1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter.
|
|
|
- Charla Rodgers
- 6 years ago
- Views:
Transcription
1 1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through 5 on the diagram below, which represents a solid material of uniform composition. 4. When this material is placed in a container of water, it sinks to the bottom of the container. Compared to the density of water, the density of this material is A) less B) greater C) the same 5. The mass of this piece of material is approximately A) 0.23 g B) 4.4 g C) 9.3 g D) 32 g 6. As air on the surface of Earth warms, the density of the air A) decreases B) increases C) remains the same 7. Under the same conditions of temperature and pressure, three different samples of the same uniform substance will have the same A) shape B) density C) mass D) volume Base your answers to questions 8 through 10 on the diagrams below, which represent two different solid, uniform materials cut into cubes A and B. 2. Which graph best represents the relationship between the mass and volume of various-sized pieces of this material? 3. If this material is heated and expands, the density of the material will A) decrease B) increase C) remain the same 8. What is the density of cube A? A) 0.2 g/cm 3 B) 5.0 g/cm 3 C) 12.8 g/cm 3 D) 64.0 g/cm 3 9. If a parcel of air is heated, its density will A) decrease B) increase C) remain the same 10. Assume cube B was broken into many irregularly shaped pieces. Compared to the density of the entire cube, the density of one of the pieces would be A) less B) greater C) the same
2 11. The data table below shows the mass and volume of three samples of the same mineral. [The density column is provided for student use.] 13. Which graph best represents the relationship between the density of a substance and its state of matter (phase) for most earth materials, excluding water? [Key: S = solid, L = liquid, G = gas] Which graph best represents the relationship between the density and the volume of these mineral samples? 12. The mineral shown below is of uniform composition and has a density of 4 grams per cubic centimeter. What is the mass of this mineral? A) 9 g B) 12 g C) 54 g D) 108 g
3 14. The cartoon below presents a humorous look at science. The correct explanation of why ice floats is that, compared to liquid water, solid ice A) has less mass B) has more mass C) is less dense D) is more dense 15. The diagram below is a cross section of an 17. The diagrams below represent two differently ice-covered lake in New York State during the shaped blocks of ice floating in water. Which month of January. Points A, B, C, and D are diagram most accurately shows the blocks of ice as locations at various levels in the lake. The they would actually float in water? temperature of the water at location D is 4 C. Which graph best represents the relationship between location and density of the ice or water? 16. Compared to the density of liquid water, the density of an ice cube is A) always less B) always greater C) always the same D) sometimes less and sometimes greater 18. A pebble has a mass of 35 grams and a volume of 14 cubic centimeters. What is its density? A) 0.4 g/cm³ B) 2.5 g/cm³ C) 490 g/cm³ D) 4.0 g/cm³ 19. The diagram below shows a glass jar containing a clear liquid and a floating rock. Which conclusion about the relative density of the rock and the liquid is true? A) The rock is less dense than the liquid. B) The rock is more dense than the liquid. C) The rock and the liquid have the same density.
4 20. The diagram at the right represents a cylinder which contains four different liquids, W, X, Y, and Z, each with a different density (D) as indicated. A piece of solid quartz having a density of 2.7 g/cm 3 is placed on the surface of liquid W. When the quartz is released, it will pass through 22. A quantity of water is frozen solid and then heated from 0ºC to 10ºC. Which statement best describes the properties of the water during this time? A) Mass and volume change. B) Volume and density change. C) Mass changes but volume remains constant. D) Volume changes but density remains constant. 23. Which graph best represents the relationship between mass and volume of a material that has a density of 5 grams per cubic centimeter? A) W, but not X, Y, or Z B) W and X, but not Y or Z C) W, X, and Y, but not Z D) W, X, Y, and Z 21. The data table below shows the density of four different mineral samples. 24. The graph below shows the relationship between the mass and volume of a mineral. A student accurately measured the mass of a sample of one of the four minerals to be grams and its volume to be 73.6 cm 3. Which mineral sample did the student measure? A) corundum B) galena C) hematite D) quartz What is the density of this mineral? A) 6.0 g/cm 3 B) 9.0 g/cm 3 C) 3.0 g/cm 3 D) 4.5 g/cm 3
5 25. A 5.0-milliliter sample of a substance has a mass of 12.5 grams. What is the mass of a 100 milliliter sample of the same substance? A) 40.0 g B) 125 g C) 250 g D) 400 g 26. As shown below, an empty 1,000.-milliliter container has a mass of grams. When filled with a liquid, the container and the liquid have a combined mass of 1,300. grams. What is the density of the liquid? A) 1.00 g/ml B) 1.05 g/ml C) 1.30 g/ml D) 0.95 g/ml 27. A black hole is a celestial feature believed to have a mass millions of times the mass of our Sun and a diameter less than the diameter of Earth. An object of such high mass and small volume would have A) a very low density B) a very high density C) an elliptical orbit with the Sun at one focal point D) an elliptical orbit with Earth at one focal point
6 Base your answers to questions 28 and 29 on the diagrams below, and your knowledge of Earth science. The diagrams represent five substances, A through E, at the same temperature. Some mass, volume, and density values are indicated for each substance. Substance C is a liquid in a graduated cylinder. [Note that 1 cubic centimeter = 1 milliliter. Objects are not drawn to scale.] 28. Water (W) was added to the graduated cylinder containing liquid C. Objects A and D were then dropped into the cylinder. Which diagram most accurately shows the resulting arrangement of these substances? A) B) C) D) 29. Which two substances could be made of the same material? A) A and B B) B and E C) C and D D) A and E
7 30. The graph below shows the relationship between mass and volume for three samples, A, B, andc, of a given material. What is the density of this material? A) 1.0 g/cm 3 B) 5.0 g/cm 3 C) 10.0 g/cm 3 D) 20.0 g/cm Base your answer to the following question on the data table below, which lists some properties of four minerals that are used as ores of zinc (Zn). A sample of sphalerite has a mass of grams. What is the volume of the sample? A) 22.7 cm 3 B) 31.4 cm 3 C) 40.0 cm 3 D) 44.0 cm 3
8 32. A rock sample has a mass of 16 grams and a volume of 8 cubic centimeters. When the rock is cut in half, what is the volume and density of each piece? A) 8 cm 3 and 0.5 g/cm 3 B) 8 cm 3 and 1.0 g/cm 3 C) 4 cm 3 and 2.0 g/cm 3 D) 4 cm 3 and 4.0 g/cm Water has its greatest density at a temperature of A) 6º C B) 10º C C) 32º C D) 4º C 34. Water has the greatest density at A) 100ºC in the gaseous phase B) 0ºC in the solid phase C) 4ºC in the solid phase D) 4ºC in the liquid phase 35. The diagram below represents a rectangular object with a mass of 450 grams. What is the density of the object? 37. An empty 250-milliliter beaker has a mass of 60 grams. When 100 milliliters of oil is added to the beaker, the total mass is 140 grams. The density of the oil is approximately A) 1.7 g/ml B) 1.4 g/ml C) 0.8 g/ml D) 0.6 g/ml A) 1 gram per cubic centimeter B) 2 grams per cubic centimeter C) 3 grams per cubic centimeter D) 4 grams per cubic centimeter 36. The diagrams below represent two solid objects A and B. with different densities. What will happen when the objects are placed in a container of water (water temperature = 4 C)? A) Both objects will sink. B) Both objects will float. C) Object A will float and object B will sink. D) Object B will float and object A will sink.
9 38. Base your answer to the following question on the bedrock cross section below, which represents part of Earth's crust where natural gas, oil, and water have moved upward through a layer of folded sandstone and filled the pore spaces at the top of the sandstone layer. The final arrangement of the natural gas, oil, and water within the sandstone was caused by differences in their A) density B) specific heat C) relative age D) radioactive half-life 39. Base your answer to the following question on the two tables below and on your knowledge of Earth science. Table 1 shows the composition, hardness, and average density of four minerals often used as gemstones. Table 2 lists the minerals in Moh's Scale of Hardness from 1 (softest) to 10 (hardest). If the mass of a spinel crystal is 9.5 grams, what is the volume of this spinel crystal? A) 0.4 cm 3 B) 2.5 cm 3 C) 5.7 cm 3 D) 36.1 cm 3
10 Base your answers to questions 40 through 42 on the diagram and table below. 40. When a sample of the mineral calcite is heated. it expands, causing its density to be A) less than 2.7 g/cm 3 B) exactly 2.7 g/cm 3 C) between 2.7 and 3.0 g/cm 3 D) greater than 3.0 g/cm Under identical conditions, several samples of the mineral pyrite are measured, and their densities are compared. The values obtained should show that A) rounded samples are more dense than rough samples B) large samples are more dense than small samples C) small samples are more dense than large samples D) all the pyrite samples have the same density 42. A student measured the mass of a sample of quartz three times. The mass was the same the first and second times, but was less the third time. This decrease in mass could have occurred before the third measurement if the sample had been A) heated and expanded B) cooled and contracted C) soaked in water D) dropped and a piece was lost 43. A student calculates the densities of five different pieces of aluminum, each having a different volume. Which graph best represents this relationship? 44. A mineral sample is found to have a density of 3.0 grams per cubic centimeter. It is then broken into two pieces, with one piece twice as large as the other. The smaller of the two pieces will have a density of A) 1.0 g/cm 3 B) 1.5 g/cm 3 C) 3.0 g/cm 3 D) 6.0 g/cm 3
11 45. The data table below shows the mass and volume of four different minerals. Which mineral has the greatest density? A) A B) B C) C D) D
Topic 1 Review Questions Set 1
 Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
DENSITY of Solids and Liquids
 Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
Student Exploration: Mineral Identification
 Name: Date: Student Exploration: Identification Vocabulary: crystal, density, hardness, homogeneous, luster, mass, mineral, streak, volume Prior Knowledge Questions (Do these BEFORE using the Gizmo.) 1.
Name: Date: Student Exploration: Identification Vocabulary: crystal, density, hardness, homogeneous, luster, mass, mineral, streak, volume Prior Knowledge Questions (Do these BEFORE using the Gizmo.) 1.
CHAPTER 2 HW PRACTICE SOLUTIONS
 CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
7. The diagram below represents cross sections of equal-size beakers A, B, and C filled with beads.
 Base your answers to questions 1 and 2 on the diagram below and on your knowledge of Earth science. The diagram represents four tubes, labeled A, B, C, and D, each containing 150 ml of sediments. Tubes
Base your answers to questions 1 and 2 on the diagram below and on your knowledge of Earth science. The diagram represents four tubes, labeled A, B, C, and D, each containing 150 ml of sediments. Tubes
Copyright 2014 Science Doodles
 Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
LESSON hat is density? 24
 LESSON hat is density? 24 Rocks are heavy. A small box of rocks has a large. Yet a box of the same size containing corks has a small. How can objects that are the same size have different es? Scientists
LESSON hat is density? 24 Rocks are heavy. A small box of rocks has a large. Yet a box of the same size containing corks has a small. How can objects that are the same size have different es? Scientists
Density Answers to the End of module questions
 IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
Density. How tightly the atoms are packed together in an object
 Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
2 Identifying Minerals
 CHAPTER 1 2 Identifying Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What seven properties can be used to identify
CHAPTER 1 2 Identifying Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What seven properties can be used to identify
Experiment #3. Density
 Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
EARTH SCIENCE 11 CHAPTER 4 NOTES HOW TO KNOW THE MINERALS. Introduction. Background
 EARTH SCIENCE 11 CHAPTER 4 NOTES HOW TO KNOW THE MINERALS KEY Introduction Much of the Earth is covered with loose rock, soil, or sand. Beneath this loose material there is always unbroken solid rock which
EARTH SCIENCE 11 CHAPTER 4 NOTES HOW TO KNOW THE MINERALS KEY Introduction Much of the Earth is covered with loose rock, soil, or sand. Beneath this loose material there is always unbroken solid rock which
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
Directions: Complete the following prior to class. Be prepared to show your work on the board.
 Worksheet L4X Student s Name Directions: Complete the following prior to class. Be prepared to show your work on the board. 1) Convert 30 ml to oz, qt, L, gal. 2) Convert 16 oz to kg, lb, tons, g. Page
Worksheet L4X Student s Name Directions: Complete the following prior to class. Be prepared to show your work on the board. 1) Convert 30 ml to oz, qt, L, gal. 2) Convert 16 oz to kg, lb, tons, g. Page
Properties of Minerals
 What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
Experiment #3. Density and Specific Gravity.
 Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
ES 106 Laboratory # 1 PROPERTIES OF WATER
 ES 106 Laboratory # 1 PROPERTIES OF WATER 1-1 Introduction What are the physical and chemical properties of water that make it so unique and necessary for living things? When you look at water, taste and
ES 106 Laboratory # 1 PROPERTIES OF WATER 1-1 Introduction What are the physical and chemical properties of water that make it so unique and necessary for living things? When you look at water, taste and
Full file at
 Chapter 2 Test Bank Completion 1. A/an is a naturally occurring inorganic solid with a characteristic chemical composition and a crystalline structure. 2. A positively charged ion is a. 3. A/an is a planar
Chapter 2 Test Bank Completion 1. A/an is a naturally occurring inorganic solid with a characteristic chemical composition and a crystalline structure. 2. A positively charged ion is a. 3. A/an is a planar
A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition.
 A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition. Naturally Occurring Formed by processes that occur in the natural world Inorganic Not
A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition. Naturally Occurring Formed by processes that occur in the natural world Inorganic Not
What is a Mineral? A Mineral is: Pure Naturally occurring Crystalline Solid
 Mineral Properties What is a Mineral? A Mineral is: Pure Naturally occurring Crystalline Solid Mineral Properties Color Streak Luster Hardness Density Crystal Shape Cleavage Fracture Fluorescence Reaction
Mineral Properties What is a Mineral? A Mineral is: Pure Naturally occurring Crystalline Solid Mineral Properties Color Streak Luster Hardness Density Crystal Shape Cleavage Fracture Fluorescence Reaction
2.3 Density and Density Calculations
 2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
EARTH SCIENCE 11 CHAPTER 4 NOTES HOW TO KNOW THE MINERALS. Introduction. Background
 EARTH SCIENCE 11 CHAPTER 4 NOTES HOW TO KNOW THE MINERALS KEY Introduction Much of the Earth is covered with loose rock, soil, or sand. Beneath this loose material there is always unbroken solid rock which
EARTH SCIENCE 11 CHAPTER 4 NOTES HOW TO KNOW THE MINERALS KEY Introduction Much of the Earth is covered with loose rock, soil, or sand. Beneath this loose material there is always unbroken solid rock which
A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition.
 Minerals Objectives List the 5 traits of minerals Identify minerals Describe characteristics of minerals Know how minerals form Describe ways that we use minerals A mineral is a naturally occurring, inorganic
Minerals Objectives List the 5 traits of minerals Identify minerals Describe characteristics of minerals Know how minerals form Describe ways that we use minerals A mineral is a naturally occurring, inorganic
Item 36 has not been slated for public release in 2011.
 S Use the power-plant diagrams and information below to answer questions 35 37. Power Plants 35. Which describes a chemical change and a physical change that take place during the production of electricity
S Use the power-plant diagrams and information below to answer questions 35 37. Power Plants 35. Which describes a chemical change and a physical change that take place during the production of electricity
NOTES Minerals.notebook. May 03, ,000. Inorganic. Solid. chemical 3,000. Oxygen. Naturally. elements. Earth's. Crystalline. crust.
 Minerals Mineral Characteristics 1. occurring 2. 3. 4. structure 5. Definite composition Naturally Mineral Characteristics 1. occurring Inorganic 2. Solid 3. Crystalline 4. structure chemical 5. Definite
Minerals Mineral Characteristics 1. occurring 2. 3. 4. structure 5. Definite composition Naturally Mineral Characteristics 1. occurring Inorganic 2. Solid 3. Crystalline 4. structure chemical 5. Definite
page - Labs 09 & 10 - Mineral Identification
 Labs 09 & 10 - Mineral Identification page - All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the
Labs 09 & 10 - Mineral Identification page - All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the
NAME: Log onto YouTube and search for jocrisci channel.
 NAME: Log onto YouTube and search for jocrisci channel. ENERGY (Videos 7.1 & 7.2 ESRT 1e, 14c) 1. You must know the differences between the three ways energy is transferred: a. What is conduction? i. Give
NAME: Log onto YouTube and search for jocrisci channel. ENERGY (Videos 7.1 & 7.2 ESRT 1e, 14c) 1. You must know the differences between the three ways energy is transferred: a. What is conduction? i. Give
Unit 2 6 th Grade PHYSICAL PROPERTIES OF MATTER
 Unit 2 6 th Grade PHYSICAL PROPERTIES OF MATTER Elements can be classified as: Metals Good conductors of heat. Good conductors of electricity. Good reflectors of light when polished (shiny luster) Malleable
Unit 2 6 th Grade PHYSICAL PROPERTIES OF MATTER Elements can be classified as: Metals Good conductors of heat. Good conductors of electricity. Good reflectors of light when polished (shiny luster) Malleable
Minerals. Mohs Scale of Hardness
 Part I: Testing Mineral Properties You will perform the following tests on each mystery mineral as directed by your teacher. Be sure to record your observations in the appropriate row of the data table.
Part I: Testing Mineral Properties You will perform the following tests on each mystery mineral as directed by your teacher. Be sure to record your observations in the appropriate row of the data table.
3 The Formation, Mining, and Use of Minerals
 CHAPTER 1 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 1 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
MINERALS MR. WHITE ADVANCED CONTENT EARTH SCIENCE
 MINERALS MR. WHITE ADVANCED CONTENT EARTH SCIENCE WHO STUDIES THE EARTH S SURFACE & ITS CHANGING SHAPE? Geologists! GEOLOGISTS STUDY minerals rocks earthquakes volcanoes plate tectonics WHAT IS A MINERAL?
MINERALS MR. WHITE ADVANCED CONTENT EARTH SCIENCE WHO STUDIES THE EARTH S SURFACE & ITS CHANGING SHAPE? Geologists! GEOLOGISTS STUDY minerals rocks earthquakes volcanoes plate tectonics WHAT IS A MINERAL?
Identifying Minerals
 Identifying Minerals Beryl, feldspar, quartz Rock-forming Minerals Rock-forming minerals Common minerals that make up most of the rocks of Earth s crust Only a few dozen members Composed mainly of the
Identifying Minerals Beryl, feldspar, quartz Rock-forming Minerals Rock-forming minerals Common minerals that make up most of the rocks of Earth s crust Only a few dozen members Composed mainly of the
v = 5.00cm x 3.00cm x 2.00cm 2. What is the volume of a marble that has a mass of 3.0 g and a density of 2.7 g/ml? Formula (density) Substitution
 Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Earth s Crust. Atoms build Molecules build Minerals build. Rocks build. Lecture 3 - Mineralogy.
 Lecture 3 - Mineralogy http://www.soest.hawaii.edu/coasts/gg101/index.html Atoms build Molecules build Minerals build Rocks build Earth s Crust Common minerals that we mine and use. Mineral Name What It
Lecture 3 - Mineralogy http://www.soest.hawaii.edu/coasts/gg101/index.html Atoms build Molecules build Minerals build Rocks build Earth s Crust Common minerals that we mine and use. Mineral Name What It
COPY. Topic 6 MINERALS. Mr. Rocco
 COPY Topic 6 MINERALS Mr. Rocco Mineral? COPY Naturally occurring in the earth Solid Definite chemical composition Atoms arranged in a specific pattern Inorganic not from once living organisms (See ESRT
COPY Topic 6 MINERALS Mr. Rocco Mineral? COPY Naturally occurring in the earth Solid Definite chemical composition Atoms arranged in a specific pattern Inorganic not from once living organisms (See ESRT
Page 1. Name:
 Name: 7574-1 - Page 1 1) The diagram below shows the result of leaving an empty, dry clay flowerpot in a full container of water for a period of time. The water level in the container dropped to level
Name: 7574-1 - Page 1 1) The diagram below shows the result of leaving an empty, dry clay flowerpot in a full container of water for a period of time. The water level in the container dropped to level
The students will be able to: define porosity and relate it to the capacity of a common object's TEKS ADDRESSED:
 AUTHORS NAMES: TITLE OF THE LESSON: Porosity and Permeability TECHNOLOGY LESSON : No DATE OF LESSON: TIME LESSON: 2 (60 min class) NAME OF COURSE Biology SOURCE OF THE LESSON: Lynn Kirby TEKS ADDRESSED:
AUTHORS NAMES: TITLE OF THE LESSON: Porosity and Permeability TECHNOLOGY LESSON : No DATE OF LESSON: TIME LESSON: 2 (60 min class) NAME OF COURSE Biology SOURCE OF THE LESSON: Lynn Kirby TEKS ADDRESSED:
The Study of Minerals (Chapter 1) - Introduction to Mineral Physical Properties and Mineral Identification
 GEOLOGY 306 Laboratory NAME: Instructor: TERRY J. BOROUGHS Lab Section: The Study of Minerals (Chapter 1) - Introduction to Mineral Physical Properties and Mineral Identification For this assignment you
GEOLOGY 306 Laboratory NAME: Instructor: TERRY J. BOROUGHS Lab Section: The Study of Minerals (Chapter 1) - Introduction to Mineral Physical Properties and Mineral Identification For this assignment you
Density What is density? How do you measure density?
 Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Science 8. Unit 1. Booklet
 Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
page - 1 Lab 9 - Mineral Identification
 Lab 9 - Mineral Identification page - 1 All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the bulk
Lab 9 - Mineral Identification page - 1 All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the bulk
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use.
 This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
Geology Physical Geology. Box of Minerals
 Geology 1403- Physical Geology Box of Minerals Minerals 1-Talc Talc O Luster: pearly/silky O Hardness: 1 O Streak: white/gray O Fracture O Color: sea green, white, gray O Uses: Talcum power, paint, ceramics,
Geology 1403- Physical Geology Box of Minerals Minerals 1-Talc Talc O Luster: pearly/silky O Hardness: 1 O Streak: white/gray O Fracture O Color: sea green, white, gray O Uses: Talcum power, paint, ceramics,
Science 8 Chapter 1 Section 1
 Science 8 Chapter 1 Section 1 Distribution of Water (pp. 8-13) How much fresh water do we have? The vast majority of water on Earth, about 97 percent, is salt water Two thirds of that fresh water supply
Science 8 Chapter 1 Section 1 Distribution of Water (pp. 8-13) How much fresh water do we have? The vast majority of water on Earth, about 97 percent, is salt water Two thirds of that fresh water supply
Lab 0.4: Density and Thickness of Aluminum Foil
 Name Block Lab 0.4: Density and Thickness of Aluminum Foil Student will be able to: Correctly use measuring instruments with accuracy Correctly apply the principles of significant figures in measurements
Name Block Lab 0.4: Density and Thickness of Aluminum Foil Student will be able to: Correctly use measuring instruments with accuracy Correctly apply the principles of significant figures in measurements
Name Class Date. Does it have a crystalline structure? Minerals are crystals. Each mineral has a certain crystal structure that is always the same.
 CHAPTER 1 1 What Is a Mineral? SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What are minerals? What determines the shape
CHAPTER 1 1 What Is a Mineral? SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What are minerals? What determines the shape
What is water? TASK: Using the three worksheets investigate what is water.
 What is water? AGE RANGES: Key stage 2 TASK: Using the three worksheets investigate what is water. OBJECTIVES: Investigate how water behaves, rain is created and how the water cycle works. CURRICULUM FIT:
What is water? AGE RANGES: Key stage 2 TASK: Using the three worksheets investigate what is water. OBJECTIVES: Investigate how water behaves, rain is created and how the water cycle works. CURRICULUM FIT:
M. It is expressed in units such a g/cc,
 Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Determining the Density of Unknown Substances
 Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
CHM101 Lab - Energy Grading Rubric
 Name Team Name CHM101 Lab - Energy Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance Printed
Name Team Name CHM101 Lab - Energy Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance Printed
SIGNIFICANT FIGURES WORKSHEET
 SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
Measurement and Density - Experiment 1
 Measurement and Density - Experiment 1 Purpose: The purpose of this experiment is to acquaint you with some common metric units of length, volume, and mass. You will also measure the density of an unknown
Measurement and Density - Experiment 1 Purpose: The purpose of this experiment is to acquaint you with some common metric units of length, volume, and mass. You will also measure the density of an unknown
Density. The purpose of this experiment is to investigate the topic of density by determining the densities of some materials.
 Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
Make Five. A game about the chemical formulas of some common minerals
 Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
Classifying Minerals. Name: Date: Class: #: Materials: Magnifying lens (2) Copper Penny (2) Glass Plate Mohs Scale Set of minerals Mineral
 Name: Date: Class: #: Classifying Minerals Materials: Magnifying lens (2) Copper Penny (2) Glass Plate Mohs Scale Set of minerals Mineral Magnet Iron Nail Streak Plates (1 white & 1 black) Identification
Name: Date: Class: #: Classifying Minerals Materials: Magnifying lens (2) Copper Penny (2) Glass Plate Mohs Scale Set of minerals Mineral Magnet Iron Nail Streak Plates (1 white & 1 black) Identification
Oceanography Page 1 of 6 Lab: Ocean Salinity and Density M.Sewell rm #70
 Oceanography Page 1 of 6 Salty Water! Description: This lab is designed to demonstrate the formation of the world s oceans and why the oceans are salty, as well as the changes that take place in density
Oceanography Page 1 of 6 Salty Water! Description: This lab is designed to demonstrate the formation of the world s oceans and why the oceans are salty, as well as the changes that take place in density
STAAR Vocabulary noticing something about the world around you. using clues to find the answer. everything everywhere. to sort into groups
 Observation noticing something about the world around you Inference using clues to find the answer Matter everything everywhere Classify to sort into groups Physical Property something you observe with
Observation noticing something about the world around you Inference using clues to find the answer Matter everything everywhere Classify to sort into groups Physical Property something you observe with
Minnesota Comprehensive Assessments-Series III
 Name Minnesota Comprehensive Assessments-Series III Science Item Sampler Grade 8 ITEM SAMPLERS ARE NOT SECURE TEST MATERIALS. THIS ITEM SAMPLER TEST BOOK MAY BE COPIED OR 18 Point State of Minnesota Copyright
Name Minnesota Comprehensive Assessments-Series III Science Item Sampler Grade 8 ITEM SAMPLERS ARE NOT SECURE TEST MATERIALS. THIS ITEM SAMPLER TEST BOOK MAY BE COPIED OR 18 Point State of Minnesota Copyright
1.4 Density. Question Paper. Subject Physics (0625) Cambridge International Examinations (CIE) General Physics A* A B C D E U
 1.4 ensity Question Paper Level IGSE Subject Physics (0625) Exam oard ambridge International Examinations (IE) Topic General Physics Sub-Topic 1.4 ensity ooklet Question Paper Time llowed: 33 minutes Score:
1.4 ensity Question Paper Level IGSE Subject Physics (0625) Exam oard ambridge International Examinations (IE) Topic General Physics Sub-Topic 1.4 ensity ooklet Question Paper Time llowed: 33 minutes Score:
The Study of Minerals (Chapter 1) Introduction to Mineral Identification: THE NON - SILICATE MINERALS
 GEOLOGY 306 Laboratory NAME: Instructor: TERRY J. BOROUGHS The Study of Minerals (Chapter 1) Introduction to Mineral Identification: THE NON - SILICATE MINERALS For this assignment you will require: a
GEOLOGY 306 Laboratory NAME: Instructor: TERRY J. BOROUGHS The Study of Minerals (Chapter 1) Introduction to Mineral Identification: THE NON - SILICATE MINERALS For this assignment you will require: a
Creation Settings. can be separated into two or more pure substances by distillation. can be separated into two or more substances by chromatography.
 Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
NOTE: If substances have the SAME VOLUME, they can have DIFFERENT MASSES.
 Grade 8 SCIENCE Chapter 8 questions Page 306 1. When particles are CLOSER TOGETHER, DENSITY is GREATER. 2. Particles in SOLIDS have FEWER SPACES between them. The ATTRACTIVE FORCES between the particles
Grade 8 SCIENCE Chapter 8 questions Page 306 1. When particles are CLOSER TOGETHER, DENSITY is GREATER. 2. Particles in SOLIDS have FEWER SPACES between them. The ATTRACTIVE FORCES between the particles
Minerals Reading with Questions (Pg. 3-7) Properties of Common Minerals Reading (Pg. 8-9 ) Crossword Puzzle (Pg. 17) Flashcard Matching
 NAME: Kick Off Activity Classification of Minerals (Pg. 2) MUST DO ALL The Foundation Minerals Reading with Questions (Pg. 3-7) Properties of Common Minerals Reading (Pg. 8-9 ) OR Prop. of Common Minerals
NAME: Kick Off Activity Classification of Minerals (Pg. 2) MUST DO ALL The Foundation Minerals Reading with Questions (Pg. 3-7) Properties of Common Minerals Reading (Pg. 8-9 ) OR Prop. of Common Minerals
4THE UNIVERSITY OF THE STATE OF NEW YORK
 4THE UNIVERSITY OF THE STATE OF NEW YORK GRADE 4 ELEMENTARY-LEVEL SCIENCE TEST WRITTEN TEST SPRING 2009 Student Name School Name Print your name and the name of your school on the lines above. The test
4THE UNIVERSITY OF THE STATE OF NEW YORK GRADE 4 ELEMENTARY-LEVEL SCIENCE TEST WRITTEN TEST SPRING 2009 Student Name School Name Print your name and the name of your school on the lines above. The test
T. Trimpe Lesson 1: Length
 T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
Density Measurements Background
 Density Measurements Background The density (ρ) of a substance or an object is defined by its mass (m) divided by its volume (V):!"#$%&' =!"##!"#$%&!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! =!! The units used
Density Measurements Background The density (ρ) of a substance or an object is defined by its mass (m) divided by its volume (V):!"#$%&' =!"##!"#$%&!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! =!! The units used
Triple Beam Balance: add the three together: 700g + 20g + 2.9g = 722.9g Metric base unit for mass is gram.
 6 th Grade 2 nd Nine Week CSA Study Guide 2015-16 SOL 6.1b: Make precise and consistent measurements and estimations. Graduated Cylinder: read from the lowest part of the water line curve. Reading: 56ml
6 th Grade 2 nd Nine Week CSA Study Guide 2015-16 SOL 6.1b: Make precise and consistent measurements and estimations. Graduated Cylinder: read from the lowest part of the water line curve. Reading: 56ml
STUDENT NAME DATE ID PERIOD GRADE 5 SCIENCE
 STUDENT NAME DATE ID PERIOD GRADE 5 SCIENCE Administered October 2006 DIRECTIONS Read each question and choose the best answer. Be sure to mark all of your answers. SAMPLE A Objects That Conduct Heat
STUDENT NAME DATE ID PERIOD GRADE 5 SCIENCE Administered October 2006 DIRECTIONS Read each question and choose the best answer. Be sure to mark all of your answers. SAMPLE A Objects That Conduct Heat
1. Hardness 2. Streak 3. Density 4. Crystal structure 5. A mineral must be a naturally occurring, inorganic. 7. c 8. f 9. a 10. d 11. g 12. e 13.
 Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
COMPOSITION OF SEAWATER
 COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
Elements and Compounds
 Chemistry Name: Partners: Date: Elements and Compounds Introduction Most of the materials you have used so far in this course were mixtures. If you could take them apart physically, you would find that
Chemistry Name: Partners: Date: Elements and Compounds Introduction Most of the materials you have used so far in this course were mixtures. If you could take them apart physically, you would find that
The Study of Minerals (Chapter 1)
 GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
The Study of Minerals (Chapter 1)
 GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
Oceans are filled with??? The Water Molecule. Hydrogen Bonding between Polar Molecules: WATER!!!!! Let s understand water then.
 Oceans are filled with??? WATER!!!!! Let s understand water then. The Water Molecule V -shaped molecule: 105º angle between H nuclei Due to covalent bonds involving Oxygen s p-orbitals P-orbitals are 90º
Oceans are filled with??? WATER!!!!! Let s understand water then. The Water Molecule V -shaped molecule: 105º angle between H nuclei Due to covalent bonds involving Oxygen s p-orbitals P-orbitals are 90º
NATURE Sunday Academy What Parts of Computers are Mined
 2013-14 NATURE Sunday Academy What Parts of Computers are Mined Activity B: Ore Body Mystery In this activity students will explore the processes of core drilling and geological testing. Students will
2013-14 NATURE Sunday Academy What Parts of Computers are Mined Activity B: Ore Body Mystery In this activity students will explore the processes of core drilling and geological testing. Students will
The table below shows some of the properties of the elements cobalt and nickel.
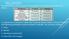 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Bulk Density Protocol
 Bulk Density Protocol Purpose To measure the bulk density of each horizon in a soil profile Overview In the field, students collect three soil samples from each horizon in a soil profile using a container
Bulk Density Protocol Purpose To measure the bulk density of each horizon in a soil profile Overview In the field, students collect three soil samples from each horizon in a soil profile using a container
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Do not write on the question paper, do not turn in the question paper. Put your answers on a new paper. Unless told otherwise.
 APES Unit 2 Packet Due Sept 10 at the beginning of class Instructions for all homework and labs: Put your name and date on top right corner of all your papers. Put the name of the problem or lab at the
APES Unit 2 Packet Due Sept 10 at the beginning of class Instructions for all homework and labs: Put your name and date on top right corner of all your papers. Put the name of the problem or lab at the
Minerals to know. Mineral Families. Calcite Fluorite Galena Kaolinite Halite Copper Talc Graphite Bornite Pyrite. Quartz. Mica. Gypsum.
 Minerals Minerals to know Calcite Fluorite Galena Kaolinite Halite Copper Talc Graphite Bornite Pyrite Mineral Families Mica Biotite Muscovite Lepidolite Gypsum Satin Spar Selenite Feldspar Pink Others
Minerals Minerals to know Calcite Fluorite Galena Kaolinite Halite Copper Talc Graphite Bornite Pyrite Mineral Families Mica Biotite Muscovite Lepidolite Gypsum Satin Spar Selenite Feldspar Pink Others
Where does the water go?
 Photo from sdherps.org. Post Visit Activity Grade Five Where does the water go? Summary Students will explore where water goes around our earth and atmosphere while learning how the sun s energy affects
Photo from sdherps.org. Post Visit Activity Grade Five Where does the water go? Summary Students will explore where water goes around our earth and atmosphere while learning how the sun s energy affects
PHYSICAL SCIENCE 0652/06
 www.xtremepapers.com Centre Number Candidate Number Name CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education PHYSICAL SCIENCE 0652/06 Paper 6 Alternative to Practical
www.xtremepapers.com Centre Number Candidate Number Name CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education PHYSICAL SCIENCE 0652/06 Paper 6 Alternative to Practical
1) An object weighs 75.7 kg. What is this weight in lbs? [1 lb = grams]
![1) An object weighs 75.7 kg. What is this weight in lbs? [1 lb = grams] 1) An object weighs 75.7 kg. What is this weight in lbs? [1 lb = grams]](/thumbs/89/98796796.jpg) CHEMISTRY 51 SPRING 2016 NAME: Exam 1 Multiple Choice (2 pts each) 1) An object weighs 75.7 kg. What is this weight in lbs? [1 lb = 453.6 grams] a) 34.3 pounds b) 167 pounds c) 343 pounds d) 16.7 pounds
CHEMISTRY 51 SPRING 2016 NAME: Exam 1 Multiple Choice (2 pts each) 1) An object weighs 75.7 kg. What is this weight in lbs? [1 lb = 453.6 grams] a) 34.3 pounds b) 167 pounds c) 343 pounds d) 16.7 pounds
NATURAL GAS Formation of a Clean Source of Energy
 NATURAL GAS Formation of a Clean Source of Energy LESSON SUMMARY This lesson explores how natural gas was formed by identifying states of matter, density, and how density factors into changes in states.
NATURAL GAS Formation of a Clean Source of Energy LESSON SUMMARY This lesson explores how natural gas was formed by identifying states of matter, density, and how density factors into changes in states.
Kinetic vs. Thermodynamic Control
 Experiment: Kinetic vs. Thermodynamic Control of rganic Reactions Kinetic vs. Thermodynamic Control During your study of reactions this year you have examined many mechanisms. You have used these mechanisms
Experiment: Kinetic vs. Thermodynamic Control of rganic Reactions Kinetic vs. Thermodynamic Control During your study of reactions this year you have examined many mechanisms. You have used these mechanisms
Thermal Conduction and Surface Area
 Chapter 16 Thermal Energy and Heat Investigation 16A Thermal Conduction and Surface Area Background Information The quantity of energy transferred by heat from a body depends on a number of physical properties
Chapter 16 Thermal Energy and Heat Investigation 16A Thermal Conduction and Surface Area Background Information The quantity of energy transferred by heat from a body depends on a number of physical properties
Water is a solid, liquid, & gas. 71% of earth s surface is water. Our body is two-thirds water. Fresh water water that is not salty and has little or
 Water is a solid, liquid, & gas. 71% of earth s surface is water. Our body is two-thirds water. Fresh water water that is not salty and has little or no taste, color, or smell. Salt water water that contains
Water is a solid, liquid, & gas. 71% of earth s surface is water. Our body is two-thirds water. Fresh water water that is not salty and has little or no taste, color, or smell. Salt water water that contains
6. In this temperature time graph for the heating of H 2O at a constant rate, the segment DE represents the
 1. Which of the following contains particles with the least freedom of motion? A) CO 2( ) B) HCl(aq) C) F 2(g) D) MgBr 2(s) E) C 6H 12O 6(aq) 2. During boiling, the temperature of a pure liquid substance
1. Which of the following contains particles with the least freedom of motion? A) CO 2( ) B) HCl(aq) C) F 2(g) D) MgBr 2(s) E) C 6H 12O 6(aq) 2. During boiling, the temperature of a pure liquid substance
UCS Mi-STAR Science 7 Semester 1 Midterm Exam Review Guide
 Name Date Hour 2018-2019 UCS Mi-STAR Science 7 Semester 1 Midterm Exam Review Guide Question How does cold air compare to warm air? Cold air weighs more than hot air. When a 9-centimeter balloon is filled
Name Date Hour 2018-2019 UCS Mi-STAR Science 7 Semester 1 Midterm Exam Review Guide Question How does cold air compare to warm air? Cold air weighs more than hot air. When a 9-centimeter balloon is filled
5 characteristics... minerals power point.notebook. September 20, Nov 17 3:48 PM. Nov 17 4:11 PM. Nov 18 9:36 AM
 Minerals! They're everywhere! What are they? Nov 17 3:48 PM To be a mineral you need to be in the special club. In order to be in the special club you need... 5 characteristics... Nov 17 4:11 PM 1. Naturally
Minerals! They're everywhere! What are they? Nov 17 3:48 PM To be a mineral you need to be in the special club. In order to be in the special club you need... 5 characteristics... Nov 17 4:11 PM 1. Naturally
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Oceans are filled with??? The Water Molecule. Water is Polar Molecule: WATER!!!!! Let s understand water then.
 Oceans are filled with??? WATER!!!!! Let s understand water then. The Water Molecule V -shaped: 105º angle between H Polar molecule: negative Oxygen and positive Hydrogen ends due to covalent bond Polar
Oceans are filled with??? WATER!!!!! Let s understand water then. The Water Molecule V -shaped: 105º angle between H Polar molecule: negative Oxygen and positive Hydrogen ends due to covalent bond Polar
16 1 Minerals and Mineral Resources
 Mining and Mineral Resources CHAPTER 16 1 Minerals and Mineral Resources 2 Mineral Exploration and Mining 3 Mining Regulations and Mine Reclamation READING WARM-UP Before you read this chapter, take a
Mining and Mineral Resources CHAPTER 16 1 Minerals and Mineral Resources 2 Mineral Exploration and Mining 3 Mining Regulations and Mine Reclamation READING WARM-UP Before you read this chapter, take a
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
GROUNDWATER. Narrative
 GROUNDWATER Narrative BIG IDEAS: Groundwater. is between the grains of sand and gravel.. moves because gravity works underground just like it does above ground.. is connected to surface water. 4. is part
GROUNDWATER Narrative BIG IDEAS: Groundwater. is between the grains of sand and gravel.. moves because gravity works underground just like it does above ground.. is connected to surface water. 4. is part
Name # Date Period! FINDING&VOLUME&AND&DENSITY& !!! !!!!!!!
 Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Why Study Minerals? blocks of Earth materials. to understanding rock formation
 Minerals I Elements of the Earth s crust Properties of minerals Crystal form; Cleavage/Fracture; Color, streak, luster; Hardness, tenacity; Specific gravity; Acid test, magnetism, etc. Why Study Minerals?
Minerals I Elements of the Earth s crust Properties of minerals Crystal form; Cleavage/Fracture; Color, streak, luster; Hardness, tenacity; Specific gravity; Acid test, magnetism, etc. Why Study Minerals?
