Molecular Characterization Indian Isolates of Pasteurella multocida Isolated from Buffalo and Cattle in India
|
|
|
- Merilyn West
- 5 years ago
- Views:
Transcription
1 International Journal of Current Microbiology and Applied Sciences ISSN: Volume 5 Number 9 (2016) pp Journal homepage: Original Research Article Molecular Characterization Indian Isolates of Pasteurella multocida Isolated from Buffalo and Cattle in India Archana Yadav 1, Anita Sharma 1, Rajesh Kumar 2, V.D.P. Rao 2 and M.K. Saxena 3 * 1 Department of Microbiology, C.B.S.H, 2 Department of Veterinary Microbiology, C.V.Sc 3 Animal Biotechnology Centre, Department of Veterinary Physiology and Biochemistry College of Veterinary and Animal Science, G. B. Pant University of Agriculture &Technology, Pantnagar , India *Corresponding author A B S T R A C T K e y w o r d s Pasteurella multocida, palindromic REP- PCR Haemorrhagic Septicaemia Article Info Accepted: 15 August 2016 Available Online: 10 September 2016 The applicability of repetitive palindromic sequences based differentiation of Pasteurella multocida serogroup B isolates involved in outbreak of haemorrhagic septesemia (HS) in Tarai region of India was evaluated. Seven isolates of Pasteurella multocida B: 2 were isolated from buffaloes and cattle. These were subjected to phenotypic and genotypic characterization. PCR assays were used for the characterization of these isolates. Repetitive extragenic palindromic REP- PCR and Enterobacterial repetitive intergenic consensus ERIC- PCR differentiated all the seven isolates into six profiles. All the isolates were found genetically distinct from standard Pasteurella multocida strain P52 (Vaccine strain of India).Two isolates shared same profile while all other isolates shown different profiles. This study provides a clear evidence of presence of more than one isolate in single outbreak of HS and also provide indication of high genetic variation among field isolates of Pasteurella and may be the reason of vaccine failure and outbreaks. Introduction Haemorrhagic Septicaemia (HS) is an important disease cattle and buffalo in India. It is by Pasteurella multocida types B: 2, B: 2, 5 and B: 5 and has high mortality rate. Conventional methods of disease diagnosis are time consuming as it requires isolation identification and differentiation through bacteriological techniques. With the advancement of molecular biology Polymerase chain reaction (PCR)-based techniques has been extensively used for detection and differentiation of pathogens (Shivshankar et al., 2001; Saxena et al., 2006; Shivchandra et al., 2006; Saxena et al., 2004, Nagagapa et al., 2007). HSB-PCR (Haemorrhagic Septicaemia serotype B specific PCR) is rapid, specific and highly sensitive, are being employed efficiently for early detection (Towensend et al., 1998) and differentiation of various isolates of P. multocida B:2 from single and different outbreaks (Biswas et al., 2004). 302
2 In the present study HS suspected animals were screened using HSB-PCR. Seven isolates were isolated from affected animals (Cattle and Buffalo). These were characterized by using conventional (biochemical, pathogenicity testing, antibiotic analysis) and molecular markers. Two DNA markers were used these were Enterobacterial repetitive intergenic consensus sequence (ERIC) and Repetitive extragenic palindromic sequence (REP) these are repetitive sequences present in family Enterobacteriacae. Out ward primers for these sequences were developed by Verosalovic et al and field isolates of Pasteurella multocida were differentiated on basis of copy number and inter sequential distances (Loubinoux et al., 1998; Saxena et al., 2006). In present study investigations was carried out during an outbreak of haemorrhagic septesemia to study genetic variation among field isolates using conventional and molecular markers with respect to design a strategic control measures. Materials and Methods Screening of samples by HSB-PCR Preparation of culture lysate Blood samples (5 ml each) and nasal discharges were collected from affected animals and healthy. Samples were inoculated in 2 ml of BHI broth for 18 hrs at 37 0 C. 1.5 ml of 18hrs growth was subjected for centrifugation. Pellet was resuspended in 100µl sterilized triple distilled water. Then tubes were boiled at C for 10 minutes and chilled immediately in ice. The tubs were centrifuged at 5000 rpm for 10 minutes to remove cell debris. 5µl of the supernatant was used as template for 25 µl of PCR reaction. HSB-PCR PCR was conducted by using KTSP 61 and KTT 72 primers (Towensend et al.1998) for the detection of Pasteurella multocida serotype B. 25 µl reaction mixture containing 5 µl of lysate, 1X PCR buffer, 1.5 mm MgCl2, 200 µm of each of dntps, 20pmol of each primer and 1 U Taq DNA polymerase was prepared. All PCR reagents were procured from MBI-Fermentas.PCR was performed with an initial denaturation step (94 0 C for 5 min) followed by 30 cycles of denaturation (94 0 C for 1 min), annealing (55 0 C for 1 min) and extension (72 0 C for 1 min, with a final) extension at 72 0 C for 10 min. Amplified product were separated by agarose gel electrophoresis (1.5% agarose in 1X TAE) at 5V/cm for 2h and stained with ethidium bromide (0.5 µg / ml). DNA fragments were observed by UV transilluminator and images were stored using gel documentation system (AlphaImager 2200 Documentation and Analysis System, Alpha Innotech Corporation, USA). Isolation of P. multocida serotype B from positive samples Positive lysates were inoculated in mice by intraperitoneal route. Heart blood was collected from died mice and streaked on plate of blood agar. Isolated single colony was used for methylene blue staining and HSB-PCR. Biochemical test Each isolate was tested for oxidase, indole and catalase tests. Growth of colony was observed on McConkey's agar and on blood agar. All the isolates were tested for fermentation of sugars glucose, fructose, 303
3 maltose, mannitol, sucrose and lactose. The isolates were tested for utilization of citrate, and gelatin. Antimicrobial sensitivity test Isolates were tested for their sensitivity against antimicrobial agents. The antibiograms was determined using a total of eleven different antibiotic discs (amoxycillin, 30 µg, pefloxacin, 5 µg, cephalexin, 30 µg, enrofloxacin, 10 µg, ciprofloxacin, 30 µg, streptomycin, 10 µg, gentamycin, 30 µg; erythromycin, 15 µg; oxytetracyclin,30 µg, chloramphenicol, 30 µg, sulphamithazole, 300 µg) on BHI agar according to disc diffusion method described by Carter and Subronto (1973). Differentiation of isolates by ERIC-PCR and REP-PCR Genomic DNA of all the isolates was extracted as per the C-Tab method described by Wilson (1987). REP-PCR was performed as per protocol described by Saxena et al., (2006) with some variations. Briefly, 25µl reaction mixture containing 50ng of genomic DNA and 20pmol of each primer in 1X PCR buffer, 1.5 mm MgCl 2, 200µM of each of the four dntps and 0.5U Taq polymerase.pcr was performed with an initial denaturation step (94 0 C for 5 min.)followed by 30 cycles of denaturation (94 0 C for 1 min.), annealing (45 0 C for 2 min.) and extension(72 0 C for 6 min.), with a final extension at 72 0 C for 10 min. Amplicons were loaded on 1.5% agarose and subjected for electrophoresis at 5V/cm for 2hrs. 100bp ladder was used as size marker. Fingerprints were stored in taggedimage filed (TIF) format with Bio ID software (Vilber Lourmat, Paris, France). Two fingerprints were considered identical if the same number of bands at the same positions were observed. ERIC-PCR was also conducted as per described by Biswas et al., (2004) with some variations. A reaction mixture of 25µl containing 50ng of genomic DNA and 20pmol of each primer in 1X PCR buffer, 1.5 mm MgCl2, 200µmol/l of each of the 4 dntps and 0.5U Taq polymerase.pcr was performed with an initial denaturation step (94 0 C for 5 min.)followed by 30 cycles of denaturation (94 0 C for 1 min.), annealing (45 0 C for 2 min.) and extension(72 0 C for 6 min.), with a final extension at 72 0 C for 10 min. Amplicons were loaded on 1.5% agarose and subjected for electrophoresis at 5V/cm for 2hrs. Results and Discussion Isolation and phenotypic characterization A total of seven strains were confirmed by HSB-PCR as P. multocida, serotype B these were designated P1, P2, P3, P4, P5, P6 and P7. All the seven isolates gave amplified products of ~620bp determining the products specific for HS causing serogroup B isolates (Figure 1). The isolated bacterial colonies on blood agar plates were small, glistening, mucoid and dewdrop- like and appeared to be Gramnegative coccobacilli when stained with Gram's stain. Agglutination was produced on serological testing with antiserum raised against whole cell antigen of P. multocida (B:2). All of the seven isolates were oxidase and catalase positive and urease negative. No growth was observed on McConkey's agar and were found to be non haemolytic on blood agar. All the isolates fermented glucose, fructose, maltose, mannitol and sucrose, but did not fermented lactose. The isolates were positive for indole test and no reaction was seen with citrate, and gelatin. 304
4 Antimicrobial sensitivity test The results indicated that all the isolates exhibited drug resistance against amoxicillin and were sensitive for enrofloxacin followed by pefloxacin, ciprofloxacin, gentamycin, erythromycin, oxytertracyclin, choramphenicol, sulphamethazole, streptomycin, cephalexin. In ERIC-PCR, amplified products were observed in all the isolates (Figure 2). However, out of the seven, five profiles were obtained and the products size varied from 128 to In ERIC type I isolates (P1 and P5) were present. In ERIC type II isolates (P4 and P6) were present. In ERIC type III, IV and V isolates P2, P3 and P7 were present respectively. Band of 488 bp were observed in all seven isolates. REP-PCR analysis of all the seven isolates revealed five different profiles (Figure 3). In REP type I isolates (P2 and P3) were present. In REP type II isolates (P4 and P6) were present. In REP type III, IV and V isolates P1, P5 and P7 were present respectively. Two bands of sizes 1080 and 784 bp were amplified in all the isolates. These may be Pasteurella-specific loci and can be exploited for the identification of P. multocida. In both ERIC and REP-PCR P52 vaccine strain revealed entirely different profile. ERIC and REP profile data was analyzed using NTSYSpc v2.0 and dendogram was constructed. On combined typing out of seven, six different profiles were observed. P4 and P6 were showing common profile. (Fig. 4). In north India the disease assume to be epizootic during rainy season (June- September) (Khera, 1979) The attempts to isolates a causative agent from various animals resulted in isolation of a few P. multocida strain from cattle and buffaloes. Though more than hundred samples were collected but few isolates were isolated. The failure to isolate the organism from many cases could be due to administration of antimicrobial drug prior to sample collection. The isolates could not be differentiated on the basis of conventional methods involving culture, pathogenicity test and biochemical characterization. Earlier similar types of finding were reported from India (Biswas et al., 2004). Isolates revealed resistance for amoxicillin which is very commonly used in India. Though isolates were found to be sensitive for many other drugs (enrofloxacin) because these are costly and not so routinely used in field cases (Yoshimura et al., 2001). Table.1 Primers sequences used in the PCR technique for identification and characterization S. No Type of PCR HSB-PCR REP PCR ERICPCR KTT72 KTSP61 REP1R 1Dt- REP2 1Dt- ERIC 1R- ERIC2- Sequences of primers used 5'AGG CTC GTT TGG ATT ATG AAG-3' 5'ATC CGC TAA CAG ACT CTC-3' 5' NNN NCG NCG NCA TCN GGC-3' 5' NCG NCT TAT CNG GCC TAC-3' 5' ATG TAA GCT CCT GGG GAT CAC-3' 5' AAG TAA GTG ACT GGG GTG AGC G-3' References Townsend et al. (1998) Versalovic et al.(1991) Versalovic et al.(1991) 305
5 Table.2 ERIC PCR profile of field isolates of Pasteurella multocida P1 P2 P3 P4 P5 P6 P7 P Table.3 REP-PCR profile of field isolates of Pasteurella multocida P1 P2 P3 P4 P5 P6 P7 P Table.4 Combined REP-ERIC profiles of isolates of P. multocida Combined profiles Strain a b c d e f g H i J k L m n o P q r s T no. P P P P P P P P a, 3649 bp; b, 2456 bp; c, 2142 bp; d, 2012 bp; e, 1860 bp; f, 1580 bp; g, 1147 bp; h, 1080 bp; i, 931 bp; j, 899bp; k, 821 bp; l, 784 bp; m, 680 bp; n, 488 bp; o,446 bp; p, 340 bp; q, 300 bp; r, 250 bp; s, 170 bp; t, 128 bp; 0,band present; 1, band absent. 306
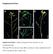
6 Fig.1 Fig.2 307
7 Fig.3 Fig.4 Dendogram of isolates of P. multocida based on combined ERIC and REP profiles. The HSB-PCR method simplifies characterization of type B isolates by detecting all three HS associated serotypes in a single process. The versatility of this assay will enable the technique to be used for routine diagnosis in countries not currently able to perform conventional methods (Saxena et al., 2002). In present study molecular methods were found to be of more efficient than conventional methods. In combined molecular typing using data produced by ERIC and REP PCR D value of 0.95 was obtained. 308
8 Our findings indicate that in outbreak the isolates which were involved, were from different origin and genetic makeup. In dendogram it can visualized that the isolates of one location are more closely related. Similar findings have been reported from India (Biswas et al., 2004) and other countries (Gunuwardana et al., 2000) (Towensend et al., 1997). Generally, oil adjuvant vaccine or alumprecipitated and aluminium hydroxide gel vaccines with a vaccine strain of P. multocida (P52) are currently used in India for control of HS. The variation at molecular level in the field isolates, in comparison to the vaccine strain, warrants an imminent strategy for controlling HS. In conclusion, we have shown H.S. is still an important disease spreading in rainy season. PCR based screening can be used effectively for detection of HS-associated serotypes of P. multocida (B:2, B:5 or B:2,5). The present investigation indicates that genetic variation of isolates of same or different origin can be visualized by ERIC and REP- PCR. References Biswas, A., S.B. Shivchandra, M.K. Saxena, A. Kumar, V.P. Singh, and Srivastava, S.K Molecular variability among the strains of Pasteurella multocida solated from an outbreak of haemorrhagic septicaemia in India. Vet. Res. Commu., 28: Carter, G.R. and Subronto, P Identification of type D strains of P. multocida with acriflavin. American J. Vet. Res., 34: Gunawardana, G.A., K.M. Townsend and Frost, A.J Molecular characterization of avian Pasteurella multocida isolates from Australia and Vietnam by REP-PCR and PFGE. Vet. Microbiol., 72: Khera, S.S The incidence and distribution of epizootic diseases in India: hemorrhagic septicemia. Bulletin of office International des Epizootics. 91 : Loubinoux, J., C. Lion, D. Grain, M. Weba, and Faou, L.E Value of ERIC- PCR for study of Pasteurella multocida strains isolated from mouth of dogs. J. Clin. Microbiol., 37: Naggapa, K., S.P. Singh, S. Tamuly, and Saxena, M.K Isolation of Salmonella. Saxena, M.K., V.P. Singh, A.A. Kumar, P. Choudhuri, V.P. Singh, S.B. Shivachandra, A. Biswas and Sharma, B REP-PCR analysis of P. multocida isolates from wild and domestic animals in India. Vet. Res. Commu., 30: Saxena, M.K., V.P. Singh, B.D. Lakhchura, A. Saxena and Shrama, B Detection of virulence in Indian Isolates of Salmonella by Polymerase Chain Reaction. Indian J. Biotechnol., 3: Shivachandra, S.B., A.A. Kumar, R. Gautam, S. Joseph, P. Chaudhuri, M.K. Saxena and Srivastav, S.K Detection of Pasteurella multocida in experimental infected embryonated chicken eggs by PCR assay. Indian J. Experimental Biol., 44: Shivshankara, N., M.K. Saxena and Singh V.P Rapid diagnosis of hemorrhagic septicemia by PCR assay. Indian Vet. J., 78: Townsend, K.M., H.J. Dawkins, and Papadimitriou, J.M REP PCR analysis of Pasteurella multocida isolates that cause hemorrhagic septicemia. Res. Vet. Sci., 63: Townsend, K.M., A.J. Frost, C.W. Lee, J. 309
9 M. Papadimitriou, and Dawkins,. H. J Development of polymerase chain reaction (PCR) assays for species and type-specific identification of P. multocida. J. Clin. Microbiol., 36: Typhimurium from poultry eggs and meat of Tarai region of Uttranchal. Indian J. Biotechnol., 6: Versalovic, J., T. Koeuth, and Lupski, J.R Distribution of repetitive DNA sequences in eubacteria and application of fingerprinting of bacterial genomes. Nucleic Acid Res., 19: Wilson, K Preparation of genomic DNA from bacteria. In: F.M. Ausabel, R.Brent, R.E. Kingston, D. D. Moore, J.G. Seidman, J.A. Smith and k. Struhl (ads). Current Protocols in Molecular Biology. unit, 2.4.1,(Wiley, New York) Yoshimura, H., M.Ishmaru, Y. S Endoh and Kojima, A Antimicrobial susceptibility of P. multocida isolated from cattle and pigs. J. Vet. Med. Infect. Dis. Vet. Public Health, 48: How to cite this article: Archana Yadav, Anita Sharma, Rajesh Kumar, V.D.P. Rao and M.K. Saxena Molecular Characterization Indian Isolates of Pasteurella multocida Isolated from Buffalo and Cattle in India. Int.J.Curr.Microbiol.App.Sci. 5(9): doi: 310
COMPARISON OF DETECTION THRESHOLD OF DIFFERENT PASTEURELLA MULTOCIDA SPECIFIC PCRs
 Indian J. Anim. Res., 46 (1) : 28-33, 2012 AGRICULTURAL RESEARCH COMMUNICATION CENTRE www.arccjournals.com / indianjournals.com COMPARISON OF DETECTION THRESHOLD OF DIFFERENT PASTEURELLA MULTOCIDA SPECIFIC
Indian J. Anim. Res., 46 (1) : 28-33, 2012 AGRICULTURAL RESEARCH COMMUNICATION CENTRE www.arccjournals.com / indianjournals.com COMPARISON OF DETECTION THRESHOLD OF DIFFERENT PASTEURELLA MULTOCIDA SPECIFIC
Differentation of closely related Vaccinal Strains of Pasteurella multocida using Polymerase Chain Reaction(PCR)
 Article 36 Differentation of closely related Vaccinal Strains of Pasteurella multocida using Polymerase Chain Reaction(PCR) Waheed ullah 1, Muhammad Abubakar* 2, Muhammad Javed Arshed 2, Syed Muhammad
Article 36 Differentation of closely related Vaccinal Strains of Pasteurella multocida using Polymerase Chain Reaction(PCR) Waheed ullah 1, Muhammad Abubakar* 2, Muhammad Javed Arshed 2, Syed Muhammad
Cat. # Product Size DS130 DynaExpress TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1
 Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
MacBlunt PCR Cloning Kit Manual
 MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
PCR Laboratory Exercise
 PCR Laboratory Exercise Advance Protocol (updated 1/2018) Introduction Detection of TPA-25 Alu by PCR A Human DNA Fingerprinting Lab Protocol 1994 Cold Spring Harbor Laboratory DNA Learning Center In this
PCR Laboratory Exercise Advance Protocol (updated 1/2018) Introduction Detection of TPA-25 Alu by PCR A Human DNA Fingerprinting Lab Protocol 1994 Cold Spring Harbor Laboratory DNA Learning Center In this
Molecular characterization and phylogenetic analysis of protease producing Streptomyces sp. isolated from mangrove sediments
 Molecular characterization and phylogenetic analysis of protease producing Streptomyces sp. isolated from mangrove sediments M. Parthasarathy and J. Joel Gnanadoss* Department of Plant Biology and Biotechnology,
Molecular characterization and phylogenetic analysis of protease producing Streptomyces sp. isolated from mangrove sediments M. Parthasarathy and J. Joel Gnanadoss* Department of Plant Biology and Biotechnology,
Rep-PCR tests for identification of bacteria
 European and Mediterranean Plant Protection Organization PM 7/100 (1) Organisation Européenne et Méditerranéenne pour la Protection des Plantes Diagnostics Diagnostic Rep-PCR tests for identification of
European and Mediterranean Plant Protection Organization PM 7/100 (1) Organisation Européenne et Méditerranéenne pour la Protection des Plantes Diagnostics Diagnostic Rep-PCR tests for identification of
Mochamad Nurcholis. Food Technology Department Agricuktural Technology Faculty Brawijaya University 2013
 Mochamad Nurcholis Food Technology Department Agricuktural Technology Faculty Brawijaya University 2013 Microbial Identification Type Conventional Identification DNA Hybridization PCR Amplification DNA
Mochamad Nurcholis Food Technology Department Agricuktural Technology Faculty Brawijaya University 2013 Microbial Identification Type Conventional Identification DNA Hybridization PCR Amplification DNA
Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH).
 Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Pasteurella multocida
 BACTOTYPE PCR Amplification Kit Pasteurella multocida Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification of
BACTOTYPE PCR Amplification Kit Pasteurella multocida Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification of
FMF NIRCA PROTOCOL STEP 1.
 FMF NIRCA PROTOCOL STEP 1. After you have isolated patient s DNA and DNA from a healthy donor (wild type), you perform a nested PCR. The primers used to amplify exon 2 and exon 10 of the mefv gene are
FMF NIRCA PROTOCOL STEP 1. After you have isolated patient s DNA and DNA from a healthy donor (wild type), you perform a nested PCR. The primers used to amplify exon 2 and exon 10 of the mefv gene are
HiPer Random Amplification of Polymorphic DNA (RAPD) Teaching Kit
 HiPer Random Amplification of Polymorphic DNA (RAPD) Teaching Kit Product Code: HTBM031 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 3.5 hours Agarose Gel Electrophoresis:
HiPer Random Amplification of Polymorphic DNA (RAPD) Teaching Kit Product Code: HTBM031 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 3.5 hours Agarose Gel Electrophoresis:
This Product Description is only valid for Lot No. 36V.
 HLA Wipe Test Negative Control Product Insert Page 1 of 8 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01 including Taq polymerase Product number: 102.101-01u without Taq polymerase
HLA Wipe Test Negative Control Product Insert Page 1 of 8 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01 including Taq polymerase Product number: 102.101-01u without Taq polymerase
qpcr Kit, DNA-free Product components 100 rxn 250 rxn Product description
 qpcr Kit, DNA-free For the PCR detection and identification of bacterial and fungal DNA using custom primers Product code A8514 Product components 100 rxn 250 rxn A 2.5x mastermix (3 mm MgCl 2 final concentration)
qpcr Kit, DNA-free For the PCR detection and identification of bacterial and fungal DNA using custom primers Product code A8514 Product components 100 rxn 250 rxn A 2.5x mastermix (3 mm MgCl 2 final concentration)
Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis *
 Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis * Laboratory of Pediatric Infectious Diseases, Department of Pediatrics and Laboratory of Medical Immunology,
Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis * Laboratory of Pediatric Infectious Diseases, Department of Pediatrics and Laboratory of Medical Immunology,
Taxonomy. Classification of microorganisms 3/12/2017. Is the study of classification. Chapter 10 BIO 220
 Taxonomy Is the study of classification Organisms are classified based on relatedness to each other Chapter 10 BIO 220 Fig. 10.1 1 Species Binomial nomenclature for species identification A eukaryotic
Taxonomy Is the study of classification Organisms are classified based on relatedness to each other Chapter 10 BIO 220 Fig. 10.1 1 Species Binomial nomenclature for species identification A eukaryotic
Engineering D66N mutant using quick change site directed mutagenesis. Harkewal Singh 09/01/2010
 Engineering D66N mutant using quick change site directed mutagenesis Harkewal Singh 09/01/2010 1 1- What is quick change site directed mutagenesis? 2- An overview of the kit contents. 3- A brief information
Engineering D66N mutant using quick change site directed mutagenesis Harkewal Singh 09/01/2010 1 1- What is quick change site directed mutagenesis? 2- An overview of the kit contents. 3- A brief information
Sequencing of DNA lesions facilitated by site-specific excision via base. excision repair DNA glycosylases yielding ligatable gaps
 Supporting information Sequencing of DNA lesions facilitated by site-specific excision via base excision repair DNA glycosylases yielding ligatable gaps Jan Riedl, Aaron M. Fleming, and Cynthia J. Burrows*
Supporting information Sequencing of DNA lesions facilitated by site-specific excision via base excision repair DNA glycosylases yielding ligatable gaps Jan Riedl, Aaron M. Fleming, and Cynthia J. Burrows*
Biotechnology Research Training
 Biotechnology Research Training NTHRYS provides Biotechnology Research Training for interested candidates at its Hyderabad facility, Telangana. Please refer below for more details including Fee strctures,
Biotechnology Research Training NTHRYS provides Biotechnology Research Training for interested candidates at its Hyderabad facility, Telangana. Please refer below for more details including Fee strctures,
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Assessment of Intra-Serotypic Diversity in Escherichia coli from Dogs Using ERIC-PCR
 DOI: 10.5958/2277-940X.2017.00104.8 Journal of Animal Research: v.7 n.4, p. 679-683. August 2017 Assessment of Intra-Serotypic Diversity in Escherichia coli from Dogs Using ERIC-PCR N. Mohammad Sharif*,
DOI: 10.5958/2277-940X.2017.00104.8 Journal of Animal Research: v.7 n.4, p. 679-683. August 2017 Assessment of Intra-Serotypic Diversity in Escherichia coli from Dogs Using ERIC-PCR N. Mohammad Sharif*,
strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular
![strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular](/thumbs/72/66361212.jpg) Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Supplemental material
 Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Molecular Identification of Key Mastitis causing Bacteria in Dairy Goats in Kenya.
 Molecular Identification of Key Mastitis causing Bacteria in Dairy Goats in Kenya. R. S. Shivairo 1, J. Matofari 2, C. I. Muleke 3, P. K. Migwi 4, E. Lugairi 1. Department of Clinical Studies, P.O. Box
Molecular Identification of Key Mastitis causing Bacteria in Dairy Goats in Kenya. R. S. Shivairo 1, J. Matofari 2, C. I. Muleke 3, P. K. Migwi 4, E. Lugairi 1. Department of Clinical Studies, P.O. Box
Y-chromosomal haplogroup typing Using SBE reaction
 Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
HiPer RT-PCR Teaching Kit
 HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
Blood direct 2x PCR Mastermix. Data sheet. Order No. BS reactions x 20 µl. (For research and in vitro applications only) Batch No.
 Data sheet Order No. BS91.222.0250 250 reactions x 20 µl Order No. BS91.222.1250 1250 reactions x 20 µl (For research and in vitro applications only) Batch No.: Best before: Appearance: Colour: 1 Description
Data sheet Order No. BS91.222.0250 250 reactions x 20 µl Order No. BS91.222.1250 1250 reactions x 20 µl (For research and in vitro applications only) Batch No.: Best before: Appearance: Colour: 1 Description
RAPD-PCR and SDS-PAGE analysis of Aeromonas hydrophila for defining molecular characterization
 Trade Science Inc. ISSN : 0974-7435 Volume 6 Issue 1 BTAIJ, 6(1), 2012 [22-26] RAPD-PCR and SDS-PAGE analysis of Aeromonas hydrophila for defining molecular characterization Agniswar Sarkar 1 *, Mousumi
Trade Science Inc. ISSN : 0974-7435 Volume 6 Issue 1 BTAIJ, 6(1), 2012 [22-26] RAPD-PCR and SDS-PAGE analysis of Aeromonas hydrophila for defining molecular characterization Agniswar Sarkar 1 *, Mousumi
Molecular characterization of Pasteurella multocida isolates obtained from poultry, ruminant, cats and dogs using RAPD and REP-PCR analysis
 Molecular Biology Research Communications 2016;5(3):123-132 Original Article Open Access Molecular characterization of Pasteurella multocida isolates obtained from poultry, ruminant, cats and dogs using
Molecular Biology Research Communications 2016;5(3):123-132 Original Article Open Access Molecular characterization of Pasteurella multocida isolates obtained from poultry, ruminant, cats and dogs using
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Pr oject Summar y. Rapid quantification of culturable and viable-but-nonculturable Escherichia coli O157:H7 in beef products using EMA-Real Time PCR
 Pr oject Summar y Rapid quantification of culturable and viable-but-nonculturable Escherichia coli O17:H7 in beef products using EMA-Real Time PCR Principal Investigator: Azlin Mustapha University of Missouri
Pr oject Summar y Rapid quantification of culturable and viable-but-nonculturable Escherichia coli O17:H7 in beef products using EMA-Real Time PCR Principal Investigator: Azlin Mustapha University of Missouri
Table of contents. I. Description...2. Kit Components...2. Storage...2. Required reagents and equipment...2. V. Protocol...3. Example Experiment...
 PCR FLT3/ITD Mutation Detection Set Cat.# 6632 Table of contents I. Description...2 II. III. IV. Kit Components...2 Storage...2 Required reagents and equipment...2 V. Protocol...3 VI. XII. Example Experiment...4
PCR FLT3/ITD Mutation Detection Set Cat.# 6632 Table of contents I. Description...2 II. III. IV. Kit Components...2 Storage...2 Required reagents and equipment...2 V. Protocol...3 VI. XII. Example Experiment...4
Chitwambi Makungu, DVM 2 John Yabe, DVM 2 Martin Simuunzab, DVM 2 Andrew Nambota, DVM 2 Hiroshi Isogai, DVM 3 Hideto Fukushi, DVM 4 Jun Yasuda, DVM 5
 Rapid Detection of Salmonella on Commercial Carcasses by Using Isothermal and Chimeric Primer- Initiated Amplification of Nucleic Acids (ICAN)-Enzyme-Linked Immunosorbent Assay (ELISA) in Zambia Emiko
Rapid Detection of Salmonella on Commercial Carcasses by Using Isothermal and Chimeric Primer- Initiated Amplification of Nucleic Acids (ICAN)-Enzyme-Linked Immunosorbent Assay (ELISA) in Zambia Emiko
Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC
 Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Legends for supplementary figures 1-3
 High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
Sexing Bovine Preimplantation Embryos by PCR
 Sexing Bovine Preimplantation Embryos by PCR Katherine E.M. Hendricks 1, Leydson F. Martins 2, Justin M. Fear 1 and Peter J. Hansen 1 1 Dept. of Animal Sciences, University of Florida and Department of
Sexing Bovine Preimplantation Embryos by PCR Katherine E.M. Hendricks 1, Leydson F. Martins 2, Justin M. Fear 1 and Peter J. Hansen 1 1 Dept. of Animal Sciences, University of Florida and Department of
Event-specific Method for the Quantification of Soybean SYHT0H2 by Real-time PCR. Validated Method
 EUROPEAN COMMISSION JOINT RESEARCH CENTRE Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification of Soybean SYHT0H2 by Real-time PCR
EUROPEAN COMMISSION JOINT RESEARCH CENTRE Institute for Health and Consumer Protection Molecular Biology and Genomics Unit Event-specific Method for the Quantification of Soybean SYHT0H2 by Real-time PCR
DETERMINATION OF THE Rh FACTOR BY PCR
 DETERMINATION OF THE Rh FACTOR BY PCR Ref.: PCR2 1. EXPERIMENT OBJECTIVE The aim of this experiment is to introduce students to the principles and practice of the Polymerase Chain Reaction (PCR) by studying
DETERMINATION OF THE Rh FACTOR BY PCR Ref.: PCR2 1. EXPERIMENT OBJECTIVE The aim of this experiment is to introduce students to the principles and practice of the Polymerase Chain Reaction (PCR) by studying
Capsular serotyping of Pasteurella multocida from various animal hosts a comparison of phenotypic and genotypic methods
 Tropical Biomedicine 28(1): 55 63 (2011) Capsular serotyping of Pasteurella multocida from various animal hosts a comparison of phenotypic and genotypic methods Arumugam, N.D. 1, Ajam, N. 1, Blackall,
Tropical Biomedicine 28(1): 55 63 (2011) Capsular serotyping of Pasteurella multocida from various animal hosts a comparison of phenotypic and genotypic methods Arumugam, N.D. 1, Ajam, N. 1, Blackall,
High Pure Technology and Silica Adsorption High Pure PCR Product Purification Kit
 for purification of DNA from PCR reactions Cat. No. 1 73 668 (50 purifications) Cat. No. 1 73 676 (50 purifications) Principle In the presence of chaotropic salt, product DNA binds selectively to glass
for purification of DNA from PCR reactions Cat. No. 1 73 668 (50 purifications) Cat. No. 1 73 676 (50 purifications) Principle In the presence of chaotropic salt, product DNA binds selectively to glass
Mycobacterium paratuberculosis
 BACTOTYPE PCR Amplification Kit Mycobacterium paratuberculosis Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification
BACTOTYPE PCR Amplification Kit Mycobacterium paratuberculosis Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification
For in vitro Veterinary Diagnostics only. DNA Extraction and PCR Detection Kit for Pasteurella multocida.
 For in vitro Veterinary Diagnostics only. DNA Extraction and PCR Detection Kit for Pasteurella multocida www.kylt.eu DIRECTION FOR USE Art. No. 31058 / 31059 Kylt Pasteurella multocida DNA Extraction and
For in vitro Veterinary Diagnostics only. DNA Extraction and PCR Detection Kit for Pasteurella multocida www.kylt.eu DIRECTION FOR USE Art. No. 31058 / 31059 Kylt Pasteurella multocida DNA Extraction and
E.Z.N.A. Microorganism Direct PCR Kit
 E.Z.N.A. Microorganism Direct PCR Kit TQ3100-00 TQ3100-01 TQ3100-02 20 preps 100 preps 500 preps June 2013 E.Z.N.A. Microorganism Direct PCR Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage
E.Z.N.A. Microorganism Direct PCR Kit TQ3100-00 TQ3100-01 TQ3100-02 20 preps 100 preps 500 preps June 2013 E.Z.N.A. Microorganism Direct PCR Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage
500U. Unit Definition. Storage Buffer. 10X PCR Buffer with Mg 2+
 Hy-Taq 500U + dntps #EZ1012 500U Concentration: 5U/μl Contents: Hy-Taq DNA Polymerase 100μl 10xPCR Buffer(Mg 2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C For research only
Hy-Taq 500U + dntps #EZ1012 500U Concentration: 5U/μl Contents: Hy-Taq DNA Polymerase 100μl 10xPCR Buffer(Mg 2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C For research only
SuperiorScript III cdna Synthesis Kit Instruction Manual
 SuperiorScript III cdna Synthesis Kit Instruction Manual Cat.# EZ405S, EZ405M SuperiorScript III cdna Synthesis Kit Table of Contents I. Description... 3 II. Kit... 4 III. Procedure... 5 IV. Control Experiment
SuperiorScript III cdna Synthesis Kit Instruction Manual Cat.# EZ405S, EZ405M SuperiorScript III cdna Synthesis Kit Table of Contents I. Description... 3 II. Kit... 4 III. Procedure... 5 IV. Control Experiment
Mastermix 16S Complete, DNA-free
 Mastermix 16S Complete, DNA-free For the PCR detection and identification of bacteria using universal 16S rdna primers For research use only Cat. No. S-020-0100 Cat. No. S-020-0250 Cat. No. S-020-1000
Mastermix 16S Complete, DNA-free For the PCR detection and identification of bacteria using universal 16S rdna primers For research use only Cat. No. S-020-0100 Cat. No. S-020-0250 Cat. No. S-020-1000
Development of Positive Control for Hepatitis B Virus
 Human Journals Research Article December 2015 Vol.:2, Issue:2 All rights are reserved by Saurabh Bandhavkar et al. Development of Positive Control for Hepatitis B Virus Keywords: Hepatitis B virus, pbluescript,
Human Journals Research Article December 2015 Vol.:2, Issue:2 All rights are reserved by Saurabh Bandhavkar et al. Development of Positive Control for Hepatitis B Virus Keywords: Hepatitis B virus, pbluescript,
Puro. Knockout Detection (KOD) Kit
 Puro Knockout Detection (KOD) Kit Cat. No. CC-03 18 Oct. 2016 Contents I. Kit Contents and Storage II. Product Overview III. Methods Experimental Outline Genomic DNA Preparation Obtain Hybrid DNA Digest
Puro Knockout Detection (KOD) Kit Cat. No. CC-03 18 Oct. 2016 Contents I. Kit Contents and Storage II. Product Overview III. Methods Experimental Outline Genomic DNA Preparation Obtain Hybrid DNA Digest
An engineered tryptophan zipper-type peptide as a molecular recognition scaffold
 SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
This Product Description is only valid for Lot No. 42R.
 HLA Wipe Test Negative Control Product Insert Page 1 of 12 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01 including Taq polymerase Product number: 102.101-01u without Taq polymerase
HLA Wipe Test Negative Control Product Insert Page 1 of 12 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01 including Taq polymerase Product number: 102.101-01u without Taq polymerase
DiAGSure Mycobacterium tuberculosis (MTB) Detection Kitit
 DiAGSure Mycobacterium tuberculosis (MTB) Detection Kitit Description: Tuberculosis (TB) is caused by the acid-fast bacterium Mycobacterium tuberculosis. Although Mycobacterium tuberculosis most commonly
DiAGSure Mycobacterium tuberculosis (MTB) Detection Kitit Description: Tuberculosis (TB) is caused by the acid-fast bacterium Mycobacterium tuberculosis. Although Mycobacterium tuberculosis most commonly
Taq + dntps #EZ U
 #EZ1012 500U Taq + dntps Concentration: 5U/μl Contents: Hy-Taq DNA polymerase 100μl 10xPCR Buffer (Mg2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C Description Hy-Taq 500U +
#EZ1012 500U Taq + dntps Concentration: 5U/μl Contents: Hy-Taq DNA polymerase 100μl 10xPCR Buffer (Mg2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C Description Hy-Taq 500U +
Manitoba Livestock Manure Management Initiative Project: MLMMI Tracing the Source of E. coli Fecal Contamination of Water Using rep-pcr.
 Manitoba Livestock Manure Management Initiative Project: MLMMI 00-02-08 Tracing the Source of E. coli Fecal Contamination of Water Using rep-pcr. Final Report Submitted by: Dr. Paul Holloway Aug. 31, 2001
Manitoba Livestock Manure Management Initiative Project: MLMMI 00-02-08 Tracing the Source of E. coli Fecal Contamination of Water Using rep-pcr. Final Report Submitted by: Dr. Paul Holloway Aug. 31, 2001
Olerup SSP HLA Wipe Test Negative Control
 HLA Wipe Test Negative Control Product Insert Page 1 of 12 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01u without Taq polymerase Lot number: 80M Expiry date: 2014-March-01 Number
HLA Wipe Test Negative Control Product Insert Page 1 of 12 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01u without Taq polymerase Lot number: 80M Expiry date: 2014-March-01 Number
Quant One Step RT-PCR Kit
 1. Quant One Step RT-PCR Kit For fast and sensitive one-step RT-PCR www.tiangen.com/en RT121221 Quant One Step RT-PCR Kit Kit Contents Cat. no. KR113 Contents Hotmaster Taq Polymerase (2.5 U/μl) Quant
1. Quant One Step RT-PCR Kit For fast and sensitive one-step RT-PCR www.tiangen.com/en RT121221 Quant One Step RT-PCR Kit Kit Contents Cat. no. KR113 Contents Hotmaster Taq Polymerase (2.5 U/μl) Quant
HCV Genotype Primer Kit
 Instruction Manual for HCV Genotype Primer Kit HCV Genotype Determination Kit for Research Purpose Thoroughly read this instruction manual before use of this kit Background Study of nucleotide sequence
Instruction Manual for HCV Genotype Primer Kit HCV Genotype Determination Kit for Research Purpose Thoroughly read this instruction manual before use of this kit Background Study of nucleotide sequence
English Version. Animal feeding stuffs - PCR typing of probiotic strains of Saccharomyces cerevisiae (yeast)
 TECHNICAL SPECIFICATION SPÉCIFICATION TECHNIQUE TECHNISCHE SPEZIFIKATION CEN/TS 15790 December 2008 ICS 07.100.30; 65.120 English Version Animal feeding stuffs - PCR typing of probiotic strains of Saccharomyces
TECHNICAL SPECIFICATION SPÉCIFICATION TECHNIQUE TECHNISCHE SPEZIFIKATION CEN/TS 15790 December 2008 ICS 07.100.30; 65.120 English Version Animal feeding stuffs - PCR typing of probiotic strains of Saccharomyces
4.0 RESULTS. 4.1 Isolation and identification
 4.0 RESULTS 4.1 Isolation and identification The isolation of s. aureus from the sample of human patients was done at by department of microbiology, Indira Gandhi Medical College, Shimla. They were confirmed
4.0 RESULTS 4.1 Isolation and identification The isolation of s. aureus from the sample of human patients was done at by department of microbiology, Indira Gandhi Medical College, Shimla. They were confirmed
Supplementary Information
 Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
For in vitro Veterinary Diagnostics only. PCR Detection Kit for Salmonella Gallinarum, Pullorum (separate detection) & DIVA 9R.
 For in vitro Veterinary Diagnostics only. PCR Detection Kit for Salmonella Gallinarum, Pullorum (separate detection) & DIVA 9R www.kylt.eu DIRECTION FOR USE Art. No. 31420 / 31421 Kylt SGP & 9R DIVA PCR
For in vitro Veterinary Diagnostics only. PCR Detection Kit for Salmonella Gallinarum, Pullorum (separate detection) & DIVA 9R www.kylt.eu DIRECTION FOR USE Art. No. 31420 / 31421 Kylt SGP & 9R DIVA PCR
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR
 Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
TD-P Revision 3.0 Creation Date: 6/9/2016 Revision Date: 8/16/2018
 Protocol TD-P Revision 3.0 Creation Date: 6/9/2016 Revision Date: 8/16/2018 Electrotransformation of Agrobacterium tumefaciens Modified from Methods in Molecular Biology Vol. 47 Introduction Agrobacterium
Protocol TD-P Revision 3.0 Creation Date: 6/9/2016 Revision Date: 8/16/2018 Electrotransformation of Agrobacterium tumefaciens Modified from Methods in Molecular Biology Vol. 47 Introduction Agrobacterium
Detection of Potential Eco-friendly Microbes to Clean-Up the Polluted Water in Sri Lanka
 Detection of Potential Eco-friendly Microbes to Clean-Up the Polluted Water in Sri Lanka K. Vivehananthan #, Y. W. Manukulasooriya and W. M. J. Y. Ekanayake Dept. of Biotechnology Faculty of Agriculture
Detection of Potential Eco-friendly Microbes to Clean-Up the Polluted Water in Sri Lanka K. Vivehananthan #, Y. W. Manukulasooriya and W. M. J. Y. Ekanayake Dept. of Biotechnology Faculty of Agriculture
2x PCR LongNova-RED PCR Master Mix
 2x PCR LongNova-RED Components RP85L 100 reactions (50 μl) RP85L-10 1000 reactions (50 μl) 2x PCR LongNova-RED 2 x 1.25 ml 20 x 1.25 ml PCR grade water 2 x 1.5 ml 20 x 1.5 ml Storage & Shiing Storage conditions
2x PCR LongNova-RED Components RP85L 100 reactions (50 μl) RP85L-10 1000 reactions (50 μl) 2x PCR LongNova-RED 2 x 1.25 ml 20 x 1.25 ml PCR grade water 2 x 1.5 ml 20 x 1.5 ml Storage & Shiing Storage conditions
DIRECTION FOR USE. Art. No / /25 reactions in vitro Diagnosticum for birds
 DIRECTION FOR USE Art. No. 31066 / 31067 100 /25 reactions in vitro Diagnosticum for birds Kylt Turkey Hemorrhagic Enteritis Virus PCR Detection Kit is for detection of the etiological agent of Infectious
DIRECTION FOR USE Art. No. 31066 / 31067 100 /25 reactions in vitro Diagnosticum for birds Kylt Turkey Hemorrhagic Enteritis Virus PCR Detection Kit is for detection of the etiological agent of Infectious
Lab Book igem Stockholm Lysostaphin. Week 9
 Lysostaphin Week 9 Summarized below are the experiments conducted this week in chronological order. Click on the experiment name to view it. To go back to this summary, click Summary in the footer. Summary
Lysostaphin Week 9 Summarized below are the experiments conducted this week in chronological order. Click on the experiment name to view it. To go back to this summary, click Summary in the footer. Summary
Designing and creating your gene knockout Background The rada gene was identified as a gene, that when mutated, caused cells to become hypersensitive
 Designing and creating your gene knockout Background The rada gene was identified as a gene, that when mutated, caused cells to become hypersensitive to ionizing radiation. However, why these mutants are
Designing and creating your gene knockout Background The rada gene was identified as a gene, that when mutated, caused cells to become hypersensitive to ionizing radiation. However, why these mutants are
Construction of a Mutant pbr322 Using Site-Directed Mutagenesis to Investigate the Exclusion Effects of pbr322 During Co-transformation with puc19
 Construction of a Mutant pbr322 Using Site-Directed Mutagenesis to Investigate the Exclusion Effects of pbr322 During Co-transformation with puc19 IVANA KOMLJENOVIC Department of Microbiology and Immunology,
Construction of a Mutant pbr322 Using Site-Directed Mutagenesis to Investigate the Exclusion Effects of pbr322 During Co-transformation with puc19 IVANA KOMLJENOVIC Department of Microbiology and Immunology,
HLA-DR TYPING OF GENOMIC DNA
 HLA-DR TYPING OF GENOMIC DNA Zofia SZCZERKOWSKA, Joanna WYSOCKA Institute of Forensic Medicine, Medical University, Gdañsk, Poland ABSTRACT: Advances in molecular biology techniques allowed for introduction
HLA-DR TYPING OF GENOMIC DNA Zofia SZCZERKOWSKA, Joanna WYSOCKA Institute of Forensic Medicine, Medical University, Gdañsk, Poland ABSTRACT: Advances in molecular biology techniques allowed for introduction
Supplemental Data Supplemental Figure 1.
 Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
SpeedSTAR HS DNA Polymerase
 Cat. # RR070A For Research Use SpeedSTAR HS DNA Polymerase Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Supplied buffers... 3 V. General reaction mixture...
Cat. # RR070A For Research Use SpeedSTAR HS DNA Polymerase Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Supplied buffers... 3 V. General reaction mixture...
Event-specific Method for the Quantification of Maize Line GA21 Using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Maize Line GA21 Using Real-time PCR Protocol 30 March 2010 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Event-specific Method for the Quantification of Maize Line GA21 Using Real-time PCR Protocol 30 March 2010 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) Fax: (905)
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com E.coli O157:H7 End-Point PCR Kit Product# EP41300 Product Insert
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com E.coli O157:H7 End-Point PCR Kit Product# EP41300 Product Insert
Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were
 1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
THE RAY- MANUAL. Instructions for the construction of complex targeting vectors using RAY (rapid assembly in yeast) Thorsten Storck December '96
 Thorsten Storck December '96 THE RAY- MANUAL Instructions for the construction of complex targeting vectors using RAY (rapid assembly in yeast) Principle of the method Genetic elements (selection markers,
Thorsten Storck December '96 THE RAY- MANUAL Instructions for the construction of complex targeting vectors using RAY (rapid assembly in yeast) Principle of the method Genetic elements (selection markers,
NESTED Sequence-based Typing (SBT) protocol for epidemiological typing of Legionella pneumophila directly from clinical samples
 NESTED Sequence-based Typing (SBT) protocol for epidemiological typing of Legionella pneumophila directly from clinical samples VERSION 2.0 SUMMARY This procedure describes the use of nested Sequence-Based
NESTED Sequence-based Typing (SBT) protocol for epidemiological typing of Legionella pneumophila directly from clinical samples VERSION 2.0 SUMMARY This procedure describes the use of nested Sequence-Based
Table of contents. I. Description II. Kit Components III. Reagents and Instruments Required IV. Storage V. Protocols...
 Table of contents I. Description... 2 II. Kit Components... 2 III. Reagents and Instruments Required... 2 IV. Storage... 2 V. Protocols... 3 VI-1. Procedure... 3 VI-2. Note... 4 VI. Control experiment...
Table of contents I. Description... 2 II. Kit Components... 2 III. Reagents and Instruments Required... 2 IV. Storage... 2 V. Protocols... 3 VI-1. Procedure... 3 VI-2. Note... 4 VI. Control experiment...
Biology 423L Nov. 6/7. Genetics in Forensic Science: Human DNA Fingerprinting Report due Nov. 21
 1 Biology 423L Nov. 6/7 Genetics in Forensic Science: Human DNA Fingerprinting Report due Nov. 21 Readings: Hartwell et al. pp. 297-302, 374-387. Nakamura Y., Carlson, K. Krapco, and R. White 1988. Isolation
1 Biology 423L Nov. 6/7 Genetics in Forensic Science: Human DNA Fingerprinting Report due Nov. 21 Readings: Hartwell et al. pp. 297-302, 374-387. Nakamura Y., Carlson, K. Krapco, and R. White 1988. Isolation
PERANAN MIKROBIOLOGI DALAM DIAGNOSIS PENYAKIT INFEKSI. dr. Agus Eka Darwinata, Ph.D.
 PERANAN MIKROBIOLOGI DALAM DIAGNOSIS PENYAKIT INFEKSI dr. Agus Eka Darwinata, Ph.D. CLINICAL MICROBIOLOGY Clinical microbiology is the discipline of detection, characterization, and quantification of
PERANAN MIKROBIOLOGI DALAM DIAGNOSIS PENYAKIT INFEKSI dr. Agus Eka Darwinata, Ph.D. CLINICAL MICROBIOLOGY Clinical microbiology is the discipline of detection, characterization, and quantification of
TaKaRa PCR Amplification Kit
 Cat. # R011 For Research Use TaKaRa PCR Amplification Kit Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 4 IV. Materials Required but not Provided... 4 V. Principle...
Cat. # R011 For Research Use TaKaRa PCR Amplification Kit Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 4 IV. Materials Required but not Provided... 4 V. Principle...
Event-specific Method for the Quantification of Oilseed Rape RF1 using Real-time PCR. Protocol
 Event-specific Method for the Quantification of Oilseed Rape RF1 using Real-time PCR Protocol 7 July 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Event-specific Method for the Quantification of Oilseed Rape RF1 using Real-time PCR Protocol 7 July 2011 Joint Research Centre Institute for Health and Consumer Protection Molecular Biology and Genomics
Determination of Antibiotic resistance and Molecular characterization of Salmonella sp isolated from Poultry samples
 ISSN: 2319-7706 Volume 3 Number 6 (2014) pp. 347-353 http://www.ijcmas.com Original Research Article Determination of Antibiotic resistance and Molecular characterization of Salmonella sp isolated from
ISSN: 2319-7706 Volume 3 Number 6 (2014) pp. 347-353 http://www.ijcmas.com Original Research Article Determination of Antibiotic resistance and Molecular characterization of Salmonella sp isolated from
Protocol Version 2. M. Mazzara, N. Foti, M. Maretti, C. Savini and G. Van den Eede. Method development: Syngenta Crop Protection AG
 EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Report on the In-House Validation of an
EUROPEAN COMMISSION DIRECTORATE GENERAL JRC JOINT RESEARCH CENTRE INSTITUTE FOR HEALTH AND CONSUMER PROTECTION COMMUNITY REFERENCE LABORATORY FOR GM FOOD AND FEED Report on the In-House Validation of an
DEC PCR KIT. SSI Diagnostica
 DEC PCR KIT SSI Diagnostica 2 DEC PCR Kit for PCR Detection of Diarrhoeagenic E. coli (DEC) for in vitro diagnostic use Application The DEC PCR Kit is for in vitro diagnostic PCR detection of diarrhoeagenic
DEC PCR KIT SSI Diagnostica 2 DEC PCR Kit for PCR Detection of Diarrhoeagenic E. coli (DEC) for in vitro diagnostic use Application The DEC PCR Kit is for in vitro diagnostic PCR detection of diarrhoeagenic
A new Strategy for Gene Deletion in Campylobacter jejuni
 Roumanian Biotechnological Letters Vol. 14, No. 3, 2009, pp. 4381-4389 Copyright 2008 Bucharest University Printed in Romania. All rights reserved Roumanian Society of Biological Sciences ORIGINAL PAPER
Roumanian Biotechnological Letters Vol. 14, No. 3, 2009, pp. 4381-4389 Copyright 2008 Bucharest University Printed in Romania. All rights reserved Roumanian Society of Biological Sciences ORIGINAL PAPER
E.Z.N.A. Plant Direct PCR Kit
 E.Z.N.A. Plant Direct PCR Kit TQ2800-00 TQ2800-01 20 preps 100 preps June 2013 E.Z.N.A. Plant Direct PCR Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Plant
E.Z.N.A. Plant Direct PCR Kit TQ2800-00 TQ2800-01 20 preps 100 preps June 2013 E.Z.N.A. Plant Direct PCR Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Plant
Hepatitis C Virus Genemer Mix
 Product Manual Hepatitis C Virus Genemer Mix Primer Pair for amplification of HCV Specific DNA Fragment Includes Internal Negative Control Primers and Template Catalog No.: 60-2003-12 Store at 20 o C For
Product Manual Hepatitis C Virus Genemer Mix Primer Pair for amplification of HCV Specific DNA Fragment Includes Internal Negative Control Primers and Template Catalog No.: 60-2003-12 Store at 20 o C For
การสอบสวนโรคต ดเช อในโรงพยาบาล
 การสอบสวนโรคต ดเช อในโรงพยาบาล MRSA Model in Rajavithi Hospital by Chanwit Tribuddharat, M.D., Ph.D. Department of Microbiology Faculty of Medicine Siriraj Hospital Mahidol University E-mail sictb@mahidol.ac.th
การสอบสวนโรคต ดเช อในโรงพยาบาล MRSA Model in Rajavithi Hospital by Chanwit Tribuddharat, M.D., Ph.D. Department of Microbiology Faculty of Medicine Siriraj Hospital Mahidol University E-mail sictb@mahidol.ac.th
Hy-Fy High Fidelity Mix (x2)
 Hy-Fy High Fidelity Mix (x2) #EZ-2021 1ml, 100rxn of 20μl Contents: 2X High Fidelity Mix 1ml Nuclease-free water 1ml Store at -20 C Shelf life: 2 years Description Hy-Fy High Fidelity Mix (x2) is a premixed,
Hy-Fy High Fidelity Mix (x2) #EZ-2021 1ml, 100rxn of 20μl Contents: 2X High Fidelity Mix 1ml Nuclease-free water 1ml Store at -20 C Shelf life: 2 years Description Hy-Fy High Fidelity Mix (x2) is a premixed,
Project 07/111 Final Report October 31, Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines
 Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
PCR analysis was performed to show the presence and the integrity of the var1csa and var-
 Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
PCR Detection of Genetically Modified (GM) Foods Protocol
 PCR Detection of Genetically Modified (GM) Foods Protocol Purpose Isolate DNA from corn-based food so that the Polymerase Chain Reaction can be used to determine whether the selected foods have been genetically
PCR Detection of Genetically Modified (GM) Foods Protocol Purpose Isolate DNA from corn-based food so that the Polymerase Chain Reaction can be used to determine whether the selected foods have been genetically
MightyAmp DNA Polymerase Ver.3
 Cat. # R076A For Research Use MightyAmp DNA Polymerase Ver.3 Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. General PCR Reaction Mix... 3 V. Primer Design...
Cat. # R076A For Research Use MightyAmp DNA Polymerase Ver.3 Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. General PCR Reaction Mix... 3 V. Primer Design...
Protocol for amplification of measles sequencing window (N-450)
 Annex 7.1 Protocol for amplification of measles sequencing window (N-450) NOTE: This document is intended to provide basic test method details and is not an SOP. Laboratories need to develop their own
Annex 7.1 Protocol for amplification of measles sequencing window (N-450) NOTE: This document is intended to provide basic test method details and is not an SOP. Laboratories need to develop their own
pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20
 pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20 1 Kit Contents Contents pgm-t Cloning Kit pgm-t Vector (50 ng/μl) 20 μl T4 DNA Ligase (3 U/μl) 20 μl 10X T4 DNA Ligation Buffer 30 μl
pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20 1 Kit Contents Contents pgm-t Cloning Kit pgm-t Vector (50 ng/μl) 20 μl T4 DNA Ligase (3 U/μl) 20 μl 10X T4 DNA Ligation Buffer 30 μl
BIO/CHEM 475 Molecular Biology Laboratory Spring 2007 Biol/Chem 475 Part 2 of Cloning Lab
 BIO/CHEM 475 Molecular Biology Laboratory Spring 2007 Biol/Chem 475 Part 2 of Cloning Lab Week 5 Analysis of pgem recombinant clones using PCR Clonecheck to determine size of insert; select clones for
BIO/CHEM 475 Molecular Biology Laboratory Spring 2007 Biol/Chem 475 Part 2 of Cloning Lab Week 5 Analysis of pgem recombinant clones using PCR Clonecheck to determine size of insert; select clones for
The Techniques of Molecular Biology: Forensic DNA Fingerprinting
 Revised Fall 2016 The Techniques of Molecular Biology: Forensic DNA Fingerprinting **Lab coat, eye goggles and gloves (nitrile or latex) are required for this lab. You will not be allowed to participate
Revised Fall 2016 The Techniques of Molecular Biology: Forensic DNA Fingerprinting **Lab coat, eye goggles and gloves (nitrile or latex) are required for this lab. You will not be allowed to participate
Cloning, In silico analysis, Expression and testing of Immune-potential of outer membrane protein ( Omp 87) of Pasteurella multocida serotype B: 2
 Available online at www.scholarsresearchlibrary.com Annals of Biological Research, 2014, 5 (7):54-61 (http://scholarsresearchlibrary.com/archive.html) ISSN 0976-1233 CODEN (USA): ABRNBW Cloning, In silico
Available online at www.scholarsresearchlibrary.com Annals of Biological Research, 2014, 5 (7):54-61 (http://scholarsresearchlibrary.com/archive.html) ISSN 0976-1233 CODEN (USA): ABRNBW Cloning, In silico
2ml of 1M stock 10x TBE (1 Litre) Tris Base 107.8g 55g (harmful, wear mask) EDTA 7.4g
 Phytoplasma Detection Protocol Buffers: Hybridisation buffer 100ml hybridisation buffer 2.92g Sodium chloride 4g Blocking reagent (add slowly while stirring) Mix at room temperature for 2 hours Can be
Phytoplasma Detection Protocol Buffers: Hybridisation buffer 100ml hybridisation buffer 2.92g Sodium chloride 4g Blocking reagent (add slowly while stirring) Mix at room temperature for 2 hours Can be
The Techniques of Molecular Biology: Forensic DNA Fingerprinting
 The Techniques of Molecular Biology: Forensic DNA Fingerprinting Revised Fall 2017 Laboratory Safety: Lab coat, long pants, closed-toe shoes, safety goggles, and nitrile or latex gloves are required. Learning
The Techniques of Molecular Biology: Forensic DNA Fingerprinting Revised Fall 2017 Laboratory Safety: Lab coat, long pants, closed-toe shoes, safety goggles, and nitrile or latex gloves are required. Learning
