB
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Investigation of the Biosynthesis of the Lasso Peptide Chaxapeptin Using an E. coli-based Production System Helena Martin-Gómez, Uwe Linne, Fernando Albericio, Judit Tulla-Puche,*
SUPPORTING INFORMATION Investigation of the Biosynthesis of the Lasso Peptide Chaxapeptin Using an E. coli-based Production System Helena Martin-Gómez, Uwe Linne, Fernando Albericio, Judit Tulla-Puche,*
Nucleotide Sequence of the Salmonella typhimurium metr Gene and
 JOURNAL OF BACTERIOLOGY, Sept. 1987, p. 3932-3937 Vol. 169, No. 9 0021-9193/87/093932-06$02.00/0 Copyright 1987, American Society for Microbiology Nucleotide Sequence of the Salmonella typhimurium metr
JOURNAL OF BACTERIOLOGY, Sept. 1987, p. 3932-3937 Vol. 169, No. 9 0021-9193/87/093932-06$02.00/0 Copyright 1987, American Society for Microbiology Nucleotide Sequence of the Salmonella typhimurium metr
TRANSCRIPTION. Renáta Schipp
 TRANSCRIPTION Renáta Schipp Gene expression Gene expression: - is the process by which information from a gene is used for the synthesis of gene products. These products are proteins, but in the case of
TRANSCRIPTION Renáta Schipp Gene expression Gene expression: - is the process by which information from a gene is used for the synthesis of gene products. These products are proteins, but in the case of
Gene synthesis by circular assembly amplification
 Gene synthesis by circular assembly amplification Duhee Bang & George M Church Supplementary figures and text: Supplementary Figure 1. Dpo4 gene (1.05kb) construction by various methods. Supplementary
Gene synthesis by circular assembly amplification Duhee Bang & George M Church Supplementary figures and text: Supplementary Figure 1. Dpo4 gene (1.05kb) construction by various methods. Supplementary
DNA.notebook March 08, DNA Overview
 DNA Overview Deoxyribonucleic Acid, or DNA, must be able to do 2 things: 1) give instructions for building and maintaining cells. 2) be copied each time a cell divides. DNA is made of subunits called nucleotides
DNA Overview Deoxyribonucleic Acid, or DNA, must be able to do 2 things: 1) give instructions for building and maintaining cells. 2) be copied each time a cell divides. DNA is made of subunits called nucleotides
Legends for supplementary figures 1-3
 High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
