Coaligned dual-channel STED nanoscopy and molecular diffusion analysis at 20 nm resolution
|
|
|
- Malcolm Evans
- 6 years ago
- Views:
Transcription
1 Supplementary Materials to Coaligned dual-channel STED nanoscopy and molecular diffusion analysis at 20 nm resolution Fabian Göttfert*, Christian A. Wurm*, Veronika Mueller*, Sebastian Berning*, Volker C. Cordes, Alf Honigmann* and Stefan W. Hell* *Department of NanoBiophotonics, Department of Cellular Logistics, Max Planck Institute for Biophysical Chemistry, Am Fassberg 11, Göttingen, Germany Address reprint requests and inquiries to SWH: Fabian Göttfert and Christian A. Wurm contributed equally to this work Determination of the absorption and emission spectra. The spectra were measured using a Varian Cary 4000 UV-VIS spectrophotometer and a Varian Cary Eclipse fluorescence spectrophotometer (Agilent Technologies, Santa Clara, CA, USA). Preparation of mammalian cell samples for confocal and STED microscopy. The cultivation of cells, antibody and immunolabelling was based on established protocols 1,2. Cultured Xenopus X177 and A6 cells were seeded on cover slips one day before fixation with formaldehyde (2.4%/ RT/ 5 min). After extraction in 0.3 % TritonX 100 in PBS and blocking in 5 % bovine serum albumin in PBS, the cells were incubated with a mouse monoclonal antibody specific for Xenopus gp210 3, novel rabbit antibodies selected for being pan-specific for most Xenopus FG repeat nucleoporins and especially targeting Nup214, Ganp, CG1, Nup98 und Nup153 in situ, and mouse monoclonal antibody QE5, targeting Nup153, Nup214 and Nup62 (Abcam, Cambridge, UK). The detection of these primary antibodies was performed using secondary antibodies (Dianova, Hamburg, Germany) custom labelled with the dyes Atto590, Atto594, Atto647N (Atto-Tec, Siegen, Germany), STAR580 and STAR635P (Abberior, Göttingen, Germany) and KK Finally, the samples were mounted in Mowiol containing DABCO. Preparation of samples for STED-FCS measurements. Glass supported lipid bilayers were prepared by spin-coating 20 µl of 2 g/l DOPC (1,2-dioleoyl-sn-glycero-3-phosphocholine, Avanti Polar Lipids) dissolved in 1 : 1 methanol/ chloroform containing ~ 0.01 mol % DSPE-PEG-KK114 onto piranha solution cleaned cover slides at 2000 rpm. After solvent evaporation under vacuum for 20 min, the bilayer was hydrated in 150 mm NaCl, 20 mm HEPES, ph 7.2 and rinsed several times until a single clean membrane remained (as determined by confocal imaging). The diffusion constant of fluorescent lipid analogues in defect-free glass supported membranes was D ~4 µm²/ s. The plasma membrane of adherent Ptk2 cells was labelled with DSPE-PEG-KK114 using bovine serum albumin (BSA) as lipid carrier. A lipid-bsa stock solution was prepared by dissolving 100 nmol of DSPE-PEG-KK114 in 10 µl ethanol and adding 1 ml of 7 g/ l fat free BSA (Sigma-Aldrich). The cells were incubated with a 1:20 dilution of the stock solution in growth medium (HDMEM) for 20 min at room temperature and then briefly washed with medium. 1
2 STED and confocal imaging. STED and the corresponding confocal microscopy were carried out by piezostage scanning (dwell times: 30 µs; pixel size: 5.9 nm) (Nano-PDQ, Mad City Labs, Madison, WI, USA) (Fig. S1). Excitation of the fluorophores was performed with two pulsed diode lasers emitting 70 ps pulses, one at 595 nm and the other one at 640 nm (both from PicoQuant, Berlin, Germany). To clean the wavefronts, the excitation beams were coupled into single mode polarization maintaining fibers. STED was performed using a frequency-doubled fiber laser (ELP DG, IPG Photonics Corporation, Oxford, MA, USA) emitting 1.2 ns-pulses at 775 nm and 20 MHz repetition rate with a maximum average power of 5 W. [Lasers with similar pulse specifications are also available from MPB Communications Inc (Montreal, Quebec, Canada) and OneFive GmbH (Zürich, Switzerland).] The average power of the STED beam in the focal plane was mw, corresponding to mw at the back aperture of the objective lens. The same STED beam was used for inhibiting fluorescence from both color channels (Fig. S1B). Triggering and synchronisation of the excitation pulses to the STED pulses was performed with a photodiode connected to a constant fraction discriminator (CFD) positioned to intercept a small reflection of the STED beam (before coupling the STED beam into a 30 m fiber) and custom-built electronics. Since the pulses of our STED laser exhibit temporal jitter, it is important to trigger each excitation pulse to the corresponding STED pulse. (This may not apply to lasers by other manufacturers due to improved pulseto-pulse stability.) In the pulse-interleaved excitation scheme, the signals from the CFD are electronically distributed to the excitation diodes, so that only every second STED pulse triggers the same excitation diode (Fig. S1C). While the coarse pulse timing of the STED and excitation lasers is set by the lengths of the fibers, the beam paths and the cables, the fine tuning is realized by electronic delay. After the fiber, the STED beam passes a polymeric phase plate (Vortex pattern, RPC Photonics, Rochester, NY, USA). In combination with a λ/4 waveplate, the phase plate is responsible for the doughnut shaped STED-focus. The excitation and the STED beams were co-aligned and coupled into a 1.4 numerical aperture oil immersion lens (NA 1.4 HCX PL APO, 100x, Leica Microsystems, Wetzlar, Germany). The fluorescence was collected by the same lens, spectrally separated and filtered into two ranges: nm and nm. The fluorescence was detected by fiber-coupled single-photon-counting modules (SPCM- AQRH13, Perkin Elmer), with their fiber cores acting as confocal pinholes. The signal from each detection channel is then passed through a home-built electronic gating unit that is triggered by the same (delayed) pulses as the excitation lasers. The time gating pipes the signal within a 10 ns window to a data acquisition card (NI PCI-6259, National Instruments, Austin, Texas, USA) (Fig. S1C). The time gating not only limits the detected fluorescence of each channel to the corresponding excitation pulse, but also suppresses the detection of signals in the first nanosecond after the excitation pulse, reducing the detected fluorescence signal to 70 % for the applied dyes ( F 3.5 ns). The loss in signal is compensated by the increased resolution as spontaneously emitted photons during the action of the STED pulse are neglected 5. [As an alternative to home-built time gating electronics we recommend time correlated single photon counting modules (e.g. by PicoQuant or Becker & Hickl, Berlin, Germany) which offer the possibility of adjusting the time window in post processing. An implementation for National Instruments 2
3 data acquisition cards with FPGA chip is an alternative option.] For better visibility, the images shown in Fig. 1, Fig. 2, and Suppl. Fig. 3 a, b were smoothed by convolution with a 2D Gaussian function (σ = 1 pixel and σ = 4 pixel for Fig. 2c). Note that stage scanning has been used here for convenience only. Coupling a (commercially available) beam-scanning device between the objective lens and the light sources, which is technically straightforward, reduces both the recording time and bleaching further. STED-FCS measurements and data analysis The 640 nm excitation laser was precisely focused into the membrane by the z-piezo-actuator of the stage. The optimal z-position was found by observing the fluorescence intensity in real-time and maximizing the amplitude of the single molecule fluctuations. The fluctuating intensities were autocorrelated with a hardware correlator card (Flex02-01D, Correlator.com, NJ), with typical measurement times of s. The measurement was repeated for increasing STED power. The resulting correlation curves were fitted using MATLAB according to a two-dimensional anomalous diffusion model 6 : G( ) 1/ N * (1 ( T (1 T )) * exp( / )) *1/(1 ( / ) ) T D where the particle number N denotes the average number of fluorescent molecules in the detection volume, T the fraction of molecules that are on average in the dark triplet state, T the triplet dwell time, D the average transit time of the molecules diffusing through the observation area, and the anomalous diffusion exponent, which is 1 for unhindered diffusion and <1 for anomalous hindered diffusion. Previous analysis 7,8 of STED-FCS curves in cell membranes have shown that anomaly exponents are < 1 in most cases. However, significant hindered diffusion with a clear non-linear relation of diffusion time vs. detection area is indicated by α < 0.7. In the case of free diffusion, the recorded diffusion time D is proportional to the observation area. Thus the FWHM of the observation area (corresponding to the resolution of the STED microscope) can be extracted for each STED power by: FWHM * /. Here, FWHM Conf D Conf Conf is given by the average diffusion time in a confocal recording, whereas the confocal FWHM conf is precisely gained by imaging 20 nm large crimson fluorescent beads (Life Technologies, Paisley, UK) in the confocal mode. For this purpose the beads are immobilized on a coverslip, treated with poly-l-lysine (0.1 % in water, Sigma-Aldrich, Steinheim, Germany) and embedded in Mowiol. Note that our measurement confirms that the STED microscopy resolution scales as FWHM FWHM 1 P / P conf STED S. The dye- and wavelength specific threshold power is determined by fitting to P s = 1.4 mw. 3
4 SUPPORTING REFERENCES 1. Hase, M. E., N. V. Kuznetsov and V. C. Cordes Amino acid substitutions of coiled-coil protein Tpr abrogate anchorage to the nuclear pore complex but not parallel, in-register homodimerization. Mol. Biol. Cell 12, Wurm, C. A., D. Neumann,, S. Jakobs Sample preparation for STED microscopy. Methods Mol. Biol. 591, Cordes, V. C., A. Gajewski, S. Stumpp and G. Krohne Immunocytochemistry of annulate lamellae: potential cell biological markers for studies of cell differentiation and pathology. Differentiation 58, Wurm, C. A., K. Kolmakov,, S. W. Hell Novel red fluorophores with superior performance in STED microscopy. Opt. Nano. 1, Moffitt, J. R., C. Osseforth and J. Michaelis Time-gating improves the spatial resolution of STED microscopy. Opt. Express 19, Schwille, P., F. J. MeyerAlmes and R. Rigler Dual-color fluorescence cross-correlation spectroscopy for multicomponent diffusional analysis in solution. Biophys. J. 72, Eggeling, C., C. Ringemann,..., S. W. Hell Direct observation of the nanoscale dynamics of membrane lipids in a living cell. Nature. 457:1159-U Mueller V, C. Ringemann,, C. Eggeling STED Nanoscopy Reveals Molecular Details of Cholesterol- and Cytoskeleton- Modulated Lipid Interactions in Living Cells. Biophys. J. 101,
5 FIGURE S1 STED setup and spectral rationale for the used fluorophores. (A) The setup uses two pulsed laser diodes (595 nm and 640 nm) for excitation and a single compact fiber laser for STED. Fluorescence is detected in two distinct wavelength ranges: 620±20 nm and 670±20 nm. The use of a single STED doughnut at 775 nm for both dyes and interleaved pulsed excitation and detection ensures nearly perfect colocalization accuracy, because the doughnut minimum predetermines the coordinate of both emitting dyes. Dichroic mirrors: DM1: z760sprdc; DM2: z585rdc; DM3: z635rdc; Notch: stop line 658 nm (Chroma, Bellows Falls, VT & Semrock, Rochester, NY). The mirrors were tuned in angle to match the laser lines. (B) The spectra of the used dyes (Atto594 and KK114) are closely overlapping. Yet the dyes can be well separated due to the distinct excitation wavelengths and the selection of the detection channels. Note that detection sensitivity can be further enhanced by replacing the 670±20 nm emission filter2 with a 650 nm long pass filter extending up to 775 nm. Detection is accomplished with avalanche photodiodes (APD). (C) Interleaved pulsed excitation and detection ensures temporal succession of the two excitation pulses in the nanosecond range, thus eliminating drift between the recordings of the two color channels, which is important when recording protein colocalization. For further details see the Supplementary Methods and Materials section. 5
6 FIGURE S2 Spectral crosstalk. Xenopus A6 cells were labeled with primary antibodies against NUP subunits in the central channel of the nuclear pore complex (NUP153, NUP214, and NUP62); fluorophore tagged secondary antibodies were appended as indicated. The crosstalk for both channels was measured under conditions similar to the recordings of Fig. 1. The conditions were kept constant for the individual recordings. The images show that crosstalk is negligible. Therefore, no data postprocessing is necessary to separate the data recorded by the two color channels. 6
7 FIGURE S3 Colocalization accuracy. To analyze the colocalization accuracy of the setup, Xenopus A6 cells were labeled with an antiserum detecting multiple NUP subunits in the central channel of the nuclear pore complex (NUP153, NUP214, and NUP62). This antiserum was in turn detected by two secondary antibody species tagged with Abberior STAR635P and Atto594, respectively. STED microscopy was performed as for the other samples. (A) Already upon overlaying the individual channels, a high degree of colocalization of the signals is visible. (B) To quantify the colocalization accuracy, we cross correlated larger images of the two channels. The correlation is maximal for no displacement of the images, a Gaussian fit confirms that the mismatch of the channels is less than 3 nm. To further demonstrate the tolerance of the colocalization accuracy to misalignment we imaged 20 nm fluorescent beads (Crimson FluoSpheres, Life Technologies) with two misaligned excitation beams (detecting at 670±20 nm). The displacement is obvious in the confocal image (C) while the STED image shows good colocalization (D). The cross correlation of the two channels shows a shift of approximately 100 nm for the confocal image and subpixel accuracy in the STED-image. Scale bars, 200 nm (A), 1000 nm (B-D) 7
8 FIGURE S4 STED imaging resolution. The resolution was determined by measuring the size of the smallest discernible features (A) as an upper bound for the resolution. (B) Line profiles across these features were extracted and fitted by Gaussians. (C) The histograms indicate a resolution of ~20 nm and ~30 nm in the raw data provided by the 670±20 nm and 620±20 nm channels respectively. 8
9 FIGURE S5 Dual color STED overview image. Overview image (smoothed raw data) of about half a nucleus labeled with the indicated antibodies. The STED image shown in Fig. 1 is an excerpt (boxed) of this overview. 9
10 Figure S6 Dual color confocal overview image. Overview image (smoothed raw data) of about half a nucleus labeled with the indicated antibodies. The confocal image shown in Fig. 1 is an excerpt (boxed) of this overview. 10
11 FIGURE S7 Anomaly exponents from FCS analysis in model and cell membranes. The anomaly exponent α from the FCS analysis presented in figure 3 A, B. In model membranes no anomaly was detected (α ~ 1). In the plasma membrane a weak anomaly was detected (α > 0.85). However, in combination with the linear relation of transit time versus detection area we conclude that diffusion of the lipid analogue was mainly free. 11
Immunofluorescence and phalloidin labeling of mammalian cells
 Immunofluorescence and phalloidin labeling of mammalian cells 2 Contents Materials for immunofluorescence and phalloidin labeling of mammalian cells...1 Immunofluorescence-labelling on cultivated adherent
Immunofluorescence and phalloidin labeling of mammalian cells 2 Contents Materials for immunofluorescence and phalloidin labeling of mammalian cells...1 Immunofluorescence-labelling on cultivated adherent
Supplementary Table 1. Components of an FCS setup (1PE and 2PE)
 Supplementary Table 1. Components of an FCS setup (1PE and 2PE) Component and function Laser source Excitation of fluorophores Microscope with xy-translation stage mounted on vibration isolated optical
Supplementary Table 1. Components of an FCS setup (1PE and 2PE) Component and function Laser source Excitation of fluorophores Microscope with xy-translation stage mounted on vibration isolated optical
MicroTime 200 STED. Super-resolution add-on for the confocal time-resolved microscopy platform
 MicroTime 200 STED Super-resolution add-on for the confocal time-resolved microscopy platform confocal STED 2 Vision The MicroTime 200... The MicroTime 200 is a high-end confocal fluorescence lifetime
MicroTime 200 STED Super-resolution add-on for the confocal time-resolved microscopy platform confocal STED 2 Vision The MicroTime 200... The MicroTime 200 is a high-end confocal fluorescence lifetime
STED microscopy with single light source. TeodoraŞcheul
 STED microscopy with single light source TeodoraŞcheul Dr. Iréne Wang, Dr. Jean-Claude Vial LIPhy, Grenoble, France Summary I. Introduction to STED microscopy II. STED with one laser source 1. Two-photon
STED microscopy with single light source TeodoraŞcheul Dr. Iréne Wang, Dr. Jean-Claude Vial LIPhy, Grenoble, France Summary I. Introduction to STED microscopy II. STED with one laser source 1. Two-photon
Performance of the Micro Photon Devices PDM 50CT SPAD detector with PicoQuant TCSPC systems
 Technical Note Performance of the Micro Photon Devices PDM 5CT SPAD detector with PicoQuant TCSPC systems Rolf Krahl, Andreas Bülter, Felix Koberling, PicoQuant GmbH These measurements were performed to
Technical Note Performance of the Micro Photon Devices PDM 5CT SPAD detector with PicoQuant TCSPC systems Rolf Krahl, Andreas Bülter, Felix Koberling, PicoQuant GmbH These measurements were performed to
UV MicroTime 200. Crossing the Border towards Deep UV Time-resolved Microscopy of Native Fluorophores
 UV MicroTime 200 Crossing the Border towards Deep UV Time-resolved Microscopy of Native Fluorophores Marcelle König 1, Sebastian Tannert 1, Sandra Orthaus 1, Volker Buschmann 1, Thomas Schönau 1, Kristian
UV MicroTime 200 Crossing the Border towards Deep UV Time-resolved Microscopy of Native Fluorophores Marcelle König 1, Sebastian Tannert 1, Sandra Orthaus 1, Volker Buschmann 1, Thomas Schönau 1, Kristian
Super Resolution Microscopy - Breaking the Diffraction Limit Radiological Research Accelerator Facility
 Super Resolution Microscopy - Breaking the Diffraction Limit Radiological Research Accelerator Facility Sabrina Campelo, Dr. Andrew Harken Outline Motivation Fluorescence Microscopy -Multiphoton Imaging
Super Resolution Microscopy - Breaking the Diffraction Limit Radiological Research Accelerator Facility Sabrina Campelo, Dr. Andrew Harken Outline Motivation Fluorescence Microscopy -Multiphoton Imaging
Nonspecific binding of 10 nm Cy5-labeled DinB on nine different surfaces, measured by the number of DinB spots over an imaging area of 2,500 µm 2.
 Supplementary Figure 1 Nonspecific binding of 10 nm Cy5-labeled DinB on nine different surfaces, measured by the number of DinB spots over an imaging area of 2,500 µm 2. Free DinB was washed out using
Supplementary Figure 1 Nonspecific binding of 10 nm Cy5-labeled DinB on nine different surfaces, measured by the number of DinB spots over an imaging area of 2,500 µm 2. Free DinB was washed out using
Supporting Information
 Supporting Information Understanding the Photophysics of the Spinach-DFHBI RNA Aptamer-Fluorogen Complex to Improve Live Cell RNA Imaging Kyu Young Han, Benjamin J. Leslie, Jingyi Fei, Jichuan Zhang, Taekjip
Supporting Information Understanding the Photophysics of the Spinach-DFHBI RNA Aptamer-Fluorogen Complex to Improve Live Cell RNA Imaging Kyu Young Han, Benjamin J. Leslie, Jingyi Fei, Jichuan Zhang, Taekjip
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1. Comparison of background levels of single-molecule measurement. (a-b) Typical levels of background signals of buffer solution at the power of 1 mw for 532 nm
Supplementary Figures Supplementary Fig. 1. Comparison of background levels of single-molecule measurement. (a-b) Typical levels of background signals of buffer solution at the power of 1 mw for 532 nm
Sample region with fluorescent labeled molecules
 FLUORESCENCE IMAGING I. Fluorescence-imaging with diffraction limited spots The resolution in optical microscopy has been hampered by the smallest spot possible (~ λ/2) that can be achieved by conventional
FLUORESCENCE IMAGING I. Fluorescence-imaging with diffraction limited spots The resolution in optical microscopy has been hampered by the smallest spot possible (~ λ/2) that can be achieved by conventional
QUICK GUIDE TO STED SAMPLE PREPARATION
 Page 1 13 Author Wernher Fouquet Date 2014-01-23 Quick Guide to STED Sample Preparation This document provides you with information about sample preparation for STED microscopy. It contains some tips and
Page 1 13 Author Wernher Fouquet Date 2014-01-23 Quick Guide to STED Sample Preparation This document provides you with information about sample preparation for STED microscopy. It contains some tips and
F* techniques: FRAP, FLIP, FRET, FLIM,
 F* techniques: FRAP, FLIP, FRET, FLIM, FCS Antonia Göhler March 2015 Fluorescence explained in the Bohr model Absorption of light (blue) causes an electron to move to a higher energy orbit. After a particular
F* techniques: FRAP, FLIP, FRET, FLIM, FCS Antonia Göhler March 2015 Fluorescence explained in the Bohr model Absorption of light (blue) causes an electron to move to a higher energy orbit. After a particular
Simultaneous multi-color, multiphoton fluorophore excitation using dual-color fiber lasers
 Multiphoton Microscopy / Fiber Laser Simultaneous multi-color, multiphoton fluorophore excitation using dual-color fiber lasers Matthias Handloser, Tim Paasch-Colberg, Bernhard Wolfring TOPTICA Photonics
Multiphoton Microscopy / Fiber Laser Simultaneous multi-color, multiphoton fluorophore excitation using dual-color fiber lasers Matthias Handloser, Tim Paasch-Colberg, Bernhard Wolfring TOPTICA Photonics
SUPPLEMENTARY INFORMATION
 Biosynthesis of Luminescent Quantum Dots in an Earthworm S.R. Stürzenbaum, a# M. Hoeckner, a# A. Panneerselvam, b J. Levitt, b J.-S. Bouillard, b S. Taniguchi, b L.-A. Dailey, d R. Ahmad Khanbeigi, d E.
Biosynthesis of Luminescent Quantum Dots in an Earthworm S.R. Stürzenbaum, a# M. Hoeckner, a# A. Panneerselvam, b J. Levitt, b J.-S. Bouillard, b S. Taniguchi, b L.-A. Dailey, d R. Ahmad Khanbeigi, d E.
SUPPLEMENTARY INFORMATION
 DOI: 1.138/NNANO.211.185 Mixing sub-attolitre volumes in a quantitative and highly parallel manner with soft matter nanofluidics Sune M. Christensen, Pierre-Yves Bolinger,Nikos S. Hatzakis, Michael W.Mortensen
DOI: 1.138/NNANO.211.185 Mixing sub-attolitre volumes in a quantitative and highly parallel manner with soft matter nanofluidics Sune M. Christensen, Pierre-Yves Bolinger,Nikos S. Hatzakis, Michael W.Mortensen
Supplementary Information. for
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supplementary Information for Nanoslitting Phase-separated Block Copolymers by Solvent Swelling
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supplementary Information for Nanoslitting Phase-separated Block Copolymers by Solvent Swelling
FLIM Fluorescence Lifetime IMaging
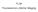 FLIM Fluorescence Lifetime IMaging Fluorescence lifetime t I(t) = F0 exp( ) τ 1 τ = k f + k nr k nr = k IC + k ISC + k bl Batiaens et al, Trends in Cell Biology, 1999 τ τ = fluorescence lifetime (~ns to
FLIM Fluorescence Lifetime IMaging Fluorescence lifetime t I(t) = F0 exp( ) τ 1 τ = k f + k nr k nr = k IC + k ISC + k bl Batiaens et al, Trends in Cell Biology, 1999 τ τ = fluorescence lifetime (~ns to
Multiplexed 3D FRET imaging in deep tissue of live embryos Ming Zhao, Xiaoyang Wan, Yu Li, Weibin Zhou and Leilei Peng
 Scientific Reports Multiplexed 3D FRET imaging in deep tissue of live embryos Ming Zhao, Xiaoyang Wan, Yu Li, Weibin Zhou and Leilei Peng 1 Supplementary figures and notes Supplementary Figure S1 Volumetric
Scientific Reports Multiplexed 3D FRET imaging in deep tissue of live embryos Ming Zhao, Xiaoyang Wan, Yu Li, Weibin Zhou and Leilei Peng 1 Supplementary figures and notes Supplementary Figure S1 Volumetric
Department of Chemistry, Center for Photochemical Sciences, Bowling Green State University,
 Supporting Information for Revealing Abrupt and Spontaneous Ruptures of Protein Native Structure under PicoNewton Compressive Force Manipulation S. Roy Chowdhury, Jin Cao, Yufan He, H. Peter Lu * Department
Supporting Information for Revealing Abrupt and Spontaneous Ruptures of Protein Native Structure under PicoNewton Compressive Force Manipulation S. Roy Chowdhury, Jin Cao, Yufan He, H. Peter Lu * Department
Supporting Information for. Electrical control of Förster energy transfer.
 1 Supporting Information for Electrical control of Förster energy transfer. Klaus Becker 1, John M. Lupton 1*, Josef Müller 1, Andrey. L. Rogach 1, Dmitri V. Talapin, Horst Weller & Jochen Feldmann 1 1
1 Supporting Information for Electrical control of Förster energy transfer. Klaus Becker 1, John M. Lupton 1*, Josef Müller 1, Andrey. L. Rogach 1, Dmitri V. Talapin, Horst Weller & Jochen Feldmann 1 1
New developments in STED Microscopy
 New developments in STED Microscopy Arnold Giske*, Jochen Sieber, Hilmar Gugel, Marcus Dyba, Volker Seyfried, Dietmar Gnass Leica Microsystems CMS, Am Friedensplatz 3, 68126 Mannheim, Germany ABSTRACT
New developments in STED Microscopy Arnold Giske*, Jochen Sieber, Hilmar Gugel, Marcus Dyba, Volker Seyfried, Dietmar Gnass Leica Microsystems CMS, Am Friedensplatz 3, 68126 Mannheim, Germany ABSTRACT
Lab 5: Optical trapping and single molecule fluorescence
 Lab 5: Optical trapping and single molecule fluorescence PI: Matt Lang Lab Instructor: Jorge Ferrer Summary Optical tweezers are an excellent experimental tool to study the biophysics of single molecule
Lab 5: Optical trapping and single molecule fluorescence PI: Matt Lang Lab Instructor: Jorge Ferrer Summary Optical tweezers are an excellent experimental tool to study the biophysics of single molecule
Biosensis P1TM Polar Lipid Tracing Reagent. Catalogue Number: TR-600-P1. For research use only, not for use in clinical and diagnostic procedures.
 Biosensis P1TM Polar Lipid Tracing Reagent Catalogue Number: TR-600-P1 For research use only, not for use in clinical and diagnostic procedures. Version: TR-600-P1/v2/Sep2018 Biosensis Pty Ltd., 51 West
Biosensis P1TM Polar Lipid Tracing Reagent Catalogue Number: TR-600-P1 For research use only, not for use in clinical and diagnostic procedures. Version: TR-600-P1/v2/Sep2018 Biosensis Pty Ltd., 51 West
Supplementary Figure 1
 Supplementary Figure 1 (A) Schematic of sequential hybridization and barcoding. (B) Schematic of the FISH images of the cell. In each round of hybridization, the same spots are detected, but the dye associated
Supplementary Figure 1 (A) Schematic of sequential hybridization and barcoding. (B) Schematic of the FISH images of the cell. In each round of hybridization, the same spots are detected, but the dye associated
Immunofluorescence Confocal Microscopy of 3D Cultures Grown on Alvetex
 Immunofluorescence Confocal Microscopy of 3D Cultures Grown on Alvetex 1.0. Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigen-specific antibodies coupled
Immunofluorescence Confocal Microscopy of 3D Cultures Grown on Alvetex 1.0. Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigen-specific antibodies coupled
Confocal Microscopy of Electronic Devices. James Saczuk. Consumer Optical Electronics EE594 02/22/2000
 Confocal Microscopy of Electronic Devices James Saczuk Consumer Optical Electronics EE594 02/22/2000 Introduction! Review of confocal principles! Why is CM used to examine electronics?! Several methods
Confocal Microscopy of Electronic Devices James Saczuk Consumer Optical Electronics EE594 02/22/2000 Introduction! Review of confocal principles! Why is CM used to examine electronics?! Several methods
Supporting Information. A Fluorogenic Resveratrol-Confined Graphene Oxide For Economic and Rapid. Detection Of Alzheimer's Disease
 Supporting Information A Fluorogenic Resveratrol-Confined Graphene Oxide For Economic and Rapid Detection Of Alzheimer's Disease Xiao-Peng He, 1 Qiong Deng, 1 Liang Cai, 1 Chang-Zheng Wang, 1,2 Yi Zang,*,2
Supporting Information A Fluorogenic Resveratrol-Confined Graphene Oxide For Economic and Rapid Detection Of Alzheimer's Disease Xiao-Peng He, 1 Qiong Deng, 1 Liang Cai, 1 Chang-Zheng Wang, 1,2 Yi Zang,*,2
PALM/STORM, BALM, STED
 PALM/STORM, BALM, STED Last class 2-photon Intro to PALM/STORM Cyanine dyes/dronpa This class Finish localization super-res BALM STED Localization microscopy Intensity Bins = pixels xx 2 = ss2 + aa 2 /12
PALM/STORM, BALM, STED Last class 2-photon Intro to PALM/STORM Cyanine dyes/dronpa This class Finish localization super-res BALM STED Localization microscopy Intensity Bins = pixels xx 2 = ss2 + aa 2 /12
ab CytoPainter ER Staining Kit Red Fluorescence
 ab139482 CytoPainter ER Staining Kit Red Fluorescence Instructions for Use Designed to detect Human endoplasmic reticulum by microscopy. This product is for research use only and is not intended for diagnostic
ab139482 CytoPainter ER Staining Kit Red Fluorescence Instructions for Use Designed to detect Human endoplasmic reticulum by microscopy. This product is for research use only and is not intended for diagnostic
CytoPainter Golgi Staining Kit Green Fluorescence
 ab139483 CytoPainter Golgi Staining Kit Green Fluorescence Instructions for Use Designed for the detection of Golgi bodies by microscopy This product is for research use only and is not intended for diagnostic
ab139483 CytoPainter Golgi Staining Kit Green Fluorescence Instructions for Use Designed for the detection of Golgi bodies by microscopy This product is for research use only and is not intended for diagnostic
Fluorescence Nanoscopy
 Fluorescence Nanoscopy Keith A. Lidke University of New Mexico panda3.phys.unm.edu/~klidke/index.html Optical Microscopy http://en.wikipedia.org/wiki/k%c3%b6hler_illumination 30 µm Fluorescent Probes Michalet
Fluorescence Nanoscopy Keith A. Lidke University of New Mexico panda3.phys.unm.edu/~klidke/index.html Optical Microscopy http://en.wikipedia.org/wiki/k%c3%b6hler_illumination 30 µm Fluorescent Probes Michalet
Fluorescence Light Microscopy for Cell Biology
 Fluorescence Light Microscopy for Cell Biology Why use light microscopy? Traditional questions that light microscopy has addressed: Structure within a cell Locations of specific molecules within a cell
Fluorescence Light Microscopy for Cell Biology Why use light microscopy? Traditional questions that light microscopy has addressed: Structure within a cell Locations of specific molecules within a cell
2-step or indirect immunofluorescence 1. Substrate on which cells are plated: plastic vs. glass; coating vs. non
 Variables in standard immunostaining protocol 2-step or indirect immunofluorescence 1. Substrate on which cells are plated: plastic vs. glass; coating vs. non 2. Plating density: sparse vs. confluent 3.
Variables in standard immunostaining protocol 2-step or indirect immunofluorescence 1. Substrate on which cells are plated: plastic vs. glass; coating vs. non 2. Plating density: sparse vs. confluent 3.
Novel red fluorophores with superior performance in STED microscopy
 Wurm et al. Optical Nanoscopy 2012, 1:7 ORIGINAL ARTICLE Novel red fluorophores with superior performance in STED microscopy Christian A Wurm 1,2*, Kirill Kolmakov 1, Fabian Göttfert 1, Haisen Ta 1, Mariano
Wurm et al. Optical Nanoscopy 2012, 1:7 ORIGINAL ARTICLE Novel red fluorophores with superior performance in STED microscopy Christian A Wurm 1,2*, Kirill Kolmakov 1, Fabian Göttfert 1, Haisen Ta 1, Mariano
Microscopy from Carl Zeiss
 Microscopy from Carl Zeiss LSM 710 In Tune with Your Application Enjoy new freedom in selecting fluorescent dyes with In Tune, the new laser system for the LSM 710. Whatever the wavelength, you can match
Microscopy from Carl Zeiss LSM 710 In Tune with Your Application Enjoy new freedom in selecting fluorescent dyes with In Tune, the new laser system for the LSM 710. Whatever the wavelength, you can match
Optical Observation - Hyperspectral Characterization of Nano-scale Materials In-situ
 Optical Observation - Hyperspectral Characterization of Nano-scale Materials In-situ Research at the nanoscale is more effective, when research teams can quickly and easily observe and characterize a wide
Optical Observation - Hyperspectral Characterization of Nano-scale Materials In-situ Research at the nanoscale is more effective, when research teams can quickly and easily observe and characterize a wide
AnaTag HiLyte Fluor 750 Microscale Protein Labeling Kit
 AnaTag HiLyte Fluor 750 Microscale Protein Labeling Kit Catalog # 72044 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor 750 SE to proteins (e.g., IgG). It provides ample
AnaTag HiLyte Fluor 750 Microscale Protein Labeling Kit Catalog # 72044 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor 750 SE to proteins (e.g., IgG). It provides ample
Fast, three-dimensional super-resolution imaging of live cells
 Nature Methods Fast, three-dimensional super-resolution imaging of live cells Sara A Jones, Sang-Hee Shim, Jiang He & Xiaowei Zhuang Supplementary Figure 1 Supplementary Figure 2 Supplementary Figure 3
Nature Methods Fast, three-dimensional super-resolution imaging of live cells Sara A Jones, Sang-Hee Shim, Jiang He & Xiaowei Zhuang Supplementary Figure 1 Supplementary Figure 2 Supplementary Figure 3
Highly GSH-sensitive SN-38 prodrug with OFF-to-ON fluorescence switch as a bifunctional anticancer agent
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Highly GSH-sensitive SN-38 prodrug with
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Highly GSH-sensitive SN-38 prodrug with
Supporting Information. Two-Photon Luminescence of Single Colloidal Gold NanoRods: Revealing the Origin of Plasmon Relaxation in Small Nanocrystals
 Supporting Information Two-Photon Luminescence of Single Colloidal Gold NanoRods: Revealing the Origin of Plasmon Relaxation in Small Nanocrystals Céline Molinaro 1, Yara El Harfouch 1, Etienne Palleau
Supporting Information Two-Photon Luminescence of Single Colloidal Gold NanoRods: Revealing the Origin of Plasmon Relaxation in Small Nanocrystals Céline Molinaro 1, Yara El Harfouch 1, Etienne Palleau
Introduction to N-STORM
 Introduction to N-STORM Dan Metcalf Advanced Imaging Manager Outline Introduction Principles of STORM Applications N-STORM overview Biological Scale Mitochondrion Microtubule Amino Acid 1Å Kinesin 1nm
Introduction to N-STORM Dan Metcalf Advanced Imaging Manager Outline Introduction Principles of STORM Applications N-STORM overview Biological Scale Mitochondrion Microtubule Amino Acid 1Å Kinesin 1nm
Femtosecond micromachining in polymers
 Femtosecond micromachining in polymers Prof. Dr Cleber R. Mendonca Daniel S. Corrêa Prakriti Tayalia Dr. Tobias Voss Dr. Tommaso Baldacchini Prof. Dr. Eric Mazur fs-micromachining focus laser beam inside
Femtosecond micromachining in polymers Prof. Dr Cleber R. Mendonca Daniel S. Corrêa Prakriti Tayalia Dr. Tobias Voss Dr. Tommaso Baldacchini Prof. Dr. Eric Mazur fs-micromachining focus laser beam inside
Supplementary Information
 Supplementary Information STED nanoscopy combined with optical tweezers reveals protein dynamics on densely covered DNA Iddo Heller, Gerrit Sitters, Onno D. Broekmans, Géraldine Farge, Carolin Menges,
Supplementary Information STED nanoscopy combined with optical tweezers reveals protein dynamics on densely covered DNA Iddo Heller, Gerrit Sitters, Onno D. Broekmans, Géraldine Farge, Carolin Menges,
Flow Cytometry - The Essentials
 Flow Cytometry - The Essentials Pocket Guide to Flow Cytometry: 1. Know your Cytometer 2. Understanding Fluorescence and Fluorophores 3. Gating Process 4. Controls 5. Optimization 6. Panel Building 7.
Flow Cytometry - The Essentials Pocket Guide to Flow Cytometry: 1. Know your Cytometer 2. Understanding Fluorescence and Fluorophores 3. Gating Process 4. Controls 5. Optimization 6. Panel Building 7.
Photon Upconversion Sensitized Nanoprobes for
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting Information Photon Upconversion Sensitized Nanoprobes for Sensing and Imaging of ph
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting Information Photon Upconversion Sensitized Nanoprobes for Sensing and Imaging of ph
Confocal Microscopy & Imaging Technology. Yan Wu
 Confocal Microscopy & Imaging Technology Yan Wu Dec. 05, 2014 Cells under the microscope What we use to see the details of the cell? Light and Electron Microscopy - Bright light / fluorescence microscopy
Confocal Microscopy & Imaging Technology Yan Wu Dec. 05, 2014 Cells under the microscope What we use to see the details of the cell? Light and Electron Microscopy - Bright light / fluorescence microscopy
Each question may have MULTIPLE correct answers. Select all that are correct.
 Knowledge Assessment Flow Cytometry Workshop, Part 1 April 20, 2015 Each question may have MULTIPLE correct answers. Select all that are correct. 1. Tandem dyes are a. highly stable fluorophores after
Knowledge Assessment Flow Cytometry Workshop, Part 1 April 20, 2015 Each question may have MULTIPLE correct answers. Select all that are correct. 1. Tandem dyes are a. highly stable fluorophores after
Graphene oxide-enhanced cytoskeleton imaging and mitosis tracking
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Supplementary information for Graphene oxide-enhanced cytoskeleton imaging and mitosis tracking
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Supplementary information for Graphene oxide-enhanced cytoskeleton imaging and mitosis tracking
Combined Use of Unnatural Amino Acids Enables Dual Color Super-Resolution Imaging of Proteins via Click Chemistry
 Supporting information Combined Use of Unnatural Amino Acids Enables Dual Color Super-Resolution Imaging of Proteins via Click Chemistry Kim-A. Saal,* [a] Frank Richter, [b] Peter Rehling, [b] Silvio O.
Supporting information Combined Use of Unnatural Amino Acids Enables Dual Color Super-Resolution Imaging of Proteins via Click Chemistry Kim-A. Saal,* [a] Frank Richter, [b] Peter Rehling, [b] Silvio O.
Elecrtonic Supplementary Information. Application of quantum dot barcodes prepared using biological self-assembly to multiplexed immunoassays
 Elecrtonic Supplementary Information Application of quantum dot barcodes prepared using biological self-assembly to multiplexed immunoassays Sakandar Rauf, Andrew Glidle and Jonathan M Cooper Department
Elecrtonic Supplementary Information Application of quantum dot barcodes prepared using biological self-assembly to multiplexed immunoassays Sakandar Rauf, Andrew Glidle and Jonathan M Cooper Department
Supporting Information: Gold nanorod plasmonic upconversion microlaser
 Supporting Information: Gold nanorod plasmonic upconversion microlaser 1 Materials Synthesis and Properties Ce Shi, Soheil Soltani, Andrea M. Armani 1.1 Nanorod synthesis First the gold nanorods (NRs)
Supporting Information: Gold nanorod plasmonic upconversion microlaser 1 Materials Synthesis and Properties Ce Shi, Soheil Soltani, Andrea M. Armani 1.1 Nanorod synthesis First the gold nanorods (NRs)
Anti-MOUSE IgG (H&L) (GOAT) Antibody DyLight 488 Conjugated (Min X Bv Ch Gt GP Ham Hs Hu Rb Rt & Sh Serum Proteins)
 Anti-MOUSE IgG (H&L) (GOAT) Antibody DyLight 488 Conjugated (Min X Bv Ch Gt GP Ham Hs Hu Rb Rt & Sh Serum Proteins) - 610-141-121 Code: 610-141-121 Size: 100 µg Product Description: Anti-MOUSE IgG (H&L)
Anti-MOUSE IgG (H&L) (GOAT) Antibody DyLight 488 Conjugated (Min X Bv Ch Gt GP Ham Hs Hu Rb Rt & Sh Serum Proteins) - 610-141-121 Code: 610-141-121 Size: 100 µg Product Description: Anti-MOUSE IgG (H&L)
ab CytoPainter ER Staining Kit Red Fluorescence
 ab139482 CytoPainter ER Staining Kit Red Fluorescence Instructions for Use Designed to detect Human endoplasmic reticulum by microscopy. This product is for research use only and is not intended for diagnostic
ab139482 CytoPainter ER Staining Kit Red Fluorescence Instructions for Use Designed to detect Human endoplasmic reticulum by microscopy. This product is for research use only and is not intended for diagnostic
Two-photon excitation STED microscopy
 Two-photon excitation STED microscopy Gael Moneron and Stefan W. Hell* Department of NanoBiophotonics, Max Planck Institute for Biophysical Chemistry, Am Fassberg 11, 37077 Göttingen, Germany *shell@gwdg.de
Two-photon excitation STED microscopy Gael Moneron and Stefan W. Hell* Department of NanoBiophotonics, Max Planck Institute for Biophysical Chemistry, Am Fassberg 11, 37077 Göttingen, Germany *shell@gwdg.de
Rice/TCU REU on Computational Neuroscience. Fundamentals of Molecular Imaging
 Rice/TCU REU on Computational Neuroscience Fundamentals of Molecular Imaging June 2, 2009 Neal Waxham 713-500-5621 m.n.waxham@uth.tmc.edu Objectives Introduction to resolution in light microscopy Brief
Rice/TCU REU on Computational Neuroscience Fundamentals of Molecular Imaging June 2, 2009 Neal Waxham 713-500-5621 m.n.waxham@uth.tmc.edu Objectives Introduction to resolution in light microscopy Brief
Living up to Life. Sample Preparation. for. Ground State Depletion (GSD) Super-resolution imaging. Protocol guide for Leica SR GSD system. Version 1.
 Sample Preparation for Ground State Depletion (GSD) Super-resolution imaging Protocol guide for Leica SR GSD system Version 1.0 1 1. Introduction Ground State Depletion (GSD) is the ultimate super-resolution
Sample Preparation for Ground State Depletion (GSD) Super-resolution imaging Protocol guide for Leica SR GSD system Version 1.0 1 1. Introduction Ground State Depletion (GSD) is the ultimate super-resolution
Real-Time Multiplex Kinase Phosphorylation Sensors in Living Cells
 Real-Time Multiplex Kinase Phosphorylation Sensors in Living Cells Nur P. Damayanti 1,2,5, Kevin Buno 3, Yi Cui 1,2,6, Sherry L. Voytik-Harbin 2-4, Roberto Pili 5, Jennifer Freeman 7 Joseph M. K. Irudayaraj
Real-Time Multiplex Kinase Phosphorylation Sensors in Living Cells Nur P. Damayanti 1,2,5, Kevin Buno 3, Yi Cui 1,2,6, Sherry L. Voytik-Harbin 2-4, Roberto Pili 5, Jennifer Freeman 7 Joseph M. K. Irudayaraj
Localization Microscopy
 Localization Microscopy Theory, Sample Prep & Practical Considerations Patrina Pellett & Ann McEvoy Applications Scientist GE Healthcare, Cell Technologies May 27 th, 2015 Localization Microscopy Talk
Localization Microscopy Theory, Sample Prep & Practical Considerations Patrina Pellett & Ann McEvoy Applications Scientist GE Healthcare, Cell Technologies May 27 th, 2015 Localization Microscopy Talk
No.49, November 2014 CONFOCAL APPLICATION LETTER. Quick Guide to STED Sample Preparation
 No.49, November 2014 CONFOCAL APPLICATION LETTER resolution Quick Guide to STED Sample Preparation 2 Quick Guide to STED Sample Preparation The guide s focus is set on the Leica TCS SP8 STED 3X with 592
No.49, November 2014 CONFOCAL APPLICATION LETTER resolution Quick Guide to STED Sample Preparation 2 Quick Guide to STED Sample Preparation The guide s focus is set on the Leica TCS SP8 STED 3X with 592
Laser-derived One-Pot Synthesis of Silicon Nanocrystals Terminated with Organic Monolayers
 Electronic Supplementary Information for: Laser-derived One-Pot Synthesis of Silicon Nanocrystals Terminated with Organic Monolayers N. Shirahata,*,a M. R. Linford, b S. Furumi, a L. Pei, b Y. Sakka, a
Electronic Supplementary Information for: Laser-derived One-Pot Synthesis of Silicon Nanocrystals Terminated with Organic Monolayers N. Shirahata,*,a M. R. Linford, b S. Furumi, a L. Pei, b Y. Sakka, a
AnaTag 5-FAM Protein Labeling Kit
 AnaTag 5-FAM Protein Labeling Kit Catalog # 72053 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate 5-FAM SE (5-carboxyfluorescein) to proteins (e.g., IgG). It provides ample materials
AnaTag 5-FAM Protein Labeling Kit Catalog # 72053 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate 5-FAM SE (5-carboxyfluorescein) to proteins (e.g., IgG). It provides ample materials
A cost-effective fluorescence detection system for pulsed laser analysis
 Susquehanna University Scholarly Commons Chemistry Faculty Publications 2-2015 A cost-effective fluorescence detection system for pulsed laser analysis J. W. Lafferty Susquehanna University N. A. Fox Susquehanna
Susquehanna University Scholarly Commons Chemistry Faculty Publications 2-2015 A cost-effective fluorescence detection system for pulsed laser analysis J. W. Lafferty Susquehanna University N. A. Fox Susquehanna
STORM/PALM. Super Resolution Microscopy 10/31/2011. Looking into microscopic world of life
 Super Resolution Microscopy STORM/PALM Bo Huang Department of Pharmaceutical Chemistry, UCSF CSHL Quantitative Microscopy, 1/31/211 Looking into microscopic world of life 1 µm 1 µm 1 nm 1 nm 1 nm 1 Å Naked
Super Resolution Microscopy STORM/PALM Bo Huang Department of Pharmaceutical Chemistry, UCSF CSHL Quantitative Microscopy, 1/31/211 Looking into microscopic world of life 1 µm 1 µm 1 nm 1 nm 1 nm 1 Å Naked
A Brief History of Light Microscopy And How It Transformed Biomedical Research
 A Brief History of Light Microscopy And How It Transformed Biomedical Research Suewei Lin Office: Interdisciplinary Research Building 8A08 Email: sueweilin@gate.sinica.edu.tw TEL: 2789-9315 Microscope
A Brief History of Light Microscopy And How It Transformed Biomedical Research Suewei Lin Office: Interdisciplinary Research Building 8A08 Email: sueweilin@gate.sinica.edu.tw TEL: 2789-9315 Microscope
AnaTag HiLyte Fluor 488 Microscale Protein Labeling Kit
 AnaTag HiLyte Fluor 488 Microscale Protein Labeling Kit Revision number: 1.3 Last updated: April 2018 Catalog # AS-72048 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor
AnaTag HiLyte Fluor 488 Microscale Protein Labeling Kit Revision number: 1.3 Last updated: April 2018 Catalog # AS-72048 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor
Wavelength LDH - P / D - _ / C / F / FA / TA - N - XXX - _ / B / M / L / XL. Narrow linewidth (on request) Tappered amplified
 LDH Series Picosecond Laser Diode Heads for PDL 800-B/-D/808/828 Wavelengths between 375 nm and 1990 nm Pulse widths as short as 40 ps (FWHM) Adjustable (average) up to 50 mw Repetition rate from single
LDH Series Picosecond Laser Diode Heads for PDL 800-B/-D/808/828 Wavelengths between 375 nm and 1990 nm Pulse widths as short as 40 ps (FWHM) Adjustable (average) up to 50 mw Repetition rate from single
OCTOPLUS QPLEX FLUORESCENCE IMAGER. for fast & powerful fast 2D Gel image acquisition
 OCTOPLUS QPLEX FLUORESCENCE IMAGER for fast & powerful fast 2D Gel image acquisition Octoplus QPLEX Fluorescence Imager The new Octoplus QPLEX fluorescence imager sets a novel standard fluorescence 2D
OCTOPLUS QPLEX FLUORESCENCE IMAGER for fast & powerful fast 2D Gel image acquisition Octoplus QPLEX Fluorescence Imager The new Octoplus QPLEX fluorescence imager sets a novel standard fluorescence 2D
For labelling sub-cellular organelles in tissue sections, cell cultures and cell free experiments using our proprietary Red fluorescence probe
 ab112127 CytoPainter F-actin Staining Kit - Red Fluorescence Instructions for Use For labelling sub-cellular organelles in tissue sections, cell cultures and cell free experiments using our proprietary
ab112127 CytoPainter F-actin Staining Kit - Red Fluorescence Instructions for Use For labelling sub-cellular organelles in tissue sections, cell cultures and cell free experiments using our proprietary
Supplementary Information. Facile fabrication of microsphere-polymer brush hierarchically. three-dimensional (3D) substrates for immunoassays
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information Facile fabrication of microsphere-polymer brush hierarchically three-dimensional
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information Facile fabrication of microsphere-polymer brush hierarchically three-dimensional
Confocal Microscopy Analyzes Cells
 Choosing Filters for Fluorescence A Laurin Publication Photonic Solutions for Biotechnology and Medicine November 2002 Confocal Microscopy Analyzes Cells Reprinted from the November 2002 issue of Biophotonics
Choosing Filters for Fluorescence A Laurin Publication Photonic Solutions for Biotechnology and Medicine November 2002 Confocal Microscopy Analyzes Cells Reprinted from the November 2002 issue of Biophotonics
EdU Cell Proliferation Kit. User Manual
 EdU Cell Proliferation Kit User Manual Ordering information: (for detailed kit content see Table 2) EdU Click Kits: Product number EdU Used fluorescent dye BCK-EdU488 BCK-EdU555 BCK-EdU594 BCK-EdU647 5
EdU Cell Proliferation Kit User Manual Ordering information: (for detailed kit content see Table 2) EdU Click Kits: Product number EdU Used fluorescent dye BCK-EdU488 BCK-EdU555 BCK-EdU594 BCK-EdU647 5
Characterizing Phenotypes of Bacteria by Staining Method
 Experiment 3 Laboratory to Biology III Diversity of Microorganisms / Wintersemester / page 1 Experiment 3 Characterizing Phenotypes of Bacteria by Staining Method Advisor NN Reading Chapters in BBOM 9
Experiment 3 Laboratory to Biology III Diversity of Microorganisms / Wintersemester / page 1 Experiment 3 Characterizing Phenotypes of Bacteria by Staining Method Advisor NN Reading Chapters in BBOM 9
SIL-based confocal fluorescence microscope for investigating individual nanostructures
 Cent. Eur. J. Phys. 9(2) 2011 293-299 DOI: 10.2478/s11534-010-0098-5 Central European Journal of Physics SIL-based confocal fluorescence microscope for investigating individual nanostructures Research
Cent. Eur. J. Phys. 9(2) 2011 293-299 DOI: 10.2478/s11534-010-0098-5 Central European Journal of Physics SIL-based confocal fluorescence microscope for investigating individual nanostructures Research
Drug Discovery Research Clinical Screening. Comparison of ELISA and AlphaScreen Assay Technologies for Measurement of Protein Expression Levels
 Drug Discovery Research Clinical Screening FLAG Expression Measurement Application Note Comparison of ELISA and AlphaScreen Assay Technologies for Measurement of Protein Expression Levels ALPHASCREEN APPLICATION
Drug Discovery Research Clinical Screening FLAG Expression Measurement Application Note Comparison of ELISA and AlphaScreen Assay Technologies for Measurement of Protein Expression Levels ALPHASCREEN APPLICATION
Characterizing Phenotypes of Bacteria by Staining Method
 Experiment 3 Laboratory to Biology III Diversity of Microorganisms / Wintersemester / page 1 Experiment Characterizing Phenotypes of Bacteria by Staining Method Advisor Reading NN Chapters 3.1, 3.7, 3.8,
Experiment 3 Laboratory to Biology III Diversity of Microorganisms / Wintersemester / page 1 Experiment Characterizing Phenotypes of Bacteria by Staining Method Advisor Reading NN Chapters 3.1, 3.7, 3.8,
ATFM 2015 ATFM Sample preparation guide
 ATFM 2016 Sample preparation guide The workshop is organized in the framework of the Czech-BioImaging research infrastructure supported by MEYS (LM2015062) 1 Table of Contents General recommendations...
ATFM 2016 Sample preparation guide The workshop is organized in the framework of the Czech-BioImaging research infrastructure supported by MEYS (LM2015062) 1 Table of Contents General recommendations...
In situ semi-quantitative assessment of single cell viability by resonance
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) In situ semi-quantitative assessment
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) In situ semi-quantitative assessment
Study Small Molecule-Membrane Protein Binding Kinetics with. Nanodisc and Charge Sensitive Optical Detection
 Support Information Study Small Molecule-Membrane Protein Binding Kinetics with Nanodisc and Charge Sensitive Optical Detection Guangzhong Ma 1,2, Yan Guan 1,3, Shaopeng Wang 1*, Han Xu 4*, Nongjian Tao
Support Information Study Small Molecule-Membrane Protein Binding Kinetics with Nanodisc and Charge Sensitive Optical Detection Guangzhong Ma 1,2, Yan Guan 1,3, Shaopeng Wang 1*, Han Xu 4*, Nongjian Tao
Toggling Between Blue and Red Emitting Fluorescent Silver Nanoclusters
 Supporting Information Toggling Between Blue and Red Emitting Fluorescent Silver Nanoclusters Uttam Anand, Subhadip Ghosh and Saptarshi Mukherjee * Department of Chemistry, Indian Institute of Science
Supporting Information Toggling Between Blue and Red Emitting Fluorescent Silver Nanoclusters Uttam Anand, Subhadip Ghosh and Saptarshi Mukherjee * Department of Chemistry, Indian Institute of Science
Protein Delivery Reagent
 Protein Delivery Reagent Cat. # Contents Quantity BP502401 BioPORTER Reagent, dried 1 tube (24 rxns.) β-galactosidase control protein 10 µg (100 µg/ml) FITC-antibody control protein 10 µg (100 µg/ml) (fluorescein-labeled
Protein Delivery Reagent Cat. # Contents Quantity BP502401 BioPORTER Reagent, dried 1 tube (24 rxns.) β-galactosidase control protein 10 µg (100 µg/ml) FITC-antibody control protein 10 µg (100 µg/ml) (fluorescein-labeled
Nature Neuroscience: doi: /nn Supplementary Figure 1. Comprehensive opto-mechanical design of the dual-axis microscope.
 Supplementary Figure 1 Comprehensive opto-mechanical design of the dual-axis microscope. (a) The complete microscope is shown to scale. The laser beam is depicted in light red. (b) A close-up view of the
Supplementary Figure 1 Comprehensive opto-mechanical design of the dual-axis microscope. (a) The complete microscope is shown to scale. The laser beam is depicted in light red. (b) A close-up view of the
AnaTag HiLyte Fluor 647 Protein Labeling Kit
 AnaTag HiLyte Fluor 647 Protein Labeling Kit Catalog # 72049 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor 647 SE to proteins (e.g., IgG). It provides ample materials
AnaTag HiLyte Fluor 647 Protein Labeling Kit Catalog # 72049 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor 647 SE to proteins (e.g., IgG). It provides ample materials
Components for Highly Advanced time-resolved fluorescence Microscopy based on Nonlinear Glass fibres
 Components for Highly Advanced time-resolved fluorescence Microscopy based on Nonlinear Glass fibres FP7 Call identifier: FP7-ICT-2011-7 Activity code: ICT-2011.3.5: Photonics Project start date and duration:
Components for Highly Advanced time-resolved fluorescence Microscopy based on Nonlinear Glass fibres FP7 Call identifier: FP7-ICT-2011-7 Activity code: ICT-2011.3.5: Photonics Project start date and duration:
AnaTag HiLyte Fluor 647 Microscale Protein Labeling Kit
 AnaTag HiLyte Fluor 647 Microscale Protein Labeling Kit Revision number: 1.3 Last updated: May 2018 Catalog # AS-72050 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor 647
AnaTag HiLyte Fluor 647 Microscale Protein Labeling Kit Revision number: 1.3 Last updated: May 2018 Catalog # AS-72050 Kit Size 3 Conjugation Reactions This kit is optimized to conjugate HiLyte Fluor 647
Propidium Iodide. Catalog Number: Structure: Molecular Formula: C 27H 34I 2N 4. Molecular Weight: CAS #
 Catalog Number: 195458 Propidium Iodide Structure: Molecular Formula: C 27H 34I 2N 4 Molecular Weight: 668.45 CAS # 25535-16-4 Physical Description: Dark red crystals Description: Reagent used for the
Catalog Number: 195458 Propidium Iodide Structure: Molecular Formula: C 27H 34I 2N 4 Molecular Weight: 668.45 CAS # 25535-16-4 Physical Description: Dark red crystals Description: Reagent used for the
Workshop advanced light microscopy
 Workshop advanced light microscopy Multi-mode confocal laser scanning microscope Jan Willem Borst Laboratory of Biochemistry Biomolecular Networks www.bic.wur.nl MicroSpectroscopy Centre Wageningen Microspectroscopy
Workshop advanced light microscopy Multi-mode confocal laser scanning microscope Jan Willem Borst Laboratory of Biochemistry Biomolecular Networks www.bic.wur.nl MicroSpectroscopy Centre Wageningen Microspectroscopy
Confocal Microscopes. Evolution of Imaging
 Confocal Microscopes and Evolution of Imaging Judi Reilly Hans Richter Massachusetts Institute of Technology Environment, Health & Safety Office Radiation Protection What is Confocal? Pinhole diaphragm
Confocal Microscopes and Evolution of Imaging Judi Reilly Hans Richter Massachusetts Institute of Technology Environment, Health & Safety Office Radiation Protection What is Confocal? Pinhole diaphragm
Low Background D-A-D Type Fluorescent Probe for Imaging of Biothiols
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2018 Electronic supplementary information Low Background D-A-D Type Fluorescent
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2018 Electronic supplementary information Low Background D-A-D Type Fluorescent
SUPPLEMENTARY FIGURES
 SYNERGISTIC STRATEGY FOR MULTICOLOR TWO-PHOTON MICROSCOPY: APPLICATION TO THE ANALYSIS OF GERMINAL CENTER REACTIONS IN VIVO ASYLKHAN RAKHYMZHAN, RUTH LEBEN, HANNA ZIMMERMANN, ROBERT GÜNTHER, PEGGY MEX,
SYNERGISTIC STRATEGY FOR MULTICOLOR TWO-PHOTON MICROSCOPY: APPLICATION TO THE ANALYSIS OF GERMINAL CENTER REACTIONS IN VIVO ASYLKHAN RAKHYMZHAN, RUTH LEBEN, HANNA ZIMMERMANN, ROBERT GÜNTHER, PEGGY MEX,
Laser Induced Fluorescence Spectroscopy and Molecular Imaging as Tools for Tumor Detection in vivo
 Laser Induced Fluorescence Spectroscopy and Molecular Imaging as Tools for Tumor Detection in vivo B. Ebert Physikalisch-Techn. Bundesanstalt, Section 8.31, PTB, Berlin, Germany Outline: Spectroscopic
Laser Induced Fluorescence Spectroscopy and Molecular Imaging as Tools for Tumor Detection in vivo B. Ebert Physikalisch-Techn. Bundesanstalt, Section 8.31, PTB, Berlin, Germany Outline: Spectroscopic
average diameter = 3 nm, from PlasmaChem) was mixed in NLCs to produce QDembedded
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information Experimental Section The blended CLC-monomer materials used to fabricate
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting information Experimental Section The blended CLC-monomer materials used to fabricate
Imaging facilities at WUR
 Imaging facilities at WUR Advanced light microscopy facilities at Wageningen UR Programme Thursday 13 June 2013 Lunch meeting organized by Cat-Agro Food 12.00 Welcome and sandwich lunch 12.10 Introduction
Imaging facilities at WUR Advanced light microscopy facilities at Wageningen UR Programme Thursday 13 June 2013 Lunch meeting organized by Cat-Agro Food 12.00 Welcome and sandwich lunch 12.10 Introduction
DETECTION OF LASER ULTRASONIC SURFACE DISPLACEMENT BY WIDE APERTURE FIBER OPTIC AMPLIFIER M.L. Rizzi and F. Corbani CESI, Milano, Italy
 DETECTION OF LASER ULTRASONIC SURFACE DISPLACEMENT BY WIDE APERTURE FIBER OPTIC AMPLIFIER M.L. Rizzi and F. Corbani CESI, Milano, Italy Abstract: In the frame of the European Project INCA, CESI is in charge
DETECTION OF LASER ULTRASONIC SURFACE DISPLACEMENT BY WIDE APERTURE FIBER OPTIC AMPLIFIER M.L. Rizzi and F. Corbani CESI, Milano, Italy Abstract: In the frame of the European Project INCA, CESI is in charge
Page 1 of 5. Product Name Label Quantity Product No. Cy 3 10 µg (~0.75 nmol) MIR Cy µg (~7.5 nmol) MIR 7901
 Page 1 of 5 Label IT RNAi Delivery Control Product Name Label Quantity Product No. Cy 3 10 µg (~0.75 nmol) MIR 7900 Label IT RNAi Delivery Control Cy 3 100 µg (~7.5 nmol) MIR 7901 Fluorescein 10 µg (~0.75
Page 1 of 5 Label IT RNAi Delivery Control Product Name Label Quantity Product No. Cy 3 10 µg (~0.75 nmol) MIR 7900 Label IT RNAi Delivery Control Cy 3 100 µg (~7.5 nmol) MIR 7901 Fluorescein 10 µg (~0.75
How to perform-control immunostaining experiment - microscopist subjective point of view. Pawel Pasierbek
 How to perform-control immunostaining experiment - microscopist subjective point of view. Pawel Pasierbek Immunolabeling and fluorescent detection became such a standard procedure in the biomedical research
How to perform-control immunostaining experiment - microscopist subjective point of view. Pawel Pasierbek Immunolabeling and fluorescent detection became such a standard procedure in the biomedical research
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Thermally Induced Recrystallization of MAPbI 3 Perovskite
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Thermally Induced Recrystallization of MAPbI 3 Perovskite
Macromolecular environments influence proteins
 Research & Development Protein Dynamics Macromolecular environments influence proteins 6 www.q-more.com/en/ q&more 01.16 Studying proteins in the presence of high concentrations of macromolecules ( molecular
Research & Development Protein Dynamics Macromolecular environments influence proteins 6 www.q-more.com/en/ q&more 01.16 Studying proteins in the presence of high concentrations of macromolecules ( molecular
Division of Chemistry and Chemical Engineering, California Institute of Technology 2
 Supporting Information Real-time, Digital LAMP with Commercial Microfluidic Chips Reveals the Interplay of Efficiency, Speed, and Background Amplification as a Function of Reaction Temperature and Time
Supporting Information Real-time, Digital LAMP with Commercial Microfluidic Chips Reveals the Interplay of Efficiency, Speed, and Background Amplification as a Function of Reaction Temperature and Time
Materials and Methods Materials Required for Fixing, Embedding and Sectioning
 Page 1 Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigenspecific antibodies coupled to fluorophores. Depending on the antibody or dye used, proteins,
Page 1 Introduction Immunofluorescence uses the recognition of cellular targets by fluorescent dyes or antigenspecific antibodies coupled to fluorophores. Depending on the antibody or dye used, proteins,
