File S1. Detailed protocol for CRISPR/Cas9 mediated genome editing with dual marker cassettes
|
|
|
- Brianne Sabina Lyons
- 5 years ago
- Views:
Transcription
1 File S1 Detailed protocol for CRISPR/Cas9 mediated genome editing with dual marker cassettes A) Preparing sgrna vectors We ve modified the strategy to create new sgrna vectors starting with the klp 12 vector as a template. The strategy from the original papers works just fine as well, but these modifications reduce the amount of work involved. We thank Arneet Saltzman for suggesting this protocol. In the new strategy we amplify around the plasmid by PCR, incorporating the new targeting sequence in a forward PCR primer, and a universal reverse primer. Following PCR amplification of the vector, the free 5 ends are phosphorylated and then ligated in sequential steps with no need to clean up the PCR product. An aliquot of the ligation reaction then goes directly into a transformation reaction. Minipreps are performed on a few clones and validated directly by sequencing. Usually nearly every clone we sequence has the new target sequence incorporated. New primer considerations sgrna specific F primer: 5 G(N20)GTTTTAGAGCTAGAAATAGCAAG 3 * Where N20 is the new 20mer targeting sequence to introduce *Notes: It has been found that a terminal GG at positions N19 and N20 can dramatically increase the efficiency of targeting (FARBOUD and MEYER 2015). The G in front of the N20 is only required if the N1 nucleotide is not a G. This G is only present to ensure that the transcription start site from the U6 promoter begins with a G. If nucleotide N1 is a G, then the primer should be of the form: 5 (N20)GTTTTAGAGCTAGAAATAGCAAG 3 sgrna universal R primer: 5 AAACATTTAGATTTGCAATTCAATTA 3 2 SI A. D. Norris et al.
2 Protocol: 1. Prepare 50 ul Phusion reaction on ice according to manufacturer s instructions: MasterMix for 1x (50uL) ddh2o xHF Buffer 10 10mM dntps 1 sgrna universal R primer(10um) 2.5 klp 12 sgrna vector (20pg/uL) 1 Phusion polymerase ul Individual items: sgrna specific F primer(10um) ul 2. PCR settings s s o 10 30s min45s 5. go to step 2, 9 times s o 30s min45s 9. go to step 6, 14 times min hold 3. Run 5 ul of PCR reaction out on agarose gel. Should see a clean band. If not you can still move forward with protocol or consider increasing cycle number to 30 or 35, although this could risk increasing chance of point mutations. 4. Purify reaction using any PCR purification column, and elute with 26 ul elution buffer with 2 min incubation on column at room temperature. 5. To purified DNA, add 3 ul 10x T4 DNA Ligase* buffer (NEB), and add 0.5 ul T4 PNK (NEB). A. D. Norris et al. 3 SI
3 *Note: 10x T4 ligase buffer is identical to 10x PNK buffer except it already has the ATP in it (1mM at 1x) (needed by both enzymes but normally not added to PNK buffer for flexibility in downstream applications). 6. Pipette or flick and quick spin to mix. Incubate 37 C for 20 min. 7. Remove 10 ul for No ligase control. 8. To rest of DNA (~19uL), add 1 ul T4 DNA ligase (NEB). Pipette or flick and quick spin to mix. 9. Incubate room temperature 1 hr. 10. Transform competent E. coli (e.g. DH5α or equivalent) with 1uL of reaction and control (No Ligase). 11. Should see many colonies on reaction plates and very few colonies on control plates. Pick 1 3 single colonies to miniprep and validate insertion of new protospacer directly by sequencing (can use M13 F primer that is available at most companies). B) Preparing homology arm repair vectors 1. Digest the vectors* and alkaline phosphatase treatment. 1a. The Pmyo 2::GFP neor loxp disruption/deletion vector is cut sequentially with SacII (Thermo) followed by cleanup with a PCR purification kit, followed by the NotI fastdigest enzyme (Thermo). SacII has much better activity in its own buffer. We usually digest a big batch of this vector so that it s good for many Gibson assembly reactions. ddh2o 10x SacII Buffer SacII enzyme Pmyo 2::GFP neor loxp 34 x ul 4uL 2uL x ul (2ug) 40 ul Incubate at 37 C for 4 hours, run through PCR purification column and elute in 34 ul elution buffer or water, then set up second digest: 10x FastDigest Buffer FastDigest NotI enzyme 4uL 2uL 4 SI A. D. Norris et al.
4 Pmyo 2::GFP neor loxp 34 ul 40 ul Incubate at 37 C for 2 hours and 30 minutes, then add 1 ul of FastAP (Thermo alkaline phosphatase), incubate at 37 C for another 30 minutes, and run sample through PCR purification column. Nanodrop DNA to assess yield. 1b. The GFP, RFP, 3xHA, and GFP::3xHA Pmyo 2::GFP neor loxp tagging vectors have been modified such that they can be cut in one step with FastDigest SpeI and FastDigest NotI enzymes (Thermo). ddh2o 10x FastDigest Buffer FastDigest SpeI enzyme FastDigest NotI enzyme Tagging vector 32 x ul 4uL 2uL 2uL x ul (2ug) 40 ul Incubate at 37 C for 2 hours and 30 minutes, then add 1 ul of FastAP (Thermo alkaline phosphatase), incubate at 37 C for another 30 minutes, and run sample through PCR purification column. Nanodrop DNA to assess yield. *NOTE: Vectors prepared this way will liberate the Pmyo 2::GFP neor selection cassette which is then left together with the vector in the same tube. These digested vector and cassette fragments are then assembled with the homology arms in one 4 piece Gibson Assembly reaction. While this does generally work well, it s less efficient in our hands than sequentially cloning in each homology arm by Gibson Assembly. As an alternative approach you can cut with the individual enzymes and insert the homology arms one at a time, which works very efficiently but has the drawback that you need multiple steps to generate the vector. In this latter approach, you need to make sure the order of inserting the homology arms is compatible with the enzymes being used to cut the vector (i.e SacII/NotI or SpeI/NotI). C) Preparing Homology arms 1. Designing primers We are cloning everything by Gibson Assembly, so primers* are designed to have 25 nucleotides of overlap at their ends to the relevant regions of the digested vectors: Pmyo 2::GFP neor loxp (disruption/deletion vector) primers Upstream F: AACGACGGCCAGTGAATTCCCGCGG(Nx) Upstream R: TTATAGGCCGCCTGATGCGCCGCGG(Nx RC) Downstream F: ATAGTTGCAGGACCACTGCGGCCGC(Nx) A. D. Norris et al. 5 SI
5 Downstream R: ATGATTACGCCAAGCTTGCGGCCGC(Nx RC) Where Nx and Nx RC are homology arm specific sense and reverse complementary sequences, respectively. GFP Pmyo 2::GFP neor loxp (GFP tagging vector) primers Upstream F: AACGACGGCCAGTGAATTCACTAGT(Nx) Upstream R: GTTCTTCTCCTTTACTCATACTAGT(Nx RC) Downstream F: TGGATGAACTATACAAAGCGGCCGC(Nx) Downstream R: ATGATTACGCCAAGCTTGCGGCCGC(Nx RC) RFP Pmyo 2::GFP neor loxp (RFP tagging vector) primers Upstream F: AACGACGGCCAGTGAATTCACTAGT(Nx) Upstream R: CTTCACCCTTTGAGACCATACTAGT(Nx RC) Downstream F: TGGATGAATTGTATAAGGCGGCCGC(Nx) Downstream R: ATGATTACGCCAAGCTTGCGGCCGC(Nx RC) 3xHA Pmyo 2::GFP neor loxp (3xHA tagging vector) primers Upstream F: AACGACGGCCAGTGAATTCACTAGT(Nx) Upstream R: CTGGAACGTCGTATGGGTAACTAGT(Nx RC) Downstream F: ACGTCCCAGATTACGCTGCGGCCGC(Nx) Downstream R: ATGATTACGCCAAGCTTGCGGCCGC(Nx RC) GFP 3xHA Pmyo 2::GFP neor loxp (GFP 3xHA tagging vector) primers Upstream F: AACGACGGCCAGTGAATTCACTAGT(Nx) Upstream R: GTTCTTCTCCTTTACTCATACTAGT(Nx RC) Downstream F: ACGTCCCAGATTACGCTGCGGCCGC(Nx) Downstream R: ATGATTACGCCAAGCTTGCGGCCGC(Nx RC) *Notes: 1) When designing homology arms, need to consider if you will need to mutate the PAM in order to avoid having the repair plasmid DNA also be a target for cleavage. For the Pmyo 2::GFP neor loxp deletion vector, we usually design our homology arms to be outside the region of cleavage, so this is typically not an issue. But for the endogenous tagging vectors, you will need to pay attention to this. 2) The primers above for the tagging vectors are designed for C terminal fusions. The GFP, RFP, and GFP 3xHA ORFs have start codons built into the vector, but they do not have stop codons (see plasmid maps for reference). You will need to pay attention to make sure your final edited locus will have appropriate start and stop codons where you want them and if you are making an N or C terminal fusion protein strain. 6 SI A. D. Norris et al.
6 The 3xHA ORF has no start or stop codons, so you will need to also design your homology arms accordingly depending on if you want to make an N or C terminal tagged fusion protein strain. 2. PCR amplification of homology arms There are several options for amplifying homology arms. We have had success with amplifying directly out of genomic DNA or plasmid/fosmid DNA. Alternatively, you could also design gblocks in situations where amplicons are difficult to obtain. We usually gel purify the PCR products if they are not very clean (i.e multiple bands on gel). D) Gibson Assembly of Homology Arms with vector and selection cassette 1. Set up assembly reaction and control in PCR tubes assembly reaction: ddh2o Cut vector + selection cassette Upstream homology arm Downstream homology arm 2x Master Assembly mix control reaction: ddh2o Cut vector + selection cassette 10 x y z ul x ul (100ng)* y ul (100ng) z ul (100ng) 10uL 20 ul 20 x ul x ul (100ng) 20 ul 2. Incubate at 50 C for 1 hour in thermal cycler and transform into DH5α or equivalent competent cells. 3. Screen and validate clones that have both homology arms and selection cassette inserted. *Notes: 1) These amounts for the assembly reaction are just rough estimates. You can scale down accordingly if you don t have as much material. Protocol suggests two fold molar excess of inserts, but it has worked well in our hands using the amounts above. 2) We have recently switched over to the NEB HiFi assembly kit since NEB claims it s more efficient for multi piece assembly with smaller amounts of material, and it s the same price as the Gibson mix. A. D. Norris et al. 7 SI
7 E) Microinjection of vectors and drug selection/screening 1. Set up injection mix: Plasmid Final concentration (ng/ul) *Peft 3::Cas9_SV40_NLS::tbb 2_UTR 50 pu6::sgrna vector 100 Repair template 50 *pcfj90 (Pmyo 2::cherry) 2.5 *pcfj104 (Pmyo 3::cherry) 5 *Notes We recommend making high quality DNA preps of the vectors with asterisks (midiprep for Cas9 vector because a lot of it is used in each injection and Invitrogen Purelink HQ for pcfj90 and pcfj104). For the repair template and sgrna vectors however, high quality DNA preps are good but can be replaced by standard minipreps with an extra purification step. When we use sgrna vectors prepped by our standard GeneJET minipreps (Thermo Scientific), we see a lot of sterility in the injected P0 mothers. Midipreps can be a bit time consuming and wasteful, especially if the sgrna and repair template vectors are only used once. We have recently taken our GeneJET miniprep prepared sgrna and repair template vectors and passed them through a Zymo clean and concentrator 5 kit and elute with water, and this effectively eliminated the sterility effect and gave really good transgenesis results. We would recommend this extra step as a convenient method for cleaning up and concentrating the sgrna vectors from standard miniprep kits. We are not sure if other standard miniprep kits give similar toxicity/sterility, but in our hands this extra cleanup step makes a huge difference from no transgenics to many. 2. Inject animals. We typically will aim for animals if injections usually lead to most P0s surviving and throwing ~25 30 Ex F1 progeny each by 24 hours post injections (we typically inject both gonad arms of most animals). In general, we aim for seeing several hundred transgenic extra chromosomal F1 progeny across all injected animals. It s recommended to inject more animals if injections do not generally yield large amounts of extra chromosomal F1 progeny. Alternatively, some extra practice at injections with one of our positive control injection mixes will be helpful for future injection success. 3. After injection, let animals recover for a half hour to one hour at room temp. Then move 3 4 adults per plate and place in 25 C incubator. 4. Day after injection and recovery, add 500 ul** of 25 mg/ml G418 (Sigma) to each plate. 8 SI A. D. Norris et al.
8 **Note A recent protocol using G418 selection states that for best selection conditions, you should weigh your agar plates (FROKJAER JENSEN et al. 2014). A standard 6 cm plate contains about 8g of agar, and that is what this 500 ul volume is designed for. If your plates weigh dramatically different, you can scale the amount of drug added. 5. Swirl drug over surface of plate to ensure complete coating. Transfer plates back to 25 C incubator with the lids removed to allow the drug solution to soak in. Check after a half hour and an hour. When plates are completely dry, return lids and continue incubation at 25 C for the next week to ten days. 6. About three to five days after adding drugs*, should primarily see transgenic animals on plates (Ex and possible integrants)**. Real CRISPR integrant animals should be distinguishable from Ex animals because they will usually have dim GFP pharyngeal expression*** that are completely fluorescent and uniform, and they will also lack the mcherry co injection marker expression. Extra chromosomal array animals will have mosaic and very bright GFP expression in the pharynx, and will have one or both of the cherry expressing markers. *Note The earliest we have seen integrants start to appear is four days post injection. This is usually most obvious by day five and six, but we have also seen later arising transgenics in days We recommend checking for insertions up until plates starve out. **Note In our hands, the G418 treatment does not lead to perfect selection, but rather serves as an enrichment scheme. We do notice non transgenic wild type escapers present in our populations. Screening for pharyngeal GFP expression is thus critical for success in the procedure. ***Note Occasionally we also observe bona fide integrants that have much brighter GFP fluorescence. These animals are still easy to spot, because they lack mcherry fluorescence markers and have uniformly bright and complete pharyngeal GFP expression. 7. Single 6 8 candidate integrants to isolate homozygous or heterozygous animals. If marker is truly heritable, you will see 75% of progeny (heterozygous) or 100% of the progeny (homozygous) with pharyngeal GFP expression. F) Excision of the dual marker cassette with Cre recombinase Homozygous animals* can be used to excise the selection cassette by microinjecting Peft 3::Cre recombinase (DICKINSON et al. 2013). With our dual marker cassette, you A. D. Norris et al. 9 SI
9 can screen for loss of GFP expression in the pharynx of F2 animals to isolate homozygous animals that have excised the selection cassette. *Note If working with essential genes and Cre mediated excision is required, it is recommended to cross insertion animals into an appropriate balancer strain to propagate heterozygotes and facilitate differentiating animals with alleles that have truly excised the dual marker cassette versus animals with the wild type allele. 1. Set up injection mix Plasmid Final concentration (ng/ul) *Peft 3::Cre (pdd104) 50 *pcfj90 (Pmyo 2::cherry) 2.5 *Note We prepare these vectors using the Invitrogen purelink HQ kit 2. Inject animals. We typically inject 5 10 animals. 3. After injection, let animals recover for a half hour to one hour at room temp. Then move 3 4 adults per plate and place in 25 C incubator. 4. Two days after injection, pick F1 progeny that are expressing the Pmyo 2::mCherry marker. Transfer 4 5 worms per plate and return for growth at 25 C. 5. Two three days after picking F1 animals, screen plates for F2 progeny that have completely lost pharyngeal GFP expression and have also lost mcherry expression. These animals should now be homozygous for the excised dual marker cassette. 6. Propagate animals for appropriate downstream applications. 10 SI A. D. Norris et al.
10 References DICKINSON, D. J., J. D. WARD, D. J. REINER and B. GOLDSTEIN, 2013 Engineering the Caenorhabditis elegans genome using Cas9 triggered homologous recombination. Nat Methods 10: FARBOUD, B., and B. J. MEYER, 2015 Dramatic enhancement of genome editing by CRISPR/Cas9 through improved guide RNA design. Genetics 199: FROKJAER JENSEN, C., M. W. DAVIS, M. SAROV, J. TAYLOR, S. FLIBOTTE et al., 2014 Random and targeted transgene insertion in Caenorhabditis elegans using a modified Mos1 transposon. Nat Methods 11: A. D. Norris et al. 11 SI
Construct Design and Cloning Guide for Cas9-triggered homologous recombination
 Construct Design and Cloning Guide for Cas9-triggered homologous recombination Written by Dan Dickinson (ddickins@live.unc.edu) and last updated December 2013. Reference: Dickinson DJ, Ward JD, Reiner
Construct Design and Cloning Guide for Cas9-triggered homologous recombination Written by Dan Dickinson (ddickins@live.unc.edu) and last updated December 2013. Reference: Dickinson DJ, Ward JD, Reiner
Protocol for cloning SEC-based repair templates using Gibson assembly and ccdb negative selection
 Protocol for cloning SEC-based repair templates using Gibson assembly and ccdb negative selection Written by Dan Dickinson (daniel.dickinson@austin.utexas.edu) and last updated January 2018. A version
Protocol for cloning SEC-based repair templates using Gibson assembly and ccdb negative selection Written by Dan Dickinson (daniel.dickinson@austin.utexas.edu) and last updated January 2018. A version
Protocols for cloning SEC-based repair templates using SapTrap assembly
 Protocols for cloning SEC-based repair templates using SapTrap assembly Written by Dan Dickinson (ddickins@live.unc.edu) and last updated July 2016. Overview SapTrap (Schwartz and Jorgensen, 2016) is a
Protocols for cloning SEC-based repair templates using SapTrap assembly Written by Dan Dickinson (ddickins@live.unc.edu) and last updated July 2016. Overview SapTrap (Schwartz and Jorgensen, 2016) is a
Protocols for cloning SEC-based repair templates using SapTrap assembly
 Protocols for cloning SEC-based repair templates using SapTrap assembly Written by Dan Dickinson (daniel.dickinson@austin.utexas.edu) and last updated January 2018. Overview SapTrap (Schwartz and Jorgensen,
Protocols for cloning SEC-based repair templates using SapTrap assembly Written by Dan Dickinson (daniel.dickinson@austin.utexas.edu) and last updated January 2018. Overview SapTrap (Schwartz and Jorgensen,
Gateway Cloning Protocol (Clough Lab Edition) This document is a modification of the Gateway cloning protocol developed by Manju in Chris Taylor's lab
 Gateway Cloning Protocol (Clough Lab Edition) This document is a modification of the Gateway cloning protocol developed by Manju in Chris Taylor's lab With the Gateway cloning system, a PCR fragment is
Gateway Cloning Protocol (Clough Lab Edition) This document is a modification of the Gateway cloning protocol developed by Manju in Chris Taylor's lab With the Gateway cloning system, a PCR fragment is
Conversion of plasmids into Gateway compatible cloning
 Conversion of plasmids into Gateway compatible cloning Rafael Martinez 14072011 Overview: 1. Select the right Gateway cassette (A, B or C). 2. Design primers to amplify the right Gateway cassette from
Conversion of plasmids into Gateway compatible cloning Rafael Martinez 14072011 Overview: 1. Select the right Gateway cassette (A, B or C). 2. Design primers to amplify the right Gateway cassette from
Supplementary Information
 Supplementary Information Vidigal and Ventura a wt locus 5 region 3 region CCTCTGCCACTGCGAGGGCGTCCAATGGTGCTTG(...)AACAGGTGGAATATCCCTACTCTA predicted deletion clone 1 clone 2 clone 3 CCTCTGCCACTGCGAGGGCGTC-AGGTGGAATATCCCTACTCTA
Supplementary Information Vidigal and Ventura a wt locus 5 region 3 region CCTCTGCCACTGCGAGGGCGTCCAATGGTGCTTG(...)AACAGGTGGAATATCCCTACTCTA predicted deletion clone 1 clone 2 clone 3 CCTCTGCCACTGCGAGGGCGTC-AGGTGGAATATCCCTACTCTA
Data Sheet Quick PCR Cloning Kit
 Data Sheet Quick PCR Cloning Kit 6044 Cornerstone Ct. West, Ste. E DESCRIPTION: The Quick PCR Cloning Kit is a simple and highly efficient method to insert any gene or DNA fragment into a vector, without
Data Sheet Quick PCR Cloning Kit 6044 Cornerstone Ct. West, Ste. E DESCRIPTION: The Quick PCR Cloning Kit is a simple and highly efficient method to insert any gene or DNA fragment into a vector, without
In this edition of Marketplace Special offers on Thermo Scientific products:
 In this edition of Marketplace Special offers on Thermo Scientific products: Cloning Enzymes and Kits FastDigest Enzymes GeneJET Nucleic Acid Purification Kits TopVision Agarose Tablets PCR Plastic Consumables
In this edition of Marketplace Special offers on Thermo Scientific products: Cloning Enzymes and Kits FastDigest Enzymes GeneJET Nucleic Acid Purification Kits TopVision Agarose Tablets PCR Plastic Consumables
Hetero-Stagger PCR Cloning Kit
 Product Name: Code No: Size: DynaExpress Hetero-Stagger PCR Cloning Kit DS150 20 reactions Kit Components: Box 1 (-20 ) phst-1 Vector, linearized Annealing Buffer Ligase Mixture phst Forward Sequence Primer
Product Name: Code No: Size: DynaExpress Hetero-Stagger PCR Cloning Kit DS150 20 reactions Kit Components: Box 1 (-20 ) phst-1 Vector, linearized Annealing Buffer Ligase Mixture phst Forward Sequence Primer
Creating pentr vectors by BP reaction
 Creating pentr vectors by BP reaction Tanya Lepikhova and Rafael Martinez 15072011 Overview: 1. Design primers to add the attb sites to gene of interest 2. Perform PCR with a high fidelity DNA polymerase
Creating pentr vectors by BP reaction Tanya Lepikhova and Rafael Martinez 15072011 Overview: 1. Design primers to add the attb sites to gene of interest 2. Perform PCR with a high fidelity DNA polymerase
Supplemental Text: Detailed protocol for generating Cas9-mediated fluorescent protein knock-ins with a self-excising selection cassette (SEC)
 Supplemental Text: Detailed protocol for generating Cas9-mediated fluorescent protein knock-ins with a self-excising selection cassette (SEC) Before the Experiment Choose the Cas9 target site 1) Identify
Supplemental Text: Detailed protocol for generating Cas9-mediated fluorescent protein knock-ins with a self-excising selection cassette (SEC) Before the Experiment Choose the Cas9 target site 1) Identify
Supplementary Methods pcfd5 cloning protocol
 Supplementary Methods cloning protocol vermilion trna grna trna grna U6:3 Terminator AmpR attb is a vector for expressing one or multiple trna-flanked Cas9 grnas under the control of the strong, ubiquitous
Supplementary Methods cloning protocol vermilion trna grna trna grna U6:3 Terminator AmpR attb is a vector for expressing one or multiple trna-flanked Cas9 grnas under the control of the strong, ubiquitous
Polymerase Chain Reaction
 Polymerase Chain Reaction Amplify your insert or verify its presence 3H Taq platinum PCR mix primers Ultrapure Water PCR tubes PCR machine A. Insert amplification For insert amplification, use the Taq
Polymerase Chain Reaction Amplify your insert or verify its presence 3H Taq platinum PCR mix primers Ultrapure Water PCR tubes PCR machine A. Insert amplification For insert amplification, use the Taq
For designing necessary primers and oligos for grna, online CRISPR designing tools can be used (e.g.
 DESIGN OF grnas For designing necessary primers and oligos for grna, online CRISPR designing tools can be used (e.g. http://crispr.mit.edu/, https://benchling.com). Assembly of grna transcriptional cassettes
DESIGN OF grnas For designing necessary primers and oligos for grna, online CRISPR designing tools can be used (e.g. http://crispr.mit.edu/, https://benchling.com). Assembly of grna transcriptional cassettes
peco TM -T7-nGST, Eco cloning Kit User Manual (Patent pending)
 peco TM -T7-nGST, Eco cloning Kit User Manual (Patent pending) Cloning PCR products for E Coli expression of N-term GST-tagged protein Cat# Contents Amounts Application IC-1004 peco-t7-ngst vector built-in
peco TM -T7-nGST, Eco cloning Kit User Manual (Patent pending) Cloning PCR products for E Coli expression of N-term GST-tagged protein Cat# Contents Amounts Application IC-1004 peco-t7-ngst vector built-in
Ben- Shahar Lab CRISPR/RMCE Protocol May 2015
 Ben- Shahar Lab CRISPR/RMCE Protocol May 2015 1. Before you begin a. Useful CRISPR overview for beginners: https://www.addgene.org/crispr/guide/ b. This protocol combines techniques described in these
Ben- Shahar Lab CRISPR/RMCE Protocol May 2015 1. Before you begin a. Useful CRISPR overview for beginners: https://www.addgene.org/crispr/guide/ b. This protocol combines techniques described in these
Protocols for cell lines using CRISPR/CAS
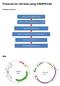 Protocols for cell lines using CRISPR/CAS Procedure overview Map Preparation of CRISPR/CAS plasmids Expression vectors for guide RNA (U6-gRNA) and Cas9 gene (CMV-p-Cas9) are ampicillin-resist ant and stable
Protocols for cell lines using CRISPR/CAS Procedure overview Map Preparation of CRISPR/CAS plasmids Expression vectors for guide RNA (U6-gRNA) and Cas9 gene (CMV-p-Cas9) are ampicillin-resist ant and stable
Perform HiFi PCR Purify PCR-mixture using PCR-clean up kit and determine concentration
 IGEM EXPERIMENT 2 CONSTRUCTION OF PSB#X#-T7CCDB Strategy Cloning Flow Chart (using restriction/ligation): 1. In silico design of cloning Design a cloning strategy (in case of insert/backbone): o Using
IGEM EXPERIMENT 2 CONSTRUCTION OF PSB#X#-T7CCDB Strategy Cloning Flow Chart (using restriction/ligation): 1. In silico design of cloning Design a cloning strategy (in case of insert/backbone): o Using
GenBuilder TM Plus Cloning Kit User Manual
 GenBuilder TM Plus Cloning Kit User Manual Cat. No. L00744 Version 11242017 Ⅰ. Introduction... 2 I.1 Product Information... 2 I.2 Kit Contents and Storage... 2 I.3 GenBuilder Cloning Kit Workflow... 2
GenBuilder TM Plus Cloning Kit User Manual Cat. No. L00744 Version 11242017 Ⅰ. Introduction... 2 I.1 Product Information... 2 I.2 Kit Contents and Storage... 2 I.3 GenBuilder Cloning Kit Workflow... 2
Recombineering Manual
 Recombineering Manual Anthony Popkie The Phiel Laboratory The Research Institute at Nationwide Childrenʼs Hospital 1 BAC Transformation BACs may be transformed into either DY380, EL250 or EL350 cells.
Recombineering Manual Anthony Popkie The Phiel Laboratory The Research Institute at Nationwide Childrenʼs Hospital 1 BAC Transformation BACs may be transformed into either DY380, EL250 or EL350 cells.
GenBuilder TM Plus Cloning Kit User Manual
 GenBuilder TM Plus Cloning Kit User Manual Cat.no L00744 Version 11242017 Ⅰ. Introduction... 2 I.1 Product Information... 2 I.2 Kit Contents and Storage... 2 I.3 GenBuilder Cloning Kit Workflow... 2 Ⅱ.
GenBuilder TM Plus Cloning Kit User Manual Cat.no L00744 Version 11242017 Ⅰ. Introduction... 2 I.1 Product Information... 2 I.2 Kit Contents and Storage... 2 I.3 GenBuilder Cloning Kit Workflow... 2 Ⅱ.
Guide-it Indel Identification Kit User Manual
 Clontech Laboratories, Inc. Guide-it Indel Identification Kit User Manual Cat. No. 631444 (120114) Clontech Laboratories, Inc. A Takara Bio Company 1290 Terra Bella Avenue, Mountain View, CA 94043, USA
Clontech Laboratories, Inc. Guide-it Indel Identification Kit User Manual Cat. No. 631444 (120114) Clontech Laboratories, Inc. A Takara Bio Company 1290 Terra Bella Avenue, Mountain View, CA 94043, USA
Vector Linearization. igem TU/e 2015 Biomedical Engineering
 igem TU/e 2015 Biomedical Engineering Eindhoven University of Technology Room: Ceres 0.04 Den Dolech 2, 5612 AZ Eindhoven The Netherlands Tel. no. +31 50 247 55 59 2015.igem.org/Team:TU_Eindhoven Vector
igem TU/e 2015 Biomedical Engineering Eindhoven University of Technology Room: Ceres 0.04 Den Dolech 2, 5612 AZ Eindhoven The Netherlands Tel. no. +31 50 247 55 59 2015.igem.org/Team:TU_Eindhoven Vector
Guide-it sgrna In Vitro Transcription and Screening Systems User Manual
 Guide-it sgrna In Vitro Transcription and Screening Systems User Manual Cat. Nos. 632638, 632639, 632635, 632636, 632637 (040618) 1290 Terra Bella Avenue, Mountain View, CA 94043, USA U.S. Technical Support:
Guide-it sgrna In Vitro Transcription and Screening Systems User Manual Cat. Nos. 632638, 632639, 632635, 632636, 632637 (040618) 1290 Terra Bella Avenue, Mountain View, CA 94043, USA U.S. Technical Support:
Hernday Lab C. albicans CRISPR System Background:
 Hernday Lab C. albicans CRISPR System Background: This document covers the plasmids and protocols used for the Hernday lab Candida albicans CRISPR system. This system allows the user to edit virtually
Hernday Lab C. albicans CRISPR System Background: This document covers the plasmids and protocols used for the Hernday lab Candida albicans CRISPR system. This system allows the user to edit virtually
Mutagenesis PCR I (Multiple Site Directed Mutagenesis)
 Mutagenesis Mutagenesis PCR I (Multiple Site Directed Mutagenesis) Mixture 25µl total reaction volume : 1. 2.5 µl of 10X Taq lligase buffer (need the NAD for Taq ligase) 2. 0.5 µl 100mM ATP 3. X µl (50-100
Mutagenesis Mutagenesis PCR I (Multiple Site Directed Mutagenesis) Mixture 25µl total reaction volume : 1. 2.5 µl of 10X Taq lligase buffer (need the NAD for Taq ligase) 2. 0.5 µl 100mM ATP 3. X µl (50-100
GenBuilder TM Cloning Kit User Manual
 GenBuilder TM Cloning Kit User Manual Cat.no L00701 Version 11242017 Ⅰ. Introduction... 2 I.1 Product Information... 2 I.2 Kit Contents and Storage... 2 I.3 GenBuilder Cloning Kit Workflow... 2 Ⅱ. DNA
GenBuilder TM Cloning Kit User Manual Cat.no L00701 Version 11242017 Ⅰ. Introduction... 2 I.1 Product Information... 2 I.2 Kit Contents and Storage... 2 I.3 GenBuilder Cloning Kit Workflow... 2 Ⅱ. DNA
Mos1 insertion. MosTIC protocol-11/2006. repair template - Mos1 transposase expression - Mos1 excision - DSB formation. homolog arm.
 MosTIC (Mos1 excision induced Transgene Instructed gene Conversion) Valérie Robert (vrobert@biologie.ens.fr) and Jean-Louis Bessereau (jlbesse@biologie.ens.fr) (November 2006) Introduction: MosTIC (Robert
MosTIC (Mos1 excision induced Transgene Instructed gene Conversion) Valérie Robert (vrobert@biologie.ens.fr) and Jean-Louis Bessereau (jlbesse@biologie.ens.fr) (November 2006) Introduction: MosTIC (Robert
Schematic representation of the endogenous PALB2 locus and gene-disruption constructs
 Supplementary Figures Supplementary Figure 1. Generation of PALB2 -/- and BRCA2 -/- /PALB2 -/- DT40 cells. (A) Schematic representation of the endogenous PALB2 locus and gene-disruption constructs carrying
Supplementary Figures Supplementary Figure 1. Generation of PALB2 -/- and BRCA2 -/- /PALB2 -/- DT40 cells. (A) Schematic representation of the endogenous PALB2 locus and gene-disruption constructs carrying
In order to make our construct pah12 (Mlra) and pah05 (venus) biobrick compatitable PCR was conducted. Following PCR mixes were prepared
 BioBrick cloning Project: BioBricks Authors: Antti Koistinen Dates: 2016-09-15 to 2016-10-06 THURSDAY, 9/15 In order to make our construct pah12 (Mlra) and pah05 (venus) biobrick compatitable PCR was conducted.
BioBrick cloning Project: BioBricks Authors: Antti Koistinen Dates: 2016-09-15 to 2016-10-06 THURSDAY, 9/15 In order to make our construct pah12 (Mlra) and pah05 (venus) biobrick compatitable PCR was conducted.
Ligation Independent Cloning (LIC) Procedure
 Ligation Independent Cloning (LIC) Procedure Ligation Independent Cloning (LIC) LIC cloning allows insertion of DNA fragments without using restriction enzymes into specific vectors containing engineered
Ligation Independent Cloning (LIC) Procedure Ligation Independent Cloning (LIC) LIC cloning allows insertion of DNA fragments without using restriction enzymes into specific vectors containing engineered
XactEdit Cas9 Nuclease with NLS User Manual
 XactEdit Cas9 Nuclease with NLS User Manual An RNA-guided recombinant endonuclease for efficient targeted DNA cleavage Catalog Numbers CE1000-50K, CE1000-50, CE1000-250, CE1001-250, CE1001-1000 Table of
XactEdit Cas9 Nuclease with NLS User Manual An RNA-guided recombinant endonuclease for efficient targeted DNA cleavage Catalog Numbers CE1000-50K, CE1000-50, CE1000-250, CE1001-250, CE1001-1000 Table of
Cold Fusion Cloning Kit. Cat. #s MC100A-1, MC101A-1. User Manual
 Fusion Cloning technology Cold Fusion Cloning Kit Store the master mixture and positive controls at -20 C Store the competent cells at -80 C. (ver. 120909) A limited-use label license covers this product.
Fusion Cloning technology Cold Fusion Cloning Kit Store the master mixture and positive controls at -20 C Store the competent cells at -80 C. (ver. 120909) A limited-use label license covers this product.
ksierzputowska.com Research Title: Using novel TALEN technology to engineer precise mutations in the genome of D. melanogaster
 Research Title: Using novel TALEN technology to engineer precise mutations in the genome of D. melanogaster Research plan: Specific aims: 1. To successfully engineer transgenic Drosophila expressing TALENs
Research Title: Using novel TALEN technology to engineer precise mutations in the genome of D. melanogaster Research plan: Specific aims: 1. To successfully engineer transgenic Drosophila expressing TALENs
Supplementary Information
 Single day construction of multi-gene circuits with 3G assembly Andrew D. Halleran 1, Anandh Swaminathan 2, and Richard M. Murray 1, 2 1. Bioengineering, California Institute of Technology, Pasadena, CA.
Single day construction of multi-gene circuits with 3G assembly Andrew D. Halleran 1, Anandh Swaminathan 2, and Richard M. Murray 1, 2 1. Bioengineering, California Institute of Technology, Pasadena, CA.
Cat # Box1 Box2. DH5a Competent E. coli cells CCK-20 (20 rxns) 40 µl 40 µl 50 µl x 20 tubes. Choo-Choo Cloning TM Enzyme Mix
 Molecular Cloning Laboratories User Manual Version 3.3 Product name: Choo-Choo Cloning Kits Cat #: CCK-10, CCK-20, CCK-096, CCK-384 Description: Choo-Choo Cloning is a highly efficient directional PCR
Molecular Cloning Laboratories User Manual Version 3.3 Product name: Choo-Choo Cloning Kits Cat #: CCK-10, CCK-20, CCK-096, CCK-384 Description: Choo-Choo Cloning is a highly efficient directional PCR
Generation of gene knockout vectors for Dictyostelium discoideum
 Generation of gene knockout vectors for Dictyostelium discoideum Instruction manual Last date of revision April 2012 2015 Version PR29-0001 PR29-0003 www.stargate.com This manual can be downloaded under
Generation of gene knockout vectors for Dictyostelium discoideum Instruction manual Last date of revision April 2012 2015 Version PR29-0001 PR29-0003 www.stargate.com This manual can be downloaded under
Farnham Lab Protocol for CRISPR/Cas 9- mediated enhancer deletion using a puromycin selection vector Created by Yu (Phoebe) Guo
 Farnham Lab Protocol for CRISPR/Cas 9- mediated enhancer deletion using a puromycin selection vector Created by Yu (Phoebe) Guo 20151024 Part A. Design grna oligos 1. Design grnas Go to Optimized CRISPR
Farnham Lab Protocol for CRISPR/Cas 9- mediated enhancer deletion using a puromycin selection vector Created by Yu (Phoebe) Guo 20151024 Part A. Design grna oligos 1. Design grnas Go to Optimized CRISPR
Guide-it sgrna In Vitro Transcription and Screening Systems User Manual
 Clontech Laboratories, Inc. Guide-it sgrna In Vitro Transcription and Screening Systems User Manual Cat. Nos. 631438, 631439 & 631440 (042114) Clontech Laboratories, Inc. A Takara Bio Company 1290 Terra
Clontech Laboratories, Inc. Guide-it sgrna In Vitro Transcription and Screening Systems User Manual Cat. Nos. 631438, 631439 & 631440 (042114) Clontech Laboratories, Inc. A Takara Bio Company 1290 Terra
Rapid amplification of cdna ends (RACE)
 Rapid amplification of cdna ends (RACE) Rapid amplification of cdna ends (RACE) is a technique used in molecular biology to obtain the full length sequence of an RNA transcript found within a cell. RACE
Rapid amplification of cdna ends (RACE) Rapid amplification of cdna ends (RACE) is a technique used in molecular biology to obtain the full length sequence of an RNA transcript found within a cell. RACE
Fast and efficient site-directed mutagenesis with Platinum SuperFi DNA Polymerase
 APPLICATION NOTE Platinum Superi Polymerase ast and efficient site-directed mutagenesis with Platinum Superi Polymerase Introduction Site-directed mutagenesis is one of the most essential techniques to
APPLICATION NOTE Platinum Superi Polymerase ast and efficient site-directed mutagenesis with Platinum Superi Polymerase Introduction Site-directed mutagenesis is one of the most essential techniques to
Illumina Sequencing Sample Preparation for use with CRISPRi/a-v2 Libraries
 Illumina Sequencing Sample Preparation for use with CRISPRi/a-v2 Libraries Overview This protocol describes the four steps required for generating sequencing samples from cells harvested from screens conducted
Illumina Sequencing Sample Preparation for use with CRISPRi/a-v2 Libraries Overview This protocol describes the four steps required for generating sequencing samples from cells harvested from screens conducted
Selected Techniques Part I
 1 Selected Techniques Part I Gel Electrophoresis Can be both qualitative and quantitative Qualitative About what size is the fragment? How many fragments are present? Is there in insert or not? Quantitative
1 Selected Techniques Part I Gel Electrophoresis Can be both qualitative and quantitative Qualitative About what size is the fragment? How many fragments are present? Is there in insert or not? Quantitative
YG1 Control. YG1 PstI XbaI. YG5 PstI XbaI Water Buffer DNA Enzyme1 Enzyme2
 9/9/03 Aim: Digestion and gel extraction of YG, YG3, YG5 and 8/C. Strain: E. coli DH5α Plasmid: Bba_J600, psbc3 4,, 3 6 7, 8 9 0, 3, 4 8/C 8/C SpeI PstI 3 3 x3 YG YG PstI XbaI.5 3.5 x YG3 YG3 PstI XbaI
9/9/03 Aim: Digestion and gel extraction of YG, YG3, YG5 and 8/C. Strain: E. coli DH5α Plasmid: Bba_J600, psbc3 4,, 3 6 7, 8 9 0, 3, 4 8/C 8/C SpeI PstI 3 3 x3 YG YG PstI XbaI.5 3.5 x YG3 YG3 PstI XbaI
An antibiotic selection marker for nematode transgenesis
 nature methods An antibiotic selection marker for nematode transgenesis Rosina Giordano-Santini, Stuart Milstein, Nenad Svrzikapa, Domena Tu, Robert Johnsen, David Baillie, Marc Vidal & Denis Dupuy Supplementary
nature methods An antibiotic selection marker for nematode transgenesis Rosina Giordano-Santini, Stuart Milstein, Nenad Svrzikapa, Domena Tu, Robert Johnsen, David Baillie, Marc Vidal & Denis Dupuy Supplementary
NZYGene Synthesis kit
 Kit components Component Concentration Amount NZYGene Synthesis kit Catalogue number: MB33901, 10 reactions GS DNA Polymerase 1U/ μl 30 μl Reaction Buffer for GS DNA Polymerase 10 150 μl dntp mix 2 mm
Kit components Component Concentration Amount NZYGene Synthesis kit Catalogue number: MB33901, 10 reactions GS DNA Polymerase 1U/ μl 30 μl Reaction Buffer for GS DNA Polymerase 10 150 μl dntp mix 2 mm
Molecular Biology Techniques Supporting IBBE
 Molecular Biology Techniques Supporting IBBE Jared Cartwright Protein Production Lab Head Contact Details: email jared.cartwright@york.ac.uk Phone 01904 328797 Presentation Aims Gene synthesis Cloning
Molecular Biology Techniques Supporting IBBE Jared Cartwright Protein Production Lab Head Contact Details: email jared.cartwright@york.ac.uk Phone 01904 328797 Presentation Aims Gene synthesis Cloning
igem LMU- Munich Munich Cloning procedure
 igem LM- Munich 2014 - igem@bio.lmu.de - http://2014.igem.org/team:lm- Munich Cloning procedure Overview of a cloning procedure: 1. Digestion of a PCR product ( insert) and a vector 2. Dephosphorylation
igem LM- Munich 2014 - igem@bio.lmu.de - http://2014.igem.org/team:lm- Munich Cloning procedure Overview of a cloning procedure: 1. Digestion of a PCR product ( insert) and a vector 2. Dephosphorylation
Targeted Gene Mutation in Rice Using a CRISPR-Cas9 System Kabin Xie 1, Bastian Minkenberg 2 and Yinong Yang 2*
 Targeted Gene Mutation in Rice Using a CRISPR-Cas9 System Kabin Xie 1, Bastian Minkenberg 2 and Yinong Yang 2* 1 Department of Plant Pathology and Environmental Microbiology, Pennsylvania State University,
Targeted Gene Mutation in Rice Using a CRISPR-Cas9 System Kabin Xie 1, Bastian Minkenberg 2 and Yinong Yang 2* 1 Department of Plant Pathology and Environmental Microbiology, Pennsylvania State University,
SUPPLEMENT MATERIALS FOR CURTIN,
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 SUPPLEMENT MATERIALS FOR CURTIN, et al. Validating genome-wide association candidates: Selecting, testing, and characterizing
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 SUPPLEMENT MATERIALS FOR CURTIN, et al. Validating genome-wide association candidates: Selecting, testing, and characterizing
Supplemental Protocols Gagnon et al., 2014
 Last updated April 15th 2014 There may be a more up-to-date version of this protocol on our website http://labs.mcb.harvard.edu/schier/index.html Protocol Page Flowchart for Cas9-mediated mutagenesis 1
Last updated April 15th 2014 There may be a more up-to-date version of this protocol on our website http://labs.mcb.harvard.edu/schier/index.html Protocol Page Flowchart for Cas9-mediated mutagenesis 1
Application of Molecular Biology tools for cloning of a foreign gene
 IFM/Kemi Linköpings Universitet September 2013/LGM Labmanual Project course Application of Molecular Biology tools for cloning of a foreign gene Table of contents Introduction... 3 Amplification of a gene
IFM/Kemi Linköpings Universitet September 2013/LGM Labmanual Project course Application of Molecular Biology tools for cloning of a foreign gene Table of contents Introduction... 3 Amplification of a gene
pgm-t Cloning Kit For direct cloning of PCR products generated by Taq DNA polymerases For research use only Cat. # : GVT202 Size : 20 Reactions
 pgm-t Cloning Kit For direct cloning of PCR products generated by Taq DNA polymerases Cat. # : GVT202 Size : 20 Reactions Store at -20 For research use only 1 pgm-t Cloning Kit Cat. No.: GVT202 Kit Contents
pgm-t Cloning Kit For direct cloning of PCR products generated by Taq DNA polymerases Cat. # : GVT202 Size : 20 Reactions Store at -20 For research use only 1 pgm-t Cloning Kit Cat. No.: GVT202 Kit Contents
Application Note: Generating GFP-Tagged Human CD81 Tetraspanin Protein Using SBI s PrecisionX SmartNuclease System And HR Tagging Vectors
 Application Note: Generating GFP-Tagged Human CD81 Tetraspanin Protein Using SBI s PrecisionX SmartNuclease System And HR Tagging Vectors Table of Contents: I. Background Pg. 1 II. Analysis of Gene Target
Application Note: Generating GFP-Tagged Human CD81 Tetraspanin Protein Using SBI s PrecisionX SmartNuclease System And HR Tagging Vectors Table of Contents: I. Background Pg. 1 II. Analysis of Gene Target
Petra, Tamannae. Made new 1:10 dilutions of P001 and P ul primer to 18 ul water
 20.7.2015 MONDAY, 7/20 Petra, Tamannae Made new 1:10 dilutions of P001 and P015. 2 ul primer to 18 ul water Made new 1 ng/µl template dilution of CAR part 1. 2 µl DNA stock and 18 µl H2O Gradient PCR for
20.7.2015 MONDAY, 7/20 Petra, Tamannae Made new 1:10 dilutions of P001 and P015. 2 ul primer to 18 ul water Made new 1 ng/µl template dilution of CAR part 1. 2 µl DNA stock and 18 µl H2O Gradient PCR for
HE Swift Cloning Kit
 HE Swift Cloning Kit For high-efficient cloning of PCR products either blunt or sticky-end Kit Contents Contents VTT-BB05 phe Vector (35 ng/µl) 20 µl T4 DNA Ligase (3 U/µl) 20 µl 2 Reaction Buffer 100
HE Swift Cloning Kit For high-efficient cloning of PCR products either blunt or sticky-end Kit Contents Contents VTT-BB05 phe Vector (35 ng/µl) 20 µl T4 DNA Ligase (3 U/µl) 20 µl 2 Reaction Buffer 100
Generating stable cell lines expressing various Cas9 proteins from AAVS1 locus
 IGI Protocol prepared by Chong Park 1 Generating stable cell lines expressing various Cas9 proteins from AAVS1 locus This is modified from original protocol from the Conklin laboratory (Mandegar et al.
IGI Protocol prepared by Chong Park 1 Generating stable cell lines expressing various Cas9 proteins from AAVS1 locus This is modified from original protocol from the Conklin laboratory (Mandegar et al.
Introducing new DNA into the genome requires cloning the donor sequence, delivery of the cloned DNA into the cell, and integration into the genome.
 Key Terms Chapter 32: Genetic Engineering Cloning describes propagation of a DNA sequence by incorporating it into a hybrid construct that can be replicated in a host cell. A cloning vector is a plasmid
Key Terms Chapter 32: Genetic Engineering Cloning describes propagation of a DNA sequence by incorporating it into a hybrid construct that can be replicated in a host cell. A cloning vector is a plasmid
Vector Linearization. igem TU/e 2016 Biomedical Engineering
 igem TU/e 2016 Biomedical Engineering Eindhoven University of Technology Room: Ceres 0.04 Den Dolech 2, 5612 AZ Eindhoven The Netherlands Tel. no. +31 50 247 55 59 2016.igem.org/Team:TU_Eindhoven Vector
igem TU/e 2016 Biomedical Engineering Eindhoven University of Technology Room: Ceres 0.04 Den Dolech 2, 5612 AZ Eindhoven The Netherlands Tel. no. +31 50 247 55 59 2016.igem.org/Team:TU_Eindhoven Vector
Enhanced Arginase production: rocf
 Enhanced Arginase production: rocf Purpose and Justification: Bacillus subtilis produces urease, which catalyses the hydrolysis of urea into ammonium and carbonate. Since the cell wall of the bacteria
Enhanced Arginase production: rocf Purpose and Justification: Bacillus subtilis produces urease, which catalyses the hydrolysis of urea into ammonium and carbonate. Since the cell wall of the bacteria
Simple protocol for gene editing using GenCrisprTM Cas9 nuclease
 Simple protocol for gene editing using GenCrisprTM Cas9 nuclease Contents Protocol Step 1: Choose the target DNA sequence Step 2: Design sgrna Step 3: Preparation for sgrna 3.1 In vitro transcription of
Simple protocol for gene editing using GenCrisprTM Cas9 nuclease Contents Protocol Step 1: Choose the target DNA sequence Step 2: Design sgrna Step 3: Preparation for sgrna 3.1 In vitro transcription of
Dramatic enhancement of genome editing by CRISPR/Cas9 through improved guide RNA design
 Genetics: Early Online, published on February 18, 215 as 1.1534/genetics.115.175166 Dramatic enhancement of genome editing by CRISPR/Cas9 through improved guide RNA design Behnom Farboud 1 and Barbara
Genetics: Early Online, published on February 18, 215 as 1.1534/genetics.115.175166 Dramatic enhancement of genome editing by CRISPR/Cas9 through improved guide RNA design Behnom Farboud 1 and Barbara
Design. Construction. Characterization
 Design Construction Characterization DNA mrna (messenger) A C C transcription translation C A C protein His A T G C T A C G Plasmids replicon copy number incompatibility selection marker origin of replication
Design Construction Characterization DNA mrna (messenger) A C C transcription translation C A C protein His A T G C T A C G Plasmids replicon copy number incompatibility selection marker origin of replication
Library Prep Using the With-Bead Method
 Target enrichment library preparation Library Prep Using the With-Bead Method Introduction This protocol is based on the method of Meyer and Kircher (2010) with modifications to accommodate the capturing
Target enrichment library preparation Library Prep Using the With-Bead Method Introduction This protocol is based on the method of Meyer and Kircher (2010) with modifications to accommodate the capturing
Molecular Cell Biology - Problem Drill 11: Recombinant DNA
 Molecular Cell Biology - Problem Drill 11: Recombinant DNA Question No. 1 of 10 1. Which of the following statements about the sources of DNA used for molecular cloning is correct? Question #1 (A) cdna
Molecular Cell Biology - Problem Drill 11: Recombinant DNA Question No. 1 of 10 1. Which of the following statements about the sources of DNA used for molecular cloning is correct? Question #1 (A) cdna
MOLECULAR BIOLOGY GREATEST HITS. Marketplace. Essentials Tour molecular biology. thermofisher.com/marketplace
 molecular biology Marketplace MOLECULAR BIOLOGY GREATEST HITS Special offers on Thermo Scientific products: Cloning kits Restriction and modifying enzymes IPTG and X-Gal DNA ladders Agarose tablets Nucleic
molecular biology Marketplace MOLECULAR BIOLOGY GREATEST HITS Special offers on Thermo Scientific products: Cloning kits Restriction and modifying enzymes IPTG and X-Gal DNA ladders Agarose tablets Nucleic
pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20
 pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20 1 Kit Contents Contents pgm-t Cloning Kit pgm-t Vector (50 ng/μl) 20 μl T4 DNA Ligase (3 U/μl) 20 μl 10X T4 DNA Ligation Buffer 30 μl
pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20 1 Kit Contents Contents pgm-t Cloning Kit pgm-t Vector (50 ng/μl) 20 μl T4 DNA Ligase (3 U/μl) 20 μl 10X T4 DNA Ligation Buffer 30 μl
Nature Biotechnology: doi: /nbt.4166
 Supplementary Figure 1 Validation of correct targeting at targeted locus. (a) by immunofluorescence staining of 2C-HR-CRISPR microinjected embryos cultured to the blastocyst stage. Embryos were stained
Supplementary Figure 1 Validation of correct targeting at targeted locus. (a) by immunofluorescence staining of 2C-HR-CRISPR microinjected embryos cultured to the blastocyst stage. Embryos were stained
Comparison of ExoSAP-IT and ExoSAP-IT Express reagents to alternative PCR cleanup methods
 WHITE PAPER ExoSAP-IT PCR cleanup reagents Comparison of ExoSAP-IT and ExoSAP-IT Express reagents to alternative PCR cleanup methods Abstract Here we present superior workflow advantages of enzymatic PCR
WHITE PAPER ExoSAP-IT PCR cleanup reagents Comparison of ExoSAP-IT and ExoSAP-IT Express reagents to alternative PCR cleanup methods Abstract Here we present superior workflow advantages of enzymatic PCR
Easi CRISPR for conditional and insertional alleles
 Easi CRISPR for conditional and insertional alleles C.B Gurumurthy, University Of Nebraska Medical Center Omaha, NE cgurumurthy@unmc.edu Types of Genome edits Gene disruption/inactivation Types of Genome
Easi CRISPR for conditional and insertional alleles C.B Gurumurthy, University Of Nebraska Medical Center Omaha, NE cgurumurthy@unmc.edu Types of Genome edits Gene disruption/inactivation Types of Genome
Version(1(*(July(14 th,(2014(
 Morrisey(Lab(Protocol:(( Generating(Large((>1kb)(Genomic(Deletions(Using(CRISPRs( bydanswarr&davefrank GeneralComments ThisprotocolisintendedtousetheCRISPR@Cas9systemforgenerationoflarge genomicdeletions(>1kb),inordertoremoveorinactivateagenomicelementof
Morrisey(Lab(Protocol:(( Generating(Large((>1kb)(Genomic(Deletions(Using(CRISPRs( bydanswarr&davefrank GeneralComments ThisprotocolisintendedtousetheCRISPR@Cas9systemforgenerationoflarge genomicdeletions(>1kb),inordertoremoveorinactivateagenomicelementof
PrecisionX Multiplex grna Cloning Kit. Cat. # CAS9-GRNA-KIT. User Manual
 PrecisionX Multiplex grna Cloning Kit Store at -20 C upon receipt A limited-use label license covers this product. By use of this product, you accept the terms and conditions outlined in the Licensing
PrecisionX Multiplex grna Cloning Kit Store at -20 C upon receipt A limited-use label license covers this product. By use of this product, you accept the terms and conditions outlined in the Licensing
Supplementary Information
 Supplementary Information Super-resolution imaging of fluorescently labeled, endogenous RNA Polymerase II in living cells with CRISPR/Cas9-mediated gene editing Won-Ki Cho 1, Namrata Jayanth 1, Susan Mullen
Supplementary Information Super-resolution imaging of fluorescently labeled, endogenous RNA Polymerase II in living cells with CRISPR/Cas9-mediated gene editing Won-Ki Cho 1, Namrata Jayanth 1, Susan Mullen
Cloning of pooled sgrnas into lentiviral vector Developed by JEV, LAG and MH Modified and updated, RAP & CYP
 Cloning of pooled sgrnas into lentiviral vector Developed by JEV, LAG and MH Modified and updated, RAP & CYP 1) Vector Preparation: plg1 library vector a) Digest 5 ug of vector with Thermo Scientific FastDigest
Cloning of pooled sgrnas into lentiviral vector Developed by JEV, LAG and MH Modified and updated, RAP & CYP 1) Vector Preparation: plg1 library vector a) Digest 5 ug of vector with Thermo Scientific FastDigest
Programmable Sequence-Specific Transcriptional Regulation of Mammalian Genome Using Designer TAL Effectors
 Supplementary Information Programmable Sequence-Specific Transcriptional Regulation of Mammalian Genome Using Designer TAL Effectors Feng Zhang 1,2,3,5,7 *,±, Le Cong 2,3,4 *, Simona Lodato 5,6, Sriram
Supplementary Information Programmable Sequence-Specific Transcriptional Regulation of Mammalian Genome Using Designer TAL Effectors Feng Zhang 1,2,3,5,7 *,±, Le Cong 2,3,4 *, Simona Lodato 5,6, Sriram
Functional Genomics Research Stream. Research Meeting: November 8, 2011 cdna Library Construction for RNA-Seq
 Functional Genomics Research Stream Research Meeting: November 8, 2011 cdna Library Construction for RNA-Seq Lab Issues Don t leave out boxes of water + ethidium bromide Minimize tip boxes in phenol waste
Functional Genomics Research Stream Research Meeting: November 8, 2011 cdna Library Construction for RNA-Seq Lab Issues Don t leave out boxes of water + ethidium bromide Minimize tip boxes in phenol waste
TrueORF TM cdna Clones and PrecisionShuttle TM Vector System
 TrueORF TM cdna Clones and PrecisionShuttle TM Vector System Application Guide Table of Contents Package Contents and Storage Conditions... 2 Related, Optional Reagents... 2 Related Products... 2 Available
TrueORF TM cdna Clones and PrecisionShuttle TM Vector System Application Guide Table of Contents Package Contents and Storage Conditions... 2 Related, Optional Reagents... 2 Related Products... 2 Available
(A) and (B) Experiments 2 and 3 (also see Table 1). Simultaneous small deletion (24bp) and restriction enzyme site (RE) insertion.
 Figure S1 Schematic representation of deletion/insertion experiments on gene Black lines represent genomic DNA. Red lines represent inserted sequence and blue lines are homology arms on template DNA. Scissors
Figure S1 Schematic representation of deletion/insertion experiments on gene Black lines represent genomic DNA. Red lines represent inserted sequence and blue lines are homology arms on template DNA. Scissors
CLONING INVERTED REPEATS IN HIGH THROUGHPUT
 CLONING INVERTED REPEATS IN HIGH THROUGHPUT This protocol is calculated for cloning one 96 well plate 1. design primers: as standard we include a EcoRI restriction site on the 5 primer and XbaI on the
CLONING INVERTED REPEATS IN HIGH THROUGHPUT This protocol is calculated for cloning one 96 well plate 1. design primers: as standard we include a EcoRI restriction site on the 5 primer and XbaI on the
MeDIP-seq library construction protocol v2 Costello Lab June Notes:
 MeDIP-seq library construction protocol v2 Costello Lab June 2010 Notes: A. For all Qiagen gel extraction steps (Qiaquick and MinElute), melt gel slice at 37º C instead of 50º (see Quail et al 2008 Nature
MeDIP-seq library construction protocol v2 Costello Lab June 2010 Notes: A. For all Qiagen gel extraction steps (Qiaquick and MinElute), melt gel slice at 37º C instead of 50º (see Quail et al 2008 Nature
3 Designing Primers for Site-Directed Mutagenesis
 3 Designing Primers for Site-Directed Mutagenesis 3.1 Learning Objectives During the next two labs you will learn the basics of site-directed mutagenesis: you will design primers for the mutants you designed
3 Designing Primers for Site-Directed Mutagenesis 3.1 Learning Objectives During the next two labs you will learn the basics of site-directed mutagenesis: you will design primers for the mutants you designed
BRED: Bacteriophage Recombineering with Electroporated DNA
 Phagehunting Program BRED: Bacteriophage Recombineering with Electroporated DNA Introduction We have developed a system for generating mutations in lytically replicating mycobacteriophages that we have
Phagehunting Program BRED: Bacteriophage Recombineering with Electroporated DNA Introduction We have developed a system for generating mutations in lytically replicating mycobacteriophages that we have
Chapter 6 - Molecular Genetic Techniques
 Chapter 6 - Molecular Genetic Techniques Two objects of molecular & genetic technologies For analysis For generation Molecular genetic technologies! For analysis DNA gel electrophoresis Southern blotting
Chapter 6 - Molecular Genetic Techniques Two objects of molecular & genetic technologies For analysis For generation Molecular genetic technologies! For analysis DNA gel electrophoresis Southern blotting
Phosphorothioate-based BioBrick cloning (Potsdam) Standard
 BBF RFC 91: phosphorothioate-based BioBrick cloning 1 Document Version 1.0, October 26, 2012 Phosphorothioate-based BioBrick cloning (Potsdam) Standard igem Team Potsdam 2012 1, Tobias Baumann 1, Kristian
BBF RFC 91: phosphorothioate-based BioBrick cloning 1 Document Version 1.0, October 26, 2012 Phosphorothioate-based BioBrick cloning (Potsdam) Standard igem Team Potsdam 2012 1, Tobias Baumann 1, Kristian
Lecture Four. Molecular Approaches I: Nucleic Acids
 Lecture Four. Molecular Approaches I: Nucleic Acids I. Recombinant DNA and Gene Cloning Recombinant DNA is DNA that has been created artificially. DNA from two or more sources is incorporated into a single
Lecture Four. Molecular Approaches I: Nucleic Acids I. Recombinant DNA and Gene Cloning Recombinant DNA is DNA that has been created artificially. DNA from two or more sources is incorporated into a single
Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis *
 Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis * Laboratory of Pediatric Infectious Diseases, Department of Pediatrics and Laboratory of Medical Immunology,
Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis * Laboratory of Pediatric Infectious Diseases, Department of Pediatrics and Laboratory of Medical Immunology,
Synthetic Biology for
 Synthetic Biology for Plasmids and DNA Digestion Plasmids Plasmids are small DNA molecules that are separate from chromosomal DNA They are most commonly found as double stranded, circular DNA Typical plasmids
Synthetic Biology for Plasmids and DNA Digestion Plasmids Plasmids are small DNA molecules that are separate from chromosomal DNA They are most commonly found as double stranded, circular DNA Typical plasmids
CELLTECHGEN For Research Only. Construction of all-in-one vector for Lenti-virus system (Example: Lenti-EF1 -Cas9-EGFP-U6 sgrna vector)
 Construction of all-in-one vector for Lenti-virus system (Example: Lenti-EF1 -Cas9-EGFP-U6 sgrna vector) Catalog number: CTG-CAS9-18 Introduction The vector Lenti-EF1 -Cas9-EGFP-U6 sgrna is designed for
Construction of all-in-one vector for Lenti-virus system (Example: Lenti-EF1 -Cas9-EGFP-U6 sgrna vector) Catalog number: CTG-CAS9-18 Introduction The vector Lenti-EF1 -Cas9-EGFP-U6 sgrna is designed for
SUPPLEMENTARY INFORMATION
 Gene replacements and insertions in rice by intron targeting using CRISPR Cas9 Table of Contents Supplementary Figure 1. sgrna-induced targeted mutations in the OsEPSPS gene in rice protoplasts. Supplementary
Gene replacements and insertions in rice by intron targeting using CRISPR Cas9 Table of Contents Supplementary Figure 1. sgrna-induced targeted mutations in the OsEPSPS gene in rice protoplasts. Supplementary
Preparing normalized cdna libraries for transcriptome sequencing (Illumina HiSeq)
 Preparing normalized cdna libraries for transcriptome sequencing (Illumina HiSeq) Last updated: Oct 28, 2016 Overview First-strand cdna is synthesized using oligo-dt containing primers and an RNA oligo
Preparing normalized cdna libraries for transcriptome sequencing (Illumina HiSeq) Last updated: Oct 28, 2016 Overview First-strand cdna is synthesized using oligo-dt containing primers and an RNA oligo
Gene Jockeying: Tricks of the Trade so that your Ligations work Every Time! Bevin Engelward
 Gene Jockeying: Tricks of the Trade so that your Ligations work Every Time! Bevin Engelward We will first review a simple example of a ligation. Know Your Vectors! -Find out as much as you can about your
Gene Jockeying: Tricks of the Trade so that your Ligations work Every Time! Bevin Engelward We will first review a simple example of a ligation. Know Your Vectors! -Find out as much as you can about your
Recitation CHAPTER 9 DNA Technologies
 Recitation CHAPTER 9 DNA Technologies DNA Cloning: General Scheme A cloning vector and eukaryotic chromosomes are separately cleaved with the same restriction endonuclease. (A single chromosome is shown
Recitation CHAPTER 9 DNA Technologies DNA Cloning: General Scheme A cloning vector and eukaryotic chromosomes are separately cleaved with the same restriction endonuclease. (A single chromosome is shown
rrna subtraction protocol for metatranscriptomics
 rrna subtraction protocol for metatranscriptomics Recommended materials/kits Price Vendor Stock Number MEGAscript transcription kit $249 Ambion AM1334 MEGAclear TM kit $89 Ambion AM1908 RNeasy MinElute
rrna subtraction protocol for metatranscriptomics Recommended materials/kits Price Vendor Stock Number MEGAscript transcription kit $249 Ambion AM1334 MEGAclear TM kit $89 Ambion AM1908 RNeasy MinElute
Diagnosis Sanger. Interpreting and Troubleshooting Chromatograms. Volume 1: Help! No Data! GENEWIZ Technical Support
 Diagnosis Sanger Interpreting and Troubleshooting Chromatograms GENEWIZ Technical Support DNAseq@genewiz.com Troubleshooting This troubleshooting guide is based on common issues seen from samples within
Diagnosis Sanger Interpreting and Troubleshooting Chromatograms GENEWIZ Technical Support DNAseq@genewiz.com Troubleshooting This troubleshooting guide is based on common issues seen from samples within
Chapter 20 Recombinant DNA Technology. Copyright 2009 Pearson Education, Inc.
 Chapter 20 Recombinant DNA Technology Copyright 2009 Pearson Education, Inc. 20.1 Recombinant DNA Technology Began with Two Key Tools: Restriction Enzymes and DNA Cloning Vectors Recombinant DNA refers
Chapter 20 Recombinant DNA Technology Copyright 2009 Pearson Education, Inc. 20.1 Recombinant DNA Technology Began with Two Key Tools: Restriction Enzymes and DNA Cloning Vectors Recombinant DNA refers
Cellulose Cross-Linker
 Cellulose Cross-Linker Project Journal 5/28/14 Found BioBrick of CBM: http://parts.igem.org/part:bba_k863111 Found BioBrick of Strep: http://parts.igem.org/part:bba_k283010 5/29/14 Researched a possible
Cellulose Cross-Linker Project Journal 5/28/14 Found BioBrick of CBM: http://parts.igem.org/part:bba_k863111 Found BioBrick of Strep: http://parts.igem.org/part:bba_k283010 5/29/14 Researched a possible
Texas A&M University-Corpus Christi CHEM4402 Biochemistry II Laboratory Laboratory 8: DNA Restriction Digest (II) and DNA Sequencing (I)
 Texas A&M University-Corpus Christi CHEM4402 Biochemistry II Laboratory Laboratory 8: DNA Restriction Digest (II) and DNA Sequencing (I) We have made considerable progress in our analysis of the gene for
Texas A&M University-Corpus Christi CHEM4402 Biochemistry II Laboratory Laboratory 8: DNA Restriction Digest (II) and DNA Sequencing (I) We have made considerable progress in our analysis of the gene for
