NOTE: If substances have the SAME VOLUME, they can have DIFFERENT MASSES.
|
|
|
- Vincent Arnold
- 5 years ago
- Views:
Transcription
1 Grade 8 SCIENCE Chapter 8 questions Page When particles are CLOSER TOGETHER, DENSITY is GREATER. 2. Particles in SOLIDS have FEWER SPACES between them. The ATTRACTIVE FORCES between the particles in a SOLID PREVENT the particles FROM COMING APART. For a LIQUID, an object moves through a FLUID pushing the particles apart because ATTRACTIVE FORCES between particles are WEAKER. 3. A CHANGE IN DENSITY of a substance ONLY OCCURS if a STATE CHANGE OCCURS. Water change its density will DECREASE if water is WARMED i.e. state changes from a LIQUID to WATER VAPOUR. 4. When every LIQUID has a GREATER DENSITY than a SOLID, the SOLID will FLOAT on it. The force of gravity upon one unit VOLUME of that liquid will be greater than the force of GRAVITY on the SOLID. GRAVITY will pull the MORE MASSIVE PARTICLES of the LIQUID to a LOWER LEVEL than the LESS MASSIVE PARTICLES of the SOLID. THIS PROCESS forces the SOLID to FLOAT on the LIQUID. NOTE: If substances have the SAME VOLUME, they can have DIFFERENT MASSES. PAGE 309 If VOLUME is the SAME and MASSES are DIFFERENT, then DENSITY MUST BE DIFFERENT. 1. GOLD is red, hot in the LIQUID STATE. At that temperature, PARTICLES are MOVING QUICKLY, ARE FAR APART, and are LESS ATTRACTED TO EACH OTHER than in the SOLID STATE. NOTE: A number of gold particles in LIQUID STATE occupies a LARGER VOLUME than the SAME number of gold particles in the SOLID STATE. LIQUID GOLD is SLIGHTLY LESS DENSE than SOLID GOLD. (Most substances are like this.) 2. Water particles are far apart and AIR particles are VERY FAR APART so the particles move more easily out of the way of a moving dolphin. 3. NOTE: Your table should be NEAT, well organized, and LABELLED. Items DENSER than water will SINK. 4. Warm air expands, become less dense and rise. Cool air contracts, become more dense and descend( drop) EMPTY SPACE has NO MATTER AT ALL. It is a COMPLETE VACUUM. AIR is mostly empty space but it does have air particles flying around in it. 7. The particles in water are CLOSER TOGETHER and MORE STRONGLY ATTRACTED TO EACH OTHER. It is HARDER to move through particles in a LIQUID like water.
2 PAGE 312 Using TABLE on page = 2.7 g/cm 3 The density of the metal is 2.7 g/cm 3 SO it is ALUMINUM. 3. = 1.26 g/ml The density of the metal is 1.26 g/ml SO it NOT water since WATER has a density of 1.0 g/ml. = g/ml The density of the gas is g/ml SO the gas is probably CARBON DIOXIDE. PAGE 313 Using table on = 0.50 cm 3 The VOLUME is 0.50 cm = ml 3 SO HELIUM would occupy 75 L. =243 ml The VOLUME of SEA WATER is 243 ml.
3 PAGE m = V x D = ( ml) x ( g/ml) = 28 g It s mass is 28 g. 2. m = V x D = (6.5 cm 3 ) x (8.92 g/cm 3 ) = g COPPER has a mass of 58 g. 3. m = V x D = (7.0 ml) x ( 0.90 g/ml) = 6.3 g MACHINE OIL has a MASS of 6.3 g. PAGE To find DENSITY, you need to measure both VOLUME and MASS of the substance. 2. MEASURE an OBJECT with an IRREGULAR SHAPE 3. To measure the VOLUME with an IRREGULAR SHAPE, put a volume of water into a graduated cylinder and measure the VOLUME ACCURATELY. Place small objects into the water, causing the water level to rise in the cylinder. Read the new volume level. THE DIFFERENCE between the FIRST VOLUME from the SECOND VOLUME LEVEL must be the volume of the irregular shape. DISPLACEMENT 4. If the mass of a PURE SUBSTANCE INCREASES, there must be MORE OF IT, THUS, the VOLUME must also INCREASE. PAGE GOLD is most dense. It is a SOLID at room temperature. 2. HYDROGEN is LEAST DENSE. It is a GAS at room temperature. 3. SOLIDS tend to be MOST DENSE. 4. GOLD is DENSER than MERCURY.
4 PAGE 321 ANALYZE ANSWERS 1. ANY substance with DENSITY LESS THAN 1.0 g/cm 3, will FLOAT on WATER. 2. ANY substance with DENSITY GREATER THAN 1.0 g/cm 3, will SINK in WATER. PAGE = 8.9 g/cm 3 The density of the metal is 8.9 g/cm 3 SO it is NICKEL. 2. = 2.7 g/cm 3 The density of the metal is 2.7 g/cm 3 ; it is ALUMINUM. 3. = 13.6 g/ml The density of the gas is 13.6g/mL SO the gas is probably MERCURY. 4. = 2.2 g/cm 3 The density is 2.6 g/cm 3 ; it is SALT 5. = 0.79 g/cm 3 The density of the gas is 0.79 g/cm 3 SO it is ETHYL ALCOHOL.
5 PAGE Density of a 2 cm 3 with mass 3.18 g: =1.59 g/cm 3 2. LEAD has a GREATER DENSITY. 3. SUBSTANCE MASS (g) VOLUME (cm 3 ) DENSITY (g/cm 3 ) MASS to VOLUME RATIO ALUMINUM SALT COPPER OAK SALT The DENSITY of WATER: 1.00 g/ml Regular soft drinks must have a greater density MORE than 1 and DIET POP must have a DENSITY of LESS than Density of a PURE SUBSTANCE is ALWAYS THE SAME (at the SAME TEMPERATURE and pressure). MASS of a PURE SUBSTANCE is DIRECTLY PROPORTIONAL to ITS VOLUME. Adding one more ml of liquid always adds precisely the SAME MASS. 6. The volume of a LIQUID is measured using a GRADUATED CYLINDER. The volume of any solid can be measured by submerging it into a liquid and measuring the displaced liquid. If the solid has a regular shape V = lwh GASES fill the container so VOLUME of a GAS is the VOLUME of the container. 7. The mass-to volume ratio of a substance is the DENSITY of the substance. 8. DISPLACEMENT of WATER can be used to measure the volume of any solid, liquid or gas, as long as the substance is NOT SOLUBLE in the water. The displaced water would have the same volume as the item displacing it.
6 9. COPPER is SOLID. Particles are very close together and strongly attracted to each other. The MERCURY atoms will NOT be able to FLOW between the copper particles. The mercury will eventually diffuse into the copper; silver copper/mercury amalgam is created which takes a very long time to happen. Chapter Review Page Density is the RATIO of MASS to UNIT VOLUME. 2. The MORE CLOSELY the particles of a pure substance are packed, the MORE DENSE the substance. The MORE MASSIVE each particles, the MORE DENSE it will be. 3. The particles are packed much more closely in a LIQUID WATER than they are in WATER VAPOUR. (At room temperature water in liquid form is 2000 times denser.) 4. A lump of solid can push aside the particles in the liquid or gas, so the solid lump can move through the liquid. 5. An example where a LIQUID state is DENSER than a solid: Liquid water is denser than solid wax 6. The volume of an irregular shaped object can be measured ONLY by DISPLACEMENT OF WATER because you can never take all the measurements you would need to calculate the volume with a formula. 7. MASS is the TOTAL QUANTITY OF MATTER in an object. DENSITY is the AMOUNT OF MASS in a GIVEN VOLUME of a substance. 8. For density of a substance to remain constant, TEMPERATURE and PRESSURE must REMAIN THE SAME. 9. A draft near a window: As warm air near a window cools down, particles move less getting closer together. They get MORE DENSE causing a draft of cold air near the window.
7 10. A) material 1 has the largest mass 50 cm 3 has a mas of 570 g. B) Material 3 takes up the MOST SPACE. 100g occupies 40 cm 3. C) Material 1 D= 11 g/cm Material 2 D= 8.0 g/cm 3 Material 3 D= 2.6 g/cm 3 Mass VS VOLUME MASS g VOLUME cm 3 B) mass to volume ratio for each measurement is g/cm 3. C) Mystery substance from Table 8.1 on page 312 is WATER. D) The line for the HIGHER DENSITY will lie ABOVE and TO THE LEFT of WATER. LOWER DENSITY line will lie BELOW and to the RIGHT of water. WATER: ICE: 12. Four methods all use a GRADUALTED CYLINDER as a measuring instrument. Pour into graduated cylinder to measure VOLUME. HALF FILL graduated cylinder.add ice cubes to water and measure the DIFFERENCE in VOLUME of WATER (the displacement) FILL water bottles to brim with water. Pour into graduated cylinder to measure its volume. Put water vapour Into a contain of known volume, like the water bottle above.
8 13. DENSITY of GOLD is GREATER than the DENSITY of FEATHERS. GOLD particles must be more densely packed than the feathers. 1 kg gold will occupy LESS space than 1 kg of feathers. 14. Density and viscosity are NOT directly related. They VARY INDEPENDENTLY of each other. Ex. Density of oil is LESS than density of WATER OILS viscosity is GREATER than viscosity of water. Density of molasses is more than density of water. Viscosity of molasses is GREATER than the viscosity of water Density depends on MASS and SIZE of each particle. VISCOSITY depends on STICKINESS of the particles and their ability to interfere or tangle with the motion of other particles.
D =? D = M = 10 g = 5 g/cm 3 M = 10 g V 2 cm 3 V = 2 cm 3 5 g/cm 3
 Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Science 8. Unit 1. Booklet
 Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
Density What is density? How do you measure density?
 Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Name # Date Period! FINDING&VOLUME&AND&DENSITY& !!! !!!!!!!
 Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Density. How tightly the atoms are packed together in an object
 Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
Experiment #3. Density
 Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
Density Answers to the End of module questions
 IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
Experiment #3. Density and Specific Gravity.
 Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
INTERACTIVE SCIENCE 1B
 INTERACTIVE SCIENCE 1B Workbook Solutions (Enrichment Edition) Chapter 6 MATTER as PARTICLES Part A Sectional Exercise 6.1 Fill in the blanks p.54 1. (a) liquid (b) gas 2. (a) Melting (b) unchanged 3.
INTERACTIVE SCIENCE 1B Workbook Solutions (Enrichment Edition) Chapter 6 MATTER as PARTICLES Part A Sectional Exercise 6.1 Fill in the blanks p.54 1. (a) liquid (b) gas 2. (a) Melting (b) unchanged 3.
v = 5.00cm x 3.00cm x 2.00cm 2. What is the volume of a marble that has a mass of 3.0 g and a density of 2.7 g/ml? Formula (density) Substitution
 Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 2 HW PRACTICE SOLUTIONS
 CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
Grade 8 SCIENCE Unit 3 FLUIDS Chapter 7 QUESTIONS
 Grade 8 SCIENCE Unit 3 FLUIDS Chapter 7 QUESTIONS Page 272 1. A FLUID is a substance that can be made to FLOW. If a material, FLOWS, it is a FLUID. If you can STIR it, it is behaving like a FLUID. 2. Particle
Grade 8 SCIENCE Unit 3 FLUIDS Chapter 7 QUESTIONS Page 272 1. A FLUID is a substance that can be made to FLOW. If a material, FLOWS, it is a FLUID. If you can STIR it, it is behaving like a FLUID. 2. Particle
Directions: Complete the following prior to class. Be prepared to show your work on the board.
 Worksheet L4X Student s Name Directions: Complete the following prior to class. Be prepared to show your work on the board. 1) Convert 30 ml to oz, qt, L, gal. 2) Convert 16 oz to kg, lb, tons, g. Page
Worksheet L4X Student s Name Directions: Complete the following prior to class. Be prepared to show your work on the board. 1) Convert 30 ml to oz, qt, L, gal. 2) Convert 16 oz to kg, lb, tons, g. Page
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
Copyright 2014 Science Doodles
 Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Topic 1 Review Questions Set 1
 Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
LESSON hat is density? 24
 LESSON hat is density? 24 Rocks are heavy. A small box of rocks has a large. Yet a box of the same size containing corks has a small. How can objects that are the same size have different es? Scientists
LESSON hat is density? 24 Rocks are heavy. A small box of rocks has a large. Yet a box of the same size containing corks has a small. How can objects that are the same size have different es? Scientists
NATURAL GAS Formation of a Clean Source of Energy
 NATURAL GAS Formation of a Clean Source of Energy LESSON SUMMARY This lesson explores how natural gas was formed by identifying states of matter, density, and how density factors into changes in states.
NATURAL GAS Formation of a Clean Source of Energy LESSON SUMMARY This lesson explores how natural gas was formed by identifying states of matter, density, and how density factors into changes in states.
Determining the Density of Unknown Substances
 Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
Physical Properties of Materials
 Physical Properties of Materials Manufacturing Materials, IE251 Dr M. Saleh King Saud University Manufacturing materials --- IE251 lect-7, Slide 1 PHYSICAL PROPERTIES OF MATERIALS 1. Volumetric and Melting
Physical Properties of Materials Manufacturing Materials, IE251 Dr M. Saleh King Saud University Manufacturing materials --- IE251 lect-7, Slide 1 PHYSICAL PROPERTIES OF MATERIALS 1. Volumetric and Melting
SIGNIFICANT FIGURES WORKSHEET
 SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
Station 1 Dry Ice Experiments / Producing CO 2 in a reaction
 Station 1 Dry Ice Experiments / Producing CO 2 in a reaction 1. WHAT IS DRY ICE Put a flat chunk of dry ice on the counter and see how easy it "floats" on top of the counter when you give it a small push.
Station 1 Dry Ice Experiments / Producing CO 2 in a reaction 1. WHAT IS DRY ICE Put a flat chunk of dry ice on the counter and see how easy it "floats" on top of the counter when you give it a small push.
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
DENSITY of Solids and Liquids
 Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
ES 106 Laboratory # 1 PROPERTIES OF WATER
 ES 106 Laboratory # 1 PROPERTIES OF WATER 1-1 Introduction What are the physical and chemical properties of water that make it so unique and necessary for living things? When you look at water, taste and
ES 106 Laboratory # 1 PROPERTIES OF WATER 1-1 Introduction What are the physical and chemical properties of water that make it so unique and necessary for living things? When you look at water, taste and
Chapter 5 Water & Seawater. Chapter 5 Water & Seawater
 Chapter 5 Water & Seawater Chapter 5 Water & Seawater Chapter Overview Water has many unique thermal and dissolving properties. Seawater is mostly water molecules but has dissolved substances. Ocean water
Chapter 5 Water & Seawater Chapter 5 Water & Seawater Chapter Overview Water has many unique thermal and dissolving properties. Seawater is mostly water molecules but has dissolved substances. Ocean water
Creation Settings. can be separated into two or more pure substances by distillation. can be separated into two or more substances by chromatography.
 Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use.
 This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
(b) Fertilizers help to increase agricultural production. chemical property physical property
 Score 1. [Chang7 1.P.011.] Do the following statements describe chemical or physical properties? (a) Oxygen gas supports combustion. chemical property physical property (b) Fertilizers help to increase
Score 1. [Chang7 1.P.011.] Do the following statements describe chemical or physical properties? (a) Oxygen gas supports combustion. chemical property physical property (b) Fertilizers help to increase
COMPOSITION OF SEAWATER
 COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
2.3 Density and Density Calculations
 2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
Quest Chapter 18. If you don t already know, check out the materials online, or read #4.
 1 (part 1 of 2) In everyday use, the word dense is often used interchangeably with the word hard. In physics, density and hardness have completely different meanings. Which object is the densest? 1. lead
1 (part 1 of 2) In everyday use, the word dense is often used interchangeably with the word hard. In physics, density and hardness have completely different meanings. Which object is the densest? 1. lead
What are the 3 ways in which energy can travel? Explain what. conduction is. Does conduction happen best in solids, liquids or gases?
 What are the 3 ways in which energy can travel? Explain what conduction is What type of materials are good conductors and what type of materials are bad conductors (good insulators)? Does conduction happen
What are the 3 ways in which energy can travel? Explain what conduction is What type of materials are good conductors and what type of materials are bad conductors (good insulators)? Does conduction happen
Homework for Unit Vocab for Unit 6; due: 2. Pg 333 (1-5), Pg 335 (1-4), Pg 337 (1-4), Pg 339 (1-6), No sentences; due:
 Unit 6 Heat Homework for Unit 6 1. Vocab for Unit 6; due: 2. Pg 333 (1-5), Pg 335 (1-4), Pg 337 (1-4), Pg 339 (1-6), No sentences; due: 3. Temperature change activity; due: 4. Heat Transfer Homework; due:
Unit 6 Heat Homework for Unit 6 1. Vocab for Unit 6; due: 2. Pg 333 (1-5), Pg 335 (1-4), Pg 337 (1-4), Pg 339 (1-6), No sentences; due: 3. Temperature change activity; due: 4. Heat Transfer Homework; due:
Introduction to Density
 Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Iÿtÿÿeÿ/ÿrh Jÿtcket. Tÿlchÿwÿeÿ
 o 0 Iÿtÿÿeÿ/ÿrh Jÿtcket Tÿlchÿwÿeÿ Elements, Compounds, and Mixtures Classify each of the pictures below by placing the correct label in the blanks below: A= Element D-- Mixture of compounds B= Compound
o 0 Iÿtÿÿeÿ/ÿrh Jÿtcket Tÿlchÿwÿeÿ Elements, Compounds, and Mixtures Classify each of the pictures below by placing the correct label in the blanks below: A= Element D-- Mixture of compounds B= Compound
Skills in Science. Lab equipment. (Always draw 2D) Drawings below are NOT to scale. Beaker - A general purpose container with a pouring lip.
 Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
The table below shows some of the properties of the elements cobalt and nickel.
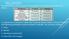 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Chemistry Quiz #1 Review
 Name: Chemistry Quiz 1 Review Page 1 of 6 Date: Please check your answers at http://leetz.weebly.com Chemistry Quiz #1 Review Part A: Density Calculations 1. The density of sodium is 0.97 g/cm 3. A sample
Name: Chemistry Quiz 1 Review Page 1 of 6 Date: Please check your answers at http://leetz.weebly.com Chemistry Quiz #1 Review Part A: Density Calculations 1. The density of sodium is 0.97 g/cm 3. A sample
Introduction to Thermal Energy
 Introduction to Thermal Energy Mar 10 7:33 AM 1 Thermal Energy vs. Temperature vs. Heat Mar 10 7:33 AM 2 Mar 10 12:53 PM 3 2 nd Law of Thermodynamics Thermal Equilibrium thermal energy is always transferred
Introduction to Thermal Energy Mar 10 7:33 AM 1 Thermal Energy vs. Temperature vs. Heat Mar 10 7:33 AM 2 Mar 10 12:53 PM 3 2 nd Law of Thermodynamics Thermal Equilibrium thermal energy is always transferred
Calculating Ballast By Troy Averill
 Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
Delaware Science Assessment Prototype: Grade 8 Integrative Item Cluster
 Student Version Delaware Science Assessment Prototype: Grade 8 Integrative Item Cluster Prepared for the Delaware Department of Education by WestEd Hot Pack Investigation #1 A student investigated how
Student Version Delaware Science Assessment Prototype: Grade 8 Integrative Item Cluster Prepared for the Delaware Department of Education by WestEd Hot Pack Investigation #1 A student investigated how
CHAPTER 5 Water and Seawater
 1 2 3 4 5 6 7 8 9 10 11 12 13 CHAPTER 5 Water and Seawater Chapter Overview Water has many unique thermal and dissolving properties. Seawater is mostly water molecules but has dissolved substances. Ocean
1 2 3 4 5 6 7 8 9 10 11 12 13 CHAPTER 5 Water and Seawater Chapter Overview Water has many unique thermal and dissolving properties. Seawater is mostly water molecules but has dissolved substances. Ocean
Density. The purpose of this experiment is to investigate the topic of density by determining the densities of some materials.
 Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
IGCSE(A*-G) Edexcel - Chemistry
 IGCSE(A*-G) Edexcel - Chemistry Principles of Chemistry States of Matter NOTES 1.1 Understand the arrangement, movement and energy of the particles in each of the three states of matter: solid, liquid
IGCSE(A*-G) Edexcel - Chemistry Principles of Chemistry States of Matter NOTES 1.1 Understand the arrangement, movement and energy of the particles in each of the three states of matter: solid, liquid
Chapters 1&2 Practice Problems
 Name Chapters 1&2 Practice Problems Multiple Choice No Calculators Allowed! 1. Which physical state of matter exhibits the greatest change in volume with changes in temperature or pressure? a) solid b)
Name Chapters 1&2 Practice Problems Multiple Choice No Calculators Allowed! 1. Which physical state of matter exhibits the greatest change in volume with changes in temperature or pressure? a) solid b)
Experiment: Measurements
 Experiment: Measurements I. INTRODUCTION Measurements are essential to experimental sciences such as chemistry, physics, biology, and geology. The measurements are usually made using the metric system
Experiment: Measurements I. INTRODUCTION Measurements are essential to experimental sciences such as chemistry, physics, biology, and geology. The measurements are usually made using the metric system
Seawater TA Initials: for finished Activity. 1 & 2 Or lose 10% of credit!
 Name: Section/ TA: Seawater TA Initials: for finished Activity. 1 & 2 Or lose 10% of credit! Seawater is an unusual substance. It is pure water mixed with various salts, trace elements, and gases. The
Name: Section/ TA: Seawater TA Initials: for finished Activity. 1 & 2 Or lose 10% of credit! Seawater is an unusual substance. It is pure water mixed with various salts, trace elements, and gases. The
SOLIDS: Mass, Volume and Density Measurements
 CHEM 1411 Lab Solids: Mass, Volume, and Density 23 SOLIDS: Mass, Volume and Density Measurements A. Overview Review Sections 1.4-1.6 in your textbook (Chemistry: The Central Science, 9th Ed., Brown, LeMay,
CHEM 1411 Lab Solids: Mass, Volume, and Density 23 SOLIDS: Mass, Volume and Density Measurements A. Overview Review Sections 1.4-1.6 in your textbook (Chemistry: The Central Science, 9th Ed., Brown, LeMay,
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
A. Description of the solid state according to the kinetic-molecular theory (KMT):
 A. Description of the solid state according to the kinetic-molecular theory (KMT): Have a definite shape and volume and a slow average kinetic energy Particles of a SOLID appear to vibrate around fixed
A. Description of the solid state according to the kinetic-molecular theory (KMT): Have a definite shape and volume and a slow average kinetic energy Particles of a SOLID appear to vibrate around fixed
Ohio s Learning Standards Grade 1 Science: Earth and Space Science - Sun, Energy and Weather The physical properties of water change.
 TEACHER GUIDE WATER: H 2 O IS AWESOME! 60 Minute Physical Science Lesson Science-to-Go! Program Grades: 1-3 Water: H 2 O is Awesome! Description Studying science takes on a Splash Factor as we introduce
TEACHER GUIDE WATER: H 2 O IS AWESOME! 60 Minute Physical Science Lesson Science-to-Go! Program Grades: 1-3 Water: H 2 O is Awesome! Description Studying science takes on a Splash Factor as we introduce
Objective: Classify metals, nonmetals, and metalloids based on their properties.
 Do Now Date: September 2, 2014 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
Do Now Date: September 2, 2014 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
1. Which of the following elements has the highest percentage by mass in nature? A. Oxygen B. Aluminium C. Nitrogen D. Silicon
 Class: F.3 ( ) Baptist Lui Ming Choi Secondary School First Term Examination (2013-2014) Date: 6 / 12 / 2013 Name: Form 3 Chemistry Time: 10:20-11:05 a.m. Answer ALL the questions. For Section A, choose
Class: F.3 ( ) Baptist Lui Ming Choi Secondary School First Term Examination (2013-2014) Date: 6 / 12 / 2013 Name: Form 3 Chemistry Time: 10:20-11:05 a.m. Answer ALL the questions. For Section A, choose
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter.
 1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
EMG 807 Post-test. 1. Workers from the electric company regularly read the meters on houses. These meters measure the amount of
 EMG 807 Post-test 1. Workers from the electric company regularly read the meters on houses. These meters measure the amount of a. Energy that we use. b. Power that we use. c. Force that we use. a. i only
EMG 807 Post-test 1. Workers from the electric company regularly read the meters on houses. These meters measure the amount of a. Energy that we use. b. Power that we use. c. Force that we use. a. i only
TEST I REVIEW. 1. All of the following are properties of antimony. Which one is not a
 TEST I REVIEW I Identify the letter of the choice that best completes the statement or answers the question. 1. All of the following are properties of antimony. Which one is not a physical property? a.
TEST I REVIEW I Identify the letter of the choice that best completes the statement or answers the question. 1. All of the following are properties of antimony. Which one is not a physical property? a.
Solids, liquids and gases
 Solids, liquids and gases Everything is made up of tiny particles called atoms. Normally, atoms join together to form groups called molecules. Molecules are always moving, even in things that look like
Solids, liquids and gases Everything is made up of tiny particles called atoms. Normally, atoms join together to form groups called molecules. Molecules are always moving, even in things that look like
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
Classifying Matter. Do they look different? Do they feel different? Do they behave the same way when they are dropped?
 Classifying Matter Standard: 5 PS1 3 Make observations and measurements to identify materials based on their properties. [Clarification Statement: Examples of materials to be identified could include baking
Classifying Matter Standard: 5 PS1 3 Make observations and measurements to identify materials based on their properties. [Clarification Statement: Examples of materials to be identified could include baking
This paper will consist of questions from Biology, Chemistry and Physics. Time allowed 1 hour.
 TRURO SCHOOL 13+ SCIENCE SAMPLE This paper will consist of questions from Biology, Chemistry and Physics Time allowed 1 hour. When told to do so, please turn over to commence the Biology section and then
TRURO SCHOOL 13+ SCIENCE SAMPLE This paper will consist of questions from Biology, Chemistry and Physics Time allowed 1 hour. When told to do so, please turn over to commence the Biology section and then
M. It is expressed in units such a g/cc,
 Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Chapter 3 - Energy and Matter Practice Problems
 Chapter 3 - Energy and Matter Practice Problems 1) Helium is a(n) A) compound. B) heterogeneous mixture. C) element. D) homogeneous mixture. E) electron. 2) Air is a(n) A) compound. B) heterogeneous mixture.
Chapter 3 - Energy and Matter Practice Problems 1) Helium is a(n) A) compound. B) heterogeneous mixture. C) element. D) homogeneous mixture. E) electron. 2) Air is a(n) A) compound. B) heterogeneous mixture.
Objective: Classify metals, nonmetals, and metalloids based on their properties.
 Do Now Date: September 6, 2016 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday, September
Do Now Date: September 6, 2016 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday, September
Chapter Overview. Water molecule. Atomic Structure. Hydrogen Bonding. Hydrogen Bonding. CHAPTER 5 Water and Seawater
 Chapter Overview CHAPTER 5 Water and Seawater Water has many unique thermal and dissolving properties. Seawater is mostly water molecules but has dissolved substances. Ocean is layered by salinity and
Chapter Overview CHAPTER 5 Water and Seawater Water has many unique thermal and dissolving properties. Seawater is mostly water molecules but has dissolved substances. Ocean is layered by salinity and
Oxygen Formula: O 2 Bonding: covalent Appearance: colourless gas. Oxygen is one of the two main gases in our atmosphere, the other being nitrogen.
 Composition of the air Air is a mixture of gases. The approximate amount if each gas in dry air is shown in the pie chart (right), but you should be aware that air also contains a variable amount of water
Composition of the air Air is a mixture of gases. The approximate amount if each gas in dry air is shown in the pie chart (right), but you should be aware that air also contains a variable amount of water
Look at the measuring cylinders. What happened to the volume of the water and the wax after freezing? the volume of water... the volume of wax...
 1. Meera poured 7 cm 3 of water into a measuring cylinder. She poured 7 cm 3 of melted wax into another measuring cylinder. She put both measuring cylinders into a freezer for 24 hours. water before freezing
1. Meera poured 7 cm 3 of water into a measuring cylinder. She poured 7 cm 3 of melted wax into another measuring cylinder. She put both measuring cylinders into a freezer for 24 hours. water before freezing
CHM101 Lab - Energy Grading Rubric
 Name Team Name CHM101 Lab - Energy Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance Printed
Name Team Name CHM101 Lab - Energy Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance Printed
, as well as some other materials. Use boiling point elevation to determine the percentages of each by mass.
 Remarks: - Use appropriate units. Note: SI units are always acceptable. - If you take apart the test, write your team number on every page in the space provided. - Sometimes, the answers won t make sense.
Remarks: - Use appropriate units. Note: SI units are always acceptable. - If you take apart the test, write your team number on every page in the space provided. - Sometimes, the answers won t make sense.
Science 8 Chapter 1 Section 1
 Science 8 Chapter 1 Section 1 Distribution of Water (pp. 8-13) How much fresh water do we have? The vast majority of water on Earth, about 97 percent, is salt water Two thirds of that fresh water supply
Science 8 Chapter 1 Section 1 Distribution of Water (pp. 8-13) How much fresh water do we have? The vast majority of water on Earth, about 97 percent, is salt water Two thirds of that fresh water supply
LAB 9A: TEMPERATURE AND HEAT, PART A
 Name: Period: LAB 9A: TEMPERATURE AND HEAT, PART A Hot and cold are familiar sensations. What happens when something hot comes in contact with something cold? Think about putting some ice cubes in a drink.
Name: Period: LAB 9A: TEMPERATURE AND HEAT, PART A Hot and cold are familiar sensations. What happens when something hot comes in contact with something cold? Think about putting some ice cubes in a drink.
HEAT and HEAT CAPACITY
 Grade VIII Sub:Physics HEAT and HEAT CAPACITY I. Tick ( ) the most appropriate answer. 1. The coolant used in car radiators is: (a) alcohol (b) benzene (c) water (d) petrol 2. During a sea breeze there
Grade VIII Sub:Physics HEAT and HEAT CAPACITY I. Tick ( ) the most appropriate answer. 1. The coolant used in car radiators is: (a) alcohol (b) benzene (c) water (d) petrol 2. During a sea breeze there
Lab Section. Experiment 2 Density. Introduction
 Experiment 2 Density Introduction Density (given the symbol d or ρ depending upon which book you read) is an intrinsic property of materials. The term intrinsic means that it is independent upon the amount
Experiment 2 Density Introduction Density (given the symbol d or ρ depending upon which book you read) is an intrinsic property of materials. The term intrinsic means that it is independent upon the amount
(3) The compound boron nitride (BN) has a high melting point (2967 ºC), high density, and is very hard. What is the best classification of this solid?
 Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
DETERMINATION OF DENSITY
 DETERMINATION OF DENSITY Dr. Barbara B. Bunn. Adapted from Experiments in General Chemistry ; Wiley 1996J.G. Wardeska, T.T-S. Huang, R.W. Kopp; used by permission. Tested by Ms. Janice Orr s Chemistry
DETERMINATION OF DENSITY Dr. Barbara B. Bunn. Adapted from Experiments in General Chemistry ; Wiley 1996J.G. Wardeska, T.T-S. Huang, R.W. Kopp; used by permission. Tested by Ms. Janice Orr s Chemistry
Objective: Classify metals, nonmetals, and metalloids based on their properties.
 Do Now Date: September 8, 2015 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
Do Now Date: September 8, 2015 Objective: Classify metals, nonmetals, and metalloids based on their properties. Draw a table listing the properties of metals, nonmetals, and metalloids. Tuesday September
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
1. Marie mixed 5 g of carbon with 5 g of lead oxide. She heated the mixture strongly for 15 minutes in a fume cupboard.
 1. Marie mixed 5 g of carbon with 5 g of lead oxide. She heated the mixture strongly for 15 minutes in a fume cupboard. After 15 minutes, Marie found some shiny beads in the mixture. a. (i) Marie collected
1. Marie mixed 5 g of carbon with 5 g of lead oxide. She heated the mixture strongly for 15 minutes in a fume cupboard. After 15 minutes, Marie found some shiny beads in the mixture. a. (i) Marie collected
Crystal Growth and Buoyancy- Driven Convection Currents
 Crystal Growth and Buoyancy- Driven Convection Currents Objective: To observe buoyancy-driven covection currents that are created as crystals grow in a crystal growing solution. Science Standards: Science
Crystal Growth and Buoyancy- Driven Convection Currents Objective: To observe buoyancy-driven covection currents that are created as crystals grow in a crystal growing solution. Science Standards: Science
*20GSD2101* Double Award Science: Chemistry. Unit C1 Foundation Tier THURSDAY 14 MAY 2015, MORNING [GSD21] *GSD21* TIME 1 hour.
![*20GSD2101* Double Award Science: Chemistry. Unit C1 Foundation Tier THURSDAY 14 MAY 2015, MORNING [GSD21] *GSD21* TIME 1 hour. *20GSD2101* Double Award Science: Chemistry. Unit C1 Foundation Tier THURSDAY 14 MAY 2015, MORNING [GSD21] *GSD21* TIME 1 hour.](/thumbs/81/82623486.jpg) Centre Number Candidate Number General Certificate of Secondary Education 2014 2015 Double Award Science: Chemistry Unit C1 Foundation Tier [GSD21] *GSD21* *G5802* *GSD21* THURSDAY 14 MAY 2015, MORNING
Centre Number Candidate Number General Certificate of Secondary Education 2014 2015 Double Award Science: Chemistry Unit C1 Foundation Tier [GSD21] *GSD21* *G5802* *GSD21* THURSDAY 14 MAY 2015, MORNING
HEAT Earth's original source of energy is our. The sun emits electro magnetic radiation in the form of rays, rays and light.
 HEAT Earth's original source of energy is our. The sun emits electro magnetic radiation in the form of rays, rays and light. of the energy from the sun is reflected directly back into space while is absorbed
HEAT Earth's original source of energy is our. The sun emits electro magnetic radiation in the form of rays, rays and light. of the energy from the sun is reflected directly back into space while is absorbed
Minnesota Comprehensive Assessments-Series III
 Name Minnesota Comprehensive Assessments-Series III Science Item Sampler Grade 5 ITEM SAMPLERS ARE NOT SECURE TEST MATERIALS. THIS ITEM SAMPLER TEST BOOK MAY BE COPIED OR DUPLICATED. 24 Point State of
Name Minnesota Comprehensive Assessments-Series III Science Item Sampler Grade 5 ITEM SAMPLERS ARE NOT SECURE TEST MATERIALS. THIS ITEM SAMPLER TEST BOOK MAY BE COPIED OR DUPLICATED. 24 Point State of
Lab Report: Thermal Energy Transfer in Mixtures
 Lab Report: Thermal Energy Transfer in Mixtures Purpose: The purpose of this experiment is to discover how exactly the final temperature of a mixture, involving a substance and hot water, is affected and
Lab Report: Thermal Energy Transfer in Mixtures Purpose: The purpose of this experiment is to discover how exactly the final temperature of a mixture, involving a substance and hot water, is affected and
Specific Heat Capacity. mass of substance Amount of Heat that goes into (or out of) a substance Specific Heat Capacity. Change in Temperature
 Specific Heat Capacity mass of substance Amount of Heat that goes into (or out of) a substance Specific Heat Capacity Change in Temperature Ice Water Diet Idea: Drink lots of Ice water, your body must
Specific Heat Capacity mass of substance Amount of Heat that goes into (or out of) a substance Specific Heat Capacity Change in Temperature Ice Water Diet Idea: Drink lots of Ice water, your body must
Density Measurements Background
 Density Measurements Background The density (ρ) of a substance or an object is defined by its mass (m) divided by its volume (V):!"#$%&' =!"##!"#$%&!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! =!! The units used
Density Measurements Background The density (ρ) of a substance or an object is defined by its mass (m) divided by its volume (V):!"#$%&' =!"##!"#$%&!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! =!! The units used
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS
 A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
UCS Mi-STAR Science 7 Semester 1 Midterm Exam Review Guide
 Name Date Hour 2018-2019 UCS Mi-STAR Science 7 Semester 1 Midterm Exam Review Guide Question How does cold air compare to warm air? Cold air weighs more than hot air. When a 9-centimeter balloon is filled
Name Date Hour 2018-2019 UCS Mi-STAR Science 7 Semester 1 Midterm Exam Review Guide Question How does cold air compare to warm air? Cold air weighs more than hot air. When a 9-centimeter balloon is filled
Unit: Water and the Ocean!
 Name: Block: ELL Notes and Activity Package Unit: Water and the Ocean Chapters in book Learning Goals page Chapter 7.1 1. I can outline the kinetic molecular theory 2. I can distinguish between solids,
Name: Block: ELL Notes and Activity Package Unit: Water and the Ocean Chapters in book Learning Goals page Chapter 7.1 1. I can outline the kinetic molecular theory 2. I can distinguish between solids,
S1 Building Blocks Summary Notes
 S1 Building Blocks Summary Notes Atoms & Molecules 1 We are developing our understanding of atoms and molecules. Atoms are the simplest building blocks of every substance in the universe. There are just
S1 Building Blocks Summary Notes Atoms & Molecules 1 We are developing our understanding of atoms and molecules. Atoms are the simplest building blocks of every substance in the universe. There are just
Understanding Life Systems
 Table of Contents Section 1 Understanding Life Systems Students will explore the components, structures, and functions of cells in plants and animals. They will learn about the postulates of the cell theory
Table of Contents Section 1 Understanding Life Systems Students will explore the components, structures, and functions of cells in plants and animals. They will learn about the postulates of the cell theory
Composition of Air. Air is a mixture of different gases. Gas % Nitrogen 78% Oxygen 21% Argon 1% Carbon Dioxide 0.04%
 The Atmosphere Composition of Air Air is a mixture of different gases Gas % Nitrogen 78% Oxygen 21% Argon 1% Carbon Dioxide 0.04% Water Vapour Varies day to day Experiment to show 1/5 of air is oxygen
The Atmosphere Composition of Air Air is a mixture of different gases Gas % Nitrogen 78% Oxygen 21% Argon 1% Carbon Dioxide 0.04% Water Vapour Varies day to day Experiment to show 1/5 of air is oxygen
Estuary Adventures. Background. Objective
 Estuary Adventures Objective Students will work in groups to understand the concept of estuaries, their importance, and the role that density plays in the mixing of fresh and salt water. Students will
Estuary Adventures Objective Students will work in groups to understand the concept of estuaries, their importance, and the role that density plays in the mixing of fresh and salt water. Students will
Draw a ring around the correct word in the box to complete the sentence.
 Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
E24 PURIFICATION OF ORGANIC COMPOUNDS Distillation, recrystallisation, melting and boiling point determination
 E24 PURIFICATION OF ORGANIC COMPOUNDS Distillation, recrystallisation, melting and boiling point determination THE TASK To learn the main techniques of purifying organic compounds. THE SKILLS By the end
E24 PURIFICATION OF ORGANIC COMPOUNDS Distillation, recrystallisation, melting and boiling point determination THE TASK To learn the main techniques of purifying organic compounds. THE SKILLS By the end
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley)
 Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
To become bigger or make bigger the amount or size of something.
 Increase Decrease (Reduce) State of Matter Property Density Energy Temperature To become bigger or make bigger the amount or size of something. To become smaller or make smaller the amount or size of something.
Increase Decrease (Reduce) State of Matter Property Density Energy Temperature To become bigger or make bigger the amount or size of something. To become smaller or make smaller the amount or size of something.
