a Ag but activity of silver ion in the membrane phase (σ)=1 Therefore, overall ISE cells containing such a membrane has and
|
|
|
- Roger Newman
- 6 years ago
- Views:
Transcription
1 Ag 2 S -based membranes behave as ion-exchangers in which Ag + conduct charge through membranes Membrane has no diffusion potential within---only two phase boundary potentials at each side of the pressed pellet. Therefore E mem = E mem = z i log a sam + a Ag Ag + int. a Ag + a Ag + but activity of silver ion in the membrane phase (σ)=1 Therefore, overall ISE cells containing such a membrane has and E cell = K log (a Ag +) sam However, such an electrode will also respond to sulfide ion activity--since: : Ag 2 S (s) -----> 2 Ag + + S = Ksp = (a Ag ) 2 a S
2 a Ag + = Ksp a S 2 = Ksp a S 2 1 / 2 E cell = K log Ksp a S 2 E cell = K ' log(a S 2 ) 1 / 2 To measure sulfide---must operate electrode in solution with high ph---ph or so; so S -2 exists at reasonable concentrations-- (S -2 + H + ---> HS - + H > H 2 S ) Detection limits determined by Ksp of Ag 2 S (s) 1.6 x therefore, at equilibrium, there is approx M Ag + in solution in equilibrium with membrane
3 Selectivity: Depends on Ksp of interfering ions with Ag + and S Ag 2 S (s) <-----> Ag + + S -2 S -2 + Hg +2 <------> HgS (s) increasing activity of Hg +2, increases activity of Ag + at surface of electrode due to lower Ksp of HgS! pot K Ag +, M + = Ksp Ag 2 S Ksp MS If Ksp MS << Ksp Ag2S ; then M will be significant interferent species for measurement of Ag + MIXED CRYSTAL Type ISEs e.g., membrane made of pressed pellet of Ag 2 S/CuS ---> electrode that can measure Cu +2 Ag 2 S (s) < > 2 Ag + + S -2 S -2 + Cu +2 <------> CuS (s)
4 -sulfide ion activity at interface of membrane controlled by solubility product of CuS-- -in turn, Ag + activity controlled by this S -2 activity. therefore: increasing Cu +2 in sample, decreases S -2 activity at electrode surface, thereby increasing Ag + activity---which is what membrane detects! For a less soluble M 2 S z, then M z+ will be interferent! pot K i, j = Ksp IS Ksp MS for low level calibrations of these M 2 S z /Ag 2 S based membrane electrodes for metal ions, must use metal ion buffers----since at conc. < µm, loss of metal ions due to adsorption on walls of vessel, becomes problem. Can also make mixed crystal type membrane elect. for measurement of given anions: Ag 2 S/AgX X = SCN -, Cl -, Br -, I -, CN - same principal----silver ion activity will be controlled by greater solubility of AgX; thus, silver ion levels will change when X is added to sample phase!
5 All of these Ag 2 S -based ISEs suffer severe limits when used in biological samples---owing to the very strong interaction of Ag + with thiol groups of cysteine residues in proteins (e.g., indeed, such sensors respond to protein levels when they are present in samples!) LaF 3 -single crystal membranes for Fluoride electrode NaF, NaCl soln LaF 3 crystal doped with Europium to improve conductivity Fluoride anion is mobile in crystal lattice--due to defects---ala the Ag 2 S crystalline membranes! Fluoride ion mobility was proven by Durst and Rosscoulometricallly generated F - through mem. with 99% efficiency.
6 equilibria at interface of crystal: LaF 3(m) < > La +3 (sam) + 3 F - (sam) Ksp = (a La )(a F ) 3 LaF 3(m) < > LaF 2 + (m) + F - equilibrium that generates phase boundary potential acts as fixed site anion exchanger with selectivity for fluoride anion Selectivity----very high for Fluoride--- K pot F,Cl = 10-4 K pot F,Br = 10-3 Again---based on Ksp of various LaA salts! OH - is major interference---la(oh) 3 --very insoluble---at high ph--looks like lots of F -
7 K pot F,OH = 10-1 in constant 10-3 M F - H + + F > HF E cell La OH - ---> La(OH) 3(s) 4 7 ph Potentiometric Gas Sensors: -use membrane electrodes behind gas permeable membrane -measure equilibrium activities of ions formed (by E cell ) within thin film of electrolyte between ISE mem. and gas permeable membrane as a function key point: called sensor because in conventional designs, the entire electrochemical cell is in one soln. behind a gas permeable membrane (only mem in contact with sample)---see Fig. on next page: 10
8 E cell reference electrodes-- (Ag/AgCl) internal electrolyte of ISE internal electrolyte of gas sensor Ion-selective membrane--(glass polymeric, etc.) gas permeable membrane (teflon,silicone rubber) thin film of electrolyte in which change in activity of ion occurs as gas reaches equilibrium across gas permeable membrane If internal ISE is ph electrode---then sensors for following gases can be made--- NH 3(g) + H 2 O <------> NH 4 + OH - SO 2(g) + H 2 O < > HSO H + CO 2(g) + H 2 O < > HCO H + 2NO 2(g) + H 2 O <------> NO NO H +
9 Theory of gas sensors---must make following assumptions: -composition of internal electrolyte is not affected by sample (concentration of ions in internal does not change due to osmotic effects, etc.) -Thin electrolyte film at the sensor s tip is an essentially isolated region from bulk internal electrolyte---only in electrolytic contact on edges. You don t want ph to change within bulk of internal electrolyte of gas sensor. - Henry s constant for gas is same for sample and internal solution and equilibrium, and hence constant E cell is achieved when the partial pressure of analyte gas is equal on both sides of gas perm. membrane. (P g = H[gas]) First potentiometric gas sensor---(for solution measurements)---severinghaus and Bradley-1957 Used only NaCl as internal electrolyte for CO 2 sensor- for blood P CO2 measurements
10 CO 2 + H 2 O <------> HCO H + K a = (a H+ ) (a HCO3 -) / P CO2 and a H + = K a P CO2 / a - HCO3 but E cell = K log (a H + (film) ) When sensor at equilibrium---p CO2(sam) = P CO2(film) therefore: E cell = K log (K a P CO2 / a HCO3 -) When no bicarbonate salt is incorporated into the internal solution---then a HCO3 - changes as the P CO2 increases---so numerator and denominator change- ====yields measured slope of < 59 mv/decade change in P CO2 However---if we add NaHCO 3 at 0.1 M in internal solution (along with NaCl---so we have Cl - for Ag/AgCl reference) ---Then both K a and a HCO3 - are constant---then E cell = K log P CO2(film)
11 To make ammonia gas sensor----use NH 4 Cl as internal electrolyte---along with same ph sensor NH 3 + H 2 O <------> NH OH - pk b = 4.7 NH 4 + < > H + + NH 3 pka = 9.3 therefore --K a = P NH3 a H +/ a NH4 + solve for a H + and substitute into E cell equation for response of inner ph electrode: E cell = K log (K a a NH4 +/ P NH3 ) therefore if NH 4 Cl is used as internal solution and NH 4 + concentration is high enough---then E cell = K log P NH3 since P NH3 = H[NH 3 ] then : E cell = K log [NH 3 ] sam
12 Detection limits of such gas sensors are generally determined by the equilibrium concentration of gas that originates from the internal solution electrolyte-- The higher the concentration of electrolyte--(bicarbonate for CO2 sensor, ammonium for NH3 sensor) the higher the detection limits----however, to achieve linear response of E cell with log [gas] to high levels of gas---then you need high electrolyte concentration. Also depends exchange between bulk internal electrolyte solution and the solution within the film-- Hence, detection limits will also depend on geometry- (if no exchange---then loss of gas from thin film can occur when soaking in solutions without the analyte gas---and this will improve the detection limits). Selectivity: only species which can permeate the outer gas permeable membrane and change the ph within the internal electrolyte film, are interferences.
Ion Concentration Controller General Information
 Document 40501, p. 1 of 8 Ion Concentration Controller General Information Common Questions Asked about Ion-Selective Electrodes 1. How often do you need to calibrate your ion-selective electrode (ISE)?
Document 40501, p. 1 of 8 Ion Concentration Controller General Information Common Questions Asked about Ion-Selective Electrodes 1. How often do you need to calibrate your ion-selective electrode (ISE)?
Lecture 11. Solubility of Ionic Compounds in Water
 Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
Chapter 9 Systematic Equilibrium
 Chapter 9 Systematic Equilibrium 1 What we Skipped Ch. 8 discussed Activity (γ) This is what should be used in equilibrium calculations rather than conc. (molarity) In Ch. 9 in later chapters, you will
Chapter 9 Systematic Equilibrium 1 What we Skipped Ch. 8 discussed Activity (γ) This is what should be used in equilibrium calculations rather than conc. (molarity) In Ch. 9 in later chapters, you will
Introduction The Van London Co. Cadmium Ion Electrode is used to measure Cadmium ions in aqueous solutions.
 GENERAL INSTRUCTIONS Introduction The Van London Co. Cadmium Ion Electrode is used to measure Cadmium ions in aqueous solutions. Required Equipment 1. An ion meter 2. Cadmium Ion Sensing Electrode 3. Polishing
GENERAL INSTRUCTIONS Introduction The Van London Co. Cadmium Ion Electrode is used to measure Cadmium ions in aqueous solutions. Required Equipment 1. An ion meter 2. Cadmium Ion Sensing Electrode 3. Polishing
Chapter 9. Dissolution and Precipitation Equilibria
 Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4 The Effects of ph on Solubility
Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4 The Effects of ph on Solubility
HI 4105 CARBON DIOXIDE. Instruction Manual HI Carbon Dioxide Ion Selective Electrode
 HI 4105 CARBON DIOXIDE Instruction Manual HI 4105 Carbon Dioxide Ion Selective Electrode 2 HI 4105 Carbon Dioxide Electrode I. Introduction: The Hanna HI 4105 Carbon Dioxide gas sensing electrode is a
HI 4105 CARBON DIOXIDE Instruction Manual HI 4105 Carbon Dioxide Ion Selective Electrode 2 HI 4105 Carbon Dioxide Electrode I. Introduction: The Hanna HI 4105 Carbon Dioxide gas sensing electrode is a
Precipitation Reactions and Gravimetric Analysis
 Precipitation Reactions and Gravimetric Analysis Titrations where the titrant forms a precipitate with the analyte. Not always so straightforward a number of requirements need to be met. Precipitation
Precipitation Reactions and Gravimetric Analysis Titrations where the titrant forms a precipitate with the analyte. Not always so straightforward a number of requirements need to be met. Precipitation
2/24/2003 OFB Chapter 9 1
 2/24/2003 OFB Chapter 9 1 Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4
2/24/2003 OFB Chapter 9 1 Chapter 9 Dissolution and Precipitation Equilibria 9-1 The Nature of Solubility Equilibria 9-2 The Solubility of Ionic Solids 9-3 Precipitation and the Solubility Product 9-4
Chapter six. Precipitate is separated by filtration, washing,drying and finally is accurately weighed.
 Gravimetric Analysis Chapter six Gravimetric Analysis :- Is one of the most accurate and precise methods of quantative analysis.it is based on the measurements of the mass of solid product of a chemical
Gravimetric Analysis Chapter six Gravimetric Analysis :- Is one of the most accurate and precise methods of quantative analysis.it is based on the measurements of the mass of solid product of a chemical
Critical Analytical Measurements for Bioreactor Optimization. controlling an organism s chemical environment leads to consistent and
 Critical Analytical Measurements for Bioreactor Optimization Mettler-Toledo Ingold, Inc., Bedford, MA Abstract Most bioreactor processes share a basic principle; optimizing and controlling an organism
Critical Analytical Measurements for Bioreactor Optimization Mettler-Toledo Ingold, Inc., Bedford, MA Abstract Most bioreactor processes share a basic principle; optimizing and controlling an organism
Chem 1120 Pretest 3 Fall 2015
 Chem 1120 Pretest 3 Fall 2015 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following solubility product expressions is incorrect? a. Cu
Chem 1120 Pretest 3 Fall 2015 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following solubility product expressions is incorrect? a. Cu
WATER QUALITY ANALYZERS SENSOR TECHNOLOGY. mv(orp) ION. Conductivity. Resistivity. Salinity TDS
 WATER QUALITY ANALYZERS mv(orp) SENSOR TECHNOLOGY ION Conductivity DO Resistivity Salinity TDS ELECTRODE LINE UP 01 02 New dome-shaped construction boosts strength in all directions! NEW STANDARD NEW SLEEVE
WATER QUALITY ANALYZERS mv(orp) SENSOR TECHNOLOGY ION Conductivity DO Resistivity Salinity TDS ELECTRODE LINE UP 01 02 New dome-shaped construction boosts strength in all directions! NEW STANDARD NEW SLEEVE
Corrosion. Lab. of Energy Conversion & Storage Materials. Produced by K. B. Kim
 Corrosion 대기환경에의한금속소재 (organic film coated steel) 의퇴화현상평가연구 Lab. of Energy Conversion & Storage Materials Produced by K. B. Kim Introduction AC Impedance Spectroscopy Application of AC Impedance to Corrosion
Corrosion 대기환경에의한금속소재 (organic film coated steel) 의퇴화현상평가연구 Lab. of Energy Conversion & Storage Materials Produced by K. B. Kim Introduction AC Impedance Spectroscopy Application of AC Impedance to Corrosion
Chapter 17 Solubility and Complex Ion Equilibria
 Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
What is ph? ph is the measurement of the Hydrogen Ion concentration in a particular solution. Indicates the Acidity or Alkalinity of a solution.
 What is ph? ph is the measurement of the Hydrogen Ion concentration in a particular solution. Indicates the Acidity or Alkalinity of a solution. Why is ph measured? Used as a process control parameter.
What is ph? ph is the measurement of the Hydrogen Ion concentration in a particular solution. Indicates the Acidity or Alkalinity of a solution. Why is ph measured? Used as a process control parameter.
Electrochemical Sensors
 Electrochemical Sensors Arthur Gildea ABC s of Electrochemistry 1/26/2012 Overview Introduction Reference electrode Biosensors and Immunosensors Potentiometric sensor Amperometric sesnor Conductometric
Electrochemical Sensors Arthur Gildea ABC s of Electrochemistry 1/26/2012 Overview Introduction Reference electrode Biosensors and Immunosensors Potentiometric sensor Amperometric sesnor Conductometric
Proxima. pco 2 and Calculated values for HCO 3 - and Base Excess on Proxima
 Operating principles The Proxima Sensor is fabricated as a single multi-analyte chip that carries an array of individual sensors, each of which measures a different analyte. pco 2 is measured by direct
Operating principles The Proxima Sensor is fabricated as a single multi-analyte chip that carries an array of individual sensors, each of which measures a different analyte. pco 2 is measured by direct
Chapter 7 Mass Transfer
 Chapter 7 Mass Transfer Mass transfer occurs in mixtures containing local concentration variation. For example, when dye is dropped into a cup of water, mass-transfer processes are responsible for the
Chapter 7 Mass Transfer Mass transfer occurs in mixtures containing local concentration variation. For example, when dye is dropped into a cup of water, mass-transfer processes are responsible for the
Corrosion and inhibition of Cu-Zn alloys in NaCl solution by using permanganate and phosphate anions
 Int. J. Electrochem. Sci., 2 (2007) 563-571 www.electrochemsci.org Corrosion and inhibition of Cu-Zn alloys in NaCl solution by using permanganate and phosphate anions S. A. M. Refaey 1, *, A. M. Abd El
Int. J. Electrochem. Sci., 2 (2007) 563-571 www.electrochemsci.org Corrosion and inhibition of Cu-Zn alloys in NaCl solution by using permanganate and phosphate anions S. A. M. Refaey 1, *, A. M. Abd El
Electrochemical cells use spontaneous redox reactions to convert chemical energy to electrical energy.
 ELECTROLYSIS: -the process of supplying electrical energy to a molten ionic compound or a solution containing ions so as to produce a chemical change (causing a non-spontaneous chemical reaction to occur).
ELECTROLYSIS: -the process of supplying electrical energy to a molten ionic compound or a solution containing ions so as to produce a chemical change (causing a non-spontaneous chemical reaction to occur).
Oxidation Reactions. This oxide will from only if thermodynamics favour a reaction of the form: M + O 2 = MO 2. Which must form rapidly (favourable(
 Oxidation of s Oxidation is a general term used to define the reaction between a metal or alloy and its environment. s or alloys are oxidised when heated to elevated temperatures es in air or highly oxidised
Oxidation of s Oxidation is a general term used to define the reaction between a metal or alloy and its environment. s or alloys are oxidised when heated to elevated temperatures es in air or highly oxidised
Surface films formed during H 2 S corrosion of pipeline steels and the effect on hydrogen permeation
 Surface films formed during H 2 S corrosion of pipeline steels and the effect on hydrogen permeation Frank Cheng Department of Mechanical Engineering University of Calgary June 1, 2017 Pipelines in sour
Surface films formed during H 2 S corrosion of pipeline steels and the effect on hydrogen permeation Frank Cheng Department of Mechanical Engineering University of Calgary June 1, 2017 Pipelines in sour
Equilibria of Slightly Soluble Ionic Compounds: We lied to you in chem 115
 Precipitation Reactions Predicting Whether a Precipitate Will Form Note the ions present in the reactants Consider all possible cation-anion combinations Use the solubility rules to decide whether any
Precipitation Reactions Predicting Whether a Precipitate Will Form Note the ions present in the reactants Consider all possible cation-anion combinations Use the solubility rules to decide whether any
Thermodynamics and Electrode Potential ME Dr. Zuhair M. Gasem
 Thermodynamics and Electrode Potential ME 472-062 Copyright Dr. Zuhair M. Gasem Corrosion Science and Engineering 2 Corrosion Science Engineering: corrosion forms, and controlling methods Chpater2 Thermodynamics
Thermodynamics and Electrode Potential ME 472-062 Copyright Dr. Zuhair M. Gasem Corrosion Science and Engineering 2 Corrosion Science Engineering: corrosion forms, and controlling methods Chpater2 Thermodynamics
Sensors for Activated Sludge Aeration Control
 October 23, 2012 Use of Ammonia and Nitrate Sensors for Activated Sludge Aeration Control Troy Morgan; Western Regional Process Analytical Manager Overview Sensors Available Optical - Nitrate / Nitrite
October 23, 2012 Use of Ammonia and Nitrate Sensors for Activated Sludge Aeration Control Troy Morgan; Western Regional Process Analytical Manager Overview Sensors Available Optical - Nitrate / Nitrite
Bucking the Industry Trend. Simple Change to Water System Saves New Jersey Manufacturer $23,000 Annually
 Simple Change to Water System Saves New Jersey Manufacturer by Kevin Preising Sales Engineer . Summary: Over the past two decades, the trend in the water treatment industry has been towards more and more
Simple Change to Water System Saves New Jersey Manufacturer by Kevin Preising Sales Engineer . Summary: Over the past two decades, the trend in the water treatment industry has been towards more and more
Electricity and Chemistry
 Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
ELECTRODES METERS LAB SUPPLIES
 ELECTRODES METERS LAB SUPPLIES PRODUCT CATALOGUE YOUR SUPPLIER GOOD CHEMISTRY THE BENCHMARK IN LONG LIFE PRECISION SINCE 1972 RECORDS / NOTES TECHNOLOGY AND INNOVATION Underpinning Ionode s reliability
ELECTRODES METERS LAB SUPPLIES PRODUCT CATALOGUE YOUR SUPPLIER GOOD CHEMISTRY THE BENCHMARK IN LONG LIFE PRECISION SINCE 1972 RECORDS / NOTES TECHNOLOGY AND INNOVATION Underpinning Ionode s reliability
Enzyme based biosensors
 Enzyme based biosensors Brief history; how it all started? 1916 First report on immobilization of proteins : adsorption of invertase on activated charcoal 1922 First glass ph electrode 1956 Clark published
Enzyme based biosensors Brief history; how it all started? 1916 First report on immobilization of proteins : adsorption of invertase on activated charcoal 1922 First glass ph electrode 1956 Clark published
Design and implementation of a microchemistry analyzer
 Pure & App/. Chem., Vol. 68, No. 10, pp. 1837-1841, 1996. Printed in Great Britain. Q 1996 IUPAC Design and implementation of a microchemistry analyzer Nina Peled, PhD i-stat Corporation 303 College Road
Pure & App/. Chem., Vol. 68, No. 10, pp. 1837-1841, 1996. Printed in Great Britain. Q 1996 IUPAC Design and implementation of a microchemistry analyzer Nina Peled, PhD i-stat Corporation 303 College Road
SELECTION GUIDE. APPLICATION GUIDE. TECHNICAL DATA. SPECIFICATIONS. OEM CORE COMPETENCIES.
 ELECTRODES & SENSORS SELECTION GUIDE. APPLICATION GUIDE. TECHNICAL DATA. SPECIFICATIONS. OEM CORE COMPETENCIES. UNEQUALLED SINCE 1972 SELECTION GUIDE Electrodes and sensors All-purpose sensors with super
ELECTRODES & SENSORS SELECTION GUIDE. APPLICATION GUIDE. TECHNICAL DATA. SPECIFICATIONS. OEM CORE COMPETENCIES. UNEQUALLED SINCE 1972 SELECTION GUIDE Electrodes and sensors All-purpose sensors with super
Table of Contents. Preface...
 Preface... xi Chapter 1. Metallurgical Thermochemistry... 1 1.1. Introduction... 1 1.2. Quantities characterizing the state of a system and its evolution... 3 1.2.1. The types of operations... 3 1.2.2.
Preface... xi Chapter 1. Metallurgical Thermochemistry... 1 1.1. Introduction... 1 1.2. Quantities characterizing the state of a system and its evolution... 3 1.2.1. The types of operations... 3 1.2.2.
Getting To Know Your ph Electrode
 1. Single vs Double Junction Electrodes feature either a single or double junction design. Single junction electrodes serve to put the reference electrode system in contact with the sample. These electrodes
1. Single vs Double Junction Electrodes feature either a single or double junction design. Single junction electrodes serve to put the reference electrode system in contact with the sample. These electrodes
Electrochemistry. Electrode types. electrodes. ph electrodes
 electrodes Electrode types ph electrodes A B C D E F G = Membrane types Each membrane of a ph electrode has its own composition, bulb configuration and thickness. Therefore, carefully study the specifications
electrodes Electrode types ph electrodes A B C D E F G = Membrane types Each membrane of a ph electrode has its own composition, bulb configuration and thickness. Therefore, carefully study the specifications
SELECTION GUIDE. APPLICATION GUIDE. TECHNICAL DATA. SPECIFICATIONS. OEM CORE COMPETENCIES.
 GOOD CHEMISTRY ELECTRODES & SENSORS SELECTION GUIDE. APPLICATION GUIDE. TECHNICAL DATA. SPECIFICATIONS. OEM CORE COMPETENCIES. UNEQUALLED SINCE 1972 SELECTION GUIDE Electrodes and sensors All-purpose sensors
GOOD CHEMISTRY ELECTRODES & SENSORS SELECTION GUIDE. APPLICATION GUIDE. TECHNICAL DATA. SPECIFICATIONS. OEM CORE COMPETENCIES. UNEQUALLED SINCE 1972 SELECTION GUIDE Electrodes and sensors All-purpose sensors
FUEL CELL CHARGE TRANSPORT
 FUEL CELL CHARGE TRANSPORT M. OLIVIER marjorie.olivier@fpms.ac.be 19/05/2008 INTRODUCTION Charge transport completes the circuit in an electrochemical system, moving charges from the electrode where they
FUEL CELL CHARGE TRANSPORT M. OLIVIER marjorie.olivier@fpms.ac.be 19/05/2008 INTRODUCTION Charge transport completes the circuit in an electrochemical system, moving charges from the electrode where they
Aluminum is a very useful metal Where does it come from? All "Bauxite" to begin with A mix of aluminum, iron, and silicon oxides
 Aluminum is a very useful metal Where does it come from? The rest of the Semester All of Chemistry Today Groups 1V-VIII All "Bauxite" to begin with A mix of aluminum, iron, and silicon oxides "Bayer process"
Aluminum is a very useful metal Where does it come from? The rest of the Semester All of Chemistry Today Groups 1V-VIII All "Bauxite" to begin with A mix of aluminum, iron, and silicon oxides "Bayer process"
Imperfections, Defects and Diffusion
 Imperfections, Defects and Diffusion Lattice Defects Week5 Material Sciences and Engineering MatE271 1 Goals for the Unit I. Recognize various imperfections in crystals (Chapter 4) - Point imperfections
Imperfections, Defects and Diffusion Lattice Defects Week5 Material Sciences and Engineering MatE271 1 Goals for the Unit I. Recognize various imperfections in crystals (Chapter 4) - Point imperfections
Munira Raja. Organic Electronics Group Department of Electrical Engineering & Electronics.
 MOS-AK/GSA ESSDERC/ESSCIRC Workshop, Bordeaux, 21 st Sept 2012 Munira Raja mraja@liv.ac.uk Organic Electronics Group Department of Electrical Engineering & Electronics Motivation Organic Electronics is
MOS-AK/GSA ESSDERC/ESSCIRC Workshop, Bordeaux, 21 st Sept 2012 Munira Raja mraja@liv.ac.uk Organic Electronics Group Department of Electrical Engineering & Electronics Motivation Organic Electronics is
VAN LONDON COMPANY COMBINATION ph ELECTRODE INSTRUCTION MANUAL
 VAN LONDON COMPANY COMBINATION ph ELECTRODE INSTRUCTION MANUAL GENERAL INFORMATION Van London combination electrodes offer the convenience of having the reference and measuring electrodes combined in a
VAN LONDON COMPANY COMBINATION ph ELECTRODE INSTRUCTION MANUAL GENERAL INFORMATION Van London combination electrodes offer the convenience of having the reference and measuring electrodes combined in a
Crystal Defects. Perfect crystal - every atom of the same type in the correct equilibrium position (does not exist at T > 0 K)
 Crystal Defects Perfect crystal - every atom of the same type in the correct equilibrium position (does not exist at T > 0 K) Real crystal - all crystals have some imperfections - defects, most atoms are
Crystal Defects Perfect crystal - every atom of the same type in the correct equilibrium position (does not exist at T > 0 K) Real crystal - all crystals have some imperfections - defects, most atoms are
What is: (a) aqua fortis, (b) aqua regia (c) Fixation of Nitrogen?
 EXERCISE.1 Question 1: What is: (a) aqua fortis, (b) aqua regia (c) Fixation of Nitrogen? Solution 1: Book Name: Selina concise (a) Aqua fortis: Nitric acid is called aqua fortis. Aqua fortis means strong
EXERCISE.1 Question 1: What is: (a) aqua fortis, (b) aqua regia (c) Fixation of Nitrogen? Solution 1: Book Name: Selina concise (a) Aqua fortis: Nitric acid is called aqua fortis. Aqua fortis means strong
Urine concentration by forward osmosis process
 The 13 th IWA specialized conference on small water and wastewater systems The 5 th IWA specialized conference on resourcesoriented sanitation September 14 th ~16 th, 216 Urine concentration by forward
The 13 th IWA specialized conference on small water and wastewater systems The 5 th IWA specialized conference on resourcesoriented sanitation September 14 th ~16 th, 216 Urine concentration by forward
AP*Chemistry Solubility Equilibria
 AP*Chemistry Solubility Equilibria SOLUBILITY EQUILIBRIA (K sp, THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Remember those solubility rules? The fine print
AP*Chemistry Solubility Equilibria SOLUBILITY EQUILIBRIA (K sp, THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Remember those solubility rules? The fine print
Microsystem Integration (March 20, 2017)
 Microsystem Integration (March 20, 2017) Mid-term exam: open notes/computers/ - 1:00 to 1:50 p.m., Wednesday, March 22. Homeworks #1 to 7. - Solutions of #1 to 7 are posted. Workshops #1 to 15. - Workshops
Microsystem Integration (March 20, 2017) Mid-term exam: open notes/computers/ - 1:00 to 1:50 p.m., Wednesday, March 22. Homeworks #1 to 7. - Solutions of #1 to 7 are posted. Workshops #1 to 15. - Workshops
Electronic Structure Methods for Complex Materials
 Electronic Structure Methods for Complex Materials The orthogonalized linear combination of atomic orbitals WAI-YIM CHING and PAUL RULIS University of Missouri-Kansas City, USA OXFORD UNIVERSITY PRESS
Electronic Structure Methods for Complex Materials The orthogonalized linear combination of atomic orbitals WAI-YIM CHING and PAUL RULIS University of Missouri-Kansas City, USA OXFORD UNIVERSITY PRESS
Sample Answers/Solutions - Study of Compounds
 Sample Answers/Solutions - Study of Compounds Hydrogen Chloride 1. It is a gas at room temperature. 2. Refer to pg.128 right side section 2. 3. If the reaction is carried out above 200 o C, then the by-product
Sample Answers/Solutions - Study of Compounds Hydrogen Chloride 1. It is a gas at room temperature. 2. Refer to pg.128 right side section 2. 3. If the reaction is carried out above 200 o C, then the by-product
Lab IV: Electrical Properties
 Lab IV: Electrical Properties Study Questions 1. How would the electrical conductivity of the following vary with temperature: (a) ionic solids; (b) semiconductors; (c) metals? Briefly explain your answer.
Lab IV: Electrical Properties Study Questions 1. How would the electrical conductivity of the following vary with temperature: (a) ionic solids; (b) semiconductors; (c) metals? Briefly explain your answer.
CHAPTER 6: INSTRUMENTATION AND CONTROL OF A BIOREACTOR
 CHAPTER 6: INSTRUMENTATION AND CONTROL OF A BIOREACTOR Bioreactor Control Temperature Dissolved oxygen ph Foam Agitator speed Nutrient addition rate Level control Bioreactor Instrumentations Temperature
CHAPTER 6: INSTRUMENTATION AND CONTROL OF A BIOREACTOR Bioreactor Control Temperature Dissolved oxygen ph Foam Agitator speed Nutrient addition rate Level control Bioreactor Instrumentations Temperature
3. [7 points] How many significant figures should there be in the answer to the following problem?
![3. [7 points] How many significant figures should there be in the answer to the following problem? 3. [7 points] How many significant figures should there be in the answer to the following problem?](/thumbs/83/87492512.jpg) 1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
IRON CARBONATE SCALE GROWTH AND THE EFFECT OF INHIBITION IN CO 2 CORROSION OF MILD STEEL
 IRON CARBONATE SCALE GROWTH AND THE EFFECT OF INHIBITION IN CO 2 CORROSION OF MILD STEEL Kunal Chokshi, Wei Sun, Srdjan Nesic Institute for Corrosion and Multiphase Technology, Ohio University 342 West
IRON CARBONATE SCALE GROWTH AND THE EFFECT OF INHIBITION IN CO 2 CORROSION OF MILD STEEL Kunal Chokshi, Wei Sun, Srdjan Nesic Institute for Corrosion and Multiphase Technology, Ohio University 342 West
Module: 9 Lecture: 39
 Module: 9 Lecture: 39 AMMONIUM CHLORIDE INTRODUCTION Ammonium chloride (NH4Cl) is white crystalline salt highly soluble in water. Solutions of ammonium chloride are mildly acidic. Sal ammoniac is a name
Module: 9 Lecture: 39 AMMONIUM CHLORIDE INTRODUCTION Ammonium chloride (NH4Cl) is white crystalline salt highly soluble in water. Solutions of ammonium chloride are mildly acidic. Sal ammoniac is a name
Direct ISE Method Method to 10.0 mg/l F Fluoride ISE
 Fluoride DOC316.53.01237 Direct ISE Method Method 8323 0.1 to 10.0 mg/l F Fluoride ISE Scope and application: For drinking water 1 and wastewater 2. 1 The equipment that is specified in this method meets
Fluoride DOC316.53.01237 Direct ISE Method Method 8323 0.1 to 10.0 mg/l F Fluoride ISE Scope and application: For drinking water 1 and wastewater 2. 1 The equipment that is specified in this method meets
EQUIPMENT AND SERVICE COMPANY, INCORPORATED FX- 300 ISE Ammonia, Nitrate, Nitrite Measurement
 FX-300 Online Ammonia, Nitrate, Nitrite Monitor and Controller The Foxcroft FX-300 with ion selective electrode technology can be used as an online monitor and control method for Ammonium (NH4+), Ammonia
FX-300 Online Ammonia, Nitrate, Nitrite Monitor and Controller The Foxcroft FX-300 with ion selective electrode technology can be used as an online monitor and control method for Ammonium (NH4+), Ammonia
INCLUSION OF POLYVINYLEDENE FLUORIDE POLYMER AS BINDING AGENT FOR GRAPHITE AND LICOO 2 GRANULES
 MATERIALS SCIENCE and TECHNOLOGY Edited by Evvy Kartini et.al. INCLUSION OF POLYVINYLEDENE FLUORIDE POLYMER AS BINDING AGENT FOR GRAPHITE AND LICOO 2 GRANULES T. Nugraha, E. Panjaitan, E. Kartini Center
MATERIALS SCIENCE and TECHNOLOGY Edited by Evvy Kartini et.al. INCLUSION OF POLYVINYLEDENE FLUORIDE POLYMER AS BINDING AGENT FOR GRAPHITE AND LICOO 2 GRANULES T. Nugraha, E. Panjaitan, E. Kartini Center
Applications of hydrogen permeable membranes in ammonia synthesis and decomposition
 Applications of hydrogen permeable membranes in ammonia synthesis and decomposition September 19, 2016 Sean-Thomas B. Lundin, Thomas F. Fuerst, Jason C. Ganley, Colin A. Wolden, J. Douglas Way Chemical
Applications of hydrogen permeable membranes in ammonia synthesis and decomposition September 19, 2016 Sean-Thomas B. Lundin, Thomas F. Fuerst, Jason C. Ganley, Colin A. Wolden, J. Douglas Way Chemical
Print version. Lecture #42 Precipitation and Dissolution: Iron & Predominance Diagrams (Stumm & Morgan, Chapt.7) Benjamin; Chapter
 Updated: 3 November 03 Print version Lecture #4 Precipitation and Dissolution: Iron & Predominance Diagrams (Stumm & Morgan, Chapt.7) Benjamin; Chapter 8.78.5 David Reckhow CEE 680 #4 opics Ferrous Carbonate
Updated: 3 November 03 Print version Lecture #4 Precipitation and Dissolution: Iron & Predominance Diagrams (Stumm & Morgan, Chapt.7) Benjamin; Chapter 8.78.5 David Reckhow CEE 680 #4 opics Ferrous Carbonate
Amperometric Biosensors. Rahul N 04/10/2014
 Amperometric Biosensors Rahul N 04/10/2014 Biosensor A biosensor is an analytical device, used for the detection of an analyte, that combines a biological component with a physicochemical detector. Bio-recognition
Amperometric Biosensors Rahul N 04/10/2014 Biosensor A biosensor is an analytical device, used for the detection of an analyte, that combines a biological component with a physicochemical detector. Bio-recognition
CHAPTER 4: Oxidation. Chapter 4 1. Oxidation of silicon is an important process in VLSI. The typical roles of SiO 2 are:
 Chapter 4 1 CHAPTER 4: Oxidation Oxidation of silicon is an important process in VLSI. The typical roles of SiO 2 are: 1. mask against implant or diffusion of dopant into silicon 2. surface passivation
Chapter 4 1 CHAPTER 4: Oxidation Oxidation of silicon is an important process in VLSI. The typical roles of SiO 2 are: 1. mask against implant or diffusion of dopant into silicon 2. surface passivation
Change in stoichiometry
 Measurement of Gas Sensor Performance Gas sensing materials: 1. Sputtered ZnO film (150 nm (Massachusetts Institute of Technology) 2. Sputtered SnO 2 film (60 nm) (Fraunhofer Institute of Physical Measurement
Measurement of Gas Sensor Performance Gas sensing materials: 1. Sputtered ZnO film (150 nm (Massachusetts Institute of Technology) 2. Sputtered SnO 2 film (60 nm) (Fraunhofer Institute of Physical Measurement
Section 4: Thermal Oxidation. Jaeger Chapter 3. EE143 - Ali Javey
 Section 4: Thermal Oxidation Jaeger Chapter 3 Properties of O Thermal O is amorphous. Weight Density =.0 gm/cm 3 Molecular Density =.3E molecules/cm 3 O Crystalline O [Quartz] =.65 gm/cm 3 (1) Excellent
Section 4: Thermal Oxidation Jaeger Chapter 3 Properties of O Thermal O is amorphous. Weight Density =.0 gm/cm 3 Molecular Density =.3E molecules/cm 3 O Crystalline O [Quartz] =.65 gm/cm 3 (1) Excellent
Chapter 2 Crystal Growth and Wafer Preparation
 Chapter 2 Crystal Growth and Wafer Preparation Professor Paul K. Chu Advantages of Si over Ge Si has a larger bandgap (1.1 ev for Si versus 0.66 ev for Ge) Si devices can operate at a higher temperature
Chapter 2 Crystal Growth and Wafer Preparation Professor Paul K. Chu Advantages of Si over Ge Si has a larger bandgap (1.1 ev for Si versus 0.66 ev for Ge) Si devices can operate at a higher temperature
a) The self-diffusion coefficient of a metal with cubic structure can be expressed as
 EXERCISES KJM5120 Chapter 5; Diffusion 1. Random (self) diffusion a) The self-diffusion coefficient of a metal with cubic structure can be expressed as 1 n D = s 6 t 2 where n/t represents the jump frequency
EXERCISES KJM5120 Chapter 5; Diffusion 1. Random (self) diffusion a) The self-diffusion coefficient of a metal with cubic structure can be expressed as 1 n D = s 6 t 2 where n/t represents the jump frequency
Corrosion Rates. OLI model basics Polarization curves
 Corrosion Rates OLI model basics Polarization curves Bulk Aqueous Phase E 0 = E 0 0 + (RT/zF zf) Π a νi i e.g. Fe Fe Fe +2 + 2e - Equilibrium Activities of species potential OLI thermo and Databank Fe
Corrosion Rates OLI model basics Polarization curves Bulk Aqueous Phase E 0 = E 0 0 + (RT/zF zf) Π a νi i e.g. Fe Fe Fe +2 + 2e - Equilibrium Activities of species potential OLI thermo and Databank Fe
Materials of Engineering ENGR 151 CORROSION ELECTRICAL PROPERTIES
 Materials of Engineering ENGR 151 CORROSION ELECTRICAL PROPERTIES more anodic (active) more cathodic (inert) GALVANIC SERIES Ranking of the reactivity of metals/alloys in seawater Platinum Gold Graphite
Materials of Engineering ENGR 151 CORROSION ELECTRICAL PROPERTIES more anodic (active) more cathodic (inert) GALVANIC SERIES Ranking of the reactivity of metals/alloys in seawater Platinum Gold Graphite
Supporting Information
 Supporting Information Influence of Natural Organic Matter Fouling and Osmotic Backwash on Pressure Retarded Osmosis Energy Production from Natural Salinity Gradients NGAI YIN YIP AND MENACHEM ELIMELECH*
Supporting Information Influence of Natural Organic Matter Fouling and Osmotic Backwash on Pressure Retarded Osmosis Energy Production from Natural Salinity Gradients NGAI YIN YIP AND MENACHEM ELIMELECH*
Chapter 7 Conclusions
 Conclusions Nowadays, much awareness has been devoted in developing sensors for monitoring the pollutants and hazardous gases in various environments. Considerable development work is being carried out
Conclusions Nowadays, much awareness has been devoted in developing sensors for monitoring the pollutants and hazardous gases in various environments. Considerable development work is being carried out
Mat E 272 Lecture 26: Oxidation and Corrosion
 Mat E 272 Lecture 26: Oxidation and Corrosion December 11, 2001 Introduction: Environmental degradation of materials is one of the most costly failure modes, accounting for over 5 percent of the total
Mat E 272 Lecture 26: Oxidation and Corrosion December 11, 2001 Introduction: Environmental degradation of materials is one of the most costly failure modes, accounting for over 5 percent of the total
PORTABLE ph METER PRICE CODE NUMBER MODEL 100
 PORTABLE METER MODEL 100 PRICE CODE NUMBER 56-0032 Refer to Bulletin A-301. OPERATION AND SERVICE GUIDE O-670A DEC. 1997 GENERAL INFORMATION Model 100 meter is designed for measuring over the entire 0
PORTABLE METER MODEL 100 PRICE CODE NUMBER 56-0032 Refer to Bulletin A-301. OPERATION AND SERVICE GUIDE O-670A DEC. 1997 GENERAL INFORMATION Model 100 meter is designed for measuring over the entire 0
Sn 2+ (aq) + 2 Ag + (aq) Sn 4+ (aq) + 2 Ag(s),
 1. Which change in oxidation number represents oxidation? A) Sn 2+ (aq) Sn 4+ (aq) B) Sn 2+ (aq) Sn(s) C) Sn 4+ (aq) Sn 2+ (aq) D) Sn 4+ (aq) Sn(s) E) Sn(s) Sn 2 (aq) 2. In the reaction Sn 2+ (aq) + 2
1. Which change in oxidation number represents oxidation? A) Sn 2+ (aq) Sn 4+ (aq) B) Sn 2+ (aq) Sn(s) C) Sn 4+ (aq) Sn 2+ (aq) D) Sn 4+ (aq) Sn(s) E) Sn(s) Sn 2 (aq) 2. In the reaction Sn 2+ (aq) + 2
Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 13. Ionic Thrills Part 3.
 Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 13. Ionic Thrills Part 3. Name(s): Element: Topics: 1. Octahedral and tetrahedral holes and the hcp lattice 2. Occupying the octahedral and tetrahedral
Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 13. Ionic Thrills Part 3. Name(s): Element: Topics: 1. Octahedral and tetrahedral holes and the hcp lattice 2. Occupying the octahedral and tetrahedral
MUR. Market Specialist
 Cyanide Methods and the 2010 MUR William Lipps Market Specialist Fundamentals of Cyanide Chemistry Free Cyanide is the CN ion and HCN, generate HCN at ph 6 + - Na C N Metal Complexes that require acid
Cyanide Methods and the 2010 MUR William Lipps Market Specialist Fundamentals of Cyanide Chemistry Free Cyanide is the CN ion and HCN, generate HCN at ph 6 + - Na C N Metal Complexes that require acid
Lecture 2. Crystals: Theory and Practice. Dr. Susan Yates Wednesday, February 2, 2011
 Lecture 2 Crystals: Theory and Practice Dr. Susan Yates Wednesday, February 2, 2011 Steps in Solving an X-ray Structure What is a Crystal? Crystal acts as an X-ray diffraction amplifier Crystals Crystals
Lecture 2 Crystals: Theory and Practice Dr. Susan Yates Wednesday, February 2, 2011 Steps in Solving an X-ray Structure What is a Crystal? Crystal acts as an X-ray diffraction amplifier Crystals Crystals
Characteristic and efficiency of PEM fuel cell and PEM electrolyser
 Related topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle and task In a PEM electrolyser, the electrolyte consists of a protonconducting membrane
Related topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle and task In a PEM electrolyser, the electrolyte consists of a protonconducting membrane
SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005
 SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005 VALE CANADA LTD. COPPER CLIFF ANALYTICAL SERVICES 18 Rink Street Copper Cliff, Ontario P0M 1N0 Canada Mr. Claude Serre Phone: 705 682 7501 CHEMICAL Valid To:
SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005 VALE CANADA LTD. COPPER CLIFF ANALYTICAL SERVICES 18 Rink Street Copper Cliff, Ontario P0M 1N0 Canada Mr. Claude Serre Phone: 705 682 7501 CHEMICAL Valid To:
Supplementary Information
 Electrochemical sensing in paper-based microfluidic devices Zhihong Nie a, Christian Nijhuis a, Jinlong Gong a, Xin Chen a, Alexander Kumachev b, Andres W. Martinez a, Max Narovlyansky a, and George M.
Electrochemical sensing in paper-based microfluidic devices Zhihong Nie a, Christian Nijhuis a, Jinlong Gong a, Xin Chen a, Alexander Kumachev b, Andres W. Martinez a, Max Narovlyansky a, and George M.
Solubility Equilibrium
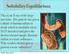 Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Corrosion Simulation. OLI Simulation Conference A. Anderko, G. Engelhardt and M.M. Lencka
 OLI Simulation Conference 2010 Corrosion Simulation Think Simulation! Harnessing the power of the OLI Engine A. Anderko, G. Engelhardt and M.M. Lencka General and localized corrosion models Single-phase
OLI Simulation Conference 2010 Corrosion Simulation Think Simulation! Harnessing the power of the OLI Engine A. Anderko, G. Engelhardt and M.M. Lencka General and localized corrosion models Single-phase
Test sticks and test papers for semi-quantitative determinations
 QUANTOFIX test sticks for semi-quantitative determinations QUANTOFIX test sticks meet the most important requirements for a modern quick-test: rapid dip and read convenient the analysis can be carried
QUANTOFIX test sticks for semi-quantitative determinations QUANTOFIX test sticks meet the most important requirements for a modern quick-test: rapid dip and read convenient the analysis can be carried
Method Excitation signal applied Wave response based on method Linear Differential pulse Square wave Cyclic Developed current recorded
 Voltammetry Electrochemistry techniques based on current (i) measurement as function of voltage (E appl ) Working electrode (microelectrode) place where redox occurs surface area few mm 2 to limit current
Voltammetry Electrochemistry techniques based on current (i) measurement as function of voltage (E appl ) Working electrode (microelectrode) place where redox occurs surface area few mm 2 to limit current
Electrospun quaternized polyvinyl alcohol nanofibers with core-shell structure and their composite in alkaline fuel cell application
 1 Electrospun quaternized polyvinyl alcohol nanofibers with core-shell structure and their composite in alkaline fuel cell application S. Jessie Lue, Ph.D. Department Head and Professor Department of Chemical
1 Electrospun quaternized polyvinyl alcohol nanofibers with core-shell structure and their composite in alkaline fuel cell application S. Jessie Lue, Ph.D. Department Head and Professor Department of Chemical
Diffusion in Solids. Why is it an important part of processing? How can the rate of diffusion be predicted for some simple cases?
 Diffusion in Solids ISSUES TO ADDRESS... How does diffusion occur? Why is it an important part of processing? How can the rate of diffusion be predicted for some simple cases? How does diffusion depend
Diffusion in Solids ISSUES TO ADDRESS... How does diffusion occur? Why is it an important part of processing? How can the rate of diffusion be predicted for some simple cases? How does diffusion depend
Development Production Sales Service
 analysis EQUIPMENT WAter Analysentechnik GmbH & Co. KG Development Production Sales Service Electrodes and Sensors portable and stationary Equipment Analysis Equipment Accessories ProduCt CatalogUE 2014
analysis EQUIPMENT WAter Analysentechnik GmbH & Co. KG Development Production Sales Service Electrodes and Sensors portable and stationary Equipment Analysis Equipment Accessories ProduCt CatalogUE 2014
SENSORS FOR MANGANESE
 SENSORS FOR MANGANESE Contents 4.1 Synthesis of the Ionophore 4.2 Sensors based on TNCR 4.3 Fabrication of the sensors 4.4 Potential measurement and calibration 4.5 Optimization studies of the two types
SENSORS FOR MANGANESE Contents 4.1 Synthesis of the Ionophore 4.2 Sensors based on TNCR 4.3 Fabrication of the sensors 4.4 Potential measurement and calibration 4.5 Optimization studies of the two types
Chapter 5: Atom and Ion Movements in Materials
 Slide 1 Chapter 5: Atom and Ion Movements in Materials 5-1 Slide 2 Learning Objectives 1. Applications of diffusion 2. Stability of atoms and ions 3. Mechanisms for diffusion 4. Activation energy for diffusion
Slide 1 Chapter 5: Atom and Ion Movements in Materials 5-1 Slide 2 Learning Objectives 1. Applications of diffusion 2. Stability of atoms and ions 3. Mechanisms for diffusion 4. Activation energy for diffusion
BIOMIMETIC MEMBRANE: EFFECT OF CONCENTRATION OF PROPANOL ON THE TRANSMEMBRANE POTENTIAL AND IMPEDANCE
 INTRODUCTION: BIOMIMETIC MEMBRANE: EFFECT OF CONCENTRATION OF PROPANOL ON THE TRANSMEMBRANE POTENTIAL AND IMPEDANCE Deng Da Department of Chemical & Environmental Engineering National University of Singapore
INTRODUCTION: BIOMIMETIC MEMBRANE: EFFECT OF CONCENTRATION OF PROPANOL ON THE TRANSMEMBRANE POTENTIAL AND IMPEDANCE Deng Da Department of Chemical & Environmental Engineering National University of Singapore
Dissolved Oxygen Probe (Order Code: DO-BTA)
 Dissolved Oxygen Probe (Order Code: DO-BTA) The Vernier Dissolved Oxygen Probe uses a Clark-type polarographic electrode to measure the concentration of dissolved oxygen in water. You can use this sensor
Dissolved Oxygen Probe (Order Code: DO-BTA) The Vernier Dissolved Oxygen Probe uses a Clark-type polarographic electrode to measure the concentration of dissolved oxygen in water. You can use this sensor
Defects and Diffusion
 Defects and Diffusion Goals for the Unit Recognize various imperfections in crystals Point imperfections Impurities Line, surface and bulk imperfections Define various diffusion mechanisms Identify factors
Defects and Diffusion Goals for the Unit Recognize various imperfections in crystals Point imperfections Impurities Line, surface and bulk imperfections Define various diffusion mechanisms Identify factors
On-Site Sodium Hypochlorite Generation Equipment
 On-Site Sodium Hypochlorite Generation Equipment Water Arabia - 2015 Electrolysis A Process that decomposes a chemical compound into its elements or produces a new compound by the action of an electrical
On-Site Sodium Hypochlorite Generation Equipment Water Arabia - 2015 Electrolysis A Process that decomposes a chemical compound into its elements or produces a new compound by the action of an electrical
Exercises. Solubility Equilibria. a. AgC 2 H 3 O 2 b. Al(OH) 3 c. Ca 3 (PO 4 ) Write balanced equations for the dissolution reactions and the
 766 Active Learning Questions These questions are designed to be used by groups of students in class. 1. Which of the following will affect the total amount of solute that can dissolve in a given amount
766 Active Learning Questions These questions are designed to be used by groups of students in class. 1. Which of the following will affect the total amount of solute that can dissolve in a given amount
Treatment Technologies
 Treatment Technologies Aeration INTRODUCTION METHODS OF AERATION Water-Fall Aerators Air Diffusion Aerators APPLICATIONS Iron and Manganese Removal Dissolved Gas Reduction LIMITATIONS Treatment Technologies
Treatment Technologies Aeration INTRODUCTION METHODS OF AERATION Water-Fall Aerators Air Diffusion Aerators APPLICATIONS Iron and Manganese Removal Dissolved Gas Reduction LIMITATIONS Treatment Technologies
Guideline for Prevention of Damage in Water Heating Installations
 VDI 2035, Part 2 Guideline for Prevention of Damage in Water Heating Installations Original document published by the Society of German Engineers Summary by Elysator Engineering AG 1. Preliminary Remarks
VDI 2035, Part 2 Guideline for Prevention of Damage in Water Heating Installations Original document published by the Society of German Engineers Summary by Elysator Engineering AG 1. Preliminary Remarks
Electricity. Characteristic and efficiency of PEM fuel cell and PEM electrolyser Stationary currents. What you need:
 Stationary currents Electricity Characteristic and efficiency of PEM fuel cell and PEM electrolyser What you can learn about Electrolysis Electrode polarisation Decomposition voltage Galvanic elements
Stationary currents Electricity Characteristic and efficiency of PEM fuel cell and PEM electrolyser What you can learn about Electrolysis Electrode polarisation Decomposition voltage Galvanic elements
Unit F FR (part B) Solubility Equilibrium, Ksp (pg 1 of 16)
 Unit F FR (part B) Solubility Equilibrium, Ksp (pg 1 of 16) 1. Answer the following questions about the solubility of some fluoride salts of alkaline earth metals. (a) A student prepares 100. ml of a saturated
Unit F FR (part B) Solubility Equilibrium, Ksp (pg 1 of 16) 1. Answer the following questions about the solubility of some fluoride salts of alkaline earth metals. (a) A student prepares 100. ml of a saturated
Effect of Chloride on Localized Corrosion Initiation of Carbon Steel in a CO 2 Aqueous Environment Xin Gao, Bruce Brown, Srdjan Nesic
 Paper No. 3880 Effect of Chloride on Localized Corrosion Initiation of Carbon Steel in a CO 2 Aqueous Environment Xin Gao, Bruce Brown, Srdjan Nesic Institute for Corrosion and Multiphase Technology Ohio
Paper No. 3880 Effect of Chloride on Localized Corrosion Initiation of Carbon Steel in a CO 2 Aqueous Environment Xin Gao, Bruce Brown, Srdjan Nesic Institute for Corrosion and Multiphase Technology Ohio
E45 Midterm 01 Fall 2007! By the 0.2% offset method (shown on plot), YS = 500 MPa
 1.!Mechanical Properties (20 points) Refer to the following stress-strain plot derived from a standard uniaxial tensile test of a high performance titanium alloy to answer the following questions. Show
1.!Mechanical Properties (20 points) Refer to the following stress-strain plot derived from a standard uniaxial tensile test of a high performance titanium alloy to answer the following questions. Show
Modeling of Galvanic Interactions Between AA5083 and Steel Under Atmospheric Condition
 Presented at the 2011 COMSOL Conference in Boston Boston, MA, Oct 13 2011 Modeling of Galvanic Interactions Between AA5083 and Steel Under Atmospheric Condition D. Mizuno a),b), Y. Shi a), and R. Kelly
Presented at the 2011 COMSOL Conference in Boston Boston, MA, Oct 13 2011 Modeling of Galvanic Interactions Between AA5083 and Steel Under Atmospheric Condition D. Mizuno a),b), Y. Shi a), and R. Kelly
SOLUBILITY STUDY GUIDE- Multiple Choice Section
 SOLUBILITY STUDY GUIDE- Multiple Choice Section Multiple Choice Section: This study guide is a compilation of questions from provincial exams since 2000. I urge you to become intimately familiar with question
SOLUBILITY STUDY GUIDE- Multiple Choice Section Multiple Choice Section: This study guide is a compilation of questions from provincial exams since 2000. I urge you to become intimately familiar with question
Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law.
 Characteristics and efficiency of PEM fuel cell TEP Related Topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle In a PEM electrolyser, the electrolyte
Characteristics and efficiency of PEM fuel cell TEP Related Topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle In a PEM electrolyser, the electrolyte
Properties of II B Group Elements
 Properties of II B Group Elements d-block Transition Elements VIIIB IIIB IVB VB VIB VIIB IB IIB Sc Ti V Cr Mn Fe Co Ni Cu Zn Y Zr Nb Mo Tc Ru Rh Pd Ag Cd La Hf Ta W Re Os Ir Pt Au Hg II B Group Elements
Properties of II B Group Elements d-block Transition Elements VIIIB IIIB IVB VB VIB VIIB IB IIB Sc Ti V Cr Mn Fe Co Ni Cu Zn Y Zr Nb Mo Tc Ru Rh Pd Ag Cd La Hf Ta W Re Os Ir Pt Au Hg II B Group Elements
