1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
|
|
|
- Jocelyn Bates
- 6 years ago
- Views:
Transcription
1 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from other materials? How are elements classified? National Science Education Standards PS 1a, 1c What Are Elements? Many materials can be broken down into simpler materials. For example, some rocks contain copper. When they are heated in a large furnace, the copper separates from the rest of the rock. Another example is breaking down water by passing electricity through it. The electric current causes hydrogen gas and oxygen gas to form. Some materials cannot be separated or broken down into other materials. An element is a pure substance that cannot be separated into simpler substances by chemical or physical methods. This is how elements are different from all other materials. A pure substance is a material in which all the basic particles are the same. For example, table salt contains particles of sodium chloride. Table salt from anywhere is the same. All pure substances, except for elements, can be broken down into simpler substances. The basic particles of an element are called atoms. Copper is an example of an element. All of the atoms in a piece of pure copper are alike. As shown in the figure below, iron is also an element. STUDY TIP Graphic Organizer In your notebook, make a Concept Map by using the terms element, substance, metal, nonmetal, and metalloid. 1. Compare How does an element differ from other pure substances? The iron atoms in the meteorite from space are the same as the iron atoms in a steel spoon. There are also iron atoms in the cereal, in the boy s braces, and even in his blood. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else. Interactive Textbook 55 Elements, Compounds, and Mixtures
2 SECTION 1 Elements continued STANDARDS CHECK PS 1a A substance has characteristic properties, such as density, a boiling point, and solubility, all of which are independent of the amount of the sample. A mixture of substances often can be separated into the original substances using one or more of the characteristic properties. 3. List What are five physical properties that are characteristics of an element? How Can Elements Be Classified? Elements can be classified based on their properties. There are two types of properties, chemical and physical. Physical properties include hardness, melting point, and density. Chemical properties include reactivity and flammability. We can use properties to tell elements apart. For example, the elements helium and krypton are both colorless, odorless, unreactive gases. However, the elements have different densities (mass per unit volume). Helium is less dense than air, so a helium balloon floats upward. Krypton is denser than air. A krypton-filled balloon, on the other hand, will sink to the floor. The Unique Properties of Elements Critical Thinking 4. Infer Compare the properties of iron with those of cobalt and nickel. How do you think cobalt and nickel are used in manufactured products? Cobalt Melting point: 1,495 C Density: 8.9 g/cm 3 Reactivity: unreactive with oxygen in the air Iron Melting point: 1,535 C Density: 7.9 g/cm 3 Reactivity: reacts by combining with oxygen in the air to form rust. Nickel Melting point: 1,455 C Density: 8.9 g/cm 3 Reactivity: unreactive with oxygen in the air 5. Explain Why can t you use density or reactivity to determine whether a sample is cobalt or nickel? The figure above shows some of the properties of three different elements. The physical properties shown are melting point, electrical and thermal conductivities, and density. Each element has other physical properties, including color, hardness, and texture. The figure also includes a chemical property: the reactivity of the element with oxygen in the air. If you had a piece of metal, you could use these properties to determine which element it is. Iron has different physical and chemical properties than the other two elements. The density of iron is much less than cobalt or nickel, and it reacts with oxygen in the air. We can also use properties to tell nickel and cobalt apart. They have the same density and reactivity, but the melting points of these two elements differ by 40 C. This property can be used to tell them apart. Interactive Textbook 56 Elements, Compounds, and Mixtures
3 SECTION 1 Elements continued How Can Elements Be Sorted? Think about all the different types of dogs that you have seen. Dogs can be classified based on different properties. These include their size, the shape of the ears, or the length of their coat. You can often determine a dog s breed just with a quick glance. The figure below shows three kinds of terriers. They are not exactly alike, but they share certain features. TAKE A LOOK 6. Describe What are some of the physical properties that describe terriers? Even though these dogs are different breeds, they have enough in common to be classified as terriers. The elements can be classified based on properties, just like the dogs in the image. There are three major categories of elements: metals, nonmetals, and metalloids. The elements iron, cobalt, and nickel are all metals. They are not exactly alike, but they have similar properties. Metals tend to be shiny solids (except mercury, which is a shiny liquid). Metals conduct electric current and heat well. Nonmetals do not conduct heat or electric current very well. Many nonmetals are gases. The solid nonmetals have a dull appearance. Metalloids have some of the properties of metals and some of the properties of nonmetals. Metalloids are important in electronics because their electrical conductivity can vary with conditions. Three Major Categories of Elements Property Metals Nonmetals Metalloids Appearance shiny dull some are shiny Conductivity of heat and electricity good poor some do Malleable can be hammered into sheets Ductile can be made into wires yes no some are somewhat malleable yes no some are somewhat ductile Brittle no yes some are 7. Identify What are the three main categories of elements? Say It Explore Applications The properties of metals make them very useful in everyday things. In groups of three or four, make a list of things that you use for cooking that are made of metal. Make another list of things used for cooking that are never made of metal. Discuss why the properties of metal determined which things are in which group. Interactive Textbook 57 Elements, Compounds, and Mixtures
4 Section 1 Review NSES PS 1a, 1c SECTION VOCABULARY element a substance that cannot be separated or broken down into simpler substances by chemical means metal an element that is shiny and conducts heat and electricity well metalloid an element that has properties of both metals and nonmetals nonmetal an element that conducts heat and electricity poorly pure substance a sample of matter, either a single element or a single compound that has definite chemical and physical properties 1. Compare How does the ability to conduct heat differ between metals and nonmetals? 2. Classify Fill in each blank to complete the table. Element Property Classification Copper shiny solid Oxygen gas Silicon electrical conductivity varies depending on conditions 3. Evaluate Assumptions Your friend tells you that all of the electric wires in your home are metals. From what you know about elements, explain whether or not this statement is true. 4. Apply Concepts Several elements are placed between panes of glass in double windows to block heat flow. Should these elements be metals or nonmetals? Why? 5. Calculate Two elements, hydrogen and helium, make up most of the atoms in the universe. 92.7% of atoms are hydrogen and 6.9% of atoms are helium. What percentage of atoms are neither hydrogen nor helium? Show your work. Interactive Textbook 58 Elements, Compounds, and Mixtures
5 Physical Science Answer Key continued Chapter 4 Elements, Compounds, and Mixtures SECTION 1 ELEMENTS 1. Elements cannot be separated into simpler substances. 2. the meteorite 3. hardness, melting point, density, boiling point, solubility 4. Because their properties are so similar to iron s, cobalt and nickel can be used where a strong metal is needed, such as in structures, tools, or vehicles. 5. These properties are too similar in the two elements to be used to tell them apart. 6. small size; short, curly hair; shape of face 7. metals, nonmetals, metalloids Review 1. Metals are good heat conductors, and nonmetals are poor heat conductors. 2. Element Property Classification Copper shiny solid metal Oxygen gas nonmetal Silicon electrical conductivity (varies depending on conditions) metalloid 3. Possible answers: Yes, because metals conduct electric current and nonmetals don t. Yes, because electric wires are made of copper or aluminum, which are metals. No, because some metalloids conduct electric current, so it may be possible to use metalloids as wires. 4. The elements are nonmetals because all of the elements that are gases are nonmetals and because metals conduct heat % (92.7% 6.9%) 0.4% SECTION 2 COMPOUNDS 1. a pure substance composed of two or more elements that are joined by chemical bonds 2. 1:4 3. Room temperature is about 25 C. This value falls between the melting point and the boiling point of each of the three compounds. 4. sodium 5. They are different. 6. heat 7. proteins and carbohydrates Review 1. The particles of a compound contain atoms of more than one element. The particles of an element are the atoms of that element. 2. physical, chemical, elements, heat or electricity 3. There was a chemical reaction with something in the air. It formed a new compound that had properties different from those of copper. 4. heat and electricity 5. 1:2 SECTION 3 MIXTURES 1. a physical change 2. The components in the mixtures are not changed. 3. the flame or the burner 4. A pure substance has the same particles throughout, so it cannot separate into layers. 5. the water 6. The ratio of components in a mixture is not fixed, but a compound always has the same elements in the same ratio. 7. the solvent 8. water 9. It is not a solution, because the metals are not spread evenly throughout the coin. 10. Oxygen, carbon dioxide, alcohol, salt, and zinc should be circled. 11. amount of solute and amount of solution 12. You add more than the solubility of sugar in water g/100 ml of water grams of solute 14. concentration milliliters of solvent concentration 55 g ml 0.11 g/ml low temperatures 16. mixing by stirring, heating the solution, crushing the solid 17. a mixture in which the particles of a material are large enough to settle out 18. by passing it through a filter 19. a mixture in which the particles are spread throughout but are not large enough to settle out Interactive Textbook Answer Key 71 Physical Science
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Chapter 11 The Periodic Table
 Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
The table below shows some of the properties of the elements cobalt and nickel.
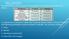 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Student Activity: Identifying Matter
 When you have completed this activity, go to Status Check. Physical Science A Unit 1 Student Activity: Identifying Matter Name Date Objectives In this activity, you will: evaluate data on different physical
When you have completed this activity, go to Status Check. Physical Science A Unit 1 Student Activity: Identifying Matter Name Date Objectives In this activity, you will: evaluate data on different physical
Metals. Key Concepts. Name Date Class. Chapter 4 Elements and the Periodic Table Section 3 Summary
 Chapter 4 Elements and the Periodic Table Section 3 Summary Metals Key Concepts What are the physical properties of metals? How does the reactivity of metals change across the periodic table? How are synthetic
Chapter 4 Elements and the Periodic Table Section 3 Summary Metals Key Concepts What are the physical properties of metals? How does the reactivity of metals change across the periodic table? How are synthetic
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet
 Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
S1 Building Blocks Summary Notes
 S1 Building Blocks Summary Notes Atoms & Molecules 1 We are developing our understanding of atoms and molecules. Atoms are the simplest building blocks of every substance in the universe. There are just
S1 Building Blocks Summary Notes Atoms & Molecules 1 We are developing our understanding of atoms and molecules. Atoms are the simplest building blocks of every substance in the universe. There are just
D =? D = M = 10 g = 5 g/cm 3 M = 10 g V 2 cm 3 V = 2 cm 3 5 g/cm 3
 Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Metals, Nonmetals, and Metalloids
 Metals, Nonmetals, and Metalloids Color and label your periodic table like the one below: Put the colored periodic table to the side, we will glue it in later Find this page, we will begin taking notes:
Metals, Nonmetals, and Metalloids Color and label your periodic table like the one below: Put the colored periodic table to the side, we will glue it in later Find this page, we will begin taking notes:
Chemistry Quiz #1 Review
 Name: Chemistry Quiz 1 Review Page 1 of 6 Date: Please check your answers at http://leetz.weebly.com Chemistry Quiz #1 Review Part A: Density Calculations 1. The density of sodium is 0.97 g/cm 3. A sample
Name: Chemistry Quiz 1 Review Page 1 of 6 Date: Please check your answers at http://leetz.weebly.com Chemistry Quiz #1 Review Part A: Density Calculations 1. The density of sodium is 0.97 g/cm 3. A sample
The City School PAF Chapter
 The City School PAF Chapter Comprehensive Worksheet December 2018 SCIENCE Class 7 Candidate Name: Index Number: Section: Branch/Campus: Date: Maximum Marks: 100 Time Allowed: 1 hour 30 minutes INSTRUCTIONS:
The City School PAF Chapter Comprehensive Worksheet December 2018 SCIENCE Class 7 Candidate Name: Index Number: Section: Branch/Campus: Date: Maximum Marks: 100 Time Allowed: 1 hour 30 minutes INSTRUCTIONS:
History of the Periodic Table
 Periodic Trends History of the Periodic Table Many scientists suspected a pattern in the order of the elements, but were not able to exactly figure it out. Dmitri Mendeleev was the first to create an accurate
Periodic Trends History of the Periodic Table Many scientists suspected a pattern in the order of the elements, but were not able to exactly figure it out. Dmitri Mendeleev was the first to create an accurate
The table shows the students suggestions about the identity of P.
 1 Three students, X, Y and Z, were told that solid P reacts with dilute acids and also conducts electricity. The table shows the students suggestions about the identity of P. Which of the students are
1 Three students, X, Y and Z, were told that solid P reacts with dilute acids and also conducts electricity. The table shows the students suggestions about the identity of P. Which of the students are
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
The table gives some information about a family of molecules in crude oil. Show information from the table in the most appropriate way on the grid.
 ## The table gives some information about a family of molecules in crude oil. NUMBER OF CARBON ATOMS IN MOLECULE MASS OF MOLECULE (atomic units) 1 16 2 30 4 58 Show information from the table in the most
## The table gives some information about a family of molecules in crude oil. NUMBER OF CARBON ATOMS IN MOLECULE MASS OF MOLECULE (atomic units) 1 16 2 30 4 58 Show information from the table in the most
Name Class Date. Does it have a crystalline structure? Minerals are crystals. Each mineral has a certain crystal structure that is always the same.
 CHAPTER 1 1 What Is a Mineral? SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What are minerals? What determines the shape
CHAPTER 1 1 What Is a Mineral? SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What are minerals? What determines the shape
METALS AND NONMETALS
 Suggested time allotment: 5 to 6 hours MODULE 5 METALS AND NONMETALS Elements are the simplest form of substances. This means that whatever you do with an element, it remains to be the same element. Its
Suggested time allotment: 5 to 6 hours MODULE 5 METALS AND NONMETALS Elements are the simplest form of substances. This means that whatever you do with an element, it remains to be the same element. Its
Science 8. Unit 1. Booklet
 Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
21.The smallest part of an element that keeps the same properties of that element. a. Element b. Compound c. Atom d. quark
 21.The smallest part of an element that keeps the same properties of that element a. Element b. Compound c. Atom d. quark 22.A substance made of 2 or more elements chemically combined a. Compound b. Mixture
21.The smallest part of an element that keeps the same properties of that element a. Element b. Compound c. Atom d. quark 22.A substance made of 2 or more elements chemically combined a. Compound b. Mixture
EXPERIMENT 5. Physical and Chemical Changes Part 1 INTRODUCTION
 EXPERIMENT 5 Physical and Chemical Changes Part 1 INTRODUCTION Matter undergoes many changes. In some cases only the form of the substance (such as physical state, size of particle, or temperature) is
EXPERIMENT 5 Physical and Chemical Changes Part 1 INTRODUCTION Matter undergoes many changes. In some cases only the form of the substance (such as physical state, size of particle, or temperature) is
Read pg Answer pg 157 #1-5, & pg159 #2-7
 4.2 Physical Properties HOMEWORK Read pg 149-159 Answer pg 157 #1-5, & pg159 #2-7 HOMEWORK Read pg 149-159 Answer pg 157 #1-5, & pg159 #2-7 Learning Goals I can describe the physical properties of matter
4.2 Physical Properties HOMEWORK Read pg 149-159 Answer pg 157 #1-5, & pg159 #2-7 HOMEWORK Read pg 149-159 Answer pg 157 #1-5, & pg159 #2-7 Learning Goals I can describe the physical properties of matter
Before Statement After
 CHAPTER 10 The Periodic Table LESSON 2 Metals What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the
CHAPTER 10 The Periodic Table LESSON 2 Metals What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the
Physical Science Chapter 19. Elements and Their Properties Quick Notes
 Physical Science Chapter 19 Elements and Their Properties Quick Notes 1 19:1 A.Metals conduct heat and electricity, reflect light (luster); are malleable ( ) Can be hammered or rolled into sheets are ductile
Physical Science Chapter 19 Elements and Their Properties Quick Notes 1 19:1 A.Metals conduct heat and electricity, reflect light (luster); are malleable ( ) Can be hammered or rolled into sheets are ductile
1. Which of the following elements has the highest percentage by mass in nature? A. Oxygen B. Aluminium C. Nitrogen D. Silicon
 Class: F.3 ( ) Baptist Lui Ming Choi Secondary School First Term Examination (2013-2014) Date: 6 / 12 / 2013 Name: Form 3 Chemistry Time: 10:20-11:05 a.m. Answer ALL the questions. For Section A, choose
Class: F.3 ( ) Baptist Lui Ming Choi Secondary School First Term Examination (2013-2014) Date: 6 / 12 / 2013 Name: Form 3 Chemistry Time: 10:20-11:05 a.m. Answer ALL the questions. For Section A, choose
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Metal and Non Metals
 Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Corresponding Physical or Chemical Property bread rising through the use of baking soda carbonates decompose to form CO 2
 Substance = a chemical (element or mixture) Substance mixture chemical process = chemical reaction (new substances result) physical process = same substances present through out Physical or chemical processes?
Substance = a chemical (element or mixture) Substance mixture chemical process = chemical reaction (new substances result) physical process = same substances present through out Physical or chemical processes?
Properties of metals
 For more awesome resources, visit us at www.savemyexams.co.uk/ Properties of metals Question Paper Level IGSE Subject hemistry (060/097) Exam oard ambridge International Examinations (IE) Topic Metals
For more awesome resources, visit us at www.savemyexams.co.uk/ Properties of metals Question Paper Level IGSE Subject hemistry (060/097) Exam oard ambridge International Examinations (IE) Topic Metals
An Organized Table Worksheet Due Thursday Name: Date: Period:
 An Organized Table Worksheet Due Thursday Name: Date: Period: The Periodic Table of Elements In 1871, the first periodic table was developed by Dmitrii Mendeleev. Mendeleev is known as the father of the
An Organized Table Worksheet Due Thursday Name: Date: Period: The Periodic Table of Elements In 1871, the first periodic table was developed by Dmitrii Mendeleev. Mendeleev is known as the father of the
Key Points. ATOMIC STRUCTURE Atom: the smallest part of an element that still has the characteristics/ properties of that element.
 The Periodic Table Learning Objective Red Yellow Green Date Completed OC3 understand what an element is and recall that all known elements are listed in the Periodic Table; (also know how to use the Table;
The Periodic Table Learning Objective Red Yellow Green Date Completed OC3 understand what an element is and recall that all known elements are listed in the Periodic Table; (also know how to use the Table;
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
CHAPTER 3 Electronic Structure and the Periodic Law
 CHAPTER 3 Electronic Structure and the Periodic Law . Metals, Nonmetals, and Metalloids Metals, Metalloids, and Nonmetals in the Periodic Table Metals, Nonmetals, and Metalloids Metals, Metalloids, and
CHAPTER 3 Electronic Structure and the Periodic Law . Metals, Nonmetals, and Metalloids Metals, Metalloids, and Nonmetals in the Periodic Table Metals, Nonmetals, and Metalloids Metals, Metalloids, and
2.2 Physical Properties
 2.2 Physical Properties A physical property is any characteristic of a material that can be observed or measured without changing the composition of the substances in the material. Examples of Physical
2.2 Physical Properties A physical property is any characteristic of a material that can be observed or measured without changing the composition of the substances in the material. Examples of Physical
Materials are all substances and include metals, ceramics and plastics as well as natural and new substances.
 National 4 Materials It is hard to imagine life without mobile gadgets such as iphones, ipads and MP3 players. Yet twenty years ago these handy gadgets such as the mobile phone where bigger and cost five
National 4 Materials It is hard to imagine life without mobile gadgets such as iphones, ipads and MP3 players. Yet twenty years ago these handy gadgets such as the mobile phone where bigger and cost five
Organizing the Elements
 Organizing the Elements In a self-service store, the products are grouped according to similar characteristics. With a logical classification system, finding and comparing products is easy. You will learn
Organizing the Elements In a self-service store, the products are grouped according to similar characteristics. With a logical classification system, finding and comparing products is easy. You will learn
Draw a ring around the correct word in the box to complete the sentence.
 Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
NOTES ON CHAPTER 4: ELEMENTS AND THE PERIODIC TABLE. 4.3 Metals
 NOTES ON CHAPTER 4: ELEMENTS AND THE PERIODIC TABLE 4.3 Metals Metals The physical properties of metals include: shininess- ability to reflect light malleability ability to be hammered or rolled into flat
NOTES ON CHAPTER 4: ELEMENTS AND THE PERIODIC TABLE 4.3 Metals Metals The physical properties of metals include: shininess- ability to reflect light malleability ability to be hammered or rolled into flat
I. PHYSICAL PROPERTIES PROPERTY METALS NON-METALS
 Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
NOTE: If substances have the SAME VOLUME, they can have DIFFERENT MASSES.
 Grade 8 SCIENCE Chapter 8 questions Page 306 1. When particles are CLOSER TOGETHER, DENSITY is GREATER. 2. Particles in SOLIDS have FEWER SPACES between them. The ATTRACTIVE FORCES between the particles
Grade 8 SCIENCE Chapter 8 questions Page 306 1. When particles are CLOSER TOGETHER, DENSITY is GREATER. 2. Particles in SOLIDS have FEWER SPACES between them. The ATTRACTIVE FORCES between the particles
Organizing the Elements
 Organizing the Elements 1 of 28 Organizing the Elements In a self-service store, the products are grouped according to similar characteristics. With a logical classification system, finding and comparing
Organizing the Elements 1 of 28 Organizing the Elements In a self-service store, the products are grouped according to similar characteristics. With a logical classification system, finding and comparing
Naming and Classifying the Elements. nobelium antimony. oxygen arsenic. phosphorus beryllium. platinum bismuth. plutonium boron.
 6.2 Naming and Classifying the Elements To learn more about the origins of the names of the elements, visit www.science.nelson.com GO The names of the elements have varied origins. Some elements are named
6.2 Naming and Classifying the Elements To learn more about the origins of the names of the elements, visit www.science.nelson.com GO The names of the elements have varied origins. Some elements are named
(b) Fertilizers help to increase agricultural production. chemical property physical property
 Score 1. [Chang7 1.P.011.] Do the following statements describe chemical or physical properties? (a) Oxygen gas supports combustion. chemical property physical property (b) Fertilizers help to increase
Score 1. [Chang7 1.P.011.] Do the following statements describe chemical or physical properties? (a) Oxygen gas supports combustion. chemical property physical property (b) Fertilizers help to increase
1. Marie mixed 5 g of carbon with 5 g of lead oxide. She heated the mixture strongly for 15 minutes in a fume cupboard.
 1. Marie mixed 5 g of carbon with 5 g of lead oxide. She heated the mixture strongly for 15 minutes in a fume cupboard. After 15 minutes, Marie found some shiny beads in the mixture. a. (i) Marie collected
1. Marie mixed 5 g of carbon with 5 g of lead oxide. She heated the mixture strongly for 15 minutes in a fume cupboard. After 15 minutes, Marie found some shiny beads in the mixture. a. (i) Marie collected
REVISION NEW LEARNING
 REVISION I have developed my knowledge of the Periodic Table by considering the properties and uses of a variety of elements relative to their positions. SCN 3-15a S2 Chemistry Metals Lesson 1 NEW LEARNING
REVISION I have developed my knowledge of the Periodic Table by considering the properties and uses of a variety of elements relative to their positions. SCN 3-15a S2 Chemistry Metals Lesson 1 NEW LEARNING
Look at the measuring cylinders. What happened to the volume of the water and the wax after freezing? the volume of water... the volume of wax...
 1. Meera poured 7 cm 3 of water into a measuring cylinder. She poured 7 cm 3 of melted wax into another measuring cylinder. She put both measuring cylinders into a freezer for 24 hours. water before freezing
1. Meera poured 7 cm 3 of water into a measuring cylinder. She poured 7 cm 3 of melted wax into another measuring cylinder. She put both measuring cylinders into a freezer for 24 hours. water before freezing
Oxygen Formula: O 2 Bonding: covalent Appearance: colourless gas. Oxygen is one of the two main gases in our atmosphere, the other being nitrogen.
 Composition of the air Air is a mixture of gases. The approximate amount if each gas in dry air is shown in the pie chart (right), but you should be aware that air also contains a variable amount of water
Composition of the air Air is a mixture of gases. The approximate amount if each gas in dry air is shown in the pie chart (right), but you should be aware that air also contains a variable amount of water
Atoms and elements/compounds and mixtures
 Medway LEA Advisory Service Atoms and elements/compounds and mixtures 8E & 8F 26 min 26 marks Q1-L3, Q2-L4, Q3-L4, Q4-L5, Q5-L6, Q6-L7 Medway LEA Advisory Service 1 0 10 C 1. (a) The drawings below show
Medway LEA Advisory Service Atoms and elements/compounds and mixtures 8E & 8F 26 min 26 marks Q1-L3, Q2-L4, Q3-L4, Q4-L5, Q5-L6, Q6-L7 Medway LEA Advisory Service 1 0 10 C 1. (a) The drawings below show
GCSE BITESIZE Examinations
 GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
Properties of Elements
 Properties of Elements Pre-Lab: (Answer questions on your data sheet.) 1. Using a periodic table, determine and write the chemical symbol for each of the eight elements listed on your data sheet. 2. Predict
Properties of Elements Pre-Lab: (Answer questions on your data sheet.) 1. Using a periodic table, determine and write the chemical symbol for each of the eight elements listed on your data sheet. 2. Predict
THE PERIODIC TABLE. Chapter 15
 THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
CLASSI ICATION OF MAT R AND HOMOGENEOUS AND HETEROGENEOUS MIXTURES
 Experiment 3 Name: CLASSI ICATION OF MAT R AND HOMOGENEOUS AND HETEROGENEOUS MIXTURES Classification of Matter A pure substance is matter with definite and constant composition with distinct chemical properties.
Experiment 3 Name: CLASSI ICATION OF MAT R AND HOMOGENEOUS AND HETEROGENEOUS MIXTURES Classification of Matter A pure substance is matter with definite and constant composition with distinct chemical properties.
Topic 9 National 4 Chemistry Summary Notes. Metals and Alloys. Materials
 Topic 9 National 4 Chemistry Summary Notes Metals and Alloys LI 1 Materials Materials are all substances and include: metals ceramics plastics natural substances novel substances. Materials can be used
Topic 9 National 4 Chemistry Summary Notes Metals and Alloys LI 1 Materials Materials are all substances and include: metals ceramics plastics natural substances novel substances. Materials can be used
Objective: Classify metals, nonmetals, and metalloids based on their properties.
 Do Now Date: September 2, 2014 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
Do Now Date: September 2, 2014 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
Creation Settings. can be separated into two or more pure substances by distillation. can be separated into two or more substances by chromatography.
 Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Classifying Matter. Do they look different? Do they feel different? Do they behave the same way when they are dropped?
 Classifying Matter Standard: 5 PS1 3 Make observations and measurements to identify materials based on their properties. [Clarification Statement: Examples of materials to be identified could include baking
Classifying Matter Standard: 5 PS1 3 Make observations and measurements to identify materials based on their properties. [Clarification Statement: Examples of materials to be identified could include baking
Properties of Elements
 Pre-Lab: (Answer questions on your data sheet.) Properties of Elements 1. Using a periodic table, determine and write the chemical symbol for each of the eight elements listed on your data sheet. 2. Predict
Pre-Lab: (Answer questions on your data sheet.) Properties of Elements 1. Using a periodic table, determine and write the chemical symbol for each of the eight elements listed on your data sheet. 2. Predict
Chapter: The Periodic Table
 Table of Contents Chapter: The Periodic Table Section 1: Introduction to the Periodic Table Section 2: Representative Elements Section 3: Transition Elements 1 Introduction to the Periodic Table Development
Table of Contents Chapter: The Periodic Table Section 1: Introduction to the Periodic Table Section 2: Representative Elements Section 3: Transition Elements 1 Introduction to the Periodic Table Development
Name Honors Chemistry / /
 Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Strong under tension and compression. Malleable. Low density. Have a dull appearance. Good conductors of electricity and heat
 Revision from Year 10: Properties of Metals and Non-Metals Read CC pp182-183 Use arrows to link the properties with the materials: Strong under tension and compression Malleable Low density Have a dull
Revision from Year 10: Properties of Metals and Non-Metals Read CC pp182-183 Use arrows to link the properties with the materials: Strong under tension and compression Malleable Low density Have a dull
2 Identifying Minerals
 CHAPTER 1 2 Identifying Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What seven properties can be used to identify
CHAPTER 1 2 Identifying Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What seven properties can be used to identify
GSHS Y9 Synoptic Test Revision Mark Scheme Booklet 1. Cells/reproduction 7A & 7B. 35 min 35 marks Q1-L3, Q2-L4, Q3-L5, Q4-L5, Q5-L6, Q6-L6
 Y9 Synoptic Test Revision Mark Scheme Booklet 1 Cells/reproduction 7A & 7B 35 min 35 marks Q1-L3, Q2-L4, Q3-L5, Q4-L5, Q5-L6, Q6-L6 1. (a) not enough light accept no light or not enough Sun 1 or no chlorophyll
Y9 Synoptic Test Revision Mark Scheme Booklet 1 Cells/reproduction 7A & 7B 35 min 35 marks Q1-L3, Q2-L4, Q3-L5, Q4-L5, Q5-L6, Q6-L6 1. (a) not enough light accept no light or not enough Sun 1 or no chlorophyll
NCERT solutions for Metals and Non Metals
 NCERT solutions for Metals and Non Metals 1 Question 1 Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv)
NCERT solutions for Metals and Non Metals 1 Question 1 Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv)
Covered with a thin layer of oxide at ordinary temperatures.
 1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
Objective: Classify metals, nonmetals, and metalloids based on their properties.
 Do Now Date: September 6, 2016 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday, September
Do Now Date: September 6, 2016 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday, September
Explain how metalloids are different from metals and nonmetals.
 Explain how metalloids are different from metals and nonmetals. Explain how the periodic table is organized based on physical and chemical properties. Where are the metals, nonmetals, and metalloids located
Explain how metalloids are different from metals and nonmetals. Explain how the periodic table is organized based on physical and chemical properties. Where are the metals, nonmetals, and metalloids located
Skills in Science. Lab equipment. (Always draw 2D) Drawings below are NOT to scale. Beaker - A general purpose container with a pouring lip.
 Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
2.2 Physical Properties
 There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that is hard to miss. A yellow color
There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that is hard to miss. A yellow color
I. PHYSICAL PROPERTIES. PROPERTY METALS NON-METALS 1.Lustre Metals have shining surface. They do not have shining surface.
 Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
Contact us:
 Class X Chapter 3 Metals and Non-metals Science Question 1: Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat.
Class X Chapter 3 Metals and Non-metals Science Question 1: Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat.
Page 2. Q1.Copper is a transition metal. (a) (i) Where is copper in the periodic table? in the central block. in Group 1. in the noble gas group
 Q1.Copper is a transition metal. (a) (i) Where is copper in the periodic table? Tick ( ) one box. in the central block in Group 1 in the noble gas group (ii) What is a property of copper? Tick ( ) one
Q1.Copper is a transition metal. (a) (i) Where is copper in the periodic table? Tick ( ) one box. in the central block in Group 1 in the noble gas group (ii) What is a property of copper? Tick ( ) one
Iÿtÿÿeÿ/ÿrh Jÿtcket. Tÿlchÿwÿeÿ
 o 0 Iÿtÿÿeÿ/ÿrh Jÿtcket Tÿlchÿwÿeÿ Elements, Compounds, and Mixtures Classify each of the pictures below by placing the correct label in the blanks below: A= Element D-- Mixture of compounds B= Compound
o 0 Iÿtÿÿeÿ/ÿrh Jÿtcket Tÿlchÿwÿeÿ Elements, Compounds, and Mixtures Classify each of the pictures below by placing the correct label in the blanks below: A= Element D-- Mixture of compounds B= Compound
SCIENCE REVISION CARDS For KS3 and Common Entrance
 SCIENCE REVISION CARDS For KS3 and Common Entrance I recommend printing the following pages onto card and cutting them into 4 equal pieces. They will then fit into a file box for revision purposes. Many
SCIENCE REVISION CARDS For KS3 and Common Entrance I recommend printing the following pages onto card and cutting them into 4 equal pieces. They will then fit into a file box for revision purposes. Many
Year 7 Chemistry HW Questions
 Year 7 Chemistry HW Questions 37 minutes 56 marks Page 1 of 15 Q1. Molly used a ph sensor to test different liquids. She dipped the probe of the sensor into each liquid and recorded the ph value in a table.
Year 7 Chemistry HW Questions 37 minutes 56 marks Page 1 of 15 Q1. Molly used a ph sensor to test different liquids. She dipped the probe of the sensor into each liquid and recorded the ph value in a table.
Processing of Non-Metals Prof. Dr. Inderdeep Singh Department of Mechanical and Industrial Engineering Indian Institute of Technology, Roorkee
 Processing of Non-Metals Prof. Dr. Inderdeep Singh Department of Mechanical and Industrial Engineering Indian Institute of Technology, Roorkee Module - 1 Engineering Materials and Processing Lecture -
Processing of Non-Metals Prof. Dr. Inderdeep Singh Department of Mechanical and Industrial Engineering Indian Institute of Technology, Roorkee Module - 1 Engineering Materials and Processing Lecture -
Materials : Metals and Non-Metals
 18 EXEMPLAR PROBLEMS 4 Materials : Metals and Non-Metals MULTIPLE CHOICE QUESTIONS 1. Which of the following is not a metal? (a) copper (c) aluminium (b) sulphur (d) iron 2. The substance that will be
18 EXEMPLAR PROBLEMS 4 Materials : Metals and Non-Metals MULTIPLE CHOICE QUESTIONS 1. Which of the following is not a metal? (a) copper (c) aluminium (b) sulphur (d) iron 2. The substance that will be
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
Metals. General information
 Metals. General information Read and copy the following text: Metals are divided into two groups: Metals that contain iron are called Ferrous Metals (Alloys). Metals that do not contain iron are called
Metals. General information Read and copy the following text: Metals are divided into two groups: Metals that contain iron are called Ferrous Metals (Alloys). Metals that do not contain iron are called
WJEC England GCSE Chemistry. Topic 11: Production, use and disposal of important chemicals and materials. Notes
 WJEC England GCSE Chemistry Topic 11: Production, use and disposal of important chemicals and materials Notes (Content in bold is for Higher Tier only) The Haber process Used to manufacture ammonia, which
WJEC England GCSE Chemistry Topic 11: Production, use and disposal of important chemicals and materials Notes (Content in bold is for Higher Tier only) The Haber process Used to manufacture ammonia, which
Objective: Classify metals, nonmetals, and metalloids based on their properties.
 Do Now Date: September 8, 2015 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
Do Now Date: September 8, 2015 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
Metals. N4 & N5 Homework Questions
 St Peter the Apostle High School Chemistry Department Metals N4 & N5 Homework Questions Answer questions as directed by your teacher. National 4 level questions are first followed by National 5 level questions.
St Peter the Apostle High School Chemistry Department Metals N4 & N5 Homework Questions Answer questions as directed by your teacher. National 4 level questions are first followed by National 5 level questions.
Page 1 of 15. Website: Mobile:
 Question 1: Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv) is a poor conductor of heat. (i) Metal that
Question 1: Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv) is a poor conductor of heat. (i) Metal that
Name # Date Period! FINDING&VOLUME&AND&DENSITY& !!! !!!!!!!
 Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Classification of matter: Properties *
 OpenStax-CNX module: m38706 1 Classification of matter: Properties * Free High School Science Texts Project This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
OpenStax-CNX module: m38706 1 Classification of matter: Properties * Free High School Science Texts Project This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
NATURAL GAS Formation of a Clean Source of Energy
 NATURAL GAS Formation of a Clean Source of Energy LESSON SUMMARY This lesson explores how natural gas was formed by identifying states of matter, density, and how density factors into changes in states.
NATURAL GAS Formation of a Clean Source of Energy LESSON SUMMARY This lesson explores how natural gas was formed by identifying states of matter, density, and how density factors into changes in states.
Elements and Compounds
 Chemistry Name: Partners: Date: Elements and Compounds Introduction Most of the materials you have used so far in this course were mixtures. If you could take them apart physically, you would find that
Chemistry Name: Partners: Date: Elements and Compounds Introduction Most of the materials you have used so far in this course were mixtures. If you could take them apart physically, you would find that
2A Chemistry - Classification of Substances
 CHEMISTRY The world is made up of a variety of substances. Some of these occur naturally in our environment, others are made through the combination of naturally occurring substances to form new materials.
CHEMISTRY The world is made up of a variety of substances. Some of these occur naturally in our environment, others are made through the combination of naturally occurring substances to form new materials.
3 The Formation, Mining, and Use of Minerals
 CHAPTER 1 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 1 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
A. Lemon juice makes the water more conducting as it conducts electricity. But when the lemon juice is not added to it, the conductivity will be low.
 Chapter 14-Chemical Effects of Electric Current Topicwise Assignment Chemical Effects of Electric Current Do Liquids Conduct Electricity? A. 1. Yes 2. Yes 3. Lemon juice is a good conductor. 4. The ends
Chapter 14-Chemical Effects of Electric Current Topicwise Assignment Chemical Effects of Electric Current Do Liquids Conduct Electricity? A. 1. Yes 2. Yes 3. Lemon juice is a good conductor. 4. The ends
NATIONAL 4 CHEMISTRY
 Farr High School NATIONAL 4 CHEMISTRY Unit 3 Chemistry in Society Question Booklet 1 Properties of Metals and Alloys 1. The grid below shows the names of some metals. You can use your data book to help
Farr High School NATIONAL 4 CHEMISTRY Unit 3 Chemistry in Society Question Booklet 1 Properties of Metals and Alloys 1. The grid below shows the names of some metals. You can use your data book to help
1. Hardness 2. Streak 3. Density 4. Crystal structure 5. A mineral must be a naturally occurring, inorganic. 7. c 8. f 9. a 10. d 11. g 12. e 13.
 Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Metals and Alloys
 Properties of Metals and Alloys 1 of 19 Boardworks Ltd 2016 Properties of Metals and Alloys 2 of 19 Boardworks Ltd 2016 What is the structure of metals? 3 of 19 Boardworks Ltd 2016 Metal particles are
Properties of Metals and Alloys 1 of 19 Boardworks Ltd 2016 Properties of Metals and Alloys 2 of 19 Boardworks Ltd 2016 What is the structure of metals? 3 of 19 Boardworks Ltd 2016 Metal particles are
CONNECTICUT SCIENCE FRAMEWORK. Grade 6
 CONNECTICUT SCIENCE FRAMEWORK Grade 6 Core Themes, Content Standards and Expected Performances Properties of Matter How does the structure of matter affect the properties and uses of materials? 6.1 - Materials
CONNECTICUT SCIENCE FRAMEWORK Grade 6 Core Themes, Content Standards and Expected Performances Properties of Matter How does the structure of matter affect the properties and uses of materials? 6.1 - Materials
Suggest one reason why spoons are electroplated. ... Why is hydrogen produced at the negative electrode and not sodium?
 Q1.This question is about electrolysis. (a) Metal spoons can be coated with silver. This is called electroplating. Suggest one reason why spoons are electroplated. (b) When sodium chloride solution is
Q1.This question is about electrolysis. (a) Metal spoons can be coated with silver. This is called electroplating. Suggest one reason why spoons are electroplated. (b) When sodium chloride solution is
Chapters 1&2 Practice Problems
 Name Chapters 1&2 Practice Problems Multiple Choice No Calculators Allowed! 1. Which physical state of matter exhibits the greatest change in volume with changes in temperature or pressure? a) solid b)
Name Chapters 1&2 Practice Problems Multiple Choice No Calculators Allowed! 1. Which physical state of matter exhibits the greatest change in volume with changes in temperature or pressure? a) solid b)
Experiment #3. Density
 Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
SAMPLE COURSE OUTLINE CHEMISTRY GENERAL YEAR 12
 SAMPLE COURSE OUTLINE CHEMISTRY GENERAL YEAR 12 Copyright School Curriculum and Standards Authority, 2015 This document apart from any third party copyright material contained in it may be freely copied,
SAMPLE COURSE OUTLINE CHEMISTRY GENERAL YEAR 12 Copyright School Curriculum and Standards Authority, 2015 This document apart from any third party copyright material contained in it may be freely copied,
4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. sulfur 16
 4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. silver 47 sulfur 16 copper 29 Some are named from the natural substance that they are
4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. silver 47 sulfur 16 copper 29 Some are named from the natural substance that they are
Microbes and Disease & Ecological Relationships
 Microbes and Disease & Ecological Relationships 8C & 8D 26 min 27 marks Q1-L4, Q2-L4, Q3-L5, Q4-L5, Q5-L6, Q6-L6 1. (a) any one from 1 bacteria do not accept 'germs' or 'microbes' viruses fungi (b) (i)
Microbes and Disease & Ecological Relationships 8C & 8D 26 min 27 marks Q1-L4, Q2-L4, Q3-L5, Q4-L5, Q5-L6, Q6-L6 1. (a) any one from 1 bacteria do not accept 'germs' or 'microbes' viruses fungi (b) (i)
GCSE MARKING SCHEME SUMMER 2016 SCIENCE - CHEMISTRY C1 4462/01/02. WJEC CBAC Ltd.
 GCSE MARKING SCHEME SUMMER 2016 SCIENCE - CHEMISTRY C1 4462/01/02 INTRODUCTION This marking scheme was used by WJEC for the 2016 examination. It was finalised after detailed discussion at examiners' conferences
GCSE MARKING SCHEME SUMMER 2016 SCIENCE - CHEMISTRY C1 4462/01/02 INTRODUCTION This marking scheme was used by WJEC for the 2016 examination. It was finalised after detailed discussion at examiners' conferences
