Supplemental Information. The UBAP1 Subunit of ESCRT-I Interacts. with Ubiquitin via a SOUBA Domain
|
|
|
- Derick Ramsey
- 6 years ago
- Views:
Transcription
1 1 Structure, Volume 20 Supplemental Information The UBAP1 Subunit of ESCRT-I Interacts with Ubiquitin via a SOUBA Domain Monica Agromayor, Nicolas Soler, Anna Caballe, Tonya Kueck, Stefan M. Freund, Mark D. Allen, Mark Bycroft, Olga Perisic, Yu Ye, Bethan McDonald, Hartmut Scheel, Kay Hofmann, Stuart J.D. Neil, Juan Martin-Serrano, and Roger L. Williams Inventory of Supplemental Information Supplemental Figures Figure S1, UBAP1 is a component of ESCRT-I, related to Figure 1 Supplemental Tables Table S1, Plasmid constructs used in this study, related to Figure 1 Table S2, sirna sequences used in this study, related to Figure 2. Table S3, Primary antibodies used in this study, related to Figure 3. Supplemental Experimental Procedures
2 Supplemental Figures 2
3 3 Figure S1, related to Figure 1. UBAP1 is a component of ESCRT-I (A) The cores of all VPS37 isoforms can form stable ESCRT-I complexes with UBAP1 when co-expressed in E. coli and co-purified by affinity chromatography and gel filtration. Coomassie stained SDS-PAGE of the purified complexes are shown. (B) Detection of endogenous UBAP1 and TSG101 in HEK293 cell lysate fractionated by gel filtration chromatography. Fractions were analysed by SDS-PAGE and
4 4 immunoblotted with either anti-ubap1 (top) or anti-tsg101 antibody (bottom). Positions of the molecular weight size markers are indicated above fraction numbers. (C) Co-precipitation assays showing binding of endogenous TSG101 to overexpressed UBAP1 and VPS28 fused to GST. 1% of the starting cell lysate and 10% of the volume eluted from the beads (pull-down) were analyzed by western blot with anti-tsg101 antibody. (D) Effects of over-expression of YFP-tagged UBAP1 constructs on HIV-1 release. 293T cells were transfected with HIV-1 proviral DNA and plasmids expressing YFP fused to UBAP1 full-length (UBAP1-FL) or truncated (UBAP and UBAP1 1-92). YFP control (empty) and YFP-TSG101 were included as negative and positive controls respectively. Infectious virus release was measured after infection of TZM-bl cells (R.L.U. relative luminescence units). Error bars indicate the standard deviation from the mean of three independent experiments. Cell lysates and extracellular virions were analyzed by western blot with anti-hiv Gag antibody. (E-H) Trans-complementation experiments showing that UBAP1 functionally recruits ESCRT-I. While not demonstrating a crucial role of UBAP1 in ESCRT-I-mediated viral budding, these experiments show that, when artificially recruited, UBAP1 can act as an adaptor that bridges Gag to the ESCRT machinery and in particular to ESCRT-I. (E) Infectivity assays following co-transfection of cells with a L-domain mutant HIV-1 proviral plasmid and plasmids expressing an HIV-1 Gag protein in which p6 was either deleted (ENX-), intact (ENX-P6), or replaced with VPS37A (ENX-VPS37A), TSG101 (ENX-TSG101), or UBAP1 (ENX-UBAP1). Infectious virus release was measured after infection of TZM-bl cells, and it is expressed as relative luminescence units (R.L.U.). Error bars indicate the standard deviation from the mean of three independent experiments. Cell lysates and extracellular virions generated were analyzed by western blot with anti-hiv Gag antibody. The positions on the blot of the precursor Pr55Gag, cleavage intermediate p25 (CA-SP1) and mature p24 (CA) proteins are indicated. (F-G) Overexpression of a catalytically
5 5 inactive form of the ATPase VPS4, whose activity is essential for all ESCRTmediated functions, strongly inhibited UBAP1-mediated complementation The same trans-complementation assay was done in the presence of either Myc tagged VPS4, HA-VPS4 mutant or empty vector (control) expressing plasmids, as indicated (F) or RNAi specific for TSG101, ALIX or non-coding control (G). The bottom images in (G) show the sirna-mediated silencing of the endogenous TSG101 and ALIX and a protein loading control as determined by Western blot analysis of the cell lysates with anti-tsg101, anti-alix, and anti-hsp90. (H) A budding deficient HIV-1 provirus and plasmids expressing HIV-1 Gag protein fused to UBAP1 wildtype (ENX-WT) or bearing point mutations (ENX-M1 to M4) were co-transfected in 293T cells. Infectious HIV-1 virion production was measured by inoculation of TZM-bl indicator cells and is expressed as relative luminescence units (R.L.U.). Error bars indicate the standard deviation from the mean of three independent experiments. Cell lysates and extracellular virions generated were analyzed by western blot with anti-hiv Gag antibody. As negative and positive controls plasmids encoding a Gag protein in which p6 was either deleted (ENX-Cont) or intact (ENX-P6) were used respectively.
6 6 Supplemental Tables Table S1: Plasmid constructs used in this study, related to Figure 1. Construct abbreviation Subunit composition ESCRT_UBAP-FL HsVPS28 full length, HsTSG101_ , HsVPS37A_ , HsUBAP1 full length with a C- terminal His6-tag in popc vector (Plasmid pop571) ESCRT_UBAP-1-92 HsVPS28_1-122, HsTSG101_ , HsVPS37A , HsUBAP1_1-92 with C-terminal His6-tag in popc vector (Plasmid pop589) UBAP1_ UBAP1_ UBAP1_( )/K415A/ K416A/E418A GST-UBAP1 GST-VPS28 GST-TSG101 HA-UBAP1 HA-UBAP1 M1 HA-UBAP1 M2 HA-UBAP1 M3 HA-UBAP1 M4 HA-VPS37A HA-VPS37B Myc-VPS28 Myc-VPS4 Myc-VPS4 DN HsUBAP1_ with an N-terminal His6 tag in popth(tev) vector (Plasmid pop673) HsUBAP1_ with an N-terminal His6 tag in popth(tev) vector (Plasmid pop674) HsUBAP1_ with an N-terminal His6 tag in popth(tev) vector (Plasmid pns11) HsUBAP1 full length in pcag-gst vector HsVPS28 full length in pcag-gst vector HsTSG101 full length in pcag-gst vector HsUBAP1 full length in pcr3.1/ha vector HsUBAP1 full length with mutation LDD/AAA in residues 17-19, in pcr3.1/ha vector HsUBAP1 full length with mutation VPF/AAA in residues 20-22, in pcr3.1/ha vector HsUBAP1 full length with mutation F28A, in pcr3.1/ha vector HsUBAP1 full length with mutation E59A, in pcr3.1/ha vector HsVPS37A full length in pcr3.1/ha vector HsVPS37B full length in pcr3.1/ha vector HsVPS28 full length in pcr3.1/myc vector HsVPS4A full length in pcr3.1/myc vector HsVPS4A full length with K173Q point mutation in pcr3.1/myc vector
7 7 Table S2: sirna sequences used in this study, related to Figure 2. Protein Sense sequence Source ALIX GAAGGAUGCUUUCGAUAAAUU Dharmacon hist1 TCGCCTTAAACTATTGGAGAA Qiagen TSG101 CCUCCAGUCUUCUCUCGUC Dharmacon UBAP1-Q3 CTCGACTATCTCTTTGCACAT Qiagen UBAP1-Q4 CAGCTAAAGTTGGTCTACCTA Qiagen Cont ON-TARGET plus sicontrol nontargeting pool (D ) Dharmacon Table S3: Primary antibodies used in this study, related to Figure 3. Antibody Mouuse anti-tubulin (clone DM1a) Mouse anti-hiv-1 p24, clone 183- H12-5C Mouse anti-tsg101, clone 4A10 Mouse anti-cd63 Mouse anti human HLA ABC:FITC, clone W6/32 Mouse anti-human CD317 (tetherin) Mouse anti-mono and polyubiquitiylated conjugates (clone FK2) Mouse anti-ha Rabbit anti-bst2 (tetherin) Rabbit anti-ha Rabbit anti-ubap1 Rabbit anti-vps37a Rabbit anti-hist1 Rabbit anti-hsp90 Source Sigma National AIDS Research and Reference Reagent Program Abcam Developmental studies Hybridoma Bank. University of Iowa AbD Serotec ebioscience Enzo Life Sciences Covance Gift from K. Strebel (Laboratory of Molecular Microbiology, NIH) Rockland Immunochemicals ProteinTech Group ProteinTech Group ProteinTech Group Santa Cruz
8 8 Supplemental Experimental Procedures Expression and purification of recombinant ESCRT-I proteins - Expression was done in C41(DE3)RIPL E. coli. Cells (6 L) were grown at 37 C in 2XTY medium containing 100 µg/ml ampicillin to an optical density of 0.8 at 600 nm. Expression was induced with 1 mm IPTG. After induction, cells were incubated over-night at 12 C, then pelleted by centrifugation at 4000 g. The pellets were resuspended in 160 ml of buffer W1 (20 mm Tris ph8.0 (4 C), 50 mm potassium phosphate ph 8.0 (4 C), 100 mm NaCl, 15 mm imidazole, 0.05% Triton X-100, 1 mm TCEP). Cells were lysed by pulsed sonication for 10 minutes. The lysate was ultracentrifuged at g for 45 min and passed through a 0.2 µm filter before loading on a HisPrep FF 16/10 Column (GE Healthcare) equilibrated in buffer W1. The column was washed with buffer W1, followed by the buffer W2 (same as W1 but without Triton X-100) and eluted with 400 mm imidazole in buffer W2. Fractions containing the four ESCRT-I subunits were pooled and loaded on a 5 ml Hitrap heparin HP column coupled to two tandem 5 ml Hitrap Q columns (GE Healthcare) equilibrated with buffer A (20 mm Tris ph 6.8 (20 C), 1 mm DTT). The complex is not retained on the heparin column but binds the Q-resin. The coupled columns were washed with 1 volume of buffer A before the heparin column was removed. Protein was eluted from the Q column using a 12 column volume gradient of 0-100% buffer B (20 mm Tris ph6.8 (20 C), 1 M NaCl, 1 mm DTT). Fractions were analyzed on SDS-PAGE, and those containing a 1:1:1:1 stoichiometric ratio of the 4 subunits were pooled and concentrated to a volume of 2 ml using a 10 kda MWCO Amicon Ultra-15 concentrator (Millipore). Concentrated proteins were loaded on a HiLoad 16/60 Superdex 200 column (GE Healthcare) equilibrated in 20 mm Tris-HCl ph 7.5, 100 mm NaCl, 2 mm DTT. Fractions corresponding to a tetrameric ESCRT-I complex containing UBAP1 were pooled and concentrated to 12.9 mg/ml.
9 9 Expression and purification of SOUBA - Expression was done as described above, except that the cells were induced at OD 600nm of 0.9 for 4 hours at 37 C. Following purification on a Ni-resin, fractions containing the SOUBA domain were pooled and subjected to TEV cleavage at mass ratio (TEV/protein) of 1/30 overnight at 4 C in order to remove the N-terminal 6His-tag. The cleaved product was passed through a HisTrap FF 5 ml to separate it from the 6His-labelled TEV protease and then loaded on a 5 ml Hitrap Q columns (GE Healthcare) equilibrated with buffer A (20 mm Tris ph 8, 1 mm DTT). Protein was eluted from the Q column, using a 10 column volume gradient of 0-100% buffer B (20 mm Tris ph 8, 1 M NaCl, 1 mm DTT). Fractions containing the protein were concentrated and loaded on a HiLoad 16/60 Superdex 75 column (GE Healthcare) equilibrated in 20 mm Tris-HCl ph 7.5, 100 mm NaCl, 2 mm DTT. SDS-PAGE of the fractions showed a pure 13.5 kda band corresponding to the SOUBA domain, which was pooled and concentrated to 21 mg/ml. Seleno-methionine substituted protein was grown in methionine-requiring B834(DE3) cells. Pre-cultures grown in 2XTY medium containing 100 μg/ml ampicillin were used to inoculate M9 minimal medium supplemented with 0.4 % w/v glucose, 100 µg/ml ampicillin, 2 mm MgSO4, 25µg/mL w/v FeSO4, 1µg/mL Riboflavin, 1µg/mL Niacinamide, 0.1µg/mL pyridoxine monohydrochloride, 1µg/mL thiamine), 0.04 mg/ml of each amino acid except methionine and 0.04 mg/ml seleno-l-methionine. After ~10 hrs at 37 C the cultures were induced with 1 mm IPTG and incubated for an additional 4 hours at 37 C before harvesting. Subsequent purification proceeded as for unlabeled protein. Crystallization of the SOUBA domain - Seleno-methionine crystals of the human SOUBA domain (UBAP ) with the K415A, K416A and E418A triple mutation designed to reduce surface enthropy charge (using SERp server: were grown by mixing 0.2 µl of the concentrated protein with 0.2 µl of a solution containing 645 mm Na/K tartrate, 271 mm LiSO4,
10 mm CHES ph 9.5, 2 mm TCEP, 5% glycerol, and 0.1 µl of seeds from native crystals obtained in similar conditions. The crystals grew in space group P1 with unit cell dimensions given in Table 1. Data collection and refinement were carried out as described in Supplementary experimental procedures. Crystallographic data collection and refinement - The diffraction data were collected at ESRF BM30 using a selenomethionine-substituted crystal and SAD methodology (Table 1). Sites for 7 selenomethionines were located using HySS and refined with SOLVE (Zwart et al., 2008). An initial model for the domain was generated using RESOLVE. This initial model was manually adjusted with COOT (Emsley and Cowtan, 2004) and refinement was carried out using PHENIX. Data collection and refinement statistics are given in Table 1. There are two molecules in the asymmetric unit that are related by a non-crystallographic two-fold axis. The two molecules in the asymmetric unit are linked to each other through a disulfide bond between two-fold related copies of residue Cys452. The structure shows density for two oxidized selenomethionines. Purification of diubiquitins K48-linked and K63-linked diubiquitins were prepared as described previously (Dong et al., 2011). Expression and purification of paramagnetically labelled monoubiquitin Wild-type monoubiquitin cloned into a modified prseta (Invitrogen) vector was mutated using a Quickchange mutagenesis kit to generate the following clones: K6C, D39C, K48C and S57C. The proteins were expressed in C41 cells and purified using ammonium sulphate precipitation, acid denaturation and gel filtration, and were kept in a reduced state throughout by using buffers containing 10 mm DTT. The mutant ubiquitins were passed though a NAP10 column to remove the DTT and immediately labeled by incubation with a 100-fold molar excess of 3-(2-iodoacetamido)-PROXYL (Sigma- Aldrich). The reaction was allowed to proceed for 12 h at room temperature before the excess label was removed using a NAP10 column. To obtain spectra
11 11 of chemically modified but not paramagnetic samples, spin-labeled samples were reduced by the addition of 10 mm ascorbic acid. All paramagnetic and chemically modified but non-paramagnetic ubiquitin samples were dialysed into PBS buffer prior to use. Coprecipitation Assays - 293T cells were transfected with GST, HA and myc expression vectors (1 g of each) using polyethylenimine (Polysciences, Inc). 36 hr later, cells were harvested and lysed in 1 ml of 50 mm Tris.HCl, ph 7.4; 150 mm NaCl; 5 mm EDTA; 5% glycerol; 1% Triton X-100 and a protease inhibitor cocktail (complete mini-edta free, Roche). Clarified lysates were incubated with glutathionesepharose beads (GE Healthcare) for 3 hr at 4 ºC and washed three times with wash buffer (50 mm Tris.HCl, ph 7.4; 150 mm NaCl; 5 mm EDTA; 5% glycerol; 0.1% Triton X-100). The bead-bound proteins were eluted by boiling in 100 μl of sodium dodecyl sulfate sample buffer and analyzed by western blotting with anti-ha monoclonal antibody. The same approach was followed for coprecipitation of proteins fused to One-strep tag with the difference that clarified lysates from cells stably expressing the tagged proteins were incubated with Strep-Tactin superflow plus (Qiagen). Multinucleation Assays HeLa cells (25000) were seeded in a 48 well plate and 2 hr after plating were transfected with 50 pmol of sirna targeting either Luciferase, UBAP1 or hist1 using Dharmafect-1 (Dharmacon). 48 hr later cells were reseeded onto glass coverslips and transfected again with 50 pmol of sirna for another 48 hr. After RNAi treatment, cells were fixed with 3% paraformaldehyde for 15 min, permeabilised with PBS/0.1% Triton X-100 for 5 min and then stained with monoclonal anti-tubulin in PBS/1% BSA for 2 hr. Alexa594 conjugated secondary antibody was applied in PBS for 1 hr. Nuclei were visualized using Hoechst and coverslips were mounted in Mowiol. Images were taken using a Leica AOBS SP2 confocal microscope. 300 cells per coverslip were scored for the presence of
12 12 more than one nucleus. Cells unambiguously connected by midbodies were considered multinucleated. Supplementary references Dong, K. C. et al. (2011). Preparation of distinct ubiquitin chain reagents of high purity and yield. Structure 19, Emsley, P., and Cowtan, K. (2004). Coot: model-building tools for molecular graphics. Acta Crystallogr D Biol Crystallogr 60, Zwart, P. H. et al. (2008). Automated structure solution with the PHENIX suite. Methods Mol Biol 426,
BACTERIAL PRODUCTION EXPRESSION METHOD OVERVIEW: PEF # GENE NAME EXPRESSION VECTOR MOLECULAR WEIGHT kda (full-length) 34.
 BACTERIAL PRODUCTION PEF # GENE NAME EXPRESSION VECTOR MOLECULAR WEIGHT 2015-XXXX XXXX pet-32a 50.9 kda (full-length) 34.0 kda (cleaved) EXPRESSION METHOD OVERVIEW: Plasmid DNA was transformed into BL21
BACTERIAL PRODUCTION PEF # GENE NAME EXPRESSION VECTOR MOLECULAR WEIGHT 2015-XXXX XXXX pet-32a 50.9 kda (full-length) 34.0 kda (cleaved) EXPRESSION METHOD OVERVIEW: Plasmid DNA was transformed into BL21
Proteins were extracted from cultured cells using a modified buffer, and immunoprecipitation and
 Materials and Methods Immunoprecipitation and immunoblot analysis Proteins were extracted from cultured cells using a modified buffer, and immunoprecipitation and immunoblot analyses with corresponding
Materials and Methods Immunoprecipitation and immunoblot analysis Proteins were extracted from cultured cells using a modified buffer, and immunoprecipitation and immunoblot analyses with corresponding
SUPPLEMENTARY INFORMATION
 The Supplementary Information (SI) Methods Cell culture and transfections H1299, U2OS, 293, HeLa cells were maintained in DMEM medium supplemented with 10% fetal bovine serum. H1299 and 293 cells were
The Supplementary Information (SI) Methods Cell culture and transfections H1299, U2OS, 293, HeLa cells were maintained in DMEM medium supplemented with 10% fetal bovine serum. H1299 and 293 cells were
nature methods Enabling IMAC purification of low abundance recombinant proteins from E. coli lysates
 nature methods Enabling IMAC purification of low abundance recombinant proteins from E. coli lysates Audur Magnusdottir, Ida Johansson, Lars-Göran Dahlgren, Pär Nordlund & Helena Berglund Supplementary
nature methods Enabling IMAC purification of low abundance recombinant proteins from E. coli lysates Audur Magnusdottir, Ida Johansson, Lars-Göran Dahlgren, Pär Nordlund & Helena Berglund Supplementary
Supporting Information
 Supporting Information Deng et al. 10.1073/pnas.1515692112 SI Materials and Methods FPLC. All fusion proteins were expressed and purified through a three-step FPLC purification protocol, as described (20),
Supporting Information Deng et al. 10.1073/pnas.1515692112 SI Materials and Methods FPLC. All fusion proteins were expressed and purified through a three-step FPLC purification protocol, as described (20),
Supplementary Information: Materials and Methods. GST and GST-p53 were purified according to standard protocol after
 Supplementary Information: Materials and Methods Recombinant protein expression and in vitro kinase assay. GST and GST-p53 were purified according to standard protocol after induction with.5mm IPTG for
Supplementary Information: Materials and Methods Recombinant protein expression and in vitro kinase assay. GST and GST-p53 were purified according to standard protocol after induction with.5mm IPTG for
Supplementary information, Figure S1A ShHTL7 interacted with MAX2 but not another F-box protein COI1.
 GR24 (μm) 0 20 0 20 GST-ShHTL7 anti-gst His-MAX2 His-COI1 PVDF staining Supplementary information, Figure S1A ShHTL7 interacted with MAX2 but not another F-box protein COI1. Pull-down assays using GST-ShHTL7
GR24 (μm) 0 20 0 20 GST-ShHTL7 anti-gst His-MAX2 His-COI1 PVDF staining Supplementary information, Figure S1A ShHTL7 interacted with MAX2 but not another F-box protein COI1. Pull-down assays using GST-ShHTL7
Strep-Spin Protein Miniprep Kit Catalog No. P2004, P2005
 INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004, P2005 Highlights Fast protocol to purify Strep-tagged proteins from cell-free extracts Screen your recombinant colonies directly for
INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004, P2005 Highlights Fast protocol to purify Strep-tagged proteins from cell-free extracts Screen your recombinant colonies directly for
Supplementary Information: Materials and Methods. Immunoblot and immunoprecipitation. Cells were washed in phosphate buffered
 Supplementary Information: Materials and Methods Immunoblot and immunoprecipitation. Cells were washed in phosphate buffered saline (PBS) and lysed in TNN lysis buffer (50mM Tris at ph 8.0, 120mM NaCl
Supplementary Information: Materials and Methods Immunoblot and immunoprecipitation. Cells were washed in phosphate buffered saline (PBS) and lysed in TNN lysis buffer (50mM Tris at ph 8.0, 120mM NaCl
Supplementary Material
 Supplementary Material Supplementary Methods Cell synchronization. For synchronized cell growth, thymidine was added to 30% confluent U2OS cells to a final concentration of 2.5mM. Cells were incubated
Supplementary Material Supplementary Methods Cell synchronization. For synchronized cell growth, thymidine was added to 30% confluent U2OS cells to a final concentration of 2.5mM. Cells were incubated
Strep-Spin Protein Miniprep Kit Catalog No. P2004 & P2005
 INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004 & P2005 Highlights Fast & Simple: Purify Strep-tagged proteins from cell-free extracts using a spin-column in 7 minutes High-Quality:
INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004 & P2005 Highlights Fast & Simple: Purify Strep-tagged proteins from cell-free extracts using a spin-column in 7 minutes High-Quality:
SUPPLEMENTARY INFORMATION. The nucleotide binding dynamics of MSH2/MSH3 are lesion-dependent.
 SUPPLEMENTARY INFORMATION The nucleotide binding dynamics of /MSH are lesion-dependent. Barbara A. L. Owen, Walter H. Lang, and Cynthia T. McMurray* mau 1 8 6 4 2 mau 8 6 4 2 mau 8 6 4 2 mau 8 7 6 5 4
SUPPLEMENTARY INFORMATION The nucleotide binding dynamics of /MSH are lesion-dependent. Barbara A. L. Owen, Walter H. Lang, and Cynthia T. McMurray* mau 1 8 6 4 2 mau 8 6 4 2 mau 8 6 4 2 mau 8 7 6 5 4
 SUPPLEMENTARY METHODS Peptides HPLC purified peptides from GL Biochem (Shanghai, China): mthsp75-k332, RYTLHY(acetyl-K)TDAPLN; PDH-K81, KADQLY(acetyl-K)QKFIRG; NADH-DH-K243, IENAYK(acetyl-K)TFLPE; ANPtransloc2-K268,
SUPPLEMENTARY METHODS Peptides HPLC purified peptides from GL Biochem (Shanghai, China): mthsp75-k332, RYTLHY(acetyl-K)TDAPLN; PDH-K81, KADQLY(acetyl-K)QKFIRG; NADH-DH-K243, IENAYK(acetyl-K)TFLPE; ANPtransloc2-K268,
Sarker et al. Supplementary Material. Subcellular Fractionation
 Supplementary Material Subcellular Fractionation Transfected 293T cells were harvested with phosphate buffered saline (PBS) and centrifuged at 2000 rpm (500g) for 3 min. The pellet was washed, re-centrifuged
Supplementary Material Subcellular Fractionation Transfected 293T cells were harvested with phosphate buffered saline (PBS) and centrifuged at 2000 rpm (500g) for 3 min. The pellet was washed, re-centrifuged
GST Fusion Protein Purification Kit
 Glutathione Resin GST Fusion Protein Purification Kit Cat. No. L00206 Cat. No. L00207 Technical Manual No. TM0185 Version 01042012 Index 1. Product Description 2. Related Products 3. Purification Procedure
Glutathione Resin GST Fusion Protein Purification Kit Cat. No. L00206 Cat. No. L00207 Technical Manual No. TM0185 Version 01042012 Index 1. Product Description 2. Related Products 3. Purification Procedure
For Research Use Only. Not for use in diagnostic procedures.
 Printed December 13, 2011 Version 1.0 For Research Use Only. Not for use in diagnostic procedures. DDDDK-tagged Protein PURIFICATION GEL with Elution Peptide (MoAb. clone FLA-1) CODE No. 3326 / 3327 PURIFICATION
Printed December 13, 2011 Version 1.0 For Research Use Only. Not for use in diagnostic procedures. DDDDK-tagged Protein PURIFICATION GEL with Elution Peptide (MoAb. clone FLA-1) CODE No. 3326 / 3327 PURIFICATION
OPPF-UK Standard Protocols: Insect Cell Purification
 OPPF-UK Standard Protocols: Insect Cell Purification Last Updated 6 th October 2016 Joanne Nettleship joanne@strubi.ox.ac.uk OPPF-UK SOP: Insect Cell Purification Table of Contents Suggested Schedule...
OPPF-UK Standard Protocols: Insect Cell Purification Last Updated 6 th October 2016 Joanne Nettleship joanne@strubi.ox.ac.uk OPPF-UK SOP: Insect Cell Purification Table of Contents Suggested Schedule...
Supplemental Materials and Methods:
 Supplemental Materials and Methods: Cloning: Oligonucleotides used in the subcloning steps are listed in Supplemental Table 1. Human FANCI (isoform 1, KIAA1794) was subcloned from pcmv6-xl4 [FANCI] in
Supplemental Materials and Methods: Cloning: Oligonucleotides used in the subcloning steps are listed in Supplemental Table 1. Human FANCI (isoform 1, KIAA1794) was subcloned from pcmv6-xl4 [FANCI] in
Supplementary methods
 Supplementary methods Cell culture, infection, transfection, and RNA interference HEK293 cells and its derivatives were grown in DMEM supplemented with 10% FBS. Various constructs were introduced into
Supplementary methods Cell culture, infection, transfection, and RNA interference HEK293 cells and its derivatives were grown in DMEM supplemented with 10% FBS. Various constructs were introduced into
PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%)
 1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%) DESCRIPTION Nickel NTA Magnetic Agarose Beads are products that allow rapid and easy small-scale purification of
1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%) DESCRIPTION Nickel NTA Magnetic Agarose Beads are products that allow rapid and easy small-scale purification of
AFFINITY HIS-TAG PURIFICATION
 DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 50% aqueous suspension containing 30 vol % ethanol. INSTRUCTIONS The resins are adapted
DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 50% aqueous suspension containing 30 vol % ethanol. INSTRUCTIONS The resins are adapted
TECHNICAL BULLETIN. HIS-Select HF Nickel Affinity Gel. Catalog Number H0537 Storage Temperature 2 8 C
 HIS-Select HF Nickel Affinity Gel Catalog Number H0537 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select High Flow (HF) is an immobilized metal-ion affinity chromatography (IMAC)
HIS-Select HF Nickel Affinity Gel Catalog Number H0537 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select High Flow (HF) is an immobilized metal-ion affinity chromatography (IMAC)
pt7ht vector and over-expressed in E. coli as inclusion bodies. Cells were lysed in 6 M
 Supplementary Methods MIG6 production, purification, inhibition, and kinase assays MIG6 segment 1 (30mer, residues 334 364) peptide was synthesized using standard solid-phase peptide synthesis as described
Supplementary Methods MIG6 production, purification, inhibition, and kinase assays MIG6 segment 1 (30mer, residues 334 364) peptide was synthesized using standard solid-phase peptide synthesis as described
Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or
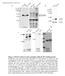 Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or absence of the acetyltransferase CBP and acetylated TDP-43
Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or absence of the acetyltransferase CBP and acetylated TDP-43
Human Viperin Causes Radical SAM Dependent Elongation of E. coli Hinting at its Physiological Role
 Supporting Information Human Viperin Causes Radical SAM Dependent Elongation of E. coli Hinting at its Physiological Role Micah T. Nelp, Anthony P. Young, Branden M. Stepanski, Vahe Bandarian* Department
Supporting Information Human Viperin Causes Radical SAM Dependent Elongation of E. coli Hinting at its Physiological Role Micah T. Nelp, Anthony P. Young, Branden M. Stepanski, Vahe Bandarian* Department
Six genes, Lsm1, Lsm2, Lsm3, Lsm5, Lsm6, and Lsm7, were amplified from the
 Supplementary information, Data S1 Methods Clones and protein preparation Six genes, Lsm1, Lsm2, Lsm3, Lsm5, Lsm6, and Lsm7, were amplified from the Saccharomyces cerevisiae genomic DNA by polymerase chain
Supplementary information, Data S1 Methods Clones and protein preparation Six genes, Lsm1, Lsm2, Lsm3, Lsm5, Lsm6, and Lsm7, were amplified from the Saccharomyces cerevisiae genomic DNA by polymerase chain
Supplementary information
 Supplementary information The E3 ligase RNF8 regulates KU80 removal and NHEJ repair Lin Feng 1, Junjie Chen 1 1 Department of Experimental Radiation Oncology, The University of Texas M. D. Anderson Cancer
Supplementary information The E3 ligase RNF8 regulates KU80 removal and NHEJ repair Lin Feng 1, Junjie Chen 1 1 Department of Experimental Radiation Oncology, The University of Texas M. D. Anderson Cancer
Supplementary Materials and Methods
 Supplementary Materials and Methods sirna sequences used in this study The sequences of Stealth Select RNAi for ALK and FLOT-1 were as follows: ALK sense no.1 (ALK): 5 -AAUACUGACAGCCACAGGCAAUGUC-3 ; ALK
Supplementary Materials and Methods sirna sequences used in this study The sequences of Stealth Select RNAi for ALK and FLOT-1 were as follows: ALK sense no.1 (ALK): 5 -AAUACUGACAGCCACAGGCAAUGUC-3 ; ALK
Supplementary Figure S1 Purification of deubiquitinases HEK293 cells were transfected with the indicated DUB-expressing plasmids.
 Supplementary Figure S1 Purification of deubiquitinases HEK293 cells were transfected with the indicated DUB-expressing plasmids. The cells were harvested 72 h after transfection. FLAG-tagged deubiquitinases
Supplementary Figure S1 Purification of deubiquitinases HEK293 cells were transfected with the indicated DUB-expressing plasmids. The cells were harvested 72 h after transfection. FLAG-tagged deubiquitinases
Structural bases for N-glycan processing by mannosidephosphorylase
 Supporting information Volume 71 (2015) Supporting information for article: Structural bases for N-glycan processing by mannosidephosphorylase Simon Ladevèze, Gianluca Cioci, Pierre Roblin, Lionel Mourey,
Supporting information Volume 71 (2015) Supporting information for article: Structural bases for N-glycan processing by mannosidephosphorylase Simon Ladevèze, Gianluca Cioci, Pierre Roblin, Lionel Mourey,
SUPPLEMENTAL MATERIAL. Supplemental Methods:
 SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
Supplementary information
 Supplementary information Table of Content: Supplementary Results... 2 Supplementary Figure S1: Experimental validation of AP-MS results by coimmunprecipitation Western blot analysis.... 3 Supplementary
Supplementary information Table of Content: Supplementary Results... 2 Supplementary Figure S1: Experimental validation of AP-MS results by coimmunprecipitation Western blot analysis.... 3 Supplementary
Mechanism of Induction and Suppression of Antiviral Immunity Directed by Virus-Derived Small RNAs in Drosophila
 Cell Host & Microbe, Volume 4 Supplemental Data Mechanism of Induction and Suppression of Antiviral Immunity Directed by Virus-Derived Small RNAs in Drosophila Roghiyh Aliyari, Qingfa Wu, Hong-Wei Li,
Cell Host & Microbe, Volume 4 Supplemental Data Mechanism of Induction and Suppression of Antiviral Immunity Directed by Virus-Derived Small RNAs in Drosophila Roghiyh Aliyari, Qingfa Wu, Hong-Wei Li,
HPV E6 oncoprotein targets histone methyltransferases for modulating specific. Chih-Hung Hsu, Kai-Lin Peng, Hua-Ci Jhang, Chia-Hui Lin, Shwu-Yuan Wu,
 1 HPV E oncoprotein targets histone methyltransferases for modulating specific gene transcription 3 5 Chih-Hung Hsu, Kai-Lin Peng, Hua-Ci Jhang, Chia-Hui Lin, Shwu-Yuan Wu, Cheng-Ming Chiang, Sheng-Chung
1 HPV E oncoprotein targets histone methyltransferases for modulating specific gene transcription 3 5 Chih-Hung Hsu, Kai-Lin Peng, Hua-Ci Jhang, Chia-Hui Lin, Shwu-Yuan Wu, Cheng-Ming Chiang, Sheng-Chung
Glutathione Resin. (Cat. # , , , ) think proteins! think G-Biosciences
 191PR 05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Glutathione Resin (Cat. # 786 280, 786 310, 786 311, 786 312) think proteins! think
191PR 05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Glutathione Resin (Cat. # 786 280, 786 310, 786 311, 786 312) think proteins! think
Supplemental Experimental Procedures
 Supplemental Experimental Procedures Cloning, expression and purification of untagged P C, Q D -N 0123, Q D- N 012 and Q D -N 01 - The DNA sequence encoding non tagged P C (starting at the Arginine 57
Supplemental Experimental Procedures Cloning, expression and purification of untagged P C, Q D -N 0123, Q D- N 012 and Q D -N 01 - The DNA sequence encoding non tagged P C (starting at the Arginine 57
SUPPLEMENTAL DATA Experimental procedures
 SUPPLEMENTAL DATA Experimental procedures Cloning, protein production and purification. The DNA sequences corresponding to residues R10-S250 (DBD) and E258-T351 (IPT/TIG) in human EBF1 (gi:31415878), and
SUPPLEMENTAL DATA Experimental procedures Cloning, protein production and purification. The DNA sequences corresponding to residues R10-S250 (DBD) and E258-T351 (IPT/TIG) in human EBF1 (gi:31415878), and
5.2 Protein purification
 Purification of a His 6 -tagged Green Fluorescent Protein (GFP). Protein purification.. Purification of a His 6 -tagged Green Fluorescent Protein (GFP) Principle You can add either a N- or C-terminal His
Purification of a His 6 -tagged Green Fluorescent Protein (GFP). Protein purification.. Purification of a His 6 -tagged Green Fluorescent Protein (GFP) Principle You can add either a N- or C-terminal His
Viral RNAi suppressor reversibly binds sirna to. outcompete Dicer and RISC via multiple-turnover
 Supplementary Data Viral RNAi suppressor reversibly binds sirna to outcompete Dicer and RISC via multiple-turnover Renata A. Rawlings 1,2, Vishalakshi Krishnan 2 and Nils G. Walter 2 * 1 Biophysics and
Supplementary Data Viral RNAi suppressor reversibly binds sirna to outcompete Dicer and RISC via multiple-turnover Renata A. Rawlings 1,2, Vishalakshi Krishnan 2 and Nils G. Walter 2 * 1 Biophysics and
Supporting Information
 Supporting Information Su et al. 10.1073/pnas.1211604110 SI Materials and Methods Cell Culture and Plasmids. Tera-1 and Tera-2 cells (ATCC: HTB- 105/106) were maintained in McCoy s 5A medium with 15% FBS
Supporting Information Su et al. 10.1073/pnas.1211604110 SI Materials and Methods Cell Culture and Plasmids. Tera-1 and Tera-2 cells (ATCC: HTB- 105/106) were maintained in McCoy s 5A medium with 15% FBS
Institute of Physics, Academy of Sciences of the Czech Republic, v.v.i., Na Slovance 2, CZ , Prague 8, Czech Republic 2
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Theoretical and experimental study of antifreeze protein AFP752, trehalose and dimethyl sulfoxide
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Theoretical and experimental study of antifreeze protein AFP752, trehalose and dimethyl sulfoxide
Protocol for induction of expression and cell lysate production
 Protocol for induction of expression and cell lysate production AV-04 Doxycyclin induction and cell lysate 1.0 Introduction / Description This method is intended for the treatment of the previously transfected
Protocol for induction of expression and cell lysate production AV-04 Doxycyclin induction and cell lysate 1.0 Introduction / Description This method is intended for the treatment of the previously transfected
Supplemental Information
 Supplemental Information Intrinsic protein-protein interaction mediated and chaperonin assisted sequential assembly of a stable Bardet Biedl syndome protein complex, the BBSome * Qihong Zhang 1#, Dahai
Supplemental Information Intrinsic protein-protein interaction mediated and chaperonin assisted sequential assembly of a stable Bardet Biedl syndome protein complex, the BBSome * Qihong Zhang 1#, Dahai
Analysing protein protein interactions using a GST-fusion protein to pull down the interacting target from the cell lysate Hong Wang and Xin Zeng
 Analysing protein protein interactions using a GST-fusion protein to pull down the interacting target from the cell lysate Hong Wang and Xin Zeng Department of Molecular Genetics, Biochemistry and Microbiology,
Analysing protein protein interactions using a GST-fusion protein to pull down the interacting target from the cell lysate Hong Wang and Xin Zeng Department of Molecular Genetics, Biochemistry and Microbiology,
Four different active promoter genes were chosen, ATXN7L2, PSRC1, CELSR2 and
 SUPPLEMENTARY MATERIALS AND METHODS Chromatin Immunoprecipitation for qpcr analysis Four different active promoter genes were chosen, ATXN7L2, PSRC1, CELSR2 and IL24, all located on chromosome 1. Primer
SUPPLEMENTARY MATERIALS AND METHODS Chromatin Immunoprecipitation for qpcr analysis Four different active promoter genes were chosen, ATXN7L2, PSRC1, CELSR2 and IL24, all located on chromosome 1. Primer
SUPPLEMENTARY INFORMATION FIGURE 1 - 1
 SUPPLEMENTARY INFORMATION FIGURE 1-1 SUPPLEMENTARY INFORMATION FIGURE 2-2 SUPPLEMENTARY INFORMATION METHODS GST-Pull-Down. Cultures of E. Coli (BL21) were transformed with pgex (Clontech) and pgex recombinant
SUPPLEMENTARY INFORMATION FIGURE 1-1 SUPPLEMENTARY INFORMATION FIGURE 2-2 SUPPLEMENTARY INFORMATION METHODS GST-Pull-Down. Cultures of E. Coli (BL21) were transformed with pgex (Clontech) and pgex recombinant
Supplementary Figure 1 - Characterization of rbag3 binding on macrophages cell surface.
 Supplementary Figure 1 - Characterization of rbag3 binding on macrophages cell surface. (a) Human PDAC cell lines were treated as indicated in Figure 1 panel F. Cells were analyzed for FITC-rBAG3 binding
Supplementary Figure 1 - Characterization of rbag3 binding on macrophages cell surface. (a) Human PDAC cell lines were treated as indicated in Figure 1 panel F. Cells were analyzed for FITC-rBAG3 binding
Antibodies against PCNA were previously described [1]. To deplete PCNA from Xenopus egg
![Antibodies against PCNA were previously described [1]. To deplete PCNA from Xenopus egg Antibodies against PCNA were previously described [1]. To deplete PCNA from Xenopus egg](/thumbs/84/90687567.jpg) Supplementary information Supplementary methods PCNA antibody and immunodepletion Antibodies against PCNA were previously described [1]. To deplete PCNA from Xenopus egg extracts, one volume of protein
Supplementary information Supplementary methods PCNA antibody and immunodepletion Antibodies against PCNA were previously described [1]. To deplete PCNA from Xenopus egg extracts, one volume of protein
Index 1. Product Description 2. Purification Procedure 3. Troubleshooting 4. Ordering Information
 High Affinity Ni-Charged Resin Cat. No. L00223 Technical Manual No. TM0217 Version 07132010 Index 1. Product Description 2. Purification Procedure 3. Troubleshooting 4. Ordering Information 1. Product
High Affinity Ni-Charged Resin Cat. No. L00223 Technical Manual No. TM0217 Version 07132010 Index 1. Product Description 2. Purification Procedure 3. Troubleshooting 4. Ordering Information 1. Product
Comparison of different methods for purification analysis of a green fluorescent Strep-tag fusion protein. Application
 Comparison of different methods for purification analysis of a green fluorescent Strep-tag fusion protein Application Petra Sebastian Meike Kuschel Stefan Schmidt Abstract This Application Note describes
Comparison of different methods for purification analysis of a green fluorescent Strep-tag fusion protein Application Petra Sebastian Meike Kuschel Stefan Schmidt Abstract This Application Note describes
Glutathione Resin. (Cat. # , , , ) think proteins! think G-Biosciences
 191PR-05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Glutathione Resin (Cat. # 786-280, 786-310, 786-311, 786-312) think proteins! think
191PR-05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Glutathione Resin (Cat. # 786-280, 786-310, 786-311, 786-312) think proteins! think
Stabilization of a virus-like particle and its application as a nanoreactor at physiological conditions
 Supporting Information Stabilization of a virus-like particle and its application as a nanoreactor at physiological conditions Lise Schoonen, b Sjors Maassen, b Roeland J. M. Nolte b and Jan C. M. van
Supporting Information Stabilization of a virus-like particle and its application as a nanoreactor at physiological conditions Lise Schoonen, b Sjors Maassen, b Roeland J. M. Nolte b and Jan C. M. van
INSECT CELL/BACULOVIRUS PRODUCTION
 INSECT CELL/BACULOVIRUS PRODUCTION PEF # GENE NAME TRANSFER VECTOR BEVS MOLECULAR WEIGHT 2015-XXXX XXXX pbac1 flashbacultra TM 36.0 kda EXPRESSION METHOD OVERVIEW: Insect cells Spodoptera frugiperda (Sf9)
INSECT CELL/BACULOVIRUS PRODUCTION PEF # GENE NAME TRANSFER VECTOR BEVS MOLECULAR WEIGHT 2015-XXXX XXXX pbac1 flashbacultra TM 36.0 kda EXPRESSION METHOD OVERVIEW: Insect cells Spodoptera frugiperda (Sf9)
Gα i Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα i Activation Assay Kit Catalog Number 80301 20 assays NewEast Biosciences, Inc 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα i Activation Assay Kit Catalog Number 80301 20 assays NewEast Biosciences, Inc 1 Table of Content Product
Innovation Moléculaire et Thérapeutique, Université François Rabelais, Tours, France *For correspondence:
 Expression and Purification of the Eukaryotic MBP-MOS1 Transposase from sf21 Insect Cells Jérôme Jaillet, Audrey Dussaussois-Montagne, Sylvaine Renault and Corinne Augé-Gouillou * Innovation Moléculaire
Expression and Purification of the Eukaryotic MBP-MOS1 Transposase from sf21 Insect Cells Jérôme Jaillet, Audrey Dussaussois-Montagne, Sylvaine Renault and Corinne Augé-Gouillou * Innovation Moléculaire
Nickel Chelating Resin Spin Columns
 326PR-02 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Nickel Chelating Resin Spin Columns A Ni-IDA IMAC resin for 6X-His Tagged Protein
326PR-02 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Nickel Chelating Resin Spin Columns A Ni-IDA IMAC resin for 6X-His Tagged Protein
RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the
 Supplementary Methods RT-PCR and real-time PCR analysis RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the manufacturer s protocol and quantified by measuring the
Supplementary Methods RT-PCR and real-time PCR analysis RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the manufacturer s protocol and quantified by measuring the
GST Elution Buffer. (Cat. # ) think proteins! think G-Biosciences
 191PR-05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name GST Elution Buffer (Cat. #786-541) think proteins! think G-Biosciences www.gbiosciences.com
191PR-05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name GST Elution Buffer (Cat. #786-541) think proteins! think G-Biosciences www.gbiosciences.com
Supporting Information
 Supporting Information Ni/NiO Core/shell Nanoparticles for Selective Binding and Magnetic Separation of Histidine-Tagged Proteins In Su Lee,, No Hyun Lee,, Jongnam Park,, Byung Hyo Kim,, Yong-Weon Yi,
Supporting Information Ni/NiO Core/shell Nanoparticles for Selective Binding and Magnetic Separation of Histidine-Tagged Proteins In Su Lee,, No Hyun Lee,, Jongnam Park,, Byung Hyo Kim,, Yong-Weon Yi,
Ni-NTA Agarose. User Manual. 320 Harbor Way South San Francisco, CA Phone: 1 (888) MCLAB-88 Fax: 1 (650)
 Ni-NTA Agarose User Manual 320 Harbor Way South San Francisco, CA 94080 Phone: 1 (888) MCLAB-88 Fax: 1 (650) 871-8796 www. Contents Introduction -----------------------------------------------------------------------
Ni-NTA Agarose User Manual 320 Harbor Way South San Francisco, CA 94080 Phone: 1 (888) MCLAB-88 Fax: 1 (650) 871-8796 www. Contents Introduction -----------------------------------------------------------------------
A General Protocol for GST Pull-down Lili Jing *
 A General Protocol for GST Pull-down Lili Jing * Department of Cell and Molecular Biology, University of Pennsylvania, Philadelphia, USA *For correspondence: lilijingcn@gmail.com [Abstract] GST pull-down
A General Protocol for GST Pull-down Lili Jing * Department of Cell and Molecular Biology, University of Pennsylvania, Philadelphia, USA *For correspondence: lilijingcn@gmail.com [Abstract] GST pull-down
TECHNICAL BULLETIN. Ni-CAM HC Resin High Capacity Nickel Chelate Affinity Matrix. Product No. N 3158 Storage Temperature 2 8 C
 Ni-CAM HC Resin High Capacity Nickel Chelate Affinity Matrix Product No. N 3158 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Ni-CAM affinity resin (Ni-CAM) is an immobilized metal-ion
Ni-CAM HC Resin High Capacity Nickel Chelate Affinity Matrix Product No. N 3158 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Ni-CAM affinity resin (Ni-CAM) is an immobilized metal-ion
Fig. S1. CrgA intracellular levels in M. tuberculosis. Ten and twenty micrograms of
 Supplementary data Fig. S1. CrgA intracellular levels in M. tuberculosis. Ten and twenty micrograms of cell free protein lysates from WT M. tuberculosis (Rv) together with various known concentrations
Supplementary data Fig. S1. CrgA intracellular levels in M. tuberculosis. Ten and twenty micrograms of cell free protein lysates from WT M. tuberculosis (Rv) together with various known concentrations
His-Spin Protein Miniprep
 INSTRUCTIONS His-Spin Protein Miniprep Catalog No. P2001 (10 purifications) and P2002 (50 purifications). Highlights Fast 5 minute protocol to purify His-tagged proteins from cell-free extracts Screen
INSTRUCTIONS His-Spin Protein Miniprep Catalog No. P2001 (10 purifications) and P2002 (50 purifications). Highlights Fast 5 minute protocol to purify His-tagged proteins from cell-free extracts Screen
The YTH domain (residues ) of human YTHDF2 (NP_ ) was subcloned
 Supplementary information, Data S1 Materials and Methods Protein Expression, Purification and Crystallization The YTH domain (residues 383-553) of human YTHDF2 (NP_057342.2) was subcloned into a modified
Supplementary information, Data S1 Materials and Methods Protein Expression, Purification and Crystallization The YTH domain (residues 383-553) of human YTHDF2 (NP_057342.2) was subcloned into a modified
Nickel-NTA Agarose Suspension
 Nickel-NTA Agarose Suspension Agarose beads for purification of His-tagged proteins Product No. A9735 Description Nickel-NTA Agarose Suspension is an agarose-based affinity chromatography resin allowing
Nickel-NTA Agarose Suspension Agarose beads for purification of His-tagged proteins Product No. A9735 Description Nickel-NTA Agarose Suspension is an agarose-based affinity chromatography resin allowing
Supplementary data. sienigma. F-Enigma F-EnigmaSM. a-p53
 Supplementary data Supplemental Figure 1 A sienigma #2 sienigma sicontrol a-enigma - + ++ - - - - - - + ++ - - - - - - ++ B sienigma F-Enigma F-EnigmaSM a-flag HLK3 cells - - - + ++ + ++ - + - + + - -
Supplementary data Supplemental Figure 1 A sienigma #2 sienigma sicontrol a-enigma - + ++ - - - - - - + ++ - - - - - - ++ B sienigma F-Enigma F-EnigmaSM a-flag HLK3 cells - - - + ++ + ++ - + - + + - -
CD93 and dystroglycan cooperation in human endothelial cell adhesion and migration
 /, Supplementary Advance Publications Materials 2016 CD93 and dystroglycan cooperation in human endothelial cell adhesion and migration Supplementary Materials Supplementary Figure S1: In ECs CD93 silencing
/, Supplementary Advance Publications Materials 2016 CD93 and dystroglycan cooperation in human endothelial cell adhesion and migration Supplementary Materials Supplementary Figure S1: In ECs CD93 silencing
Supplementary methods Shoc2 In Vitro Ubiquitination Assay
 Supplementary methods Shoc2 In Vitro Ubiquitination Assay 35 S-labelled Shoc2 was prepared using a TNT quick Coupled transcription/ translation System (Promega) as recommended by manufacturer. For the
Supplementary methods Shoc2 In Vitro Ubiquitination Assay 35 S-labelled Shoc2 was prepared using a TNT quick Coupled transcription/ translation System (Promega) as recommended by manufacturer. For the
Protocol for in vitro transcription
 Protocol for in vitro transcription Assemble the reaction at room temperature in the following order: Component 10xTranscription Buffer rntp T7 RNA Polymerase Mix grna PCR DEPC H 2 O volume 2μl 2μl 2μl
Protocol for in vitro transcription Assemble the reaction at room temperature in the following order: Component 10xTranscription Buffer rntp T7 RNA Polymerase Mix grna PCR DEPC H 2 O volume 2μl 2μl 2μl
Cdc42 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
Supplementary Table 1. The Q-PCR primer sequence is summarized in the following table.
 Supplementary Table 1. The Q-PCR primer sequence is summarized in the following table. Name Sequence (5-3 ) Application Flag-u ggactacaaggacgacgatgac Shared upstream primer for all the amplifications of
Supplementary Table 1. The Q-PCR primer sequence is summarized in the following table. Name Sequence (5-3 ) Application Flag-u ggactacaaggacgacgatgac Shared upstream primer for all the amplifications of
AminTRAP HIS Prepacked Column
 INDEX Ordering Information... 3 Intended Use... 3 Product Description... 3 Purification Procedure... 4 Sample Preparation... 5 Sample Purification... 6 Analysis... 6 Regeneration Procedure... 6 Use and
INDEX Ordering Information... 3 Intended Use... 3 Product Description... 3 Purification Procedure... 4 Sample Preparation... 5 Sample Purification... 6 Analysis... 6 Regeneration Procedure... 6 Use and
Supplementary material for: Materials and Methods:
 Supplementary material for: Iron-responsive degradation of iron regulatory protein 1 does not require the Fe-S cluster: S.L. Clarke, et al. Materials and Methods: Fe-S Cluster Reconstitution: Cells treated
Supplementary material for: Iron-responsive degradation of iron regulatory protein 1 does not require the Fe-S cluster: S.L. Clarke, et al. Materials and Methods: Fe-S Cluster Reconstitution: Cells treated
Native mass spectrometry of recombinant proteins from crude cell lysates. Supporting Information
 Native mass spectrometry of recombinant proteins from crude cell lysates Jinrui Gan, Gili Ben-Nissan, Galina Arkind, Mark Tarnavsky, Devin Trudeau, Lianet Noda Garcia, Dan S. Tawfik and Michal Sharon Department
Native mass spectrometry of recombinant proteins from crude cell lysates Jinrui Gan, Gili Ben-Nissan, Galina Arkind, Mark Tarnavsky, Devin Trudeau, Lianet Noda Garcia, Dan S. Tawfik and Michal Sharon Department
Growth, Purification, and Characterization of P450 cam
 1. Cell growth without isotopic labeling Growth, Purification, and Characterization of P450 cam Growth medium Per liter (all components are previously sterilized by either autoclave or filtration): 5 M9
1. Cell growth without isotopic labeling Growth, Purification, and Characterization of P450 cam Growth medium Per liter (all components are previously sterilized by either autoclave or filtration): 5 M9
Coleman et al., Supplementary Figure 1
 Coleman et al., Supplementary Figure 1 BrdU Merge G1 Early S Mid S Supplementary Figure 1. Sequential destruction of CRL4 Cdt2 targets during the G1/S transition. HCT116 cells were synchronized by sequential
Coleman et al., Supplementary Figure 1 BrdU Merge G1 Early S Mid S Supplementary Figure 1. Sequential destruction of CRL4 Cdt2 targets during the G1/S transition. HCT116 cells were synchronized by sequential
INSTRUCTIONS The resins are adapted to work mainly in native conditions like denaturing.
 1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE Nickel NTA Agarose Beads DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 50% aqueous
1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE Nickel NTA Agarose Beads DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 50% aqueous
Introduction to Affinity Chromatography (AC) By: Sunsanee Yoojun Bang Trading 1992 Co., Ltd.
 Introduction to Affinity Chromatography (AC) By: Sunsanee Yoojun Bang Trading 1992 Co., Ltd. Content What is affinity chromatography (AC)? What is AC used for? Principles Experimental set up Summary What
Introduction to Affinity Chromatography (AC) By: Sunsanee Yoojun Bang Trading 1992 Co., Ltd. Content What is affinity chromatography (AC)? What is AC used for? Principles Experimental set up Summary What
Supporting Information
 Supporting Information Koh et al. 10.1073/pnas.1212917110 SI Materials and Methods Protein Purification. N-terminal His 6 -Dicer was purified as previously described with several modifications (1). After
Supporting Information Koh et al. 10.1073/pnas.1212917110 SI Materials and Methods Protein Purification. N-terminal His 6 -Dicer was purified as previously described with several modifications (1). After
SERVA Ni-NTA Magnetic Beads
 INSTRUCTION MANUAL SERVA Ni-NTA Magnetic Beads Magnetic beads for Affinity Purification of His-Tag Fusion Proteins (Cat. No. 42179) SERVA Electrophoresis GmbH - Carl-Benz-Str. 7-69115 Heidelberg Phone
INSTRUCTION MANUAL SERVA Ni-NTA Magnetic Beads Magnetic beads for Affinity Purification of His-Tag Fusion Proteins (Cat. No. 42179) SERVA Electrophoresis GmbH - Carl-Benz-Str. 7-69115 Heidelberg Phone
Supplemental material
 Supplemental material THE JOURNAL OF CELL BIOLOGY Taylor et al., http://www.jcb.org/cgi/content/full/jcb.201403021/dc1 Figure S1. Representative images of Cav 1a -YFP mutants with and without LMB treatment.
Supplemental material THE JOURNAL OF CELL BIOLOGY Taylor et al., http://www.jcb.org/cgi/content/full/jcb.201403021/dc1 Figure S1. Representative images of Cav 1a -YFP mutants with and without LMB treatment.
Ubiquitin (76 aa) UB genes encode linear fusions of UB either to itself (poly-ub genes) or to other proteins these fusions are cleaved by
 Ubiquitin (76 aa) UB genes encode linear fusions of UB either to itself (poly-ub genes) or to other proteins these fusions are cleaved by deubiquitylases (DUBs) yielding mature Ub deubiquitylase FLAG 3
Ubiquitin (76 aa) UB genes encode linear fusions of UB either to itself (poly-ub genes) or to other proteins these fusions are cleaved by deubiquitylases (DUBs) yielding mature Ub deubiquitylase FLAG 3
Supplemental Experimental Procedures
 Supplemental Experimental Procedures Generation of BLM protein segments and mutants. For immunoprecipitation experiments with topoisomerase IIα, BLM N-terminal segments were generated by PCR amplification
Supplemental Experimental Procedures Generation of BLM protein segments and mutants. For immunoprecipitation experiments with topoisomerase IIα, BLM N-terminal segments were generated by PCR amplification
Preparation of Pneumococcal Proteins for Western Blot Analysis Maria João Frias *, José Melo-Cristino and Mário Ramirez *
 Preparation of Pneumococcal Proteins for Western Blot Analysis Maria João Frias *, José Melo-Cristino and Mário Ramirez * Instituto de Microbiologia, Instituto de Medicina Molecular, Faculdade de Medicina,
Preparation of Pneumococcal Proteins for Western Blot Analysis Maria João Frias *, José Melo-Cristino and Mário Ramirez * Instituto de Microbiologia, Instituto de Medicina Molecular, Faculdade de Medicina,
ASPP1 Fw GGTTGGGAATCCACGTGTTG ASPP1 Rv GCCATATCTTGGAGCTCTGAGAG
 Supplemental Materials and Methods Plasmids: the following plasmids were used in the supplementary data: pwzl-myc- Lats2 (Aylon et al, 2006), pretrosuper-vector and pretrosuper-shp53 (generous gift of
Supplemental Materials and Methods Plasmids: the following plasmids were used in the supplementary data: pwzl-myc- Lats2 (Aylon et al, 2006), pretrosuper-vector and pretrosuper-shp53 (generous gift of
Purification of GST-tagged proteins using PureCube Glutathione Agarose
 Purification GST-tagged proteins using PureCube Glutathione Agarose Overview This protocol describes the generation a cleared lysate from 200 ml E. coli cell culture, and the purification GST-tagged proteins
Purification GST-tagged proteins using PureCube Glutathione Agarose Overview This protocol describes the generation a cleared lysate from 200 ml E. coli cell culture, and the purification GST-tagged proteins
OPPF-UK Standard Protocols: Mammalian Expression
 OPPF-UK Standard Protocols: Mammalian Expression Joanne Nettleship joanne@strubi.ox.ac.uk Table of Contents 1. Materials... 3 2. Cell Maintenance... 4 3. 24-Well Transient Expression Screen... 5 4. DNA
OPPF-UK Standard Protocols: Mammalian Expression Joanne Nettleship joanne@strubi.ox.ac.uk Table of Contents 1. Materials... 3 2. Cell Maintenance... 4 3. 24-Well Transient Expression Screen... 5 4. DNA
Ab SpinTrap Ab Buffer Kit
 GE Healthcare Data File 28-9020-30 AB Protein Sample Preparation Ab SpinTrap Ab Buffer Kit The Ab SpinTrap and the Protein G HP SpinTrap are identical columns. The difference between the article numbers
GE Healthcare Data File 28-9020-30 AB Protein Sample Preparation Ab SpinTrap Ab Buffer Kit The Ab SpinTrap and the Protein G HP SpinTrap are identical columns. The difference between the article numbers
Arf6 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Arf6 Activation Assay Kit Catalog Number: 82401 20 assays NewEast Biosciences 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Arf6 Activation Assay Kit Catalog Number: 82401 20 assays NewEast Biosciences 1 Table of Content Product
SUPPLEMENTARY INFORMATION
 Supplementary Methods Protein expression and in vitro binding studies. Recombinant baculovirus carrying GST, GST-mCC, GST-mSec, GST-mSecA and GST-mSecD were generated according to the manufacturer s instructions
Supplementary Methods Protein expression and in vitro binding studies. Recombinant baculovirus carrying GST, GST-mCC, GST-mSec, GST-mSecA and GST-mSecD were generated according to the manufacturer s instructions
Cobalt Chelating Resin
 078PR-05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Cobalt Chelating Resin A Co-IDA IMAC resin for 6X-His Tagged Protein Purification
078PR-05 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Cobalt Chelating Resin A Co-IDA IMAC resin for 6X-His Tagged Protein Purification
1. Cross-linking and cell harvesting
 ChIP is a powerful tool that allows the specific matching of proteins or histone modifications to regions of the genome. Chromatin is isolated and antibodies to the antigen of interest are used to determine
ChIP is a powerful tool that allows the specific matching of proteins or histone modifications to regions of the genome. Chromatin is isolated and antibodies to the antigen of interest are used to determine
Firefly luciferase mutants as sensors of proteome stress
 Nature Methods Firefly luciferase mutants as sensors of proteome stress Rajat Gupta, Prasad Kasturi, Andreas Bracher, Christian Loew, Min Zheng, Adriana Villella, Dan Garza, F Ulrich Hartl & Swasti Raychaudhuri
Nature Methods Firefly luciferase mutants as sensors of proteome stress Rajat Gupta, Prasad Kasturi, Andreas Bracher, Christian Loew, Min Zheng, Adriana Villella, Dan Garza, F Ulrich Hartl & Swasti Raychaudhuri
HOOK 6X His Protein Purification (Yeast)
 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Purification (Yeast) For The Purification of His Tagged Proteins from
G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Purification (Yeast) For The Purification of His Tagged Proteins from
Title: Direct and high throughput (HT) interactions on the ribosomal surface by iria
 Title: Direct and high throughput (HT) interactions on the ribosomal surface by iria Authors: Elisa Pesce 1, Claudia Minici 2, Jochen Baβler 3, Ed Hurt 3, Massimo Degano 2, Piera Calamita 1, and Stefano
Title: Direct and high throughput (HT) interactions on the ribosomal surface by iria Authors: Elisa Pesce 1, Claudia Minici 2, Jochen Baβler 3, Ed Hurt 3, Massimo Degano 2, Piera Calamita 1, and Stefano
HOOK 6X His Protein Purification (Bacteria)
 182PR-02 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Purification (Bacteria) For The Purification Of His-Tagged Proteins
182PR-02 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Purification (Bacteria) For The Purification Of His-Tagged Proteins
HOOK 6X His Protein Spin Purification (Bacteria)
 222PR 03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Spin Purification (Bacteria) For the Purification of His Tagged
222PR 03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Spin Purification (Bacteria) For the Purification of His Tagged
Solutions to 7.02 Quiz II 10/27/05
 Solutions to 7.02 Quiz II 10/27/05 Class Average = 83 Standard Deviation = 9 Range Grade % 87-100 A 43 74-86 B 39 55-73 C 17 > 54 D 1 Question 1 (56 points) While studying deep sea bacteria, you discover
Solutions to 7.02 Quiz II 10/27/05 Class Average = 83 Standard Deviation = 9 Range Grade % 87-100 A 43 74-86 B 39 55-73 C 17 > 54 D 1 Question 1 (56 points) While studying deep sea bacteria, you discover
A sensitive direct human telomerase activity assay Scott B Cohen & Roger R Reddel
 A sensitive direct human telomerase activity assay Scott B Cohen & Roger R Reddel Supplementary figure and text: Supplementary Figure 1 Titration of the sheep polyclonal htert antibody. Supplementary Methods
A sensitive direct human telomerase activity assay Scott B Cohen & Roger R Reddel Supplementary figure and text: Supplementary Figure 1 Titration of the sheep polyclonal htert antibody. Supplementary Methods
