NEWSLETTERNEWSLETTERNEW
|
|
|
- Elijah Marsh
- 6 years ago
- Views:
Transcription
1 NEWSLETTERNEWSLETTERNEW ACuteTox April Research Project for Alternative Testing CONTACT: CECILIA CLEMEDSON, DEPT. OF NEUROCHEM., STOCKHOLM UNIV., SE STOCKHOLM, SWEDEN, CECILIA.CLEMEDSON@NEUROCHEM.SU.SE The ACuteTox prevalidation phase has started As a result of the below described statistical analysis, potential assays for the ACuteTox testing strategy have been selected (see Table 1) according to their reproducibility and reliability as well as to their potential to classify chemicals into the official acute oral toxicity classes (GHS 1 global harmonisation system and CLP Regulation in EU). The selected tests are w challenged with a new set of 32 coded chemicals in a pre-validation study. During this last phase of the ACuteTox project the main focus will be on the assessment of the predictive capacity of three proposed tiered testing strategies and the identification of the combination that gives the best prediction. What is the ACuteTox project? ACuteTox is an integrated project funded by the EU 6FP, with the aim to develop and pre-validate a simple and robust testing strategy based on in vitro and computer models for the prediction of acute oral systemic toxicity and classification of chemicals into the different EU and GHS 1 toxicity classes. Fast, cheaper and scientifically better tests for testing chemicals, cosmetics, and other products are urgently needed to replace the animal acute oral toxicity tests used today for regulatory purposes. Table 1. The best performing in vitro assays, which were selected as candidates for the final tiered testing strategy, are further evaluated in the pre-validation phase. Selected assay The neutral red uptake assay using the 3T3 fibroblast (3T3/NRU) Target (workpackage involved) General basal cytotoxicity (WP2) The cytokine release assay using whole blood (IL-1, IL-6, TNF-alpha) Haemotoxicity (WP 4) Cell differentiation in cord blood-derived cells (CBC/CFU-GM) Haemotoxicity (WP 4) Gene expression (GFAP, HSP-32, MBP and NF-H) in primary rat brain aggregate s (see Figure 1) Uridine incorporation measuring the total mrna synthesis in primary rat brain aggregate s Cytomic panel measuring oxidative stress (intracellular peroxidative activity, intracellular levels of superoxide anion, oxidized DNA base 8-oxoguanine) in HepG2, and A.704 cells Cytomic panel for cytotoxicity screening (intracellular Ca 2+ levels, mitochondrial membrane potential, plasma membrane potential) in HepG2, SH- SY5Y and A.704 cells Neurotoxicity (WP 7.1) Neurotoxicity (WP 7.1) New endpoints (WP 4) New endpoints (WP 4) The MTT assay using primary rat hepatocytes Metabolism (WP 6) Kinetic parameters: volume of distribution, protein binding, clearance, and oral absorption (Caco-2 cells) for the estimation of the oral dose from the effective concentration observed in vitro The estimation of compound passage through the blood-brain barrier using neuronal networks (for neurotoxicity assays) (see Figure 2) Biokinetics (WP5) Biokinetics (WP5) EU contract. LSHB-CT
2 NEWSLETTERNEWSLETTERNEW Background Starting in 2005, 24 European laboratories have tested 57 reference compounds in 75 in vitro assays, including assessment of basal cytotoxicity, organ- and system-specific toxicity, as well as analysis of biokinetic parameters. The generated data were used to assess the withinlaboratory variability, the preliminary predictive capacity and in some cases also the betweenlaboratory variability of each in vitro assay. The outcome of this phase of the project is a large toolbox of in vitro methods, some of them evaluated to the level of prevalidation (Table 2 in Appendix). The first approach to analyse the in vitro data: multivariate PLS analyses The in vitro IC 50 values calculated in each laboratory were correlated with (LC 50 blood concentration values) and animal (oral LD 50 values for rodents) data. The results showed that the combination of 3 in vitro assays (3T3 cells/neutral red uptake, Normal keraticytes/neutral red uptake, Brain re-aggregation s/gene expression) slightly improve the correlation between in vitro IC 50 and in vivo LD 50 (R 2 =0.59) and in vitro IC 50 and LC 50 (R 2 =0.71) compared to a single cytotoxicity assay (3T3 cells/neutral red uptake). However, a number of misclassified compounds were identified using this combination of assays. When the in vitro data were correlated with animal LD 50 data, physostigmine, warfarin, sodium selenate, parathion, nicotine and epinephrine were under-predicted, while amiodarone, 17-ethenylestradiol, sodium lauryl sulphate, cadmium chloride and carbamazepine were over-predicted. When the in vitro data were correlated with the LC 50 data, colchicine, nicotine, lindane, atropine, acetonitrile, strychnine, malathion, cyclosporine, and parathion were under predicted. Pentachlorphel, isopropyl alcohol, 2,4 dichlorphexyacid, dichlorvos, diquat dibromide and acetylsalicylic acid were overpredicted. By including kinetic data (volume of distribution, protein binding, clearance, and oral absorption) in the in vitro models, the predication of rat LD 50 values could be increased from R 2 =0,59 to R 2 =0,70. However, the combination of three assays did t improve the prediction of the different EU and GHS 1 toxicity classes when compared to a single cytotoxicity assay (e.g. 3T3 cells/neutral red uptake). Based on the results from this multivariate analysis, it was concluded that further statistical analysis of data was needed in order to select the best performing methods for the testing strategy. Figure 1. Re-aggregated brain cell s. Top row: the s are kept in Erlenmayer flasks under constant rotation in an incubator. Bottom row: Microphotographs of rat brain aggregates (20 days in vitro) EU contract. LSHB-CT
3 NEWSLETTERNEWSLETTERNEW Additional statistical analyses aimed to select assays for the testing strategy The selection of the in vitro/in silico methods for the prevalidation exercise from the total number of assays performed in the ACuteTox project was based on an ad hoc analysis of variability, repeatability and reproducibility of the single assays, as well as the assessment of preliminary predictive capacity using univariate and multivariate CART analyses (Table 2 in Appendix). The first step of the analysis consisted in the recalculation of EC 20, EC 50 or LOEC values for every experiment using a standard approach. The assays were then compared with respect to their reproducibility and reliability by evaluating the variability of the assays at the level of raw response data and at the level of the estimated EC 20, EC 50 or LOEC values. Since the ultimate aim of the statistical analysis for ACuteTox is the classification of chemicals into the official acute oral toxicity categories (EU and GHS 1 ), the Classification and Regression Trees (CART) was used as the classification algorithm of choice. CART is a n-parametric technique that produces either classification or regression trees, depending on whether the dependent variable is categorical or numeric, respectively. The application of the CART methodology meets with the approach suggested by the initial aim of ACuteTox, namely to improve the in vitro-in vivo correlation by evaluating existing outliers in order to introduce further parameters (ADE, metabolism, organ specificity) which might improve the correlation. The CART analyses were performed using the untransformed EC 50, EC 20 and LOEC values from the in vitro assays. In addition, for the neurotoxicity assays, a blood brain barrier (BBB) 1 The Globally Harmonized System (GHS) is an internationally standardized system for classifying chemicals. For acute oral toxicity the GHS categories are assigned based on the LD50 values: GHS 1 (LD50 < 5 mg/kg b.w., fatal if swallowed); GHS 2 (LD50 > 5 mg/kg < 50 mg/kg b.w., fatal if swallowed); GHS 3 (LD50 > 50 mg/kg < 300 mg/kg b.w., toxic if swallowed); GHS 4 (LD50 > 300 mg/kg < 2000 mg/kg b.w., harmful if swallowed); GHS 5 (LD50 > 2000 mg/kg b.w., may be harmful if swallowed; or LD50 > 5000 mg/kg, t classified). transformation was performed. Ather approach was used in which the EC 50, EC 20 and LOEC values from the in vitro assays were first transformed to rat LD 50 values using a transformation algorithm suggested by WP5 (kinetic transformations), which took into account, the intestinal absorption, protein binding, clearance and volume of distribution. After derivation of a CART tree, misclassification rate was estimated from the data. Twelve CART multivariate models have been investigated and three potential testing strategies have been identified. The best CART multivariate model found until w classified 65% (37 of the 57 reference chemicals) correctly into the 5 GHS acute oral toxicity classes. Toxicity was under-predicted for five compounds, by one hazard class for three compounds, by two classes for one compound and by three classes for one compound. Accordingly, toxicity was over-predicted for 15 of the compounds tested, by one hazard class for ten compounds, by two classes for three compounds and by three classes for two compounds. This misclassification rate is expected to be too optimistic because the correct classification rates are estimated from the training set. An unbiased estimate of the misclassification rate will be obtained from the application of the three selected CART trees to independent data, such as the data of the challenging exercise (prevalidation) that will involve testing of a new set of chemicals. The database of the project The Acutoxbase database is a working database that allows ACuteTox partners to submit and share in vitro and in vivo data on. The structure of the database is outd in the publication Kinsner-Ovaskainen et al, Toxicol In Vitro (3): The largest section of the database is of the part containing the in vitro data from all assays evaluated in the project. It also contains in vivo LD 50 data on the 97 project chemicals (50% of which were drugs and 30% industrial chemicals, 20% others) as well as data ( lethal blood concentrations). All in vivo data were collected from the literature. EU contract. LSHB-CT
4 NEWSLETTERNEWSLETTERNEW It is envisaged that Acutoxbase will be made publicly available after the project is terminated. Parts of the data collected in the database have been already published: The LC 50 data are available in the publication by Sjöström et al. Toxicol in Vitro 22 (2008) The summary of the 6 cytotoxicity assays (including all values) is presented in Clothier et al., ATLA 36, , 2008 The IC 50, IC 70 and IC 90 values for the chemicals tested in CFU-GM assay in Cerrato et al. Toxicol Appl Pharmacol 238, Effects on GABA-A receptor, GABA transport and Glutamate transport in primary d cortical neurons (neurotoxicity assays, EC 50 values) are summarized in Galofre et al. Neurotoxicol Teratol. 2010, 32(1): Evaluation of transcriptional activity of caspase-3 gene as a marker of acute neurotoxicity in rat cerebellar granular cells (IC 50 values published in Folch et al., Toxicol In Vitro. 2009). Neurofunctional endpoints assessed in cells for estimation of acute systemic toxicity are presented in Gustafsson et al. Toxicol Appl Pharmacol in press doi: /j.taap The publication on the analysis of the in vivo rat and mouse data and the summary tables of the LD 50 values is in preparation. Next Newsletter will be published in June 2010 More information about the project and publications can be found at Figure 2. Strategy used to develop a neural network model to predict the passage of the blood-brain barrier. The key steps of this methodology are: I) codification using CODES program, II) reduction of dimensions of the dynamic matrix, III) training of the neural model and IV) model validation EU contract. LSHB-CT
5 Table 2 In vitro assays evaluated in the different Work Packages (WPs) of the ACuteTox project. The Table presents the status of the assays, including an indication whether within-laboratory reproducibility, between-laboratory reproducibility and preliminary predictive capacity were evaluated. In vitro assay Test system Within-lab reproducibility Between-lab reproducibility Preliminary predictive capacity Candidates for tiered testing strategy Cytotoxicity ATP content HL-60 Protein content (CBQCA assay) HepG2 Protein content (CBQCA assay) Fa32 rat Neutral red uptake Neutral red uptake Balb/3T3 mouse fibroblasts Normal keraticytes Neutral red uptake Fa32 rat New cell systems, new endpoints Multiplexed flow cytometry analysis of cytokine release (IL- 12p70, IFN-γ, IL-2, IL-10, IL-8, IL-6, IL- 4, IL-5, IL-1β, TNF-α, TNF-β) ELISA analysis of cytokines IL-5, IFN-γ and TNF-α release Human peripheral blood monuclear cells (PBMC) Human peripheral blood monuclear cells (PBMC) Cytokine release (IL- 1, IL-6, TNF-α) Human whole blood Colony forming unitgranulocyte/macroph age (CFU-GM) Cytomic Panel for Cytotoxicity Screening including: Human cord blood cells Performed in three s: - A.704 kidney EU contract. LSHB-CT
6 - intracellular Ca 2+ (Fluo-4 probe) - mitochondrial membrane potential (rhodamine123) - plasma membrane potential (DIBAC probe) - intracellular lipid content (BODIPY probe) Cytomic Panel for Oxidative Stress Screening including: - intracellular peroxides - mitochondrial generation of superoxide - intracellular levels of the oxidized DNA base 8-Oxo- Guanine adecarcima - HepG2 - Performed in three s: - A.704 kidney adecarcima - HepG2 - Biokinetics PAMPA assay Not available Intestinal absorption (permeability assay) Intestinal absorption (toxicity assay - 14 C- Mannitol) Intestinal absorption (toxicity assay Lucifer yellow) Caco-2 intestinal cell * Caco-2 intestinal cell * Caco-2 intestinal cell *, a) Blood-brain barrier passage (permeability assay) Blood-brain barrier model ** Blood-brain barrier (toxicity assay Lucifer yellow) Blood-brain barrier model ** Blood-brain barrier (toxicity assay - 14 C- sucrose) Blood-brain barrier model ** Aqueous solubility Not available See te a) Plasma protein Pooled See te a) EU contract. LSHB-CT
7 binding plasma*** Chromatographic Hydrophobicity Index (CHI) Not available Metabolic stability Rat liver microsomes**** See te a) Metabolic stability Human liver microsomes**** See te a) Metabolic stability hepatocytes See te a) Metabolic stability Cryopreserved hepatocytes See te a) Neurotoxicity Alamar Blue assay See te b) Mitochondrial membrane potential See te b) AChE inhibition Pure enzyme See te b) AChE inhibition LDH leakage See te b) LDH leakage Rat brain slices See te b) Ca 2+ overload See te b) GABA-A receptor function Primary mouse cortical neurons GABA uptake Primary mouse cortical neurons See te b) Cell membrane potential (CMP) Primary mouse cortical neurons See te b) EU contract. LSHB-CT
8 Glutamate uptake Primary mouse See te b) Cell membrane potential (CMP) Noradrenalin uptake See te b) Voltage operated Ca 2+ channel function See te b) Acetylcho receptor function See te b) Gene expression (GFAP, MBP, NF-H, NF-M, PPAR-gamma, HSP-32, inos) Enzyme inhibition/activation (ChAT, GS, AChE, LDH, 2,3-CNP) See te b) Methionine uptake (protein synthesis) Uridine uptake (RNA synthesis) 2-deoxyglucose uptake Caspase-3 mrna expression MTT assay See te b) Glutamate induced cytosolic Ca 2+ increase See te b) EU contract. LSHB-CT
9 ROS production See te b) LDH release See te b) Microarray quantitative mrna expression analyses of 31 genes See te b) Nephrotoxicity Alamar Blue Transepithelial electrical resistance (TEER) LLCPK-1 renal epithelial cell LLCPK-1 renal epithelial cell Hepatotoxicity, metabolism and bioactivation MTT assay MTT assay MTT assay Rat hepatocytes Balb/3T3 mouse fibroblasts HepG2 a) WP5 - These assays provided input data for the PBBK modelling (biokinetics) b) WP7.1 only 20 chemicals have been tested. * Three different protocols for culturing Caco-2 cells and for the assessment of toxicity and prediction of intestinal absorption in Caco-2 cells have been evaluated in WP5. The cell models were: Caco- 2/TC-7 clone, CacoReady TM (Advancell, Spain), Caco-2 from ATCC. ** Two different models of Blood-brain barrier (BBB), as well as three different protocols for the assessment of toxicity and prediction of BBB passage have been evaluated in WP5 *** Two different protocol for plasma protein binding were used in WP5 **** Two different protocol for rat and microsomal stability were used in WP5 EU contract. LSHB-CT
Workshop: Applying Exposure Science to Increase the Utility of Non-Animal Data in Efficacy and Safety Testing, 16 Feb. 2017
 Workshop: Applying Exposure Science to Increase the Utility of Non-Animal Data in Efficacy and Safety Testing, 16 Feb. 2017 Presentation Outline Should we account for kinetics in vitro and in vivo for
Workshop: Applying Exposure Science to Increase the Utility of Non-Animal Data in Efficacy and Safety Testing, 16 Feb. 2017 Presentation Outline Should we account for kinetics in vitro and in vivo for
Value Scale (1-5) 1 I. Final Product Profile Characteristics of the product required for patient care
 In this example, the majority of the compounds was active in the xenograft model and showed "drug-like" properties from in-silico and experimental data. However, the animal studies showed moderate toxicity
In this example, the majority of the compounds was active in the xenograft model and showed "drug-like" properties from in-silico and experimental data. However, the animal studies showed moderate toxicity
vitro Toxicology Analysis
 Anthony D Baxter, CEO New High Content Screening Methods Applied to in vitro Toxicology Analysis Better decisions. Earlier in the game. Enquiries@cyprotex.com Business overview A uniquely packaged ADMET
Anthony D Baxter, CEO New High Content Screening Methods Applied to in vitro Toxicology Analysis Better decisions. Earlier in the game. Enquiries@cyprotex.com Business overview A uniquely packaged ADMET
Our Business. in vitro / in vivo. in vitro / in vivo ADME. in vitro / in vivo. Toxicology. Cell Battery V79. Analytical Services. Service.
 GenPharmTox is one of the few CROs who can guarantee the success of our project within the given time framework and on the terms agreed. Dr. Michael Runkel, Apogepha GmbH 3 3 In vitro 3.1. Phototoxicity
GenPharmTox is one of the few CROs who can guarantee the success of our project within the given time framework and on the terms agreed. Dr. Michael Runkel, Apogepha GmbH 3 3 In vitro 3.1. Phototoxicity
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/30138 holds various files of this Leiden University dissertation Author: Driessen, Marja Title: Evaluation of the zebrafish embryo as an alternative model
Cover Page The handle http://hdl.handle.net/1887/30138 holds various files of this Leiden University dissertation Author: Driessen, Marja Title: Evaluation of the zebrafish embryo as an alternative model
Preclinical Drug Development
 Preclinical Drug Development A guidance prepared by From a 2004 NIH Summit Workshop: A major reason for the tremendous cost of drug development is the high rate of drug candidate failure during clinical
Preclinical Drug Development A guidance prepared by From a 2004 NIH Summit Workshop: A major reason for the tremendous cost of drug development is the high rate of drug candidate failure during clinical
Strategies for Assessment of Immunotoxicology in Preclinical Drug Development
 Strategies for Assessment of Immunotoxicology in Preclinical Drug Development Rebecca Brunette, PhD Scientist, Analytical Biology SNBL USA Preclinical Immunotoxicology The study of evaluating adverse effects
Strategies for Assessment of Immunotoxicology in Preclinical Drug Development Rebecca Brunette, PhD Scientist, Analytical Biology SNBL USA Preclinical Immunotoxicology The study of evaluating adverse effects
Maximizing opportunities towards achieving clinical success D R U G D I S C O V E R Y. Report Price Publication date
 F o r a c l e a r e r m a r k e t p e r s p e c t i v e Early Stage Drug Safety Strategies & Risk Management Maximizing opportunities towards achieving clinical success D R U G D I S C O V E R Y Report
F o r a c l e a r e r m a r k e t p e r s p e c t i v e Early Stage Drug Safety Strategies & Risk Management Maximizing opportunities towards achieving clinical success D R U G D I S C O V E R Y Report
PK and PK/PD Guided Starting Dose Selection for First-In-Human Trials. Sylvia Zhao ( 赵子微 ) Translational Clinical Oncology Novartis
 PK and PK/PD Guided Starting Dose Selection for First-In-Human Trials Sylvia Zhao ( 赵子微 ) Translational Clinical Oncology Novartis Disclaimer Contents are the opinion of the author and not that of Novartis
PK and PK/PD Guided Starting Dose Selection for First-In-Human Trials Sylvia Zhao ( 赵子微 ) Translational Clinical Oncology Novartis Disclaimer Contents are the opinion of the author and not that of Novartis
The Long Range Science Strategy (LRSS) of Cosmetics Europe
 The Long Range Science Strategy (LRSS) of Cosmetics Europe Repeated dose systemic toxicity 2016 Bas Blaauboer Aim of LRSS: Development of a strategy to determine safety of cosmetic ingredients and products
The Long Range Science Strategy (LRSS) of Cosmetics Europe Repeated dose systemic toxicity 2016 Bas Blaauboer Aim of LRSS: Development of a strategy to determine safety of cosmetic ingredients and products
Chapter - 9 IN VITRO CYTOTOXICITY ASSAY OF ZERUMBONE AND MDM3:1
 Chapter - 9 IN VITRO CYTOTOXICITY ASSAY OF ZERUMBONE AND MDM3:1 9.1 INTRODUCTION 9.2 CHAPTER OBJECTIVE 9.3 MATERIALS AND METHODS 9.4 RESULT 9.5 DISCUSSION In Vitro Cytotoxicity Assay of Zerumbone and MDM
Chapter - 9 IN VITRO CYTOTOXICITY ASSAY OF ZERUMBONE AND MDM3:1 9.1 INTRODUCTION 9.2 CHAPTER OBJECTIVE 9.3 MATERIALS AND METHODS 9.4 RESULT 9.5 DISCUSSION In Vitro Cytotoxicity Assay of Zerumbone and MDM
Alternative (Non-animal) Methods for Chemicals Testing: Current Status and Future Prospects
 Alternative (Non-animal) Methods for Chemicals Testing: Current Status and Future Prospects A Report prepared by ECVAM and the ECVAM Working Group on Chemicals Edited by Andrew Worth and Michael Balls
Alternative (Non-animal) Methods for Chemicals Testing: Current Status and Future Prospects A Report prepared by ECVAM and the ECVAM Working Group on Chemicals Edited by Andrew Worth and Michael Balls
Cosmetics Europe LRSS Programme
 Cosmetics Europe LRSS Programme 2016-20 Rob Taalman Objectives of CE Research To deliver tools & strategies for animal-free safety assessment Accurate Robust Efficient ultimately accepted by regulators
Cosmetics Europe LRSS Programme 2016-20 Rob Taalman Objectives of CE Research To deliver tools & strategies for animal-free safety assessment Accurate Robust Efficient ultimately accepted by regulators
HUMAN CYSTIC ECHINOCOCCOSIS RESEARCH IN CENTRAL AND EASTERN SOCIETIES (HERACLES) KICK OFF MEETING, DECEMBER 2013
 HUMAN CYSTIC ECHINOCOCCOSIS RESEARCH IN CENTRAL AND EASTERN SOCIETIES (HERACLES) KICK OFF MEETING, 17-18 DECEMBER 2013 PARTNER 7 PENDIK VETERINARY CONTROL INSTITUTE, DEPARTMENT OF PARASITOLOGY, TURKEY
HUMAN CYSTIC ECHINOCOCCOSIS RESEARCH IN CENTRAL AND EASTERN SOCIETIES (HERACLES) KICK OFF MEETING, 17-18 DECEMBER 2013 PARTNER 7 PENDIK VETERINARY CONTROL INSTITUTE, DEPARTMENT OF PARASITOLOGY, TURKEY
Extrapolation of in vitro effects to in vivo embryotoxicity Basic requirements for prediction model development. Han van de Sandt, PhD
 Extrapolation of in vitro effects to in vivo embryotoxicity Basic requirements for prediction model development Han van de Sandt, PhD TNO: Netherlands Organisation for Applied Scientific Research Established
Extrapolation of in vitro effects to in vivo embryotoxicity Basic requirements for prediction model development Han van de Sandt, PhD TNO: Netherlands Organisation for Applied Scientific Research Established
Introduction to Drug Development in Commercializing Biomedical Technology
 Introduction to Drug Development in Commercializing Biomedical Technology Kevin W. Hunt, Ph.D. Director of Biopharmaceutical Product Development Office of Technology Development and Head of Translational
Introduction to Drug Development in Commercializing Biomedical Technology Kevin W. Hunt, Ph.D. Director of Biopharmaceutical Product Development Office of Technology Development and Head of Translational
Human embryonic stem cells for in-vitro developmental toxicity testing
 Human embryonic stem cells for in-vitro developmental toxicity testing HESI workshop on alternatives assays for developmental toxicity Raimund Strehl Cellartis The company Founded in early 2001, University
Human embryonic stem cells for in-vitro developmental toxicity testing HESI workshop on alternatives assays for developmental toxicity Raimund Strehl Cellartis The company Founded in early 2001, University
Transfection Reagents That Really Work
 Transfection Reagents That Really Work non-toxic, biodegradable, highly efficient, serum-compatible Available for DNA,, protein, stem cells www.clontech.com/xf P L A S M I D T R A N S F E C T I O N R E
Transfection Reagents That Really Work non-toxic, biodegradable, highly efficient, serum-compatible Available for DNA,, protein, stem cells www.clontech.com/xf P L A S M I D T R A N S F E C T I O N R E
PROTOCOL. Monoamine Oxidase B Enzyme Specific Activity Assay Kit (human) MS747 Rev.0 DESCRIPTION ADDITIONAL MATERIALS REQUIRED
 PROTOCOL Monoamine Oxidase B Enzyme Specific Activity Assay Kit (human) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 MS747 Rev.0 DESCRIPTION MAOB Enzyme Specific Activity Assay Kit Sufficient materials
PROTOCOL Monoamine Oxidase B Enzyme Specific Activity Assay Kit (human) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 MS747 Rev.0 DESCRIPTION MAOB Enzyme Specific Activity Assay Kit Sufficient materials
Comparison of three cytokine release assays (CRA) for hazard. syndrome potential of monoclonal
 Comparison of three cytokine release assays (CRA) for hazard identification of cytokine release syndrome potential of monoclonal antibody (mab) therapeutics ti Madeline Fort HESI ITC Cytokine Release Assay
Comparison of three cytokine release assays (CRA) for hazard identification of cytokine release syndrome potential of monoclonal antibody (mab) therapeutics ti Madeline Fort HESI ITC Cytokine Release Assay
Investigation of an In Vitro Method for Protein Hazard Characterization
 Investigation of an In Vitro Method for Protein Hazard Characterization Bryan Delaney, PhD, DABT, Fellow, ATS DuPont Pioneer Johnston, Iowa bryan.delaney@pioneer.com 515.535.7086 Conflict of Interest Statement
Investigation of an In Vitro Method for Protein Hazard Characterization Bryan Delaney, PhD, DABT, Fellow, ATS DuPont Pioneer Johnston, Iowa bryan.delaney@pioneer.com 515.535.7086 Conflict of Interest Statement
ELISPOT and FLUOROSPOT kits
 ELISPOT and FLUOROSPOT kits Interleukins Interferons Granzymes and perforins TNF superfamily ligands and receptors Apoptosis markers And many more... ELISPOT and FLUOROSPOT: a cell-based assay to assess
ELISPOT and FLUOROSPOT kits Interleukins Interferons Granzymes and perforins TNF superfamily ligands and receptors Apoptosis markers And many more... ELISPOT and FLUOROSPOT: a cell-based assay to assess
NEURONAL CELL CULTURE MATRIX FOR BETTER MAINTENANCE AND SURVIVAL OF NEURONAL CELL CULTURES IN TISSUE CULTURE.
 NEURONAL CELL CULTURE MATRIX FOR BETTER MAINTENANCE AND SURVIVAL OF NEURONAL CELL CULTURES IN TISSUE CULTURE. D. R. Aguirre, N. DiMassa, Chrystal Johnson, H. Eran, R. Perez, C.V.R. Sharma, M.V.R. Sharma,
NEURONAL CELL CULTURE MATRIX FOR BETTER MAINTENANCE AND SURVIVAL OF NEURONAL CELL CULTURES IN TISSUE CULTURE. D. R. Aguirre, N. DiMassa, Chrystal Johnson, H. Eran, R. Perez, C.V.R. Sharma, M.V.R. Sharma,
Cell Health and Viability Assays Real-time automated measurements of cell health and viability inside your incubator
 INCUCYTE LIVE-CELL ANALYSIS SYSTEM Cell Health and Viability Assays Real-time automated measurements of cell health and viability inside your incubator See what your cells are doing and when they do it
INCUCYTE LIVE-CELL ANALYSIS SYSTEM Cell Health and Viability Assays Real-time automated measurements of cell health and viability inside your incubator See what your cells are doing and when they do it
Pharmacology. Chatchai Chinpaisal, Ph.D. Department of Pharmacology and Toxicology, Faculty of Pharmacy, Silpakorn University.
 Pharmacology Chatchai Chinpaisal, Ph.D. Department of Pharmacology and Toxicology, Faculty of Pharmacy, Silpakorn University. 1 PHARMACODYNAMIC STUDIES A. Primary pharmacodynamics primary action in target
Pharmacology Chatchai Chinpaisal, Ph.D. Department of Pharmacology and Toxicology, Faculty of Pharmacy, Silpakorn University. 1 PHARMACODYNAMIC STUDIES A. Primary pharmacodynamics primary action in target
Peptide libraries: applications, design options and considerations. Laura Geuss, PhD May 5, 2015, 2:00-3:00 pm EST
 Peptide libraries: applications, design options and considerations Laura Geuss, PhD May 5, 2015, 2:00-3:00 pm EST Overview 1 2 3 4 5 Introduction Peptide library basics Peptide library design considerations
Peptide libraries: applications, design options and considerations Laura Geuss, PhD May 5, 2015, 2:00-3:00 pm EST Overview 1 2 3 4 5 Introduction Peptide library basics Peptide library design considerations
Cytomics in Action: Cytokine Network Cytometry
 Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
PCR Arrays. An Advanced Real-time PCR Technology to Empower Your Pathway Analysis
 PCR Arrays An Advanced Real-time PCR Technology to Empower Your Pathway Analysis 1 Table of Contents 1. Introduction to the PCR Arrays 2. How PCR Arrays Work 3. Performance Data from PCR Arrays 4. Research
PCR Arrays An Advanced Real-time PCR Technology to Empower Your Pathway Analysis 1 Table of Contents 1. Introduction to the PCR Arrays 2. How PCR Arrays Work 3. Performance Data from PCR Arrays 4. Research
Oroxcell Cellular Toxicity
 Oroxcell 2009 Cellular Toxicity Table of contents Cellular Toxicity 1. Episkin Episkin model by SkinEthic Skin corrosion Skin irritation 2. Reconstructed Human Epidermis (RHE) RHE model by SkinEthic Validation
Oroxcell 2009 Cellular Toxicity Table of contents Cellular Toxicity 1. Episkin Episkin model by SkinEthic Skin corrosion Skin irritation 2. Reconstructed Human Epidermis (RHE) RHE model by SkinEthic Validation
Toxicological Assessment of (silver) Nanomaterials: Challenges and Pitfalls
 Dr Helinor Johnston, Nano Safety Research Group Toxicological Assessment of (silver) Nanomaterials: Challenges and Pitfalls BfR Conference: Nanosilver Feb 2012 h.johnston@hw.ac.uk Nanotoxicology Nanotechnology
Dr Helinor Johnston, Nano Safety Research Group Toxicological Assessment of (silver) Nanomaterials: Challenges and Pitfalls BfR Conference: Nanosilver Feb 2012 h.johnston@hw.ac.uk Nanotoxicology Nanotechnology
Transfection Reagent INTRODUCTION SPECIFICATIONS MATERIALS. For Research Use Only. Materials Supplied. Materials required, but not supplied
 TransIT-Neural Transfection Reagent INTRODUCTION TransIT-Neural Transfection Reagent is specifically optimized to provide exceptional transfection efficiency of plasmid DNA in various neural cell types.
TransIT-Neural Transfection Reagent INTRODUCTION TransIT-Neural Transfection Reagent is specifically optimized to provide exceptional transfection efficiency of plasmid DNA in various neural cell types.
INTRODUCTION TO PHARMACOLOGY
 INTRODUCTION TO PHARMACOLOGY Pharmacology is the study of how chemicals interact with the body Endogenous hormones, growth factors, etc Exogenous drugs Two areas of study Pharmacodynamics Interaction of
INTRODUCTION TO PHARMACOLOGY Pharmacology is the study of how chemicals interact with the body Endogenous hormones, growth factors, etc Exogenous drugs Two areas of study Pharmacodynamics Interaction of
Justification Document for the Selection of a CoRAP Substance
 Justification Document for the Selection of a CoRAP Substance Substance Name (public name): 1,1'-(isopropylidene)bis[3,5-dibromo- 4-(2,3-dibromopropoxy)benzene] EC Number: 244-617-5 CAS Number: 21850-44-2
Justification Document for the Selection of a CoRAP Substance Substance Name (public name): 1,1'-(isopropylidene)bis[3,5-dibromo- 4-(2,3-dibromopropoxy)benzene] EC Number: 244-617-5 CAS Number: 21850-44-2
Proteomics. Areas of Application for Proteomics. Most Commonly Used Proteomics Techniques: Limitations: Examples
 Proteomics Areas of Application for Proteomics Most Commonly Used Proteomics Techniques: Antibody arrays Protein activity arrays 2-D gels ICAT technology SELDI Limitations: protein sources surfaces and
Proteomics Areas of Application for Proteomics Most Commonly Used Proteomics Techniques: Antibody arrays Protein activity arrays 2-D gels ICAT technology SELDI Limitations: protein sources surfaces and
Convoy TM Transfection Reagent
 Convoy TM Transfection Reagent Catalog No.11103 0.25ml (40-80 transfections in 35mm dishes) Catalog No.11105 0.5 ml (80-165 transfections in 35mm dishes) Catalog No.11110 1.0 ml (165-330 transfections
Convoy TM Transfection Reagent Catalog No.11103 0.25ml (40-80 transfections in 35mm dishes) Catalog No.11105 0.5 ml (80-165 transfections in 35mm dishes) Catalog No.11110 1.0 ml (165-330 transfections
Stem cell transfection guide
 APPLICATION NOTE Stem cell transfection guide Gene delivery solutions Introduction Stem cells continue to show immense promise for the future of regenerative medicine and personalized therapeutic treatments.
APPLICATION NOTE Stem cell transfection guide Gene delivery solutions Introduction Stem cells continue to show immense promise for the future of regenerative medicine and personalized therapeutic treatments.
ab Mitochondrial Viability Assay
 ab129732 Mitochondrial Viability Assay Instructions for Use For measuring mitochondrial/cellular viability in high throughput This product is for research use only and is not intended for diagnostic use.
ab129732 Mitochondrial Viability Assay Instructions for Use For measuring mitochondrial/cellular viability in high throughput This product is for research use only and is not intended for diagnostic use.
Summary of Mutagenic Toxicity Test Results for EvaGreen
 Summary of Mutagenic Toxicity Test Results for EvaGreen Compiled by Biotium, Inc. from the results of an independent testing service: Litron Laboratories, Inc., Rochester, NY Overview When our scientists
Summary of Mutagenic Toxicity Test Results for EvaGreen Compiled by Biotium, Inc. from the results of an independent testing service: Litron Laboratories, Inc., Rochester, NY Overview When our scientists
The Road to Functional Bioanalysis: Development and Validation of a Cell-Based Assay for Neutralizing Anti-Drug Antibody Analysis
 The Road to Functional Bioanalysis: Development and Validation of a Cell-Based Assay for Neutralizing Anti-Drug Antibody Analysis Christelle Pythoud, PhD 2 nd EBF YSS, Barcelona Spain Celerion Switzerland
The Road to Functional Bioanalysis: Development and Validation of a Cell-Based Assay for Neutralizing Anti-Drug Antibody Analysis Christelle Pythoud, PhD 2 nd EBF YSS, Barcelona Spain Celerion Switzerland
Stem Cell Services. Driving Innovation for Stem Cell Researchers
 Driving Innovation for Stem Cell Researchers Stem Cell Services Partner with us and have access to the most advanced and comprehensive stem cell services available today. 675 W. Kendall St. Cambridge,
Driving Innovation for Stem Cell Researchers Stem Cell Services Partner with us and have access to the most advanced and comprehensive stem cell services available today. 675 W. Kendall St. Cambridge,
TITLE: Novel Small-Molecule Inhibitor of Tyk2: Lucrative Therapeutic Target in Lupus
 AWARD NUMBER: W81XWH-16-1-0609 TITLE: Novel Small-Molecule Inhibitor of Tyk2: Lucrative Therapeutic Target in Lupus PRINCIPAL INVESTIGATOR: Abhishek Trigunaite CONTRACTING ORGANIZATION: SRI International
AWARD NUMBER: W81XWH-16-1-0609 TITLE: Novel Small-Molecule Inhibitor of Tyk2: Lucrative Therapeutic Target in Lupus PRINCIPAL INVESTIGATOR: Abhishek Trigunaite CONTRACTING ORGANIZATION: SRI International
Agilent RapidFire 360 High-throughput Mass Spectrometry System. Innovative Integrated Technology
 Agilent RapidFire 360 High-throughput Mass Spectrometry System Innovative Integrated Technology Agilent RapidFire High-throughput Mass Spectrometry system Streamline workflows and eliminate bottlenecks
Agilent RapidFire 360 High-throughput Mass Spectrometry System Innovative Integrated Technology Agilent RapidFire High-throughput Mass Spectrometry system Streamline workflows and eliminate bottlenecks
Xfect Protein Transfection Reagent
 Xfect Protein Transfection Reagent Mammalian Expression Systems Rapid, high-efficiency, low-toxicity protein transfection Transfect a large amount of active protein Virtually no cytotoxicity, unlike lipofection
Xfect Protein Transfection Reagent Mammalian Expression Systems Rapid, high-efficiency, low-toxicity protein transfection Transfect a large amount of active protein Virtually no cytotoxicity, unlike lipofection
drug discovery: Where are we now? How did we RSC February 2013
 The role of pharmacokinetics in drug discovery: Where are we now? How did we get th here??where are we going? Peter Webborn RSC February 2013 Why conduct PK studies in animals? The primary purpose of pre-clinical
The role of pharmacokinetics in drug discovery: Where are we now? How did we get th here??where are we going? Peter Webborn RSC February 2013 Why conduct PK studies in animals? The primary purpose of pre-clinical
Supporting Online Material, Matsumoto et al.
 Supporting Online Material, Matsumoto et al. Material and Methods Library. Poly(A) + mrna was purified from RAW264.7 cells stimulated with murine IFN-γ (100 units/ml) and bacterial LPS (100 ng/ml) for
Supporting Online Material, Matsumoto et al. Material and Methods Library. Poly(A) + mrna was purified from RAW264.7 cells stimulated with murine IFN-γ (100 units/ml) and bacterial LPS (100 ng/ml) for
Green Fluorescent Protein (GFP) Purification. Hydrophobic Interaction Chromatography
 Green Fluorescent Protein (GFP) Purification Hydrophobic Interaction Chromatography What is the GFP gene? GFP is a green fluorescent protein that is normally found in jellyfish. It has been engineered
Green Fluorescent Protein (GFP) Purification Hydrophobic Interaction Chromatography What is the GFP gene? GFP is a green fluorescent protein that is normally found in jellyfish. It has been engineered
US EPA Perspectives on GHS SCHC Fall Meeting. Kaitlin Keller, Pesticide Re-evaluation Division Office of Pesticide Programs, USEPA September 27, 2017
 1 US EPA Perspectives on GHS SCHC Fall Meeting Kaitlin Keller, Pesticide Re-evaluation Division Office of Pesticide Programs, USEPA September 27, 2017 2 Background: GHS GHS was adopted by the UN in 2003;
1 US EPA Perspectives on GHS SCHC Fall Meeting Kaitlin Keller, Pesticide Re-evaluation Division Office of Pesticide Programs, USEPA September 27, 2017 2 Background: GHS GHS was adopted by the UN in 2003;
Relative cost of hepatocytes
 Relative cost of hepatocytes - WORKPACKAGE 8 PROGENITOR CELLS FOR BIO- ARTIFICIAL LIVER UNEW, Charité, SCS StemCellSystems WP2 System design and medical device regulatory requirements WP7 Communications,
Relative cost of hepatocytes - WORKPACKAGE 8 PROGENITOR CELLS FOR BIO- ARTIFICIAL LIVER UNEW, Charité, SCS StemCellSystems WP2 System design and medical device regulatory requirements WP7 Communications,
Rat TNF-α ELISA REF. Ver5.0 RUO. ELISA Set for Accurate Quantitation from Cell Culture Supernatant, Serum, Plasma or Other Bodily Fluids
 REF : KB3145 Ver5.0 RUO ELISA Set for Accurate Quantitation from Cell Culture Supernatant, Serum, Plasma or Other Bodily Fluids RUO For Research Use Only REF Catalog Number Store At Manufactured By Expiry
REF : KB3145 Ver5.0 RUO ELISA Set for Accurate Quantitation from Cell Culture Supernatant, Serum, Plasma or Other Bodily Fluids RUO For Research Use Only REF Catalog Number Store At Manufactured By Expiry
OxiSelect Monoamine Oxidase Assay Kit (Colorimetric)
 Product Manual OxiSelect Monoamine Oxidase Assay Kit (Colorimetric) Catalog Number XPX-5006 XPX-5006-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Monoamine
Product Manual OxiSelect Monoamine Oxidase Assay Kit (Colorimetric) Catalog Number XPX-5006 XPX-5006-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Monoamine
Figure S1. Figure S2. Figure S3 HB Anti-FSP27 (COOH-terminal peptide) Ab. Anti-GST-FSP27(45-127) Ab.
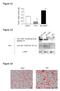 / 36B4 mrna ratio Figure S1 * 2. 1.6 1.2.8 *.4 control TNFα BRL49653 Figure S2 Su bw AT p iw Anti- (COOH-terminal peptide) Ab Blot : Anti-GST-(45-127) Ab β-actin Figure S3 HB2 HW AT BA T Figure S4 A TAG
/ 36B4 mrna ratio Figure S1 * 2. 1.6 1.2.8 *.4 control TNFα BRL49653 Figure S2 Su bw AT p iw Anti- (COOH-terminal peptide) Ab Blot : Anti-GST-(45-127) Ab β-actin Figure S3 HB2 HW AT BA T Figure S4 A TAG
Enzymes Part III: regulation I. Dr. Mamoun Ahram Summer, 2017
 Enzymes Part III: regulation I Dr. Mamoun Ahram Summer, 2017 Mechanisms of regulation Expression of isoenzymes Regulation of enzymatic activity Inhibitors Conformational changes Allostery Modulators Reversible
Enzymes Part III: regulation I Dr. Mamoun Ahram Summer, 2017 Mechanisms of regulation Expression of isoenzymes Regulation of enzymatic activity Inhibitors Conformational changes Allostery Modulators Reversible
Proteomics. Areas of Application for Proteomics Most Commonly Used Proteomics Techniques: Limitations: Examples
 Proteomics Areas of Application for Proteomics Most Commonly Used Proteomics Techniques: Antibody arrays Protein activity arrays 2-D gels ICAT technology SELDI Limitations: protein sources surfaces and
Proteomics Areas of Application for Proteomics Most Commonly Used Proteomics Techniques: Antibody arrays Protein activity arrays 2-D gels ICAT technology SELDI Limitations: protein sources surfaces and
ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions. Step 3
 European Medicines Agency June 2009 EMEA/CHMP/ICH/380636/2009 ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions Step 3 NOTE FOR GUIDANCE
European Medicines Agency June 2009 EMEA/CHMP/ICH/380636/2009 ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions Step 3 NOTE FOR GUIDANCE
SANIPEDIA Research Lab
 SANIPEDIA Research Lab Cells and In Vitro Assays Catalog Index A. CELL PORTFOLIO 3 B. GENERAL ASSAYS 4 B.1 WHOLE CELL ASSAYS 4 B.1.1. Viability and Toxicity 4 B.1.2. Proliferation 4 B.1.3. Apoptosis 4
SANIPEDIA Research Lab Cells and In Vitro Assays Catalog Index A. CELL PORTFOLIO 3 B. GENERAL ASSAYS 4 B.1 WHOLE CELL ASSAYS 4 B.1.1. Viability and Toxicity 4 B.1.2. Proliferation 4 B.1.3. Apoptosis 4
ingenio electroporation kits & solution
 ingenio electroporation kits & solution Electroporation x DEFINITION and OPTIMIZATION What is ELECTROPORATION? Electroporation is a physical method of nucleic acid transfer wherein the cells and nucleic
ingenio electroporation kits & solution Electroporation x DEFINITION and OPTIMIZATION What is ELECTROPORATION? Electroporation is a physical method of nucleic acid transfer wherein the cells and nucleic
Measurement of Hematopoietic Stem Cell Potency Prior to Transplantation
 WHITE PAPER Measurement of Hematopoietic Stem Cell Potency Prior to Transplantation February, 2009 This White Paper is a forward-looking statement. It represents the present state of the art and future
WHITE PAPER Measurement of Hematopoietic Stem Cell Potency Prior to Transplantation February, 2009 This White Paper is a forward-looking statement. It represents the present state of the art and future
MitoBiogenesis In-Cell ELISA Kit (Colorimetric)
 PROTOCOL MitoBiogenesis In-Cell ELISA Kit (Colorimetric) DESCRIPTION 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 MS643 Rev.2 For identifying inhibitors and activators of mitochondrial biogenesis
PROTOCOL MitoBiogenesis In-Cell ELISA Kit (Colorimetric) DESCRIPTION 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 MS643 Rev.2 For identifying inhibitors and activators of mitochondrial biogenesis
Nanomedicine: An Innovative Approach Towards Treating Tuberculosis
 Nanomedicine: An Innovative Approach Towards Treating Tuberculosis Polymeric shell H. SWAI 1, L. KALOMBO, B. SEMETE and L. KATATA Encapsulation and Delivery Research Group Polymers and Composites, Materials
Nanomedicine: An Innovative Approach Towards Treating Tuberculosis Polymeric shell H. SWAI 1, L. KALOMBO, B. SEMETE and L. KATATA Encapsulation and Delivery Research Group Polymers and Composites, Materials
Alternative Testverfahren und intelligente Teststrategien Position der EU-Kommission. Thomas Hartung & ECVAM Team.
 Alternative Testverfahren und intelligente Teststrategien Position der EU-Kommission Thomas Hartung & ECVAM Team Institute for Health and Consumer Protection (IHCP) Ispra (Va), Italy http://ecvam.jrc.it
Alternative Testverfahren und intelligente Teststrategien Position der EU-Kommission Thomas Hartung & ECVAM Team Institute for Health and Consumer Protection (IHCP) Ispra (Va), Italy http://ecvam.jrc.it
FDA Perspective on the Preclinical Development of Cancer Vaccines
 FDA Perspective on the Preclinical Development of Cancer Vaccines Richard D. McFarland Ph.D., M.D. Medical Officer CBER/OCTGT/DCEPT mcfarlandr@cber.fda.gov Cancer Vaccine Clinical Trials Workshop Alexandria,
FDA Perspective on the Preclinical Development of Cancer Vaccines Richard D. McFarland Ph.D., M.D. Medical Officer CBER/OCTGT/DCEPT mcfarlandr@cber.fda.gov Cancer Vaccine Clinical Trials Workshop Alexandria,
Cell Health and Mechanistic Toxicity Assays
 Cell Health and Mechanistic Toxicity Assays Zhong Yu, PhD Product Manager BeNeLux & Nordic Zhong.yu@promeg.com March 2015 Outline Bioluminescent assays Live and Dead Cell Assays Apoptosis Stress Events
Cell Health and Mechanistic Toxicity Assays Zhong Yu, PhD Product Manager BeNeLux & Nordic Zhong.yu@promeg.com March 2015 Outline Bioluminescent assays Live and Dead Cell Assays Apoptosis Stress Events
Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section B and ONE question from Section C.
 UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2013-2014 CELL BIOLOGY BIO-2B06 Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in
UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2013-2014 CELL BIOLOGY BIO-2B06 Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in
green B 1 ) into a single unit to model the substrate in this reaction. enzyme
 Teacher Key Objectives You will use the model pieces in the kit to: Simulate enzymatic actions. Explain enzymatic specificity. Investigate two types of enzyme inhibitors used in regulating enzymatic activity.
Teacher Key Objectives You will use the model pieces in the kit to: Simulate enzymatic actions. Explain enzymatic specificity. Investigate two types of enzyme inhibitors used in regulating enzymatic activity.
Cells and Tissues. Overview CELLS
 Cells and Tissues WIll The basic unit of structure and function in the human body is the cell. Each of a cell's parts, or organelles, as well as the entire cell, is organized to perform a specific function.
Cells and Tissues WIll The basic unit of structure and function in the human body is the cell. Each of a cell's parts, or organelles, as well as the entire cell, is organized to perform a specific function.
leading the way in research & development
 leading the way in research & development people. passion. possibilities. ABBVIE 2 immunology AbbVie Immunology has a demonstrated record of success in identifying and developing both small molecule and
leading the way in research & development people. passion. possibilities. ABBVIE 2 immunology AbbVie Immunology has a demonstrated record of success in identifying and developing both small molecule and
PRECLINICAL DRUG DISCOVERY AND DEVELOPMENT BIOBOOT CAMP 2016
 PRECLINICAL DRUG DISCOVERY AND DEVELOPMENT BIOBOOT CAMP 2016 1 FROM DISCOVERY TO COMMERCIALIZATION At least 10 years on average Average spend of $1.4B Less than 12% of drugs entering Phase I are approved
PRECLINICAL DRUG DISCOVERY AND DEVELOPMENT BIOBOOT CAMP 2016 1 FROM DISCOVERY TO COMMERCIALIZATION At least 10 years on average Average spend of $1.4B Less than 12% of drugs entering Phase I are approved
Xeno-Free Systems for hesc & hipsc. Facilitating the shift from Stem Cell Research to Clinical Applications
 Xeno-Free Systems for hesc & hipsc Facilitating the shift from Stem Cell Research to Clinical Applications NutriStem Defined, xeno-free (XF), serum-free media (SFM) specially formulated for growth and
Xeno-Free Systems for hesc & hipsc Facilitating the shift from Stem Cell Research to Clinical Applications NutriStem Defined, xeno-free (XF), serum-free media (SFM) specially formulated for growth and
APPROACHES TO IMPROVING THE PERFORMANCE OF MAMMALIAN CELL CULTURES FOR PROTEIN PRODUCTION
 BioLOGIC USA BOSTON, 20 th OCTOBER 2004 APPROACHES TO IMPROVING THE PERFORMANCE OF MAMMALIAN CELL CULTURES FOR PROTEIN PRODUCTION Dr Robert Gay Lonza Biologics 2004 The Challenge of the MAb Market Global
BioLOGIC USA BOSTON, 20 th OCTOBER 2004 APPROACHES TO IMPROVING THE PERFORMANCE OF MAMMALIAN CELL CULTURES FOR PROTEIN PRODUCTION Dr Robert Gay Lonza Biologics 2004 The Challenge of the MAb Market Global
A Level. A Level Biology. Cells, Microscopes, Cell Cycle and Immunity Questions. AQA, OCR, Edexcel. Name: Total Marks: Page 1
 AQA, OCR, Edexcel A Level A Level Biology Cells, Microscopes, Cell Cycle and Immunity Questions Name: Total Marks: Page 1 Q1.The diagram shows a eukaryotic cell. (a) Complete the table by giving the letter
AQA, OCR, Edexcel A Level A Level Biology Cells, Microscopes, Cell Cycle and Immunity Questions Name: Total Marks: Page 1 Q1.The diagram shows a eukaryotic cell. (a) Complete the table by giving the letter
The TetraQ Difference. Quality Preclinical Drug Development Solutions. Provide advice and tailored solutions, not just a menu of choices
 A leading Australian contract research organisation (CRO) providing integrated preclinical drug development services to the biotech and pharmaceutical industries The TetraQ Difference Located at The University
A leading Australian contract research organisation (CRO) providing integrated preclinical drug development services to the biotech and pharmaceutical industries The TetraQ Difference Located at The University
WST-1 Cell Proliferation Assay Kit
 WST-1 Cell Proliferation Assay Kit Item No. 10008883 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
WST-1 Cell Proliferation Assay Kit Item No. 10008883 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
The Challenges of Designing Cyclic Prodrugs of Opioid Peptides that Permeate the Intestinal Mucosa and the Blood-Brain Barrier
 The Challenges of Designing Cyclic Prodrugs of pioid Peptides that Permeate the Intestinal Mucosa and the Blood-Brain Barrier Ronald T. Borchardt Department of Pharmaceutical Chemistry The University of
The Challenges of Designing Cyclic Prodrugs of pioid Peptides that Permeate the Intestinal Mucosa and the Blood-Brain Barrier Ronald T. Borchardt Department of Pharmaceutical Chemistry The University of
Long-Term Hepatotoxicity Studies using Cultured Human ipsc- Derived Hepatocytes
 A p p l i c a t i o n N o t e Long-Term Hepatotoxicity Studies using Cultured Human ipsc- Derived Hepatocytes Brad Larson and Peter Banks, BioTek Instruments, Inc., Winooski, VT USA Coby Carlson and Michael
A p p l i c a t i o n N o t e Long-Term Hepatotoxicity Studies using Cultured Human ipsc- Derived Hepatocytes Brad Larson and Peter Banks, BioTek Instruments, Inc., Winooski, VT USA Coby Carlson and Michael
Method Review by the Interagency Coordinating Committee for the Validation of Alternative Methods (ICCVAM)
 Method Review by the Interagency Coordinating Committee for the Validation of Alternative Methods (ICCVAM) ICCVAM was created as an ad-hoc committee in 1994 and became a standing Committee in 2000. ICCVAM
Method Review by the Interagency Coordinating Committee for the Validation of Alternative Methods (ICCVAM) ICCVAM was created as an ad-hoc committee in 1994 and became a standing Committee in 2000. ICCVAM
INTRODUCTION PATRICK POULIN, 1,2 SAMI HADDAD 2
 RESEARCH ARTICLE Pharmacokinetics, Pharmacodynamics and Drug Transport and Metabolism Albumin and Uptake of Drugs in Cells: Additional Validation Exercises of a Recently Published Equation that Quantifies
RESEARCH ARTICLE Pharmacokinetics, Pharmacodynamics and Drug Transport and Metabolism Albumin and Uptake of Drugs in Cells: Additional Validation Exercises of a Recently Published Equation that Quantifies
Can HTRF technology answer to different scientific questions? An academic with industrial standards point of view
 Plate-forme de Chimie Biologique Intégrative de Strasbourg UMS 3286 CNRS-UdS Can HTRF technology answer to different scientific questions? An academic with industrial standards point of view Pascal Villa
Plate-forme de Chimie Biologique Intégrative de Strasbourg UMS 3286 CNRS-UdS Can HTRF technology answer to different scientific questions? An academic with industrial standards point of view Pascal Villa
64 CuCl 2 in 50 µl 0.1N NaOAc buffer, and 20 µg of each DOTA-antibody conjugate in 40 µl
 Number of DOTA per antibody The average number of DOTA chelators per antibody was measured using a reported procedure with modifications (1,2). Briefly, nonradioactive CuCl 2 (80-fold excess of DOTA antibodies)
Number of DOTA per antibody The average number of DOTA chelators per antibody was measured using a reported procedure with modifications (1,2). Briefly, nonradioactive CuCl 2 (80-fold excess of DOTA antibodies)
Ingenio Electroporation Kits and Solution
 Protocol for MIR 08, 09, 01, 0111, 0112, 0113, 0114, 011, 0116, 0117, 0118, 0119 Quick Reference Protocol, SDS and Certificate of Analysis available at mirusbio.com/0111 INTRODUCTION Ingenio Electroporation
Protocol for MIR 08, 09, 01, 0111, 0112, 0113, 0114, 011, 0116, 0117, 0118, 0119 Quick Reference Protocol, SDS and Certificate of Analysis available at mirusbio.com/0111 INTRODUCTION Ingenio Electroporation
INNOVATIVE PRODUCTS FOR NEUROSCIENCE RESEARCH
 INNOVATIVE PRODUCTS FOR NEUROSCIENCE RESEARCH NEURAL CELLS & MEDIA STEM CELLS & CULTURE REAGENTS TRANSFECTION CUSTOM SERVICES NEUROSCIENCE PRODUCTS CATALOG TABLE OF CONTENTS Neuroscience research demands
INNOVATIVE PRODUCTS FOR NEUROSCIENCE RESEARCH NEURAL CELLS & MEDIA STEM CELLS & CULTURE REAGENTS TRANSFECTION CUSTOM SERVICES NEUROSCIENCE PRODUCTS CATALOG TABLE OF CONTENTS Neuroscience research demands
Structure and content of an IMPD. What is required for first into man trial?
 What is required for first into man? The EU IMPD Thomas Sudhop, MD Scope Structure and content of an IMPD What is required for first into man trial? Only for IMPs that do not have a marketing authorisation
What is required for first into man? The EU IMPD Thomas Sudhop, MD Scope Structure and content of an IMPD What is required for first into man trial? Only for IMPs that do not have a marketing authorisation
For identifying inhibitors and activators of mitochondrial biogenesis in adherent cultured cells.
 ab110216 MitoBiogenesis TM In-Cell ELISA Kit (IR) Instructions for Use For identifying inhibitors and activators of mitochondrial biogenesis in adherent cultured cells. This product is for research use
ab110216 MitoBiogenesis TM In-Cell ELISA Kit (IR) Instructions for Use For identifying inhibitors and activators of mitochondrial biogenesis in adherent cultured cells. This product is for research use
Universal Methyltransferase Activity Assay Kit
 ab139433 Universal Methyltransferase Activity Assay Kit Instructions for Use A complete kit for the screening of candidate compounds that may alter normal Methyltransferase activity. This product is for
ab139433 Universal Methyltransferase Activity Assay Kit Instructions for Use A complete kit for the screening of candidate compounds that may alter normal Methyltransferase activity. This product is for
Tips for Multiplexing Cell-Based Assays:
 Tips for Multiplexing Cell-Based Assays: Plan for success Fall 2010 Click this icon to view speakers notes for each slide. 2010, Promega Corporation. Considerations for Successful Cell-Based Assays Assay
Tips for Multiplexing Cell-Based Assays: Plan for success Fall 2010 Click this icon to view speakers notes for each slide. 2010, Promega Corporation. Considerations for Successful Cell-Based Assays Assay
Rat TNF-alpha ELISA Kit(KT20661) User Manual. For research use only. Not intended for diagnostic testing.
 Rat TNF-alpha ELISA Kit(KT20661) User Manual For research use only. Not intended for diagnostic testing. www.abgent.com TABLE OF CONTENTS I. Introduction...2 II. Reagents.... 2 III. Storage.. 3 IV. Additional
Rat TNF-alpha ELISA Kit(KT20661) User Manual For research use only. Not intended for diagnostic testing. www.abgent.com TABLE OF CONTENTS I. Introduction...2 II. Reagents.... 2 III. Storage.. 3 IV. Additional
Testing of chemicals for classification according to REACH and GHS
 Testing of chemicals for classification according to REACH and GHS Norbert Bornatowicz, Head Toxicology Seibersdorf Labor GmbH 2444 Seibersdorf, AUSTRIA Overview on the presentation Classification: IUCLID
Testing of chemicals for classification according to REACH and GHS Norbert Bornatowicz, Head Toxicology Seibersdorf Labor GmbH 2444 Seibersdorf, AUSTRIA Overview on the presentation Classification: IUCLID
How antimicrobial agents work
 Physical and Chemical Control of Microbes Physical Agents heat or radiation Chemical Agents disinfectants or antiseptics Important Terms 1. Sterilization process of killing all viable microbes 2. Bactericide
Physical and Chemical Control of Microbes Physical Agents heat or radiation Chemical Agents disinfectants or antiseptics Important Terms 1. Sterilization process of killing all viable microbes 2. Bactericide
ab Complex I Enzyme Activity Microplate Assay Kit
 ab109721 Complex I Enzyme Activity Microplate Assay Kit Instructions for Use For the analysis of Complex I enzyme activity from Human, Rat, Mouse and Bovine samples This product is for research use only
ab109721 Complex I Enzyme Activity Microplate Assay Kit Instructions for Use For the analysis of Complex I enzyme activity from Human, Rat, Mouse and Bovine samples This product is for research use only
Testing of chemicals for classification according to REACH and GHS
 Testing of chemicals for classification according to REACH and GHS Norbert Bornatowicz, Head Toxicology Seibersdorf Labor GmbH 2444 Seibersdorf, AUSTRIA Overview on the presentation Classification: IUCLID
Testing of chemicals for classification according to REACH and GHS Norbert Bornatowicz, Head Toxicology Seibersdorf Labor GmbH 2444 Seibersdorf, AUSTRIA Overview on the presentation Classification: IUCLID
Introduction to Assay Development
 Introduction to Assay Development A poorly designed assay can derail a drug discovery program before it gets off the ground. While traditional bench-top assays are suitable for basic research and target
Introduction to Assay Development A poorly designed assay can derail a drug discovery program before it gets off the ground. While traditional bench-top assays are suitable for basic research and target
Development of paediatric formulations - points to consider
 Development of paediatric formulations - points to consider Workshop on Paediatric Formulations II London, 8 November 2011 Presented by: Ann Marie Kaukonen Scientific Administrator, Paediatric Medicines,
Development of paediatric formulations - points to consider Workshop on Paediatric Formulations II London, 8 November 2011 Presented by: Ann Marie Kaukonen Scientific Administrator, Paediatric Medicines,
IN VITRO TOXICITY TESTING: TECHNOLOGIES AND GLOBAL MARKETS
 IN VITRO TOXICITY TESTING: TECHNOLOGIES AND GLOBAL MARKETS PHM017E January 2014 Robert Hunter Project Analyst ISBN: 1-56965-661-4 BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215
IN VITRO TOXICITY TESTING: TECHNOLOGIES AND GLOBAL MARKETS PHM017E January 2014 Robert Hunter Project Analyst ISBN: 1-56965-661-4 BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215
Biowaivers and Harmonization Guidelines for Class I and 3 Drugs: Biowaiver Case Studies
 Biowaivers and Harmonization Guidelines for Class I and 3 Drugs: Biowaiver Case Studies Barbara M. Davit, Merck & Co., Inc. 3 rd FDA/PQRI Conference on Advancing Product Quality March 22, 2017 Disclaimer
Biowaivers and Harmonization Guidelines for Class I and 3 Drugs: Biowaiver Case Studies Barbara M. Davit, Merck & Co., Inc. 3 rd FDA/PQRI Conference on Advancing Product Quality March 22, 2017 Disclaimer
Chicken EpithelialGut CellLines 1
 Chicken EpithelialGut CellLines 1 Content 01 Introduction p 3 02 Characterization p 5 03 Infection and inhibition p 6 04 Protein expression system p 8 05 NutriProof p 10 06 Contact p 12 01...which came
Chicken EpithelialGut CellLines 1 Content 01 Introduction p 3 02 Characterization p 5 03 Infection and inhibition p 6 04 Protein expression system p 8 05 NutriProof p 10 06 Contact p 12 01...which came
amaxa Peptide Transfection Control 2-3 Product specifications 2 Storage and stability 3 Product use limitations 3 Intended use 3
 Contents amaxa Peptide Transfection Control 2-3 Product specifications 2 Storage and stability 3 Product use limitations 3 Intended use 3 Background Information 4 Cell culture 4 Supporting Data 5-8 Equipment
Contents amaxa Peptide Transfection Control 2-3 Product specifications 2 Storage and stability 3 Product use limitations 3 Intended use 3 Background Information 4 Cell culture 4 Supporting Data 5-8 Equipment
TRANSFEX - SUPERIOR GENE EXPRESSION FOR HARD-TO-TRANSFECT CELL TYPES. Kevin Grady Product Line Business Manager ASCB Vendor Showcase Dec.
 TRANSFEX - SUPERIOR GENE EXPRESSION FOR HARD-TO-TRANSFECT CELL TYPES Kevin Grady Product Line Business Manager ASCB Vendor Showcase Dec. 15, 2013 Outline Overview of transfection TransfeX Primary/hTERT
TRANSFEX - SUPERIOR GENE EXPRESSION FOR HARD-TO-TRANSFECT CELL TYPES Kevin Grady Product Line Business Manager ASCB Vendor Showcase Dec. 15, 2013 Outline Overview of transfection TransfeX Primary/hTERT
Material Safety Data Sheet
 Material Safety Data Sheet 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Manufacturer: Mitsubishi Chemical Medience Corp. PATHFAST D-Dimer Address : Regulatory Affairs Dept. (Product No.:
Material Safety Data Sheet 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Manufacturer: Mitsubishi Chemical Medience Corp. PATHFAST D-Dimer Address : Regulatory Affairs Dept. (Product No.:
European Medicines Agency Evaluation of Medicines for Human Use COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT
 European Medicines Agency Evaluation of Medicines for Human Use London, 27 July 2005 EMEA/CHMP/89249/2004 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT Guideline on the Clinical Investigation
European Medicines Agency Evaluation of Medicines for Human Use London, 27 July 2005 EMEA/CHMP/89249/2004 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) DRAFT Guideline on the Clinical Investigation
Cellometer Vision CBA
 Features of the Vision CBA Image Cytometry System All-in-One System Basic cell counting, primary cell viability, and cellbased assays. See for Yourself Why the Top Ten Pharmaceutical Companies Trust Cellometer
Features of the Vision CBA Image Cytometry System All-in-One System Basic cell counting, primary cell viability, and cellbased assays. See for Yourself Why the Top Ten Pharmaceutical Companies Trust Cellometer
