2.2 Physical Properties
|
|
|
- Frederick Lambert
- 6 years ago
- Views:
Transcription
1 There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that is hard to miss. A yellow color and a tart taste are two properties of lemonade.
2 Examples of Physical Properties What are some examples of physical properties? A physical property is any characteristic of a material that can be observed or measured without changing the composition of the substances in the material. Viscosity, conductivity, malleability, hardness, melting point, boiling point, and density are examples of physical properties.
3 Examples of Physical Properties Viscosity The tendency of a liquid to keep from flowing is called its viscosity. Thick liquids, such as corn syrup and honey, have a high viscosity. Thin liquids, such as vinegar and water, have a low viscosity.
4 Examples of Physical Properties Conductivity A material s ability to allow heat to flow is called conductivity. Materials that have a high conductivity, such as metals, are called conductors. Good conductors of heat are usually also good conductors of electricity.
5 Examples of Physical Properties Malleability The ability of a solid to be hammered without shattering is malleability. Most metals, such as gold, are malleable. An ice cube or piece of glass breaks into small pieces when struck with a hammer. Solids that shatter when struck are brittle, not malleable.
6 Examples of Physical Properties Hardness One material can scratch another material if it is harder than the other material. A kitchen knife can scratch a copper sheet because stainless steel is harder than copper. The material used to sharpen the knife blade must be harder than stainless steel. Diamond is the hardest known material.
7 Examples of Physical Properties This Tlingit carver is using an adze to carve a canoe from Western red cedar. Red cedar is a relatively soft wood. The adze is hard.
8 Examples of Physical Properties Melting and Boiling Points The temperature at which a material changes state is a physical property. The temperature at which a substance changes from solid to liquid (melts) is its melting point. The temperature at which a substance changes from liquid to gas (boils) is its boiling point.
9 Examples of Physical Properties Melting and Boiling Points Which of these substances are liquids at room temperature (20 C, or 68 F)? Answer:
10 Examples of Physical Properties Melting and Boiling Points Which of these substances are liquids at room temperature (20 C, or 68 F)? Answer: octane, water, and acetic acid
11 Examples of Physical Properties Density The ratio of the mass of a substance to its volume is its density. Density can be used to test the purity of a substance. Silver has a density of 10.5 g/cm 3. A coin with a density of 9.9 g/cm 3 is not made from silver, or it contains substances in addition to silver.
12 Using Physical Properties How can knowing the physical properties of matter be useful? Physical properties are used to identify a material, to choose a material for a specific purpose, or to separate the substances in a mixture.
13 Using Physical Properties Using Properties to Identify Materials A material can be identified by its properties. Decide which properties to test. Do tests on a sample of the unknown material. Compare the results with the data reported for known materials.
14 Using Physical Properties Using Properties to Choose Materials Properties determine which materials are chosen for which uses. For example, shoelaces must be flexible, that is they must be able to bend without breaking. They must also be durable, that is, they must be able to withstand repeated use.
15 Using Physical Properties Laces in hiking boots are usually made of nylon or leather, not from wood.
16 Using Properties to Separate Mixtures What processes are used to separate mixtures? Filtration and distillation are two common separation methods.
17 Using Properties to Separate Mixtures Filtration You can separate hot tea from loose tea leaves by pouring the mixture through a strainer. Filtration is a process that separates materials based on the size of their particles.
18 Using Properties to Separate Mixtures These students filter (sift) dirt through a wire screen to locate small objects. Particles of dirt are small enough to pass through the holes, but objects such as broken bits of pottery are too large.
19 Using Properties to Separate Mixtures Distillation Sometimes all the particles in a solution are small enough to pass through a filter. Distillation is a process that separates the substances in a solution based on their boiling points.
20 Recognizing Physical Changes The change of water from a liquid to a gas during boiling is a physical change. A physical change occurs when some of the properties of a material change, but the substances in the material remain the same.
21 Recognizing Physical Changes During a physical change, the size and shape of a material can change but not the composition. Some examples include melting butter in a pan crumpling a piece of paper slicing a tomato
22 Recognizing Physical Changes Some but not all physical changes can be reversed. Braiding hair is a reversible change. Cutting hair cannot be reversed.
23 Assessment Questions 1. Which of the following is not a physical property? a. density b. boiling point c. flammability d. conductivity
24 Assessment Questions 1. Which of the following is not a physical property? a. density b. boiling point c. flammability d. conductivity ANS: C
25 Assessment Questions 2. Which of these materials is not malleable? a. copper b. aluminum c. glass d. gold
26 Assessment Questions 2. Which of these materials is not malleable? a. copper b. aluminum c. glass d. gold ANS: C
27 Assessment Questions 3. In choosing a material for use as a wire to carry electric current, which physical property would be most important? a. conductivity b. malleability c. hardness d. boiling point
28 Assessment Questions 3. In choosing a material for use as a wire to carry electric current, which physical property would be most important? a. conductivity b. malleability c. hardness d. boiling point ANS: A
29 Assessment Questions 4. Which of these statements best describes a physical change in a pure substance? a. The substance changes into one or more new substances. b. Some of the properties of the substance change, but the material remains the same. c. The properties of the material do not change, and the material remains the same. d. The substance is separated into two or more simpler substances.
30 Assessment Questions 4. Which of these statements best describes a physical change in a pure substance? a. The substance changes into one or more new substances. b. Some of the properties of the substance change, but the material remains the same. c. The properties of the material do not change, and the material remains the same. d. The substance is separated into two or more simpler substances. ANS: B
31 Assessment Questions 1. The process of filtration uses the difference in boiling points of substances to separate a mixture. True False
32 Assessment Questions 1. The process of filtration uses the difference in boiling points of substances to separate a mixture. True False ANS: F, distillation
2.2 Physical Properties. Physical Properties. Notes due EOC
 Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
7.3 Bonding in Metals > Chapter 7 Ionic and Metallic Bonding. 7.3 Bonding in Metals. 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds
 Chapter 7 Ionic and Metallic Bonding 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds 7.3 Bonding in Metals 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY & YOU What
Chapter 7 Ionic and Metallic Bonding 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds 7.3 Bonding in Metals 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY & YOU What
Properties of Metals and Alloys
 Properties of Metals and Alloys 1 of 19 Boardworks Ltd 2016 Properties of Metals and Alloys 2 of 19 Boardworks Ltd 2016 What is the structure of metals? 3 of 19 Boardworks Ltd 2016 Metal particles are
Properties of Metals and Alloys 1 of 19 Boardworks Ltd 2016 Properties of Metals and Alloys 2 of 19 Boardworks Ltd 2016 What is the structure of metals? 3 of 19 Boardworks Ltd 2016 Metal particles are
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
D =? D = M = 10 g = 5 g/cm 3 M = 10 g V 2 cm 3 V = 2 cm 3 5 g/cm 3
 Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Metal and Non Metals
 Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Name Honors Chemistry / /
 Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Corresponding Physical or Chemical Property bread rising through the use of baking soda carbonates decompose to form CO 2
 Substance = a chemical (element or mixture) Substance mixture chemical process = chemical reaction (new substances result) physical process = same substances present through out Physical or chemical processes?
Substance = a chemical (element or mixture) Substance mixture chemical process = chemical reaction (new substances result) physical process = same substances present through out Physical or chemical processes?
5. MINERAL PROPERTIES, IDENTIFICATION, & USES
 LAST NAME (ALL IN CAPS): FIRST NAME: 5. MINERAL PROPERTIES, IDENTIFICATION, & USES Instructions: Refer to Laboratory 3 in your Lab Manual on pages 73-110 to answer the questions in this work sheet. Your
LAST NAME (ALL IN CAPS): FIRST NAME: 5. MINERAL PROPERTIES, IDENTIFICATION, & USES Instructions: Refer to Laboratory 3 in your Lab Manual on pages 73-110 to answer the questions in this work sheet. Your
1. METALS 2. FERRIC METALS 3. NON-FERRIC METALS 4. WORKING WITH METALS 5. METAL FORMING TECHNIQUES 6. ENVIRONMENTAL IMPACT OF METAL EXTRACTION
 METALS: 1. METALS 2. FERRIC METALS 3. NON-FERRIC METALS 4. WORKING WITH METALS 5. METAL FORMING TECHNIQUES 6. ENVIRONMENTAL IMPACT OF METAL EXTRACTION 1. METALS: Metals are chemical elements found in nature
METALS: 1. METALS 2. FERRIC METALS 3. NON-FERRIC METALS 4. WORKING WITH METALS 5. METAL FORMING TECHNIQUES 6. ENVIRONMENTAL IMPACT OF METAL EXTRACTION 1. METALS: Metals are chemical elements found in nature
The table shows the students suggestions about the identity of P.
 1 Three students, X, Y and Z, were told that solid P reacts with dilute acids and also conducts electricity. The table shows the students suggestions about the identity of P. Which of the students are
1 Three students, X, Y and Z, were told that solid P reacts with dilute acids and also conducts electricity. The table shows the students suggestions about the identity of P. Which of the students are
Chapter 3 - Energy and Matter Practice Problems
 Chapter 3 - Energy and Matter Practice Problems 1) Helium is a(n) A) compound. B) heterogeneous mixture. C) element. D) homogeneous mixture. E) electron. 2) Air is a(n) A) compound. B) heterogeneous mixture.
Chapter 3 - Energy and Matter Practice Problems 1) Helium is a(n) A) compound. B) heterogeneous mixture. C) element. D) homogeneous mixture. E) electron. 2) Air is a(n) A) compound. B) heterogeneous mixture.
Properties of Minerals
 What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet
 Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
THE PERIODIC TABLE. Chapter 15
 THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
Natural Science. and. Technology. Grade 5
 1 Natural Science and Technology Grade 5 2 Contents Topic 6 Metals and Non-metals Unit 1 Properties of metals Unit 2 Properties of non- metals Topic 7 Uses of metals Unit 1 Other properties of metals Unit
1 Natural Science and Technology Grade 5 2 Contents Topic 6 Metals and Non-metals Unit 1 Properties of metals Unit 2 Properties of non- metals Topic 7 Uses of metals Unit 1 Other properties of metals Unit
There are about 3,000 known minerals, only about 30 are common. The most common are quartz,feldspar,mica, and calcite.
 There are about 3,000 known minerals, only about 30 are common. The most common are quartz,feldspar,mica, and calcite. These minerals make up most of the rocks found in the Earth s crust. Most of the Earth
There are about 3,000 known minerals, only about 30 are common. The most common are quartz,feldspar,mica, and calcite. These minerals make up most of the rocks found in the Earth s crust. Most of the Earth
Chemistry Quiz #1 Review
 Name: Chemistry Quiz 1 Review Page 1 of 6 Date: Please check your answers at http://leetz.weebly.com Chemistry Quiz #1 Review Part A: Density Calculations 1. The density of sodium is 0.97 g/cm 3. A sample
Name: Chemistry Quiz 1 Review Page 1 of 6 Date: Please check your answers at http://leetz.weebly.com Chemistry Quiz #1 Review Part A: Density Calculations 1. The density of sodium is 0.97 g/cm 3. A sample
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
Physical Behavior of Metals
 Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
Draw a ring around the correct word in the box to complete the sentence.
 Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
Unit 2M.3 & 2M.4: Natural and man-made materials and changing materials
 Unit 2M.3 & 2M.4: Natural and man-made materials and changing materials Natural and man-made materials. Shaping materials Heating materials Science Skills Observing Classifying Predicting Experimenting
Unit 2M.3 & 2M.4: Natural and man-made materials and changing materials Natural and man-made materials. Shaping materials Heating materials Science Skills Observing Classifying Predicting Experimenting
1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these characteristics of mercury physical or chemical properties?
 Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
(A) Copper. (B) Zinc. (C) Iron. (D) Wood. (A) Zinc. (B) Wood. (C) Rubber. (D) Plastic.
 Downloaded from MATERIALS : METALS AND NON-METALS 1.Give one example of each: metals and non-metals. 2.Name the metal, which is the best conductor of heat and electricity. 3.Name the property by which
Downloaded from MATERIALS : METALS AND NON-METALS 1.Give one example of each: metals and non-metals. 2.Name the metal, which is the best conductor of heat and electricity. 3.Name the property by which
page - Labs 09 & 10 - Mineral Identification
 Labs 09 & 10 - Mineral Identification page - All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the
Labs 09 & 10 - Mineral Identification page - All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the
2.3 Density and Density Calculations
 2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
Free Electron Model What kind of interactions hold metal atoms together? How does this explain high electrical and thermal conductivity?
 Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Mechanical Lustrous
Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Mechanical Lustrous
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
INDEX METALS FERRIC METALS NON-FERRIC METALS WORKING WITH METALS METAL FORMING TECHNIQUES ENVIRONMENTAL IMPACT OF METAL EXTRACTION
 METALS INDEX METALS FERRIC METALS NON-FERRIC METALS WORKING WITH METALS METAL FORMING TECHNIQUES ENVIRONMENTAL IMPACT OF METAL EXTRACTION METALS CHEMICAL ELEMENTS FOUND IN NATURE IN SOLID STATE AT ROOM
METALS INDEX METALS FERRIC METALS NON-FERRIC METALS WORKING WITH METALS METAL FORMING TECHNIQUES ENVIRONMENTAL IMPACT OF METAL EXTRACTION METALS CHEMICAL ELEMENTS FOUND IN NATURE IN SOLID STATE AT ROOM
The table below shows some of the properties of the elements cobalt and nickel.
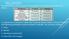 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
001- Engineering Skills Principles-1. Chapter - 03 Material
 001- Engineering Skills Principles-1 Chapter - 03 Material 1 Objective of Chapter 3.0 (Material) The aim of this section is to enable the candidate to: 1. Identify metals and compare their properties.
001- Engineering Skills Principles-1 Chapter - 03 Material 1 Objective of Chapter 3.0 (Material) The aim of this section is to enable the candidate to: 1. Identify metals and compare their properties.
2. Crystallization. A. Background
 2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
1. Hardness 2. Streak 3. Density 4. Crystal structure 5. A mineral must be a naturally occurring, inorganic. 7. c 8. f 9. a 10. d 11. g 12. e 13.
 Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Mark E. Damon - All Rights Reserved
 2001 - All rights Reserved markedamon@hotmail.com Another Presentation *Edited by Wilmeth Elementary s awesome 5 th grade teachers Directions: Click View From Beginning to get started Scroll through the
2001 - All rights Reserved markedamon@hotmail.com Another Presentation *Edited by Wilmeth Elementary s awesome 5 th grade teachers Directions: Click View From Beginning to get started Scroll through the
Homework for Unit Vocab for Unit 6; due: 2. Pg 333 (1-5), Pg 335 (1-4), Pg 337 (1-4), Pg 339 (1-6), No sentences; due:
 Unit 6 Heat Homework for Unit 6 1. Vocab for Unit 6; due: 2. Pg 333 (1-5), Pg 335 (1-4), Pg 337 (1-4), Pg 339 (1-6), No sentences; due: 3. Temperature change activity; due: 4. Heat Transfer Homework; due:
Unit 6 Heat Homework for Unit 6 1. Vocab for Unit 6; due: 2. Pg 333 (1-5), Pg 335 (1-4), Pg 337 (1-4), Pg 339 (1-6), No sentences; due: 3. Temperature change activity; due: 4. Heat Transfer Homework; due:
Chapter 11 The Periodic Table
 Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
Covered with a thin layer of oxide at ordinary temperatures.
 1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
page - 1 Lab 9 - Mineral Identification
 Lab 9 - Mineral Identification page - 1 All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the bulk
Lab 9 - Mineral Identification page - 1 All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the bulk
Experimental techniques
 Unit 1 Experimental techniques In this unit you will come to understand the general principles behind various experimental techniques. Experimental investigation is a very important aspect of Physical
Unit 1 Experimental techniques In this unit you will come to understand the general principles behind various experimental techniques. Experimental investigation is a very important aspect of Physical
1 Graphite is a form of carbon. (a) Graphite is used as a lubricant. Write down one property of graphite that explains why it is used as a lubricant.
 1 Graphite is a form of carbon. (a) Graphite is used as a lubricant. Write down one property of graphite that explains why it is used as a lubricant.... [1] (b) Graphite conducts electricity. Explain how.
1 Graphite is a form of carbon. (a) Graphite is used as a lubricant. Write down one property of graphite that explains why it is used as a lubricant.... [1] (b) Graphite conducts electricity. Explain how.
H N 2. Decolorizing carbon O. O Acetanilide
 Experiment 1: Recrystallization of Acetanilide Reading Assignment Mohrig 2 4 (Glassware, Reagents, & Heating) & 14 15 (Melting Point & Recrystallization) The purification of organic compounds is a tedious,
Experiment 1: Recrystallization of Acetanilide Reading Assignment Mohrig 2 4 (Glassware, Reagents, & Heating) & 14 15 (Melting Point & Recrystallization) The purification of organic compounds is a tedious,
Bonding and Limestone Questions
 Bonding and Limestone Questions 56 minutes 56 marks Page of 26 Q. Sodium is a reactive element. (a) Complete the diagram to show the electronic structure of a sodium atom. (2) (b) Sodium reacts with chlorine
Bonding and Limestone Questions 56 minutes 56 marks Page of 26 Q. Sodium is a reactive element. (a) Complete the diagram to show the electronic structure of a sodium atom. (2) (b) Sodium reacts with chlorine
AQA GCSE Design and Technology 8552
 AQA GCSE Design and Technology 8552 Metals and alloys Unit 3 Materials and their working properties 3 Objectives Know the primary sources of materials for producing metals and alloys Be able to recognise
AQA GCSE Design and Technology 8552 Metals and alloys Unit 3 Materials and their working properties 3 Objectives Know the primary sources of materials for producing metals and alloys Be able to recognise
solvent diffusion dissolving soluble
 What do we call it when a liquid changes into a solid? What do we call it when a liquid turns into a gas? What do we call it when a gas turns into a liquid? What do we call the solid that dissolves in
What do we call it when a liquid changes into a solid? What do we call it when a liquid turns into a gas? What do we call it when a gas turns into a liquid? What do we call the solid that dissolves in
UNCONVENTIONAL MACHINING PROCESS UNIT 1 INTRODUCTION. Prepared by S. SENTHIL KUMAR AP / MECH SVCET
 UNCONVENTIONAL MACHINING PROCESS UNIT 1 INTRODUCTION Prepared by S. SENTHIL KUMAR AP / MECH SVCET INTRODUCTION Conventional machining process Metal is removed by means of tool which is harder than work
UNCONVENTIONAL MACHINING PROCESS UNIT 1 INTRODUCTION Prepared by S. SENTHIL KUMAR AP / MECH SVCET INTRODUCTION Conventional machining process Metal is removed by means of tool which is harder than work
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
Downloaded from
 Fun with Magnets 1 1.What happens when the north pole of a magnet is placed near the north pole of another magnet? 2.Which of the following cannot be attracted by a magnet? (A) Wood. (B) Nickel. (C)
Fun with Magnets 1 1.What happens when the north pole of a magnet is placed near the north pole of another magnet? 2.Which of the following cannot be attracted by a magnet? (A) Wood. (B) Nickel. (C)
GCSE BITESIZE Examinations
 GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
Extracting and using metals. ores. native. Only the most unreactive metals such as gold and platinum are found as native metals.
 Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
2 Identifying Minerals
 CHAPTER 1 2 Identifying Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What seven properties can be used to identify
CHAPTER 1 2 Identifying Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What seven properties can be used to identify
Free Electron Model What kind of interactions hold metal atoms together? How does this explain high electrical and thermal conductivity?
 Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Free Electron
Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Free Electron
Minerals. This study packet belongs to
 Minerals This study packet belongs to What are minerals? To understand what minerals are, you need to know a bit about what makes up our Earth. Our Earth is made up of about 109 pure substances called.
Minerals This study packet belongs to What are minerals? To understand what minerals are, you need to know a bit about what makes up our Earth. Our Earth is made up of about 109 pure substances called.
CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1
 CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1 TECHNIQUES: chemical and physical changes, reactions, observations READING: PHYSICAL AND CHEMICAL CHANGES e.g. Tro chapter 1 SAFETY: Safety
CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1 TECHNIQUES: chemical and physical changes, reactions, observations READING: PHYSICAL AND CHEMICAL CHANGES e.g. Tro chapter 1 SAFETY: Safety
Separating Mixtures WORKSHEETS
 Attention Teachers KEEP IT SIMPLE SCIENCE Separating Year 7 Chemical Sciences WORKSHEETS KISS topic number Topic 03.7C KISS Worksheets are designed to consolidate students knowledge & understanding and/or
Attention Teachers KEEP IT SIMPLE SCIENCE Separating Year 7 Chemical Sciences WORKSHEETS KISS topic number Topic 03.7C KISS Worksheets are designed to consolidate students knowledge & understanding and/or
OC30 Conduct a qualitative experiment to detect the presence of dissolved solids in water samples, and test water for hardness (soap test)
 Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
SMAW. Shielded metal arc welding (SMAW) is commonly referred to as stick welding
 SMAW EQUIPMENT SMAW Shielded metal arc welding (SMAW) is commonly referred to as stick welding An electric arc between the stick electrode and the base metal creates heat. Heat melts the base metal and
SMAW EQUIPMENT SMAW Shielded metal arc welding (SMAW) is commonly referred to as stick welding An electric arc between the stick electrode and the base metal creates heat. Heat melts the base metal and
Iron filings (Fe) 56g IRON + SULPHUR IRON SULPHIDE
 W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
6.4 The Structure of Metals. Vocabulary metallic bond alloy
 Section 6.4 6.4 The Structure of Metals 1 FOCUS Objectives 6.4.1 Describe the structure and strength of bonds in metals. 6.4.2 Relate the properties of metals to their structure. 6.4.3 Define an alloy
Section 6.4 6.4 The Structure of Metals 1 FOCUS Objectives 6.4.1 Describe the structure and strength of bonds in metals. 6.4.2 Relate the properties of metals to their structure. 6.4.3 Define an alloy
CHAPTER 9. The Uses of Sulphuric Acid
 CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
v = 5.00cm x 3.00cm x 2.00cm 2. What is the volume of a marble that has a mass of 3.0 g and a density of 2.7 g/ml? Formula (density) Substitution
 Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Assistant Lecturer. Sahar Mohammed Shakir Assistant Lecturer. Abdul Hafeedh Hameed Assistant Lecturer. Ali Basim
 Assistant Lecturer Sahar Mohammed Shakir Assistant Lecturer Abdul Hafeedh Hameed Assistant Lecturer Ali Basim Solid organic cpd.s when isolated from organic reaction are impure; they are contaminated with
Assistant Lecturer Sahar Mohammed Shakir Assistant Lecturer Abdul Hafeedh Hameed Assistant Lecturer Ali Basim Solid organic cpd.s when isolated from organic reaction are impure; they are contaminated with
Name Class Date. Does it have a crystalline structure? Minerals are crystals. Each mineral has a certain crystal structure that is always the same.
 CHAPTER 1 1 What Is a Mineral? SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What are minerals? What determines the shape
CHAPTER 1 1 What Is a Mineral? SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: What are minerals? What determines the shape
PHASE CHANGES. Time Temperature Observations. Name(s)
 3 5 PHASE CHANGES PHASE CHANGES Name(s) The activities presented here focus on the energy changes that occur in substances undergoing a phase change. The first activity will take the most time to complete.
3 5 PHASE CHANGES PHASE CHANGES Name(s) The activities presented here focus on the energy changes that occur in substances undergoing a phase change. The first activity will take the most time to complete.
Year 7 Chemistry HW Questions
 Year 7 Chemistry HW Questions 37 minutes 56 marks Page 1 of 15 Q1. Molly used a ph sensor to test different liquids. She dipped the probe of the sensor into each liquid and recorded the ph value in a table.
Year 7 Chemistry HW Questions 37 minutes 56 marks Page 1 of 15 Q1. Molly used a ph sensor to test different liquids. She dipped the probe of the sensor into each liquid and recorded the ph value in a table.
Mon. Tues. Wed. Thurs. Fri. AM or PM B
 Name: (cf. Honesty Declaration Statement on page 20) Laboratory Day (circle) Lab Room Locker Lab. Session (circle) Lab. Section Mon. Tues. Wed. Thurs. Fri. AM or PM B Date experiment is performed MARK:
Name: (cf. Honesty Declaration Statement on page 20) Laboratory Day (circle) Lab Room Locker Lab. Session (circle) Lab. Section Mon. Tues. Wed. Thurs. Fri. AM or PM B Date experiment is performed MARK:
Solids SECTION Critical Thinking
 SECTION 10.3 Solids A gas has neither a definite volume nor a definite shape. A liquid has a definite volume, but not a definite shape. A solid, the third state, has a definite volume and a definite shape.
SECTION 10.3 Solids A gas has neither a definite volume nor a definite shape. A liquid has a definite volume, but not a definite shape. A solid, the third state, has a definite volume and a definite shape.
A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition.
 A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition. Naturally Occurring Formed by processes that occur in the natural world Inorganic Not
A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition. Naturally Occurring Formed by processes that occur in the natural world Inorganic Not
أت ارش. Dr. Abdel-Wahab El-Morsy Faculty of Engineering - Rabigh
 Basic Workshop 1 أت ارش There are thousands of materials available for use in engineering applications. Most materials fall into one of three classes that are based on the atomic bonding forces of a particular
Basic Workshop 1 أت ارش There are thousands of materials available for use in engineering applications. Most materials fall into one of three classes that are based on the atomic bonding forces of a particular
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
Anglon Tools Tools for ANGL ROTARY HANDPC Soft rubber pads available in 4 different diameters (ø 0 mm, ø mm, ø 30 mm, ø mm) are available. Pressure se
 inimo One Anglon Tools inimo Anglon System -- Versatility & Performance inimo Anglon angle grinders, with angles of 90 and degrees, are suitable for polishing flat, curved and end surfaces. Great for areas
inimo One Anglon Tools inimo Anglon System -- Versatility & Performance inimo Anglon angle grinders, with angles of 90 and degrees, are suitable for polishing flat, curved and end surfaces. Great for areas
Chapter 26: Canada s Mineral Wealth May 8, Monday, May 12, 14
 Chapter 26: Canada s Mineral Wealth May 8, 2014. What kind of mining products do you have on you today? Types of minerals a mineral is a naturally occurring, pure, non-living substance found in rocks TYPES
Chapter 26: Canada s Mineral Wealth May 8, 2014. What kind of mining products do you have on you today? Types of minerals a mineral is a naturally occurring, pure, non-living substance found in rocks TYPES
Classifying Minerals. Name: Date: Class: #: Materials: Magnifying lens (2) Copper Penny (2) Glass Plate Mohs Scale Set of minerals Mineral
 Name: Date: Class: #: Classifying Minerals Materials: Magnifying lens (2) Copper Penny (2) Glass Plate Mohs Scale Set of minerals Mineral Magnet Iron Nail Streak Plates (1 white & 1 black) Identification
Name: Date: Class: #: Classifying Minerals Materials: Magnifying lens (2) Copper Penny (2) Glass Plate Mohs Scale Set of minerals Mineral Magnet Iron Nail Streak Plates (1 white & 1 black) Identification
Introduction to Density
 Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
METALS AND THEIR COMPOUNDS
 METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
Installation Guide Wall Panels
 Installation Guide Wall Panels Jointless or Dry-Stacked Installation (Read Prior to installation) The MStone system is a jointless / dry-stacked installation where panels and corners fit tightly together
Installation Guide Wall Panels Jointless or Dry-Stacked Installation (Read Prior to installation) The MStone system is a jointless / dry-stacked installation where panels and corners fit tightly together
These conditions must be maintained during transport to the laboratory.
 A successful laboratory analysis starts in the field. The result of an analysis is as good as the representativeness of the soil sample used. The sampling procedure is basically determined by the analyses
A successful laboratory analysis starts in the field. The result of an analysis is as good as the representativeness of the soil sample used. The sampling procedure is basically determined by the analyses
Measurement Methods and Calculations to Determine Internal Deposit Stress. Frank H. Leaman Specialty Testing & Development, Inc.
 Measurement Methods and Calculations to Determine Internal Deposit Stress Frank H. Leaman Specialty Testing & Development, Inc. York, PA Methods for Deposit Stress Determination Bent Strip (simple beam
Measurement Methods and Calculations to Determine Internal Deposit Stress Frank H. Leaman Specialty Testing & Development, Inc. York, PA Methods for Deposit Stress Determination Bent Strip (simple beam
3th GRADE MINIMUM CONTENTS UDI 3: MATERIALS & MACHINES (9)
 MATERIALS What are materials? 3th GRADE MINIMUM CONTENTS UDI 3: MATERIALS & MACHINES (9) Materials are the substances that we use to make something. We use material for building, making tools, making clothes,
MATERIALS What are materials? 3th GRADE MINIMUM CONTENTS UDI 3: MATERIALS & MACHINES (9) Materials are the substances that we use to make something. We use material for building, making tools, making clothes,
The Technological World. Chapter 12
 The Technological World Chapter 12 Chapter 12 Manufacturing Technical Objects There have been many inventions that have improved the quality of our lives. We will refer to these inventions as technical
The Technological World Chapter 12 Chapter 12 Manufacturing Technical Objects There have been many inventions that have improved the quality of our lives. We will refer to these inventions as technical
ArenNano Black Stainless black sol gel on Stainless Steel
 black sol gel on Stainless Steel 1 Context of the NANOSOL project Transparent protection layer Top coat Top coat + protection Primer Metal Protection of organic layers Coating for home appliances Anti
black sol gel on Stainless Steel 1 Context of the NANOSOL project Transparent protection layer Top coat Top coat + protection Primer Metal Protection of organic layers Coating for home appliances Anti
ME -215 ENGINEERING MATERIALS AND PROCESES
 ME -215 ENGINEERING MATERIALS AND PROCESES Instructor: Office: MEC325, Tel.: 973-642-7455 E-mail: samardzi@njit.edu PROPERTIES OF MATERIALS Chapter 3 Materials Properties STRUCTURE PERFORMANCE PROCESSING
ME -215 ENGINEERING MATERIALS AND PROCESES Instructor: Office: MEC325, Tel.: 973-642-7455 E-mail: samardzi@njit.edu PROPERTIES OF MATERIALS Chapter 3 Materials Properties STRUCTURE PERFORMANCE PROCESSING
Electron Microscopy (EM) Grid
 Anirban Som 25-01-14 Instrumental technique presentation Electron Microscopy (EM) Grid What I will talk about Some basic topics about EM grid Home-made grid preparation Grid cleaning Carbon coating and
Anirban Som 25-01-14 Instrumental technique presentation Electron Microscopy (EM) Grid What I will talk about Some basic topics about EM grid Home-made grid preparation Grid cleaning Carbon coating and
Alloys and Solid Solutions
 Alloys and Solid Solutions Chemistry 123 Spring 2008 Dr. Woodward Solutions Solid Solution 14 Carat Gold Liquid Solution Vodka Gaseous Solution Air Solution = A homogeneous mixture 1 Alloys An alloy is
Alloys and Solid Solutions Chemistry 123 Spring 2008 Dr. Woodward Solutions Solid Solution 14 Carat Gold Liquid Solution Vodka Gaseous Solution Air Solution = A homogeneous mixture 1 Alloys An alloy is
Physical Properties of Minerals
 1 of 8 9/20/2002 9:30 AM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy Physical Properties of Minerals This document last updated on 20-Sep-2002 Although we have discussed x-ray identification
1 of 8 9/20/2002 9:30 AM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy Physical Properties of Minerals This document last updated on 20-Sep-2002 Although we have discussed x-ray identification
Metals And Their Properties- Physical and Chemical
 Metals And Their Properties- Physical and Chemical All the things around us are made of 100 or so elements. These elements were classified by Lavoisier in to metals and non-metals by studying their properties.
Metals And Their Properties- Physical and Chemical All the things around us are made of 100 or so elements. These elements were classified by Lavoisier in to metals and non-metals by studying their properties.
85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO 3
 Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
MATERIALS SCIENCE-44 Which point on the stress-strain curve shown gives the ultimate stress?
 MATERIALS SCIENCE 43 Which of the following statements is FALSE? (A) The surface energy of a liquid tends toward a minimum. (B) The surface energy is the work required to create a unit area of additional
MATERIALS SCIENCE 43 Which of the following statements is FALSE? (A) The surface energy of a liquid tends toward a minimum. (B) The surface energy is the work required to create a unit area of additional
Machinability is the ease with which a given material may be worked with a cutting tool
 Machinability Machinability is the ease with which a given material may be worked with a cutting tool Machinability ratings (MR) provide and understanding of the severity of a metalworking operation in
Machinability Machinability is the ease with which a given material may be worked with a cutting tool Machinability ratings (MR) provide and understanding of the severity of a metalworking operation in
Mixtures in Industry
 3.7 Mixtures in Industry Key Question: How are mixtures separated in industry? Many industries separate mixtures to make pure products. In this section, you will learn about three industries that separate
3.7 Mixtures in Industry Key Question: How are mixtures separated in industry? Many industries separate mixtures to make pure products. In this section, you will learn about three industries that separate
An Organized Table Worksheet Due Thursday Name: Date: Period:
 An Organized Table Worksheet Due Thursday Name: Date: Period: The Periodic Table of Elements In 1871, the first periodic table was developed by Dmitrii Mendeleev. Mendeleev is known as the father of the
An Organized Table Worksheet Due Thursday Name: Date: Period: The Periodic Table of Elements In 1871, the first periodic table was developed by Dmitrii Mendeleev. Mendeleev is known as the father of the
Chapter: The Periodic Table
 Table of Contents Chapter: The Periodic Table Section 1: Introduction to the Periodic Table Section 2: Representative Elements Section 3: Transition Elements 1 Introduction to the Periodic Table Development
Table of Contents Chapter: The Periodic Table Section 1: Introduction to the Periodic Table Section 2: Representative Elements Section 3: Transition Elements 1 Introduction to the Periodic Table Development
Mathematics and Science in Schools in Sub-Saharan Africa
 Mathematics and Science in Schools in Sub-Saharan Africa MATERIAL SCIENCE METAL ALLOYS Metals have many advantages Strong Malleable Conductors Shiny Metal Customizing Cold-working and heat treating metals
Mathematics and Science in Schools in Sub-Saharan Africa MATERIAL SCIENCE METAL ALLOYS Metals have many advantages Strong Malleable Conductors Shiny Metal Customizing Cold-working and heat treating metals
1-Materials Science & Materials Engineering
 1-Materials Science & Materials Engineering 1-1-Structure & Properties Relationship (Materials Science or Materials Engineering) Processing Structure Properties Performance Sub Atomic Atomic Sub Atomic
1-Materials Science & Materials Engineering 1-1-Structure & Properties Relationship (Materials Science or Materials Engineering) Processing Structure Properties Performance Sub Atomic Atomic Sub Atomic
: Series 300 Stainless Steel Wire Brushes
 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name : Series 300 Stainless Steel Wire Brushes 1.2. Relevant identified uses of the substance
SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name : Series 300 Stainless Steel Wire Brushes 1.2. Relevant identified uses of the substance
A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition.
 Minerals Objectives List the 5 traits of minerals Identify minerals Describe characteristics of minerals Know how minerals form Describe ways that we use minerals A mineral is a naturally occurring, inorganic
Minerals Objectives List the 5 traits of minerals Identify minerals Describe characteristics of minerals Know how minerals form Describe ways that we use minerals A mineral is a naturally occurring, inorganic
NCERT solutions for Metals and Non Metals
 NCERT solutions for Metals and Non Metals 1 Question 1 Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv)
NCERT solutions for Metals and Non Metals 1 Question 1 Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv)
Nickel Electroplating
 Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
