DENSITY OF MATTER. To experimentally determine the densities of given solids and liquids.
|
|
|
- Russell Watkins
- 5 years ago
- Views:
Transcription
1 DENSITY OF MATTER PURPOSE: To experimentally determine the densities of iven solids and liquids. EQUIPMENT: Triple-beam balance, vernier caliper, raduated, lass block, metal, metal sphere, wooden sphere, rock sample, ethyl alcohol. INTRODUCTION: You may have heard it said that iron is "heavier" than aluminum and that both are heavier than water. However, a piece of aluminum the size of a battleship obviously weihs more than an iron cannonball, and the water in the Atlantic Ocean outweihs them both. What we really mean is that iron has a reater density than aluminum, and both are denser than water. is an important property of matter because it is a measure of how compact an object is, i.e. how much mass is contained within the space occupied by the object. is defined to be the ratio of the mass of an object to the volume of the object: mass() density ( ) = cm volume( cm ) In the metric system, the units of density are rams per cubic centimeter (/cm ) or kilorams per cubic meter (k/m ). Table E ives the accepted values of densities of some common substances. PROCEDURE: Part A. Densities of Reular Shaped Solids 1. Usin the balance, measure the mass (to the nearest 0.1 ram) of the lass block, metal, metal sphere, and wooden sphere. Record in the data table. 2. Usin the vernier caliper, measure the dimensions (to the nearest 0.01 cm) of the lass block, metal, metal sphere, and wooden sphere. Record in the data table.. Compute and record the volume of the objects from the formulas iven on the data sheet. 4. Calculate the density of each object by takin the ratio of the mass to the volume.
2 Part B. Densities of Irreular Shaped Solids 1. Usin the balance, measure the mass of the rock sample. Record in the data table alon with the mass of the metal from Part A. 2. Fill a raduated about half full of water. Record the initial water level at the bottom of the meniscus. It is important to estimate the level to the nearest 0.1 ml. Usin a wire or a strin carefully lower the metal into the raduated. Record the final water level to the nearest 0.1 ml. Since 1 ml is practically the same as 1 cm, the difference between the levels is the volume of the object placed in the water.. Repeat step 2 usin the rock sample. Because some water may have been removed, the initial level should be re-measured. 4. Calculate the volume of each object from the chane in water level. 5. Calculate the density of each object. Part C. Densities of Liquids 1. Measure and record the mass of a dry raduated. 2. Fill the about /4 full of water. Record the volume of the water to the nearest 0.1 ml = 0.1 cm. Aain read the level at the bottom of the meniscus.. Measure and record the mass of the plus water. Determine the mass of the water by subtraction. 4. Repeat steps 2 and usin alcohol instead of water. 5. Calculate the density of each liquid. Part D. Comparison of Densities 1. List each experimental density in the data table in increasin order (smallest density first). 2. Record the accepted value in the data table and compute the per cent error. % Error Accepted Measured Accepted x 100 Note: The actual densities of some substances (wood, etc.) have a wide rane due to different compositions. Do not compute errors for these substances.
3 QUESTIONS: 1. The Earth can be considered a sphere of diameter 1.28 x 10 9 cm and mass 5.98 x rams. Calculate the averae density of Earth matter. =. The density of the rock sample used in this experiment is than the averae density of earth material. If the density of the rock sample is typical of rock material close to the earth surface, then interior rock must have density than surface rock. This is due to reater in the Earth's interior.
4 DATA SHEET: Table A. Densities of Reular Shaped Solids Object Mass () Dimensions (cm) Volume (cm ) (/cm ) L= lass block W= H= metal L= D= metal sphere D= wooden sphere D= Volume of block ( lenth)( width)( heiht) LDH Volume of.14( diameter) 4 2 ( lenth) 2 D L 4 Volume of sphere.14 ( diameter) 6 D 6 mass volume Table B. Densities of Irreular Shaped Solids Object Mass () Water level (ml) Volume (cm ) cm metal final = initial = difference = rock sample final = initial = difference =
5 Table C. Densities of Liquids Substance Mass () Volume (cm ) cm plus liquid = water = liquid = plus liquid = alcohol = liquid = Table D. Comparison of Densities Substance Experimental (/cm ) Accepted (/cm ) % Error Table E. Densities of Substances (accepted value) Substance (/cm ) Substance (/cm ) air zinc 7.1 wood (pine) tin 7. alcohol (ethyl) 0.79 steel 7.8 water 1.0 brass 8.4 lass copper 8.9 aluminum 2.7 lead 11.
Refer to the Appendices at the back of your lab manual to review laboratory techniques, significant figures and statistical treatment of data.
 LABORATORY EXPERIMENT A: Volume of a Solid Cylinder PRELABORATORY ASSIGNMENT: Refer to the Appendices at the back of your lab manual to review laboratory techniques, sinificant fiures and statistical treatment
LABORATORY EXPERIMENT A: Volume of a Solid Cylinder PRELABORATORY ASSIGNMENT: Refer to the Appendices at the back of your lab manual to review laboratory techniques, sinificant fiures and statistical treatment
DENSITY of Solids and Liquids
 Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
Answer the pre-lab questions that appear at the end of this lab exercise.
 Experiment 3 Enery and Heat Capacity Pre-lab Assinment Before comin to lab: Read the lab thorouhly. Answer the pre-lab questions that appear at the end of this lab exercise. Backround The oal of this lab
Experiment 3 Enery and Heat Capacity Pre-lab Assinment Before comin to lab: Read the lab thorouhly. Answer the pre-lab questions that appear at the end of this lab exercise. Backround The oal of this lab
SOLIDS: Mass, Volume and Density Measurements
 CHEM 1411 Lab Solids: Mass, Volume, and Density 23 SOLIDS: Mass, Volume and Density Measurements A. Overview Review Sections 1.4-1.6 in your textbook (Chemistry: The Central Science, 9th Ed., Brown, LeMay,
CHEM 1411 Lab Solids: Mass, Volume, and Density 23 SOLIDS: Mass, Volume and Density Measurements A. Overview Review Sections 1.4-1.6 in your textbook (Chemistry: The Central Science, 9th Ed., Brown, LeMay,
Density. The purpose of this experiment is to investigate the topic of density by determining the densities of some materials.
 Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
T. Trimpe Lesson 1: Length
 T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
D =? D = M = 10 g = 5 g/cm 3 M = 10 g V 2 cm 3 V = 2 cm 3 5 g/cm 3
 Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Copyright 2014 Science Doodles
 Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
CHAPTER 2 HW PRACTICE SOLUTIONS
 CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
T. Trimpe Lesson 1: Length
 T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
Lab 0.4: Density and Thickness of Aluminum Foil
 Name Block Lab 0.4: Density and Thickness of Aluminum Foil Student will be able to: Correctly use measuring instruments with accuracy Correctly apply the principles of significant figures in measurements
Name Block Lab 0.4: Density and Thickness of Aluminum Foil Student will be able to: Correctly use measuring instruments with accuracy Correctly apply the principles of significant figures in measurements
Measurement and Density - Experiment 1
 Measurement and Density - Experiment 1 Purpose: The purpose of this experiment is to acquaint you with some common metric units of length, volume, and mass. You will also measure the density of an unknown
Measurement and Density - Experiment 1 Purpose: The purpose of this experiment is to acquaint you with some common metric units of length, volume, and mass. You will also measure the density of an unknown
Density What is density? How do you measure density?
 Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
SIGNIFICANT FIGURES WORKSHEET
 SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
Experiment #3. Density
 Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
Introduction to Density
 Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Metals. General information
 Metals. General information Read and copy the following text: Metals are divided into two groups: Metals that contain iron are called Ferrous Metals (Alloys). Metals that do not contain iron are called
Metals. General information Read and copy the following text: Metals are divided into two groups: Metals that contain iron are called Ferrous Metals (Alloys). Metals that do not contain iron are called
Experiment #3. Density and Specific Gravity.
 Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
Cu-AgNO 3 Lab Date: Hour: Question: What is the mole ratio of copper consumed to silver produced in the reaction?
 Chemistry Cu-ANO 3 Lab Name: Date: Hour: Partner: Question: What is the mole ratio of copper consumed to silver produced in the reaction? Hypothesis: (answer usin complete sentences) Research (answer questions
Chemistry Cu-ANO 3 Lab Name: Date: Hour: Partner: Question: What is the mole ratio of copper consumed to silver produced in the reaction? Hypothesis: (answer usin complete sentences) Research (answer questions
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use.
 This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
2.3 Density and Density Calculations
 2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
Determining the Density of Unknown Substances
 Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
There are 2 different types of numbers Exact -Measured
 Significant Figures When measuring or using our calculators we must determine the correct answer; our calculators are mindless drones and don t know the correct answer. There are 2 different types of numbers
Significant Figures When measuring or using our calculators we must determine the correct answer; our calculators are mindless drones and don t know the correct answer. There are 2 different types of numbers
v = 5.00cm x 3.00cm x 2.00cm 2. What is the volume of a marble that has a mass of 3.0 g and a density of 2.7 g/ml? Formula (density) Substitution
 Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Science and Health CLASSIFYING MATERIALS INTO CONDUCTORS AND INSULATORS
 5 Module 32 Science and Health CLASSIFYING MATERIALS INTO CONDUCTORS AND INSULATORS A DepEd-BEAM Distance Learning Program supported by the Australian Agency for International Development To the Learner
5 Module 32 Science and Health CLASSIFYING MATERIALS INTO CONDUCTORS AND INSULATORS A DepEd-BEAM Distance Learning Program supported by the Australian Agency for International Development To the Learner
Directions: Complete the following prior to class. Be prepared to show your work on the board.
 Worksheet L4X Student s Name Directions: Complete the following prior to class. Be prepared to show your work on the board. 1) Convert 30 ml to oz, qt, L, gal. 2) Convert 16 oz to kg, lb, tons, g. Page
Worksheet L4X Student s Name Directions: Complete the following prior to class. Be prepared to show your work on the board. 1) Convert 30 ml to oz, qt, L, gal. 2) Convert 16 oz to kg, lb, tons, g. Page
Biomass Calorimeter Energy from Biomass
 Biomass Calorimeter Enery from Biomass Introduction Have you ever noticed the nutrition label located on the packain of the food you buy? One of the first thins listed on the label are the calories per
Biomass Calorimeter Enery from Biomass Introduction Have you ever noticed the nutrition label located on the packain of the food you buy? One of the first thins listed on the label are the calories per
Experiment 10 Uniform Circular Motion
 Experiment 0 Whenever a body moves in a circular path, a force directed toward the center of the circle must act on the body to keep it moving in this path. That force is called centripetal force. The
Experiment 0 Whenever a body moves in a circular path, a force directed toward the center of the circle must act on the body to keep it moving in this path. That force is called centripetal force. The
Topic 1 Review Questions Set 1
 Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
Metal Stock Identification
 Metal Work Description Metal is produced in many different types, shapes, and forms. This activity plan provides examples of stock forms that might be found in a metal shop. Many of these shapes are common
Metal Work Description Metal is produced in many different types, shapes, and forms. This activity plan provides examples of stock forms that might be found in a metal shop. Many of these shapes are common
LESSON hat is density? 24
 LESSON hat is density? 24 Rocks are heavy. A small box of rocks has a large. Yet a box of the same size containing corks has a small. How can objects that are the same size have different es? Scientists
LESSON hat is density? 24 Rocks are heavy. A small box of rocks has a large. Yet a box of the same size containing corks has a small. How can objects that are the same size have different es? Scientists
The table below shows some of the properties of the elements cobalt and nickel.
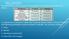 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Experiment: Measurements
 Experiment: Measurements I. INTRODUCTION Measurements are essential to experimental sciences such as chemistry, physics, biology, and geology. The measurements are usually made using the metric system
Experiment: Measurements I. INTRODUCTION Measurements are essential to experimental sciences such as chemistry, physics, biology, and geology. The measurements are usually made using the metric system
Practice with Properties of Matter. Practice with Physical and Chemical Changes. Practice with Mixtures and Pure Substances
 Chapter 1 Practice Problems Practice with Properties of Matter Indicate whether each of the following statements describes a physical or a chemical property. a) the normal color of elemental bromine is
Chapter 1 Practice Problems Practice with Properties of Matter Indicate whether each of the following statements describes a physical or a chemical property. a) the normal color of elemental bromine is
2. Use the Styrofoam cutter to cut the Styrofoam. Take your time so that your beam has the appropriate dimensions over the entire beam.
 Structures Activity 8 Beams and Cantilevers In the fist part of this activity you will determine the relationship between the dimensions of a beam and the deflection of the beam under a load. In the second
Structures Activity 8 Beams and Cantilevers In the fist part of this activity you will determine the relationship between the dimensions of a beam and the deflection of the beam under a load. In the second
ES 106 Laboratory # 1 PROPERTIES OF WATER
 ES 106 Laboratory # 1 PROPERTIES OF WATER 1-1 Introduction What are the physical and chemical properties of water that make it so unique and necessary for living things? When you look at water, taste and
ES 106 Laboratory # 1 PROPERTIES OF WATER 1-1 Introduction What are the physical and chemical properties of water that make it so unique and necessary for living things? When you look at water, taste and
Density. How tightly the atoms are packed together in an object
 Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
Creation Settings. can be separated into two or more pure substances by distillation. can be separated into two or more substances by chromatography.
 Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Name # Date Period! FINDING&VOLUME&AND&DENSITY& !!! !!!!!!!
 Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Density Answers to the End of module questions
 IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
Engineering Materials
 Engineering Materials PREPARED BY IAT Curriculum Unit August 2010 Institute of Applied Technology, 2010 Module Objectives After the completion of this module, the student will be able to: Explain the difference
Engineering Materials PREPARED BY IAT Curriculum Unit August 2010 Institute of Applied Technology, 2010 Module Objectives After the completion of this module, the student will be able to: Explain the difference
Unit C: Forest Management. Lesson 3: Measuring Timber Stands
 Unit C: Forest Management Lesson 3: Measuring Timber Stands 1 Terms Basal area Board foot dbh Diameter at breast height Log rules Lumber overrun Taper Wedge prism 2 How do I calculate board feet? A board
Unit C: Forest Management Lesson 3: Measuring Timber Stands 1 Terms Basal area Board foot dbh Diameter at breast height Log rules Lumber overrun Taper Wedge prism 2 How do I calculate board feet? A board
Dimensions Mechanical and Performance Requirements. Round Spacers Hex Spacers Round Threaded Female Standoffs...
 Spacers & Standoffs imensions Mechanical and Performance Requirements Round Spacers... 295 Hex Spacers... 296 Round hreaded emale Standoffs... 297 Hex hreaded emale Standoffs... 298 Hex & Round hreaded
Spacers & Standoffs imensions Mechanical and Performance Requirements Round Spacers... 295 Hex Spacers... 296 Round hreaded emale Standoffs... 297 Hex hreaded emale Standoffs... 298 Hex & Round hreaded
Lesson 26: Volume and Surface Area
 NYS COMMON CORE MATHEMATICS CURRICULUM Lesson 6 7 3 Student Outcomes Students solve real-world and mathematical problems involving volume and surface areas of threedimensional objects composed of cubes
NYS COMMON CORE MATHEMATICS CURRICULUM Lesson 6 7 3 Student Outcomes Students solve real-world and mathematical problems involving volume and surface areas of threedimensional objects composed of cubes
Density (d) is a property of a substance equal to the ratio of its mass (m) to its volume (V): d = m V
 Experiment 2 Name: DEN 14 Si TY In this experiment, you will learn about density and practice the proper way to report measurements and calculated quantities. Properties Properties of a substance can be
Experiment 2 Name: DEN 14 Si TY In this experiment, you will learn about density and practice the proper way to report measurements and calculated quantities. Properties Properties of a substance can be
Downloaded from
 Fun with Magnets 1 1.What happens when the north pole of a magnet is placed near the north pole of another magnet? 2.Which of the following cannot be attracted by a magnet? (A) Wood. (B) Nickel. (C)
Fun with Magnets 1 1.What happens when the north pole of a magnet is placed near the north pole of another magnet? 2.Which of the following cannot be attracted by a magnet? (A) Wood. (B) Nickel. (C)
Analysis of Alum, AlK(SO 4 ) 2 12H 2 O AP Chemistry Laboratory #2
 Analysis of Alum, AlK(SO 4 ) 2 12H 2 O AP Chemistry Laboratory #2 Catalo No. AP6354 Publication No. 6354A Introduction When a compound is synthesized, tests are carried out to confirm whether the compound
Analysis of Alum, AlK(SO 4 ) 2 12H 2 O AP Chemistry Laboratory #2 Catalo No. AP6354 Publication No. 6354A Introduction When a compound is synthesized, tests are carried out to confirm whether the compound
Geometric formulas for volume include: Where r is the radius of the cylinder and h is the height. Volume of a rectangular prism = LxWxH
 Supplement Density The density of an object is defined as its mass per unit of volume. The formula below may be used to calculate the density of an object: Density = mass/volume Volume may be measured
Supplement Density The density of an object is defined as its mass per unit of volume. The formula below may be used to calculate the density of an object: Density = mass/volume Volume may be measured
Engineering Materials
 Engineering Materials PREPARED BY Academic Services August 2011 Applied Technology High Schools, 2011 Module Objectives After the completion of this module, the student will be able to: Explain the difference
Engineering Materials PREPARED BY Academic Services August 2011 Applied Technology High Schools, 2011 Module Objectives After the completion of this module, the student will be able to: Explain the difference
Choosing building materials
 Choosing building materials clay (click for more) straw bale copper stone Plastic (click for more) metals (click for more) bamboo wood (click for more) sand Can I reduce the environmental impact of my
Choosing building materials clay (click for more) straw bale copper stone Plastic (click for more) metals (click for more) bamboo wood (click for more) sand Can I reduce the environmental impact of my
Lab Section. Experiment 2 Density. Introduction
 Experiment 2 Density Introduction Density (given the symbol d or ρ depending upon which book you read) is an intrinsic property of materials. The term intrinsic means that it is independent upon the amount
Experiment 2 Density Introduction Density (given the symbol d or ρ depending upon which book you read) is an intrinsic property of materials. The term intrinsic means that it is independent upon the amount
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
INDEX METALS FERRIC METALS NON-FERRIC METALS WORKING WITH METALS METAL FORMING TECHNIQUES ENVIRONMENTAL IMPACT OF METAL EXTRACTION
 METALS INDEX METALS FERRIC METALS NON-FERRIC METALS WORKING WITH METALS METAL FORMING TECHNIQUES ENVIRONMENTAL IMPACT OF METAL EXTRACTION METALS CHEMICAL ELEMENTS FOUND IN NATURE IN SOLID STATE AT ROOM
METALS INDEX METALS FERRIC METALS NON-FERRIC METALS WORKING WITH METALS METAL FORMING TECHNIQUES ENVIRONMENTAL IMPACT OF METAL EXTRACTION METALS CHEMICAL ELEMENTS FOUND IN NATURE IN SOLID STATE AT ROOM
Experiment 5 Uniform Circular Motion
 Experiment 5 Uniform Circular Motion Whenever a body moves in a circular path, a force directed toward the center of the circle must act on the body to keep it moving in this path. That force is called
Experiment 5 Uniform Circular Motion Whenever a body moves in a circular path, a force directed toward the center of the circle must act on the body to keep it moving in this path. That force is called
PHARMACOPOEIAL DISCUSSION GROUP BULK AND TAPPED DENSITY OF SOLIDS
 PHARMACOPOEIAL DISCUSSION GROUP BULK AND TAPPED DENSITY OF SOLIDS It is understood that sign-off covers the technical content of the draft and each party will adapt it as necessary to conform to the usual
PHARMACOPOEIAL DISCUSSION GROUP BULK AND TAPPED DENSITY OF SOLIDS It is understood that sign-off covers the technical content of the draft and each party will adapt it as necessary to conform to the usual
DCHA DOWNHOLE COUPON HOLDER ASSEMBLY
 DCHA DOWNHOLE COUPON HOLDER ASSEMBLY User Manual Rohrback Cosasco Systems, Inc. 11841 E. Smith Avenue Santa Fe Springs, CA 90670 Tel: (562) 949-0123 (800) 635-6898 Fax: (562) 949-3065 E-mail: sales@cosasco.com
DCHA DOWNHOLE COUPON HOLDER ASSEMBLY User Manual Rohrback Cosasco Systems, Inc. 11841 E. Smith Avenue Santa Fe Springs, CA 90670 Tel: (562) 949-0123 (800) 635-6898 Fax: (562) 949-3065 E-mail: sales@cosasco.com
3.3 Minerals. Describe the characteristics that define minerals.
 3.3 Minerals Describe the characteristics that define minerals. Are you a mineral? There used to be a TV commercial that said "you are what you eat." If that s true - and to some extent it is - then you
3.3 Minerals Describe the characteristics that define minerals. Are you a mineral? There used to be a TV commercial that said "you are what you eat." If that s true - and to some extent it is - then you
Density of Matter. Fast Track GRASP Math Packet. Photo by Ben Stephenson (Wikimedia Commons) Version 1.0 Released 12/4/2018
 Density of Matter Fast Track GRASP Math Packet Photo by Ben Stephenson (Wikimedia Commons) Version 1.0 Released 12/4/2018 This Fast Track GRASP Math Packet was made possible through support from the New
Density of Matter Fast Track GRASP Math Packet Photo by Ben Stephenson (Wikimedia Commons) Version 1.0 Released 12/4/2018 This Fast Track GRASP Math Packet was made possible through support from the New
Density of Matter. Fast Track GRASP Math Packet. Photo by Ben Stephenson (Wikimedia Commons) Version 1.2 Released 1/16/2019
 Density of Matter Fast Track GRASP Math Packet Photo by Ben Stephenson (Wikimedia Commons) Version 1.2 Released 1/16/2019 This Fast Track GRASP Math Packet was made possible through support from the New
Density of Matter Fast Track GRASP Math Packet Photo by Ben Stephenson (Wikimedia Commons) Version 1.2 Released 1/16/2019 This Fast Track GRASP Math Packet was made possible through support from the New
PERFORMANCE TEST REPORT. Rendered to: HILTI CORPORATION. PRODUCT: Extreme Weather Window & Door Insulating Foam Sealant
 PERFORMANCE TEST REPORT Rendered to: HILTI CORPORATION PRODUCT: Extreme Weather Window & Door Insulating Foam Sealant Report No.: G1169.02-106-31 Report Date: 11/16/16 Test Record Retention Date: 10/25/20
PERFORMANCE TEST REPORT Rendered to: HILTI CORPORATION PRODUCT: Extreme Weather Window & Door Insulating Foam Sealant Report No.: G1169.02-106-31 Report Date: 11/16/16 Test Record Retention Date: 10/25/20
CRYSTAL STRUCTURE TERMS
 CRYSTAL STRUCTURE TERMS crystalline material - a material in which atoms, ions, or molecules are situated in a periodic 3-dimensional array over large atomic distances (all metals, many ceramic materials,
CRYSTAL STRUCTURE TERMS crystalline material - a material in which atoms, ions, or molecules are situated in a periodic 3-dimensional array over large atomic distances (all metals, many ceramic materials,
CITY OF BRISTOL DECK CONSTRUCTION GUIDE ACCORDING TO THE 2012 IRC WITH THE 2016 CT AMMENDMENTS
 CITY OF BRISTOL DECK CONSTRUCTION GUIDE ACCORDING TO THE 2012 IRC WITH THE 2016 CT AMMENDMENTS To construct an attached deck in to a residential home in the City of Bristol, you must provide the following:
CITY OF BRISTOL DECK CONSTRUCTION GUIDE ACCORDING TO THE 2012 IRC WITH THE 2016 CT AMMENDMENTS To construct an attached deck in to a residential home in the City of Bristol, you must provide the following:
Class XII Chapter 1 The Solid State Chemistry
 Class XII Chapter 1 The Solid State Chemistry Question 1.1: Define the term 'amorphous'. Give a few examples of amorphous solids. Amorphous solids are the solids whose constituent particles are of irregular
Class XII Chapter 1 The Solid State Chemistry Question 1.1: Define the term 'amorphous'. Give a few examples of amorphous solids. Amorphous solids are the solids whose constituent particles are of irregular
M. It is expressed in units such a g/cc,
 Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
European Pharmacopoeia
 i PHARMACOPOEIAL DISCUSSION GROUP REV 1 - CORR 1 G02 Bulk density and tapped density of powders It is understood that sign-off covers the technical content of the draft and each party will adapt it as
i PHARMACOPOEIAL DISCUSSION GROUP REV 1 - CORR 1 G02 Bulk density and tapped density of powders It is understood that sign-off covers the technical content of the draft and each party will adapt it as
CHAPTER 1. Conversions. General Plan for Converting Measurements. State the relationship between the unit given and the unit sought as an equality.
 CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
The weight of the bell in kilograms is ~2,174. The radius is about 7mm or 0.007m.
 Problem Set 3 Due: Tuesday, February 7, 11:00 AM 1. Yield/Shear on Storke Tower Suppose the larest bell in Storke Tower, which weihs 4,793 pounds, needs to be replaced. A crane is brouht in to remove the
Problem Set 3 Due: Tuesday, February 7, 11:00 AM 1. Yield/Shear on Storke Tower Suppose the larest bell in Storke Tower, which weihs 4,793 pounds, needs to be replaced. A crane is brouht in to remove the
Chapter 2 Timber Cruising
 Chapter 2 Timber Cruising Timber cruising is a procedure for determining the gross and net volume and value (timber quality) for a tract of timber. Cruising involves measuring tree diameter and height
Chapter 2 Timber Cruising Timber cruising is a procedure for determining the gross and net volume and value (timber quality) for a tract of timber. Cruising involves measuring tree diameter and height
To become bigger or make bigger the amount or size of something.
 Increase Decrease (Reduce) State of Matter Property Density Energy Temperature To become bigger or make bigger the amount or size of something. To become smaller or make smaller the amount or size of something.
Increase Decrease (Reduce) State of Matter Property Density Energy Temperature To become bigger or make bigger the amount or size of something. To become smaller or make smaller the amount or size of something.
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley)
 Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Unit I PART I. MATH TOOLS FOR CHEMISTRY I. The Metric System The metric system is the scientific system of units of measurement
 CHEMISTRY 100 LECTURE Unit I PART I. MATH TOOLS FOR CHEMISTRY I. The Metric System The metric system is the scientific system of units of measurement Length Volume Mass METRIC BASIC UNITS LENGTH MASS VOLUME
CHEMISTRY 100 LECTURE Unit I PART I. MATH TOOLS FOR CHEMISTRY I. The Metric System The metric system is the scientific system of units of measurement Length Volume Mass METRIC BASIC UNITS LENGTH MASS VOLUME
COURSE: METAL CASTING. Module No. 7: TESTING OF SAND
 COURSE: METAL CASTING Module No. 7: TESTING OF SAND Lecture No-3 Compactibility, Shatter index, Compression strength and Permeability in Green sands Determination of Compactability of Green Sand The compactability
COURSE: METAL CASTING Module No. 7: TESTING OF SAND Lecture No-3 Compactibility, Shatter index, Compression strength and Permeability in Green sands Determination of Compactability of Green Sand The compactability
1. Hardness 2. Streak 3. Density 4. Crystal structure 5. A mineral must be a naturally occurring, inorganic. 7. c 8. f 9. a 10. d 11. g 12. e 13.
 Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Specific Heat Activity
 Title of Lesson Subject Area Age or Grade Level Specific Heat Activity Understanding energy transfers within the lense of specific heat 7 th grade This lab is designed to be an easy way to teach students
Title of Lesson Subject Area Age or Grade Level Specific Heat Activity Understanding energy transfers within the lense of specific heat 7 th grade This lab is designed to be an easy way to teach students
Introduction to Thermal Energy
 Introduction to Thermal Energy Mar 10 7:33 AM 1 Thermal Energy vs. Temperature vs. Heat Mar 10 7:33 AM 2 Mar 10 12:53 PM 3 2 nd Law of Thermodynamics Thermal Equilibrium thermal energy is always transferred
Introduction to Thermal Energy Mar 10 7:33 AM 1 Thermal Energy vs. Temperature vs. Heat Mar 10 7:33 AM 2 Mar 10 12:53 PM 3 2 nd Law of Thermodynamics Thermal Equilibrium thermal energy is always transferred
STAAR Vocabulary noticing something about the world around you. using clues to find the answer. everything everywhere. to sort into groups
 Observation noticing something about the world around you Inference using clues to find the answer Matter everything everywhere Classify to sort into groups Physical Property something you observe with
Observation noticing something about the world around you Inference using clues to find the answer Matter everything everywhere Classify to sort into groups Physical Property something you observe with
CHEMISTRY - CLUTCH CH.1 - INTRO TO GENERAL CHEMISTRY.
 !! www.clutchprep.com CONCEPT: MATTER Chemistry is the study of matter and the changes it undergoes, with the being its basic functional unit. When two or more of these elements chemically bond together
!! www.clutchprep.com CONCEPT: MATTER Chemistry is the study of matter and the changes it undergoes, with the being its basic functional unit. When two or more of these elements chemically bond together
CHEMISTRY - MCQUARRIE 4E CH.1 - CHEMISTRY & THE SCIENTIFIC METHOD.
 !! www.clutchprep.com CONCEPT: MATTER Chemistry is the study of matter and the changes it undergoes, with the being its basic functional unit. When two or more of these elements chemically bond together
!! www.clutchprep.com CONCEPT: MATTER Chemistry is the study of matter and the changes it undergoes, with the being its basic functional unit. When two or more of these elements chemically bond together
Construction, alteration, removal, or repair of any plumbing system.
 PERMITS AND INSPECTION PERMITS (IRC R0 IPC 06) REQUIRED (IRC R 0. IPC 06.) Construction, alteration, removal, or repair of any plumbing system. APPLICATION (IRC R0. IPC 06.) Submit application to local
PERMITS AND INSPECTION PERMITS (IRC R0 IPC 06) REQUIRED (IRC R 0. IPC 06.) Construction, alteration, removal, or repair of any plumbing system. APPLICATION (IRC R0. IPC 06.) Submit application to local
EXPERIMENT 1: Thermochemistry
 EXPERIMENT 1: Thermochemistry Heat Flows Associated with Physical Processes Laboratory Techniques You will be expected to measure temperatures precisely and quickly in addition to becomin familiar with
EXPERIMENT 1: Thermochemistry Heat Flows Associated with Physical Processes Laboratory Techniques You will be expected to measure temperatures precisely and quickly in addition to becomin familiar with
Chemistry: The Central Science, 12e (Brown et al.) Chapter 1 Introduction: Matter and Measurement. 1.1 Multiple-Choice Questions
 Chemistry: The Central Science, 12e (Brown et al.) Chapter 1 Introduction: Matter and Measurement 1.1 Multiple-Choice Questions 1) In the following list, only is not an example of matter. A) planets B)
Chemistry: The Central Science, 12e (Brown et al.) Chapter 1 Introduction: Matter and Measurement 1.1 Multiple-Choice Questions 1) In the following list, only is not an example of matter. A) planets B)
Recommended Resources: The following resources may be useful in teaching this
 Unit C: Forest Management Lesson 3: Measuring Timber Stands Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives: 1. Explain how to calculate
Unit C: Forest Management Lesson 3: Measuring Timber Stands Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives: 1. Explain how to calculate
MATERIAL SPECIFICATION FOR STANDARD HIGHWAY FENCE COMPONENTS
 ONTARIO PROVINCIAL STANDARD SPECIFICATION METRIC OPSS.MUNI 1540 NOVEMBER 2016 MATERIAL SPECIFICATION FOR STANDARD HIGHWAY FENCE COMPONENTS TABLE OF CONTENTS 1540.01 SCOPE 1540.02 REFERENCES 1540.03 DEFINITIONS
ONTARIO PROVINCIAL STANDARD SPECIFICATION METRIC OPSS.MUNI 1540 NOVEMBER 2016 MATERIAL SPECIFICATION FOR STANDARD HIGHWAY FENCE COMPONENTS TABLE OF CONTENTS 1540.01 SCOPE 1540.02 REFERENCES 1540.03 DEFINITIONS
Making the Right Choice Metal Detection, X-Ray or Both. Robert Rogers Senior Advisor for Food Safety & Regulation Mettler Toledo
 1 Making the Right Choice Metal Detection, X-Ray or Both Robert Rogers Senior Advisor for Food Safety & Regulation Mettler Toledo IFSQN 5/20/16 Robert Rogers Making the Right Choice Metal Detection, X-Ray
1 Making the Right Choice Metal Detection, X-Ray or Both Robert Rogers Senior Advisor for Food Safety & Regulation Mettler Toledo IFSQN 5/20/16 Robert Rogers Making the Right Choice Metal Detection, X-Ray
CONSTRUCTION SPECIFICATION FOR STANDARD HIGHWAY FENCE
 ONTARIO PROVINCIAL STANDARD SPECIFICATION METRIC OPSS 540 APRIL 2007 CONSTRUCTION SPECIFICATION FOR STANDARD HIGHWAY FENCE TABLE OF CONTENTS 540.01 SCOPE 540.02 REFERENCES 540.03 DEFINITIONS 540.04 SUBMISSION
ONTARIO PROVINCIAL STANDARD SPECIFICATION METRIC OPSS 540 APRIL 2007 CONSTRUCTION SPECIFICATION FOR STANDARD HIGHWAY FENCE TABLE OF CONTENTS 540.01 SCOPE 540.02 REFERENCES 540.03 DEFINITIONS 540.04 SUBMISSION
Metals and Semi-Conductors
 Metals and Semi-Conductors Unit Cells for Cubic Close- Packed Spheres Hexagonal Close- Packed Spheres Inorganic Chemistry Chapter 1: Table 3.2 What are the characteristics of the metals that might be correlated
Metals and Semi-Conductors Unit Cells for Cubic Close- Packed Spheres Hexagonal Close- Packed Spheres Inorganic Chemistry Chapter 1: Table 3.2 What are the characteristics of the metals that might be correlated
Art caption: Natural resources such as rocks are mined in rock quarries (KWOR-eez) like this one. DRAFT
 1 OBSERVING EARTH S RESOURCES INVESTIGATION Humans use a lot of materials found naturally on Earth. There are many materials, including metals such as copper and woods such as pine. Materials that are
1 OBSERVING EARTH S RESOURCES INVESTIGATION Humans use a lot of materials found naturally on Earth. There are many materials, including metals such as copper and woods such as pine. Materials that are
History of the Periodic Table
 Periodic Trends History of the Periodic Table Many scientists suspected a pattern in the order of the elements, but were not able to exactly figure it out. Dmitri Mendeleev was the first to create an accurate
Periodic Trends History of the Periodic Table Many scientists suspected a pattern in the order of the elements, but were not able to exactly figure it out. Dmitri Mendeleev was the first to create an accurate
TEST I REVIEW. 1. All of the following are properties of antimony. Which one is not a
 TEST I REVIEW I Identify the letter of the choice that best completes the statement or answers the question. 1. All of the following are properties of antimony. Which one is not a physical property? a.
TEST I REVIEW I Identify the letter of the choice that best completes the statement or answers the question. 1. All of the following are properties of antimony. Which one is not a physical property? a.
GROUNDING SPECIFICATIONS FOR thermoweld EXOTHERMIC WELDING SYSTEM
 GROUNDING SPECIFICATIONS FOR thermoweld EXOTHERMIC WELDING SYSTEM 1. Purpose This specification recommends criteria for use of thermoweld products to provide reliable permanent connections for utility,
GROUNDING SPECIFICATIONS FOR thermoweld EXOTHERMIC WELDING SYSTEM 1. Purpose This specification recommends criteria for use of thermoweld products to provide reliable permanent connections for utility,
1. Plastic products that are formed into a permanent shape by heat and pressure and may not be altered after curing are produced by which process?
 Student Name: Teacher: Date: District: Rowan Assessment: 9_12 T and I IV22 - Drafting - Engnrng II Test 1 Description: Unit C - Manufacturing 1 Form: 501 1. Plastic products that are formed into a permanent
Student Name: Teacher: Date: District: Rowan Assessment: 9_12 T and I IV22 - Drafting - Engnrng II Test 1 Description: Unit C - Manufacturing 1 Form: 501 1. Plastic products that are formed into a permanent
Standard Test Procedures Manual
 STP 205-5 Standard Test Procedures Manual 1. SCOPE 1.1. Description of Test 1.2. Application of Test This method describes the procedure for determining the relationship between the moisture content and
STP 205-5 Standard Test Procedures Manual 1. SCOPE 1.1. Description of Test 1.2. Application of Test This method describes the procedure for determining the relationship between the moisture content and
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter.
 1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
EXPERIMENT 3 THE SYNTHESIS OF COPPER SULFIDE
 EXPERIMENT 3 THE SYNTHESIS OF COPPER SULFIDE When heated together, copper and sulfur combine to form a sulfide of copper. In this assignment, you will heat a known mass of copper with excess sulfur in
EXPERIMENT 3 THE SYNTHESIS OF COPPER SULFIDE When heated together, copper and sulfur combine to form a sulfide of copper. In this assignment, you will heat a known mass of copper with excess sulfur in
Lesson Version 1.0 1/15 2 1
 Lesson 2 2-1 Version 1.0 1/15 2 1 Learning Outcomes Describe the difference between compaction and density and wet & dry density Calculate unit weight Describe the Proctor and LBR Tests and their use Identify
Lesson 2 2-1 Version 1.0 1/15 2 1 Learning Outcomes Describe the difference between compaction and density and wet & dry density Calculate unit weight Describe the Proctor and LBR Tests and their use Identify
MATERIAL SPECIFICATION FOR STANDARD HIGHWAY FENCE COMPONENTS
 ONTARIO PROVINCIAL STANDARD SPECIFICATION METRIC OPSS 1540 NOVEMBER 2012 MATERIAL SPECIFICATION FOR STANDARD HIGHWAY FENCE COMPONENTS TABLE OF CONTENTS 1540.01 SCOPE 1540.02 REFERENCES 1540.03 DEFINITIONS
ONTARIO PROVINCIAL STANDARD SPECIFICATION METRIC OPSS 1540 NOVEMBER 2012 MATERIAL SPECIFICATION FOR STANDARD HIGHWAY FENCE COMPONENTS TABLE OF CONTENTS 1540.01 SCOPE 1540.02 REFERENCES 1540.03 DEFINITIONS
RITES Lab, Western Region, 210, Amit Industrial Estate, 61, Dr. S.S. Rao Road, Parel, Mumbai, Maharashtra
 Last Amended on - Page 1 of 5 I. METALS & ALLOYS 1. Low-Alloy Steel Carbon ASTM E 415-08/ Manganese ASTM E 415-08/ Silicon ASTM E 415-08/ Sulphur ASTM E 415-08/ Phosphorous ASTM E 415-08/ Chromium ASTM
Last Amended on - Page 1 of 5 I. METALS & ALLOYS 1. Low-Alloy Steel Carbon ASTM E 415-08/ Manganese ASTM E 415-08/ Silicon ASTM E 415-08/ Sulphur ASTM E 415-08/ Phosphorous ASTM E 415-08/ Chromium ASTM
