Supplemental Information. Spatial and Temporal Regulation of Receptor. Endocytosis in Neuronal Dendrites Revealed
|
|
|
- Jeffrey Hubbard
- 6 years ago
- Views:
Transcription
1 Cell Reports, Volume 18 Supplemental Information Spatial and Temporal Regulation of Receptor Endocytosis in Neuronal Dendrites Revealed by Imaging of Single Vesicle Formation Morgane Rosendale, Damien Jullié, Daniel Choquet, and David Perrais
2 Figure S1 related to Figure 1: The ph 5.5 solution does not affect TfR endocytosis in neurons A, Example of an endocytic event detected with the pph assay in a neuron (DIV13) transfected with TfR-SEP. Labelled transferrin (Tfn-A568, 5 µg/ml) is co-applied in both ph 5.5 and ph 7.4 perfusion channels. Note the colocalization of TfR-SEP at ph 7.4 and the Tfn-A568 signal. B, Average fluorescence of scission events in the green (dark green ph 7.4, light green ph 5.5) and red channels, aligned to the time of vesicle detection (n = 1169 events in 8 cells. Gray area indicates 95% confidence intervals for significant enrichment. C, Top: images of neurons after 10 minutes of Tfn-A568 application without (left) or with (right) the pph assay. Images taken with wide field epifluorescence microscopy with the same illumination and camera settings and displayed with the same contrast. Bottom, average Tfn-A568 in the soma and proximal dendrites of neurons without (n = 10) or with (n = 9) the pph assay. The average fluorescence values were not significantly different (p = 0.22). D, Kinetics of Tfn-A568 fluorescence accumulation in cells submitted (grey curve, n = 9 cells) or not (black curve, n = 10 cells) to the pph assay as described in C. Normalization done at 1 min after the start of the recording. E, Example trace of current amplitudes evoked by sequential application of acidic solution during the pph assay, recorded in a neurons voltage clamped at -70 mv. After 2 minutes, amiloride (500 µm) was added to the two solutions (ph 7.4 and 5.5) of the application pipette. Inset: currents evoked by acidic solution at time points indicated in the main figure. Note that the fast decrease in current amplitude observed at the beginning of the recording and the partial recovery after washout of amiloride can be explained by a desensitization of ASICs. F, Normalized current amplitude measured at the peak evoked by the pph assay before, during and after application of amiloride (n = 6 cells). G, Normalized cumulative frequency of events detected with the pph assay before and during application of amiloride. H, The average frequency of detected endocytic events is the same before (693 events) and during (n = 673 events) application of amiloride (p = 0.81).
3 Figure S2 related to Figure 1: Internalization of TfR-SEP monitored with Trypan Purple A, Consecutive images taken at 100 ms intervals of a neuron transfected with TfR-SEP during the application and washout of Trypan Purple (5 mm). Note the rapid quenching of most of the extracellular fluorescence, with some dot-like structures still visible B, Example voltage recordings of a neuron (15 DIV) before and during the application of Trypan. Responses to injection of -100, 0 and +100 pa currents for 200 ms show that the membrane resistance and neuronal excitability are not changed by the application of Trypan. In 10 neurons recorded, the membrane resistances calculated from current injections was 309 ± 43 and 313 ± 47 MΩ before and after Trypan, respectively, and were not significantly different (p = 0.82). The numbers of evoked action potentials were 3.8 ± 0.5 and 3.9 ± 0.5 before and after Trypan, respectively, and were not significantly different (p = 0.76). C, Example of an event detected with the ptry protocol at time 0 in a neuron transfected with TfR- SEP. D, Average fluorescence, aligned to the time of vesicle detection for 1022 events in 8 neurons of fluorescence without (top, HBS) and with 5 mm Trypan (bottom). Grey area indicates 95% confidence intervals for significant enrichment. E, Average neuron fluorescence outside of low ph or Trypan application during the pph (black, n = 7) or the ptry (purple, n = 5) protocols. F, cumulative event frequency normalized to 100 at 3 minutes. Note the progressive decrease in event frequency with the ptry protocol, which goes down to 68 ± 6 % of initial frequency in the final 6-9 minutes of recording, whereas the frequency remains stable for the pph assay (108 ±17 % of initial frequency).
4 Figure S3 related to Figure 1: Monitoring stimulated endocytosis of SEP-β2AR. A, Portion of a dendrite transfected with SEP-β2AR and Clc-mCh before (top) and during (bottom) application of 10 µm isoproterenol. The SEP-β2AR fluorescence increased 1.8 ± 0.2-folds at 1263 CCSs in 5 neurons. B, Example event detected at time 0 during isoproterenol application. Note the formation of a cluster visible 2 min before vesicle formation while the clathrin cluster does not change. C, Normalized cumulative frequency of events (552 events in 5 cells). D, Average fluorescence in the green channel at the indicated ph, aligned to the time of vesicle detection, before (left, 52 events) and during isoproterenol application (right, 352 events). The fluorescence at ph 5.5 at the time of vesicle formation reflects receptor loading in vesicles. It does not vary significantly during agonist application (626 ± 77 a.u. during baseline; 786 ± 33 a.u. in the presence of isoproterenol; p = 0.41 Kolmogorov-Smirnov test).
5 Figure S4 related to Figure 2: AMPARs are internalized at CCSs. A, Example event detected at time 0 in a neuron cotransfected with SEP-GluA1 and Clc-mCherry. On the right is the merged image of SEP-GluA1 at ph 7.4 and Clc (averages of 10 frames around the event). White cross indicates the location of the event. B, Average fluorescence of SEP-GluA1 at the indicated ph, and of Clc-mCherry, aligned to the time of vesicle detection (53 events in 5 cells). Gray areas represent the 95% confidence interval of randomized data indicating clathrin enrichment at the site of vesicle formation.. C,D, Same as A and B for neurons transfected with SEP-GluA2, GluA1 and Clc-mCherry. Averages of 266 events recorded in 16 cells.
6 Figure S5 related to Figure 4: Internalization of SEP-GluA2 visualized with the pulsed Trypan protocol is stimulated by NMDA application. A, Currents evoked by application of 20 µm NMDA and 10 µm glycine in a solution containing 0.3 mm Mg 2+ (chemltd stimulation), at ph 7.4 for 2 s then at ph 5.5 and back to ph 7.4. Note the complete reversal of inhibition. The neuron was voltage clamped at -60 mv. B, Same as A with the application of Trypan (5 mm) instead of low ph. The inhibition by Trypan is completely reversible as well. C, Inhibition of NMDA currents measured just before and at the end of quencher application by ph 5.5 solution and Trypan. These inhibitions are not significantly different (p = 0.68). D, Illustration of the paradigm used to visualize the formation of endocytic vesicles by ptry before, during and after stimulation of neurons with the cltd protocol. Bleaching was performed at ph 5.5 for neurons expressing SEP-GluA2 and AP-GluA1. See materials and methods for details. E, Two GluA2 example events. The first event, detected at time 0, occurs in the dendritic shaft. 44 seconds later, a second event occurs in a spine neck. F, Average ratio of event frequency normalized to baseline (n = 5 cells). G, Normalized cumulative frequency of endocytic events during NMDA application. The Y-axis origin starts at 100, which corresponds to the normalized cumulative frequency in the control period. H, Average fluorescence of dendritic regions of the cells used in Figure 4 at ph 7.4 (grey) and ph 5.5 (green). I, Average fluorescence of dendritic regions of the cells used in Figure S5D-G in HBS (grey) and HBS + Trypan (purple).
7 Supplemental Experimental procedures Primary neuronal cultures and transfection Neurons were prepared and cultured following the Banker protocol (Kaech and Banker, 2006). In brief, dissociated hippocampal neurons from embryonic day 18 rat embryos were plated on 18 mm poly-d-lysinecoated glass coverslips at a density of cells/ml in MEM containing 10 % horse serum (Invitrogen) for 3 h, then cultured in Neurobasal medium supplemented with 2 mm glutamine and 10 % Neuromix (PAA), B27 (Gibco) or SM1 supplement (Stem Cell) on a feeder layer of glial cells at 37 C in 5 % CO2 for 13 to 21 days. TfR-SEP, SEP-β2AR, SEP-GluA1, Clc-mCh and Homer1c-RFP (tandem dimer Tomato) plasmids have been described previously (Petrini et al., 2009; Letellier et al., 2014; Merrifield et al., 2005; Taylor et al., 2011; Jullie et al., 2014). SEP-GluA2 was obtained by inserting the SEP sequence after the signal peptide of rat GluA2 coding sequence using AgeI/NheI restriction sites. This SEP-GluA2 insert was then cloned into the pbi-tet-on vector (Clontech) by MluI and EcoRI. AP-GluA1 was generated by introducing a biotin ligase acceptor peptide (AP) tag (GGCCTGAACGACATCTTCGAGGCCCAGAAGATCGAGTGGCACGAG) on the N-terminal part of rat GluA1 after the signal peptide. The AP-GluA1 insert was cloned in a psynaav vector (expression under the human Synapsin promoter) between EcoRI/HindIII restriction sites. Neurons were transfected at 7-10 DIV except when expressing Dyn1-mCh (transfected at DIV 13, i.e. 24h before the experiment to prevent mcherry induced clusterization). Transfection was performed using Effectene (Qiagen) following the company protocol except for SEP-GluA2 expressing neurons used for chemltd experiments under the pph assay which were transfected using a calcium phosphate technique (Wienisch and Klingauf, 2006). Briefly, the culture medium was removed and replaced with Fresh, unsupplemented Neurobasal medium min prior to transfection. The calcium phosphate (125 mm final concentration)/dna precipitate was formed in BES buffered saline (in mm: 50 BES, 280 NaCl, 1,5 NA 2HPO 4.2H 2O; ph 7.07) for min. The precipitate was added dropwise to the cells. Following a min transfection, during which a fine sandy precipitate covered the cells, the cultures were washed in HBSS and returned to the original culture medium. These neurons were systematically cotransfected with a tetracycline transactivator plasmid (Clontech) to allow for the inducible expression of the protein by doxycycline (2 µg/ml 48 hr prior to an imaging session) as well as with AP-GluA1 to favor the heterodimerization and membrane export of the overexpressed AMPAR subunits. Fluorescence Imaging Imaging was performed at 35 C on a TIRF microscope previously described (Shen et al., 2014) on neurons at DIV (Figures 1, S1, S2 and S3) or at DIV (Figures 2, 3, 4 and S4). Cells were perfused with HEPES buffered saline solution (HBS) with, in mm: 120 NaCl, 2 KCl, 2 MgCl 2, 2 CaCl 2, 5 D-glucose and 10 HEPES, and adjusted to ph 7.4 and mosm. For the pph assay, MES buffered saline solution (MBS) was prepared similarly by replacing HEPES with MES and adjusting the ph to 5.5. (Salts from Sigma Aldrich). HBS and MBS were perfused locally around the recorded cell using a 2-way borosilicate glass pipette. Local perfusion solutions were supplemented with (in µm) 3 Na 2-Ascorbate, 500 amiloride (when applicable), 0.5 TTX and 2% v/v Neuromix (PAA laboratories) or B27 (Gibco) on the day of the experiment. For the ptry protocol, 5 mm Trypan purple (Interchim) were diluted from a 0.1 M stock solution in HBS. After Trypan dilution, the osmolarity was not changed and the ph was 7.2. Chemical LTD was induced by using HBS/MBS solutions containing 0.3 mm MgCl 2 (instead of 2) supplemented with 20 µm NMDA (Abcam) and 10 µm Glycine (Sigma). For control experiments, D-APV (50µM) was added to all local perfusion solutions (baseline, stimulation and washout) as well as in the main bath HBS perfusion. The fluorescence of neurons transfected with SEP-GluA2 and unlabelled-glua1 was distributed throughout the neuronal membrane, including spines and PSDs labelled with Homer1c-RFP. However, when
8 Trypan or MBS was applied to these neurons, the fluorescence was only partially quenched, especially in the dendritic shaft consistent with the presence of a non-acidic intracellular compartment (also see (Rathje et al., 2013)). This residual background fluorescence added to the less complete quenching of surface receptors by Trypan than by ph 5.5 solution prevented the efficient detection of endocytic events. To reduce this artefact, data from Figure S4 were acquired after bleaching intracellular receptors of whole dendrites as in Petrini et al. (2009) with a FRAP module (ILas system, Roper Scientific). During this step, neurons were bathed in ph 5.5 solution in the presence of amiloride to render SEP-tagged receptors located on the plasma membrane nonfluorescent, thus sparing them from bleaching. Neurons were then allowed to recover in normal solution for 10 minutes. Patch-clamp recordings Recording pipettes (resistance, 3-5 MΩ) were filled with a solution containing the following (in mm): 110 KCH 3SO 3, 2 KCl, 1 EGTA, 0.1 CaCl 2, 10 HEPES, 4 NaATP, 0.4 NaGTP and 5 Na 2Phosphocreatine, adjusted to ph 7.2 with KOH ( mosm). Cells were recorded on the same microscope and using the same HBS solution as for fluorescence imaging. They were voltage clamped at -60 mv or kept in current clamp mode around -60 mv with an EPC10 patch-clamp amplifier (HEKA). See figure legends (Figures S1 and S3) for more details. Image analysis Semi-automatic detection of endocytic events was performed as described previously for cell lines (Shen et al., 2014; Taylor et al., 2011b). In short, a sudden, punctate, fluorescence increase appearing in ph 5.5 images was detected as being an endocytic event if 1) it was visible for more than 3 frames (i.e. 8 seconds), and 2) it appeared at the same location as a pre-existing fluorescence cluster detectable in ph 7.4 images. Candidate events were then validated by visual inspection in a random order to avoid any bias during cell stimulation. In neurons, the proportion of automatically detected events validated by visual inspection (57.1 ± 10.6 % for TfR- SEP events in this study) was smaller than for cell lines (~80 %, Taylor et al. 2011, Shen et al. 2014). Indeed, we saw a greater contribution of moving intracellular vesicles presumably because transport tracks in small dendrites are closer to the membrane than in larger cell bodies, and vesicles travelling along them become visible within the field of TIRF illumination. In addition, we saw a sizeable amount of exocytic events (characterized by the sudden appearance of fluorescence at ph 7.4), followed by the appearance of the same organelle at ph 5.5. We have characterized this type of kiss-and-run event previously (Jullié et al., 2014). They are independent of clathrin and dynamin, thus independent of clathrin mediated endocytosis, and were not included in this study. Event frequency was expressed per cell surface area measured on the cell mask. The cell mask was determined by the reunion of the thresholded image of SEP tagged receptors at ph 7.4 and the thresholded image of synapses (i.e. Homer1c-tdTom) when applicable. Determination of the number of events per CCS (Figure 1H and related text) was performed using Clc-mCherry images acquired at ph 5.5 throughout each pph recording to reduce the contribution of fluorescence emanating from TfR-SEP surface clusters due to spectral bleed-through from the green to the red channel. These images were averaged into a single image for segmentation. An endocytic event was considered as originating from a CCS if it was detected less than 750 nm away from its center. Cumulative distance plots were obtained by measuring the Euclidian distance between the center of an object A (a CCS, a PSD or an event) and the closest pixel identified in segmented images as belonging to an object B (a PSD or a CCS). Randomized datasets were obtained by generating 1000 cumulative plots of distances for n A randomly scattered objects within the cell mask, where n A is the number of objects A detected. The 95 % upper and lower percentiles of this randomized dataset are represented in grey, and the median as a black line. Parts of the measured cumulative distance plots outside of the gray area of the randomized dataset were considered a specific enrichment, i.e. close to PSDs for GluA1 and GluA2 events (Fig 2F,I; Fig 3F; Fig
9 4C,D) and for CCSs (Fig 2L). Histogram representations associated to these graphs were obtained by subtracting the median of the randomized distance plots to the distance plot of actually detected objects. Excess of objects A occurring closer than chance to an object B was quantified as the sum of the first positive 100 nm bins of this histogram (i.e nm distances). We further assessed significant differences between the two distributions with the non-parametric Kolmogorov-Smirnov (KS) test with a significance level of Fluorescence quantification of events (Figures 1C, 1F, 2D, 4E, S1B, S2D, S3D, S4B,D) was performed as in (Shen et al., 2014). In short, each value is calculated as the mean intensity in a 2-pixel radius circle centered on the detection to which the local background intensity is subtracted (the local background is taken as the 20 th to 80 th percentile of fluorescence in an annulus of 5 to 2 pixel outer and inner radii centered on the detection). The grey area indicates represented on the graphs are a display of the 95% confidence intervals for significant enrichment obtained from randomized fluorescence data. Overall cellular fluorescence intensity (Figure S1C,D, S2E) was measured in a mask determined for each cell by thresholding in images taken in HBS (ph 7.4, no Trypan) during 10 min of pph or pulsed-trypan recordings. Neurons transfected with fluorescent AMPAR subunits (SEP-GluA1 or SEP-GluA2) sometimes displayed very low frequencies of detectable events, yielding in many cases only a few validated events per recording, and often no events in the control period of 3 minutes, precluding a reliable measure of frequency and its modulation by stimulation. This could result from poor signal to noise images due to the fact that many AMPA receptors are located in weakly acidic intracellular organelles, making their SEP tag visible even during extracellular application of ph 5.5 buffer. Therefore, we kept for analysis only those cells where more than 15 events were automatically detected within 8-10 minute recordings. The final dataset was as follows: for cells cotransfected with Homer1c-tdTom, we kept 8 SEP-GluA1 transfected cells with 16 to 187 automatic detections (yielding 6 to 55 validated events) out of 14 recorded cells; we kept 14 SEP-GluA2 transfected cells with 62 to 268 automatic detections (yielding 10 to 39 validated events) out of 58 recorded cells. For cells co-transfected with clc-mcherry, we kept 4 SEP-GluA1 transfected cells with 31 to 54 automatic detections (yielding 10 to 14 validated events) out of 13 recorded cells; we kept 12 SEP-GluA2 transfected cells with 21 to 160 automatic detections (yielding 8 to 53 validated events) out of 24 recorded cells. On the other hand all recorded cells transfected with TfR-SEP or SEP-β2AR could be analyzed, most probably because their intracellular fluorescence was very weak, consistent with the fact that these receptors are mostly found in acidic recycling endosomes rather than in the more neutral endoplasmic reticulum. Supplemental References Kaech, S., and Banker, G. (2006). Culturing hippocampal neurons. Nat. Protoc. 1, Letellier, M., Elramah, S., Mondin, M., Soula, A., Penn, A., Choquet, D., Landry, M., Thoumine, O., and Favereaux, A. (2014). mir-92a regulates expression of synaptic GluA1-containing AMPA receptors during homeostatic scaling. Nat. Neurosci. 17, Wienisch, M., and Klingauf, J. (2006). Vesicular proteins exocytosed and subsequently retrieved by compensatory endocytosis are nonidentical. Nat. Neurosci. 9,
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Moutin et al., http://www.jcb.org/cgi/content/full/jcb.201110101/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Tagged Homer1a and Homer are functional and display different
Supplemental material Moutin et al., http://www.jcb.org/cgi/content/full/jcb.201110101/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Tagged Homer1a and Homer are functional and display different
Stargazin regulates AMPA receptor trafficking through adaptor protein. complexes during long term depression
 Supplementary Information Stargazin regulates AMPA receptor trafficking through adaptor protein complexes during long term depression Shinji Matsuda, Wataru Kakegawa, Timotheus Budisantoso, Toshihiro Nomura,
Supplementary Information Stargazin regulates AMPA receptor trafficking through adaptor protein complexes during long term depression Shinji Matsuda, Wataru Kakegawa, Timotheus Budisantoso, Toshihiro Nomura,
Supplemental figures (1-9) Gu et al. ADF/Cofilin-Mediated Actin Dynamics Regulate AMPA Receptor Trafficking during Synaptic Plasticity
 Supplemental figures (1-9) Gu et al. ADF/Cofilin-Mediated Actin Dynamics Regulate AMPA Receptor Trafficking during Synaptic Plasticity Supplemental figure 1. The quantification method to determine the
Supplemental figures (1-9) Gu et al. ADF/Cofilin-Mediated Actin Dynamics Regulate AMPA Receptor Trafficking during Synaptic Plasticity Supplemental figure 1. The quantification method to determine the
Supplementary Figure 1. Serial deletion mutants of BLITz.
 Supplementary Figure 1 Serial deletion mutants of BLITz. (a) Design of TEVseq insertion into J -helix. C-terminal end of J -helix was serially deleted and replaced by TEVseq. TEV cleavage site is labeled
Supplementary Figure 1 Serial deletion mutants of BLITz. (a) Design of TEVseq insertion into J -helix. C-terminal end of J -helix was serially deleted and replaced by TEVseq. TEV cleavage site is labeled
15 June 2011 Supplementary material Bagriantsev et al.
 Supplementary Figure S1 Characterization of K 2P 2.1 (TREK-1) GOF mutants A, Distribution of the positions of mutated nucleotides, represented by a red x, from a pool of 18 unselected K 2P 2.1 (KCNK2)
Supplementary Figure S1 Characterization of K 2P 2.1 (TREK-1) GOF mutants A, Distribution of the positions of mutated nucleotides, represented by a red x, from a pool of 18 unselected K 2P 2.1 (KCNK2)
CHO-Tet herg Cell Line
 F B SYS GmbH CHO-Tet herg Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO-Tet herg Cells Page 2 TABLE OF CONTENTS 1 BACKGROUND...3 1.1 Drug-induced QT Prolongation...3 1.2 Regulatory Issues...3
F B SYS GmbH CHO-Tet herg Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO-Tet herg Cells Page 2 TABLE OF CONTENTS 1 BACKGROUND...3 1.1 Drug-induced QT Prolongation...3 1.2 Regulatory Issues...3
Assembly of synapses by neuronal adhesion molecules: single molecule studies
 Assembly of synapses by neuronal adhesion molecules: single molecule studies Olivier Thoumine Interdisciplinary Institute of Neurosciences CNRS - University of Bordeaux Connectivity in the brain 300 nm
Assembly of synapses by neuronal adhesion molecules: single molecule studies Olivier Thoumine Interdisciplinary Institute of Neurosciences CNRS - University of Bordeaux Connectivity in the brain 300 nm
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10016 Supplementary discussion on binding site density for protein complexes on the surface: The density of biotin sites on the chip is ~10 3 biotin-peg per µm 2. The biotin sites are
doi:10.1038/nature10016 Supplementary discussion on binding site density for protein complexes on the surface: The density of biotin sites on the chip is ~10 3 biotin-peg per µm 2. The biotin sites are
CHO K V 4.3 Cell Line
 B SYS GmbH CHO K V 4.3 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO K V 4.3 Cells Page 2 TABLE OF CONTENTS 1. PRODUCT SHIPMENT...3 1.1. Product Format...3 1.2. Mycoplasma Certificate...3 1.3.
B SYS GmbH CHO K V 4.3 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO K V 4.3 Cells Page 2 TABLE OF CONTENTS 1. PRODUCT SHIPMENT...3 1.1. Product Format...3 1.2. Mycoplasma Certificate...3 1.3.
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 PCR-genotyping of the three mouse models used in this study and controls for behavioral experiments after semi-chronic Pten inhibition. a-c. DNA from App/Psen1 (a), Pten tg (b) and
Supplementary Figure 1 PCR-genotyping of the three mouse models used in this study and controls for behavioral experiments after semi-chronic Pten inhibition. a-c. DNA from App/Psen1 (a), Pten tg (b) and
CHO herg-duo Cell Line
 B SYS GmbH CHO herg-duo Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO herg DUO Cells Page 2 TABLE OF CONTENTS 1. BACKGROUND...3 1.1. DRUG-INDUCED QT PROLONGATION...3 1.2. REGULATORY ISSUES...3
B SYS GmbH CHO herg-duo Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO herg DUO Cells Page 2 TABLE OF CONTENTS 1. BACKGROUND...3 1.1. DRUG-INDUCED QT PROLONGATION...3 1.2. REGULATORY ISSUES...3
FluoVolt Membrane Potential Kit
 FluoVolt Membrane Potential Kit Catalog no. F10488 Table 1 Contents and storage Material Amount Storage Stability FluoVolt, 1000X in DMSO (Component A) PowerLoad Concentrate, 100X (Component B) 50 μl 500
FluoVolt Membrane Potential Kit Catalog no. F10488 Table 1 Contents and storage Material Amount Storage Stability FluoVolt, 1000X in DMSO (Component A) PowerLoad Concentrate, 100X (Component B) 50 μl 500
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/325/5941/756/dc1 Supporting Online Material for Synaptic Integration in Tuft Dendrites of Layer 5 Pyramidal Neurons: A New Unifying Principle Matthew E. Larkum,* Thomas
www.sciencemag.org/cgi/content/full/325/5941/756/dc1 Supporting Online Material for Synaptic Integration in Tuft Dendrites of Layer 5 Pyramidal Neurons: A New Unifying Principle Matthew E. Larkum,* Thomas
CHO Na V 1.4 Cell Line
 B SYS GmbH CHO Na V 1.4 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO Na V 1.4 Cells Page 2 TABLE OF CONTENTS 1. B'SYS Na V 1.4 CELL LINE...2 1.1 THE VOLTAGE GATED SODIUM CHANNELS NA V 1.4...2
B SYS GmbH CHO Na V 1.4 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO Na V 1.4 Cells Page 2 TABLE OF CONTENTS 1. B'SYS Na V 1.4 CELL LINE...2 1.1 THE VOLTAGE GATED SODIUM CHANNELS NA V 1.4...2
Serine phosphorylation of ephrinb2 regulates trafficking of synaptic
 Serine phosphorylation of ephrinb2 regulates trafficking of synaptic AMPA receptors Clara L. Essmann, Elsa Martinez, Julia C. Geiger, Manuel Zimmer, Matthias H. Traut, Valentin Stein, Rüdiger Klein and
Serine phosphorylation of ephrinb2 regulates trafficking of synaptic AMPA receptors Clara L. Essmann, Elsa Martinez, Julia C. Geiger, Manuel Zimmer, Matthias H. Traut, Valentin Stein, Rüdiger Klein and
CHO-hERG DUO optimized for QPatch
 Application Report CHO-hERG DUO optimized for QPatch Functional validation and performance data for the CHO herg DUO cell line available from. This cell line is made in collaboration with B SYS GmbH (Switzerland).
Application Report CHO-hERG DUO optimized for QPatch Functional validation and performance data for the CHO herg DUO cell line available from. This cell line is made in collaboration with B SYS GmbH (Switzerland).
Cor.4U on QPatch Cells provided by AxioGenesis
 Application Report: Cor.4U on QPatch This report shows how cardiomyocytes from Axiogenesis can be used on the QPatch. Action potentials can be induced in a controlled and reproducible manner using a mix
Application Report: Cor.4U on QPatch This report shows how cardiomyocytes from Axiogenesis can be used on the QPatch. Action potentials can be induced in a controlled and reproducible manner using a mix
Supplementary information
 Supplementary information Supplementary Figure 1 (a) EM image of the pyramidal layer of wt mice. CA3 pyramidal neurons were selected according to their typical alignment, size and shape for subsequent
Supplementary information Supplementary Figure 1 (a) EM image of the pyramidal layer of wt mice. CA3 pyramidal neurons were selected according to their typical alignment, size and shape for subsequent
MeCP2. MeCP2/α-tubulin. GFP mir1-1 mir132
 Conservation Figure S1. Schematic showing 3 UTR (top; thick black line), mir132 MRE (arrow) and nucleotide sequence conservation (vertical black lines; http://genome.ucsc.edu). a GFP mir1-1 mir132 b GFP
Conservation Figure S1. Schematic showing 3 UTR (top; thick black line), mir132 MRE (arrow) and nucleotide sequence conservation (vertical black lines; http://genome.ucsc.edu). a GFP mir1-1 mir132 b GFP
Supplemental Table 1: Sequences of real time PCR primers. Primers were intronspanning
 Symbol Accession Number Sense-primer (5-3 ) Antisense-primer (5-3 ) T a C ACTB NM_001101.3 CCAGAGGCGTACAGGGATAG CCAACCGCGAGAAGATGA 57 HSD3B2 NM_000198.3 CTTGGACAAGGCCTTCAGAC TCAAGTACAGTCAGCTTGGTCCT 60
Symbol Accession Number Sense-primer (5-3 ) Antisense-primer (5-3 ) T a C ACTB NM_001101.3 CCAGAGGCGTACAGGGATAG CCAACCGCGAGAAGATGA 57 HSD3B2 NM_000198.3 CTTGGACAAGGCCTTCAGAC TCAAGTACAGTCAGCTTGGTCCT 60
Cytotoxicity of Botulinum Neurotoxins Reveals a Direct Role of
 Supplementary Information Cytotoxicity of Botulinum Neurotoxins Reveals a Direct Role of Syntaxin 1 and SNAP-25 in Neuron Survival Lisheng Peng, Huisheng Liu, Hongyu Ruan, William H. Tepp, William H. Stoothoff,
Supplementary Information Cytotoxicity of Botulinum Neurotoxins Reveals a Direct Role of Syntaxin 1 and SNAP-25 in Neuron Survival Lisheng Peng, Huisheng Liu, Hongyu Ruan, William H. Tepp, William H. Stoothoff,
Detection of Action Potentials In Vitro by Changes in Refractive Index
 Detection of Action Potentials In Vitro by Changes in Refractive Index David Kleinfeld 1 and Arthur LaPorta 2 1 - Department of Physics, University of California, La Jolla, CA 92093-0319. 2- Laboratory
Detection of Action Potentials In Vitro by Changes in Refractive Index David Kleinfeld 1 and Arthur LaPorta 2 1 - Department of Physics, University of California, La Jolla, CA 92093-0319. 2- Laboratory
Nature Neuroscience: doi: /nn Supplementary Figure 1
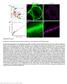 Supplementary Figure 1 Nanoscale localization precision and relative quantification of CB 1 receptors by 3D-STORM imaging (a) Schematic representation of the experimental paradigm for combined confocal/storm
Supplementary Figure 1 Nanoscale localization precision and relative quantification of CB 1 receptors by 3D-STORM imaging (a) Schematic representation of the experimental paradigm for combined confocal/storm
Supporting Information
 Supporting Information Stavru et al. 0.073/pnas.357840 SI Materials and Methods Immunofluorescence. For immunofluorescence, cells were fixed for 0 min in 4% (wt/vol) paraformaldehyde (Electron Microscopy
Supporting Information Stavru et al. 0.073/pnas.357840 SI Materials and Methods Immunofluorescence. For immunofluorescence, cells were fixed for 0 min in 4% (wt/vol) paraformaldehyde (Electron Microscopy
Neuronal Activity and CaMKII Regulate Kinesin-Mediated Transport of Synaptic AMPARs
 Neuron Supplemental Information Neuronal Activity and CaMKII Regulate Kinesin-Mediated Transport of Synaptic AMPARs Frédéric J. Hoerndli, Rui Wang, Jerry E. Mellem, Angy Kallarackal, Penelope J. Brockie,
Neuron Supplemental Information Neuronal Activity and CaMKII Regulate Kinesin-Mediated Transport of Synaptic AMPARs Frédéric J. Hoerndli, Rui Wang, Jerry E. Mellem, Angy Kallarackal, Penelope J. Brockie,
University of Bristol - Explore Bristol Research
 Lee, K., Kim, H., An, K., Kwon, O-B., Park, S., Cha, J. H.,... Kim, H-S. (2016). Replenishment of microrna-188-5p restores the synaptic and cognitive deficits in 5XFAD Mouse Model of Alzheimer s Disease.
Lee, K., Kim, H., An, K., Kwon, O-B., Park, S., Cha, J. H.,... Kim, H-S. (2016). Replenishment of microrna-188-5p restores the synaptic and cognitive deficits in 5XFAD Mouse Model of Alzheimer s Disease.
Long-term dynamics of CA1 hippocampal place codes
 Long-term dynamics of CA1 hippocampal place codes Yaniv Ziv, Laurie D. Burns, Eric D. Cocker, Elizabeth O. Hamel, Kunal K. Ghosh, Lacey J. Kitch, Abbas El Gamal, and Mark J. Schnitzer Supplementary Fig.
Long-term dynamics of CA1 hippocampal place codes Yaniv Ziv, Laurie D. Burns, Eric D. Cocker, Elizabeth O. Hamel, Kunal K. Ghosh, Lacey J. Kitch, Abbas El Gamal, and Mark J. Schnitzer Supplementary Fig.
Methanol fixation allows better visualization of Kal7. To compare methods for
 Supplementary Data Methanol fixation allows better visualization of Kal7. To compare methods for visualizing Kal7 in dendrites, mature cultures of dissociated hippocampal neurons (DIV21) were fixed with
Supplementary Data Methanol fixation allows better visualization of Kal7. To compare methods for visualizing Kal7 in dendrites, mature cultures of dissociated hippocampal neurons (DIV21) were fixed with
Masayoshi Honda, Jeehae Park, Robert A. Pugh, Taekjip Ha, and Maria Spies
 Molecular Cell, Volume 35 Supplemental Data Single-Molecule Analysis Reveals Differential Effect of ssdna-binding Proteins on DNA Translocation by XPD Helicase Masayoshi Honda, Jeehae Park, Robert A. Pugh,
Molecular Cell, Volume 35 Supplemental Data Single-Molecule Analysis Reveals Differential Effect of ssdna-binding Proteins on DNA Translocation by XPD Helicase Masayoshi Honda, Jeehae Park, Robert A. Pugh,
Fast, three-dimensional super-resolution imaging of live cells
 Nature Methods Fast, three-dimensional super-resolution imaging of live cells Sara A Jones, Sang-Hee Shim, Jiang He & Xiaowei Zhuang Supplementary Figure 1 Supplementary Figure 2 Supplementary Figure 3
Nature Methods Fast, three-dimensional super-resolution imaging of live cells Sara A Jones, Sang-Hee Shim, Jiang He & Xiaowei Zhuang Supplementary Figure 1 Supplementary Figure 2 Supplementary Figure 3
1.7 on Nanion's SyncroPatch 384PE. The electrophysiology team at Nanion Technologies GmbH, Munich, Germany. Cells were kindly provided by Anaxon.
 Channel: h 1.7 Cells: CHO Tools: SyncroPatch 384PE Characterization of h 1.7 on Nanion's SyncroPatch 384PE The electrophysiology team at, Munich, Germany. Cells were kindly provided by Anaxon. Summary
Channel: h 1.7 Cells: CHO Tools: SyncroPatch 384PE Characterization of h 1.7 on Nanion's SyncroPatch 384PE The electrophysiology team at, Munich, Germany. Cells were kindly provided by Anaxon. Summary
Figure legends for supplement
 Figure legends for supplement Supplemental Figure 1 Characterization of purified and recombinant proteins Relevant fractions related the final stage of the purification protocol(bingham et al., 1998; Toba
Figure legends for supplement Supplemental Figure 1 Characterization of purified and recombinant proteins Relevant fractions related the final stage of the purification protocol(bingham et al., 1998; Toba
Challenges to measuring intracellular Ca 2+ Calmodulin: nature s Ca 2+ sensor
 Calcium Signals in Biological Systems Lecture 3 (2/9/0) Measuring intracellular Ca 2+ signals II: Genetically encoded Ca 2+ sensors Henry M. Colecraft, Ph.D. Challenges to measuring intracellular Ca 2+
Calcium Signals in Biological Systems Lecture 3 (2/9/0) Measuring intracellular Ca 2+ signals II: Genetically encoded Ca 2+ sensors Henry M. Colecraft, Ph.D. Challenges to measuring intracellular Ca 2+
Amaxa Basic Neuron SCN Nucleofector Kit
 Amaxa Basic Neuron SCN Nucleofector Kit For Primary Mouse Hippocampal Neurons (Small-Cell-Number) Embryonic mouse hippocampal neurons transfected using the Nucleofector Technology A B Approximately 100.000
Amaxa Basic Neuron SCN Nucleofector Kit For Primary Mouse Hippocampal Neurons (Small-Cell-Number) Embryonic mouse hippocampal neurons transfected using the Nucleofector Technology A B Approximately 100.000
LiveReceptor AMPAR <Endogenous AMPAR Labeling Reagent>
 9-7 Hongo 2-Chome, Bunkyo-Ku Tokyo 113-0033, Japan LiveReceptor AMPAR Product Background Neurotransmitter receptors including glutamate receptors and GABA receptors
9-7 Hongo 2-Chome, Bunkyo-Ku Tokyo 113-0033, Japan LiveReceptor AMPAR Product Background Neurotransmitter receptors including glutamate receptors and GABA receptors
PRODUCT DESCRIPTION Recombinant HEK293 cell line expressing the four subunits α1, β1, δ and ε of the human nicotinic acetylcholine receptor type I.
 PRODUCT DATASHEET PrecisION hnachr α1/β1/δ/ε-hek Recombinant Cell Line Catalog Number: CYL3052 PRODUCT DESCRIPTION Recombinant HEK293 cell line expressing the four subunits α1, β1, δ and ε of the human
PRODUCT DATASHEET PrecisION hnachr α1/β1/δ/ε-hek Recombinant Cell Line Catalog Number: CYL3052 PRODUCT DESCRIPTION Recombinant HEK293 cell line expressing the four subunits α1, β1, δ and ε of the human
Amaxa Basic Neuron SCN Nucleofector Kit
 Amaxa Basic Neuron SCN Nucleofector Kit For Primary Neural Cells (Small Cell Number) SCN Nucleofector Kits are compatible with Nucleofector ll Devices of serial version S with software version S4 4 or
Amaxa Basic Neuron SCN Nucleofector Kit For Primary Neural Cells (Small Cell Number) SCN Nucleofector Kits are compatible with Nucleofector ll Devices of serial version S with software version S4 4 or
Amaxa Chicken Neuron Nucleofector Kit
 Amaxa Chicken Neuron Nucleofector Kit For Primary Chicken Hippocampal Neurons Primary dissociated chicken hippocampal neurons, prepared from chicken embryos (E7) as mixed glial cultures. Example for Nucleofection
Amaxa Chicken Neuron Nucleofector Kit For Primary Chicken Hippocampal Neurons Primary dissociated chicken hippocampal neurons, prepared from chicken embryos (E7) as mixed glial cultures. Example for Nucleofection
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Integrative Biology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Table S1. Definition of quantitative cellular features
Electronic Supplementary Material (ESI) for Integrative Biology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Table S1. Definition of quantitative cellular features
Ratiometric Calcium Assay Kit
 BD Technical Data Sheet Ratiometric Calcium Assay Kit Product Information Catalog Number: 644244 Components: Ratiometric Calcium Indicator, 10 vials, lyophilized 10X Signal Enhancer, 100 ml Description
BD Technical Data Sheet Ratiometric Calcium Assay Kit Product Information Catalog Number: 644244 Components: Ratiometric Calcium Indicator, 10 vials, lyophilized 10X Signal Enhancer, 100 ml Description
< Supporting Information >
 SUPPORTING INFORMATION 1 < Supporting Information > Discovery of autophagy modulators through the construction of high-content screening platform via monitoring of lipid droplets Sanghee Lee, Eunha Kim,
SUPPORTING INFORMATION 1 < Supporting Information > Discovery of autophagy modulators through the construction of high-content screening platform via monitoring of lipid droplets Sanghee Lee, Eunha Kim,
Supplementary Information Cyclin Y inhibits plasticity-induced AMPA receptor exocytosis and LTP
 Supplementary Information Cyclin Y inhibits plasticity-induced AMPA receptor exocytosis and LTP Eunsil Cho, Dong-Hyun Kim, Young-Na Hur, Daniel J. Whitcomb, Philip Regan, Jung-Hwa Hong, Hanna Kim, Young
Supplementary Information Cyclin Y inhibits plasticity-induced AMPA receptor exocytosis and LTP Eunsil Cho, Dong-Hyun Kim, Young-Na Hur, Daniel J. Whitcomb, Philip Regan, Jung-Hwa Hong, Hanna Kim, Young
SUPPLEMENTARY NOTE 3. Supplememtary Note 3, Wehr et al., Monitoring Regulated Protein-Protein Interactions Using Split-TEV 1
 SUPPLEMENTARY NOTE 3 Fluorescent Proteolysis-only TEV-Reporters We generated TEV reporter that allow visualizing TEV activity at the membrane and in the cytosol of living cells not relying on the addition
SUPPLEMENTARY NOTE 3 Fluorescent Proteolysis-only TEV-Reporters We generated TEV reporter that allow visualizing TEV activity at the membrane and in the cytosol of living cells not relying on the addition
Turn Plasticity Distinguishes Different Modes of Amyloid-β Aggregation
 SUPPORTING INFORMATION Turn Plasticity Distinguishes Different Modes of Amyloid-β Aggregation Nasrollah Rezaei-Ghaleh, Mehriar Amininasab, Karin Giller, Sathish Kumar, Anne Stündl, Anja Schneider, Stefan
SUPPORTING INFORMATION Turn Plasticity Distinguishes Different Modes of Amyloid-β Aggregation Nasrollah Rezaei-Ghaleh, Mehriar Amininasab, Karin Giller, Sathish Kumar, Anne Stündl, Anja Schneider, Stefan
Supplementary
 Supplementary information Supplementary Material and Methods Plasmid construction The transposable element vectors for inducible expression of RFP-FUS wt and EGFP-FUS R521C and EGFP-FUS P525L were derived
Supplementary information Supplementary Material and Methods Plasmid construction The transposable element vectors for inducible expression of RFP-FUS wt and EGFP-FUS R521C and EGFP-FUS P525L were derived
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure S1. Generation of a synaptobrevin2-mrfp knock-in mouse. (a) Targeting strategy of Syb2-mRFP knock-in mouse leaving the synaptobrevin2 gene locus intact except
SUPPLEMENTARY FIGURES Supplementary Figure S1. Generation of a synaptobrevin2-mrfp knock-in mouse. (a) Targeting strategy of Syb2-mRFP knock-in mouse leaving the synaptobrevin2 gene locus intact except
LINGO-1, A TRANSMEMBRANE SIGNALING PROTEIN, INHIBITS OLIGODENDROCYTE DIFFERENTIATION AND MYELINATION THROUGH INTERCELLULAR SELF- INTERACTIONS.
 Supplemental Data: LINGO-1, A TRANSMEMBRANE SIGNALING PROTEIN, INHIBITS OLIGODENDROCYTE DIFFERENTIATION AND MYELINATION THROUGH INTERCELLULAR SELF- INTERACTIONS. Scott Jepson, Bryan Vought, Christian H.
Supplemental Data: LINGO-1, A TRANSMEMBRANE SIGNALING PROTEIN, INHIBITS OLIGODENDROCYTE DIFFERENTIATION AND MYELINATION THROUGH INTERCELLULAR SELF- INTERACTIONS. Scott Jepson, Bryan Vought, Christian H.
3T3-L1 Differentiation Protocol
 3T3-L1 Differentiation Protocol Written by Eisuke Kato on 2013/10/09 MATERIALS Dulbecco's Modified Eagles Medium (D-MEM) (High Glucose) with L-Glutamine and Phenol Red High glucose (Wako Chem 044-29765,
3T3-L1 Differentiation Protocol Written by Eisuke Kato on 2013/10/09 MATERIALS Dulbecco's Modified Eagles Medium (D-MEM) (High Glucose) with L-Glutamine and Phenol Red High glucose (Wako Chem 044-29765,
DCLK-immunopositive. Bars, 100 µm for B, 50 µm for C.
 Supplementary Figure S1. Characterization of rabbit polyclonal anti-dclk antibody. (A) Immunoblotting of COS7 cells transfected with DCLK1-GFP and DCLK2-GFP expression plasmids probed with anti-dclk antibody
Supplementary Figure S1. Characterization of rabbit polyclonal anti-dclk antibody. (A) Immunoblotting of COS7 cells transfected with DCLK1-GFP and DCLK2-GFP expression plasmids probed with anti-dclk antibody
Supplemental Methods:
 Supplemental Methods: Cell Culture 84A human mammary epithelial cells (HMEC s) were a kind gift from Martha Stampfer (Lawrence Berkeley Laboratory, Berkeley CA). Cells were maintained in DFCI- medium supplemented
Supplemental Methods: Cell Culture 84A human mammary epithelial cells (HMEC s) were a kind gift from Martha Stampfer (Lawrence Berkeley Laboratory, Berkeley CA). Cells were maintained in DFCI- medium supplemented
Hippocampal Neuron Dissociation Transfection and Culture in Microfluidics Chambers Yang Geng *
 Hippocampal Neuron Dissociation Transfection and Culture in Microfluidics Chambers Yang Geng * Department of Pediatrics and Bioengineering, Stanford University School of Medicine, Stanford, USA *For correspondence:
Hippocampal Neuron Dissociation Transfection and Culture in Microfluidics Chambers Yang Geng * Department of Pediatrics and Bioengineering, Stanford University School of Medicine, Stanford, USA *For correspondence:
Cell Surface-Anchored Fluorescent Aptamer Sensor Enables. Imaging of Chemical Transmitter Dynamics
 Supporting Information for Cell Surface-Anchored Fluorescent Aptamer Sensor Enables Imaging of Chemical Transmitter Dynamics Takeshi Tokunaga, Shigeyuki Namiki, Katsuhiro Yamada, Takahiro Imaishi, Hiroshi
Supporting Information for Cell Surface-Anchored Fluorescent Aptamer Sensor Enables Imaging of Chemical Transmitter Dynamics Takeshi Tokunaga, Shigeyuki Namiki, Katsuhiro Yamada, Takahiro Imaishi, Hiroshi
Regulation of Acetylcholine Receptor Clustering by ADF/Cofilin- Directed Vesicular Trafficking
 Regulation of Acetylcholine Receptor Clustering by ADF/Cofilin- Directed Vesicular Trafficking Chi Wai Lee, Jianzhong Han, James R. Bamburg, Liang Han, Rachel Lynn, and James Q. Zheng Supplementary Figures
Regulation of Acetylcholine Receptor Clustering by ADF/Cofilin- Directed Vesicular Trafficking Chi Wai Lee, Jianzhong Han, James R. Bamburg, Liang Han, Rachel Lynn, and James Q. Zheng Supplementary Figures
BD Ratiometric Calcium Assay Kit
 BD Technical Data Sheet BD Ratiometric Calcium Assay Kit Product Information Catalog Number: 644243 Components: Ratiometric Calcium Indicator, 1 vial, lyophilized 10X Signal Enhancer, 10 ml Calcium Assay
BD Technical Data Sheet BD Ratiometric Calcium Assay Kit Product Information Catalog Number: 644243 Components: Ratiometric Calcium Indicator, 1 vial, lyophilized 10X Signal Enhancer, 10 ml Calcium Assay
Supplementary Information (Ha, et. al) Supplementary Figures Supplementary Fig. S1
 Supplementary Information (Ha, et. al) Supplementary Figures Supplementary Fig. S1 a His-ORMDL3 ~ 17 His-ORMDL3 GST-ORMDL3 - + - + IPTG GST-ORMDL3 ~ b Integrated Density (ORMDL3/ -actin) 0.4 0.3 0.2 0.1
Supplementary Information (Ha, et. al) Supplementary Figures Supplementary Fig. S1 a His-ORMDL3 ~ 17 His-ORMDL3 GST-ORMDL3 - + - + IPTG GST-ORMDL3 ~ b Integrated Density (ORMDL3/ -actin) 0.4 0.3 0.2 0.1
Supporting Information
 Supporting Information Deng et al. 10.1073/pnas.1515692112 SI Materials and Methods FPLC. All fusion proteins were expressed and purified through a three-step FPLC purification protocol, as described (20),
Supporting Information Deng et al. 10.1073/pnas.1515692112 SI Materials and Methods FPLC. All fusion proteins were expressed and purified through a three-step FPLC purification protocol, as described (20),
SUPPLEMENTARY INFORMATION
 DOI: 1.138/NNANO.211.185 Mixing sub-attolitre volumes in a quantitative and highly parallel manner with soft matter nanofluidics Sune M. Christensen, Pierre-Yves Bolinger,Nikos S. Hatzakis, Michael W.Mortensen
DOI: 1.138/NNANO.211.185 Mixing sub-attolitre volumes in a quantitative and highly parallel manner with soft matter nanofluidics Sune M. Christensen, Pierre-Yves Bolinger,Nikos S. Hatzakis, Michael W.Mortensen
In vivo recording, forepaw denervation, and isolation of slices: Methods for mapping the forepaw/lower jaw border in anesthetized adult rat primary
 Supplementary Methods In vivo recording, forepaw denervation, and isolation of slices: Methods for mapping the forepaw/lower jaw border in anesthetized adult rat primary somatosensory cortex (S1), forepaw
Supplementary Methods In vivo recording, forepaw denervation, and isolation of slices: Methods for mapping the forepaw/lower jaw border in anesthetized adult rat primary somatosensory cortex (S1), forepaw
MEFs were treated with the indicated concentrations of LLOMe for three hours, washed
 Supplementary Materials and Methods Cell Fractionation MEFs were treated with the indicated concentrations of LLOMe for three hours, washed with ice-cold PBS, collected by centrifugation, and then homogenized
Supplementary Materials and Methods Cell Fractionation MEFs were treated with the indicated concentrations of LLOMe for three hours, washed with ice-cold PBS, collected by centrifugation, and then homogenized
Current ( pa) Current (pa) Voltage (mv) Voltage ( mv)
 Current ( pa) 3000 2000 1000 0-1000 -2000-3000 a -400-200 0 200 400 Voltage (mv) P1 P2 P3 P4 P5 P6 P7 P8 P9 P10 P11 P12 P13 P14 P15 P16 P17 P18 Average Current (pa) 4500 3000 1500 0-1500 -3000-4500 b -400-200
Current ( pa) 3000 2000 1000 0-1000 -2000-3000 a -400-200 0 200 400 Voltage (mv) P1 P2 P3 P4 P5 P6 P7 P8 P9 P10 P11 P12 P13 P14 P15 P16 P17 P18 Average Current (pa) 4500 3000 1500 0-1500 -3000-4500 b -400-200
APPLICATION SPECIFIC PROTOCOL CELL MIGRATION FOR ADHERENT CELLS
 APPLICATION SPECIFIC PROTOCOL CELL MIGRATION FOR ADHERENT CELLS AIM 3D Cell Culture Chips are very useful for the study of 3D cell invasion and migration. The chips are not only suitable for endpoint measurement;
APPLICATION SPECIFIC PROTOCOL CELL MIGRATION FOR ADHERENT CELLS AIM 3D Cell Culture Chips are very useful for the study of 3D cell invasion and migration. The chips are not only suitable for endpoint measurement;
Fig. S1. Endocytosis of extracellular cargo does not depend on dia. (A) To visualize endocytosis, fluorescently labelled wheat germ agglutinin
 Fig. S1. Endocytosis of extracellular cargo does not depend on dia. (A) To visualize endocytosis, fluorescently labelled wheat germ agglutinin (WGA-Alexa555) was injected into the extracellular perivitteline
Fig. S1. Endocytosis of extracellular cargo does not depend on dia. (A) To visualize endocytosis, fluorescently labelled wheat germ agglutinin (WGA-Alexa555) was injected into the extracellular perivitteline
Chapter One. Construction of a Fluorescent α5 Subunit. Elucidation of the unique contribution of the α5 subunit is complicated by several factors
 4 Chapter One Construction of a Fluorescent α5 Subunit The significance of the α5 containing nachr receptor (α5* receptor) has been a challenging question for researchers since its characterization by
4 Chapter One Construction of a Fluorescent α5 Subunit The significance of the α5 containing nachr receptor (α5* receptor) has been a challenging question for researchers since its characterization by
Normalization of Agilent Seahorse XF Data by In-situ Cell Counting Using a BioTek Cytation 5
 Normalization of Agilent Seahorse XF Data by In-situ Cell Counting Using a BioTek Cytation Application Note Authors Yoonseok Kam 1, Ned Jastromb 1, Joe Clayton, Paul Held, and Brian P. Dranka 1 1 Agilent
Normalization of Agilent Seahorse XF Data by In-situ Cell Counting Using a BioTek Cytation Application Note Authors Yoonseok Kam 1, Ned Jastromb 1, Joe Clayton, Paul Held, and Brian P. Dranka 1 1 Agilent
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Monteiro et al., http://www.jcb.org/cgi/content/full/jcb.201306162/dc1 Figure S1. 3D deconvolution microscopy analysis of WASH and exocyst
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Monteiro et al., http://www.jcb.org/cgi/content/full/jcb.201306162/dc1 Figure S1. 3D deconvolution microscopy analysis of WASH and exocyst
Pado, a fluorescent protein with proton channel activity can optically monitor membrane potential,
 Pado, a fluorescent protein with proton channel activity can optically monitor membrane potential, intracellular ph, and map gap junctions. Bok Eum Kang and Bradley J. Baker Supplemental figure 1. S1.
Pado, a fluorescent protein with proton channel activity can optically monitor membrane potential, intracellular ph, and map gap junctions. Bok Eum Kang and Bradley J. Baker Supplemental figure 1. S1.
Supporting Information for: DNA-based delivery vehicles: ph-controlled disassembly and cargo release by Jung-Won Keum and Harry Bermudez
 Supporting Information for: DNA-based delivery vehicles: ph-controlled disassembly and cargo release by Jung-Won Keum and Harry Bermudez DNA sequences Strand Sequence 1- GGGTTAGGGTTAGGGTTAGGGAGGGTTAGGGTTAGGGTTAGGGAGGGTTAGGGTTAGGGTTAGGG
Supporting Information for: DNA-based delivery vehicles: ph-controlled disassembly and cargo release by Jung-Won Keum and Harry Bermudez DNA sequences Strand Sequence 1- GGGTTAGGGTTAGGGTTAGGGAGGGTTAGGGTTAGGGTTAGGGAGGGTTAGGGTTAGGGTTAGGG
Visualizing mechanical tension across membrane receptors with a fluorescent sensor
 Nature Methods Visualizing mechanical tension across membrane receptors with a fluorescent sensor Daniel R. Stabley, Carol Jurchenko, Stephen S. Marshall, Khalid S. Salaita Supplementary Figure 1 Fabrication
Nature Methods Visualizing mechanical tension across membrane receptors with a fluorescent sensor Daniel R. Stabley, Carol Jurchenko, Stephen S. Marshall, Khalid S. Salaita Supplementary Figure 1 Fabrication
Combining Ca 2+ Imaging with L-glutamate Photorelease
 Author manuscript, published in "Cold Spring Harb Protoc 2013;2013(12):1165-8" DOI : 10.1101/pdb.prot073122 PROTOCOL 2 Title page Combining Ca 2+ Imaging with L-glutamate Photorelease SHORT TITLE: Ca 2+
Author manuscript, published in "Cold Spring Harb Protoc 2013;2013(12):1165-8" DOI : 10.1101/pdb.prot073122 PROTOCOL 2 Title page Combining Ca 2+ Imaging with L-glutamate Photorelease SHORT TITLE: Ca 2+
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Legends for Supplementary Tables. Supplementary Table 1. An excel file containing primary screen data. Worksheet 1, Normalized quantification data from a duplicated screen: valid
SUPPLEMENTARY INFORMATION Legends for Supplementary Tables. Supplementary Table 1. An excel file containing primary screen data. Worksheet 1, Normalized quantification data from a duplicated screen: valid
Supplementary Figure 1.
 Supplementary Figure 1. VEH NI Supplementary Figure 1. Cannula tip placement in mice infused with vehicle (VEH) or anisomycin (NI). Cannula tip placement in mice infused with VEH (open circles) or NI (solid
Supplementary Figure 1. VEH NI Supplementary Figure 1. Cannula tip placement in mice infused with vehicle (VEH) or anisomycin (NI). Cannula tip placement in mice infused with VEH (open circles) or NI (solid
Supplementary Materials and Methods
 Supplementary Materials and Methods sirna sequences used in this study The sequences of Stealth Select RNAi for ALK and FLOT-1 were as follows: ALK sense no.1 (ALK): 5 -AAUACUGACAGCCACAGGCAAUGUC-3 ; ALK
Supplementary Materials and Methods sirna sequences used in this study The sequences of Stealth Select RNAi for ALK and FLOT-1 were as follows: ALK sense no.1 (ALK): 5 -AAUACUGACAGCCACAGGCAAUGUC-3 ; ALK
Supporting Information
 Supporting Information Wang et al. 10.1073/pnas.1008382107 SI Methods Tissue Fixation. Adult rats, mice, and Oprd1 exon 1-deleted mice (Jackson Lab) were anesthetized and perfused with 20 ml of warm saline,
Supporting Information Wang et al. 10.1073/pnas.1008382107 SI Methods Tissue Fixation. Adult rats, mice, and Oprd1 exon 1-deleted mice (Jackson Lab) were anesthetized and perfused with 20 ml of warm saline,
University of Dundee. Published in: Proceedings of the National Academy of Sciences. DOI: /pnas Publication date: 2017
 University of Dundee Adenylate cyclase A acting on PKA mediates induction of stalk formation by cyclic diguanylate at the Dictyostelium organizer Chen, Zhi-Hui; Singh, Reema; Cole, Christian; Lawal, Hajara
University of Dundee Adenylate cyclase A acting on PKA mediates induction of stalk formation by cyclic diguanylate at the Dictyostelium organizer Chen, Zhi-Hui; Singh, Reema; Cole, Christian; Lawal, Hajara
Direct Imaging of APP Proteolysis in Living Cells
 Direct Imaging of APP Proteolysis in Living Cells Niccoló Parenti, Ambra Del Grosso, Claudia Antoni, Marco Cecchini, Renato Corradetti, Francesco S. Pavone, Martino Calamai Supplementary Information Supplementary
Direct Imaging of APP Proteolysis in Living Cells Niccoló Parenti, Ambra Del Grosso, Claudia Antoni, Marco Cecchini, Renato Corradetti, Francesco S. Pavone, Martino Calamai Supplementary Information Supplementary
IncuCyte S3 Neuronal Activity Assay
 IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
Supplementary Figure 1. Pathologically phosphorylated Tau is localized to the presynaptic terminals of Alzheimer s disease (AD) patient brains.
 Supplementary Figure 1. Pathologically phosphorylated Tau is localized to the presynaptic terminals of Alzheimer s disease (AD) patient brains. Ultrathin (70 nm) sections of healthy control or AD patient
Supplementary Figure 1. Pathologically phosphorylated Tau is localized to the presynaptic terminals of Alzheimer s disease (AD) patient brains. Ultrathin (70 nm) sections of healthy control or AD patient
Supplementary material 1: DNA tracing
 Supplementary material 1: DNA tracing Figure S1:Typical AFM image showing DNA molecules relaxed when deposited with Mg 2+ DNA molecules that appear to have a higher or larger end (indicated by a red arrow
Supplementary material 1: DNA tracing Figure S1:Typical AFM image showing DNA molecules relaxed when deposited with Mg 2+ DNA molecules that appear to have a higher or larger end (indicated by a red arrow
IncuCyte S3 Neuronal Activity Assay
 IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Rainero et al., http://www.jcb.org/cgi/content/full/jcb.201109112/dc1 Figure S1. The expression of DGK- is reduced upon transfection
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Rainero et al., http://www.jcb.org/cgi/content/full/jcb.201109112/dc1 Figure S1. The expression of DGK- is reduced upon transfection
SUMMER SCHOOL LABORATORY ACTIVITIES
 SUMMER SCHOOL LABORATORY ACTIVITIES ACTIVITIES Monday Tuesday Wednesday Thursday 18 th 19 th 20 th 21 st 1 and 2 A B C D 3 and 4 B C D A 5 and 6 C D A B 7 and 8 D A B C The students are divided into 4
SUMMER SCHOOL LABORATORY ACTIVITIES ACTIVITIES Monday Tuesday Wednesday Thursday 18 th 19 th 20 th 21 st 1 and 2 A B C D 3 and 4 B C D A 5 and 6 C D A B 7 and 8 D A B C The students are divided into 4
In Vitro Study of Receptor-Mediated Silica Nanoparticles Delivery across Blood-Brain Barrier
 Supporting Information In Vitro Study of Receptor-Mediated Silica Nanoparticles Delivery across Blood-Brain Barrier Yang Song, Dan Du, Lei Li, Jun Xu, Prashanta Dutta *,, and Yuehe Lin *, School of Mechanical
Supporting Information In Vitro Study of Receptor-Mediated Silica Nanoparticles Delivery across Blood-Brain Barrier Yang Song, Dan Du, Lei Li, Jun Xu, Prashanta Dutta *,, and Yuehe Lin *, School of Mechanical
CHO-Nav1.5. On QPatch. Application Report:
 Application Report: CHO-Nav1.5 On QPatch The sodium ion channel Nav1.5 is expressed as in integral membrane protein and contains a tetrodotoxinresistant voltage-gated sodium channel subunit.the encoded
Application Report: CHO-Nav1.5 On QPatch The sodium ion channel Nav1.5 is expressed as in integral membrane protein and contains a tetrodotoxinresistant voltage-gated sodium channel subunit.the encoded
FLUORESCENCE TRACKING OF CARDIAC MYOCYTE EXCITATION
 FLUORESCENCE TRACKING OF CARDIAC MYOCYTE EXCITATION Phys 173 / BGGN 266 Amy Davenport and Laurent Palmatier 06/12/09 ABSTRACT Cultured cardiac myocytes were fluorescently tagged with fluo-4am and stimulated
FLUORESCENCE TRACKING OF CARDIAC MYOCYTE EXCITATION Phys 173 / BGGN 266 Amy Davenport and Laurent Palmatier 06/12/09 ABSTRACT Cultured cardiac myocytes were fluorescently tagged with fluo-4am and stimulated
Regulation of Synaptic Structure and Function by FMRP- Associated MicroRNAs mir-125b and mir-132
 Neuron, Volume 65 Regulation of Synaptic Structure and Function by FMRP- Associated MicroRNAs mir-125b and mir-132 Dieter Edbauer, Joel R. Neilson, Kelly A. Foster, Chi-Fong Wang, Daniel P. Seeburg, Matthew
Neuron, Volume 65 Regulation of Synaptic Structure and Function by FMRP- Associated MicroRNAs mir-125b and mir-132 Dieter Edbauer, Joel R. Neilson, Kelly A. Foster, Chi-Fong Wang, Daniel P. Seeburg, Matthew
Supplemental material
 Supplemental material THE JOURNAL OF CELL BIOLOGY Taylor et al., http://www.jcb.org/cgi/content/full/jcb.201403021/dc1 Figure S1. Representative images of Cav 1a -YFP mutants with and without LMB treatment.
Supplemental material THE JOURNAL OF CELL BIOLOGY Taylor et al., http://www.jcb.org/cgi/content/full/jcb.201403021/dc1 Figure S1. Representative images of Cav 1a -YFP mutants with and without LMB treatment.
Membrane Potential Assays Using the ValiScreen Human Kv1.3 Voltage-Gated K + Channel Cell Line on the EnVision Multilabel Plate Reader
 TECHNICAL BRIEF ValiScreen Stable Recombinant Ion Channel Cell Line and the EnVision Multilabel Plate Reader Membrane Potential Assays Using the ValiScreen Human Kv1.3 Voltage-Gated K + Channel Cell Line
TECHNICAL BRIEF ValiScreen Stable Recombinant Ion Channel Cell Line and the EnVision Multilabel Plate Reader Membrane Potential Assays Using the ValiScreen Human Kv1.3 Voltage-Gated K + Channel Cell Line
sirna Transfection Into Primary Neurons Using Fuse-It-siRNA
 sirna Transfection Into Primary Neurons Using Fuse-It-siRNA This Application Note describes a protocol for sirna transfection into sensitive, primary cortical neurons using Fuse-It-siRNA. This innovative
sirna Transfection Into Primary Neurons Using Fuse-It-siRNA This Application Note describes a protocol for sirna transfection into sensitive, primary cortical neurons using Fuse-It-siRNA. This innovative
RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the
 Supplementary Methods RT-PCR and real-time PCR analysis RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the manufacturer s protocol and quantified by measuring the
Supplementary Methods RT-PCR and real-time PCR analysis RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the manufacturer s protocol and quantified by measuring the
Supplementary Figure 1. Thin layer chromatography of R18 salts with different counterions. The mobility of the R18 salts with TPB counterions is much
 Supplementary Figure 1. Thin layer chromatography of R18 salts with different counterions. The mobility of the R18 salts with TPB counterions is much higher with perchlorate, showing their much higher
Supplementary Figure 1. Thin layer chromatography of R18 salts with different counterions. The mobility of the R18 salts with TPB counterions is much higher with perchlorate, showing their much higher
Nature Medicine doi: /nm.2548
 Supplementary Table 1: Genotypes of offspring and embryos from matings of Pmm2 WT/F118L mice with Pmm2 WT/R137H mice total events Pmm2 WT/WT Pmm2 WT/R137H Pmm2 WT/F118L Pmm2 R137H/F118L offspring 117 (100%)
Supplementary Table 1: Genotypes of offspring and embryos from matings of Pmm2 WT/F118L mice with Pmm2 WT/R137H mice total events Pmm2 WT/WT Pmm2 WT/R137H Pmm2 WT/F118L Pmm2 R137H/F118L offspring 117 (100%)
Supplementary information
 Supplementary information Table of Content: Supplementary Results... 2 Supplementary Figure S1: Experimental validation of AP-MS results by coimmunprecipitation Western blot analysis.... 3 Supplementary
Supplementary information Table of Content: Supplementary Results... 2 Supplementary Figure S1: Experimental validation of AP-MS results by coimmunprecipitation Western blot analysis.... 3 Supplementary
THE JOURNAL OF CELL BIOLOGY
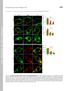 Supplemental Material THE JOURNAL OF CELL BIOLOGY Yeung et al., http://www.jcb.org/cgi/content/full/jcb.200903020/dc1 Figure S1. Assessment of the surface charge of maturing phagosomes. (A C and F H) RAW
Supplemental Material THE JOURNAL OF CELL BIOLOGY Yeung et al., http://www.jcb.org/cgi/content/full/jcb.200903020/dc1 Figure S1. Assessment of the surface charge of maturing phagosomes. (A C and F H) RAW
ratmrp2 PREDIVEZ Protocol CAT. NO. SBPV03
 ratmrp2 PREDIVEZ Protocol CAT. NO. SBPV03 Page 1 of 14 Determination of the interaction of drugs with rat Mrp2 using the rat Mrp2 Fluorescent PREDIVEZ Kit For the following membrane product: -HEK293-VT
ratmrp2 PREDIVEZ Protocol CAT. NO. SBPV03 Page 1 of 14 Determination of the interaction of drugs with rat Mrp2 using the rat Mrp2 Fluorescent PREDIVEZ Kit For the following membrane product: -HEK293-VT
SUPPLEMENTARY INFORMATION
 a 14 12 Densitometry (AU) 1 8 6 4 2 t b 16 NMHC-IIA GAPDH NMHC-IIB Densitometry (AU) 14 12 1 8 6 4 2 1 nm 1 nm 1 nm 1 nm sirna 1 nm 1 nm Figure S1 S4 Quantification of protein levels. (a) The microtubule
a 14 12 Densitometry (AU) 1 8 6 4 2 t b 16 NMHC-IIA GAPDH NMHC-IIB Densitometry (AU) 14 12 1 8 6 4 2 1 nm 1 nm 1 nm 1 nm sirna 1 nm 1 nm Figure S1 S4 Quantification of protein levels. (a) The microtubule
Human CRAC (STIM1/ORAI1) Ion Channel Cell Line
 TECHNICAL DATA SHEET PhotoScreen Ion Channel Cell Line Caution: For Laboratory Use. A product for research purposes only Human CRAC (STIM1/ORAI1) Ion Channel Cell Line Product No.: AX-013-PCL Lot No.:
TECHNICAL DATA SHEET PhotoScreen Ion Channel Cell Line Caution: For Laboratory Use. A product for research purposes only Human CRAC (STIM1/ORAI1) Ion Channel Cell Line Product No.: AX-013-PCL Lot No.:
To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well
 Supplemental Information: Supplemental Methods: Cell culture To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well in 96 well Primaria plates in GNS media and incubated at
Supplemental Information: Supplemental Methods: Cell culture To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well in 96 well Primaria plates in GNS media and incubated at
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. Two-hybrid screening, anti-rab39b specificity and GST-RAB39A pull down. (a) Open reading frame of human RAB39B cdna cloned into pdb-leu and used as bait in
Supplementary Figures Supplementary Figure 1. Two-hybrid screening, anti-rab39b specificity and GST-RAB39A pull down. (a) Open reading frame of human RAB39B cdna cloned into pdb-leu and used as bait in
Page 1 of 5. Product Name Label Quantity Product No. Cy 3 10 µg (~0.75 nmol) MIR Cy µg (~7.5 nmol) MIR 7901
 Page 1 of 5 Label IT RNAi Delivery Control Product Name Label Quantity Product No. Cy 3 10 µg (~0.75 nmol) MIR 7900 Label IT RNAi Delivery Control Cy 3 100 µg (~7.5 nmol) MIR 7901 Fluorescein 10 µg (~0.75
Page 1 of 5 Label IT RNAi Delivery Control Product Name Label Quantity Product No. Cy 3 10 µg (~0.75 nmol) MIR 7900 Label IT RNAi Delivery Control Cy 3 100 µg (~7.5 nmol) MIR 7901 Fluorescein 10 µg (~0.75
CNGC-encoded guard cell Ca 2+ -permeable channels
 Supplemental data CNGC-encoded guard cell Ca 2+ -permeable channels Julian I. Schroeder Division of Biological Sciences, Cell and Developmental Biology Section, University of California San Diego, 9500
Supplemental data CNGC-encoded guard cell Ca 2+ -permeable channels Julian I. Schroeder Division of Biological Sciences, Cell and Developmental Biology Section, University of California San Diego, 9500
