Webinar: Genome Editing using Nucleofector Technology
|
|
|
- Emma Washington
- 6 years ago
- Views:
Transcription
1 Pharma&Biotech BioResearch Webinar: Genome Editing using Nucleofector Technology Efficient Transfection of ZFN, TALEN and CRISPR/Cas In collaboration with: Lonza Walkersville, Inc., Walkersville, MD / 2014 Lonza
2 Speakers Dr. Claudia Schwartz Senior Scientific Support Specialist, Lonza Erika Holroyd ZFN and CRISPR Technical Specialist Moderation: Dr. Saskia Ihle, Scientific Support Specialist, Lonza 2
3 Agenda Basic Introduction to Genome Editing Tools Introduction to Nucleofector Technology Application Examples TALEN-based Genome Editing ZFN- and CRIPSR-based Genome Editing (by Sigma-Aldrich) Technical Tips For ZFN/CRISPR (by Sigma-Aldrich) For Nucleofection Summary 4
4 Methods for Modulating Gene Expression DNA RNA Protein Transcription Translation Zinc Finger Nucleases TALENs CRISPRs Transposons antisense Oligos Plasmids sirna shrna mirna Small Molecules mabs Aptamers 5
5 What is Genome Editing? Using engineered nucleases to introduce DNA insertions, deletions or replacements at sequence specific sites in a genome Engineered chimeric nucleases contain A non-specific DNA cleavage module (nuclease*): induces double-strand breaks in the genomic DNA targeted, which stimulates the DNA repair process Sequence-specific DNA binding domains (ZFN or TALE protein or CRISPRgRNA): can be customized to recognize virtually any sequence *Other options under evaluation: nickases, recombinases, transposases, transcription activators/repressors.. 6
6 Genome Editing Possibilities Add/delete TF binding sites Insert constitutive or inducible promoter Generate point mutation Fuse a reporter gene Delete/mutate promoter Disrupt gene (knockout) Insert new exon to generate gene fusions Add/delete mirna control element Transcription factor Promoter Exon UTR Gene knockouts Deletions of large sections of DNA Point mutations Incorporation of unique restriction sites Integration of loxp sites Integration of expression cassettes Adapted from Sigma-Aldrich with permission 7
7 Genome Editing Process Double-strand Break Breaks induce DNA repair and increase mutagenesis frequencies by >1000 fold DSB = Double strand break Plus donor DNA NHEJ = Non-homologous end joining Targeted mutagenesis/deletion HDR = Homology directed repair Targeted insertion/replacement 8
8 Genome Editing Process Example: ZFN DSB = Double strand break Plus donor DNA NHEJ = Non-homologous end joining Targeted mutagenesis/deletion HDR = Homology directed repair Targeted insertion/replacement 9
9 Genome Editing Process Example: TALEN DSB = Double strand break Plus donor DNA NHEJ = Non-homologous end joining Targeted mutagenesis/deletion HDR = Homology directed repair Targeted insertion/replacement 10
10 Genome Editing Process Example: CRISPR/Cas9 DSB = Double strand break Plus donor DNA NHEJ = Non-homologous end joining Targeted mutagenesis/deletion HDR = Homology directed repair Targeted insertion/replacement 11
11 Genome Editing Tools ZFN, TALEN and CRISPR ZFN TALEN CRISPR Nuclease Fok 1 Fok 1 Cas9 DNA Binding Via ZF protein TALE protein GuideRNA (grna) Type Fusion protein Fusion protein Protein + RNA High effort to modify for new targeting site High effort to modify for new targeting site Easy to modify Multiple targeting possible Binding Site 2 sites (15 or 18 bp each) High specificity Low risk for off-target effects 2 sites ( 13 bp each) High specificity Low risk for off-target effects 1 site (18-20 bp + 3bp NGG) Lower specificity Higher risk for off-target effects Summarized from: Gaj T et al (2013) Trends in Biotechnology 31(7):
12 Genome Editing Requires Co-Transfection ZFN or TALEN CRIPSR/Cas 2x or or Fok1 ZFN fusion or Fok1-TALE fusion Repair plasmid or ssodn (optional) Cas9 + grna Repair plasmid or ssodn (optional) 2x or or Fok1 ZFN fusion mrna or Fok1-TALE fusion mrna Repair plasmid or ssodn (optional) Cas9 grna Repair plasmid or ssodn (optional) or Cas9 grna PCR cassette Repair plasmid or ssodn (optional) 13
13 Agenda Basic Introduction to Genome Editing Tools Introduction to Nucleofector Technology Application Examples TALEN-based Genome Editing ZFN- and CRIPSR-based Genome Editing (by Sigma-Aldrich) Technical Tips For ZFN/CRISPR (by Sigma-Aldrich) For Nucleofection Summary 14
14 Nucleofection Principle Notebook Control Unit High transfection efficiency combined with low mortality DNA is directed into the nucleus giving faster gene expression Nucleofection Principle Other transfection methods 15
15 Components of Nucleofector Technology Notebook Control Unit A Unique Combination: Nucleofector Device Specific Nucleofector Kits Detailed optimized protocols Enabling excellent transfection performance combined with high functionality! For more information, check out: 16
16 More Than 80 Primary Cell Types Successfully Transfected % Transfection Efficiency Analysis time: h post Nucleofection Notebook Control Unit Reporter genes H-2K k egfp (customer data) maxgfp Reporter Protein GFP/YFP (customer data) 17
17 Agenda Basic Introduction to Genome Editing Tools Introduction to Nucleofector Technology Application Examples TALEN-based Genome Editing ZFN- and CRIPSR-based Genome Editing (by Sigma-Aldrich) Technical Tips For ZFN/CRISPR (by Sigma-Aldrich) For Nucleofection Summary 18
18 Nucleofection of TALEN or CRISPR/Cas9 into ips Cells Yang L et al (2013) Optimization of scarless human stem cell genome editing. NAR 41: (George Church Lab) Functional re-coding of TALEs (retalens) that simplify TALE synthesis Optimization of ssodn donor design Using Nucleofection to transfect hipscs: Comparison of integration efficiencies of retalens and Cas9-gRNA targeting CCR5 Combined optimized ssodns with both methods for introducing specific mutations Result: retalens Cas9-gRNA NHEJ 0.4% 3% HDR with ssodn 0.6% 1% Both nuclease platforms serve as robust tools for genome editing, but Cas9-gRNA achieved 7-8 x higher NHEJ 19
19 Nucleofection for Genome Editing Further Applications Tool Authors Citation Year Cell type ZFN Fung H et al PLOS ONE May 2011, e hesc Zou J et al Blood 117: ipsc Zou J et al Blood 118: ipsc Torikai H et al Blood 119(24): Human TC Liu X et al PLOS ONE, May 2012, e hes Schjoldager K PNAS 109: HepG2 Wang J et al Genome Res 22: K562 Ou W et al PLOS ONE, Nov 2013, e ipsc Qu X et al Nucleic Acids Res 41: HIV-infected PBL + CD4 T cells Richter S et al PLOS ONE, Jun 2013, e HTC116 + H460 Robbez-Masson LJ et al PLOS ONE 8(11):e MCF7 Samsonov A et al PLOS ONE, July 2013, e A549 Toscano MG et al Dis Model Mech 6: K562 Genovese P et al. Nature 510:235ff 2014 hcd34 TALEN Piganeau M et al Genome Res 23: hes and Jurkat cells Yang L et al Nucleic Acids Res 41: ipsc Zhu F et al. Nucleic Acids Res /nar/gkt ipsc + H9 hes CRISPR Petit Cs et al J Cell Biol 202: HeLa Ran FA et al Cell 154: various cell lines, e.g. HEK293FT Ran FA et al* Nat Prot 8(11): HUES62 + HEK293 *Provides a comprehensive protocol for use of the 4D-Nucleofector with CRIPS/Cas 20
20 Agenda Basic Introduction to Genome Editing Tools Introduction to Nucleofector Technology Application Examples TALEN-based Genome Editing ZFN- and CRIPSR-based Genome Editing (by Sigma-Aldrich) Technical Tips For ZFN/CRISPR (by Sigma-Aldrich) For Nucleofection Summary 21
21 PTEN Knockout with ZFN MCF10A - PTEN (-/-), clone A2A9 Genotype: del 19 / del 2 ZFN Binding site in upper case RED ZFN cut site in lowercase red CCATAACCCACCACAGCTAGAACTTATCAAACCCTtttgtGAAGATCTTGACCAATGGCTAAGTGAAGATGACAATCATG wt CCATAACCCACCACAGCTAGAACTTATC TTGACCAATGGCTAAGTGAAGATGACAATCATG -19 CCATAACCCACCACAGCTAGAACTTATCAAACCC- - ttgtgaagatcttgaccaatggctaagtgaagatgacaatcatg -2 22
22 PTEN Knockout with ZFN ELISA assay PTEN in wild type vs. knockout cell line luminus Conc. Lysate ug/ml Cytolysis of MCF10A vs. PTEN Knockout Targets % Cytolysis Ratio CTL to Target 23
23 Targeted Integration with Zinc Finger Nucleases xxxdonor Plasmid 24
24 Endogenous Tagging of TUBA1B Treatment of RFP-tagged TUBA1B MCF10A with paclitaxel - Paclitaxel, 0 min + Paclitaxel, 3 min + Paclitaxel, 26 min + Paclitaxel, 57 min + Paclitaxel, 98 min + Paclitaxel, 165 min Wild Type Clone RFP Clone 25
25 Multiple Modifications in the Same Cell BFP-Lamin RFP-Actin Overlay GFP-Tubulin DIC Sigma R&D 26
26 CRISPR Nickase System for Enhanced Specificity D10A 27
27 Cas9-D10A paired Nickase Configurations Nicking Cas9 Nicking Cas9 3 grna1 CUCCGCCCCACCUCCCCCAG-5 5 CCGGAGGCGGGGTGGAGGGGGTCGGGGCTCGCGGCGTCGCACTGAAACTTTTCGTCCAACTTCTGGGCTGTTCTCGCTTCGG < PAM spacing = 76 bp > (recommended limits = 30 to 150 bp) 3 GGCCTCCGCCCCACCTCCCCCAGCCCCGAGCGCCGCAGCGTGACTTTGAAAAGCAGGTTGAAGACCCGACAAGAGCGAAGCC 5 -CUUCUGGGCUGUUCUCGCUU grna2 3 28
28 Reduced Off-Target Activity with Cas9 Nickase Cas9 nuclease D10A nickase sgrna as2 sgrna s Indel (%) On-target Off-target Paired Cas9 nickase eliminates detectable modification at off-target location Sigma-Aldrich, unpublished data 29
29 Agenda Basic Introduction to Genome Editing Tools Introduction to Nucleofector Technology Application Examples TALEN-based Genome Editing ZFN- and CRIPSR-based Genome Editing (by Sigma-Aldrich) Technical Tips For ZFN/CRISPR (by Sigma-Aldrich) For Nucleofection Summary 30
30 Mismatch Detection Assay and Clone Screening General Guidelines when Deriving a Clone Modification <1%, do not single cell clone Modification = 1-3%, screen >300 clones (or use selection methods) Modification > 3%, screen at least 100 clones Percent of modified alleles in the transfected population 31
31 Enhancing ZFN Cutting Efficiency Using Cold Shock Culturing Doyon, Y., et al., Nature Methods 7, (2010) 32
32 Sorting for a Pool With Increased Activity 33
33 Targeted Integration of a MCS to AAVS1 Site in 11 Cell Lines Kb M ZFN DNA + Donor WT TI Donor alone WT 1. A DU HCT HEK HeLa 6. HepG2 7. IMR90 8. K LNCap 10. MCF7 11. U-2OS 34
34 ZFN-directed DNA Integration Rates Vary Between Cell Lines TI of a 50 bp MCS 1 TI of 3 kb 2 TI of 5 kb 2 Cell Lines Nucleofection Electroporation Lipid Nucleofection Nucleofection K562 30% 40% 20% HEK % 10% 6% HeLa 3% 5% 10% A % 10-20% 10% 1.4% 0.3% MCF7 3-5% ~5% HCT % 10-20% 3.3%*/ 5.2% 0.4%*/ <0.35% U-2 OS 30% IMR 90 ~5% LNCap 5-10% DU % HepG2 5-10% 1) Pool analysis 2) Single cell cloning * In this experiment, transfection efficiency was only around 20% 35
35 Agenda Basic Introduction to Genome Editing Tools Introduction to Nucleofector Technology Application Examples TALEN-based Genome Editing ZFN- and CRIPSR-based Genome Editing (by Sigma-Aldrich) Technical Tips For ZFN/CRISPR (by Sigma-Aldrich) For Nucleofection Summary 36
36 Nucleofection Tips and Tricks Optimal Nucleofection Conditions Depending on the cell type, first verify optimal conditions (given in the ready-to-use protocol) using pmaxgfp Control or determine optimal conditions (e.g. for ipsc clones) 37
37 Nucleofection Tips and Tricks Co-Transfection Optimize total substrate amount and ratio Example ranges from publications: ZFN Plasmid mrna Donor Plasmid: µg/µl each µg/µl each µg/µl TALEN Plasmid Donor Plasmid: Donor dsdna (lin) Donor ssodn µg/µl each µg/µl 0.1 µg/µl 10 µm CRISPR Plasmid grna PCR Cassette Donor dsdna (lin) Donor ssodn Cas9: µg/µl grna: µg/µl 0.5 ng/µl µg/µl µm Cas9/gRNA: µg/µl 38
38 Nucleofection Tips and Tricks Nucleofection of mrna Follow the same protocol and use the same program that is used for the transfection of DNA with your cells The mrna should be capped and poly-adenylated Optimal amount has to be titrated, might be higher than for plasmid 2 µg to 20 µg of each When higher amount is required the volume should not exceed 10% of the total sample volume Depending on the cell type (e.g. for DCs) it might be best to add the mrna directly into the empty cuvettes and then add the cell-solution mix on top. After adding the cell suspension transfect the sample immediately 39
39 Agenda Basic Introduction to Genome Editing Tools Introduction to Nucleofector Technology Application Examples TALEN-based Genome Editing ZFN- and CRIPSR-based Genome Editing (by Sigma-Aldrich) Technical Tips For ZFN/CRISPR (by Sigma-Aldrich) For Nucleofection Summary 40
40 Summary Nucleofection for Genome Editing Non-viral technology allows easy adaptation to different genome editing systems, be it ZFN, TALEN, or CRISPR Substrate flexibility and simple co-transfection due to same conditions for various substrates A reliable, proven technology, with > 30 genome editing publications and used by renowned players in the field Successful combination of ZFN and CRISPR tools from Sigma Aldrich with Nucleofector Technology for the generation of genetically modified cell lines 41
41 Sources of Information For Nucleofector Technology Our Online Databases Citations: Cell Transfection: Optimized Protocols: Transfection Experts Scientific Support Team EU: Scientific Support Team US: (toll free) For more information visit 42
42 Sources of Information For Genome Editing Tools Sigma-Aldrich Genome Editing Information Zinc Finger Nucleases: CRISPRs: Technical Experts ZFN Support: CRISPR Support: 43
43 Questions & Answers To submit a question: Please use the Questions feature located in your control panel, which you can access by clicking on the orange arrow on the right hand side of your screen. 44
44 Interested in Learning More? Do not miss Lonza s 2015 webinars series! Subscribe to our BioResearch Webinar Information service and receive personal event invitations per Sign up for our Cell Culture or Transfection enews Follow us on LinkedIn 45
45 Thank You for Your Kind Attention Disclaimer: Lonza is not responsible for the content of the Sigma-Aldrich slides. Sigma-Aldrich is not responsible for the content of the Lonza slides. The views and opinions expressed by one company do not necessarily reflect or represent the views and opinions of the other.
Genome Editing using Nucleofector Technology Technical Reference Guide
 BioResearch Genome Editing using Nucleofect Technology Technical Reference Guide This guideline provides a brief background on various genome editing tools and describes how to establish Lonza s Nucleofect
BioResearch Genome Editing using Nucleofect Technology Technical Reference Guide This guideline provides a brief background on various genome editing tools and describes how to establish Lonza s Nucleofect
CRISPR/Cas9 Genome Editing: Transfection Methods
 CRISPR/ Genome Editing: Transfection Methods For over 20 years Mirus Bio has developed and manufactured high performance transfection products and technologies. That expertise is now being applied to the
CRISPR/ Genome Editing: Transfection Methods For over 20 years Mirus Bio has developed and manufactured high performance transfection products and technologies. That expertise is now being applied to the
Return to Web Version
 Page 1 of 7 Page 1 of 7 Return to Web Version ZFN Technology Biowire Volume 10 Article 1 Have your genomic work cut out for you The genomes of several organisms, including humans, have been sequenced,
Page 1 of 7 Page 1 of 7 Return to Web Version ZFN Technology Biowire Volume 10 Article 1 Have your genomic work cut out for you The genomes of several organisms, including humans, have been sequenced,
Genomic Editing, Nucleofection, and the Creation of Next-Generation Cell-Based Assays
 Genomic Editing, Nucleofection, and the Creation of Next-Generation Cell-Based Assays Gregory Alberts, Ph.D. Global Subject Matter Expert Lonza Pharma Bioscience Solutions Content Nucleofection and Next-Generation
Genomic Editing, Nucleofection, and the Creation of Next-Generation Cell-Based Assays Gregory Alberts, Ph.D. Global Subject Matter Expert Lonza Pharma Bioscience Solutions Content Nucleofection and Next-Generation
Applications For CRISPR-Cas9 Stable Cell Lines
 Applications For CRISPR-Cas9 Stable Cell Lines Presenter: March 22, 2017 Ed Davis, Ph.D. Senior Application Scientist GeneCopoeia, Inc. GeneCopoeia products & services Functional Genomics & Cell Biology
Applications For CRISPR-Cas9 Stable Cell Lines Presenter: March 22, 2017 Ed Davis, Ph.D. Senior Application Scientist GeneCopoeia, Inc. GeneCopoeia products & services Functional Genomics & Cell Biology
Genome Engineering with ZFNs, TALENs and CRISPR/Cas9
 Genome Engineering with ZFNs, TALENs and CRISPR/Cas9 Designer Endonucleases ZFNs (zinc finger nucleases), TALENs (transcription activator-like effector nucleases) and CRISPR/Cas9 (clustered regularly interspaced
Genome Engineering with ZFNs, TALENs and CRISPR/Cas9 Designer Endonucleases ZFNs (zinc finger nucleases), TALENs (transcription activator-like effector nucleases) and CRISPR/Cas9 (clustered regularly interspaced
Genome editing. Knock-ins
 Genome editing Knock-ins Experiment design? Should we even do it? In mouse or rat, the HR-mediated knock-in of homologous fragments derived from a donor vector functions well. However, HR-dependent knock-in
Genome editing Knock-ins Experiment design? Should we even do it? In mouse or rat, the HR-mediated knock-in of homologous fragments derived from a donor vector functions well. However, HR-dependent knock-in
CRISPR/Cas9 Gene Editing Tools
 CRISPR/Cas9 Gene Editing Tools - Separations Simply Spectacular INDELS Identify indels Determine if one or both copies of your gene have indels The Guide-it Genotype Confirmation Kit: Simple detection
CRISPR/Cas9 Gene Editing Tools - Separations Simply Spectacular INDELS Identify indels Determine if one or both copies of your gene have indels The Guide-it Genotype Confirmation Kit: Simple detection
Transfection of CRISPR/Cas9 Nuclease NLS ribonucleoprotein (RNP) into adherent mammalian cells using Lipofectamine RNAiMAX
 Transfection of CRISPR/Cas9 Nuclease NLS ribonucleoprotein (RNP) into adherent mammalian cells using Lipofectamine RNAiMAX INTRODUCTION The CRISPR/Cas genome editing system consists of a single guide RNA
Transfection of CRISPR/Cas9 Nuclease NLS ribonucleoprotein (RNP) into adherent mammalian cells using Lipofectamine RNAiMAX INTRODUCTION The CRISPR/Cas genome editing system consists of a single guide RNA
Using CRISPR for genetic alteration
 Using CRISPR for genetic alteration Joffrey Mianné. j.mianne@har.mrc.ac.uk Mary Lyon Centre, MRC Harwell. CRISPR/Cas origins Origin of the CRISPR/Cas system: Clustered-Regularly Interspaced Short Palindromic
Using CRISPR for genetic alteration Joffrey Mianné. j.mianne@har.mrc.ac.uk Mary Lyon Centre, MRC Harwell. CRISPR/Cas origins Origin of the CRISPR/Cas system: Clustered-Regularly Interspaced Short Palindromic
User Instructions:Transfection-ready CRISPR/Cas9 Reagents. Target DNA. NHEJ repair pathway. Nucleotide deletion. Nucleotide insertion Gene disruption
 User Instructions:Transfection-ready CRISPR/Cas9 Reagents Background Introduction to CRISPR/Cas9 genome editing In bacteria and archaea, clustered regularly interspaced short palindromic repeats (CRISPR)
User Instructions:Transfection-ready CRISPR/Cas9 Reagents Background Introduction to CRISPR/Cas9 genome editing In bacteria and archaea, clustered regularly interspaced short palindromic repeats (CRISPR)
Amplicons, Heteroduplexes and Enzymes - Proper Processing Elevates Detection of CRISPR Gene Editing Events
 Amplicons, Heteroduplexes and Enzymes - Proper Processing Elevates Detection of CRISPR Gene Editing Events Steve Siembieda, MS MBA VP Commercialization ABRF Conference February 2015 What Is CRISPR? Clustered
Amplicons, Heteroduplexes and Enzymes - Proper Processing Elevates Detection of CRISPR Gene Editing Events Steve Siembieda, MS MBA VP Commercialization ABRF Conference February 2015 What Is CRISPR? Clustered
CRISPR/Cas9 Gene Editing Tools
 CRISPR/Cas9 Gene Editing Tools - Guide-it Products for Successful CRISPR/Cas9 Gene Editing - Why choose Guide-it products? Optimized methods designed for speed and ease of use Complete kits that don t
CRISPR/Cas9 Gene Editing Tools - Guide-it Products for Successful CRISPR/Cas9 Gene Editing - Why choose Guide-it products? Optimized methods designed for speed and ease of use Complete kits that don t
Applications of Cas9 nickases for genome engineering
 application note genome editing Applications of Cas9 nickases for genome engineering Shuqi Yan, Mollie Schubert, Maureen Young, Brian Wang Integrated DNA Technologies, 17 Commercial Park, Coralville, IA,
application note genome editing Applications of Cas9 nickases for genome engineering Shuqi Yan, Mollie Schubert, Maureen Young, Brian Wang Integrated DNA Technologies, 17 Commercial Park, Coralville, IA,
Genome Editing: Cas9 Stable Cell Lines for CRISPR sgrna Validation, Library Screening, and More. Ed Davis, Ph.D.
 TECHNICAL NOTE Genome Editing: Cas9 Stable Cell Lines for CRISPR sgrna Validation, Library Screening, and More Introduction Ed Davis, Ph.D. The CRISPR-Cas9 system has become greatly popular for genome
TECHNICAL NOTE Genome Editing: Cas9 Stable Cell Lines for CRISPR sgrna Validation, Library Screening, and More Introduction Ed Davis, Ph.D. The CRISPR-Cas9 system has become greatly popular for genome
CRISPR RNA-guided activation of endogenous human genes
 CRISPR RNA-guided activation of endogenous human genes Morgan L Maeder, Samantha J Linder, Vincent M Cascio, Yanfang Fu, Quan H Ho, J Keith Joung Supplementary Figure 1 Comparison of VEGF activation induced
CRISPR RNA-guided activation of endogenous human genes Morgan L Maeder, Samantha J Linder, Vincent M Cascio, Yanfang Fu, Quan H Ho, J Keith Joung Supplementary Figure 1 Comparison of VEGF activation induced
Introduction to CRISPR/Cas9 Background DNA Cleavage and Repair (NHEJ and HDR) Alternative Cas9 Variants Delivery of Cas9 and sgrna Library Products
 Introduction to CRISPR/Cas9 Background DNA Cleavage and Repair (NHEJ and HDR) Alternative Cas9 Variants Delivery of Cas9 and sgrna Library Products which one is right for you? CRISPR Workflow abm s Toolbox
Introduction to CRISPR/Cas9 Background DNA Cleavage and Repair (NHEJ and HDR) Alternative Cas9 Variants Delivery of Cas9 and sgrna Library Products which one is right for you? CRISPR Workflow abm s Toolbox
Easi CRISPR for conditional and insertional alleles
 Easi CRISPR for conditional and insertional alleles C.B Gurumurthy, University Of Nebraska Medical Center Omaha, NE cgurumurthy@unmc.edu Types of Genome edits Gene disruption/inactivation Types of Genome
Easi CRISPR for conditional and insertional alleles C.B Gurumurthy, University Of Nebraska Medical Center Omaha, NE cgurumurthy@unmc.edu Types of Genome edits Gene disruption/inactivation Types of Genome
CRISPRseek Workshop Design of target-specific guide RNAs in CRISPR-Cas9 genome-editing systems
 April 2008 CRISPRseek Workshop Design of target-specific guide RNAs in CRISPR-Cas9 genome-editing systems Lihua Julie Zhu August 1st 2014 Outline Background and Motives CRISPRseek Functionality Dependency
April 2008 CRISPRseek Workshop Design of target-specific guide RNAs in CRISPR-Cas9 genome-editing systems Lihua Julie Zhu August 1st 2014 Outline Background and Motives CRISPRseek Functionality Dependency
Effect of Different CRISPR/Cas9 Variants on Repair Pathway Choice
 Effect of Different CRISPR/Cas9 Variants on Repair Pathway Choice Cecilia Cotta-Ramusino www.editasmedicine.com 1 Cas9 Stimulates the Endogenous Repair Pathways WT Cas9 DSB C-NHEJ Resection 3 Locus Unaltered
Effect of Different CRISPR/Cas9 Variants on Repair Pathway Choice Cecilia Cotta-Ramusino www.editasmedicine.com 1 Cas9 Stimulates the Endogenous Repair Pathways WT Cas9 DSB C-NHEJ Resection 3 Locus Unaltered
Introduction and History of Genome Modification. Adam Clore, PhD Director, Synthetic Biology Design
 Introduction and History of Genome Modification Adam Clore, PhD Director, Synthetic Biology Design Early Non-site Directed Genome Modification Homologous recombination in yeast TARGET GENE 5 Arm URA3 3
Introduction and History of Genome Modification Adam Clore, PhD Director, Synthetic Biology Design Early Non-site Directed Genome Modification Homologous recombination in yeast TARGET GENE 5 Arm URA3 3
Application Note: Generating GFP-Tagged Human CD81 Tetraspanin Protein Using SBI s PrecisionX SmartNuclease System And HR Tagging Vectors
 Application Note: Generating GFP-Tagged Human CD81 Tetraspanin Protein Using SBI s PrecisionX SmartNuclease System And HR Tagging Vectors Table of Contents: I. Background Pg. 1 II. Analysis of Gene Target
Application Note: Generating GFP-Tagged Human CD81 Tetraspanin Protein Using SBI s PrecisionX SmartNuclease System And HR Tagging Vectors Table of Contents: I. Background Pg. 1 II. Analysis of Gene Target
Genome edi3ng with the CRISPR-Cas9 system
 CRISPR-Cas9 Genome Edi3ng Bootcamp AHA Council on Func3onal Genomics and Transla3onal Biology Narrated video link: hfps://youtu.be/h18hmftybnq Genome edi3ng with the CRISPR-Cas9 system Kiran Musunuru,
CRISPR-Cas9 Genome Edi3ng Bootcamp AHA Council on Func3onal Genomics and Transla3onal Biology Narrated video link: hfps://youtu.be/h18hmftybnq Genome edi3ng with the CRISPR-Cas9 system Kiran Musunuru,
IMPROVEMENT OF CRISPR GENE EDITING EFFICIENCY AND BEYONDS
 IMPROVEMENT OF CRISPR GENE EDITING EFFICIENCY AND BEYONDS YONGLUN LUO (ALUN) ALUN@BIOMED.AU.DK VIB, NOV. 21. 2017 Associate Professor, Department of Biomedicine, Aarhus University, Denmark Executive Director,
IMPROVEMENT OF CRISPR GENE EDITING EFFICIENCY AND BEYONDS YONGLUN LUO (ALUN) ALUN@BIOMED.AU.DK VIB, NOV. 21. 2017 Associate Professor, Department of Biomedicine, Aarhus University, Denmark Executive Director,
A Guide to CRISPR/Cas9
 Genome editing and beyond freepik A Guide to CRISPR/Cas9 The latest advance in genomic DNA editing is the Clustered Regularly Interspaced Short Palindromic Repeat (CRISPR)/Cas9 system. This simple-touse
Genome editing and beyond freepik A Guide to CRISPR/Cas9 The latest advance in genomic DNA editing is the Clustered Regularly Interspaced Short Palindromic Repeat (CRISPR)/Cas9 system. This simple-touse
New Plant Breeding Technologies
 New Plant Breeding Technologies Ricarda A. Steinbrecher, PhD EcoNexus / ENSSER Berlin, 07 May 2015 r.steinbrecher@econexus.info distributed by EuropaBio What are the NPBTs? *RNAi *Epigenetic alterations
New Plant Breeding Technologies Ricarda A. Steinbrecher, PhD EcoNexus / ENSSER Berlin, 07 May 2015 r.steinbrecher@econexus.info distributed by EuropaBio What are the NPBTs? *RNAi *Epigenetic alterations
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 ATP1A1 variants with in-frame deletions are enriched in ouabain-resistant cell populations. (a) Total editing efficacy along with spectrum and frequency of individual indels as determined
Supplementary Figure 1 ATP1A1 variants with in-frame deletions are enriched in ouabain-resistant cell populations. (a) Total editing efficacy along with spectrum and frequency of individual indels as determined
Optimizing CRISPR ribonucleoprotein components for precision genome editing
 Optimizing CRISPR ribonucleoprotein components for precision genome editing Justin Barr Sr. Product Manager, Integrated DNA Technologies VIB CRISPR User Meeting 18 September 2018 1 Agenda Overview of CRISPR-Cas9
Optimizing CRISPR ribonucleoprotein components for precision genome editing Justin Barr Sr. Product Manager, Integrated DNA Technologies VIB CRISPR User Meeting 18 September 2018 1 Agenda Overview of CRISPR-Cas9
Simple protocol for gene editing using GenCrisprTM Cas9 nuclease
 Simple protocol for gene editing using GenCrisprTM Cas9 nuclease Contents Protocol Step 1: Choose the target DNA sequence Step 2: Design sgrna Step 3: Preparation for sgrna 3.1 In vitro transcription of
Simple protocol for gene editing using GenCrisprTM Cas9 nuclease Contents Protocol Step 1: Choose the target DNA sequence Step 2: Design sgrna Step 3: Preparation for sgrna 3.1 In vitro transcription of
Getting started with CRISPR: A review of gene knockout and homology-directed repair
 Getting started with CRISPR: A review of gene knockout and homology-directed repair Justin Barr Product Manager, Functional Genomics Integrated DNA Technologies 1 Agenda: Getting started with CRISPR Background
Getting started with CRISPR: A review of gene knockout and homology-directed repair Justin Barr Product Manager, Functional Genomics Integrated DNA Technologies 1 Agenda: Getting started with CRISPR Background
Genome Editing with Programmable Nucleases. Jin-Soo Kim Department of Chemistry Seoul National University
 Genome Editing with Programmable Nucleases Jin-Soo Kim Department of Chemistry Seoul National University 1 Method of the Year 2011: Engineered Nucleases RNA-guided Cas9 Endonuclease 3 FokI and the First
Genome Editing with Programmable Nucleases Jin-Soo Kim Department of Chemistry Seoul National University 1 Method of the Year 2011: Engineered Nucleases RNA-guided Cas9 Endonuclease 3 FokI and the First
PLNT2530 (2018) Unit 9. Genome Editing
 PLNT2530 (2018) Unit 9 Genome Editing Unless otherwise cited or referenced, all content of this presenataion is licensed under the Creative Commons License Attribution Share-Alike 2.5 Canada 1 Genome Editing
PLNT2530 (2018) Unit 9 Genome Editing Unless otherwise cited or referenced, all content of this presenataion is licensed under the Creative Commons License Attribution Share-Alike 2.5 Canada 1 Genome Editing
Testing Non-Transgenic CRISPR Technology for Wheat Improvement 13 TH IWGS - TULLN, AUSTRIA
 Testing Non-Transgenic CRISPR Technology for Wheat Improvement KALI M BRANDT, HILARY L GUNN, BRETT L BUSCHKE, ADAM HEESACKER, NATHALIA MORET TI, ALEXANDER KARASEV, ROBERT S ZEMETRA 13 TH IWGS - TULLN,
Testing Non-Transgenic CRISPR Technology for Wheat Improvement KALI M BRANDT, HILARY L GUNN, BRETT L BUSCHKE, ADAM HEESACKER, NATHALIA MORET TI, ALEXANDER KARASEV, ROBERT S ZEMETRA 13 TH IWGS - TULLN,
NEXT-GENERATION METHODS AND TOOLS FOR PLANT GENOME EDITING. Tomáš Čermák VIB CRISPR-CAS User meeting September 18th 2018
 NEXT-GENERATION METHODS AND TOOLS FOR PLANT GENOME EDITING Tomáš Čermák VIB CRISPR-CAS User meeting September 18th 2018 Outline 1. How can we edit plant genomes? 2. What are the tools we can use? 3. How
NEXT-GENERATION METHODS AND TOOLS FOR PLANT GENOME EDITING Tomáš Čermák VIB CRISPR-CAS User meeting September 18th 2018 Outline 1. How can we edit plant genomes? 2. What are the tools we can use? 3. How
Designing CRISPR mediated Gene disruptions with gblocks Gene Fragments
 Designing CRISPR mediated Gene disruptions with gblocks Gene Fragments Adam Clore, PhD Manager, Synthetic Biology Design Integrated DNA technology Typical CRISPR timeline in Mammalian cell lines Design
Designing CRISPR mediated Gene disruptions with gblocks Gene Fragments Adam Clore, PhD Manager, Synthetic Biology Design Integrated DNA technology Typical CRISPR timeline in Mammalian cell lines Design
Efficient generation of conditional knockout mice by CLICK
 Efficient generation of conditional knockout mice by CLICK Tomoji Mashimo Genome Editing Research and Development (R&D) Center and Institute of Experimental Animal Sciences, Graduate School of Medicine,
Efficient generation of conditional knockout mice by CLICK Tomoji Mashimo Genome Editing Research and Development (R&D) Center and Institute of Experimental Animal Sciences, Graduate School of Medicine,
Genome editing as a new powerful tool for wheat breeding
 Genome editing as a new powerful tool for wheat breeding Vladimir Nekrasov 15 th WGIN Stakeholders Meeting Applying CRISPR Cas9 technology in model and crop plants Model plant: Crop plants: Nicotiana
Genome editing as a new powerful tool for wheat breeding Vladimir Nekrasov 15 th WGIN Stakeholders Meeting Applying CRISPR Cas9 technology in model and crop plants Model plant: Crop plants: Nicotiana
Lentiviral CRISPR guild RNA Cloning Kit for constructing CRISPR targeting grna lentivectors
 Lentiviral CRISPR guild RNA Cloning Kit for constructing CRISPR targeting grna lentivectors Cat# Product Name Amount Application grna-h1-gb grna-h1-gp grna-h1-rb grna-h1-rp grna-h1-puro grna-h1-bsd grna-u6-gb
Lentiviral CRISPR guild RNA Cloning Kit for constructing CRISPR targeting grna lentivectors Cat# Product Name Amount Application grna-h1-gb grna-h1-gp grna-h1-rb grna-h1-rp grna-h1-puro grna-h1-bsd grna-u6-gb
Optimized, chemically-modified crrna:tracrrna complexes for CRISPR gene editing
 Optimized, chemically-modified crrna:tracrrna complexes for CRISPR gene editing Mark Behlke MD, PhD Chief Scientific Officer February 24, 2016 1 Implementing CRISPR/Cas9 gene editing 2 To focus on RNA
Optimized, chemically-modified crrna:tracrrna complexes for CRISPR gene editing Mark Behlke MD, PhD Chief Scientific Officer February 24, 2016 1 Implementing CRISPR/Cas9 gene editing 2 To focus on RNA
ksierzputowska.com Research Title: Using novel TALEN technology to engineer precise mutations in the genome of D. melanogaster
 Research Title: Using novel TALEN technology to engineer precise mutations in the genome of D. melanogaster Research plan: Specific aims: 1. To successfully engineer transgenic Drosophila expressing TALENs
Research Title: Using novel TALEN technology to engineer precise mutations in the genome of D. melanogaster Research plan: Specific aims: 1. To successfully engineer transgenic Drosophila expressing TALENs
Eric Paul Bennett, MSc, Dr.med. Cellular Indel Profiles and Dynamics CRISPR/Cas9 delivery formats: Applying in vitro methodologies ex vivo
 Copenhagen Center for Glycomics/Department of Odontology Eric Paul Bennett, MSc, Dr.med. Cellular Indel Profiles and Dynamics CRISPR/Cas9 delivery formats: Applying in vitro methodologies ex vivo Eric
Copenhagen Center for Glycomics/Department of Odontology Eric Paul Bennett, MSc, Dr.med. Cellular Indel Profiles and Dynamics CRISPR/Cas9 delivery formats: Applying in vitro methodologies ex vivo Eric
Supplementary Information. Optimization of the production of knock-in alleles by CRISPR/Cas9 microinjection into the mouse zygote
 Supplementary Information Supplementary Figures S1 to S5 and Tables S1 to S3. Optimization of the production of knock-in alleles by CRISPR/Cas9 microinjection into the mouse zygote Aurélien Raveux, Sandrine
Supplementary Information Supplementary Figures S1 to S5 and Tables S1 to S3. Optimization of the production of knock-in alleles by CRISPR/Cas9 microinjection into the mouse zygote Aurélien Raveux, Sandrine
CRISPR: A Simple Tool for Answering Complex Questions
 CRISPR: A Simple Tool for Answering Complex Questions www.bcm.edu/cbass c_bass@bcm.edu 713-798-8987 CRISPR-Cas systems CRISPRs: Clustered Regularly Interspaced Short Palindromic Repeats Cas: The accompanying
CRISPR: A Simple Tool for Answering Complex Questions www.bcm.edu/cbass c_bass@bcm.edu 713-798-8987 CRISPR-Cas systems CRISPRs: Clustered Regularly Interspaced Short Palindromic Repeats Cas: The accompanying
CRISPR: hot, hot, hot
 CRISPR: hot, hot, hot 166 CRISPR is the latest technique for genome engineering and is generating tons of excitement due to its versatility, high specificity, and ease of use. CRISPR stands for clustered
CRISPR: hot, hot, hot 166 CRISPR is the latest technique for genome engineering and is generating tons of excitement due to its versatility, high specificity, and ease of use. CRISPR stands for clustered
CRISPR/Cas9: Tools and Applications for Eukaryotic Genome Editing
 CRISPR/Cas9: Tools and Applications for Eukaryotic Genome Editing Fei Ann Ran Broad Institute Cambridge, Massachusetts ran@fas.harvard.edu I will provide some background on the CRISPR/Cas9 technology,
CRISPR/Cas9: Tools and Applications for Eukaryotic Genome Editing Fei Ann Ran Broad Institute Cambridge, Massachusetts ran@fas.harvard.edu I will provide some background on the CRISPR/Cas9 technology,
Guide-it Indel Identification Kit User Manual
 Clontech Laboratories, Inc. Guide-it Indel Identification Kit User Manual Cat. No. 631444 (120114) Clontech Laboratories, Inc. A Takara Bio Company 1290 Terra Bella Avenue, Mountain View, CA 94043, USA
Clontech Laboratories, Inc. Guide-it Indel Identification Kit User Manual Cat. No. 631444 (120114) Clontech Laboratories, Inc. A Takara Bio Company 1290 Terra Bella Avenue, Mountain View, CA 94043, USA
SUPPLEMENTARY INFORMATION
 Gene replacements and insertions in rice by intron targeting using CRISPR Cas9 Table of Contents Supplementary Figure 1. sgrna-induced targeted mutations in the OsEPSPS gene in rice protoplasts. Supplementary
Gene replacements and insertions in rice by intron targeting using CRISPR Cas9 Table of Contents Supplementary Figure 1. sgrna-induced targeted mutations in the OsEPSPS gene in rice protoplasts. Supplementary
Innovative Trait Development Tools in Plant Breeding will be Crucial for Doubling Global Agricultural Productivity by 2050
 Innovative Trait Development Tools in Plant Breeding will be Crucial for Doubling Global Agricultural Productivity by 2050 Greg Gocal, Ph.D., Senior Vice President, Research and Development CRISPR Precision
Innovative Trait Development Tools in Plant Breeding will be Crucial for Doubling Global Agricultural Productivity by 2050 Greg Gocal, Ph.D., Senior Vice President, Research and Development CRISPR Precision
Bart Williams, PhD Van Andel Research Center
 A History of Genome Editing in the Laboratory Implications for Translational Applications Bart Williams, PhD Van Andel Research Center Introduction by Matthew Denenberg, MD DeVos Childrens Hospital Disclosures:
A History of Genome Editing in the Laboratory Implications for Translational Applications Bart Williams, PhD Van Andel Research Center Introduction by Matthew Denenberg, MD DeVos Childrens Hospital Disclosures:
Generating stable cell lines expressing various Cas9 proteins from AAVS1 locus
 IGI Protocol prepared by Chong Park 1 Generating stable cell lines expressing various Cas9 proteins from AAVS1 locus This is modified from original protocol from the Conklin laboratory (Mandegar et al.
IGI Protocol prepared by Chong Park 1 Generating stable cell lines expressing various Cas9 proteins from AAVS1 locus This is modified from original protocol from the Conklin laboratory (Mandegar et al.
Improving CRISPR-Cas9 Gene Knockout with a Validated Guide RNA Algorithm
 Improving CRISPR-Cas9 Gene Knockout with a Validated Guide RNA Algorithm Anja Smith Director R&D Dharmacon, part of GE Healthcare Imagination at work crrna:tracrrna program Cas9 nuclease Active crrna is
Improving CRISPR-Cas9 Gene Knockout with a Validated Guide RNA Algorithm Anja Smith Director R&D Dharmacon, part of GE Healthcare Imagination at work crrna:tracrrna program Cas9 nuclease Active crrna is
Construct Design and Cloning Guide for Cas9-triggered homologous recombination
 Construct Design and Cloning Guide for Cas9-triggered homologous recombination Written by Dan Dickinson (ddickins@live.unc.edu) and last updated December 2013. Reference: Dickinson DJ, Ward JD, Reiner
Construct Design and Cloning Guide for Cas9-triggered homologous recombination Written by Dan Dickinson (ddickins@live.unc.edu) and last updated December 2013. Reference: Dickinson DJ, Ward JD, Reiner
CRISPR Design Considera1ons
 CRISPR Design Considera1ons Shifra Ben-Dor Bioinforma1cs Unit Life Sciences Core Facili1es Weizmann Ins1tute of Science June 2018 Jinek et al, elife 2013;2:e00471 Cradick et al Outline Define your
CRISPR Design Considera1ons Shifra Ben-Dor Bioinforma1cs Unit Life Sciences Core Facili1es Weizmann Ins1tute of Science June 2018 Jinek et al, elife 2013;2:e00471 Cradick et al Outline Define your
Targeted Gene Therapy in the Treatment of X-linked Hyper-IgM Syndrome. Caroline Kuo, MD Pediatric Allergy & Immunology Clinical Instructor
 Targeted Gene Therapy in the Treatment of X-linked Hyper-IgM Syndrome Caroline Kuo, MD Pediatric Allergy & Immunology Clinical Instructor 1 Disclosures None. Hyper-immunoglobulin M syndromes Heterogeneous
Targeted Gene Therapy in the Treatment of X-linked Hyper-IgM Syndrome Caroline Kuo, MD Pediatric Allergy & Immunology Clinical Instructor 1 Disclosures None. Hyper-immunoglobulin M syndromes Heterogeneous
XTN TALENs Site-Specific Nucleases
 Site-Specific Nucleases User Manual Transposagen Biopharmaceuticals Inc. 535 W. Second St. Suite L102 Lexington, KY 40508 info@transposagenbio.com 859-428-8561 Contents I. Introduction and Background...
Site-Specific Nucleases User Manual Transposagen Biopharmaceuticals Inc. 535 W. Second St. Suite L102 Lexington, KY 40508 info@transposagenbio.com 859-428-8561 Contents I. Introduction and Background...
XactEdit Cas9 Nuclease with NLS User Manual
 XactEdit Cas9 Nuclease with NLS User Manual An RNA-guided recombinant endonuclease for efficient targeted DNA cleavage Catalog Numbers CE1000-50K, CE1000-50, CE1000-250, CE1001-250, CE1001-1000 Table of
XactEdit Cas9 Nuclease with NLS User Manual An RNA-guided recombinant endonuclease for efficient targeted DNA cleavage Catalog Numbers CE1000-50K, CE1000-50, CE1000-250, CE1001-250, CE1001-1000 Table of
CRISPR/Cas9 Mouse Production
 CRISPR/Cas9 Mouse Production Emory Transgenic and Gene Targeting Core http://cores.emory.edu/tmc Tamara Caspary, Ph.D. Scientific Director Teresa Quackenbush --- Lab Operations and Communications Coordinator
CRISPR/Cas9 Mouse Production Emory Transgenic and Gene Targeting Core http://cores.emory.edu/tmc Tamara Caspary, Ph.D. Scientific Director Teresa Quackenbush --- Lab Operations and Communications Coordinator
Bi Lecture 3 Loss-of-function (Ch. 4A) Monday, April 8, 13
 Bi190-2013 Lecture 3 Loss-of-function (Ch. 4A) Infer Gene activity from type of allele Loss-of-Function alleles are Gold Standard If organism deficient in gene A fails to accomplish process B, then gene
Bi190-2013 Lecture 3 Loss-of-function (Ch. 4A) Infer Gene activity from type of allele Loss-of-Function alleles are Gold Standard If organism deficient in gene A fails to accomplish process B, then gene
Allele-specific locus binding and genome editing by CRISPR at the
 Supplementary Information Allele-specific locus binding and genome editing by CRISPR at the p6ink4a locus Toshitsugu Fujita, Miyuki Yuno, and Hodaka Fujii Supplementary Figure Legends Supplementary Figure
Supplementary Information Allele-specific locus binding and genome editing by CRISPR at the p6ink4a locus Toshitsugu Fujita, Miyuki Yuno, and Hodaka Fujii Supplementary Figure Legends Supplementary Figure
character design & illustration: ryuji nouguchi Special Edition
 character design & illustration: ryuji nouguchi Special Edition Hmm... my sirna experiment worked, and now I want to know more about this gene. Can I completely knock out its expression in cells? Hello
character design & illustration: ryuji nouguchi Special Edition Hmm... my sirna experiment worked, and now I want to know more about this gene. Can I completely knock out its expression in cells? Hello
Barley as a model for cereal engineering and genome editing. Wendy Harwood
 Barley as a model for cereal engineering and genome editing Wendy Harwood MonoGram 29 th April 2015 www.bract.org BRACT Transformation Platform Over-expression of single genes RNAi based silencing Promoter
Barley as a model for cereal engineering and genome editing Wendy Harwood MonoGram 29 th April 2015 www.bract.org BRACT Transformation Platform Over-expression of single genes RNAi based silencing Promoter
Protocols for cell lines using CRISPR/CAS
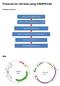 Protocols for cell lines using CRISPR/CAS Procedure overview Map Preparation of CRISPR/CAS plasmids Expression vectors for guide RNA (U6-gRNA) and Cas9 gene (CMV-p-Cas9) are ampicillin-resist ant and stable
Protocols for cell lines using CRISPR/CAS Procedure overview Map Preparation of CRISPR/CAS plasmids Expression vectors for guide RNA (U6-gRNA) and Cas9 gene (CMV-p-Cas9) are ampicillin-resist ant and stable
Humanized Cas9 endonuclease expression lentivirus for CRISPR. Cat# Product Name Amounts
 Humanized Cas9 endonuclease expression lentivirus for CRISPR Cat# Product Name Amounts LVP681 LVP681-PBS LVP682 LVP682-PBS LVP683 LVP683-PBS LVP678 LVP678-PBS LVP679 LVP679-PBS LVP680 LVP680-PBS LVP707
Humanized Cas9 endonuclease expression lentivirus for CRISPR Cat# Product Name Amounts LVP681 LVP681-PBS LVP682 LVP682-PBS LVP683 LVP683-PBS LVP678 LVP678-PBS LVP679 LVP679-PBS LVP680 LVP680-PBS LVP707
Experimental genetics - 2 Partha Roy
 Partha Roy Experimental genetics - 2 Making genetically altered animal 1) Gene knock-out k from: a) the entire animal b) selected cell-type/ tissue c) selected cell-type/tissue at certain time 2) Transgenic
Partha Roy Experimental genetics - 2 Making genetically altered animal 1) Gene knock-out k from: a) the entire animal b) selected cell-type/ tissue c) selected cell-type/tissue at certain time 2) Transgenic
Use of Gene Editing Technologies in Rodents. Carlisle P. Landel, Ph.D.
 Use of Gene Editing Technologies in Rodents Carlisle P. Landel, Ph.D. The Mouse as A Model Mammal Small, easy to maintain, fecund Well understood genetics Similarity to humans >90% Availability of inbred
Use of Gene Editing Technologies in Rodents Carlisle P. Landel, Ph.D. The Mouse as A Model Mammal Small, easy to maintain, fecund Well understood genetics Similarity to humans >90% Availability of inbred
Supplementary Figure 1. Nature Structural & Molecular Biology: doi: /nsmb.3494
 Supplementary Figure 1 Pol structure-function analysis (a) Inactivating polymerase and helicase mutations do not alter the stability of Pol. Flag epitopes were introduced using CRISPR/Cas9 gene targeting
Supplementary Figure 1 Pol structure-function analysis (a) Inactivating polymerase and helicase mutations do not alter the stability of Pol. Flag epitopes were introduced using CRISPR/Cas9 gene targeting
CRISPR/Cas9 engineering and bioinformatics. Leopold Parts 10/05/2018
 CRISPR/Cas9 engineering and bioinformatics Leopold Parts 10/05/2018 CRISPR/Cas9 engineering and bioinformatics CRISPR/Cas9 background Genome engineering in olden days: RE and HR DNA break repair as a basic
CRISPR/Cas9 engineering and bioinformatics Leopold Parts 10/05/2018 CRISPR/Cas9 engineering and bioinformatics CRISPR/Cas9 background Genome engineering in olden days: RE and HR DNA break repair as a basic
Protocol CRISPR Genome Editing In Cell Lines
 Protocol CRISPR Genome Editing In Cell Lines Protocol 2: HDR donor plasmid applications (gene knockout, gene mutagenesis, gene tagging, Safe Harbor ORF knock-in) Notes: 1. sgrna validation: GeneCopoeia
Protocol CRISPR Genome Editing In Cell Lines Protocol 2: HDR donor plasmid applications (gene knockout, gene mutagenesis, gene tagging, Safe Harbor ORF knock-in) Notes: 1. sgrna validation: GeneCopoeia
Alt-Ring the genome with CRISPR- Cas9
 Alt-Ring the genome with CRISPR- Cas9 Genome editing done easily and fast CHIA Jin Ngee Regional Application Specialist 1 Integrated DNA Technologies The Custom Biology Company 2 IDT was founded by a scientist
Alt-Ring the genome with CRISPR- Cas9 Genome editing done easily and fast CHIA Jin Ngee Regional Application Specialist 1 Integrated DNA Technologies The Custom Biology Company 2 IDT was founded by a scientist
CRISPR GENOMIC SERVICES PRODUCT CATALOG
 CRISPR GENOMIC SERVICES PRODUCT CATALOG DESIGN BUILD ANALYZE The experts at Desktop Genetics can help you design, prepare and manufacture all of the components needed for your CRISPR screen. We provide
CRISPR GENOMIC SERVICES PRODUCT CATALOG DESIGN BUILD ANALYZE The experts at Desktop Genetics can help you design, prepare and manufacture all of the components needed for your CRISPR screen. We provide
Genome Editing: Considerations for Therapeutic Applications Cecilia Fernandez TIDES,
 Genome Editing: Considerations for Therapeutic Applications Cecilia Fernandez TIDES, 2017 www.editasmedicine.com 1 The Potential to Repair Any Broken Gene Transformative new category of medicines for genetically-defined
Genome Editing: Considerations for Therapeutic Applications Cecilia Fernandez TIDES, 2017 www.editasmedicine.com 1 The Potential to Repair Any Broken Gene Transformative new category of medicines for genetically-defined
Supplementary Figures and Figure legends
 Supplementary Figures and Figure legends 3 Supplementary Figure 1. Conditional targeting construct for the murine Satb1 locus with a modified FLEX switch. Schematic of the wild type Satb1 locus; the conditional
Supplementary Figures and Figure legends 3 Supplementary Figure 1. Conditional targeting construct for the murine Satb1 locus with a modified FLEX switch. Schematic of the wild type Satb1 locus; the conditional
Cell line CDH1 mrna AS-paRNA S-paRNA
 a b Cell line mrna AS-paRNA S-paRNA HCT116 P53 +/+ 39.47 + + HCT116 P53 -/- 35.36 + + LNCAP 1562.4 + - MCF7 C1 1864.76 + - MCF7 C2 1812.81 + - Supplementary Figure 1. Promoter-associated transcripts at
a b Cell line mrna AS-paRNA S-paRNA HCT116 P53 +/+ 39.47 + + HCT116 P53 -/- 35.36 + + LNCAP 1562.4 + - MCF7 C1 1864.76 + - MCF7 C2 1812.81 + - Supplementary Figure 1. Promoter-associated transcripts at
New Plant Breeding Techniques Group 1 Targeted Mutagenesis
 WORKSHOP COMPERATIVE SITUATION OF NEW PLANT BREEDING TECHNIQUES 12-13 SEPTEMBER 2011 SEVILLE, SPAIN New Plant Breeding Techniques Group 1 Targeted Mutagenesis Maria Lusser Joint Research Centre, European
WORKSHOP COMPERATIVE SITUATION OF NEW PLANT BREEDING TECHNIQUES 12-13 SEPTEMBER 2011 SEVILLE, SPAIN New Plant Breeding Techniques Group 1 Targeted Mutagenesis Maria Lusser Joint Research Centre, European
CRISPR Ribonucleoprotein (RNP) User Manual
 CRISPR Ribonucleoprotein (RNP) User Manual Description This user manual describes how to use GenScript s CRISPR Ribonucleoprotein (RNP) products for targeted genome editing. The RNP system is comprised
CRISPR Ribonucleoprotein (RNP) User Manual Description This user manual describes how to use GenScript s CRISPR Ribonucleoprotein (RNP) products for targeted genome editing. The RNP system is comprised
A Survey of Genetic Methods
 IBS 8102 Cell, Molecular, and Developmental Biology A Survey of Genetic Methods January 24, 2008 DNA RNA Hybridization ** * radioactive probe reverse transcriptase polymerase chain reaction RT PCR DNA
IBS 8102 Cell, Molecular, and Developmental Biology A Survey of Genetic Methods January 24, 2008 DNA RNA Hybridization ** * radioactive probe reverse transcriptase polymerase chain reaction RT PCR DNA
Transfection of neural stem cells with Lipofectamine Stem Transfection Reagent in StemPro medium
 TRANSFECTION PROTOCOL Lipofectamine Stem Transfection Reagent Transfection of neural stem cells with Lipofectamine Stem Transfection Reagent in StemPro medium NSC media, passaging reagents, and complexation
TRANSFECTION PROTOCOL Lipofectamine Stem Transfection Reagent Transfection of neural stem cells with Lipofectamine Stem Transfection Reagent in StemPro medium NSC media, passaging reagents, and complexation
SURVEY AND SUMMARY How to create state-of-the-art genetic model systems: strategies for optimal CRISPR-mediated genome editing
 Published online 28 June 2018 Nucleic Acids Research, 2018, Vol. 46, No. 13 6435 6454 doi: 10.1093/nar/gky571 SURVEY AND SUMMARY How to create state-of-the-art genetic model systems: strategies for optimal
Published online 28 June 2018 Nucleic Acids Research, 2018, Vol. 46, No. 13 6435 6454 doi: 10.1093/nar/gky571 SURVEY AND SUMMARY How to create state-of-the-art genetic model systems: strategies for optimal
Supplementary Information
 Supplementary Information Super-resolution imaging of fluorescently labeled, endogenous RNA Polymerase II in living cells with CRISPR/Cas9-mediated gene editing Won-Ki Cho 1, Namrata Jayanth 1, Susan Mullen
Supplementary Information Super-resolution imaging of fluorescently labeled, endogenous RNA Polymerase II in living cells with CRISPR/Cas9-mediated gene editing Won-Ki Cho 1, Namrata Jayanth 1, Susan Mullen
Mouse Engineering Technology. Musculoskeletal Research Center 2016 Summer Educational Series David M. Ornitz Department of Developmental Biology
 Mouse Engineering Technology Musculoskeletal Research Center 2016 Summer Educational Series David M. Ornitz Department of Developmental Biology Core service and new technologies Mouse ES core Discussions
Mouse Engineering Technology Musculoskeletal Research Center 2016 Summer Educational Series David M. Ornitz Department of Developmental Biology Core service and new technologies Mouse ES core Discussions
Surrogate reporter-based enrichment of cells containing RNA-guided Cas9 nucleaseinduced
 Supplementary Data Surrogate reporter-based enrichment of cells containing RNA-guided Cas9 nucleaseinduced mutations Suresh Ramakrishna 1, Seung Woo Cho 2, Sojung Kim 2, Myungjae Song 1, Ramu Gopalappa
Supplementary Data Surrogate reporter-based enrichment of cells containing RNA-guided Cas9 nucleaseinduced mutations Suresh Ramakrishna 1, Seung Woo Cho 2, Sojung Kim 2, Myungjae Song 1, Ramu Gopalappa
Amaxa Cell Line Nucleofector Kit L
 Amaxa Cell Line Nucleofector Kit L For 3T3-L1 (adipocytes) [ATCC CL-173, cryopreserved] Mouse embryonal fibroblast, differentiated into adipocytes; Fibroblast-like cells before differentiation; adipocyte-like
Amaxa Cell Line Nucleofector Kit L For 3T3-L1 (adipocytes) [ATCC CL-173, cryopreserved] Mouse embryonal fibroblast, differentiated into adipocytes; Fibroblast-like cells before differentiation; adipocyte-like
Cell Monitoring and Remote Recording: The CytoSMART System
 Pharma&Biotech BioResearch Cell Monitoring and Remote Recording: The CytoSMART System Webinar 22 September 2015 / Speaker: Sean Fuerst 23 September 2015 / Speaker: Dr. Claudia Schwartz Lonza Cologne GmbH,
Pharma&Biotech BioResearch Cell Monitoring and Remote Recording: The CytoSMART System Webinar 22 September 2015 / Speaker: Sean Fuerst 23 September 2015 / Speaker: Dr. Claudia Schwartz Lonza Cologne GmbH,
Comparison of Commercial Transfection Reagents: Cell line optimized transfection kits for in vitro cancer research.
 Comparison of Commercial Transfection Reagents: Cell line optimized transfection kits for in vitro cancer research. by Altogen Labs, 11200 Manchaca Road, Suite 203 Austin TX 78748 USA Tel. (512) 433-6177
Comparison of Commercial Transfection Reagents: Cell line optimized transfection kits for in vitro cancer research. by Altogen Labs, 11200 Manchaca Road, Suite 203 Austin TX 78748 USA Tel. (512) 433-6177
Inhibition of HSV-1 Replication by Gene Editing Strategy. Running Title: Targeting HSV-1 Infection by CRISPR/Cas9
 SUPPLEMENTARY MATERIALS 2/4/16 Inhibition of HSV-1 Replication by Gene Editing Strategy Running Title: Targeting HSV-1 Infection by CRISPR/Cas9 Pamela C. Roehm 1,2,3*, Masoud Shekarabi 1, Hassen Wollebo
SUPPLEMENTARY MATERIALS 2/4/16 Inhibition of HSV-1 Replication by Gene Editing Strategy Running Title: Targeting HSV-1 Infection by CRISPR/Cas9 Pamela C. Roehm 1,2,3*, Masoud Shekarabi 1, Hassen Wollebo
Protocol for tissue-specific gene disruption in zebrafish
 Protocol for tissue-specific gene disruption in zebrafish Overview This protocol describes a method to inactivate genes in zebrafish in a tissue-specific manner. It can be used to analyze mosaic loss-of-function
Protocol for tissue-specific gene disruption in zebrafish Overview This protocol describes a method to inactivate genes in zebrafish in a tissue-specific manner. It can be used to analyze mosaic loss-of-function
Characterization and Optimization of the CRISPR/Cas System for Applications in Genome Engineering
 Characterization and Optimization of the CRISPR/Cas System for Applications in Genome Engineering The Harvard community has made this article openly available. Please share how this access benefits you.
Characterization and Optimization of the CRISPR/Cas System for Applications in Genome Engineering The Harvard community has made this article openly available. Please share how this access benefits you.
Transfection of pluripotent stem cells with Lipofectamine Stem reagent in mtesr1 Medium on Geltrex matrix
 TRANSFECTION PROTOCOL Lipofectamine Stem Transfection Reagent Transfection of pluripotent stem cells with Lipofectamine Stem reagent in mtesr1 Medium on Geltrex matrix PSC growth medium, passaging reagents,
TRANSFECTION PROTOCOL Lipofectamine Stem Transfection Reagent Transfection of pluripotent stem cells with Lipofectamine Stem reagent in mtesr1 Medium on Geltrex matrix PSC growth medium, passaging reagents,
CRISPR Genome Editing: Considerations for Therapeutic Applications
 CRISPR Genome Editing: Considerations for Therapeutic Applications November 9, 2017 Cecilia Fernandez editasmedicine.com 1 Medicines that Aim to Repair Any Broken Gene Potential to create the next major
CRISPR Genome Editing: Considerations for Therapeutic Applications November 9, 2017 Cecilia Fernandez editasmedicine.com 1 Medicines that Aim to Repair Any Broken Gene Potential to create the next major
Incorporating SeqStudio Genetic Analyzer and Sanger sequencing into genome editing workflows
 Incorporating SeqStudio Genetic Analyzer and Sanger sequencing into genome editing workflows Stephen Jackson, Ph.D 27 May 2017 The world leader in serving science Key Applications for Genome Editing Research
Incorporating SeqStudio Genetic Analyzer and Sanger sequencing into genome editing workflows Stephen Jackson, Ph.D 27 May 2017 The world leader in serving science Key Applications for Genome Editing Research
Cationic lipid-mediated delivery of proteins enables efficient proteinbased genome editing in vitro and in vivo
 correction notice Nat. Biotechnol. 33, 73 8 (215) Cationic lipid-mediated delivery of proteins enables efficient proteinbased genome editing in vitro and in vivo John A Zuris, David B Thompson, Yilai Shu,
correction notice Nat. Biotechnol. 33, 73 8 (215) Cationic lipid-mediated delivery of proteins enables efficient proteinbased genome editing in vitro and in vivo John A Zuris, David B Thompson, Yilai Shu,
Protocol for cloning SEC-based repair templates using Gibson assembly and ccdb negative selection
 Protocol for cloning SEC-based repair templates using Gibson assembly and ccdb negative selection Written by Dan Dickinson (daniel.dickinson@austin.utexas.edu) and last updated January 2018. A version
Protocol for cloning SEC-based repair templates using Gibson assembly and ccdb negative selection Written by Dan Dickinson (daniel.dickinson@austin.utexas.edu) and last updated January 2018. A version
TRANSGENIC ANIMALS. -transient transfection of cells -stable transfection of cells. - Two methods to produce transgenic animals:
 TRANSGENIC ANIMALS -transient transfection of cells -stable transfection of cells - Two methods to produce transgenic animals: 1- DNA microinjection - random insertion 2- embryonic stem cell-mediated gene
TRANSGENIC ANIMALS -transient transfection of cells -stable transfection of cells - Two methods to produce transgenic animals: 1- DNA microinjection - random insertion 2- embryonic stem cell-mediated gene
PrecisionX T7 Cas9 SmartNuclease Vector System (Tagged and Untagged) Catalog#s CAS7xxA/R/G-1. User Manual
 PrecisionX T7 Cas9 SmartNuclease Vector System (Tagged and Untagged) Catalog#s CAS7xxA/R/G-1 User Manual Store at -20 C upon receipt A limited-use label license covers this product. By use of this product,
PrecisionX T7 Cas9 SmartNuclease Vector System (Tagged and Untagged) Catalog#s CAS7xxA/R/G-1 User Manual Store at -20 C upon receipt A limited-use label license covers this product. By use of this product,
Nature Biotechnology: doi: /nbt.4166
 Supplementary Figure 1 Validation of correct targeting at targeted locus. (a) by immunofluorescence staining of 2C-HR-CRISPR microinjected embryos cultured to the blastocyst stage. Embryos were stained
Supplementary Figure 1 Validation of correct targeting at targeted locus. (a) by immunofluorescence staining of 2C-HR-CRISPR microinjected embryos cultured to the blastocyst stage. Embryos were stained
Homology-directed repair with Dharmacon Edit-R CRISPR-Cas9 reagents and single-stranded DNA oligos
 APPLICATION NOTE Homology-directed repair with Dharmacon Edit-R CRISPR- reagents and single-stranded DNA oligos John A. Schiel, Eldon Chou, Maren Mayer, Emily M. Anderson, and Anja van Brabant Smith Horizon
APPLICATION NOTE Homology-directed repair with Dharmacon Edit-R CRISPR- reagents and single-stranded DNA oligos John A. Schiel, Eldon Chou, Maren Mayer, Emily M. Anderson, and Anja van Brabant Smith Horizon
Nature Protocols: doi: /nprot Supplementary Figure 1. Efficient genome editing in human pluripotent stem cells.
 Supplementary Figure 1 Efficient genome editing in human pluripotent stem cells. Our genome editing protocol was highly efficient in an additional pluripotent human cell line (CHB10) and at several different
Supplementary Figure 1 Efficient genome editing in human pluripotent stem cells. Our genome editing protocol was highly efficient in an additional pluripotent human cell line (CHB10) and at several different
GENOME EDITING OF HUMAN PLURIPOTENT STEM CELLS
 TECHNICAL BULLETIN GENOME EDITING OF HUMAN PLURIPOTENT STEM CELLS Using the ArciTect CRISPR-Cas9 System Introduction The ability of human embryonic stem (ES) and induced pluripotent stem (ips) cells to
TECHNICAL BULLETIN GENOME EDITING OF HUMAN PLURIPOTENT STEM CELLS Using the ArciTect CRISPR-Cas9 System Introduction The ability of human embryonic stem (ES) and induced pluripotent stem (ips) cells to
Supplementary Figures Montero et al._supplementary Figure 1
 Montero et al_suppl. Info 1 Supplementary Figures Montero et al._supplementary Figure 1 Montero et al_suppl. Info 2 Supplementary Figure 1. Transcripts arising from the structurally conserved subtelomeres
Montero et al_suppl. Info 1 Supplementary Figures Montero et al._supplementary Figure 1 Montero et al_suppl. Info 2 Supplementary Figure 1. Transcripts arising from the structurally conserved subtelomeres
A Practical Guide to CRISPR-Mediated Gene Tagging with a Bioluminescent Peptide
 A Practical Guide to CRISPR-Mediated Gene Tagging with a Bioluminescent Peptide Marie K. Schwinn, PhD Senior Research Scientist For Research Use Only. Not for use in diagnostic procedures. Why endogenous
A Practical Guide to CRISPR-Mediated Gene Tagging with a Bioluminescent Peptide Marie K. Schwinn, PhD Senior Research Scientist For Research Use Only. Not for use in diagnostic procedures. Why endogenous
