A-Site Deficient (Pr0.6Sr0.4)(1-s)Fe0.8Co0.2O3-delta Perovskites as Solid Oxide Fuel Cell Cathodes
|
|
|
- Elizabeth Moore
- 6 years ago
- Views:
Transcription
1 Downloaded from orbit.dtu.dk on: Apr 21, 218 A-Site Deficient (Pr.6Sr.4)(1-)Fe.8Co.2O3-delta Perovkite a Solid Oxide Fuel Cell Cathode Kammer Hanen, Kent Publihed in: Journal of the Electrochemical Society Link to article, DOI: / Publication date: 29 Document Verion Publiher' PDF, alo known a Verion of record Link back to DTU Orbit Citation (APA): Kammer Hanen, K. (29). A-Site Deficient (Pr.6Sr.4)(1-)Fe.8Co.2O3-delta Perovkite a Solid Oxide Fuel Cell Cathode. Journal of the Electrochemical Society, 156(1), B1257-B126. DOI: / General right Copyright and moral right for the publication made acceible in the public portal are retained by the author and/or other copyright owner and it i a condition of acceing publication that uer recognie and abide by the legal requirement aociated with thee right. Uer may download and print one copy of any publication from the public portal for the purpoe of private tudy or reearch. You may not further ditribute the material or ue it for any profit-making activity or commercial gain You may freely ditribute the URL identifying the publication in the public portal If you believe that thi document breache copyright pleae contact u providing detail, and we will remove acce to the work immediately and invetigate your claim.
2 Downloaded 3 Jun 21 to Reditribution ubject to ECS licene or copyright; ee /29/156 1 /B1257/4/$25. The Electrochemical Society A-Site Deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 Perovkite a Solid Oxide Fuel Cell Cathode K. Kammer Hanen*,z Fuel Cell and Solid State Chemitry Diviion, Riø National Laboratory for Sutainable Energy, Technical Univerity of Denmark, DK-4 Rokilde, Denmark B1257 Five A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite =.1,.5,.1,.15, and.2 were yntheized uing the glycine-nitrate proce. The perovkite were characterized with powder X-ray diffraction XRD, dilatometry, four-point dc conductivity meaurement, and electrochemical impedance pectrocopy uing cone-haped electrode on a Ce.9 Gd.1 O 1.95 electrolyte. XRD revealed that only the compound with =.1 and.5 were of ingle phae. The other compound contained an additional phae in the form of a Co Fe pinel. The thermal expanion coefficient decreaed ytematically with an increae in. The total conductivity followed the mall polaron hopping proce below a certain characteritic temperature. The total conductivity decreaed with increaing. It wa likewie hown that the polarization reitance found a minimum for the compound with =.5, with a total area pecific reitance more than 3 time lower than the weakly A-ite deficient Pr.6 Sr.4.99 Fe.8 Co.2 O 3 perovkite. 29 The Electrochemical Society. DOI: / All right reerved. Manucript ubmitted May 26, 29; revied manucript received June 29, 29. Publihed Augut 2, 29. The olid oxide fuel cell SOFC i a high temperature generator of heat and electricity. One major obtacle that mut be overcome, if the SOFC i to be commercialized, i lowering of the operation temperature. 1 To make thi poible, new electrode material are needed. On the cathode ide Co- or Fe Co-baed electrode have the highet activity toward the reduction of oxygen in an SOFC, 2 even though example of other ytem with an equal electrochemical activity are known. 3 In thi ene two compoition have been extenively invetigated in the literature, Sm.5 Sr.5 CoO 3 and La.6 Sr.4 Fe.8 Co.2 O An area pecific reitance ASR of.18 cm 2 at 6 C ha been reached by a compoite of Sm.5 Sr.5 CoO 3 and Ce 1 x Gd x O 2 CGO. 4 Thi i till not a high enough performance at 6 C, and better electrode material mut be developed. Recently, even better/lower ASR value have been obtained on a Ba.5 Sr.5 Fe.2 Co.8 O 3 -baed cathode. 1 An ASR of le than.1 cm 2 at 6 C wa obtained. However, electrode made with thi compound are not table over time. 11,12 A few tudie of A-ite deficient Fe Co-baed perovkite have been reported It wa hown that the conductivity depend on the A-ite toichiometry. 13 Likewie, it wa hown that the thermal expanion coefficient TEC decreae when the A-ite toichiometry i lowered. From powder X-ray diffraction XRD it wa concluded that the A-ite deficient perovkite were rhombohedral and ingle phae. 13 Reult of the electrochemitry of the A-ite deficient La.6 Sr.4 1 Fe.8 Co.2 O 3 were reported in Ref. 15. The activity wa trongly dependent on. Thee A-ite deficient perovkite conited of two cubic perovkite phae and one pinel phae. 15 The trongly A-ite deficient perovkite La.6 Sr.4.9 Fe.8 Co.2 O 3 ha been invetigated a porou compoite electrode with Ce.9 Gd.1 O 1.95 CGO1 with relatively good reult, a an ASR value of.19 cm 2 wa obtained on a CGO1 electrolyte at 6 C. 14 Thi i better than the ASR value obtained on the toichiometric La.6 Sr.4 Fe.8 Co.2 O 3 perovkite. 9 Even though the perovkite wa trongly A-ite deficient, the perovkite wa not table together with yttria-tabilized zirconia. In thi tudy a erie of A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite =.1,.5,.1,.15, and.2 wa yntheized and invetigated a SOFC cathode by uing electrochemical impedance pectrocopy EIS and cone-haped electrode. The ue of cone-haped electrode i very convenient when tudying SOFC electrode The contact area of the cone-haped electrode can be determined uing Newman formula 2 * Electrochemical Society Active Member. z kkha@rioe.dtu.dk 1 r = 4R 1 where R i the intercept at the real axi at high frequency and i the pecific conductivity of the electrolyte. r i the radiu of the contact between the cone-haped electrode and the electrolyte, from which the contact area can be calculated. The polarization reitance on cone-haped electrode i much higher than the polarization reitance on porou planar electrode. Fe Co-baed cathode with Pr are of particular interet a they perform better than Fe Co-baed cathode with other A-ite cation. 21 Experimental Synthei of the Pr.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite wa done uing the glycine-nitrate route. 22 Aqueou olution of the metal nitrate were mixed in an appropriate ratio in a beaker. Glycine wa then added. The olution were then heated on a hot plate until they ignited. The reulting powder were tranferred to alumina crucible and were calcined at 11 C/12 h in air in a box furnace. The following tarting compound were ued a metal nitrate: Pr NO 3 3 Alfa Aear, 99.9%, Sr NO 3 2 Alfa Aear, 99%, Fe NO 3 3 Alfa Aear, 98%, and Co NO 3 2 Alfa Aear, 99.8%. To evaluate the phae purity of the calcined powder, powder XRD uing a Stoe diffractometer equipped with Cu K radiation wa performed. The diffractogram were recorded in the range 2 : 2 8. The bar for the dilatometry and conductivity meaurement and the cylinder for the fabrication of the cone-haped electrode were made a follow: The powder were preed in appropriate die bar and cylinder and intered at 125 C/12 h in a box furnace in air. The bar were then machined into mm bar and the cylinder were machined into cone-haped electrode. The conehaped electrode had a bae of 7.5 mm and ide with an angle of 45. The denity of the bar wa determined with the Archimede method. The CGO1 pellet ued for the electrochemical meaurement wa made a follow: CGO1 powder Rhodia wa mixed with tearic acid and glycerine in a ball mill with ethanol. After drying the powder wa preed to a dik in an appropriate die and intered at 15 C/2 h in air in a box furnace. The tearic acid wa added a a lubricant and the glycerine wa added a a binder. The dilatometry meaurement were performed by uing a Netzh 42 dilatometer in the temperature range of 3 1 C with heating and cooling ramp of 2 C/min. The bar were kept at 1 C for 2 h before cooling down. The four-point dc conductivity meaurement were alo performed in air uing a Keithley micro-ohmmeter together with a peronal computer PC controlling the ohmmeter and the furnace temperature. An in-houe PC Linux program, wa ued for the collection of the data temperature and reitance. The
3 Downloaded 3 Jun 21 to Reditribution ubject to ECS licene or copyright; ee B T/ o C Rel. int. /% σ Τ / Scm -1 *K 1e+6 1e =.1 =.5 =.1 =.15 = θ /degree Figure 1. Powder XRD pattern of the compound Pr.6 Sr.4.95 Fe.8 Co.2 O 3 recorded in air at room temperature. ample were heated and cooled with a ramp of 5 C/min. The temperature range panned wa 5 1 C. At every 5 C the ample were kept at temperature for 2 h. The reitance wa meaured every fifth minute. The characterization of the cone-haped electrode were done in a etup decribed in Ref. 23. EIS wa ued to characterize the cone-haped electrode. The EIS wa meaured in the range of 1 MHz.5 Hz with five point meaured at each decade. An amplitude of 36 mv wa ued throughout. An amplitude of 36 mv wa needed to obtain data of ufficient quality. The meaurement were done at 8, 7, and 6 C in the given order. The cone were equilibrated at temperature for 24 h before the meaurement. 24 h were ufficient for equilibration. The meaurement were done in air. For the EIS meaurement a Solartron 126 in tand-alone mode wa ued. The EIS data were treated in the PC- DOS program equivcrt by Boukamp. 24 Three RQ component were ued in the fitting. Q i a contant phae element with the admittance Y = Y j / n 2 where Y i an admittance, i the angular frequency, j i the imaginary unit, and n i the frequency exponent. Reult A reult from the powder XRD i found in Fig. 1. From powder XRD only the ample with =.1 and.5 are ingle phae. The Table I. Unit cell parameter of A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite meaured with powder XRD in air at room temperature. A Å B Å C Å e+4,5,1,15,2,25,3,35 1 / T1/K Figure 2. Color online Total conductivitie a a function of temperature for the A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite. The conductivity drop with increaing. The conductivitie have been corrected uing the Bruggeman aymmetric model. 25 other ample contain an additional phae in the form of a Co Febaed pinel phae. A determined from XRD, the amount of the pinel phae increae with increaing. The maximum amount of foreign phae i around 1% for =.2. The unit cell parameter are found in Table I. The unit cell parameter how an irregular change with. The peak are rather broad. Thi i probably due to an inhomogeneou ditribution of the cation. For the intered bar a denity in exce of 92% of the theoretical denity wa obtained. The reult of the dilatometry meaurement are found in Table II. Oberve that the TEC decreae with decreaing toichiometry on the A-ite. The conductivity of the ample i alo dependent on the A-ite toichiometry. The conductivity a a function of temperature i plotted in Fig. 2. The conductivity i highet for the compound with =.1 at all temperature. For all the ample an increae in conductivity with an increae in temperature, until a certain temperature after which the conductivity decreae, i oberved. The maximum in conductivity i found for the compound with =.1, and it give a value of 42 S cm 1 at 52 C. An example of an EIS pectrum i een in Fig. 3, together with the reult of the fitting. The pectrum conit of three arc. The variation in the total ASR with at a temperature of 6 C i found in Fig. 4. The total ASR find a minimum for the compound with =.5 with an ASR of 1.72 cm 2 at 6 C. The total ASR value at the three meaured temperature are found in Table III. The contribution to the total ASR of the individual arc at 6 C are found in Table IV, together with the n value. The n value are the frequency exponent given in Eq. 2. Dicuion The A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite differ from the A-ite deficient La.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite. 15 In A-ite deficient lanthanum-baed perovkite, two perovkite phae are preent along with a Fe Co-baed pinel Table II. TEC value of A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 perovkite in air from 1 to 9 C. TEC 1 6 K
4 Downloaded 3 Jun 21 to Reditribution ubject to ECS licene or copyright; ee B1259 -Z imag / Ω phae. The two perovkite phae in the A-ite deficient lanthanumbaed perovkite are two phae with different amount of La, Sr, Fe, and Co. In praeodymium-baed perovkite, only one perovkite phae i preent along with a Co Fe-baed pinel phae. The variation in the TEC with i ytematic, a the TEC decreae almot linearly with increaing A-ite deficiency. Thi i probably due to the impuritie with the pinel phae, and becaue cobalt i lot from the perovkite phae 16 Fe-baed perovkite have much lower TEC than Fe Co-baed perovkite. 17 The change in TEC could alo be due to a change in the point defect concentration. The TEC value found for the A-ite deficient Pr-baed perovkite are higher than the TEC for CGO electrolyte. 26 However, ASR / Ωcm e+4 4e+4 6e+4 8e+4 1e+5 Z real / Ω Figure 3. Color online EIS of the compound Pr.6 Sr.4.9 Fe.8 Co.2 O 3 at 6 C in air. Circle are meaured data. Solid line i fitted data. Characteritic frequencie are marked in(pr.6 Sr.4 ) 1- Fe.8 Co.2 O 3-δ Figure 4. Total ASR value a a function of meaured in air at 6 C with EIS. The uncertaintie on the contact area are around 1 15%. Table III. Total ASR value for A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 at three different temperature in air meaured with EIS. The value are given in cm 2. 6 C C C compoite electrode made by the A-ite deficient perovkite and CGO might be of ue a cathode on a CGO electrolyte. The electronic conductivity of the compound change from a metallic to a emiconductor-like behavior when the temperature i lowered, that i, the electronic conductivity increae until a certain temperature i reached after which the electronic conductivity decreae. The conductivity behavior a a function of temperature below a certain characteritic temperature indicate that the conductivity i due to the mall polaron hopping mechanim 27 with the following relationhip = A/kT v exp E a /kt 3 where A i a material contant, k i Boltzmann contant, and E a i the activation energy. The exponent v i 1 for the adiabatic mall polaron hopping proce and 3/2 for the nonadiabatic mall polaron hopping proce. The equation with v=3/2 fit the meaured data bet, in contrat to what ha been oberved by Tai et al. 28,29 but in agreement with Hanen and Hanen 15 for imilar material. The activation energy E a of the total conductivity, below the temperature of maximum conductivity, i around.1 ev for all the compound. The decreae in the total conductivity above a certain characteritic temperature can be explained a follow. When the temperature i increaed, the perovkite loe oxygen. When oxygen vacancie are creted in the perovkite tructure, a decoupling of the overlap between the d-orbital of the tranition metal and the p-orbital of oxygen i the reult. Thi can explain the lowered conductivity at high temperature. 28,29 Not only do the oxygen vacancie hinder the polaron hopping but they alo alter the other defect concentration which lead to a lowering of the concentration of charge carrier. Thi i the main effect. The calculated temperature of maximum conductivity i alo higher than the oberved temperature of maximum conductivity. The calculated temperature can be found uing the equation E a = kt max 4 The calculated temperature of maximum conductivity i around 115 K for all the compound, compared to the oberved temperature of maximum conductivity of approximately 8 K. Thi i alo an indication that the oxygen toichiometry i lowered upon heating of the perovkite. The conductivity behavior found in thi tudy i almot the ame a found in the literature. 13,15 The variation in the total conductivity with can be undertood a follow. When i increaed, cobalt i lot from the perovkite phae, a oberved for A-ite deficient lanthanum-baed perovkite 15 and confirmed by XRD. Fe Co-baed perovkite have a higher conductivity than Fe-baed perovkite. 3 Thi therefore lower the conductivity. Alo, the preence of a pinel lower the conductivity a Co Fe-baed pinel have a much lower conductivity than Fe Co-baed perovkite. 31 Oxide ionic conductivity change a a function of. In the literature it wa hown that a lowered A-ite occupancy greatly influence the oxygen toichiometry, oxygen ion vacancy, and cation vacancy concentration for manganite-baed perovkite. 32 However, no literature data on the defect chemitry of the A-ite deficient Fe Co-baed perovkite have been given. In principle, a lowered A-ite occupancy can lead to the creation of both oxide ion vacan- Table IV. Contribution to the total ASR of the individual arc for A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 meaured at 6 C in air with EIS. The value are given in cm 2. 6 C ASR n ASR n ASR n ASR n ASR n High Medium Low
5 Downloaded 3 Jun 21 to Reditribution ubject to ECS licene or copyright; ee B126 Table V. Chemical capacitance of A-ite deficient Pr.6 Sr.4 1 Fe.8 Co.2 O 3 at 6 C in air meaured with EIS. cie and cation vacancie in the perovkite tructure, but thi ha never been oberved for perovkite; alo, the powder XRD data ugget that exce Fe Co i expelled from the perovkite tructure when i increaed. If, however, both anion and cation vacancie are created in the perovkite tructure, thi probably lower the oxide ionic conductivity due to the attraction between cation and anion vacancie. The magnitude of the ASR i very high on cone-haped electrode compared to porou compoite electrode of the ame material. Thi i partly becaue cone-haped electrode are not compoite electrode, and partly due to the interface between the electrode electrolyte, which i not optimized in the cone-haped electrode. The geometrical configuration of the cone-haped electrode limit the number of contact point at the electrode electrolyte interface. Alo, the cone-haped electrode i dene and therefore ha a low urface area. The interpretation of the EIS data can be done according to the mechanim uggeted and confirmed by Siebert et al. 33 We are well aware of other mechanim uggeted in the literature, but thi mechanim fit the data bet. In thi mechanim the high frequency arc i due to the exchange of oxide ion at the electrode electrolyte interface. The equivalent capacity of thi arc can be calculated uing the equation 34 C = R 1 n /n Y 1/n 5 The magnitude of the equivalent capacity of thi arc i between.5 and 1 F cm 2 for all the compound, uggeting that thi arc i due to a double-layer effect. 35 The n value are all cloe to.9 for all the compound, which again indicate that it i the ame type of reaction that occur on all the compound. The medium frequency arc i normally attributed to the diffuion of oxide anion through the bulk of the electrode. 33 Thi arc find it minimum for the compound with =.5, indicating that thi compound ha the highet oxide ionic conductivity. The magnitude of the low frequency arc i alo dependent on, the magnitude being mallet for the compound with =.5. The low frequency arc i due to a low redox reaction at the urface of the electrode. 33 One poible explanation for thi behavior could be the lowering of the urface egregation of A-ite cation, increaing the amount of the catalytic active pecie Fe and Co on the urface of the electrode material. That the magnitude of the low frequency arc increae with a further decreae in i perhap due to the lo of cobalt from the perovkite phae. The equivalent capacity of thi arc can be calculated for the high frequency arc uing Eq. 5. The value are given in Table V. They all fall in the range for a chemical capacitance, a depicted in Ref. 36. Furthermore it eem like the magnitude of the three arc i coupled; they all decreae or increae in the ame manner with a change in. Thi parameter could either be the oxide ionic conductivity or the urface egregation of praeodymium, or perhap both. The electrochemical behavior of the A-ite deficient Pr-baed perovkite i markedly different from the behavior of the A-ite deficient La-baed perovkite. The effect of A-ite deficiency i much le pronounced for the Pr-baed perovkite than for the Labaed perovkite. 8 Thi could be due to le urface egregation for the praeodymium-baed perovkite than for the lanthanum-baed perovkite. Praeodymium perhap fit better in the perovkite tructure than lanthanum, lowering the importance of A-ite deficiency. It could alo be due to the ability of praeodymium to C low F cm change it oxidation tate. It could be that praeodymium ha ome catalytic activity in itelf toward the reduction of oxygen. Thi ha been hown for other ytem. 37 Concluion The TEC decreae almot linearly with, probably due to the lo of cobalt from the perovkite phae. The total conductivity decreae a a function of increaing. The effect of A-ite ubtoichiometry on the activity of praeodymium-baed perovkite toward the reduction of oxygen i reflected in a lowered ASR for the compound with =.5. Thi i thought to be due to lowering of the urface egregation of praeodymium. Acknowledgment Colleague at the Fuel Cell and Solid State Chemitry Diviion are thanked for fruitful dicuion. Financial upport from Energinet.dk through PSO-R&D project no i gratefully acknowledged. Technical Univerity of Denmark aited in meeting the publication cot of thi article. Reference 1. N. Q. Minh, J. Am. Ceram. Soc., 76, J. M. Ralph, C. Roignol, and R. Kumar, J. Electrochem. Soc., 15, A G. Xia and M. Liu, Adv. Mater. (Weinheim, Ger.), 14, C. Xia, W. Rauch, F. Chen, and M. Liu, Solid State Ionic, 149, Y. Liu, W. Rauch, S. Zha, and M. Liu, Solid State Ionic, 166, C. Xia and M. Liu, Solid State Ionic, 144, Y. Liu, W. Rauch, S. Zha, and M. Liu, Chem. Mater., 16, M. Sahibzada, S. J. Benon, R. A. Rudkin, and J. A. Kilner, Solid State Ionic, , E. P. Murray, M. J. Sever, and S. A. Barnett, Solid State Ionic, 148, Z. Shao and S. M. Haile, Nature (London), 431, A. Yan, M. Cheng, Y. Dong, W. Yang, V. Maragou, S. Song, and P. Tiakara, Appl. Catal., B, 66, A. Yan, V. Maragou, A. Arico, M. Cheng, and P. Tiakara, Appl. Catal., B, 76, G. Ch. Kotogloudi and Ch. Ftiko, Solid State Ionic, 126, W. G. Wang and M. Mogenen, Solid State Ionic, 176, K. K. Hanen and K. V. Hanen, Solid State Ionic, 178, S.-I. Hahimoto, K. Kammer, P. H. Laren, F. W. Poulen, and M. Mogenen, Solid State Ionic, 176, K. Kammer, L. Mikkelen, and J. B. Bilde-Sørenen, J. Solid State Electrochem., 1, K. Kammer, Solid State Ionic, 177, S.-I. Hahimoto, K. Kammer, F. W. Poulen, and M. Mogenen, J. Alloy Compd., 428, J. Newman, J. Electrochem. Soc., 113, I. Ruiz de Larramendi, D. G. Lama, M. D. Cabeza, J. I. Ruiz de Larramendi, N. E. Walöe de Reca, and T. Rojo, J. Power Source, In pre. 22. L. A. Chick, L. R. Pederon, G. D. Maupin, J. L. Bate, L. E. Thoma, and G. J. Exarho, Mater. Lett., 1, M. Juhl, M. Mogenen, T. Jacoben, B. Zachau-Chritianen, N. Thorup, and E. Skou, Proceeding of the 4th International Sympoium on Solid oxide fuel cell (SOFC-IV), Yokohama, June 18 23, M. Dokiya, O. Yamamoto, H. Tagawa and S. C. Singhal, Editor, The Electrochemical Society, Pennington, NJ, Vol. PV 95-1, p B. A. Boukamp, Solid State Ionic, 2, D. A. G. Bruggeman, Ann. Phy., 416, N. Kim, B.-H. Kim, and D. Lee, J. Power Source, 9, J. M. Wimmer and I. Branky, in Electrical Conductivity in Ceramic and Gla, N. M. Tallan, Editor, Chap. 4, Pergamon, New York L.-W. Tai, M. M. Narallah, H. U. Anderon, D. M. Sparlin, and S. R. Sehlin, Solid State Ionic, 76, L.-W. Tai, M. M. Narallah, H. U. Anderon, D. M. Sparlin, and S. R. Sehlin, Solid State Ionic, 76, M. V. Patrakeev, J. A. Bahteeva, E. B. Mitberg, I. A. Leonidov, V. L. Kozhevnikov, and K. R. Poeppelmeier, J. Solid State Chem., 172, D. S. Erickon and T. O. Maon, J. Solid State Chem., 59, F. W. Poulen, Solid State Ionic, 129, E. Siebert, A. Hammouche, and M. Kleitz, Electrochim. Acta, 4, T. Jacoben, B. Zachau-Chritianen, L. Bay, and S. Skaarup, in Proceeding of the 17th International Sympoium on Material Science, F. W. Poulen, N. Bonano, S. Linderoth, M. Mogenen, and B. Zachau-Chritianen, Editor, Riø National Laboratory, p N. L. Roberton and J. N. Michael, J. Electrochem. Soc., 138, S. B. Adler, J. A. Lane, and B. C. H. Steele, J. Electrochem. Soc., 143, R. M. L. Werchmeiter, K. K. Hanen, and M. Mogenen, To be publihed.
Cobalt-free Composite Ba 0.5 Sr 0.5 Fe 0.9 Ni 0.1 O 3 δ Ce 0.8 Sm 0.2 O 2 δ as Cathode for Intermediate-Temperature Solid Oxide Fuel Cell
 J. Mater. Sci. Technol., 212, 28(9), 828 832. Cobalt-free Composite Ba.5 Sr.5 Fe.9 Ni.1 O 3 δ Ce.8 Sm.2 O 2 δ as Cathode for Intermediate-Temperature Solid Oxide Fuel Cell Xiangfeng Chu 1), Feng Liu 1),
J. Mater. Sci. Technol., 212, 28(9), 828 832. Cobalt-free Composite Ba.5 Sr.5 Fe.9 Ni.1 O 3 δ Ce.8 Sm.2 O 2 δ as Cathode for Intermediate-Temperature Solid Oxide Fuel Cell Xiangfeng Chu 1), Feng Liu 1),
PHYSICAL PROPERTIES OF La 0.9 Sr 0.1 Cr 1-X Ni X O 3-δ (X = 0-0.6) SYNTHESIZED VIA CITRATE GEL COMBUSTION
 PHYSICAL PROPERTIES OF La 0.9 Sr 0.1 Cr 1-X Ni X O 3-δ (X = 0-0.6) SYNTHESIZED VIA CITRATE GEL COMBUSTION Anuchit Ruangvittayanon * and Sutin Kuharuangrong Received: Sept 29, 2009; Revised: Nov 17, 2009;
PHYSICAL PROPERTIES OF La 0.9 Sr 0.1 Cr 1-X Ni X O 3-δ (X = 0-0.6) SYNTHESIZED VIA CITRATE GEL COMBUSTION Anuchit Ruangvittayanon * and Sutin Kuharuangrong Received: Sept 29, 2009; Revised: Nov 17, 2009;
Lawrence Berkeley National Laboratory Lawrence Berkeley National Laboratory
 Lawrence Berkeley National Laboratory Lawrence Berkeley National Laboratory Title LSM-YSZ Cathode with Reaction-Infiltrated Nanoparticle Permalink http://echolarhip.org/uc/item/2714v00f Author Lu, Chun
Lawrence Berkeley National Laboratory Lawrence Berkeley National Laboratory Title LSM-YSZ Cathode with Reaction-Infiltrated Nanoparticle Permalink http://echolarhip.org/uc/item/2714v00f Author Lu, Chun
Modeling Liquid Phase Sintering of Hard metal powder compacts
 ing Liquid Phae Sintering of Hard metal powder compact Shama Shamaundar *, Maheha Siddegowda*, Pavanachand Chigurupati**, Rengarajan Raghavan***, Rameh Rao. S. *** * ProSIM AFTC # 326, 8th Main, III Stage,
ing Liquid Phae Sintering of Hard metal powder compact Shama Shamaundar *, Maheha Siddegowda*, Pavanachand Chigurupati**, Rengarajan Raghavan***, Rameh Rao. S. *** * ProSIM AFTC # 326, 8th Main, III Stage,
Equilibrium Sediment Transport and Evolution Trend Simulation of the Lower Yellow River
 Senor & Tranducer, Vol. 21, Special Iue, May 213, pp. 135-141 Senor & Tranducer 213 by IFSA http://www.enorportal.com Equilibrium Sediment Tranport and Evolution Trend Simulation of the Lower Yellow River
Senor & Tranducer, Vol. 21, Special Iue, May 213, pp. 135-141 Senor & Tranducer 213 by IFSA http://www.enorportal.com Equilibrium Sediment Tranport and Evolution Trend Simulation of the Lower Yellow River
Management Science Letters
 Management Science Letter 2 (202) 247 252 Content lit available at GrowingScience Management Science Letter homepage: www.growingscience.com/ml An empirical tudy to meaure the impact of loan aignment for
Management Science Letter 2 (202) 247 252 Content lit available at GrowingScience Management Science Letter homepage: www.growingscience.com/ml An empirical tudy to meaure the impact of loan aignment for
Electrical and Ionic Transport Properties. (1) Laboratoire de Recherches sur la Réactivité des Solides
 (La 0.8 Sr 0.2 )(Mn 1-y Fe y )O 3±δ Oxides for ITSOFC Cathode Materials? Electrical and Ionic Transport Properties M. Petitjean (1), G. Caboche (1), E. Siebert (2), L. Dessemond (2), L.-C. Dufour (1) (1)
(La 0.8 Sr 0.2 )(Mn 1-y Fe y )O 3±δ Oxides for ITSOFC Cathode Materials? Electrical and Ionic Transport Properties M. Petitjean (1), G. Caboche (1), E. Siebert (2), L. Dessemond (2), L.-C. Dufour (1) (1)
Typical Albedo ALBEDO 1/14/13. Albedo. Disposition of Solar Radiation at Earth s Surface
 Introduction to Climatology GEOGAPHY 300 Dipoition of Solar adiation at Earth Surface Tom Giambelluca Univerity of Hawai i at Mānoa eflection: Albedo i a key property of urface controlling the urface energy
Introduction to Climatology GEOGAPHY 300 Dipoition of Solar adiation at Earth Surface Tom Giambelluca Univerity of Hawai i at Mānoa eflection: Albedo i a key property of urface controlling the urface energy
Land-Surface Models, Page 1
 Land-Surface Model Introduction A land-urface model mut be able to accurately depict the interaction of the atmophere with the underlying urface land a well a the interaction of the ub-urface, or ubtrate,
Land-Surface Model Introduction A land-urface model mut be able to accurately depict the interaction of the atmophere with the underlying urface land a well a the interaction of the ub-urface, or ubtrate,
3.4 BUTT FUSION WELDING
 3.4 BUTT FUSION 3.4.1 INTRODUCTION The butt welding proce conit of the joining of two component (pipe and/or fitting) of equal diameter and thickne in which the urface to be welded are heated until melting
3.4 BUTT FUSION 3.4.1 INTRODUCTION The butt welding proce conit of the joining of two component (pipe and/or fitting) of equal diameter and thickne in which the urface to be welded are heated until melting
Ionic Conductivity and Solid Electrolytes II: Materials and Applications
 Ionic Conductivity and Solid Electrolytes II: Materials and Applications Chemistry 754 Solid State Chemistry Lecture #27 June 4, 2003 References A. Manthiram & J. Kim Low Temperature Synthesis of Insertion
Ionic Conductivity and Solid Electrolytes II: Materials and Applications Chemistry 754 Solid State Chemistry Lecture #27 June 4, 2003 References A. Manthiram & J. Kim Low Temperature Synthesis of Insertion
Paul Addo, Beatriz Molero-Sanchez, Aligul Buyukaksoy, Scott Paulson and Viola Birss
 10.1149/06602.0219ecst The Electrochemical Society Sulfur Tolerance of La 0.3 M 0.7 Fe 0.7 Cr 0.3 O 3-δ (M = Sr, Ca) Solid Oxide Fuel Cell Anodes Paul Addo, Beatriz Molero-Sanchez, Aligul Buyukaksoy, Scott
10.1149/06602.0219ecst The Electrochemical Society Sulfur Tolerance of La 0.3 M 0.7 Fe 0.7 Cr 0.3 O 3-δ (M = Sr, Ca) Solid Oxide Fuel Cell Anodes Paul Addo, Beatriz Molero-Sanchez, Aligul Buyukaksoy, Scott
Functionally graded cathodes fabricated by sol-gel/slurry coating for honeycomb SOFCs
 Solid State Ionics 176 (2005) 25 31 www.elsevier.com/locate/ssi Functionally graded cathodes fabricated by sol-gel/slurry coating for honeycomb SOFCs Shaowu Zha, Yuelan Zhang, Meilin Liu* School of Materials
Solid State Ionics 176 (2005) 25 31 www.elsevier.com/locate/ssi Functionally graded cathodes fabricated by sol-gel/slurry coating for honeycomb SOFCs Shaowu Zha, Yuelan Zhang, Meilin Liu* School of Materials
Effect of Starting Materials on the Characteristics of (La 1-x Sr x ) Mn 1+y O 3-δ Powder Synthesized by GNP
 Korea-America nano forum Effect of Starting Materials on the Characteristics of (La 1-x Sr x ) Mn 1+y O 3-δ Powder Synthesized by GNP 2007. 04. 26 orea nstitute of eramic ngineering & echnology MI-Jai
Korea-America nano forum Effect of Starting Materials on the Characteristics of (La 1-x Sr x ) Mn 1+y O 3-δ Powder Synthesized by GNP 2007. 04. 26 orea nstitute of eramic ngineering & echnology MI-Jai
Sintering Behavior of Porous Nanostructured Sr-Doped Lanthanum Manganite as SOFC Cathode Material
 Int. J. Nanosci. Nanotechnol., Vol. 9, No. 2, June 2013, pp. 71-76 Sintering Behavior of Porous Nanostructured Sr-Doped Lanthanum Manganite as SOFC Cathode Material H. Tamaddon *, A. Maghsoudipour Ceramics
Int. J. Nanosci. Nanotechnol., Vol. 9, No. 2, June 2013, pp. 71-76 Sintering Behavior of Porous Nanostructured Sr-Doped Lanthanum Manganite as SOFC Cathode Material H. Tamaddon *, A. Maghsoudipour Ceramics
Effect of Cobalt Doping on Physical Properties of ZnO Nanoparticles
 CPUH-Reearch Journal: 06, (), 47-5 ISSN (nline): 455-6076 http://www.cpuh.in/academic/academic_journal.php Effect of Cobalt oping on Phyical Propertie of Zn Nanoparticle Neha Sharma, Shaveta Thakur, Ruchita
CPUH-Reearch Journal: 06, (), 47-5 ISSN (nline): 455-6076 http://www.cpuh.in/academic/academic_journal.php Effect of Cobalt oping on Phyical Propertie of Zn Nanoparticle Neha Sharma, Shaveta Thakur, Ruchita
Address for Correspondence
 Reearch Paper ENERGY CONSERVATION IN MUD HOUSE AS COMPARED TO BRICK WALL BUILDING IN INDIA Subhah Mihra 1, Dr. J A Umani 2 Addre for Correpondence 1 Ph.d Scholar, 2 Profeor, Department of Mechanical Engineering,
Reearch Paper ENERGY CONSERVATION IN MUD HOUSE AS COMPARED TO BRICK WALL BUILDING IN INDIA Subhah Mihra 1, Dr. J A Umani 2 Addre for Correpondence 1 Ph.d Scholar, 2 Profeor, Department of Mechanical Engineering,
6/6/2012. HR Training and Development. Content. Training: concept. Training: concept. Training: concept. Training and Development: Concept
 HR Training and Development UNIT 5 Content Concept and need of HR training and development Training need aement HR training: objective and method (on-the-job and off-the-job). Evaluation of training program
HR Training and Development UNIT 5 Content Concept and need of HR training and development Training need aement HR training: objective and method (on-the-job and off-the-job). Evaluation of training program
M A S O N R Y. Revised Spring Engineering Notes For Design With Concrete Block Masonry
 A S O N R Y Revied Spring 007 Engineering Note For Deign With Concrete Block aonry C H R O N I C L E S To rectify the ituation, the Spring 007 article i being reiued. We apologize for any inconvenience
A S O N R Y Revied Spring 007 Engineering Note For Deign With Concrete Block aonry C H R O N I C L E S To rectify the ituation, the Spring 007 article i being reiued. We apologize for any inconvenience
Model of Integrated Production and Delivery Batch Scheduling Under JIT Environment to Minimize Inventory Cost
 Proceeding of the 2014 International Conference on Indutrial Engineering and Operation Management Bali, Indoneia, January 7 9, 2014 Model of Integrated Production and Delivery Batch Scheduling Under JIT
Proceeding of the 2014 International Conference on Indutrial Engineering and Operation Management Bali, Indoneia, January 7 9, 2014 Model of Integrated Production and Delivery Batch Scheduling Under JIT
Management Science Letters
 Management Science Letter 2 (2012) 3049 3054 Content lit available at GrowingScience Management Science Letter homepage: www.growingscience.com/ml Identification and prioritization of hazardou material
Management Science Letter 2 (2012) 3049 3054 Content lit available at GrowingScience Management Science Letter homepage: www.growingscience.com/ml Identification and prioritization of hazardou material
arxiv: v2 [physics.soc-ph] 8 Oct 2009
![arxiv: v2 [physics.soc-ph] 8 Oct 2009 arxiv: v2 [physics.soc-ph] 8 Oct 2009](/thumbs/75/72611811.jpg) Human group formation in online guild and offline gang driven by common team dynamic arxiv:082.2299v2 [phyic.oc-ph] 8 Oct 2009 Neil F. Johnon, Chen Xu 2,3, Zhenyuan Zhao, Nicola Ducheneaut 4, Nichola Yee
Human group formation in online guild and offline gang driven by common team dynamic arxiv:082.2299v2 [phyic.oc-ph] 8 Oct 2009 Neil F. Johnon, Chen Xu 2,3, Zhenyuan Zhao, Nicola Ducheneaut 4, Nichola Yee
Research Article. Hydration process study on the alkali-activated slag cementing materials
 Available online wwwjocprcom Journal of Chemical and Pharmaceutical Reearch,, ():88 Reearch Article ISSN : 975738 CODEN(USA) : JCPRC5 Hydration proce tudy on the alkaliactivated lag cementing material
Available online wwwjocprcom Journal of Chemical and Pharmaceutical Reearch,, ():88 Reearch Article ISSN : 975738 CODEN(USA) : JCPRC5 Hydration proce tudy on the alkaliactivated lag cementing material
Cerámica y Vidrio A R T I C U L O
 B O L E T I N D E L A S O C I E D A D E S P A Ñ O L A D E Cerámica y Vidrio A R T I C U L O A view on organic binder effect on technical propertie of ceramic Rachig ring Y. BEYGI KHOSROWSHAHI, A. SALEM
B O L E T I N D E L A S O C I E D A D E S P A Ñ O L A D E Cerámica y Vidrio A R T I C U L O A view on organic binder effect on technical propertie of ceramic Rachig ring Y. BEYGI KHOSROWSHAHI, A. SALEM
Structural Properties of NiO-CGO Composites Precursor Prepared via Combustion Synthesis Route
 Asian Journal of Chemistry Vol. 21, No. 10 (2009), S157-161 Structural Properties of NiO-CGO Composites Precursor Prepared via Combustion Synthesis Route PANKAJ KALRA# ANIRUDH P. SINGH and AJAY KUMAR #
Asian Journal of Chemistry Vol. 21, No. 10 (2009), S157-161 Structural Properties of NiO-CGO Composites Precursor Prepared via Combustion Synthesis Route PANKAJ KALRA# ANIRUDH P. SINGH and AJAY KUMAR #
Polycaprolactone Foam for Scaffold Development
 STR/4/6/FT Polycaprolactone Foam for Scaffold Development B. Y. Tay, M. H. Myint and F. L. Ng Abtract The tudy invetigate the proceing of biodegradable porou caffold of polycaprolactone (PCL). A commercially
STR/4/6/FT Polycaprolactone Foam for Scaffold Development B. Y. Tay, M. H. Myint and F. L. Ng Abtract The tudy invetigate the proceing of biodegradable porou caffold of polycaprolactone (PCL). A commercially
a) The self-diffusion coefficient of a metal with cubic structure can be expressed as
 EXERCISES KJM5120 Chapter 5; Diffusion 1. Random (self) diffusion a) The self-diffusion coefficient of a metal with cubic structure can be expressed as 1 n D = s 6 t 2 where n/t represents the jump frequency
EXERCISES KJM5120 Chapter 5; Diffusion 1. Random (self) diffusion a) The self-diffusion coefficient of a metal with cubic structure can be expressed as 1 n D = s 6 t 2 where n/t represents the jump frequency
Challenges of Developing ISO Sampling Standards
 Challenge of Developing ISO Sampling Standard Ralph Holme CSIRO Mineral Down Under Flaghip Chair ISO/TC 10/SC 1 Sampling Iron Ore Chair ISO/TC 7/SC 4 Sampling Coal and Coke Convenor ISO/TC 183/WG 9 Sampling
Challenge of Developing ISO Sampling Standard Ralph Holme CSIRO Mineral Down Under Flaghip Chair ISO/TC 10/SC 1 Sampling Iron Ore Chair ISO/TC 7/SC 4 Sampling Coal and Coke Convenor ISO/TC 183/WG 9 Sampling
Concentration profiles of gold inside activated carbon during adsorption
 Concentration profile of gold inide activated carbon during adorption by N.M. Vegter*, R.F. Sandenbergh*, and A.J. Botha Synopi The gold concentration profile which develop inide the activated carbon particle
Concentration profile of gold inide activated carbon during adorption by N.M. Vegter*, R.F. Sandenbergh*, and A.J. Botha Synopi The gold concentration profile which develop inide the activated carbon particle
Pollution prevention with chemical process simulators: the generalized waste reduction (WAR) algorithm full version
 Computer and Chemical Engineering 23 (1999) 623 634 Pollution prevention with chemical proce imulator: the generalized wate reduction (WAR) algorithm full verion Heriberto Cabeza *, Jane C. Bare, Subir
Computer and Chemical Engineering 23 (1999) 623 634 Pollution prevention with chemical proce imulator: the generalized wate reduction (WAR) algorithm full verion Heriberto Cabeza *, Jane C. Bare, Subir
In Situ IonicÕElectric Conductivity Measurement of La 0.55 Li 0.35 TiO 3 Ceramic at Different Li Insertion Levels
 A1196 Journal of The Electrochemical Society, 151 8 A1196-A1201 2004 0013-4651/2004/151 8 /A1196/6/$7.00 The Electrochemical Society, Inc. In Situ IonicÕElectric Conductivity Measurement of La 0.55 Li
A1196 Journal of The Electrochemical Society, 151 8 A1196-A1201 2004 0013-4651/2004/151 8 /A1196/6/$7.00 The Electrochemical Society, Inc. In Situ IonicÕElectric Conductivity Measurement of La 0.55 Li
2nd International Conference on Production and Industrial Engineering CPIE-2010
 nd International Conference on Production and Indutrial Engineering CPIE- 59 Actie/Paie Damping Technique for iration control: - FEM aed deign and experimental iration control Sanji Kumar Raji Kumar Rakeh
nd International Conference on Production and Indutrial Engineering CPIE- 59 Actie/Paie Damping Technique for iration control: - FEM aed deign and experimental iration control Sanji Kumar Raji Kumar Rakeh
The University of Bradford Institutional Repository
 he Univerity of Bradford Intitutional Repoitory http://bradcholar.brad.ac.uk hi work i made available online in accordance with publiher policie. Pleae refer to the repoitory record for thi item and our
he Univerity of Bradford Intitutional Repoitory http://bradcholar.brad.ac.uk hi work i made available online in accordance with publiher policie. Pleae refer to the repoitory record for thi item and our
THE POTENTIAL AND COST OF CARBON SEQUESTRATION IN AGRICULTURAL SOIL; EMPIRICAL STUDY OF DYNAMIC MODEL IN THE MIDWESTERN U.S.
 THE POTENTIAL AND COST OF CARBON SEQUESTRATION IN AGRICULTURAL SOIL; EMPIRICAL STUDY OF DYNAMIC MODEL IN THE MIDWESTERN U.S. DISSERTATION Preented in Partial Fulfillment of the Requirement for the Degree
THE POTENTIAL AND COST OF CARBON SEQUESTRATION IN AGRICULTURAL SOIL; EMPIRICAL STUDY OF DYNAMIC MODEL IN THE MIDWESTERN U.S. DISSERTATION Preented in Partial Fulfillment of the Requirement for the Degree
High Temperature Co-electrolysis of Steam and CO2 in an SOC stack: Performance and Durability
 Downloaded from orbit.dtu.dk on: Nov 26, 2017 High Temperature Co-electrolysis of Steam and CO2 in an SOC stack: Performance and Durability Chen, Ming; Høgh, Jens Valdemar Thorvald; Nielsen, Jens Ulrik
Downloaded from orbit.dtu.dk on: Nov 26, 2017 High Temperature Co-electrolysis of Steam and CO2 in an SOC stack: Performance and Durability Chen, Ming; Høgh, Jens Valdemar Thorvald; Nielsen, Jens Ulrik
Electronic supplementary information. Efficient energy storage capabilities promoted by hierarchically MnCo 2 O 4
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information Efficient energy storage capabilities promoted by hierarchically
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information Efficient energy storage capabilities promoted by hierarchically
GMACE Pilot #4: Adjusting the National Reliability Input Data
 INTERBULL BULLETIN NO. 48. Berlin, Germany, May 20 21, 2014 GMACE Pilot #4: Adjuting the National Reliability Input Data P. G. Sullivan 1 and J. H. Jakoben 2 1 Canadian Dairy Network, Guelph, ON, Canada
INTERBULL BULLETIN NO. 48. Berlin, Germany, May 20 21, 2014 GMACE Pilot #4: Adjuting the National Reliability Input Data P. G. Sullivan 1 and J. H. Jakoben 2 1 Canadian Dairy Network, Guelph, ON, Canada
Concept of Heat Recovery from Exhaust Gases
 IOP Conference Serie: Material Science and Engineering PAPER OPEN ACCESS Concept of Heat Recovery from Exhaut Gae To cite thi article: Maria Bukowka et al 2017 IOP Conf. Ser.: Mater. Sci. Eng. 245 052057
IOP Conference Serie: Material Science and Engineering PAPER OPEN ACCESS Concept of Heat Recovery from Exhaut Gae To cite thi article: Maria Bukowka et al 2017 IOP Conf. Ser.: Mater. Sci. Eng. 245 052057
Preparation and ionic conductivity of new Bi 12.5 Lu 1.5 ReO 24.5 phase
 Preparation and ionic conductivity of new Bi 12.5 Lu 1.5 ReO 24.5 phase A.N. Bryzgalova 1, N.I. Matskevich 1*, Th. Wolf 2, C. Greaves 3 1 Nikolaev Institute of Inorganic Chemistry, Siberian Branch of the
Preparation and ionic conductivity of new Bi 12.5 Lu 1.5 ReO 24.5 phase A.N. Bryzgalova 1, N.I. Matskevich 1*, Th. Wolf 2, C. Greaves 3 1 Nikolaev Institute of Inorganic Chemistry, Siberian Branch of the
Tom-Reiel Heggedal and Karl Jacobsen
 Dicuion Paper No. 536, April 2008 Statitic Norway, Reearch Department Tom-Reiel eggedal and Karl Jacoben Timing of innovation policie when carbon emiion are retricted: an applied general equilibrium analyi
Dicuion Paper No. 536, April 2008 Statitic Norway, Reearch Department Tom-Reiel eggedal and Karl Jacoben Timing of innovation policie when carbon emiion are retricted: an applied general equilibrium analyi
On the Onset of the Flash Transition in Single Crystals of Cubic Zirconia. Yanhao Dong
 On the Onset of the Flash Transition in Single Crystals of Cubic Zirconia Yanhao Dong (dongya@seas.upenn.edu) Department of Materials Science and Engineering, University of Pennsylvania, Philadelphia,
On the Onset of the Flash Transition in Single Crystals of Cubic Zirconia Yanhao Dong (dongya@seas.upenn.edu) Department of Materials Science and Engineering, University of Pennsylvania, Philadelphia,
Supplementary Material (ESI) for Chemical Communications. Solid-state single-crystal-to-single-crystal transformation from a 2D
 Supplementary Material (ESI) for Chemical Communications Solid-state single-crystal-to-single-crystal transformation from a 2D layer to a 3D framework mediated by lattice iodine release Yuan-Chun He, a
Supplementary Material (ESI) for Chemical Communications Solid-state single-crystal-to-single-crystal transformation from a 2D layer to a 3D framework mediated by lattice iodine release Yuan-Chun He, a
Gas Processing Expander
 Expander Operated Ga Proceing April, 2015 Ga Proceing Expander Colder Proce Temperature and Maximized Compreor Uptime with Helidyne Expander Skid. Specification: Flowrate 1-10 mmcfd Max. Preure 1,440 pi
Expander Operated Ga Proceing April, 2015 Ga Proceing Expander Colder Proce Temperature and Maximized Compreor Uptime with Helidyne Expander Skid. Specification: Flowrate 1-10 mmcfd Max. Preure 1,440 pi
The Nernst-Einstein equation indicates that the ratio β /D for a given material varies only with temperature. Calculate β/d for oxygen ions in Zr 0.
 The Nernst-Einstein equation indicates that the ratio β /D for a given material varies only with temperature. Calculate β/d for oxygen ions in 0.8 Y 0.2 1.9 at 800 C. 1 The Nernst-Einstein equation indicates
The Nernst-Einstein equation indicates that the ratio β /D for a given material varies only with temperature. Calculate β/d for oxygen ions in 0.8 Y 0.2 1.9 at 800 C. 1 The Nernst-Einstein equation indicates
SINCE the 1960s, conceptual models are in use to facilitate
 IEEE TRNSTIONS ON SYSTEMS, MN, ND YBERNETIS PRT Study into the Factor that Influence the Undertandability of Buine Proce Model Hajo. Reijer and Jan Mendling btract Buine proce model are key artifact in
IEEE TRNSTIONS ON SYSTEMS, MN, ND YBERNETIS PRT Study into the Factor that Influence the Undertandability of Buine Proce Model Hajo. Reijer and Jan Mendling btract Buine proce model are key artifact in
X-ray Diffraction Study on Structural Characteristics of Pure and Doped Perovskite BaTiO 3
 Egypt. J. Solids, Vol. (31), No. (1), (2008) 55 X-ray Diffraction Study on Structural Characteristics of Pure and Doped Perovskite BaTiO 3 F. F. Hammad 1, A. K. Mohamed 1 and A. M. El-Shabiny 2 1) Inorganic
Egypt. J. Solids, Vol. (31), No. (1), (2008) 55 X-ray Diffraction Study on Structural Characteristics of Pure and Doped Perovskite BaTiO 3 F. F. Hammad 1, A. K. Mohamed 1 and A. M. El-Shabiny 2 1) Inorganic
Dependence of potentiometric oxygen sensing characteristics on the nature of electrodes
 Sensors and Actuators B 113 (2006) 162 168 Dependence of potentiometric oxygen sensing characteristics on the nature of electrodes Ramasamy Ramamoorthy, Sheikh A. Akbar, Prabir. K. Dutta Center for Industrial
Sensors and Actuators B 113 (2006) 162 168 Dependence of potentiometric oxygen sensing characteristics on the nature of electrodes Ramasamy Ramamoorthy, Sheikh A. Akbar, Prabir. K. Dutta Center for Industrial
Ab initio modelling of Y O cluster formation in g-fe lattice
 Phy. Statu Solidi B 253, No. 11, 2136 2143 (2016) / DOI 10.1002/pb.201600472 Ab initio modelling of Y O cluter formation in g-fe lattice phyica tatu olidi baic olid tate phyic Alekej Gopejenko *,1, Yuri
Phy. Statu Solidi B 253, No. 11, 2136 2143 (2016) / DOI 10.1002/pb.201600472 Ab initio modelling of Y O cluter formation in g-fe lattice phyica tatu olidi baic olid tate phyic Alekej Gopejenko *,1, Yuri
Aalborg Universitet. Published in: I E E E Transactions on Industrial Electronics. DOI (link to publication from Publisher): /TIE.2016.
 Aalborg Univeritet Droop Control with Improved Diturbance Adaption for PV Sytem with Two Power Converion Stage Liu, Hongpeng; Loh, Poh Chiang; Wang, Xiongfei; Yang, Yongheng; Wang, Wei; Xu, Dianguo Publihed
Aalborg Univeritet Droop Control with Improved Diturbance Adaption for PV Sytem with Two Power Converion Stage Liu, Hongpeng; Loh, Poh Chiang; Wang, Xiongfei; Yang, Yongheng; Wang, Wei; Xu, Dianguo Publihed
Maintaining ISO Compliance in Automated Procedures
 Maintaining ISO 1705 Compliance in Automated Procedure Preenter & Author: Jorge Martin Fluke Corporation PO 9090 M/S 6-30 Everett, WA, USA 9806 Phone: (45) 446 6477; Fax: (45) 446 6390 Email: jmartin@flukecom
Maintaining ISO 1705 Compliance in Automated Procedure Preenter & Author: Jorge Martin Fluke Corporation PO 9090 M/S 6-30 Everett, WA, USA 9806 Phone: (45) 446 6477; Fax: (45) 446 6390 Email: jmartin@flukecom
Antibody light chain variable domains and their biophysically improved versions for human immunotherapy
 Paper Type mab 6:1, 219 235; January/February 2014; 2014 Lande Biocience Report Antibody light chain variable domain and their biophyically improved verion for human immunotherapy Dae Young Kim 1, Rebecca
Paper Type mab 6:1, 219 235; January/February 2014; 2014 Lande Biocience Report Antibody light chain variable domain and their biophyically improved verion for human immunotherapy Dae Young Kim 1, Rebecca
Software Reverse Engineering Tool for Object Oriented Programming
 Software Revere Engineering Tool for Object Oriented Programming D.M.Thakore Department of Computer Engineering Bharati vidyapeeth Deemed Univerity College of Engineering, Pune-43, Maharahtra, India S.J.Sarde
Software Revere Engineering Tool for Object Oriented Programming D.M.Thakore Department of Computer Engineering Bharati vidyapeeth Deemed Univerity College of Engineering, Pune-43, Maharahtra, India S.J.Sarde
Thermochemical behavior of nano-sized aluminum-coated nickel particles
 J Nanopart Re (2014) 16:2392 DOI 10.1007/11051-014-2392-4 RESEARCH PAPER Thermochemical behavior of nano-ized aluminum-coated nickel particle Dilip Sriniva Sundaram Puneeh Puri Vigor Yang Received: 17
J Nanopart Re (2014) 16:2392 DOI 10.1007/11051-014-2392-4 RESEARCH PAPER Thermochemical behavior of nano-ized aluminum-coated nickel particle Dilip Sriniva Sundaram Puneeh Puri Vigor Yang Received: 17
Solubility of Carbohydrates in Subcritical Water
 Solubility of Carbohydrate in Subcritical Water Fernando Montañé 1, Tiziana Fornari 2, Elena Ibáñez 1, Keerthi Sriniva 3 Dongfang Zhang 3 and Jerry W. King *3 1 Intituto de Fermentacione Indutriale (CSIC).
Solubility of Carbohydrate in Subcritical Water Fernando Montañé 1, Tiziana Fornari 2, Elena Ibáñez 1, Keerthi Sriniva 3 Dongfang Zhang 3 and Jerry W. King *3 1 Intituto de Fermentacione Indutriale (CSIC).
Effect of Anodizing Potential on the Surface Morphology and Corrosion Property of AZ31 Magnesium Alloy
 Materials Transactions, Vol. 51, No. 6 (21) pp. 119 to 1113 #21 The Japan Institute of Metals Effect of Anodizing Potential on the Surface Morphology and Corrosion Property of AZ31 Magnesium Alloy S. A.
Materials Transactions, Vol. 51, No. 6 (21) pp. 119 to 1113 #21 The Japan Institute of Metals Effect of Anodizing Potential on the Surface Morphology and Corrosion Property of AZ31 Magnesium Alloy S. A.
A1104 Effects of sintering temperature on composition, microstructure and electrochemical performance of spray pyrolysed LSC thin film cathodes
 A1104 Effects of sintering temperature on composition, microstructure and electrochemical performance of spray pyrolysed LSC thin film cathodes Omar Pecho 1,2 Lorenz Holzer 1, Zhèn Yáng 2, Julia Martynczuk
A1104 Effects of sintering temperature on composition, microstructure and electrochemical performance of spray pyrolysed LSC thin film cathodes Omar Pecho 1,2 Lorenz Holzer 1, Zhèn Yáng 2, Julia Martynczuk
parameter n in a power law σ p O 2 1/n
 substance: NiO 6 property: defects in pure NiO,conductivity defects: NiO is approximately stoichiometric at the Ni/NiO phase boundary, but at higher oxygen partial pressures, it is cation deficient, the
substance: NiO 6 property: defects in pure NiO,conductivity defects: NiO is approximately stoichiometric at the Ni/NiO phase boundary, but at higher oxygen partial pressures, it is cation deficient, the
OPTIMIZATION OF ALUMINIUM BLANK SAND CASTING PROCESS BY USING TAGUCHI S ROBUST DESIGN METHOD
 International Journal for Quality reearch UDK 669.76 Original Scientific Paper (.0) OPTIMIZATION OF ALUMINIUM BLANK SAND CASTING PROCESS BY USING TAGUCHI S ROBUST DESIGN METHOD Mekonnen Liben Nekere )
International Journal for Quality reearch UDK 669.76 Original Scientific Paper (.0) OPTIMIZATION OF ALUMINIUM BLANK SAND CASTING PROCESS BY USING TAGUCHI S ROBUST DESIGN METHOD Mekonnen Liben Nekere )
(12) Patent Application Publication (10) Pub. No.: US 2003/ A1
 US 20030204424A1 (19) United State (12) Patent Application Publication (10) Pub. No.: US 2003/0204424 A1 Koller et al. (43) Pub. Date: Oct. 30, 2003 (54) MANAGEMENT BY OBJECTIVES (52) U.S. Cl.... 705/7
US 20030204424A1 (19) United State (12) Patent Application Publication (10) Pub. No.: US 2003/0204424 A1 Koller et al. (43) Pub. Date: Oct. 30, 2003 (54) MANAGEMENT BY OBJECTIVES (52) U.S. Cl.... 705/7
A Strategy for Achieving High-performance with SOFC Ceramic Anodes
 University of Pennsylvania ScholarlyCommons Departmental Papers (CBE) Department of Chemical & Biomolecular Engineering January 2007 A Strategy for Achieving High-performance with SOFC Ceramic Anodes Michael
University of Pennsylvania ScholarlyCommons Departmental Papers (CBE) Department of Chemical & Biomolecular Engineering January 2007 A Strategy for Achieving High-performance with SOFC Ceramic Anodes Michael
Laurea Magistrale in Scienza dei Materiali. Materiali Inorganici Funzionali. Electrolytes: Ceria
 Laurea Magistrale in Scienza dei Materiali Materiali Inorganici Funzionali Electrolytes: Ceria Prof. Antonella Glisenti - Dip. Scienze Chimiche - Università degli Studi di Padova Bibliography 1. N.Q. Minh,
Laurea Magistrale in Scienza dei Materiali Materiali Inorganici Funzionali Electrolytes: Ceria Prof. Antonella Glisenti - Dip. Scienze Chimiche - Università degli Studi di Padova Bibliography 1. N.Q. Minh,
Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supplementary Information CO 2 -to-co Conversion on Layered Persovskite
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supplementary Information CO 2 -to-co Conversion on Layered Persovskite
Synthesis and characterization of Aln 2 O 4 indates, A ˆ Mg, Ca, Sr, Ba
 JOURNAL OF MATERIALS SCIENCE LETTERS 17 (1998) 619±623 Synthesis and characterization of Aln 2 O 4 indates, A ˆ Mg, Ca, Sr, Ba S. ESTHER DALI, V. V. S. S. SAI SUNDAR, M. JAYACHANDRAN, M. J. CHOCKALINGAM
JOURNAL OF MATERIALS SCIENCE LETTERS 17 (1998) 619±623 Synthesis and characterization of Aln 2 O 4 indates, A ˆ Mg, Ca, Sr, Ba S. ESTHER DALI, V. V. S. S. SAI SUNDAR, M. JAYACHANDRAN, M. J. CHOCKALINGAM
Southern Africa Labour and Development Research Unit
 Southern Africa Labour and Development Reearch Unit Entrepreneurhip and the Buine Environment in Africa: An Application to Ethiopia by Zuzana Brixiová and Mthuli Ncube Working Paper Serie Number 146 About
Southern Africa Labour and Development Reearch Unit Entrepreneurhip and the Buine Environment in Africa: An Application to Ethiopia by Zuzana Brixiová and Mthuli Ncube Working Paper Serie Number 146 About
ENVE 301 Environmental Engineering Unit Operations. CHAPTER: 5 Aeration
 ENVE 301 Environmental Engineering Unit Operation CHAPTER: 5 Aeration Ait. Prof. Bilge Alpalan Kocamemi Marmara Univerity Department of Environmental Engineering Itanbul, Turkey 1 Aeration Primary application
ENVE 301 Environmental Engineering Unit Operation CHAPTER: 5 Aeration Ait. Prof. Bilge Alpalan Kocamemi Marmara Univerity Department of Environmental Engineering Itanbul, Turkey 1 Aeration Primary application
Dynamic compressive and splitting tensile tests on mortar using split Hopkinson pressure bar technique
 730 Dynamic compreive and plitting tenile tet on mortar uing plit Hopkinon preure bar technique Abtract Dynamic compreive and tenile propertie of mortar under impact loading were invetigated experimentally
730 Dynamic compreive and plitting tenile tet on mortar uing plit Hopkinon preure bar technique Abtract Dynamic compreive and tenile propertie of mortar under impact loading were invetigated experimentally
Protective Properties of High Temperature Oxide Films on Ni-based Superalloys in 3.5% NaCl Solution
 J. Mater. Sci. Technol., Vol.23 No.4, 2007 541 Protective Properties of High Temperature Oxide Films on Ni-based Superalloys in 3.5% NaCl Solution Huiping BAI and Fuhui WANG State Key Laboratory for Corrosion
J. Mater. Sci. Technol., Vol.23 No.4, 2007 541 Protective Properties of High Temperature Oxide Films on Ni-based Superalloys in 3.5% NaCl Solution Huiping BAI and Fuhui WANG State Key Laboratory for Corrosion
Lab 1 The Defect Structure of cubic Bi 2 O 3 Nb 2 O 5 Solid Solutions
 Lab 1 The Defect Structure of cubic Bi 2 O 3 Nb 2 O 5 Solid Solutions 27-202 Fall 2002 Objective The objective of this lab is to identify the defect structure and charge compensation mechanism in Bi 2
Lab 1 The Defect Structure of cubic Bi 2 O 3 Nb 2 O 5 Solid Solutions 27-202 Fall 2002 Objective The objective of this lab is to identify the defect structure and charge compensation mechanism in Bi 2
Abstract. 1 Introduction
 Automatic conflict detection and reolution in metrorail ytem: evaluation approach for MARCO EU project G.F. D'Addio, M. Mazzucchelli, S. Savio Dipartimento di Ingegneria Elettrica, Univerita di Genova,
Automatic conflict detection and reolution in metrorail ytem: evaluation approach for MARCO EU project G.F. D'Addio, M. Mazzucchelli, S. Savio Dipartimento di Ingegneria Elettrica, Univerita di Genova,
All-solid-state Li battery using a light-weight solid electrolyte
 All-solid-state Li battery using a light-weight solid electrolyte Hitoshi Takamura Department of Materials Science, Graduate School of Engineering, Tohoku University Europe-Japan Symposium, Electrical
All-solid-state Li battery using a light-weight solid electrolyte Hitoshi Takamura Department of Materials Science, Graduate School of Engineering, Tohoku University Europe-Japan Symposium, Electrical
Lydia Fawcett Imperial College London, Department of Materials Submitted for examination for the degree of Doctor of Philosophy
 Electrochemical Performance and Compatibility of La 2 NiO 4+δ Electrode Material with La 0.8 Sr 0.2 Ga 0.8 Mg 0.2 O 3-δ Electrolyte for Solid Oxide Electrolysis Lydia Fawcett Imperial College London, Department
Electrochemical Performance and Compatibility of La 2 NiO 4+δ Electrode Material with La 0.8 Sr 0.2 Ga 0.8 Mg 0.2 O 3-δ Electrolyte for Solid Oxide Electrolysis Lydia Fawcett Imperial College London, Department
Effects of calcium impurity on phase relationship, ionic conductivity and microstructure of Na + -β/β -alumina solid electrolyte
 Bull. Mater. Sci., Vol. 39, No. 3, June 216, pp. 729 735. DOI 1.17/s1234-16-1199-6 c Indian Academy of Sciences. Effects of calcium impurity on phase relationship, ionic conductivity and microstructure
Bull. Mater. Sci., Vol. 39, No. 3, June 216, pp. 729 735. DOI 1.17/s1234-16-1199-6 c Indian Academy of Sciences. Effects of calcium impurity on phase relationship, ionic conductivity and microstructure
Corrosion Rate Measurement on C-Steel
 Measurements of corrosion rate on Carbon-steel using Electrochemical (potentiodynamic Polarization, EIS etc.) technique. Corrosion Rate Measurement on C-Steel Abdullah Al Ashraf 1. Introduction: The degradation
Measurements of corrosion rate on Carbon-steel using Electrochemical (potentiodynamic Polarization, EIS etc.) technique. Corrosion Rate Measurement on C-Steel Abdullah Al Ashraf 1. Introduction: The degradation
Migrating, corrosion-inhibiting admixtures for reinforced structures. Increases durability and dramatically reduces corrosion.
 Migrating Corroion Inhibitor! Migrating Corroion Inhibitor! MCI Admixture Migrating, corroion-inhibiting admixture for reinforced tructure. Increae durability and dramatically reduce corroion. Q U A EXCELLENCE
Migrating Corroion Inhibitor! Migrating Corroion Inhibitor! MCI Admixture Migrating, corroion-inhibiting admixture for reinforced tructure. Increae durability and dramatically reduce corroion. Q U A EXCELLENCE
Electrical Properties of the Ferrite SrFeO y at High Temperatures
 Journal of Solid State Chemistry 158, 320}326 (2000) doi:10.1006/jssc.2001.9120, available online at http://www.idealibrary.com on Electrical Properties of the Ferrite SrFeO y at High Temperatures V. L.
Journal of Solid State Chemistry 158, 320}326 (2000) doi:10.1006/jssc.2001.9120, available online at http://www.idealibrary.com on Electrical Properties of the Ferrite SrFeO y at High Temperatures V. L.
Evaluation of Tests To Assess Stripping Potential of Asphalt Concrete Mixtures
 18 TRASPORTATIO RESEARCH RECORD 1171 Evaluation of Tet To Ae Stripping Potential of Aphalt Concrete Mixture FRAZIER PARKER JR., AD FouAD A. GHARAYBEH Stre pedetal, boll, and Indirect tenile tet were evaluated
18 TRASPORTATIO RESEARCH RECORD 1171 Evaluation of Tet To Ae Stripping Potential of Aphalt Concrete Mixture FRAZIER PARKER JR., AD FouAD A. GHARAYBEH Stre pedetal, boll, and Indirect tenile tet were evaluated
J. Mater. Sci. Technol., 2010, 26(11),
 J. Mater. Sci. Technol., 2010, 26(11), 1016-1020. Effects of Current Density on the Microstructure and the Corrosion Resistance of Alumina Coatings Embedded with SiC Nano-particles Produced by Micro-arc
J. Mater. Sci. Technol., 2010, 26(11), 1016-1020. Effects of Current Density on the Microstructure and the Corrosion Resistance of Alumina Coatings Embedded with SiC Nano-particles Produced by Micro-arc
SOLIK Li-hochleitende Keramiken für all-solid-state Batterien
 SOLIK Li-hochleitende Keramiken für all-solid-state Batterien Dr. Ningxin ZHANG Electric Drive Technologies Center for Low-Emission Transport Austrian Institute of Technology GmbH Outline Basic Data Project
SOLIK Li-hochleitende Keramiken für all-solid-state Batterien Dr. Ningxin ZHANG Electric Drive Technologies Center for Low-Emission Transport Austrian Institute of Technology GmbH Outline Basic Data Project
Novel concept of rechargeable battery using iron oxide nanorods. anode and nickel hydroxide cathode in aqueous electrolyte
 Supplementary Information for: Novel concept of rechargeable battery using iron oxide nanorods anode and nickel hydroxide cathode in aqueous electrolyte Zhaolin Liu *, Siok Wei Tay and Xu Li Institute
Supplementary Information for: Novel concept of rechargeable battery using iron oxide nanorods anode and nickel hydroxide cathode in aqueous electrolyte Zhaolin Liu *, Siok Wei Tay and Xu Li Institute
Participation, Growth and Social Poverty: Social Capital in a Homogeneous
 The Open Economic Journal, 2008,, -3 Open Acce Participation, Growth and Social Poverty: Social Capital in a Homogeneou Society Angelo Antoci *,, Pier Luigi Sacco 2 and Paolo Vanin 3 DEIR, Univerity of
The Open Economic Journal, 2008,, -3 Open Acce Participation, Growth and Social Poverty: Social Capital in a Homogeneou Society Angelo Antoci *,, Pier Luigi Sacco 2 and Paolo Vanin 3 DEIR, Univerity of
Solid-state thermoelectric (TE) converters are recently
 Experimental Studies on Anisotropic Thermoelectric Properties and Structures of n-type Bi 2 Te 2.7 Se 0.3 Xiao Yan, Bed Poudel, Yi Ma, W. S. Liu, G. Joshi, Hui Wang, Yucheng Lan, Dezhi Wang, Gang Chen,*,
Experimental Studies on Anisotropic Thermoelectric Properties and Structures of n-type Bi 2 Te 2.7 Se 0.3 Xiao Yan, Bed Poudel, Yi Ma, W. S. Liu, G. Joshi, Hui Wang, Yucheng Lan, Dezhi Wang, Gang Chen,*,
Narrow Gap Welding, Electrogas - and. Electroslag Welding
 6. Narrow Gap Welding, Electroga - and Electrolag Welding 00 6. Narrow Gap Welding, Electroga- and Electrolag Welding 7 Up to thi day, there i no univeral agreement about the definition of the term Narrow
6. Narrow Gap Welding, Electroga - and Electrolag Welding 00 6. Narrow Gap Welding, Electroga- and Electrolag Welding 7 Up to thi day, there i no univeral agreement about the definition of the term Narrow
Preparation of Porous NiO-Ce 0.8 Sm 0.2 O 1.9 Ceramics for Anode-supported Low-temperature Solid Oxide Fuel Cells
 J. Mater. Sci. Technol., 21, 26(6), 523-528. Preparation of Porous NiO-Ce.8 Sm.2 O 1.9 Ceramics for Anode-supported Low-temperature Solid Oxide Fuel Cells Han Chen, Kui Cheng, Zhicheng Wang, Wenjian Weng,
J. Mater. Sci. Technol., 21, 26(6), 523-528. Preparation of Porous NiO-Ce.8 Sm.2 O 1.9 Ceramics for Anode-supported Low-temperature Solid Oxide Fuel Cells Han Chen, Kui Cheng, Zhicheng Wang, Wenjian Weng,
Electronic Supporting Information
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supporting Information Copyright Royal Society of Chemistry, London,
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supporting Information Copyright Royal Society of Chemistry, London,
Microstructural Evolution of Ti-Mo-Ni-C Powder by Mechanical Alloying
 Materials Transactions, Vol. 50, No. 1 (2009) pp. 117 to 122 #2009 The Japan Institute of Metals Microstructural Evolution of -Mo-Ni-C Powder by Mechanical Alloying Hiroyuki Hosokawa, Kiyotaka Kato, Koji
Materials Transactions, Vol. 50, No. 1 (2009) pp. 117 to 122 #2009 The Japan Institute of Metals Microstructural Evolution of -Mo-Ni-C Powder by Mechanical Alloying Hiroyuki Hosokawa, Kiyotaka Kato, Koji
S T A N D A R D. ASAE EP496.3 FEB2006 Agricultural Machinery Management
 SE EP496.3 FEB2006 gricultural Machinery Management S T N D R D SBE i a profeional and technical organization, of member worldwide, who are dedicated to advancement of engineering applicable to agricultural,
SE EP496.3 FEB2006 gricultural Machinery Management S T N D R D SBE i a profeional and technical organization, of member worldwide, who are dedicated to advancement of engineering applicable to agricultural,
Adaptive Description of ATM Traffic Flows 1
 Adaptive Decription of ATM Traffic Flow 1 Juergen Rochol Mouriac Halen Diemer (juergen@inf.ufrg.br) (mouriac@univate.br) Univeridade Federal do Rio Grande do Sul - UFRGS Intituto de Informática - Departamento
Adaptive Decription of ATM Traffic Flow 1 Juergen Rochol Mouriac Halen Diemer (juergen@inf.ufrg.br) (mouriac@univate.br) Univeridade Federal do Rio Grande do Sul - UFRGS Intituto de Informática - Departamento
Power to Gas (& liquids)
 Downloaded from orbit.dtu.dk on: Jan 28, 2018 Power to Gas (& liquids) Holtappels, Peter Publication date: 2013 Link back to DTU Orbit Citation (APA): Holtappels, P. (2013). Power to Gas (& liquids) [Sound/Visual
Downloaded from orbit.dtu.dk on: Jan 28, 2018 Power to Gas (& liquids) Holtappels, Peter Publication date: 2013 Link back to DTU Orbit Citation (APA): Holtappels, P. (2013). Power to Gas (& liquids) [Sound/Visual
MICROFIL Injection Compounds
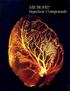 MICROFIL Injection Compound MICROFIL compound will fill and opacify microvacular and other pace of non-urviving animal and potmortem tiue under phyiological injection preure. The continuou, cloed vacular
MICROFIL Injection Compound MICROFIL compound will fill and opacify microvacular and other pace of non-urviving animal and potmortem tiue under phyiological injection preure. The continuou, cloed vacular
International Journal of Chemical Studies
 ISSN: 2321-4902 Volume 1 Issue 1 Online Available at www.chemijournal.com International Journal of Chemical Studies Calculation of Diffusion Coefficients and Layer Thickness for Oxidation the Ferrocene
ISSN: 2321-4902 Volume 1 Issue 1 Online Available at www.chemijournal.com International Journal of Chemical Studies Calculation of Diffusion Coefficients and Layer Thickness for Oxidation the Ferrocene
Conductivity Studies of PMMA/Al 2 O 3 Composite
 Conductivity Studies of PMMA/Al 2 O 3 Composite S.Devikala*, P. Kamaraj and M. Arthanreeswari. Department of chemistry, SRM University, Kattankulathur, Tamil Nadu, India *Corresponding author s mail id:
Conductivity Studies of PMMA/Al 2 O 3 Composite S.Devikala*, P. Kamaraj and M. Arthanreeswari. Department of chemistry, SRM University, Kattankulathur, Tamil Nadu, India *Corresponding author s mail id:
Modelling of a small scale reciprocating ORC expander for cogeneration applications
 Downloaded from orbit.dtu.dk on: Nov 22, 2017 Modelling of a small scale reciprocating ORC expander for cogeneration applications Wronski, Jorrit; Oudkerk, Jean-Francois ; Haglind, Fredrik Publication
Downloaded from orbit.dtu.dk on: Nov 22, 2017 Modelling of a small scale reciprocating ORC expander for cogeneration applications Wronski, Jorrit; Oudkerk, Jean-Francois ; Haglind, Fredrik Publication
Conductivity and Dielectric Studies of PMMA Composites
 Chem Sci Trans., 2013, 2(S1), S129-S134 Chemical Science Transactions DOI:10.7598/cst2013.26 ISSN/E-ISSN: 2278-3458/2278-3318 RESEARCH ARTICLE Conductivity and Dielectric Studies of PMMA Composites S.
Chem Sci Trans., 2013, 2(S1), S129-S134 Chemical Science Transactions DOI:10.7598/cst2013.26 ISSN/E-ISSN: 2278-3458/2278-3318 RESEARCH ARTICLE Conductivity and Dielectric Studies of PMMA Composites S.
CHARACTARISTICS OF DAMAGE AND FRACTURE PROCESS OF SOLID OXIDE FUEL CELLS UNDER SIMULATED OPERATING CONDITIONS BY USING AE METHOD
 CHARACTARISTICS OF DAMAGE AND FRACTURE PROCESS OF SOLID OXIDE FUEL CELLS UNDER SIMULATED OPERATING CONDITIONS BY USING AE METHOD KAZUHISA SATO 1), TOSHIYUKI HASHIDA 2), HIROO YUGAMI 3), KEIJI YASHIRO 1),
CHARACTARISTICS OF DAMAGE AND FRACTURE PROCESS OF SOLID OXIDE FUEL CELLS UNDER SIMULATED OPERATING CONDITIONS BY USING AE METHOD KAZUHISA SATO 1), TOSHIYUKI HASHIDA 2), HIROO YUGAMI 3), KEIJI YASHIRO 1),
Behavior of dielectric constant and dielectric loss tangent in Cd 2+ and Cr 3+ substituted magnesium ferrites
 Available online at www.pelagiaresearchlibrary.com Advances in Applied Science Research, 2013, 4(4):335-339 ISSN: 0976-8610 CODEN (USA): AASRFC Behavior of dielectric constant and dielectric loss tangent
Available online at www.pelagiaresearchlibrary.com Advances in Applied Science Research, 2013, 4(4):335-339 ISSN: 0976-8610 CODEN (USA): AASRFC Behavior of dielectric constant and dielectric loss tangent
CANOPY TRANSPIRATION RESPONSE TO ENVIRONMENTAL VARIATIONS IN PLATYCLADUS ORIENTALIS: PROPERTIES AND MODELLING
 Pak. J. Bot., 44(2): 541-545, 2012. CANOPY TRANSPIRATION RESPONSE TO ENVIRONMENTAL VARIATIONS IN PLATYCLADUS ORIENTALIS: PROPERTIES AND MODELLING LEI HAN 1,2, KANGNING HE 2*, XINGBO HU 2 AND DONG ZHANG
Pak. J. Bot., 44(2): 541-545, 2012. CANOPY TRANSPIRATION RESPONSE TO ENVIRONMENTAL VARIATIONS IN PLATYCLADUS ORIENTALIS: PROPERTIES AND MODELLING LEI HAN 1,2, KANGNING HE 2*, XINGBO HU 2 AND DONG ZHANG
High temperature thermoelectric properties of the Ca 32x B x Co 4 O 9 system
 J Mater Sci: Mater Electron (2013) 24:4406 4410 DOI 10.1007/s10854-013-1417-6 High temperature thermoelectric properties of the Ca 32x B x Co 4 O 9 system S. Demirel S. Altin M. A. Aksan Received: 19 June
J Mater Sci: Mater Electron (2013) 24:4406 4410 DOI 10.1007/s10854-013-1417-6 High temperature thermoelectric properties of the Ca 32x B x Co 4 O 9 system S. Demirel S. Altin M. A. Aksan Received: 19 June
Further development of indicators in the performance measurement frameworks
 Meeting of the Board 8 10 March 2016 Songdo, Incheon, Republic of Korea Proviional agenda item 20 GCF/B.12/13 1March 2016 Further development of indicator in the performance meaurement framework Summary
Meeting of the Board 8 10 March 2016 Songdo, Incheon, Republic of Korea Proviional agenda item 20 GCF/B.12/13 1March 2016 Further development of indicator in the performance meaurement framework Summary
The Arcor/Bagley Merger and the Argentine Biscuit Market: Price Increases vs. Cost Reductions
 The rcor/agley Merger and the rgentine icuit Market: rice Increae v. Cot Reduction Germán Coloma* Thi paper analyze the behavior of the rgentine bicuit market during 2003-06 to find out whether any important
The rcor/agley Merger and the rgentine icuit Market: rice Increae v. Cot Reduction Germán Coloma* Thi paper analyze the behavior of the rgentine bicuit market during 2003-06 to find out whether any important
