Identification, cloning, and nucleotide sequencing of the ornithine
|
|
|
- Jonathan Palmer
- 6 years ago
- Views:
Transcription
1 Proc. Natl. Acad. Sci. USA Vol. 9, pp , August 993 Biochemistry dentification, cloning, and nucleotide sequencing of the ornithine decarboxylase antizyme gene of Escherichia coli E. S. CANELLAKS*, A. A. PATERAKSt, S.-C. HUANG*, C. A. PANAGOTDS*t, AND D. A. KYRAKDSt *Department of Pharmacology, Yale University School of Medicine, New Haven, CT 65; and tlaboratory of Biochemistry, School of Chemistry, Aristotelian University of Thessaloniki, 54 6 Thessaloniki, Greece Communicated by Philip Siekevitz, April 5, 993 ABSTRACT The ornithine decarboxylase antizyme gene of Escherichia coli was identified by immunological screening of an E. coli genomic library. A 6.4-kilobase fragment containing the antizyme gene was subdoned and sequenced. The open reading frame encoding the antizyme was identified on the basis of its ability to direct the synthesis of immunoreactive antizyme. Antizyme shares significant homology with bacterial transcriptional activators of the two-component regulatory system family; these systems consist of a "sensor" kinase and a transcriptional regulator. The open reading frame next to antizyme is homologous to sensor kinases. Antizyme overproduction inhibits the activities of both ornithine and arginine decarboxylases without affecting their protein levels. Extracts from E. coi bearing an antizyme gene-containing plasmid exhibit increased antizyme activity. These data strongly suggest that (i) the cloned gene encodes the ornithine decarboxylase antizyme and (it) antizyme is a bifunctional protein serving as both an inhibitor of polyamine biosynthesis as well as a transcriptional regulator of an as yet unknown set of genes. Polyamines are necessary components of nearly all living cells, from bacteria to plant to human cells. Although their specific functions remain elusive, they are required components of the cell milieu since polyamine depletion results in severe reductions in cell growth and, in some instances, in cell death (for review, see refs. and 2). Regulation of polyamine biosynthesis is complex and the key biosynthetic enzyme omithine decarboxylase (ODC; L-ornithine carboxylyase, EC 4...7) is probably one of the most highly regulated enzymes (3). t has been shown that the levels and/or the activity of ODC can be modulated both positively and negatively at the transcriptional, translational, and posttranslational levels (for review, see ref. 4). ODC is regulated posttranslationally by covalent modification, degradation, or interaction with inhibitor proteins (antizymes; Az) (5). Az are defined as noncompetitive protein inhibitors of ODC whose synthesis is induced by polyamines (6, 7). Although inhibition of ODC by Az is reversible and active ODC can be recovered from inactive ODC-Az complexes under certain conditions (8), there is also substantial evidence involving Az in ODC degradation (9, ). n Escherichia coli, the posttranslational control of the activities of the polyamine biosynthetic enzymes ODC and arginine decarboxylase (ADC; L-arginine carboxy-lyase, EC 4...9) by Az-like proteins has been studied by our group for a number of years (8,, 2). t is now established that three proteins, whose intracellular levels increase when the polyamine concentrations increase, participate in the negative regulation of both ODC and ADC (). Two of these inhibitors are basic proteins and they have been identified as the ribosomal proteins S2 and L34 (2). The third inhibitor, the E. coli Az, has an acidic apparent p and a molecular weight of 49,5 The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" in accordance with 8 U.S.C. 734 solely to indicate this fact. 729 (). The study of the Az has been very difficult because of its low abundance and because its N terminus is blocked (unpublished results). We now report the isolation of the E. coli Az gene, its location on the E. coli chromosome, as well as the identification and sequence of the open reading frame (ORF) encoding the Az. MATERALS AND METHODS Bacterial Strains, Plasmids, and Phages. E. coli MG655 (A-, F-) (3); KL527 [MG655 gyra, A (spea-speb)97, A(speC-glc)63] (4); DH5a, HB, TG-, and LE392; phages M3mpl8 and M3mpl9 (5); and plasmids puc8, puc9, and pc2h (6) were used in this work. Protein Purification and Antibody Production. ODC and ADC were purified to homogeneity by standard methods (7). Az was purified as described (). Antibodies to Az were produced by injecting a female New Zealand White rabbit subcutaneously with 3,ug of Az in complete Freund's adjuvant. After 4 weeks,,ug of Az in incomplete Freund's adjuvant was injected and blood was drawn days later. ODC and ADC antibodies have been described (8). Extracts, Enzyme Assays, and mmunoblotting. Extracts were prepared as described (4) and protein concentrations were determined (9). ODC, ADC, and Az activity assays and unit definitions have been described (8, 7). Variations in the in vitro ODC assays, probably due to its sensitivity to activating and inhibitor nucleotides (2), were prevented by including mm GTP. All assays were performed in triplicate. SDS/PAGE and immunoblotting were performed by standard methods (2, 22). mmunoadsorption of Az. E. coli extracts enriched in Az were prepared by adjusting the ph of E. coli lysates to.8 (), stirring for 3 min at 4 C, and dialyzing the soluble proteins against buffer A [2 mm Tris-HCl, ph 7.4/% (vol/vol) glycerol/i mm dithiothreitol]. The extracts (5 mg of protein) were applied to.5 ml of protein A-Sepharose (Pharmacia) columns containing bound anti-az or preimmune immunoglobulins, and they were circulated for 4 hr at 4 C. Each column was washed with ml of buffer A containing.5 M NaCl, followed by 5 ml of buffer A. Elution was performed with. M glycine HCl (ph 2.6). Subcloning and Sequencing. The 6.4-kb BamH fragment, containing the Az gene, was digested with several restriction enzymes (see Fig. 2), and the resulting fragments were cloned in M3mpl8 and M3mpl9 phages (5). Recombinant plaques were identified by hybridization (23). Single- Abbreviations: ODC, ornithine decarboxylase; ADC, arginine decarboxylase; Az, antizyme(s); ORF, open reading frame; GST, glutathione S-transferase. TTo whom reprint requests should be addressed at: Department of Microbiology, College of Physicians and Surgeons, Columbia University, New York, NY 32. The sequence reported in this paper has been deposited in the GenBank data base (accession no. L378).
2 73 Biochemistry: Canellakis et al. stranded phage DNA was isolated as described (5). Both DNA strands were sequenced with the Sequenase kit (version 2.) from United States Biochemical. Computer analyses were performed with either DNA STRDER. (24) or MAcDNAss version. (Hitachi Software Engineering, Brisbane, CA) programs. The National Biomedical Resource Foundation-Protein dentification Resource data base, on CD-ROM, was searched for sequence homologies using MAcDNASs. Generation of Glutathione S-Transferase (GST)-Az Fusion Protein. A 72-bp Pvu l/ecor fragment (bp 294-2; see Fig. 3) was ligated to plasmid pgex-3x (25), which had been cut with Sma and EcoR. The resulting recombinant plasmid pcpc-xazl produced a GST-Az fusion protein, which contained amino acids of Az. RESULTS Characterization of the Anti-Az Antibody. We have previously shown that the antibody raised against Az recognizes a 5-kDa protein whose levels increase upon E. coli growth in the presence of polyamines (26). To further demonstrate that this protein is Az, we tested the ability of immobilized anti-az antibody to bind it from Az-enriched cell extracts. The eluate from the anti-az immunoaffinity column exhibited strong Az activity, whereas no activity was detected in the eluate from a control column (Fig. ). This finding indicates that the protein recognized by the antibody is Az. dentification of Clones Containing the Az Gene and Subcloning. mmunological screening of an E. coli genomic library in A phage (27) was used to identify the Az gene. This procedure was chosen because of difficulties in obtaining N-terminal or internal peptide sequences of the Az protein. The library was subdivided into groups of 2 consecutive clones. Liquid cultures of E. coli LE392 were infected with these clone mixtures and allowed to grow until lysis. The lysates were analyzed by SDS/PAGE and immunoblotting. The infection procedure was repeated with individual clones from the positive group. Two consecutive clones, SA and 4F2 (27), expressed a 5-kDa immunoreactive protein. These two clones share a common chromosomal region located at the 48th minute of the E. coli linkage map (28). DNA from phage 4F2 was subcloned in plasmid puc9. E. coli DH5a transformed with the resulting recombinant plasmids were screened by immunoblotting. The plasmid Table. Effect of Az overproduction on ODC and ADC activity Specific activity (% activity) ODC Strain ADC - GTP + GTP MG (%o) 4 (%) 35 (%) MG655/pUC9 4.9 (95%) 3 (93%) 33 (5%) MG655/pUC-Az 2.4 (46%) 7 (5%) 2 (6%) Specific activities are expressed as nmol of CO2 produced per mg of protein per 6-min incubation. Preparation of cell extracts and assays for ODC and ADC activities were performed as described. Numbers represent average of three independent experiments, each performed in triplicate. Variation between experiments was <5%. described in this study (puc-az) contains a 6.4-kb BamH fragment. Az s Overproduced in puc-az-transformed Ceils. To ensure that the cloned gene was indeed the Az gene, E. coli KL527 (ODC-, ADC-) (4) was transformed with puc-az. Az was partially purified until the perchloric acid step () to remove ribosomal proteins S2 and L34, which also inhibit ODC and could interfere with the assay (2). The specific activity of Az in these extracts was 4-6 units per mg of protein, which is 5- to 8-fold higher than that of extracts from control cells (results not shown). This agrees well with the extent of Az protein overproduction (5- to -fold) in these cells (data not shown). The ability of the overproduced protein to inhibit ODC and ADC activities in wild-type cells was also tested. The activities of endogenous ODC and ADC were decreased 5% in lysates from E. coli MG655 transformed with puc-az (Table ). mmunoblot analysis of the extracts indicated that the protein levels of these two enzymes were not decreased in the Az-overproducing cells (results not shown). Therefore, the decreased activities of ODC and ADC result from posttranslational inhibition and not from decreased amounts. Sequence of the Az Gene. The 6.4-kb insert was sequenced as described. The sequencing strategy and partial restriction map are shown in Fig. 2. Two ORFs, which could encode proteins with sizes similar to that ofaz, were identified in the sequenced region (Fig. 2c). Transcription of both ORFs proceeded in the same direction. One part ofthe sequence (bp ) overlaps with the published sequence of the rcsc gene (29), which is also located in the 48th minute of the E. coli genetic map. The nucleotide and deduced amino acid sequences of ORF2, which encodes Az (see below), are given in Fig. 3. a Proc. Natl. Acad. Sci. USA 9 (993) Kb H B E S E B law Frcoton Number b i -e- e e b ii FG.. Elution of Az from an immunoaffinity column. Prepara- denote direction of sequencing. (c) ORF map. The truncated ORF, tion of extracts and absorption on the column Mvere performed as which encodes protein rcsc (29) as well as a truncated unknown ORF described. Az activity was assayed by mixing 2.5 J from each (ORFX), is shown. ORFs with sizes smaller than.5 kb are not fraction with 8 units ( ng) of purified ODC. shown. c ORFX -4q. Az ORF ORF rcsc *._. FG. 2. (a) Partial restriction map of the sequenced fragment _9r < containing the Az gene. E, EcoR; S, Sal ; H, Hindll; B, BamH (b) Sequencing strategy is presented. Clones used in sequencing were generated by digesting the 6.4-kb BamH fragment with (i) EcoR alone orin combination withalu, (ii) EcoRV, or (iii) Hinc. Arrows
3 Biochemistry: Canellakis et al. Proc. Natl. Acad. Sci. USA 9 (993) ACC TGT TTG CCG ATA TTC ACC CTG ATG TGG TGT TGA TGG ATA TCC GCA TGC RGA TGG ACG GCA TCA A G CAC TAA AGG AGA TGC GCA GCC ATG AGA CCC GGA CAC CCG TTA TTC TGA&TGACGG CCT ATG CGG AAG TGG AAA CCG CCG GTC GAA GCG CTA Met Arg Lys Trp Lys Pro Pro Val Glu Ala Leu CGC TGC GGA CGC TTC GAC TAT GTT ATT AAA CCG TTT GAT CTC GAT GAG TTG AAT Arg Cys Gly Arg Phe Asp Tyr Val le Lys Pro Phe Asp Leu Asp Glu Leu Asn TTA ATC GTT CAG CGC GCT TTA CAA CTC CAG TCA ATG AAA AAA GAG ATC CGT CAT Leu le Val Gln Arg Ala Leu Gln Leu Gln Ser Met Lys Lys Glu le Arg His CTG CAC CAG GCA CTG AGC ACC AGC TGG CAA TGG GGG CAC ATT CTC ACC AAC AGC Leu His Gln Ala Leu Ser Thr Ser Trp Gln Trp Gly His le Leu Thr Asn Ser CCG GCG ATG ATG GAC ATC TGC AAA GAC ACC GCC AAA ATT GCC CTT TCT CAG GCC Pro Ala Met Met Asp le Cys Lys Asp Thr Ala Lys le Ala Leu Ser Gln Ala AGC GTC TTG ATT AGC GGT GAA AGC GGC ACC GGG AAA GAG TTG ATT GCC AGA GCG Ser Val Leu le Ser Gly Glu Ser Gly Thr Gly Lys Glu Leu le Ala Arg Ala ATT CAC TAC AAT TCG CGG CGG GCA AAG GGG CCG TTC ATT AAA GTC AAC TGC GCG le His Tyr Asn Ser Arg Arg Ala Lys Gly Pro Phe le Lys Val Asn Cys Ala GCG CTG CCG GAA TCG TTG CTC GAA AGT GAA CTG TTT GGT CAT GAA AAA GGT GCA Ala Leu Pro Glu Ser Leu Leu Glu Ser Glu Leu Phe Gly His Glu Lys Gly Ala TTT ACT GGT GCA CAA ACC TTG CGT CAG GGA TTA TTT GAA CGA GCC AAC GAA GGT Phe Thr Gly Ala Gln Thr Leu Arg Gln Gly Leu Phe Glu Arg Ala Asn Glu Gly ACT CTG CTC CTC GAC GAA ATT GGC GAA ATG CCG CTG GTA CTA CAA GCC AAA TTA Thr Leu Leu Leu Asp Glu le Gly Glu Met Pro Leu Val Leu Gln Ala Lys Leu CTA CGC ATT CTA CAG GAA CGG GAA TTT GAA CGG ATT GGC GGC CAT CAG ACC ATA Leu Arg le Leu Gln Glu Arg Glu Phe Glu Arg le Gly Gly His Gln Thr le AAA GTT GAT ATC CGC ATC ATT GCT GCC ACC AAC CGC GAC TTG CAG GCA ATG GTA Lys Val Asp le Arg le le Ala Ala Thr Asn Arg Asp Leu Gln Ala Met Val AAA GAA GGC ACC TTC CGT GAA GAT CTC TTT TAT CGC CTT AAC GTT ATT CAT TTA Lys Glu Gly Thr Phe Arg Glu Asp Leu Phe Tyr Arg Leu Asn Val le His Leu ATA CTG CCG CCT CTG CGC GAT CGC CGG GAA GAT ATT TCC CTG TTA GCT AAT CAC le Leu Pro Pro Leu Arg Asp Arg Arg Glu Asp le Ser Leu Leu Ala Asn His TTT TTG CAA AAA TTC AGT AGT GAG AAT CAG CGC GAT ATT ATC GAC ATC GAT CCG Phe Leu Gln Lys Phe Ser Ser Glu Asn Gln Arg Asp le le Asp le Asp Pro ATG GCA ATG TCA CTG CTT ACC GCC TGG TCA TGG CCG GGA AAT ATT CGA GAG CTT Met Ala Met Ser Leu Leu Thr Ala Trp Ser Trp Pro Gly Asn le Arg Glu Leu TCC AAC GTT ATT GAA CGC GCC GTC GTG ATG AAT TCA GGC CCG ATC ATT TTT TCT Ser Asn Val le Glu Arg Ala Val Val Met Asn Ser Gly Pro le le Phe Ser GAG GAT CTT CCG CCA CAG ATT CGT CAG CCA GTC TGT AAT GCT GGC GAG GTA AAA Glu Asp Leu Pro Pro Gln le Arg Gln Pro Val Cys Asn Ala Gly Glu Val Lys ACA GCC CCT GTC GGT GAG CGT AAT TTA AAA GAG GAA ATT AAA CGC GTC GAA AAA Thr Ala Pro Val Gly Glu Arg Asn Leu Lys Glu Glu le Lys Arg Val Glu Lys CGC ATC ATT ATG GAA GTG CTG GAA CAA CAA GAA GGA AAC CGA ACC CGC ACT GCT Arg le le Met Glu Val Leu Glu Gln Gln Glu Gly Asn Arg Thr Arg Thr Ala TTA ATG CTG GGC ATC AGT CGC CGT GCA TTG ATG TAT AAA CTC CAG GAA TAC GGT Leu Met Leu Gly le Ser Arg Arg Ala Leu Met Tyr Lys Leu Gln Glu Tyr Gly ATC GAT CCG GCG GAT GTA TAA CAC CAA AAC TTG CTA TGC AGA AAT TTG CAC AG 372 le Asp Pro Ala Asp Val *** 297 GG CAA TTT TCT GCA TAG CCG CTC ATT CTC CTT ATA AAT CCC CAT CCA ATT TAT FG. 3. Nucleotide and deduced amino acid sequence of the Az gene. Sequence is in reverse orientation to that given in Fig. 2. Two boxes located upstream of the Az ORF indicate sequences with homology to - promoter regions recognized by the transcription factor ff54. A putative Shine-Dalgarno sequence preceding the Az initiator codon is underlined. A palindromic sequence downstream of the Az termination codon is also underlined. ORF2 Encodes the Az. The 6.4-kb BamH fragment was digested with Sal (Fig. 2) and the resulting fragments were cloned in plasmid pc2h. Since Sal cleaves within ORF, it was expected that if ORFi encoded Az neither of the two resulting plasmids would express Az. This was not the case, since cells transformed with plasmid pcpc-az carrying the 4.2-kb Sal /BamH fragment including the ORF2 overproduced Az (Fig. 4). This suggested that ORF2 encodes Az. The 6.4-kb BamH fragment was also digested with EcoR, which cleaves within both ORFi and ORF2 coding sequences, and the fragments were subcloned in puc9. None of the resulting recombinant plasmids overproduced Az (data not shown). This nullifies the possibility that one of the minor ORFs contained in the 4.2-kb BamH/Sal fragment regulates Az expression in trans. The Az gene (ORF2) is preceded by a weak Shine- Dalgarno sequence 6 bp upstream from the initiator AUG codon (Fig. 3). t encodes a 378-amino acid protein with a predicted molecular weight of 43,45. The codon usage ofthe Az gene indicates that it contains a large number of infrequently used synonymic codons (3) (data not shown). This may explain, in part, the low abundance of Az, since such codons correspond to rare trna-isoaccepting species in E. coli (3). Calculation of the p of Az based on its amino acid composition resulted in a basic p value of 8.2. This is in apparent disagreement with the acidic nature of the Az protein, which was measured by native isoelectric focusing (). Denaturing two-dimensional gel electrophoresis of extracts from Az-overproducing cells, followed by immunoblot, revealed that Az protein exists in three isoelectric forms with p values between 6. and 6.5 (data not shown). These data indicate that Az is modified posttranslationally (also see below). Az s Homologous with Transcriptional Regulators of the Two-Component System Family. Homology searches between the Az protein sequence and sequences existing in protein data bases indicate that Az shares striking sequence homologies with bacterial transcriptional activators (Fig. 5). These proteins, which include, among others, the gene products of ntrc from Klebsiella pneumoniae, dctd from Rhizobium meliloti, xylr from Pseudomonas putida, nifa from Bradyrhizobium japonicum, flbd from Caulobacter crescentus, and hydg and J7hlA from E. coli, are members of two-component transcriptional regulatory systems (3-33). The synthesis of these transcription factors is regulated by alternative a factors of the (J54 (rpon) family. Although the start point ofaz gene transcription was not determined in the present study, there are two sequences with considerable homology to o54 - regions located 33 and 23 bp upstream of the initiator AUG of the Az gene (Fig. 3). No homologies to -35 sequences were found. To exclude the possibility that ORF2 encodes a transcriptional activator of the Az gene, we subcloned a portion of the ORF2 gene, encoding amino acids , in plasmid
4 732 Biochemistry: Canellakis et al Proc. Natl. Acad. Sci. USA 9 (993) P 2 M kda 7 kda - 48 kda - 29 kda 7 kda _ is to... 29,, -p- DnciK GroEL..GST-Az i- -+ -GST FG. 4. ORF2 encodes Az. The 6.4-kb BamH fragment was digested with Sal and subcloned in plasmid pc2h. Plasmids containing either a 2.2-kb insert (pcpc-j) or a 4.2-kb insert with the intact ORF2 (pcpc-az) were selected. Extracts from E. coli DH5a transformed with the indicated plasmids were analyzed by SDS/ PAGE and immunoblotting. Anti-Az antibody was used at : dilution. Extracts from cells transformed with pc2h (negative control) (lane ; 2,ug), puc-az (positive control) (lane 2; 2,ug), pcpc-j (lane 3; 2,ug), and pcpc-az (lane 4; 2,ug) were used. Positions of size markers used (prestained high range; BRL) are indicated. pgex-3x and expressed it as a fusion protein with GST (25). The resulting full size GST-Az fusion protein (52 kda) and a smaller degradation product were recognized both by anti-az and anti-gst antibodies on immunoblots (Fig. 6). This E E :. 5 -W ox E :. 5.c ] nifa x ntrc ha.x \.. A A xylr fbd ; 5 e '\ dct D FG. 5. Homology plots of Az versus several transcriptional regulators. Az amino acid number is indicated on the vertical axis; transcriptional regulator is indicated on the horizontal axis. FG. 6. A GST-ORF2 fusion protein is recognized both by anti-az and by anti-gst antibodies. Glutathione-Sepharose affinitypurified proteins from control pgex-3x or recombinant pcpc- XAzl-transformed E. coli were analyzed on SDS/% polyacrylamide gels, and either they were stained with Coomassie brilliant blue (lanes P, M,, and 2) or they were electrophoretically transferred to nitrocellulose membranes and probed with anti-gst (lane 3) or anti-az (lane 4) antibodies. Prestained size markers (lane P) were from BRL; unstained size markers (lane M) were from Pharmacia. proves that ORF2 is the structural gene for Az. Two proteins of approximately 6 and 7 kda copurify with GST-Az, but not with GST alone, in the affinity chromatography step (Fig. 6). These proteins were identified immunologically as the heat-shock proteins GroEL (HSP6) and DnaK (HSP7) (34) (data not shown). We do not know whether this Az binding to heat shock proteins has any physiological significance or whether the overexpressed GST-Az protein is partially denatured. nterestingly, the predicted 69-amino acid, acidic protein (predicted p 5.8) that is encoded by ORF, has significant homologies with several "sensor" kinases oftwo-component regulatory systems (data not shown). We do not know whether ORFl kinase regulates Az. DSCUSSON The gene encoding the E. coli Az was identified by immunological screening. The Az gene maps at the 48th minute of the E. coli linkage map (28). Extracts from cells transformed with plasmid puc-az exhibited a 5- to 8-fold higher Az specific activity than those from control cells. Such extracts also exhibited a 5- to -fold increase in the levels of Az protein. However, ODC and ADC activities in Azoverproducing extracts are reduced only to -5% of the control cells. This more modest inhibition may be due either to a variety of activators that participate in the regulation of ODC and ADC (2) or to the presence of sequestered or inactive forms of Az. Treatment of E. coli extracts at ph.8 and perchloric acid precipitation (), which were used prior to measuring the Az activity, may reactivate Az by disrupting nonproductive complexes. The N-terminal amino acid of this protein was blocked as was that of Az from nonoverproducing cells. The fact that the cloned gene, which was selected on the basis of immunological cross-reactivity of its product with antibodies raised against Az, can direct the synthesis of a protein that behaves functionally as Az indicates that it is indeed the Az gene. The mammalian Az has been implicated not only in inhibition of ODC but also in its targeting for degradation (9,, 35). Although we have not directly tested the effect of E. coli Az overproduction on ODC stability, the fact that ODC protein levels are not reduced in the Az overproducers indicates that E. coli Az may behave differently than its mammalian counterpart. The predicted pl of Az is 8.2. This is in disagreement with its previously reported acidic pl of 3.5 (). When analyzed
5 Biochemistry: Canellakis et al. by denaturing two-dimensional gel electrophoresis, the Az from overproducing cells was found to consist of three isoelectric forms with p , significantly lower than that predicted from the amino acid composition. This indicates that Az is modified posttranslationally-e.g., by phosphorylation. Such modification could explain the discrepancy, although it is also possible that the previously performed native isoelectric focusing resulted in an erroneous p value. Native isoelectric focusing, unlike denaturing isoelectric focusing, does not disrupt protein-protein or protein-dna complexes. Thus, when proteins complexed with other proteins or nucleic acids are examined by this technique, anomalous p values may be obtained. Although no homology was found between the E. coli and rat Az (36) sequences, significant homology was found between E. coli Az and bacterial transcriptional regulators belonging in the two-component regulator system family (3-33). These systems are usually composed of a sensor kinase and a transcriptional regulator. Activation of the sensor kinase results in its autophosphorylation on a histidyl residue and subsequent phosphorylation of the transcription factor on an aspartyl residue by protein-protein phosphate transfer. Phosphorylation of the transcriptional regulator is usually required for its activation. Activated transcriptional regulators bind to transcriptional enhancers (37) and they activate RNA polymerase, which contains oj54 (33) as or factor. n this respect, it is interesting that ORF shares significant homologies with several sensor kinases. Thus, it is possible that ORF encodes a kinase that regulates the activity of Az by phosphorylation. The apparent dual function of Az is reminiscent of our earlier findings that the S2 and L34 ribosomal proteins also act as inhibitors of ODC and ADC (2). Which gene(s) is regulated by the postulated transcription factor activity of Az, what are the signals for Az activation, and what is its relevance to the polyamine content of E. coli remain to be determined. The above questions need to be addressed at the biochemical as well as genetic levels. We thank Dr. Y. Kohara for the E. coli library, Dr. A. Gragerov for the GroEL antibodies, Dr. A. Sarris for helpful discussions, and Dr. M. E. Gottessman for critically reading the manuscript. This work was supported by United States Public Health Service Grant CA and by United States Public Health Service Career Award 5 K6 GM37 (to E.S.C.).. Tabor, C. W. & Tabor, H. (984) Annu. Rev. Biochem. 53, Tabor, C. W. & Tabor, H. (985) Microbiol. Rev. 49, Pegg, A. E. (986) Biochem. J. 234, Davis, R. H., Morris, D. R. & Coffino, P. (992) Microbiol. Rev. 56, Hayashi, S.-. & Canellakis, E. S. (989) in Ornithine Decarboxylase: Biology, Enzymology and Molecular Genetics, ed. Hayashi, S.-. (Pergamon, New York), pp Fong, W. F., Heller, J. S. & Canellakis, E. S. (9) Biochim. Biophys. Acta 428, Proc. Natl. Acad. Sci. USA 9 (993) Heller, J. S., Fong, W. F. & Canellakis, E. S. (9) Proc. Natl. Acad. Sci. USA 73, Kyriakidis, D. A., Heller, J. S. & Canellakis, E. S. (978) Proc. Natl. Acad. Sci. USA 75, Canellakis, E. S. & Hayashi, S.. (989) in The Physiology of Polyamines, eds. Bachrach, U. & Heimer, Y. M. (CRC, Boca Raton, FL), Vol., pp Murakami, Y., Matsufuji, S., Miyazaki, Y. & Hayashi, S.. (992) J. Biol. Chem. 267, Heller, J. S., Kyriakidis, D. A. & Canellakis, E. S. (983) Biochim. Biophys. Acta, Panagiotidis, C. A. & Canellakis, E. S. (984) J. Biol. Chem. 259, Guyer, M. S., Reed, R. R., Steitz, J. A. & Low, K. B. (98) Cold Spring Harbor Symp. Quant. Biol. 45, Panagiotidis, C. A., Blackburn, S., Low, K. B. & Canellakis, E. S. (987) Proc. Natl. Acad. Sci. USA 84, Messing, J. (983) Methods Enzymol., March, J. L., Erfle, M. & Wykes, E. J. (984) Gene 32, Morris, D. R. & Boeker, E. A. (983) Methods Enzymol. 94, Huang, S. C., Panagiotidis, C. A. & Canellakis, E. S. (99) Proc. Natl. Acad. Sci. USA 87, Bradford, M. M. (9) Anal. Biochem. 72, Holtta, E., Janne, J. & Pispa, J. (974) Biochem. Biophys. Res. Commun. 59, Laemmli, U. K. (97) Nature (London) 227, Towbin, H., Staehelin, T. & Gordon, J. (979) Proc. Natl. Acad. Sci. USA, Maniatis, T., Fritsch, E. F. & Sambrook, J. (989) Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Lab. Press, Plainview, NY). 24. Marck, C. (988) Nucleic Acids Res. 6, Smith, D. B. & Johnson, K. S. (988) Gene 67, Panagiotidis, C. A., Huang, S. C., Tsirka, S. A., Kyriakidis, D. A. & Canellakis, E. S. (989) in Progress in Polyamine Research: Novel Biochemical, Pharmacological and Clinical Aspects, eds. Zappia, V. & Pegg, A. E. (Plenum, New York), Vol. 25, pp Kohara, Y., Akiyama, K. & sono, K. (987) Cell 5, Bachmann, B. J. (987) in Escherichia coli and Salmonella Typhimurium: Cellular and Molecular Biology, ed. Neidhardt, F. C. (Am. Soc. Microbiol., Washington, DC), Vol. 2, pp Stout, V. & Gottesman, S. (99) J. Bacteriol. 72, Konigsberg, W. & Godson, G. N. (983) Proc. Natl. Acad. Sci. USA 8, Bourret, R. B., Borkovich, K. A. & Simon, M.. (99) Annu. Rev. Biochem. 6, Kustu, S., Santero, E., Keener, J., Popham, D. & Weiss, D. (989) Microbiol. Rev. 53, Saier, M. H., Wu, L. F. & Reizer, J. (99) Trends Biochem. Sci. 5, Neidhardt, F. C. & VanBogelen, R. A. (987) in Escherichia coli and Salmonella Typhimurium: Cellular and Molecular Biology, ed. Neidhardt, F. C. (Am. Soc. Microbiol., Washington, DC), Vol. 2, pp Li, X. & Coffino, P. (992) Mol. Cell. Biol. 2, Miyazaki, Y., Matsufuji, S. & Hayashi, S.. (992) Gene 3, Popham, D. L., Szeto, D., Keener, J. & Kustu, J. (989) Science 243,
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Materials Protein synthesis kit. This kit consists of 24 amino acids, 24 transfer RNAs, four messenger RNAs and one ribosome (see below).
 Protein Synthesis Instructions The purpose of today s lab is to: Understand how a cell manufactures proteins from amino acids, using information stored in the genetic code. Assemble models of four very
Protein Synthesis Instructions The purpose of today s lab is to: Understand how a cell manufactures proteins from amino acids, using information stored in the genetic code. Assemble models of four very
ORFs and genes. Please sit in row K or forward
 ORFs and genes Please sit in row K or forward https://www.flickr.com/photos/teseum/3231682806/in/photostream/ Question: why do some strains of Vibrio cause cholera and others don t? Methods Mechanisms
ORFs and genes Please sit in row K or forward https://www.flickr.com/photos/teseum/3231682806/in/photostream/ Question: why do some strains of Vibrio cause cholera and others don t? Methods Mechanisms
Disease and selection in the human genome 3
 Disease and selection in the human genome 3 Ka/Ks revisited Please sit in row K or forward RBFD: human populations, adaptation and immunity Neandertal Museum, Mettman Germany Sequence genome Measure expression
Disease and selection in the human genome 3 Ka/Ks revisited Please sit in row K or forward RBFD: human populations, adaptation and immunity Neandertal Museum, Mettman Germany Sequence genome Measure expression
Project 07/111 Final Report October 31, Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines
 Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
Project 07/111 Final Report October 31, 2007. Project Title: Cloning and expression of porcine complement C3d for enhanced vaccines Project Leader: Dr Douglas C. Hodgins (519-824-4120 Ex 54758, fax 519-824-5930)
NAME:... MODEL ANSWER... STUDENT NUMBER:... Maximum marks: 50. Internal Examiner: Hugh Murrell, Computer Science, UKZN
 COMP710, Bioinformatics with Julia, Test One, Thursday the 20 th of April, 2017, 09h30-11h30 1 NAME:...... MODEL ANSWER... STUDENT NUMBER:...... Maximum marks: 50 Internal Examiner: Hugh Murrell, Computer
COMP710, Bioinformatics with Julia, Test One, Thursday the 20 th of April, 2017, 09h30-11h30 1 NAME:...... MODEL ANSWER... STUDENT NUMBER:...... Maximum marks: 50 Internal Examiner: Hugh Murrell, Computer
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR
 Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Lecture 11: Gene Prediction
 Lecture 11: Gene Prediction Study Chapter 6.11-6.14 1 Gene: A sequence of nucleotides coding for protein Gene Prediction Problem: Determine the beginning and end positions of genes in a genome Where are
Lecture 11: Gene Prediction Study Chapter 6.11-6.14 1 Gene: A sequence of nucleotides coding for protein Gene Prediction Problem: Determine the beginning and end positions of genes in a genome Where are
Expression of Recombinant Proteins
 Expression of Recombinant Proteins Uses of Cloned Genes sequencing reagents (eg, probes) protein production insufficient natural quantities modify/mutagenesis library screening Expression Vector Features
Expression of Recombinant Proteins Uses of Cloned Genes sequencing reagents (eg, probes) protein production insufficient natural quantities modify/mutagenesis library screening Expression Vector Features
G+C content. 1 Introduction. 2 Chromosomes Topology & Counts. 3 Genome size. 4 Replichores and gene orientation. 5 Chirochores.
 1 Introduction 2 Chromosomes Topology & Counts 3 Genome size 4 Replichores and gene orientation 5 Chirochores 6 7 Codon usage 121 marc.bailly-bechet@univ-lyon1.fr Bacterial genome structures Introduction
1 Introduction 2 Chromosomes Topology & Counts 3 Genome size 4 Replichores and gene orientation 5 Chirochores 6 7 Codon usage 121 marc.bailly-bechet@univ-lyon1.fr Bacterial genome structures Introduction
Lecture 19A. DNA computing
 Lecture 19A. DNA computing What exactly is DNA (deoxyribonucleic acid)? DNA is the material that contains codes for the many physical characteristics of every living creature. Your cells use different
Lecture 19A. DNA computing What exactly is DNA (deoxyribonucleic acid)? DNA is the material that contains codes for the many physical characteristics of every living creature. Your cells use different
Supplementary. Table 1: Oligonucleotides and Plasmids. complementary to positions from 77 of the SRα '- GCT CTA GAG AAC TTG AAG TAC AGA CTG C
 Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
Supplementary Table 1: Oligonucleotides and Plasmids 913954 5'- GCT CTA GAG AAC TTG AAG TAC AGA CTG C 913955 5'- CCC AAG CTT ACA GTG TGG CCA TTC TGC TG 223396 5'- CGA CGC GTA CAG TGT GGC CAT TCT GCT G
RPA-AB RPA-C Supplemental Figure S1: SDS-PAGE stained with Coomassie Blue after protein purification.
 RPA-AB RPA-C (a) (b) (c) (d) (e) (f) Supplemental Figure S: SDS-PAGE stained with Coomassie Blue after protein purification. (a) RPA; (b) RPA-AB; (c) RPA-CDE; (d) RPA-CDE core; (e) RPA-DE; and (f) RPA-C
RPA-AB RPA-C (a) (b) (c) (d) (e) (f) Supplemental Figure S: SDS-PAGE stained with Coomassie Blue after protein purification. (a) RPA; (b) RPA-AB; (c) RPA-CDE; (d) RPA-CDE core; (e) RPA-DE; and (f) RPA-C
Supplemental Data Supplemental Figure 1.
 Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Codon Bias with PRISM. 2IM24/25, Fall 2007
 Codon Bias with PRISM 2IM24/25, Fall 2007 from RNA to protein mrna vs. trna aminoacid trna anticodon mrna codon codon-anticodon matching Watson-Crick base pairing A U and C G binding first two nucleotide
Codon Bias with PRISM 2IM24/25, Fall 2007 from RNA to protein mrna vs. trna aminoacid trna anticodon mrna codon codon-anticodon matching Watson-Crick base pairing A U and C G binding first two nucleotide
Supplementary Figure 1A A404 Cells +/- Retinoic Acid
 Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
Supplementary Figure 1A A44 Cells +/- Retinoic Acid 1 1 H3 Lys4 di-methylation SM-actin VEC cfos (-) RA (+) RA 14 1 1 8 6 4 H3 Lys79 di-methylation SM-actin VEC cfos (-) RA (+) RA Supplementary Figure
Supporting information for Biochemistry, 1995, 34(34), , DOI: /bi00034a013
 Supporting information for Biochemistry, 1995, 34(34), 10807 10815, DOI: 10.1021/bi00034a013 LESNIK 10807-1081 Terms & Conditions Electronic Supporting Information files are available without a subscription
Supporting information for Biochemistry, 1995, 34(34), 10807 10815, DOI: 10.1021/bi00034a013 LESNIK 10807-1081 Terms & Conditions Electronic Supporting Information files are available without a subscription
Supplemental material
 Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Homework. A bit about the nature of the atoms of interest. Project. The role of electronega<vity
 Homework Why cited articles are especially useful. citeulike science citation index When cutting and pasting less is more. Project Your protein: I will mail these out this weekend If you haven t gotten
Homework Why cited articles are especially useful. citeulike science citation index When cutting and pasting less is more. Project Your protein: I will mail these out this weekend If you haven t gotten
Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis
 1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
Protein Structure Analysis
 BINF 731 Protein Structure Analysis http://binf.gmu.edu/vaisman/binf731/ Iosif Vaisman COMPUTATIONAL BIOLOGY COMPUTATIONAL STRUCTURAL BIOLOGY COMPUTATIONAL MOLECULAR BIOLOGY BIOINFORMATICS STRUCTURAL BIOINFORMATICS
BINF 731 Protein Structure Analysis http://binf.gmu.edu/vaisman/binf731/ Iosif Vaisman COMPUTATIONAL BIOLOGY COMPUTATIONAL STRUCTURAL BIOLOGY COMPUTATIONAL MOLECULAR BIOLOGY BIOINFORMATICS STRUCTURAL BIOINFORMATICS
Hes6. PPARα. PPARγ HNF4 CD36
 SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
SUPPLEMENTARY INFORMATION Supplementary Table Positions and Sequences of ChIP primers -63 AGGTCACTGCCA -79 AGGTCTGCTGTG Hes6-0067 GGGCAaAGTTCA ACOT -395 GGGGCAgAGTTCA PPARα -309 GGCTCAaAGTTCAaGTTCA CPTa
Det matematisk-naturvitenskapelige fakultet
 UNIVERSITETET I OSLO Det matematisk-naturvitenskapelige fakultet Exam in: MBV4010 Arbeidsmetoder i molekylærbiologi og biokjemi I MBV4010 Methods in molecular biology and biochemistry I Day of exam: Friday
UNIVERSITETET I OSLO Det matematisk-naturvitenskapelige fakultet Exam in: MBV4010 Arbeidsmetoder i molekylærbiologi og biokjemi I MBV4010 Methods in molecular biology and biochemistry I Day of exam: Friday
Cat. # Product Size DS130 DynaExpress TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1
 Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were
 1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
Multiplexing Genome-scale Engineering
 Multiplexing Genome-scale Engineering Harris Wang, Ph.D. Department of Systems Biology Department of Pathology & Cell Biology http://wanglab.c2b2.columbia.edu Rise of Genomics An Expanding Toolbox Esvelt
Multiplexing Genome-scale Engineering Harris Wang, Ph.D. Department of Systems Biology Department of Pathology & Cell Biology http://wanglab.c2b2.columbia.edu Rise of Genomics An Expanding Toolbox Esvelt
Primer Design Workshop. École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria
 Primer Design Workshop École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria Scenario You have discovered the presence of a novel endophy5c organism living inside the cells
Primer Design Workshop École d'été en géné-que des champignons 2012 Dr. Will Hintz University of Victoria Scenario You have discovered the presence of a novel endophy5c organism living inside the cells
PCR analysis was performed to show the presence and the integrity of the var1csa and var-
 Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Arabidopsis actin depolymerizing factor AtADF4 mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB
 Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
Supplemental Data. mir156-regulated SPL Transcription. Factors Define an Endogenous Flowering. Pathway in Arabidopsis thaliana
 Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH).
 Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Table S1. Bacterial strains (Related to Results and Experimental Procedures)
 Table S1. Bacterial strains (Related to Results and Experimental Procedures) Strain number Relevant genotype Source or reference 1045 AB1157 Graham Walker (Donnelly and Walker, 1989) 2458 3084 (MG1655)
Table S1. Bacterial strains (Related to Results and Experimental Procedures) Strain number Relevant genotype Source or reference 1045 AB1157 Graham Walker (Donnelly and Walker, 1989) 2458 3084 (MG1655)
National PHL TB DST Reference Center PSQ Reporting Language Table of Contents
 PSQ Reporting Language Table of Contents Document Page Number PSQ for Rifampin 2-6 Comparison table for rpob Codon Numbering 2 rpob mutation list (new numbering system) 3-5 rpob interpretations 6 PSQ for
PSQ Reporting Language Table of Contents Document Page Number PSQ for Rifampin 2-6 Comparison table for rpob Codon Numbering 2 rpob mutation list (new numbering system) 3-5 rpob interpretations 6 PSQ for
Supplementary Information. Construction of Lasso Peptide Fusion Proteins
 Supplementary Information Construction of Lasso Peptide Fusion Proteins Chuhan Zong 1, Mikhail O. Maksimov 2, A. James Link 2,3 * Departments of 1 Chemistry, 2 Chemical and Biological Engineering, and
Supplementary Information Construction of Lasso Peptide Fusion Proteins Chuhan Zong 1, Mikhail O. Maksimov 2, A. James Link 2,3 * Departments of 1 Chemistry, 2 Chemical and Biological Engineering, and
strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular
![strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular](/thumbs/72/66361212.jpg) Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
ΔPDD1 x ΔPDD1. ΔPDD1 x wild type. 70 kd Pdd1. Pdd3
 Supplemental Fig. S1 ΔPDD1 x wild type ΔPDD1 x ΔPDD1 70 kd Pdd1 50 kd 37 kd Pdd3 Supplemental Fig. S1. ΔPDD1 strains express no detectable Pdd1 protein. Western blot analysis of whole-protein extracts
Supplemental Fig. S1 ΔPDD1 x wild type ΔPDD1 x ΔPDD1 70 kd Pdd1 50 kd 37 kd Pdd3 Supplemental Fig. S1. ΔPDD1 strains express no detectable Pdd1 protein. Western blot analysis of whole-protein extracts
Dierks Supplementary Fig. S1
 Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
Dierks Supplementary Fig. S1 ITK SYK PH TH K42R wt K42R (kinase deficient) R29C E42K Y323F R29C E42K Y323F (reduced phospholipid binding) (enhanced phospholipid binding) (reduced Cbl binding) E42K Y323F
Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC
 Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/10/494/eaan6284/dc1 Supplementary Materials for Activation of master virulence regulator PhoP in acidic ph requires the Salmonella-specific protein UgtL Jeongjoon
www.sciencesignaling.org/cgi/content/full/10/494/eaan6284/dc1 Supplementary Materials for Activation of master virulence regulator PhoP in acidic ph requires the Salmonella-specific protein UgtL Jeongjoon
PROTEIN SYNTHESIS Study Guide
 PART A. Read the following: PROTEIN SYNTHESIS Study Guide Protein synthesis is the process used by the body to make proteins. The first step of protein synthesis is called Transcription. It occurs in the
PART A. Read the following: PROTEIN SYNTHESIS Study Guide Protein synthesis is the process used by the body to make proteins. The first step of protein synthesis is called Transcription. It occurs in the
Y-chromosomal haplogroup typing Using SBE reaction
 Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system
 Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system Dr. Tim Welsink Molecular Biology Transient Gene Expression OUTLINE Short
Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system Dr. Tim Welsink Molecular Biology Transient Gene Expression OUTLINE Short
 www.lessonplansinc.com Topic: Gene Mutations WS Summary: Students will learn about frame shift mutations and base substitution mutations. Goals & Objectives: Students will be able to demonstrate how mutations
www.lessonplansinc.com Topic: Gene Mutations WS Summary: Students will learn about frame shift mutations and base substitution mutations. Goals & Objectives: Students will be able to demonstrate how mutations
Genomics and Gene Recognition Genes and Blue Genes
 Genomics and Gene Recognition Genes and Blue Genes November 1, 2004 Prokaryotic Gene Structure prokaryotes are simplest free-living organisms studying prokaryotes can give us a sense what is the minimum
Genomics and Gene Recognition Genes and Blue Genes November 1, 2004 Prokaryotic Gene Structure prokaryotes are simplest free-living organisms studying prokaryotes can give us a sense what is the minimum
Supporting Online Information
 Supporting Online Information Isolation of Human Genomic DNA Sequences with Expanded Nucleobase Selectivity Preeti Rathi, Sara Maurer, Grzegorz Kubik and Daniel Summerer* Department of Chemistry and Chemical
Supporting Online Information Isolation of Human Genomic DNA Sequences with Expanded Nucleobase Selectivity Preeti Rathi, Sara Maurer, Grzegorz Kubik and Daniel Summerer* Department of Chemistry and Chemical
for Programmed Chemo-enzymatic Synthesis of Antigenic Oligosaccharides
 Supporting Information Design of α-transglucosidases of Controlled Specificity for Programmed Chemo-enzymatic Synthesis of Antigenic Oligosaccharides Elise Champion ±,,,, Isabelle André ±,,, Claire Moulis
Supporting Information Design of α-transglucosidases of Controlled Specificity for Programmed Chemo-enzymatic Synthesis of Antigenic Oligosaccharides Elise Champion ±,,,, Isabelle André ±,,, Claire Moulis
Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR
 Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
Supplemental Dataset Supplemental Table 1. Mutant ADAMTS3 alleles detected in HEK293T clone 4C2. DNA sequence Amino acid sequence WT CCTGTCACTTTGGTTGATAGC MVLLSLWLIAAALVEVR Allele 1 CCTGTC------------------GATAGC
PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells
 Supplementary Information for: PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells Ju Hye Jang 1, Hyun Kim 2, Mi Jung Jang 2, Ju Hyun Cho 1,2,* 1 Research Institute
Supplementary Information for: PGRP negatively regulates NOD-mediated cytokine production in rainbow trout liver cells Ju Hye Jang 1, Hyun Kim 2, Mi Jung Jang 2, Ju Hyun Cho 1,2,* 1 Research Institute
Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of
 Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of the cell line) were immunostained for HA, acetylated
Supplementary Figure 1. Localization of MST1 in RPE cells. Proliferating or ciliated HA- MST1 expressing RPE cells (see Fig. 5b for establishment of the cell line) were immunostained for HA, acetylated
Supporting Information
 Supporting Information Transfection of DNA Cages into Mammalian Cells Email: a.turberfield@physics.ox.ac.uk Table of Contents Supporting Figure 1 DNA tetrahedra used in transfection experiments 2 Supporting
Supporting Information Transfection of DNA Cages into Mammalian Cells Email: a.turberfield@physics.ox.ac.uk Table of Contents Supporting Figure 1 DNA tetrahedra used in transfection experiments 2 Supporting
SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer
 TEACHER S GUIDE SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer SYNOPSIS This activity uses the metaphor of decoding a secret message for the Protein Synthesis process. Students teach themselves
TEACHER S GUIDE SAY IT WITH DNA: Protein Synthesis Activity by Larry Flammer SYNOPSIS This activity uses the metaphor of decoding a secret message for the Protein Synthesis process. Students teach themselves
Legends for supplementary figures 1-3
 High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
Lezione 10. Bioinformatica. Mauro Ceccanti e Alberto Paoluzzi
 Lezione 10 Bioinformatica Mauro Ceccanti e Alberto Paoluzzi Dip. Informatica e Automazione Università Roma Tre Dip. Medicina Clinica Università La Sapienza Lezione 10: Sintesi proteica Synthesis of proteins
Lezione 10 Bioinformatica Mauro Ceccanti e Alberto Paoluzzi Dip. Informatica e Automazione Università Roma Tre Dip. Medicina Clinica Università La Sapienza Lezione 10: Sintesi proteica Synthesis of proteins
Gene synthesis by circular assembly amplification
 Gene synthesis by circular assembly amplification Duhee Bang & George M Church Supplementary figures and text: Supplementary Figure 1. Dpo4 gene (1.05kb) construction by various methods. Supplementary
Gene synthesis by circular assembly amplification Duhee Bang & George M Church Supplementary figures and text: Supplementary Figure 1. Dpo4 gene (1.05kb) construction by various methods. Supplementary
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Anti-Pim-1 (Cat#3247), anti-met (Cat#3127), anti-ron (Cat#2654), Anti-EGFR
 Supplementary Methods Antibodies Anti-Pim-1 (Cat#3247), anti-met (Cat#3127), anti-ron (Cat#2654), Anti-EGFR (Cat#2646), anti-igf1r (Cat#3018), anti-insr (Cat#3020), anti-akt (pan, Cat#4691), anti-phospho-akt
Supplementary Methods Antibodies Anti-Pim-1 (Cat#3247), anti-met (Cat#3127), anti-ron (Cat#2654), Anti-EGFR (Cat#2646), anti-igf1r (Cat#3018), anti-insr (Cat#3020), anti-akt (pan, Cat#4691), anti-phospho-akt
Supporting Information
 Supporting Information Table S1. Oligonucleotide sequences used in this work Oligo DNA A B C D CpG-A CpG-B CpG-C CpG-D Sequence 5 ACA TTC CTA AGT CTG AAA CAT TAC AGC TTG CTA CAC GAG AAG AGC CGC CAT AGT
Supporting Information Table S1. Oligonucleotide sequences used in this work Oligo DNA A B C D CpG-A CpG-B CpG-C CpG-D Sequence 5 ACA TTC CTA AGT CTG AAA CAT TAC AGC TTG CTA CAC GAG AAG AGC CGC CAT AGT
Supporting Information
 Supporting Information Barderas et al. 10.1073/pnas.0801221105 SI Text: Docking of gastrin to Constructed scfv Models Interactive predocking of the 4-WL-5 motif into the central pocket observed in the
Supporting Information Barderas et al. 10.1073/pnas.0801221105 SI Text: Docking of gastrin to Constructed scfv Models Interactive predocking of the 4-WL-5 motif into the central pocket observed in the
MacBlunt PCR Cloning Kit Manual
 MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1 Characterization of GSCs. a. Immunostaining of primary GSC spheres from GSC lines. Nestin (neural progenitor marker, red), TLX (green). Merged images of nestin,
Supplementary Figures Supplementary Fig. 1 Characterization of GSCs. a. Immunostaining of primary GSC spheres from GSC lines. Nestin (neural progenitor marker, red), TLX (green). Merged images of nestin,
Creation of A Caspese-3 Sensing System Using A Combination of Split- GFP and Split-Intein
 Supplementary Information Creation of A Caspese-3 Sensing System Using A Combination of Split- GFP and Split-Intein Seiji Sakamoto,* Mika Terauchi, Anna Hugo, Tanner Kim, Yasuyuki Araki and Takehiko Wada*
Supplementary Information Creation of A Caspese-3 Sensing System Using A Combination of Split- GFP and Split-Intein Seiji Sakamoto,* Mika Terauchi, Anna Hugo, Tanner Kim, Yasuyuki Araki and Takehiko Wada*
Thr Gly Tyr. Gly Lys Asn
 Your unique body characteristics (traits), such as hair color or blood type, are determined by the proteins your body produces. Proteins are the building blocks of life - in fact, about 45% of the human
Your unique body characteristics (traits), such as hair color or blood type, are determined by the proteins your body produces. Proteins are the building blocks of life - in fact, about 45% of the human
Overexpression Normal expression Overexpression Normal expression. 26 (21.1%) N (%) P-value a N (%)
 SUPPLEMENTARY TABLES Table S1. Alteration of ZNF322A protein expression levels in relation to clinicopathological parameters in 123 Asian and 74 Caucasian lung cancer patients. Asian patients Caucasian
SUPPLEMENTARY TABLES Table S1. Alteration of ZNF322A protein expression levels in relation to clinicopathological parameters in 123 Asian and 74 Caucasian lung cancer patients. Asian patients Caucasian
evaluated with UAS CLB eliciting UAS CIT -N Libraries increase in the
 Supplementary Figures Supplementary Figure 1: Promoter scaffold library assemblies. Many ensembless of libraries were evaluated in this work. As a legend, the box outline color in top half of the figure
Supplementary Figures Supplementary Figure 1: Promoter scaffold library assemblies. Many ensembless of libraries were evaluated in this work. As a legend, the box outline color in top half of the figure
2
 1 2 3 4 5 6 7 Supplemental Table 1. Magnaporthe oryzae strains generated in this study. Strain background Genotype Strain name Description Guy-11 H1:RFP H1:RFP Strain expressing Histone H1- encoding gene
1 2 3 4 5 6 7 Supplemental Table 1. Magnaporthe oryzae strains generated in this study. Strain background Genotype Strain name Description Guy-11 H1:RFP H1:RFP Strain expressing Histone H1- encoding gene
Supplemental Information. Human Senataxin Resolves RNA/DNA Hybrids. Formed at Transcriptional Pause Sites. to Promote Xrn2-Dependent Termination
 Supplemental Information Molecular Cell, Volume 42 Human Senataxin Resolves RNA/DNA Hybrids Formed at Transcriptional Pause Sites to Promote Xrn2-Dependent Termination Konstantina Skourti-Stathaki, Nicholas
Supplemental Information Molecular Cell, Volume 42 Human Senataxin Resolves RNA/DNA Hybrids Formed at Transcriptional Pause Sites to Promote Xrn2-Dependent Termination Konstantina Skourti-Stathaki, Nicholas
Genomic Sequence Analysis using Electron-Ion Interaction
 University of Aizu, Graduation Thesis. March, 25 s1985 1 Genomic Sequence Analysis using Electron-Ion Interaction Potential Masumi Kobayashi s1985 Supervised by Hiroshi Toyoizumi Abstract This paper proposes
University of Aizu, Graduation Thesis. March, 25 s1985 1 Genomic Sequence Analysis using Electron-Ion Interaction Potential Masumi Kobayashi s1985 Supervised by Hiroshi Toyoizumi Abstract This paper proposes
Supplementary Information
 Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
FROM DNA TO GENETIC GENEALOGY Stephen P. Morse
 1. GENES, CHROMOSOMES, AND DNA Chromosomes FROM DNA TO GENETIC GENEALOGY Stephen P. Morse (steve@stevemorse.org) Every human cell = 46 chromosomes (1 to 22 in pairs, 2 sex chromosomes) Male: sex chromosomes
1. GENES, CHROMOSOMES, AND DNA Chromosomes FROM DNA TO GENETIC GENEALOGY Stephen P. Morse (steve@stevemorse.org) Every human cell = 46 chromosomes (1 to 22 in pairs, 2 sex chromosomes) Male: sex chromosomes
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Investigation of the Biosynthesis of the Lasso Peptide Chaxapeptin Using an E. coli-based Production System Helena Martin-Gómez, Uwe Linne, Fernando Albericio, Judit Tulla-Puche,*
SUPPORTING INFORMATION Investigation of the Biosynthesis of the Lasso Peptide Chaxapeptin Using an E. coli-based Production System Helena Martin-Gómez, Uwe Linne, Fernando Albericio, Judit Tulla-Puche,*
II 0.95 DM2 (RPP1) DM3 (At3g61540) b
 Table S2. F 2 Segregation Ratios at 16 C, Related to Figure 2 Cross n c Phenotype Model e 2 Locus A Locus B Normal F 1 -like Enhanced d Uk-1/Uk-3 149 64 36 49 DM2 (RPP1) DM1 (SSI4) a Bla-1/Hh-0 F 3 111
Table S2. F 2 Segregation Ratios at 16 C, Related to Figure 2 Cross n c Phenotype Model e 2 Locus A Locus B Normal F 1 -like Enhanced d Uk-1/Uk-3 149 64 36 49 DM2 (RPP1) DM1 (SSI4) a Bla-1/Hh-0 F 3 111
DNA sentences. How are proteins coded for by DNA? Materials. Teacher instructions. Student instructions. Reflection
 DNA sentences How are proteins coded for by DNA? Deoxyribonucleic acid (DNA) is the molecule of life. DNA is one of the most recognizable nucleic acids, a double-stranded helix. The process by which DNA
DNA sentences How are proteins coded for by DNA? Deoxyribonucleic acid (DNA) is the molecule of life. DNA is one of the most recognizable nucleic acids, a double-stranded helix. The process by which DNA
Introduction to Bioinformatics Dr. Robert Moss
 Introduction to Bioinformatics Dr. Robert Moss Bioinformatics is about searching biological databases, comparing sequences, looking at protein structures, and more generally, asking biological questions
Introduction to Bioinformatics Dr. Robert Moss Bioinformatics is about searching biological databases, comparing sequences, looking at protein structures, and more generally, asking biological questions
Supplemental Data. Bennett et al. (2010). Plant Cell /tpc
 BRN1 ---------MSSSNGGVPPGFRFHPTDEELLHYYLKKKISYEKFEMEVIKEVDLNKIEPWDLQDRCKIGSTPQNEWYFFSHKDRKYPTGS 81 BRN2 --------MGSSSNGGVPPGFRFHPTDEELLHYYLKKKISYQKFEMEVIREVDLNKLEPWDLQERCKIGSTPQNEWYFFSHKDRKYPTGS 82 SMB
BRN1 ---------MSSSNGGVPPGFRFHPTDEELLHYYLKKKISYEKFEMEVIKEVDLNKIEPWDLQDRCKIGSTPQNEWYFFSHKDRKYPTGS 81 BRN2 --------MGSSSNGGVPPGFRFHPTDEELLHYYLKKKISYQKFEMEVIREVDLNKLEPWDLQERCKIGSTPQNEWYFFSHKDRKYPTGS 82 SMB
hcd1tg/hj1tg/ ApoE-/- hcd1tg/hj1tg/ ApoE+/+
 ApoE+/+ ApoE-/- ApoE-/- H&E (1x) Supplementary Figure 1. No obvious pathology is observed in the colon of diseased ApoE-/me. Colon samples were fixed in 1% formalin and laid out in Swiss rolls for paraffin
ApoE+/+ ApoE-/- ApoE-/- H&E (1x) Supplementary Figure 1. No obvious pathology is observed in the colon of diseased ApoE-/me. Colon samples were fixed in 1% formalin and laid out in Swiss rolls for paraffin
An engineered tryptophan zipper-type peptide as a molecular recognition scaffold
 SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
SUPPLEMENTARY MATERIALS AND METHODS. E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1)
 SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
Table S1. Sequences of mutagenesis primers used to create altered rdpa- and sdpa genes
 Supplementary Table and Figures for Structural Basis for the Enantiospecificities of R- and S-Specific Phenoxypropionate/α-Ketoglutarate Dioxygenases by Tina A. Müller, Maria I. Zavodszky, Michael Feig,
Supplementary Table and Figures for Structural Basis for the Enantiospecificities of R- and S-Specific Phenoxypropionate/α-Ketoglutarate Dioxygenases by Tina A. Müller, Maria I. Zavodszky, Michael Feig,
7.03 Problem Set 3 Due before 5 PM on Wednesday, October 18 Hand in answers in recitation section or in the box outside of
 7.03 Problem Set 3 Due before 5 PM on Wednesday, October 18 Hand in answers in recitation section or in the box outside of 68-120 1. The following DNA sequence fragment comes from the middle of a bacterial
7.03 Problem Set 3 Due before 5 PM on Wednesday, October 18 Hand in answers in recitation section or in the box outside of 68-120 1. The following DNA sequence fragment comes from the middle of a bacterial
MCB421 FALL2005 EXAM#3 ANSWERS Page 1 of 12. ANSWER: Both transposon types form small duplications of adjacent host DNA sequences.
 Page 1 of 12 (10pts) 1. There are two mechanisms for transposition used by bacterial transposable elements: replicative (Tn3) and non-replicative (Tn5 and Tn10). Compare and contrast the two mechanisms
Page 1 of 12 (10pts) 1. There are two mechanisms for transposition used by bacterial transposable elements: replicative (Tn3) and non-replicative (Tn5 and Tn10). Compare and contrast the two mechanisms
SUPPORTING INFORMATION FILE
 Intrinsic and extrinsic connections of Tet3 dioxygenase with CXXC zinc finger modules Nan Liu, Mengxi Wang, Wen Deng, Christine S. Schmidt, Weihua Qin, Heinrich Leonhardt and Fabio Spada Department of
Intrinsic and extrinsic connections of Tet3 dioxygenase with CXXC zinc finger modules Nan Liu, Mengxi Wang, Wen Deng, Christine S. Schmidt, Weihua Qin, Heinrich Leonhardt and Fabio Spada Department of
S4B fluorescence (AU)
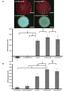 A S4B fluorescence (AU) S4B fluorescence (AU) dsbb csgba csgd dsbb csgba bcsa 5000 * NS NS 4000 * 3000 2000 1000 0 ΔcsgBAΔbcsA ΔcsgDΔdsbBΔbcsA ΔcsgBA ΔdsbBΔcsgBA ΔcsgDΔdsbB B -1000 4000 * * NS 3500 * 3000
A S4B fluorescence (AU) S4B fluorescence (AU) dsbb csgba csgd dsbb csgba bcsa 5000 * NS NS 4000 * 3000 2000 1000 0 ΔcsgBAΔbcsA ΔcsgDΔdsbBΔbcsA ΔcsgBA ΔdsbBΔcsgBA ΔcsgDΔdsbB B -1000 4000 * * NS 3500 * 3000
Supplemental Table 1. Primers used for PCR.
 Supplemental Table 1. Primers used for PCR. Gene Type Primer Sequence Genotyping and semi-quantitative RT-PCR F 5 -TTG CCC GAT CAC CAT CTG TA-3 rwa1-1 R 5 -TGT AGC GAT CAA GGC CTG ATC TAA-3 LB 5 -TAG CAT
Supplemental Table 1. Primers used for PCR. Gene Type Primer Sequence Genotyping and semi-quantitative RT-PCR F 5 -TTG CCC GAT CAC CAT CTG TA-3 rwa1-1 R 5 -TGT AGC GAT CAA GGC CTG ATC TAA-3 LB 5 -TAG CAT
SUPPLEMENTARY INFORMATION
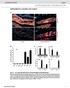 doi: 10.1038/nature07182 SUPPLEMENTAL FIGURES AND TABLES Fig. S1. myf5-expressing cells give rise to brown fat depots and skeletal muscle (a) Perirenal BAT from control (cre negative) and myf5-cre:r26r3-yfp
doi: 10.1038/nature07182 SUPPLEMENTAL FIGURES AND TABLES Fig. S1. myf5-expressing cells give rise to brown fat depots and skeletal muscle (a) Perirenal BAT from control (cre negative) and myf5-cre:r26r3-yfp
INTRODUCTION TO THE MOLECULAR GENETICS OF THE COLOR MUTATIONS IN ROCK POCKET MICE
 The Making of the The Fittest: Making of the Fittest Natural Selection Natural and Adaptation Selection and Adaptation Educator Materials TEACHER MATERIALS INTRODUCTION TO THE MOLECULAR GENETICS OF THE
The Making of the The Fittest: Making of the Fittest Natural Selection Natural and Adaptation Selection and Adaptation Educator Materials TEACHER MATERIALS INTRODUCTION TO THE MOLECULAR GENETICS OF THE
The B1 Protein Guides the Biosynthesis of a Lasso Peptide
 The B1 Protein Guides the Biosynthesis of a Lasso Peptide Shaozhou Zhu 1,2, Christopher D. Fage 1, Julian D. Hegemann 1, Andreas Mielcarek 1, Dushan Yan 1, Uwe Linne 1 & Mohamed A. Marahiel*,1 1 Department
The B1 Protein Guides the Biosynthesis of a Lasso Peptide Shaozhou Zhu 1,2, Christopher D. Fage 1, Julian D. Hegemann 1, Andreas Mielcarek 1, Dushan Yan 1, Uwe Linne 1 & Mohamed A. Marahiel*,1 1 Department
Degenerate Code. Translation. trna. The Code is Degenerate trna / Proofreading Ribosomes Translation Mechanism
 Translation The Code is Degenerate trna / Proofreading Ribosomes Translation Mechanism Degenerate Code There are 64 possible codon triplets There are 20 naturally-encoding amino acids Several codons specify
Translation The Code is Degenerate trna / Proofreading Ribosomes Translation Mechanism Degenerate Code There are 64 possible codon triplets There are 20 naturally-encoding amino acids Several codons specify
Complexity of the Ruminococcus flavefaciens FD-1 cellulosome reflects an expansion of family-related protein-protein interactions
 Complexity of the Ruminococcus flavefaciens FD-1 cellulosome reflects an expansion of family-related protein-protein interactions Vered Israeli-Ruimy 1,*, Pedro Bule 2,*, Sadanari Jindou 3, Bareket Dassa
Complexity of the Ruminococcus flavefaciens FD-1 cellulosome reflects an expansion of family-related protein-protein interactions Vered Israeli-Ruimy 1,*, Pedro Bule 2,*, Sadanari Jindou 3, Bareket Dassa
Causes and Effects of N-Terminal Codon Bias in Bacterial Genes. Mikk Eelmets Journal Club
 Causes and Effects of N-Terminal Codon Bias in Bacterial Genes Mikk Eelmets Journal Club 21.2.214 Introduction Ribosomes were first observed in the mid-195s (Nobel Prize in 1974) Nobel Prize in 29 for
Causes and Effects of N-Terminal Codon Bias in Bacterial Genes Mikk Eelmets Journal Club 21.2.214 Introduction Ribosomes were first observed in the mid-195s (Nobel Prize in 1974) Nobel Prize in 29 for
Supporting Information
 upporting Information hiota et al..73/pnas.159218 I Materials and Methods Yeast trains. Yeast strains used in this study are described in Table 1. TOM22FLAG, a yeast haploid strain for expression of C-terminally
upporting Information hiota et al..73/pnas.159218 I Materials and Methods Yeast trains. Yeast strains used in this study are described in Table 1. TOM22FLAG, a yeast haploid strain for expression of C-terminally
Glutathione (GSH)-Decorated Magnetic Nanoparticles for Binding Glutathione-S-transferase (GST) Fusion Protein and Manipulating Live Cells
 Glutathione (GSH)-Decorated Magnetic Nanoparticles for Binding Glutathione-S-transferase (GST) Fusion Protein and Manipulating Live Cells Yue Pan, Marcus J. C. Long, Xinming Li, Junfeng Shi, Lizbeth Hedstrom,
Glutathione (GSH)-Decorated Magnetic Nanoparticles for Binding Glutathione-S-transferase (GST) Fusion Protein and Manipulating Live Cells Yue Pan, Marcus J. C. Long, Xinming Li, Junfeng Shi, Lizbeth Hedstrom,
PCR-based Markers and Cut Flower Longevity in Carnation
 PCRbased Markers and Cut Flower Longevity in Carnation Laura De Benedetti, Luca Braglia, Simona Bruna, Gianluca Burchi *, Antonio Mercuri and Tito Schiva Istituto Sperimentale per la Floricoltura, Corso
PCRbased Markers and Cut Flower Longevity in Carnation Laura De Benedetti, Luca Braglia, Simona Bruna, Gianluca Burchi *, Antonio Mercuri and Tito Schiva Istituto Sperimentale per la Floricoltura, Corso
CHE-3H84: Protein Engineering Past Exam Papers
 CHE-3H84: Protein Engineering Past Exam Papers Sorted by Topic then Year Knowledge-Based Engineering of Proteins, Large Scale Production of Recombinant Proteins, and Protein Purification Dr. Hemmings 2006/7
CHE-3H84: Protein Engineering Past Exam Papers Sorted by Topic then Year Knowledge-Based Engineering of Proteins, Large Scale Production of Recombinant Proteins, and Protein Purification Dr. Hemmings 2006/7
Supplemental Data. Distinct Pathways for snorna and mrna Termination
 Molecular Cell, Volume 24 Supplemental Data Distinct Pathways for snorna and mrna Termination Minkyu Kim, Lidia Vasiljeva, Oliver J. Rando, Alexander Zhelkovsky, Claire Moore, and Stephen Buratowski A
Molecular Cell, Volume 24 Supplemental Data Distinct Pathways for snorna and mrna Termination Minkyu Kim, Lidia Vasiljeva, Oliver J. Rando, Alexander Zhelkovsky, Claire Moore, and Stephen Buratowski A
SUPPLEMENTAL TABLE S1. Additional descriptions of plasmid constructions and the oligonucleotides used Plasmid or Oligonucleotide
 SUPPLEMENTAL TABLE S1. Additional descriptions of plasmid constructions and the oligonucleotides used Plasmid or Oligonucleotide former/ working Description a designation Plasmids pes213a b pes213-tn5
SUPPLEMENTAL TABLE S1. Additional descriptions of plasmid constructions and the oligonucleotides used Plasmid or Oligonucleotide former/ working Description a designation Plasmids pes213a b pes213-tn5
BioInformatics and Computational Molecular Biology. Course Website
 BioInformatics and Computational Molecular Biology Course Website http://bioinformatics.uchc.edu What is Bioinformatics Bioinformatics upgrades the information content of biological measurements. Discovery
BioInformatics and Computational Molecular Biology Course Website http://bioinformatics.uchc.edu What is Bioinformatics Bioinformatics upgrades the information content of biological measurements. Discovery
Supporting Information. Trifluoroacetophenone-Linked Nucleotides and DNA for Studying of DNA-protein Interactions by 19 F NMR Spectroscopy
 Supporting Information Trifluoroacetophenone-Linked Nucleotides and DNA for Studying of DNA-protein Interactions by 19 F NMR Spectroscopy Agata Olszewska, Radek Pohl and Michal Hocek # * Institute of Organic
Supporting Information Trifluoroacetophenone-Linked Nucleotides and DNA for Studying of DNA-protein Interactions by 19 F NMR Spectroscopy Agata Olszewska, Radek Pohl and Michal Hocek # * Institute of Organic
Chapter 13 Chromatin Structure and its Effects on Transcription
 Chapter 13 Chromatin Structure and its Effects on Transcription Students must be positive that they understand standard PCR. There is a resource on the web for this purpose. Warn them before this class.
Chapter 13 Chromatin Structure and its Effects on Transcription Students must be positive that they understand standard PCR. There is a resource on the web for this purpose. Warn them before this class.
Search for and Analysis of Single Nucleotide Polymorphisms (SNPs) in Rice (Oryza sativa, Oryza rufipogon) and Establishment of SNP Markers
 DNA Research 9, 163 171 (2002) Search for and Analysis of Single Nucleotide Polymorphisms (SNPs) in Rice (Oryza sativa, Oryza rufipogon) and Establishment of SNP Markers Shinobu Nasu, Junko Suzuki, Rieko
DNA Research 9, 163 171 (2002) Search for and Analysis of Single Nucleotide Polymorphisms (SNPs) in Rice (Oryza sativa, Oryza rufipogon) and Establishment of SNP Markers Shinobu Nasu, Junko Suzuki, Rieko
