Supporting Information for. Monovalent vs Divalent Cation Diffusion in Thiospinel Ti 2 S 4
|
|
|
- Malcolm Walsh
- 6 years ago
- Views:
Transcription
1 Supporting Information for Monovalent vs Divalent Cation Diffusion in Thiospinel Ti 2 S 4 Patrick Bonnick a, Xiaoqi Sun a, Ka-Cheong Lau b, Chen Liao b and Linda F. Nazar a * a Department of Chemistry and the Waterloo Institute of Nanotechnology, University of Waterloo, Waterloo, Ontario N2L 3G1, Canada b Chemical Science and Engineering, Argonne National Laboratory, 9700 S. Cass Avenue, Lemont, IL 60439, USA * Corresponding author: Prof. Linda F. Nazar, lfnazar@uwaterloo ca S1
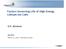
2 Experimental Methods Material synthesis Spinel CuTi 2 S 4 was synthesized as explained elsewhere. 1 3 Briefly, inside an Ar-filled glove box, stoichiometric amounts of powders of the elements were mixed together and pressed into 12 mm diam. pellets totaling 1 g each, which were then cut in half and sealed inside an evacuated quartz tube (8 mm diameter, 12 cm long). The tube was heated at 1 C/min to 700 C, where it was held for 7 days before being cooled naturally and returned to the glove box where the powder product was removed from the tube, reground, re-pelletized and resealed back into a new tube. The tube was reheated to 700 C and held there for another 7 days. This second iteration improves the purity of the product CuTi 2 S 4. Cu + ions were removed by oxidizing CuTi 2 S 4 in dry acetonitrile using twice the stoichiometric amount of Br 2 required and stirring the mixture in a sealed vial at room temperature for 3 days. The resulting Cu 0.1 Ti 2 S 4 particles ranged from 1 10 µm in diameter and were otherwise characterized previously. 1 Electrode fabrication Inside an Ar-filled glove box, about 50 mg of Cu 0.1 Ti 2 S 4 powder was pressed at 6 tonnes in a 12 mm diameter pellet die to form the electrode. Sometimes, a 12 mm diameter, stainless steel 316, 150-size wire mesh was incorporated to help hold the pellet together and improve the conductivity of the pellet. Diffusion experiments performed on pellets without a wire mesh yielded the same results as one with a wire mesh, albeit with messier data and a more fragile pellet. Cell fabrication Conflat cells were designed by Periyapperuma et al. 4 and purchased from DPM Solutions Inc. (Design Prove Machine Solutions, Hebbville, NS, Canada). They were assembled inside an Ar-filled glove box using three layers of (dry) glass fiber filter paper as separators and either Mg or Li foil as the reference and counter electrodes. The electrolyte for the Li x Ti 2 S 4 cells was 1 M LiPF 6 in 1:1 EC:DMC, acquired from BASF. Electrolyte synthesis The electrolyte for Mg x Ti 2 S 4 cells was 0.5 M Mg(CB 11 H 12 ) 2 in tetraglyme, made by Ka- Cheong Lau and Chen Liao of Argonne National Lab (ANL). A previously reported method was modified for the preparation of Mg(CB 11 H 12 ) 2 in tetraglyme electrolyte. 5 A mixture of THF (5 ml), Mg powder (99.5%, 325 mesh; 3.99 g, 164 mmol), and [HNMe 3 ][CB 11 H 12 ] (2.00 g, 9.85 mmol) were stirred at 60 C for 1 h. After 1 h, additional THF (30 ml) was added to the reaction S2
3 mixture. A grey slurry was formed as the reaction proceeded. The reaction was monitored by 1 H NMR of aliquots of the supernatant of the slurry, following the disappearance of Me 3 NH + resonance at δ 3.19 in acetone-d 6. After completion, the slurry was filtered through a mediumporosity frit, and the solid was washed with THF. The solid was dissolved in DME (20 ml) to form a grey slurry, and the slurry was filtered through Celite on a medium-porosity frit to give a clear filtrate. The filtrate was collected and volatiles were removed at 60 C to give colorless to white crystals of [Mg(DME) 3 ](CB 11 H 12 ) 2 (2.51 g, 4.32 mmol, 44 % yield). Anhydrous tetraglyme was added to [Mg(DME) 3 ](CB 11 H 12 ) 2, and the mixture was stirred at room temperature for 12 h to form a clear solution of 0.5 M Mg(CB 11 H 12 ) 2 in tetraglyme. Figure S1 shows a cyclic voltammogram (CV) of the electrolyte, demonstrating the plating and stripping behaviour. current density (ma cm -2 ) CE = 100% Potential (V) Figure S1. Cyclic voltammogram of Mg deposition and stripping from a solution of 0.5 M Mg(CB 11 H 12 ) 2 in tetraglyme using a Pt working electrode, Mg counter electrode and Mg reference electrode at room temperature. The sweep rate was 100 mv/s. Electrochemical experiments Galvanic Intermittent Titration Technique (GITT) and the Galvanic Alternating Pulse (GAP) technique The theory behind GITT experiments has been covered in detail by Weppner, Huggins and Wen, and interested readers are directed to their papers. 6 8 GAP is a GITT experiment where the polarity of the pulse is reversed each pulse so that the electrode is theoretically returned to the same state of charge after every second pulse. How to perform a GITT and GAP experiment, along with how to analyze the data, is covered by Talaie et al. 9 and so will not be repeated here. We will, however, list the experimental parameters used in the GITT experiments that generated Figures 1 and 2 in the main text, as well as in the GAP experiments S3
4 used to generate Figure 4 and the data in Table 1. For all cells, a Bio-logic VMP3 potentiostat was used to control the current and collect potential data from 3-electrode Conflat cells, employing pellet working electrodes (WE), foil reference and counter electrodes, separators and electrolyte as described above. GITT with Li x Ti 2 S 4 (Figures 1a and 2a): Working electrode (WE) = mg Ti 2 S 4 pellet Data points were recorded every 10 mv or 10 s during current pulses Discharge at -240 μa (C/44) 0.5 h (Δx = ), OCV 4h, repeat until OCV < 2.4 V vs Li Discharge at -240 μa for 1.0 h (Δx = ), OCV 3 h, repeat until OCV < 1.88 V vs Li Discharge at -240 μa for 0.5 h, OCV 2 h, repeat until OCV < 1.80 V vs Li Charge at 240 μa for 0.5 h, OCV 2 h, repeat until OCV > 1.90 V vs Li Charge at 240 μa for 1.0 h, OCV 3 h, repeat until OCV > 2.40 V vs Li Charge at 240 μa for 0.5 h, OCV 2 h, repeat until OCV > 2.55 V vs Li Charge at 100 μa for 0.5 h, OCV 4 h, repeat until OCV > 2.65 V vs Li GITT with Mg x Ti 2 S 4 (Figures 1b and 2b): Working electrode (WE) = mg Ti 2 S 4 pellet Data points were recorded every 10 mv or 10 s during current pulses Discharge at -150 μa (C/76) 2 h (Δx = ), OCV 4 h, repeat until OCV < 1.10 V vs Mg Discharge at -75 μa (C/150) 2 h (Δx = ), OCV 4 h, repeat until OCV < 0.90 V vs Mg Charge at 150 μa for 2.0 h, OCV 4 h, repeat until OCV > 1.50 V vs Mg Charge at 75 μa for 2.0 h, OCV 4 h, repeat until OCV > 1.62 V vs Mg In the above details, small steps in x were used near the beginning and end of the V curve to probe the steeper slopes in these regions. The decision to use a shorter time or smaller current was based on the overpotential; the Li x Ti 2 S 4 pellet had a smaller overpotential, so there was no concern that the potential would rise to levels where the electrolyte would oxidize during charge pulses. This was not the case with the Mg x Ti 2 S 4 pellet, which had larger overpotentials and an electrolyte with a smaller electrochemical window. Hence, to avoid the potential descending low enough to plate Mg (< 0.1 V vs Mg) or high enough to oxidize the electrolyte (> 2.5 V vs Mg), the current was halved at the beginning and end of the V curve where the overpotentials were much larger than in the middle section. To form Figure 1, the OCV at the end of each rest period was extracted and plotted vs x to form the quasi-equilibrium (so named because the OCV period ranged from 2 to 4 h instead of being infinite) voltage curves shown in Figure 1. Each individual pulse was then re-plotted on a V vs t graph, beginning at t = 0. These new plots had linear sections in regions of S4
5 approximately 15 < t < 25 for Li x Ti 2 S 4, and 25 < t < 35 for Mg x Ti 2 S 4. The slope of these linear sections (dv/d t) along with the slope of the equilibrium voltage curve (dv/dx, Figure 1) were used to calculate the self-diffusion coefficients for both Li + and Mg 2+ (D Li and D Mg ) as a function of x: 6,9 This data was plotted in Figure 2. = 4 GAP Experimental Methods with Mg x Ti 2 S 4 (Figure 4 and Table 1):. Working electrode (WE) = 40 to 50 mg Ti 2 S 4 pellets with a SS wire mesh Data points were collected every 10 mv or 5 s during each pulse and every 2 mv or 3 min during each OCV period Pulses lasted 30 min and OCV periods lasted 2.5 hours Current pulses: 90 μa, -90 μa, 120 μa, -120 μa, 150 μa, -150 μa, 180 μa, -180 μa, 210 μa and -210 μa Temperature was maintained with a Binder convection oven. The GAP experiment was repeated at 40, 50, 60, 70 and 80 C. GAP Experimental Methods with Li x Ti 2 S 4 (Table 1): Working electrode (WE) = 90 to 100 mg Ti 2 S 4 pellets Data points were collected every 10 mv or 5 s during each pulse and every 2 mv or 3 min during each OCV period Pulses lasted 30 min and OCV periods lasted 2 hours Current pulses: 90 μa, -90 μa, 120 μa, -120 μa, 210 μa, -210 μa, 240 μa and -240 μa Temperature was maintained with a Binder convection oven. The GAP experiment was repeated at 40, 50, 60, 70 and 80 C. To form Figure 4, dv/d t was extracted from each current pulse and plotted vs the current. The slope of this plot (dv/d t/i) was then used to calculate D Mg or Li as per above, for a particular temperature. The oven temperature was then changed and the cell was allowed to reach equilibrium (i.e. the potential levelled out) before the next iteration of current pulses were initiated. Since the diffusion coefficient follows an Arrhenius dependence on temperature, 9,10 =, +, S5
6 a plot of ln(d Mg or Li ) vs 1/T should yield a straight line where E a,diff can be extracted from the slope. Supplementary Figures Figure S2. Pictures of Mg dendrites taken during cell disassembly. (A) Dendrites passing through all three layers of glass fiber filter paper separator used in the Conflat, 3-electrode cells. (B) Evidence that Celgard microporous separator did not stop the growth of dendrites. This picture shows a torn piece of glass fiber filter paper separator still attached by Mg dendrites to a Celgard 3501 separator, which is still attached to the Mg foil disk below it. The electrolyte was 0.5 M Mg(CB 11 H 12 ) 2 in tetraglyme and the current was 130 μa/cm 2. a -16 Chemical Diffusion Coefficient 1x10-7 b -16 Self-Diffusion Coefficient 1x10-7 ln(d C Mg or Li) x10-8 1x10-9 1x x10-11 D C Mg or Li (cm2 /s) ln(d Mg or Li ) x10-8 1x10-9 1x x10-11 D Mg or Li (cm 2 /s) Mg Cell Discharge Mg Cell Discharge 25 C Mg Cell Charge Mg Cell Charge 25 C Li Cell Discharge Li Cell Discharge 25 C Li Cell Charge Li Cell Charge 25 C / T (1000 / K) 1x / T (1000 / K) 1x10-12 Figure S3. Extrapolation of Arrhenius data for Li x Ti 2 S 4 /Li and Mg x Ti 2 S 4 /Mg cells down to 25 C. a) Chemical diffusion coefficients. b) Self-diffusion coefficients. Recall D C = WD, where W = thermodynamic factor. 6 Mg ions appear to diffuse 100 to 1000 times slower at room temperature than Li ions in Ti 2 S 4. S6
7 References (1) Sun, X.; Bonnick, P.; Duffort, V.; Liu, M.; Rong, Z.; Persson, K. A.; Ceder, G.; Nazar, L. F. A High Capacity Thiospinel Cathode for Mg Batteries. Energy Environ. Sci. 2016, 9, (2) Bruce, P. G.; Saidi, M. Y. Variation of the Preexponential Factor and Activation Energy for Lithium Diffusion in Cubic Titanium Disulfide. J. Solid State Chem. 1990, 88, (3) Soheilnia, N.; Kleinke, K. M.; Dashjav, E.; Cuthbert, H. L.; Greedan, J. E.; Kleinke, H. Crystal Structure and Physical Properties of a New CuTi 2 S 4 Modification in Comparison to the Thiospinel. Inorg. Chem. 2004, 43, (4) Periyapperuma, K.; Tran, T. T.; Trussler, S.; Ioboni, D.; Obrovac, M. N. Conflat Two and Three Electrode Electrochemical Cells. J. Electrochem. Soc. 2014, 161, A2182 A2187. (5) McArthur, S. G.; Geng, L.; Guo, J.; Lavallo, V. Cation Reduction and Comproportionation as Novel Strategies to Produce High Voltage, Halide Free, Carborane Based Electrolytes for Rechargeable Mg Batteries. Inorg. Chem. Front. 2015, 2, (6) Weppner, W.; Huggins, R. A. Determination of the Kinetic Parameters of Mixed- Conducting Electrodes and Application to the System Li 3 Sb. J. Electrochem. Soc. 1977, 124, (7) Wen, C. J.; Boukamp, B. A.; Huggins, R. A.; Weppner, W. Thermodynamic and Mass Transport Properties of LiAl. J. Electrochem. Soc. 1979, 126, (8) Weppner, W.; Huggins, R. A. Electrochemical Methods for Determining Kinetic Properties of Solids. Annu. Rev. Mater. Sci. 1978, 8, (9) Talaie, E.; Bonnick, P.; Sun, X.; Pang, Q.; Liang, X.; Nazar, L. F. Methods and Protocols for Electrochemical Energy Storage Materials Research. Chem. Mater. 2017, 29, (10) Van der Ven, A.; Ceder, G.; Asta, M.; Tepesch, P. D. First-Principles Theory of Ionic Diffusion with Nondilute Carriers. Phys. Rev. B 2001, 64, S7
Electronic Supplementary Material (ESI) for Chemical Communications This journal is The Royal Society of Chemistry 2013
 Sodium-ion battery based on ion exchange membranes as electrolyte and separator Chengying Cao, Weiwei Liu, Lei Tan, Xiaozhen Liao and Lei Li* School of Chemical and Chemistry Engineering, Shanghai Jiaotong
Sodium-ion battery based on ion exchange membranes as electrolyte and separator Chengying Cao, Weiwei Liu, Lei Tan, Xiaozhen Liao and Lei Li* School of Chemical and Chemistry Engineering, Shanghai Jiaotong
Electronic supplementary information. Efficient energy storage capabilities promoted by hierarchically MnCo 2 O 4
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information Efficient energy storage capabilities promoted by hierarchically
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information Efficient energy storage capabilities promoted by hierarchically
Supporting Information
 Supporting Information Low-Temperature Molten-Salt Production of Silicon Nanowires by the Electrochemical Reduction of CaSiO 3 Yifan Dong, Tyler Slade, Matthew J. Stolt, Linsen Li, Steven N. Girard, Liqiang
Supporting Information Low-Temperature Molten-Salt Production of Silicon Nanowires by the Electrochemical Reduction of CaSiO 3 Yifan Dong, Tyler Slade, Matthew J. Stolt, Linsen Li, Steven N. Girard, Liqiang
Lithium Potassium Manganese Mixed Metal Oxide Material for Rechargeable Electrochemical Cells
 Lithium Potassium Manganese Mixed Metal Oxide Material for Rechargeable Electrochemical Cells Terrill B. Atwater 1,2 and Alvin J. Salkind 2,3 1 US Army RDECOM, CERDEC, Ft. Monmouth NJ 2 Rutgers University,
Lithium Potassium Manganese Mixed Metal Oxide Material for Rechargeable Electrochemical Cells Terrill B. Atwater 1,2 and Alvin J. Salkind 2,3 1 US Army RDECOM, CERDEC, Ft. Monmouth NJ 2 Rutgers University,
Novel concept of rechargeable battery using iron oxide nanorods. anode and nickel hydroxide cathode in aqueous electrolyte
 Supplementary Information for: Novel concept of rechargeable battery using iron oxide nanorods anode and nickel hydroxide cathode in aqueous electrolyte Zhaolin Liu *, Siok Wei Tay and Xu Li Institute
Supplementary Information for: Novel concept of rechargeable battery using iron oxide nanorods anode and nickel hydroxide cathode in aqueous electrolyte Zhaolin Liu *, Siok Wei Tay and Xu Li Institute
Supporting Information
 Supporting Information Experimental Methods Pt ALD. The precursor used for ALD was trimethyl-methylcyclopentadienyl-platinum(iv) (MeCpPtMe 3 ) (Strem Chemicals, 99%), which has been widely reported for
Supporting Information Experimental Methods Pt ALD. The precursor used for ALD was trimethyl-methylcyclopentadienyl-platinum(iv) (MeCpPtMe 3 ) (Strem Chemicals, 99%), which has been widely reported for
Supplemental Information. A Low-Cost and High-Energy Hybrid. Iron-Aluminum Liquid Battery Achieved. by Deep Eutectic Solvents
 JOUL, Volume 1 Supplemental Information A Low-Cost and High-Energy Hybrid Iron-Aluminum Liquid Battery Achieved by Deep Eutectic Solvents Leyuan Zhang, Changkun Zhang, Yu Ding, Katrina Ramirez-Meyers,
JOUL, Volume 1 Supplemental Information A Low-Cost and High-Energy Hybrid Iron-Aluminum Liquid Battery Achieved by Deep Eutectic Solvents Leyuan Zhang, Changkun Zhang, Yu Ding, Katrina Ramirez-Meyers,
High Performance Lithium Battery Anodes Using Silicon Nanowires
 Supporting Online Materials For High Performance Lithium Battery Anodes Using Silicon Nanowires Candace K. Chan, Hailin Peng, Gao Liu, Kevin McIlwrath, Xiao Feng Zhang, Robert A. Huggins and Yi Cui * *To
Supporting Online Materials For High Performance Lithium Battery Anodes Using Silicon Nanowires Candace K. Chan, Hailin Peng, Gao Liu, Kevin McIlwrath, Xiao Feng Zhang, Robert A. Huggins and Yi Cui * *To
In situ generation of Li 2 FeSiO 4 coating on MWNT as a high rate cathode material for lithium ion batteries
 Supporting Information: In situ generation of Li 2 FeSiO 4 coating on MWNT as a high rate cathode material for lithium ion batteries Yi Zhao, Jiaxin Li, Ning Wang, Chuxin Wu, Yunhai Ding, Lunhui Guan*
Supporting Information: In situ generation of Li 2 FeSiO 4 coating on MWNT as a high rate cathode material for lithium ion batteries Yi Zhao, Jiaxin Li, Ning Wang, Chuxin Wu, Yunhai Ding, Lunhui Guan*
Electroactive Polymer for Controlling Overcharge in Lithium-Ion Batteries
 PSI-SR-1261 Electroactive Polymer for Controlling Overcharge in Lithium-Ion Batteries A. Newman R. Pawle K. White J. Lennhoff A. Newman, R. Pawle, K. White, J. Lennhoff, "Electroactive Polymer for Controlling
PSI-SR-1261 Electroactive Polymer for Controlling Overcharge in Lithium-Ion Batteries A. Newman R. Pawle K. White J. Lennhoff A. Newman, R. Pawle, K. White, J. Lennhoff, "Electroactive Polymer for Controlling
Supporting Information. Investigation of the Reversible Intercalation/Deintercalation of Al
 Supporting Information Investigation of the Reversible Intercalation/Deintercalation of Al into the Novel Li 3 VO 4 @C Microsphere Composite Cathode Material for Aluminum-Ion Batteries Jiali Jiang, He
Supporting Information Investigation of the Reversible Intercalation/Deintercalation of Al into the Novel Li 3 VO 4 @C Microsphere Composite Cathode Material for Aluminum-Ion Batteries Jiali Jiang, He
ELECTROCHEMICAL REDUCTION OF TITANIUM DIOXIDE THIN FILM IN LiCl-KCl-CaCl 2 EUTECTIC MELT
 ELECTROCHEMICAL REDUCTION OF TITANIUM DIOXIDE THIN FILM IN LiCl-KCl-CaCl 2 EUTECTIC MELT Yasushi Katayama Department of Applied Chemistry, Faculty of Science and Technology, Keio University 3-14-1, Hiyoshi,
ELECTROCHEMICAL REDUCTION OF TITANIUM DIOXIDE THIN FILM IN LiCl-KCl-CaCl 2 EUTECTIC MELT Yasushi Katayama Department of Applied Chemistry, Faculty of Science and Technology, Keio University 3-14-1, Hiyoshi,
SOLIK Li-hochleitende Keramiken für all-solid-state Batterien
 SOLIK Li-hochleitende Keramiken für all-solid-state Batterien Dr. Ningxin ZHANG Electric Drive Technologies Center for Low-Emission Transport Austrian Institute of Technology GmbH Outline Basic Data Project
SOLIK Li-hochleitende Keramiken für all-solid-state Batterien Dr. Ningxin ZHANG Electric Drive Technologies Center for Low-Emission Transport Austrian Institute of Technology GmbH Outline Basic Data Project
Supplementary Information
 Supplementary Information Comparative study on Na 2 MnPO 4 F and Li 2 MnPO 4 F for rechargeable battery cathodes Sung-Wook Kim a,b, Dong-Hwa Seo a, Hyung-Sub Kim c, Kyu-Young Park d, and Kisuk Kang a,b*
Supplementary Information Comparative study on Na 2 MnPO 4 F and Li 2 MnPO 4 F for rechargeable battery cathodes Sung-Wook Kim a,b, Dong-Hwa Seo a, Hyung-Sub Kim c, Kyu-Young Park d, and Kisuk Kang a,b*
Supplementary information. performance Li-ion battery
 Supplementary information The investigation of Ni(OH) 2 /Ni as anode for high performance Li-ion battery Shibing Ni a, Xiaohu Lv a, Tao Li a, Xuelin Yang a,and Lulu Zhang a College of Mechanical and Material
Supplementary information The investigation of Ni(OH) 2 /Ni as anode for high performance Li-ion battery Shibing Ni a, Xiaohu Lv a, Tao Li a, Xuelin Yang a,and Lulu Zhang a College of Mechanical and Material
EMA4303/5305 Electrochemical Engineering Lecture 05 Applications (1)
 EMA4303/5305 Electrochemical Engineering Lecture 05 Applications (1) Prof. Zhe Cheng Mechanical & Materials Engineering Florida International University Corrosion Definition Electrochemical attack of metals
EMA4303/5305 Electrochemical Engineering Lecture 05 Applications (1) Prof. Zhe Cheng Mechanical & Materials Engineering Florida International University Corrosion Definition Electrochemical attack of metals
Nickel Electroplating
 Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
Single-crystalline LiFePO 4 Nanosheets for High-rate Li-ion Batteries
 /8 SUPPORTING INFORMATION Single-crystalline LiFePO 4 Nanosheets for High-rate Li-ion Batteries Yu Zhao, Lele Peng, Borui Liu, Guihua Yu* Materials Science and Engineering Program and Department of Mechanical
/8 SUPPORTING INFORMATION Single-crystalline LiFePO 4 Nanosheets for High-rate Li-ion Batteries Yu Zhao, Lele Peng, Borui Liu, Guihua Yu* Materials Science and Engineering Program and Department of Mechanical
Supplementary Information
 Electronic Supplementary Material (ESI) for Materials Chemistry Frontiers. This journal is the Partner Organisations 2017 Supplementary Information Self-Standing Bi 2 O 3 Nanoparticles/Carbon Nanofiber
Electronic Supplementary Material (ESI) for Materials Chemistry Frontiers. This journal is the Partner Organisations 2017 Supplementary Information Self-Standing Bi 2 O 3 Nanoparticles/Carbon Nanofiber
Electronic Supplementary Information. Synthesis and crystal structure of a rare square-planar Co (II) complex of a hydroxyamidinate ligand.
 Electronic Supplementary Information Synthesis and crystal structure of a rare square-planar Co (II) complex of a hydroxyamidinate ligand. Mihaela Cibian, a Sofia Derossi, a and Garry S. Hanan* a Département
Electronic Supplementary Information Synthesis and crystal structure of a rare square-planar Co (II) complex of a hydroxyamidinate ligand. Mihaela Cibian, a Sofia Derossi, a and Garry S. Hanan* a Département
In Situ IonicÕElectric Conductivity Measurement of La 0.55 Li 0.35 TiO 3 Ceramic at Different Li Insertion Levels
 A1196 Journal of The Electrochemical Society, 151 8 A1196-A1201 2004 0013-4651/2004/151 8 /A1196/6/$7.00 The Electrochemical Society, Inc. In Situ IonicÕElectric Conductivity Measurement of La 0.55 Li
A1196 Journal of The Electrochemical Society, 151 8 A1196-A1201 2004 0013-4651/2004/151 8 /A1196/6/$7.00 The Electrochemical Society, Inc. In Situ IonicÕElectric Conductivity Measurement of La 0.55 Li
Electrochemical properties of alkal. Citation Electrochimica Acta (2010), 55(3):
 KURENAI : Kyoto University Researc Title Electrochemical properties of alkal bis(trifluoromethylsulfonyl)amides Author(s) Kubota, Keigo; Tamaki, Kenichiro; N Takuya; Hagiwara, Rika Citation Electrochimica
KURENAI : Kyoto University Researc Title Electrochemical properties of alkal bis(trifluoromethylsulfonyl)amides Author(s) Kubota, Keigo; Tamaki, Kenichiro; N Takuya; Hagiwara, Rika Citation Electrochimica
Carbon Nanotube-Based Supercapacitors with Excellent AC-Line
 SUPPORTING INFORMATION FOR: Carbon Nanotube-Based Supercapacitors with Excellent AC-Line Filtering and Rate Capability via Improved Interfacial Impedance Yverick Rangom, Xiaowu (Shirley) Tang*, and Linda
SUPPORTING INFORMATION FOR: Carbon Nanotube-Based Supercapacitors with Excellent AC-Line Filtering and Rate Capability via Improved Interfacial Impedance Yverick Rangom, Xiaowu (Shirley) Tang*, and Linda
components and inorganic Li salts from PST-90. When contacting with Li metal, PST-
 Supplementary Figure 1. Proposed mechanism for the formation of organic components and inorganic Li salts from PST-90. When contacting with Li metal, PST- 1 90 first generates high order organopolysulfide
Supplementary Figure 1. Proposed mechanism for the formation of organic components and inorganic Li salts from PST-90. When contacting with Li metal, PST- 1 90 first generates high order organopolysulfide
Terephthalonitrile-derived nitrogen-rich networks for high
 Electronic Supplementary Information Terephthalonitrile-derived nitrogen-rich networks for high performance supercapacitors Long Hao, a Bin Luo, a Xianglong Li, a Meihua Jin, a Yan Fang, a Zhihong Tang,
Electronic Supplementary Information Terephthalonitrile-derived nitrogen-rich networks for high performance supercapacitors Long Hao, a Bin Luo, a Xianglong Li, a Meihua Jin, a Yan Fang, a Zhihong Tang,
Factors Governing Life of High-Energy Lithium-Ion Cells
 Factors Governing Life of High-Energy Lithium-Ion Cells D.P. Abraham IBA 2013 March 11, 2013 Barcelona, Spain Research sponsors are both Government and Private Sector 2 Diagnostics Overview Use of characterization
Factors Governing Life of High-Energy Lithium-Ion Cells D.P. Abraham IBA 2013 March 11, 2013 Barcelona, Spain Research sponsors are both Government and Private Sector 2 Diagnostics Overview Use of characterization
Supplementary Information.
 Supplementary Information. This file contains additional experimental and analytical information. The organic ionic plastic crystals were synthesised according to the literature procedures. 1-4 Chemicals:
Supplementary Information. This file contains additional experimental and analytical information. The organic ionic plastic crystals were synthesised according to the literature procedures. 1-4 Chemicals:
Tutorial Corrosion II. Electrochemical characterization with EC-Lab techniques
 Tutorial Corrosion II Electrochemical characterization with EC-Lab techniques 1 OUTLINE 1. Introduction 2. Types of corrosion a) Uniform corrosion b) Localized corrosion 3. Corrosion experiment 4. EC-Lab
Tutorial Corrosion II Electrochemical characterization with EC-Lab techniques 1 OUTLINE 1. Introduction 2. Types of corrosion a) Uniform corrosion b) Localized corrosion 3. Corrosion experiment 4. EC-Lab
IBM Almaden June 27, Seongmin Ha, Dongho Koo, Kyu Tae Lee * Chemical and Biological Engineering Seoul National University
 IBM Almaden June 27, 2017 Seongmin Ha, Dongho Koo, Kyu Tae Lee * Chemical and Biological Engineering Seoul National University (ktlee@snu.ac.kr) 1) Introduction 2) Failure mechanism of a redox mediator
IBM Almaden June 27, 2017 Seongmin Ha, Dongho Koo, Kyu Tae Lee * Chemical and Biological Engineering Seoul National University (ktlee@snu.ac.kr) 1) Introduction 2) Failure mechanism of a redox mediator
Electronic Supporting Information
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supporting Information Copyright Royal Society of Chemistry, London,
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supporting Information Copyright Royal Society of Chemistry, London,
All-solid-state Li battery using a light-weight solid electrolyte
 All-solid-state Li battery using a light-weight solid electrolyte Hitoshi Takamura Department of Materials Science, Graduate School of Engineering, Tohoku University Europe-Japan Symposium, Electrical
All-solid-state Li battery using a light-weight solid electrolyte Hitoshi Takamura Department of Materials Science, Graduate School of Engineering, Tohoku University Europe-Japan Symposium, Electrical
Soft silicon anodes for lithium ion batteries
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2014 Supplementary Information Soft silicon anodes for lithium ion batteries Qizhen
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2014 Supplementary Information Soft silicon anodes for lithium ion batteries Qizhen
Morphology controlled synthesis of monodispersed manganese. sulfide nanocrystals and their primary application for supercapacitor
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Morphology controlled synthesis of monodispersed manganese sulfide nanocrystals
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Morphology controlled synthesis of monodispersed manganese sulfide nanocrystals
Red Phosphorus Nano-Dots on Reduced Graphene Oxide as Flexible High-Performance Anode for Sodium-Ion Batteries
 Red Phosphorus Nano-Dots on Reduced Graphene Oxide as Flexible High-Performance Anode for Sodium-Ion Batteries Yihang Liu 1, Anyi Zhang 2, Chenfei Shen 2, Qingzhou Liu 2, Xuan Cao 2, Yuqiang Ma 2, Liang
Red Phosphorus Nano-Dots on Reduced Graphene Oxide as Flexible High-Performance Anode for Sodium-Ion Batteries Yihang Liu 1, Anyi Zhang 2, Chenfei Shen 2, Qingzhou Liu 2, Xuan Cao 2, Yuqiang Ma 2, Liang
Three-dimensional NiFe Layered Double Hydroxide Film for Highefficiency
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Three-dimensional NiFe Layered Double Hydroxide Film for Highefficiency Oxygen Evolution Reaction
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Three-dimensional NiFe Layered Double Hydroxide Film for Highefficiency Oxygen Evolution Reaction
International Journal of Chemical Studies
 ISSN: 2321-4902 Volume 1 Issue 1 Online Available at www.chemijournal.com International Journal of Chemical Studies Calculation of Diffusion Coefficients and Layer Thickness for Oxidation the Ferrocene
ISSN: 2321-4902 Volume 1 Issue 1 Online Available at www.chemijournal.com International Journal of Chemical Studies Calculation of Diffusion Coefficients and Layer Thickness for Oxidation the Ferrocene
Supporting Information. Christina W. Li and Matthew W. Kanan* *To whom correspondence should be addressed.
 Supporting Information CO 2 Reduction at Low Overpotential on Cu Electrodes Resulting from the Reduction of Thick Cu 2 O Films Christina W. Li and Matthew W. Kanan* *To whom correspondence should be addressed.
Supporting Information CO 2 Reduction at Low Overpotential on Cu Electrodes Resulting from the Reduction of Thick Cu 2 O Films Christina W. Li and Matthew W. Kanan* *To whom correspondence should be addressed.
PVP-Functionalized Nanometer Scale Metal Oxide Coatings for. Cathode Materials: Successful Application to LiMn 2 O 4 Spinel.
 PVP-Functionalized Nanometer Scale Metal Oxide Coatings for Cathode Materials: Successful Application to LiMn 2 O 4 Spinel Nanoparticles Hyesun Lim, Jaephil Cho* Department of Applied Chemistry Hanyang
PVP-Functionalized Nanometer Scale Metal Oxide Coatings for Cathode Materials: Successful Application to LiMn 2 O 4 Spinel Nanoparticles Hyesun Lim, Jaephil Cho* Department of Applied Chemistry Hanyang
Cycle life performance of lithium-ion pouch cells
 Journal of Power Sources 158 (2006) 679 688 Cycle life performance of lithium-ion pouch cells Karthikeyan Kumaresan, Qingzhi Guo, Premanand Ramadass, Ralph E. White Department of Chemical Engineering,
Journal of Power Sources 158 (2006) 679 688 Cycle life performance of lithium-ion pouch cells Karthikeyan Kumaresan, Qingzhi Guo, Premanand Ramadass, Ralph E. White Department of Chemical Engineering,
Electrochemical and Transport Properties of Ions in Mixtures of. Electroactive Ionic Liquid and Propylene Carbonate with a Lithium
 Supporting Information Electrochemical and Transport Properties of Ions in Mixtures of Electroactive Ionic Liquid and Propylene Carbonate with a Lithium Salt for Lithium-ion Batteries Bruno Gélinas, Myriann
Supporting Information Electrochemical and Transport Properties of Ions in Mixtures of Electroactive Ionic Liquid and Propylene Carbonate with a Lithium Salt for Lithium-ion Batteries Bruno Gélinas, Myriann
Supplementary Figure 1. Photographs of the Suaeda glauca (S. glauca) Bunge at different stages of metal ion absorption. (a) Photographs of S.
 1 2 3 4 5 6 7 Supplementary Figure 1. Photographs of the Suaeda glauca (S. glauca) Bunge at different stages of metal ion absorption. (a) Photographs of S. glauca after absorption of tin salt. (b) Photographs
1 2 3 4 5 6 7 Supplementary Figure 1. Photographs of the Suaeda glauca (S. glauca) Bunge at different stages of metal ion absorption. (a) Photographs of S. glauca after absorption of tin salt. (b) Photographs
to enable Lithium metal electrodes IBA2013, Barcelona, Spain
 Fluorine free ionic liquid electrolytes to enable Lithium metal electrodes IBA2013, Barcelona, Spain A. S. Best, Martin (Hyun Gook) YOON, G. H. Lane, Y. Shekibi, P. C. Howlett, M. Forsyth & D. R. MacFarlane
Fluorine free ionic liquid electrolytes to enable Lithium metal electrodes IBA2013, Barcelona, Spain A. S. Best, Martin (Hyun Gook) YOON, G. H. Lane, Y. Shekibi, P. C. Howlett, M. Forsyth & D. R. MacFarlane
Ionic Conductivity and Solid Electrolytes II: Materials and Applications
 Ionic Conductivity and Solid Electrolytes II: Materials and Applications Chemistry 754 Solid State Chemistry Lecture #27 June 4, 2003 References A. Manthiram & J. Kim Low Temperature Synthesis of Insertion
Ionic Conductivity and Solid Electrolytes II: Materials and Applications Chemistry 754 Solid State Chemistry Lecture #27 June 4, 2003 References A. Manthiram & J. Kim Low Temperature Synthesis of Insertion
Safe, Inexpensive, Long Life, High Power and Efficiency Batteries For Grid Scale Energy Storage Applications
 Safe, Inexpensive, Long Life, High Power and Efficiency Batteries For Grid Scale Energy Storage Applications Investigators Yi Cui, Associate Professor; Robert Huggins, Professor; Mauro Pasta, Postdoctoral
Safe, Inexpensive, Long Life, High Power and Efficiency Batteries For Grid Scale Energy Storage Applications Investigators Yi Cui, Associate Professor; Robert Huggins, Professor; Mauro Pasta, Postdoctoral
Comparison between the article and script of thesis
 Comparison between the article and script of thesis All nanomaterials and a part of results presented and discussed in this article titled Efficient multi-metallic anode catalysts in a PEM water electrolyzer
Comparison between the article and script of thesis All nanomaterials and a part of results presented and discussed in this article titled Efficient multi-metallic anode catalysts in a PEM water electrolyzer
Facile, mild and fast thermal-decomposition reduction of graphene oxide in air and its application in high-performance lithium batteries
 Facile, mild and fast thermal-decomposition reduction of graphene oxide in air and its application in high-performance lithium batteries Zhong-li Wang, Dan Xu, Yun Huang, Zhong Wu, Li-min Wang and Xin-bo
Facile, mild and fast thermal-decomposition reduction of graphene oxide in air and its application in high-performance lithium batteries Zhong-li Wang, Dan Xu, Yun Huang, Zhong Wu, Li-min Wang and Xin-bo
IRON REDOX EQUILIBRIUM AND DIFFUSIVITY IN MIXED ALKALI-ALKALINE EARTH-SILICA GLASS MELTS
 Original papers IRON REDOX EQUILIBRIUM AND DIFFUSIVITY IN MIXED ALKALI-ALKALINE EARTH-SILICA GLASS MELTS KI-DONG KIM Department of Materials Science and Engineering, Kunsan National University, Kunsan
Original papers IRON REDOX EQUILIBRIUM AND DIFFUSIVITY IN MIXED ALKALI-ALKALINE EARTH-SILICA GLASS MELTS KI-DONG KIM Department of Materials Science and Engineering, Kunsan National University, Kunsan
Amorphous Metallic Glass as New High Power and Energy Density Anodes For Lithium Ion Rechargeable Batteries
 Amorphous Metallic Glass as New High Power and Energy Density Anodes For Lithium Ion Rechargeable Batteries Meng Ying 1, Li Yi 1,2, E. M. Arroyo 3 and G. Ceder 1,3 1. Singapore-MIT Alliance, Advanced Materials
Amorphous Metallic Glass as New High Power and Energy Density Anodes For Lithium Ion Rechargeable Batteries Meng Ying 1, Li Yi 1,2, E. M. Arroyo 3 and G. Ceder 1,3 1. Singapore-MIT Alliance, Advanced Materials
a) The self-diffusion coefficient of a metal with cubic structure can be expressed as
 EXERCISES KJM5120 Chapter 5; Diffusion 1. Random (self) diffusion a) The self-diffusion coefficient of a metal with cubic structure can be expressed as 1 n D = s 6 t 2 where n/t represents the jump frequency
EXERCISES KJM5120 Chapter 5; Diffusion 1. Random (self) diffusion a) The self-diffusion coefficient of a metal with cubic structure can be expressed as 1 n D = s 6 t 2 where n/t represents the jump frequency
Corrosion Rate Measurement on C-Steel
 Measurements of corrosion rate on Carbon-steel using Electrochemical (potentiodynamic Polarization, EIS etc.) technique. Corrosion Rate Measurement on C-Steel Abdullah Al Ashraf 1. Introduction: The degradation
Measurements of corrosion rate on Carbon-steel using Electrochemical (potentiodynamic Polarization, EIS etc.) technique. Corrosion Rate Measurement on C-Steel Abdullah Al Ashraf 1. Introduction: The degradation
Salts purification and redox potential measurement for the molten LiF-ThF 4 -UF 4 mixture Presented by Valery K. Afonichkin
 First ACSEPT International Workshop Lisbon, Portugal, 31 March 2 April 2010 Salts purification and redox potential measurement for the molten LiF-ThF 4 -UF 4 mixture Presented by Valery K. Afonichkin ISTC#
First ACSEPT International Workshop Lisbon, Portugal, 31 March 2 April 2010 Salts purification and redox potential measurement for the molten LiF-ThF 4 -UF 4 mixture Presented by Valery K. Afonichkin ISTC#
INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES. Introduction. Electrochemistry Revised 4/28/14
 INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES Introduction Electrochemical Cells In this part of the experiment, four half cells are created by immersing metal strips of zinc, copper,
INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES Introduction Electrochemical Cells In this part of the experiment, four half cells are created by immersing metal strips of zinc, copper,
Understanding Cation-Disordered Cathode Materials Based on Percolation Theory and Ligand Field Theory
 2016 ECS Prime meeting (10/5/2016, 8:40 9:00 am) Understanding Cation-Disordered Cathode Materials Based on Percolation Theory and gand Field Theory Jinhyuk Lee, Dong-Hwa Seo, Alexander Urban, Gerbrand
2016 ECS Prime meeting (10/5/2016, 8:40 9:00 am) Understanding Cation-Disordered Cathode Materials Based on Percolation Theory and gand Field Theory Jinhyuk Lee, Dong-Hwa Seo, Alexander Urban, Gerbrand
Supporting Information for Manuscript B516757D
 Supporting Information for Manuscript B516757D 1. UV-Vis absorption spectra Absorbance (a.u.) 0.4 0.2 5F 6F 7F 0.0 300 400 500 Wavelength (nm) Figure S1 UV-Vis spectra of, 5F, 6F and 7F in CHCl 3 solutions
Supporting Information for Manuscript B516757D 1. UV-Vis absorption spectra Absorbance (a.u.) 0.4 0.2 5F 6F 7F 0.0 300 400 500 Wavelength (nm) Figure S1 UV-Vis spectra of, 5F, 6F and 7F in CHCl 3 solutions
Studies on Alloys and Composites that Undergo Anomalous Codeposition
 Studies on Alloys and Composites that Undergo Anomalous Codeposition Electrochemical of of South Columbia, SC908 09, 1998 Electrochemical Systems Studied FeNi alloys and FeNiSiO composites Electrodeposition
Studies on Alloys and Composites that Undergo Anomalous Codeposition Electrochemical of of South Columbia, SC908 09, 1998 Electrochemical Systems Studied FeNi alloys and FeNiSiO composites Electrodeposition
Fundamental Chemistry of Sion Power Li/S Battery. Yuriy Mikhaylik Sion Power Corporation, 9040 South Rita Road, Tucson, Arizona, 85747, USA
 Fundamental Chemistry of Sion Power Li/S Battery Yuriy Mikhaylik Sion Power Corporation, 9040 South Rita Road, Tucson, Arizona, 85747, USA Outline Thermodynamics of Li-S Discharge-charge mechanism in the
Fundamental Chemistry of Sion Power Li/S Battery Yuriy Mikhaylik Sion Power Corporation, 9040 South Rita Road, Tucson, Arizona, 85747, USA Outline Thermodynamics of Li-S Discharge-charge mechanism in the
Electrochemical Methods
 Electrochemical Methods John F. Berry June 19th, 2014 Why perform electrochemistry? Me Me N N N 3 Fe III O N N Me O [(Me 3 cyclam-acetate)fen 3 ]PF 6 Current Why perform electrochemistry? Me Me N N N 3
Electrochemical Methods John F. Berry June 19th, 2014 Why perform electrochemistry? Me Me N N N 3 Fe III O N N Me O [(Me 3 cyclam-acetate)fen 3 ]PF 6 Current Why perform electrochemistry? Me Me N N N 3
Preparation of Bi-Based Ternary Oxide Photoanodes, BiVO 4,
 Preparation of Bi-Based Ternary Oxide Photoanodes, BiVO 4, Bi 2 WO 6 and Bi 2 Mo 3 O 12, Using Dendritic Bi Metal Electrodes Donghyeon Kang, a, Yiseul Park, a, James C. Hill, b and Kyoung-Shin Choi a,*
Preparation of Bi-Based Ternary Oxide Photoanodes, BiVO 4, Bi 2 WO 6 and Bi 2 Mo 3 O 12, Using Dendritic Bi Metal Electrodes Donghyeon Kang, a, Yiseul Park, a, James C. Hill, b and Kyoung-Shin Choi a,*
Stability of Sodium Couple in Organic and Inorganic Molten Salt Electrolytes Investigated with Electrochemical Quartz Crystal Microbalance
 A346 0013-4651/2001/148 4 /A346/5/$7.00 The Electrochemical Society, Inc. Stability of Sodium Couple in Organic and Inorganic Molten Salt Electrolytes Investigated with Electrochemical Quartz Crystal Microbalance
A346 0013-4651/2001/148 4 /A346/5/$7.00 The Electrochemical Society, Inc. Stability of Sodium Couple in Organic and Inorganic Molten Salt Electrolytes Investigated with Electrochemical Quartz Crystal Microbalance
Spray Drying Method for Large-Scale and High. Performance Silicon Negative Electrodes in Li-ion. Batteries
 SUPPORTING INFORMATION Spray Drying Method for Large-Scale and High Performance Silicon Negative Electrodes in Li-ion Batteries Dae Soo Jung, Tae Hoon Hwang, Seung Bin Park, and Jang Wook Choi,,* Graduate
SUPPORTING INFORMATION Spray Drying Method for Large-Scale and High Performance Silicon Negative Electrodes in Li-ion Batteries Dae Soo Jung, Tae Hoon Hwang, Seung Bin Park, and Jang Wook Choi,,* Graduate
IN SITU MONITORING OF REINFORCEMENT CORROSION BY MEANS OF ELECTROCHEMICAL METHODS
 ABSTRACT IN SITU MONITORING OF REINFORCEMENT CORROSION BY MEANS OF ELECTROCHEMICAL METHODS Oskar Klinghoffer, M.Sc. FORCE Institute, Park Allé 345, DK-2605 Brøndby, Denmark The well-known and usually adapted
ABSTRACT IN SITU MONITORING OF REINFORCEMENT CORROSION BY MEANS OF ELECTROCHEMICAL METHODS Oskar Klinghoffer, M.Sc. FORCE Institute, Park Allé 345, DK-2605 Brøndby, Denmark The well-known and usually adapted
Corrosion characteristics of mild steel in aqueous solution of formic acid containing some acetic acid
 Indian Journal of Chemical Technology Vol. 15, March 2008, pp. 174-179 Corrosion characteristics of mild steel in aqueous solution of formic acid containing some acetic acid S K Singh, Ashim K Mukherjee*
Indian Journal of Chemical Technology Vol. 15, March 2008, pp. 174-179 Corrosion characteristics of mild steel in aqueous solution of formic acid containing some acetic acid S K Singh, Ashim K Mukherjee*
J. Mater. Sci. Technol., 2010, 26(11),
 J. Mater. Sci. Technol., 2010, 26(11), 1016-1020. Effects of Current Density on the Microstructure and the Corrosion Resistance of Alumina Coatings Embedded with SiC Nano-particles Produced by Micro-arc
J. Mater. Sci. Technol., 2010, 26(11), 1016-1020. Effects of Current Density on the Microstructure and the Corrosion Resistance of Alumina Coatings Embedded with SiC Nano-particles Produced by Micro-arc
1.Is the onsite potential affected by the voltage scanning rate? Or, is it a thermodynamically controlled process or kinetics dominant?
 Reviewers' comments: Reviewer #1 (Remarks to the Author): In this paper, Hersbach et al. reported an impressive study of the anisotropic etching of Pt electrodes at the onset of cathodic corrosion. This
Reviewers' comments: Reviewer #1 (Remarks to the Author): In this paper, Hersbach et al. reported an impressive study of the anisotropic etching of Pt electrodes at the onset of cathodic corrosion. This
Electronic Supplementary Information (ESI) for
 Electronic Supplementary Information (ESI) for Binder-free CNT network/mos 2 composite as high performance anode material in lithium ion battery Congxiang Lu, ab Wen-wen Liu b, Hong Li c and Beng Kang
Electronic Supplementary Information (ESI) for Binder-free CNT network/mos 2 composite as high performance anode material in lithium ion battery Congxiang Lu, ab Wen-wen Liu b, Hong Li c and Beng Kang
2. Crystallization. A. Background
 2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons.
 Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons. A.(16) An electrochemical cell is prepared with a strip of manganese metal dipping in to a 1.0 M MnSO 4 solution
Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons. A.(16) An electrochemical cell is prepared with a strip of manganese metal dipping in to a 1.0 M MnSO 4 solution
Summer School June 2-4 th 2015
 MAT4BAT Advanced materials for batteries Summer School June 2-4 th 2015 «Electrode formulation and processing» Dane Sotta (CEA-Liten, France) Mat4Bat Summer School Dane Sotta (CEA) June 3 rd 2015 1 Outline
MAT4BAT Advanced materials for batteries Summer School June 2-4 th 2015 «Electrode formulation and processing» Dane Sotta (CEA-Liten, France) Mat4Bat Summer School Dane Sotta (CEA) June 3 rd 2015 1 Outline
CHARACTERISTICS OF ELECTRIC-DOUBLE LAYER CAPACITOR OF THE METAL-POROUS CARBON COMPOSITE FROM ION EXCHANGE RESIN
 CHARACTERISTICS OF ELECTRIC-DOUBLE LAYER CAPACITOR OF THE METAL-POROUS CARBON COMPOSITE FROM ION EXCHANGE RESIN Akinori Muto, Norimasa Yamada, Md. A. Uddin, Kenji Kojima*, Satoshi Ibaraki *, Chisato Marumo
CHARACTERISTICS OF ELECTRIC-DOUBLE LAYER CAPACITOR OF THE METAL-POROUS CARBON COMPOSITE FROM ION EXCHANGE RESIN Akinori Muto, Norimasa Yamada, Md. A. Uddin, Kenji Kojima*, Satoshi Ibaraki *, Chisato Marumo
Supporting Information
 Supporting Information A New Polymorph of Na2MnP2O7 as a 3.6 V Cathode Material for Sodium-ion Batteries Prabeer Barpanda, * Tian Ye, Maxim Avdeev, Sai-Cheong Chung, Atsuo Yamada * Department of Chemical
Supporting Information A New Polymorph of Na2MnP2O7 as a 3.6 V Cathode Material for Sodium-ion Batteries Prabeer Barpanda, * Tian Ye, Maxim Avdeev, Sai-Cheong Chung, Atsuo Yamada * Department of Chemical
Schottky Tunnel Contacts for Efficient Coupling of Photovoltaics and Catalysts
 Schottky Tunnel Contacts for Efficient Coupling of Photovoltaics and Catalysts Christopher E. D. Chidsey Department of Chemistry Stanford University Collaborators: Paul C. McIntyre, Y.W. Chen, J.D. Prange,
Schottky Tunnel Contacts for Efficient Coupling of Photovoltaics and Catalysts Christopher E. D. Chidsey Department of Chemistry Stanford University Collaborators: Paul C. McIntyre, Y.W. Chen, J.D. Prange,
What happens if we connect Zn and Pt in HCl solution? Corrosion of platinum (Pt) in HCl. 1. If Zn and Pt are not connected
 Corrosion of platinum (Pt) in HCl Now if we place a piece of Pt in HCl, what will happen? Pt does not corrode does not take part in the electrochemical reaction Pt is a noble metal Pt acts as a reference
Corrosion of platinum (Pt) in HCl Now if we place a piece of Pt in HCl, what will happen? Pt does not corrode does not take part in the electrochemical reaction Pt is a noble metal Pt acts as a reference
PERFORMANCE ANALYSIS OF BENCHMARK PLANT FOR SELECTIVE LITHIUM RECOVERY FROM SEAWATER
 PERFORMANCE ANALYSIS OF BENCHMARK PLANT FOR SELECTIVE LITHIUM RECOVERY FROM SEAWATER Kazuharu YOSHIZUKA Faculty of Environmental Engineering, The University of Kitakyushu Marek HOLBA, Takeshi YASUNAGA,
PERFORMANCE ANALYSIS OF BENCHMARK PLANT FOR SELECTIVE LITHIUM RECOVERY FROM SEAWATER Kazuharu YOSHIZUKA Faculty of Environmental Engineering, The University of Kitakyushu Marek HOLBA, Takeshi YASUNAGA,
Low Cost Bipolar Plates for Large Scale PEM Electrolyzers
 1 Low Cost Bipolar Plates for Large Scale PEM Electrolyzers A. S. Gago, A. S. Ansar, P. Gazdzicki, N. Wagner, J. Arnold, K. A. Friedrich Electrochemical Energy Technology Institute of Engineering Thermodynamics
1 Low Cost Bipolar Plates for Large Scale PEM Electrolyzers A. S. Gago, A. S. Ansar, P. Gazdzicki, N. Wagner, J. Arnold, K. A. Friedrich Electrochemical Energy Technology Institute of Engineering Thermodynamics
Journal of Power Sources
 Journal of Power Sources 196 (2011) 1442 1448 Contents lists available at ScienceDirect Journal of Power Sources journal homepage: www.elsevier.com/locate/jpowsour Strain accommodation and potential hysteresis
Journal of Power Sources 196 (2011) 1442 1448 Contents lists available at ScienceDirect Journal of Power Sources journal homepage: www.elsevier.com/locate/jpowsour Strain accommodation and potential hysteresis
A R C H I V E S O F M E T A L L U R G Y A N D M A T E R I A L S Volume Issue 2 DOI: /amm
 A R C H I V E S O F M E T A L L U R G Y A N D M A T E R I A L S Volume 60 2015 Issue 2 DOI: 10.1515/amm-2015-0086 S.M. SHIN, G.J. JUNG, WOO-JIN LEE, C.Y. KANG, J.P. WANG, RECOVERY OF ELECTRODIC POWDER
A R C H I V E S O F M E T A L L U R G Y A N D M A T E R I A L S Volume 60 2015 Issue 2 DOI: 10.1515/amm-2015-0086 S.M. SHIN, G.J. JUNG, WOO-JIN LEE, C.Y. KANG, J.P. WANG, RECOVERY OF ELECTRODIC POWDER
Experiment 13: Determination of Molecular Weight by Freezing Point Depression
 1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
A NOVEL METHOD FOR THE IMPROVEMENT IN THERMOELECTRIC PROPERTY OF TIN OXIDE THIN FILMS AND ITS APPLICATION IN GAS SENSING
 INTERNATIONAL JOURNAL ON SMART SENSING AND INTELLIGENT SYSTEMS, VOL. 1, NO. 2, JUNE 2008 A NOVEL METHOD FOR THE IMPROVEMENT IN THERMOELECTRIC PROPERTY OF TIN OXIDE THIN FILMS AND ITS APPLICATION IN GAS
INTERNATIONAL JOURNAL ON SMART SENSING AND INTELLIGENT SYSTEMS, VOL. 1, NO. 2, JUNE 2008 A NOVEL METHOD FOR THE IMPROVEMENT IN THERMOELECTRIC PROPERTY OF TIN OXIDE THIN FILMS AND ITS APPLICATION IN GAS
Tailor Made Carbon and Graphite Based Anode Materials for Lithium Ion Batteries. Heribert Walter, Battery+Storage 2013
 Tailor Made Carbon and Graphite Based Anode Materials for Lithium Ion Batteries Heribert Walter, Battery+Storage 2013 Agenda SGL Group at a Glance Anode Materials Overview Material Synthesis and Modification
Tailor Made Carbon and Graphite Based Anode Materials for Lithium Ion Batteries Heribert Walter, Battery+Storage 2013 Agenda SGL Group at a Glance Anode Materials Overview Material Synthesis and Modification
Solef. Solef PVDF Aqueous Dispersions. for Lithium Batteries
 Solef Solef PVDF Aqueous Dispersions for Lithium Batteries Innovative Polymerization Technology Solef PVDF is a partially fluorinated, semi-crystalline polymer with excellent thermo-mechanical and chemical
Solef Solef PVDF Aqueous Dispersions for Lithium Batteries Innovative Polymerization Technology Solef PVDF is a partially fluorinated, semi-crystalline polymer with excellent thermo-mechanical and chemical
ADVANCED AP PLACEMENT CHEMISTRY. Activity Series. Introduction. Objective. Chemicals and Equipment
 ADVANCED AP PLACEMENT CHEMISTRY Introduction Activity Series An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal
ADVANCED AP PLACEMENT CHEMISTRY Introduction Activity Series An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal
Supplementary Material for Chitin and chitosan dissolving. in ionic liquids as reversible sorbents of CO 2
 Supplementary Material for Chitin and chitosan dissolving in ionic liquids as reversible sorbents of CO 2 Haibo Xie a, Suobo Zhang* a, Shenghai Li b a State Key Laboratory of Polymer Physics and Chemistry,
Supplementary Material for Chitin and chitosan dissolving in ionic liquids as reversible sorbents of CO 2 Haibo Xie a, Suobo Zhang* a, Shenghai Li b a State Key Laboratory of Polymer Physics and Chemistry,
Supporting Information for Fluorinated Ethylene Carbonate as Electrolyte Additive for Rechargeable Na Batteries
 Supporting Information for Fluorinated Ethylene Carbonate as Electrolyte Additive for Rechargeable Na Batteries Shinichi Komaba,* a Toru Ishikawa, a Naoaki Yabuuchi, a Wataru Murata, a Atsushi Ito, b and
Supporting Information for Fluorinated Ethylene Carbonate as Electrolyte Additive for Rechargeable Na Batteries Shinichi Komaba,* a Toru Ishikawa, a Naoaki Yabuuchi, a Wataru Murata, a Atsushi Ito, b and
PHYSICAL PROPERTIES OF La 0.9 Sr 0.1 Cr 1-X Ni X O 3-δ (X = 0-0.6) SYNTHESIZED VIA CITRATE GEL COMBUSTION
 PHYSICAL PROPERTIES OF La 0.9 Sr 0.1 Cr 1-X Ni X O 3-δ (X = 0-0.6) SYNTHESIZED VIA CITRATE GEL COMBUSTION Anuchit Ruangvittayanon * and Sutin Kuharuangrong Received: Sept 29, 2009; Revised: Nov 17, 2009;
PHYSICAL PROPERTIES OF La 0.9 Sr 0.1 Cr 1-X Ni X O 3-δ (X = 0-0.6) SYNTHESIZED VIA CITRATE GEL COMBUSTION Anuchit Ruangvittayanon * and Sutin Kuharuangrong Received: Sept 29, 2009; Revised: Nov 17, 2009;
The electrodeposition of Zn-Mo and Zn-Sn-Mo alloys from citrate electrolytes
 Honorata Kazimierczak The electrodeposition of Zn-Mo and Zn-Sn-Mo alloys from citrate electrolytes Supervisor: Assoc. Prof. Piotr Ozga The electrodeposition of Zn-Mo and Zn-Sn-Mo alloys from citrate electrolytes
Honorata Kazimierczak The electrodeposition of Zn-Mo and Zn-Sn-Mo alloys from citrate electrolytes Supervisor: Assoc. Prof. Piotr Ozga The electrodeposition of Zn-Mo and Zn-Sn-Mo alloys from citrate electrolytes
Experiment 3 * Thermochromism in the Ionic Conductor, Cu 2 HgI 4
 Experiment 3 * Thermochromism in the Ionic Conductor, Cu 2 HgI 4 *This lab taken from Teaching General Chemistry; A Materials Science Companion, Eds. Ellis, A.B.; Geselbracht, M.J.; Johnson, B.J.; Lisensky,
Experiment 3 * Thermochromism in the Ionic Conductor, Cu 2 HgI 4 *This lab taken from Teaching General Chemistry; A Materials Science Companion, Eds. Ellis, A.B.; Geselbracht, M.J.; Johnson, B.J.; Lisensky,
ELECTROCHEMICAL BEHAVIOURS OF LANTHANIDE FLUORIDES IN THE ELECTROLYSIS SYSTEM WITH LiF-NaF-KF SALT
 ELECTROCHEMICAL BEHAVIOURS OF LANTHANIDE FLUORIDES IN THE ELECTROLYSIS SYSTEM WITH LiF-NaF-KF SALT Joon-Bo Shim, Sung-Chan Hwang, Eung-Ho Kim, Young-Ho Kang, Byung-Jik Lee and Jae-Hyung Yoo Korea Atomic
ELECTROCHEMICAL BEHAVIOURS OF LANTHANIDE FLUORIDES IN THE ELECTROLYSIS SYSTEM WITH LiF-NaF-KF SALT Joon-Bo Shim, Sung-Chan Hwang, Eung-Ho Kim, Young-Ho Kang, Byung-Jik Lee and Jae-Hyung Yoo Korea Atomic
Natural Graphite versus Synthetic, Silicon and Others in Lithium Ion Battery Anodes
 Natural Graphite versus Synthetic, Silicon and Others in Lithium Ion Battery Anodes George C Hawley President George C Hawley & Associates Supermin123@hotmail.ca Biography George C. Hawley & Associates
Natural Graphite versus Synthetic, Silicon and Others in Lithium Ion Battery Anodes George C Hawley President George C Hawley & Associates Supermin123@hotmail.ca Biography George C. Hawley & Associates
Supporting Information. Oxidation State of Cross-over Manganese Species on the Graphite Electrode of Lithium-ion Cells
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is The Royal Society of Chemistry 2014 Supporting Information Oxidation State of Cross-over Manganese Species
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is The Royal Society of Chemistry 2014 Supporting Information Oxidation State of Cross-over Manganese Species
Lower Cost Higher Performance Graphite for LIBs. Prepared by: Dr. Edward R. Buiel President and CEO Coulometrics, LLC. Date: March 23, 2017
 Lower Cost Higher Performance Graphite for LIBs Prepared by: Dr. Edward R. Buiel President and CEO Coulometrics, LLC. Date: March 23, 2017 Outline Company overview Review of natural graphite resources
Lower Cost Higher Performance Graphite for LIBs Prepared by: Dr. Edward R. Buiel President and CEO Coulometrics, LLC. Date: March 23, 2017 Outline Company overview Review of natural graphite resources
Supporting Information. Low temperature synthesis of silicon carbide nanomaterials using
 Supporting Information Low temperature synthesis of silicon carbide nanomaterials using solid-state method Mita Dasog, Larissa F. Smith, Tapas K. Purkait and Jonathan G. C. Veinot * Department of Chemistry,
Supporting Information Low temperature synthesis of silicon carbide nanomaterials using solid-state method Mita Dasog, Larissa F. Smith, Tapas K. Purkait and Jonathan G. C. Veinot * Department of Chemistry,
for New Energy Materials and Devices; Beijing National Laboratory for Condense Matter Physics,
 Electronic Supplementary Information Highly efficient core shell CuInS 2 /Mn doped CdS quantum dots sensitized solar cells Jianheng Luo, a Huiyun Wei, a Qingli Huang, a Xing Hu, a Haofei Zhao, b Richeng
Electronic Supplementary Information Highly efficient core shell CuInS 2 /Mn doped CdS quantum dots sensitized solar cells Jianheng Luo, a Huiyun Wei, a Qingli Huang, a Xing Hu, a Haofei Zhao, b Richeng
Supporting Information for. Nitric Oxide Reactivity of Copper(II) Complexes of Bidentate Amine Ligands: Effect of. Substitution on Ligand Nitrosation
 1 Supporting Information for Nitric Oxide Reactivity of Copper(II) Complexes of Bidentate Amine Ligands: Effect of Substitution on Ligand Nitrosation Moushumi Sarma and Biplab Mondal Department of Chemistry,
1 Supporting Information for Nitric Oxide Reactivity of Copper(II) Complexes of Bidentate Amine Ligands: Effect of Substitution on Ligand Nitrosation Moushumi Sarma and Biplab Mondal Department of Chemistry,
Multi-step and multi-component organometallic synthesis in one pot using orthogonal mechanochemical reactions
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2014 Supporting Information Multi-step and multi-component organometallic synthesis in one pot
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2014 Supporting Information Multi-step and multi-component organometallic synthesis in one pot
Supporting Information. Electrochemical Formation of a p-n Junction on Thin Film Silicon Deposited in Molten Salt
 Supporting Information Electrochemical Formation of a p-n Junction on Thin Film Silicon Deposited in Molten Salt Xingli Zou, Li Ji, Xiao Yang, Taeho Lim, Edward T. Yu, and Allen J. Bard Experimental section
Supporting Information Electrochemical Formation of a p-n Junction on Thin Film Silicon Deposited in Molten Salt Xingli Zou, Li Ji, Xiao Yang, Taeho Lim, Edward T. Yu, and Allen J. Bard Experimental section
ACCELERATED CORROSION TESTING OF GALVANIC COUPLES. James F. Dante, Josh Averett, Fritz Friedersdorf, and Christy Vestal
 ACCELERATED CORROSION TESTING OF GALVANIC COUPLES James F. Dante, Josh Averett, Fritz Friedersdorf, and Christy Vestal Luna Innovations 706 Forest St. Suite A Charlottesville, VA 22903 dantej@lunainnovations.com
ACCELERATED CORROSION TESTING OF GALVANIC COUPLES James F. Dante, Josh Averett, Fritz Friedersdorf, and Christy Vestal Luna Innovations 706 Forest St. Suite A Charlottesville, VA 22903 dantej@lunainnovations.com
Synthesis and Evaluation of Electrocatalysts for Fuel Cells
 Synthesis and Evaluation of Electrocatalysts for Fuel Cells Jingguang Chen Center for Catalytic Science and Technology (CCST) Department of Chemical Engineering University of Delaware Newark, DE 19711
Synthesis and Evaluation of Electrocatalysts for Fuel Cells Jingguang Chen Center for Catalytic Science and Technology (CCST) Department of Chemical Engineering University of Delaware Newark, DE 19711
One-Pot Surface Engineering of Battery Electrode. Materials with Metallic SWCNT-Enriched, Ivy-
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information One-Pot Surface Engineering of Battery
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information One-Pot Surface Engineering of Battery
Hydrothermal synthesis of nano-silicon from a silica sol and its use in lithium ion batteries
 Electronic Supplementary Material Hydrothermal synthesis of nano-silicon from a silica sol and its use in lithium ion batteries Jianwen Liang, Xiaona Li, Yongchun Zhu ( ), Cong Guo, and Yitai Qian ( )
Electronic Supplementary Material Hydrothermal synthesis of nano-silicon from a silica sol and its use in lithium ion batteries Jianwen Liang, Xiaona Li, Yongchun Zhu ( ), Cong Guo, and Yitai Qian ( )
Effects of Lead on Tin Whisker Elimination
 Effects of Lead on Tin Whisker Elimination Wan Zhang and Felix Schwager Rohm and Haas Electronic Materials Lucerne, Switzerland inemi Tin Whisker Workshop at ECTC 0 May 30, 2006, in San Diego, CA Efforts
Effects of Lead on Tin Whisker Elimination Wan Zhang and Felix Schwager Rohm and Haas Electronic Materials Lucerne, Switzerland inemi Tin Whisker Workshop at ECTC 0 May 30, 2006, in San Diego, CA Efforts
