Lecture 17 Guest Lecturer this week. Prof. Greg Ravizza
|
|
|
- Norah Reed
- 6 years ago
- Views:
Transcription
1 Lecture 17 Guest Lecturer this week. Prof. Greg Ravizza Migrating Reservoirs in the Hydrologic Cycle - Inorganic solubility examples from Rain and River chemistry Migrating Reservoirs in the Hydrologic Cycle We study rain and river compositions to learn about: how the cumulative effects of terrigenous weathering are superimposed on local watershed rain compositions. rates for material movement through the hydrosphere rates at which the land masses of earth are eroded anthropogenic effects. 1
2 Rain Rain is the starting point composition for water in a given watershed. Rivers and ground water compositions in turn reflect weathering and biological processes, followed by anthropogenic effects. For both rain and river water, dissolved solutes reflect the abundance of different types of solids in the environments and their solubility, both of which are highly variable. Rain Why does rain form? The atmosphere becomes supersaturated in water vapor at local conditions of temperature and pressure 2
3 Rain water delivers new water to a watershed. Its composition reflects fresh and weathered rocks of the watershed. natural and non-natural particulates in the atmosphere local gas fluxes to the atmosphere Aerosols: Rain drops form by nucleation on the surface of very fine (:m size) particles in the atmosphere called aerosols, which partly or completely dissolve. Natural sources: Marine -sea salt Terrestrial -dust -soils -volcanic emissions -pollen/other plant materials -smoke non-natural (Anthropogenic) sources: industrial agricultural -particulates/soot -automobile emissions -bakeries/restaurants dust/soils -agricultural fertilizers Particulates/Aerosols are produced naturally by wind mobilization of fine components of terrigenous matter and by drying + mobilization of marine aerosols. Only the small particles (F) are transported significant distances from their source. Solid particulates contribute to rain in 2 distinct processes: a. rainout removal of particulates during droplet nucleation (occurs in clouds) b. washout removal of particulates as raindrops fall to the ground (occurs below 3
4 Surface Water Chemistry changes from rain to river: river water has: Average North American rain and river water compared: a huge increase in SiO 2 and HCO 3- (due to congruent dissolution of silicates and carbonates) a large increase in Ca (for same reasons) a relative deficit of K, due to retention in clays other ions have increased in concentration but their ratios to each other are similar to rain ratios. Particulate control on global and local rain composition Rain of dominantly marine or continental origins are distinct The mostly marine ions are: Cl - = Na + >Mg 2+ The mostly terrigenous ions are: K + > Ca 2+ > SO 2-4 > NO 2-3 = NH 4+, Al +3 = SiO 2 (aq) The presence of Al +3 or SiO 2 (aq) (both highly ph dependent) indicates continental origin for a rain water, but their proportions relative to other solutes are complicated, so they are not used to quantitatively source rain water. 4
5 Particulate control on global and local rain composition Marine aerosols formed from drying of sea water dominate rain compositions over the oceans, coasts and islands like Hawaii. These aerosols contain the major sea water ions in the same proportion as they occur in the oceans. The marine component is rained out progressively with distance from the coast. Compositions become more terrestrial towards the centers of large land masses as more ions are derived from terrigenous materials. Pollution in Rain After isolating the likely marine input, the remainder is considered to be a combination of: local terrigenous components anthropogenic components Both are dominated by local effects and can be perturbed by human activities, such as: land cultivation industrial activities general urbanization The Pollutive component can be: "more than normal" amounts of ions in otherwise natural proportions more ions in some other proportion. ions or molecules not naturally observed 5
6 Except for Ca, all the other major ions of sea water behave conservatively (elemental ratios are constant throughout the oceans). We use ions such as Cl - and Na +, to determine the amount of marine aerosol dissolved in rainwater, since they have almost no non-marine natural sources. These then are a good starting point for understanding the sources of other ions in a particular rain water. Note the similarities, particularly for Cl - /Na + ratio of various marine rains (ion concentrations are ratioed to Na +, although I prefer ratios to Cl - ). Each other constituent of rain can be compared to the Cl - content to determine excesses or deficits relative to marine components. Excesses or deficits can be understood by looking at: the proportion of the ion added the potential terrigenous particulate sources the governing solubility reactions involved considering non-marine anthropogenic sources of Cl - from industrial inputs and road salting in cold regions. [ion] (sea water ratio in g/g) x [Cl - ] (in weight units) = [ion] (from sea water) [Cl - ] [total ion] - [ion] (from sea water) = [ion] (non-marine) if [ion](non-marine) >1 there is an excess if [ion](non-marine) <1 there is a deficit (or excess Cl - ) 6
7 The strong rainout gradient for the marine component is easy to see in a contour map of Cl - concentration in rain over 1 year over the continental US. Na + distribution is more (next page) complicated. Comparing Na + /Cl - rain to Na + /Cl - seawater shows slight "excess" Na everywhere. Large Na excesses occur near regions where dust/soils/clays add Na + to the marine component. The farther inland one gets, the easier it is to produce "excess" Na since the absolute Cl - content is much lower. 7
8 The more complex continental US: [Ca 2+ ] map derives mostly from CaCO 3 and CaSO 4 dissolution. Lo Hi Hi Lo Calcium in US rain. After subtracting the small marine component using Ca/Cl sea water, the rest of the Ca is sourced by comparing Ca content, rain ph and SO CaCO 3 dissolution neutralizes rain acidity, raising ph. CaSO 4 dissolution does not affect ph. CaCO 3 is the dominant source of Ca in most continental rain waters. We can tell this because high [Ca 2+ ] is usually accompanied by slightly higher ph. Exceptions are in arid regions (e.g., southwestern US), where CaSO 4 is prevalent in dry lake beds and soils. CaSO 4 particles add Ca to rain without the base CO 3 2-, so that ph is not shifted as [Ca 2+ ] increases along with [SO 4 2- ]. We must take care to distinguish high sulfate from this process as opposed to sulfate that is high from SO 3 addition to produce "acid rain". 8
9 Rain summary: We can then understand the "starting" composition of water that enters a particular watershed by isolating marine, local terrigenous and anthropogenic components. The farther from the coast, the less marine component to the rain. Example from the Amazon Basin: Concentrations of all rain components drop with distance from the coast, reflecting rainout of marine particulates as storm clouds move inland. Terrigenous components such as SO 2-4 and Ca 2+ increase at various points further inland. The terrigenous component is dominated by local effects and can be perturbed by human activities (resulting in "more than normal" amounts of ions in otherwise natural proportions or more ions in some other proportion. Rivers Rivers reflect the net integrated effects over a watershed of meteoric water interactions with solids in that environment. Rivers transport material in suspended and dissolved forms. These two materials are necessarily directly related in a particular river. 9
10 TDS (dissolved solids) chemistry reflects input from: inorganic reactions biological processes various water sources -rain - ground water discharge - lake discharge Suspended load is mostly independent of watershed discharge rate. Instead it reflects: watershed basin relief (river gradient= height/ distance) Basin relief reflects average time since last local orogeny and local rock type. Younger and steeper mountains have rivers with mostly higher suspended load and have high erosion rates. watershed basin area rock type temperature TABLE 5.1 Major Rivers that Flow to the Sea, Listed in Order of Discharge Source: Berner and Berner, Global Environment, 1996 Annual Discharge Dissolved Solids (Tg/yr) Suspended Solids (Tg/yr) Dissolved/ Suspended ratio Drainage Area (10 6 km 2 ) River Location Water (km 3 /yr) 1. Amazon S. America Lots of Lots of Zaire (Congo) Africa water dissolved load Orinoco S. America Yangtze (Chiang) Asia (China) Brahmaputra Asia Mississippi N. America (400) Yenisei Asia (Russia) Lena Asia (Russia) Mekong Asia (Vietnam) Ganges Asia St. Lawrence N. America Parana S. America lrrawaddy Asia (Burma) Mackenzie N. America Columbia N. America (15) Indus Asia (India) (250) Red (Hungho) Asia (Vietnam) 123? 130? 0.12 Huanghe (Yellow) Asia (China) Note: Tributaries are excluded. Tg = 10 6 tons = g. Sources: Water and suspended solids from Milliman and Meade (1983) and Milliman and Syvitski (1992). Dissolved solids calculated from Table 5.7 and Pinet and Souriau (1988) (for the Irrawaddy). Suspended load values in parentheses indicate pre-dam values. Lots of sediment Not much sediment 10
11 Suspended load variations around the globe: Human effects on sediment load increased erosion due to agriculture increased erosion due to deforestation decreased load transport due to dams decreased load transport due bank stabilization increased surface runoff due to urbanization (which also decreases groundwater recharge). 11
12 Sources of major ions in river water Table 5.11 Sources of Major Elements in World River water (in Percent Actual Concentrations) source: Berner and Berner, Global Environment (1996) Atmos. Cyclic Weathering Element Salt Carbonates Silicates Evaporites a Pollution b Ca C (HCO - 3 ) <<1 61 c 37 c 0 2 Na Cl S (sulfate) 2 d d 54 Mg <<1 8 K SiO 2 (aq) << a. also includes NaCl from shales and thermal springs b. values from Meybeck (1979) except sulfate, which is based on a calculation given in the text c. for carbonates, 34% from calcite and doliomite and 27% from soil CO2; for silicates, all 37% from soil CO2; thus, total HCO3 - from soil (atmospheric) CO2 = 64% (see also Table 5.13) d. other sources of river sulfate: natural biogenic emissions to atmosphere delivered to land, 3%; volcanism, 8%; pyrite weathering, 11% The weathering and cyclic salt columns probably contain no real surprises to you (although cyclic salt estimates vary enormously in the literature) The pollution column may seem striking. These estimates rely on making a budget for riverine TDS from suspected natural sources, and then accounting for any "excesses" as pollutant inputs. Solids Controls on river composition Riverine Dissolved load reflects local precipitation climate the extent of local weathering ion solubility at various conditions: T ph TOC (total organic carbon). Suspended load reflects rock type climate the suspended load is mostly composed of: weathering-resistant minerals (which can contain both high and low solubility elements) low solubility elements in secondary minerals organic particulates 12
13 Climate dependency T affects rate of dissolution, elemental solubility and secondary mineral stability T and H 2 O affects the local biology (biomass and type) Riverine TOC increases as biomass increases Type affects depth of physical weathering by roots, burrows, etc.. Climate dependency example: Weathering of the same silicate rock will create in a watershed: tropical climate: much higher suspended load proportions of Al and Fe to other elements due to more intense rock weathering and incongruent dissolution. temperate climate: much greater relative amounts of Ca, Mg, K, Na to Al and Fe in s-load because weathering is less intense Rock Types Effect on Riverine TDS: sedimentary rock > volcanic rock > crystalline rock (intrusive and metamorphic) Note: Chemical Denudation Rate is a measure of TDS flux per unit time out of a watershed. Greater runoff and high TDS both increase overall transport. 13
14 We see rock type and climate effects together in dissolved SiO 2, which in most natural waters is essentially all is from silicate rocks. Dissolution rate depends on temperature and rock type. Temperature dependence involves 2 factors. As T increases : dissolution rate increases biological production of dissolved organic acids increases Dissolved silica is highest in waters of tropical volcanic terrains such as Hawaii 14
Water Resources on PEI: an overview and brief discussion of challenges
 Water Resources on PEI: an overview and brief discussion of challenges Components: Components and links Atmospheric water Surface water (including glacial water) Groundwater Links: Precipitation (atm(
Water Resources on PEI: an overview and brief discussion of challenges Components: Components and links Atmospheric water Surface water (including glacial water) Groundwater Links: Precipitation (atm(
Oceans OUTLINE. Reading: White, Chapter 15 Today Finish estuaries and particles, then: 1. The oceans: currents, stratification and chemistry
 Oceans OUTLINE Reading: White, Chapter 15 Today Finish estuaries and particles, then: 1. The oceans: currents, stratification and chemistry Next Time Salinity Exercise bring something to calculate with
Oceans OUTLINE Reading: White, Chapter 15 Today Finish estuaries and particles, then: 1. The oceans: currents, stratification and chemistry Next Time Salinity Exercise bring something to calculate with
Unit 5 Lesson 1 What Is the Water Cycle? Copyright Houghton Mifflin Harcourt Publishing Company
 Water on the Move warm up 1 Water on the Move About three-fourths of Earth s surface is covered by water. Water on the Move Video!!! Water on the Move Water moves between Earth s surface and the atmosphere
Water on the Move warm up 1 Water on the Move About three-fourths of Earth s surface is covered by water. Water on the Move Video!!! Water on the Move Water moves between Earth s surface and the atmosphere
The Carbon cycle. Atmosphere, terrestrial biosphere and ocean are constantly exchanging carbon
 The Carbon cycle Atmosphere, terrestrial biosphere and ocean are constantly exchanging carbon The oceans store much more carbon than the atmosphere and the terrestrial biosphere The oceans essentially
The Carbon cycle Atmosphere, terrestrial biosphere and ocean are constantly exchanging carbon The oceans store much more carbon than the atmosphere and the terrestrial biosphere The oceans essentially
7.014 Lecture 20: Biogeochemical Cycles April 1, 2007
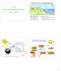 Global Nutrient Cycling - Biogeochemical Cycles 7.14 Lecture 2: Biogeochemical Cycles April 1, 27 Uptake Bioelements in Solution Weathering Precipitation Terrestrial Biomass Decomposition Volatile Elements
Global Nutrient Cycling - Biogeochemical Cycles 7.14 Lecture 2: Biogeochemical Cycles April 1, 27 Uptake Bioelements in Solution Weathering Precipitation Terrestrial Biomass Decomposition Volatile Elements
Ch. 5 - Nutrient Cycles and Soils
 Ch. 5 - Nutrient Cycles and Soils What are Nutrient (biogeochemical) Cycles? a process by which nutrients are recycled between living organisms and nonliving environment. The three general types of nutrient
Ch. 5 - Nutrient Cycles and Soils What are Nutrient (biogeochemical) Cycles? a process by which nutrients are recycled between living organisms and nonliving environment. The three general types of nutrient
Introduction. Welcome to the Belgium Study Abroad Program. Courses:
 Introduction Welcome to the Belgium Study Abroad Program Courses: AGSM 335: Soil and Water Management BAEN 460: Principals of Environmental Hydrology BAEN 460 / AGSM 335 Combined lecture and HW sessions
Introduction Welcome to the Belgium Study Abroad Program Courses: AGSM 335: Soil and Water Management BAEN 460: Principals of Environmental Hydrology BAEN 460 / AGSM 335 Combined lecture and HW sessions
Ch 18. Hydrologic Cycle and streams. Tom Bean
 Ch 18. Hydrologic Cycle and streams Tom Bean Wednesday s outline 1. the hydrologic cycle reservoirs cycling between them Evaporation and the atmosphere 2. Surface hydrology infiltration and soil moisture
Ch 18. Hydrologic Cycle and streams Tom Bean Wednesday s outline 1. the hydrologic cycle reservoirs cycling between them Evaporation and the atmosphere 2. Surface hydrology infiltration and soil moisture
Water is everywhere on Earth oceans, glaciers, rivers, lakes, air,
 Section 6.1 6.1 Running Water 1 FOCUS Section Objectives 6.1 Explain how the water cycle circulates Earth s water supply in an unending cycle. 6.2 Explain how the water cycle is kept in balance. 6.3 Describe
Section 6.1 6.1 Running Water 1 FOCUS Section Objectives 6.1 Explain how the water cycle circulates Earth s water supply in an unending cycle. 6.2 Explain how the water cycle is kept in balance. 6.3 Describe
Earth & Space Science (Water Cycle) Grade 7 Science Grade 7 Science Start Date: November 04, 2013 End Date : November 22, 2013
 Unit Overview Hydrologic Cycle, Thermal Energy Transfer. Content Elaborations The movement of water through the spheres of E water changes state and energy is transferred, it water transfers from the hydrosphere
Unit Overview Hydrologic Cycle, Thermal Energy Transfer. Content Elaborations The movement of water through the spheres of E water changes state and energy is transferred, it water transfers from the hydrosphere
Freshwater. 260 Points Total
 9 SURFACE WATER SECTION 9.1 Surface Water Movement In your textbook, read about surface water and the way in which it moves sediment. Complete each statement. (13 points) 1. An excessive amount of water
9 SURFACE WATER SECTION 9.1 Surface Water Movement In your textbook, read about surface water and the way in which it moves sediment. Complete each statement. (13 points) 1. An excessive amount of water
WATER AND THE HYDROLOGIC CYCLE
 WATER AND THE HYDROLOGIC CYCLE Summary Water is essential for the support of life and can be considered as a fundamental environmental good. Water is needed to support human habitation, grow crops and
WATER AND THE HYDROLOGIC CYCLE Summary Water is essential for the support of life and can be considered as a fundamental environmental good. Water is needed to support human habitation, grow crops and
CHAPTER 13 OUTLINE The Hydrologic Cycle and Groundwater. Hydrologic cycle. Hydrologic cycle cont.
 CHAPTER 13 OUTLINE The Hydrologic Cycle and Groundwater Does not contain complete lecture notes. To be used to help organize lecture notes and home/test studies. Hydrologic cycle The hydrologic cycle is
CHAPTER 13 OUTLINE The Hydrologic Cycle and Groundwater Does not contain complete lecture notes. To be used to help organize lecture notes and home/test studies. Hydrologic cycle The hydrologic cycle is
OCN 201 Chemical Oceanography Class Notes, Fall 2014 The origin of sea salt Chris Measures, Department of Oceanography
 OCN 201 Chemical Oceanography Class Notes, Fall 2014 The origin of sea salt Chris Measures, Department of Oceanography 1 Introduction Everyone knows that the sea is salty but what exactly is the salt in
OCN 201 Chemical Oceanography Class Notes, Fall 2014 The origin of sea salt Chris Measures, Department of Oceanography 1 Introduction Everyone knows that the sea is salty but what exactly is the salt in
Chapter 2: Aquifers and groundwater
 Chapter 2: Aquifers and groundwater Groundwater movement through aquifers is driven by differences in groundwater levels or pressure and is controlled by how porous the material is that it passes through.
Chapter 2: Aquifers and groundwater Groundwater movement through aquifers is driven by differences in groundwater levels or pressure and is controlled by how porous the material is that it passes through.
Lakes, Primary Production, Budgets and Cycling
 OCN 401-Biogeochemical Systems Lecture #10 (9.22.11) Lakes, Primary Production, Budgets and Cycling (Schlesinger: Chapter 7) 1. Primary Production and Nutrient Cycling in Lakes Physical aspects and nomenclature
OCN 401-Biogeochemical Systems Lecture #10 (9.22.11) Lakes, Primary Production, Budgets and Cycling (Schlesinger: Chapter 7) 1. Primary Production and Nutrient Cycling in Lakes Physical aspects and nomenclature
The role of freshwater ecosystems in carbon and nutrient cycling on the catchment scale. LSUE external launch Steven Bouillon
 The role of freshwater ecosystems in carbon and nutrient cycling on the catchment scale LSUE external launch Steven Bouillon Why are freshwater ecosystems important? CO 2 efflux 0.8 Pg C y -1 ~1.9 Pg C
The role of freshwater ecosystems in carbon and nutrient cycling on the catchment scale LSUE external launch Steven Bouillon Why are freshwater ecosystems important? CO 2 efflux 0.8 Pg C y -1 ~1.9 Pg C
GLY 155 Introduction to Physical Geology, W. Altermann. Grotzinger Jordan. Understanding Earth. Sixth Edition
 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 17: THE HYDROLOGIC CYCLE AND GROUNDWATER 2011 by W. H. Freeman and Company Chapter 17 The Hydrologic Cycle and Groundwater 1 About the Hydrologic
Grotzinger Jordan Understanding Earth Sixth Edition Chapter 17: THE HYDROLOGIC CYCLE AND GROUNDWATER 2011 by W. H. Freeman and Company Chapter 17 The Hydrologic Cycle and Groundwater 1 About the Hydrologic
Air & Water Lesson 2. Chapter 6 Conserving Our Resources
 Air & Water Lesson 2 Chapter 6 Conserving Our Resources Objectives Summarize the importance of air. Describe the water cycle. Main Idea Living things use air and water to carry out their life processes.
Air & Water Lesson 2 Chapter 6 Conserving Our Resources Objectives Summarize the importance of air. Describe the water cycle. Main Idea Living things use air and water to carry out their life processes.
Ecosystems: Nutrient Cycles
 Ecosystems: Nutrient Cycles Greeks, Native Peoples, Buddhism, Hinduism use(d) Earth, Air, Fire, and Water as the main elements of their faith/culture Cycling in Ecosystems the Hydrologic Cycle What are
Ecosystems: Nutrient Cycles Greeks, Native Peoples, Buddhism, Hinduism use(d) Earth, Air, Fire, and Water as the main elements of their faith/culture Cycling in Ecosystems the Hydrologic Cycle What are
Hydrology and Water Quality. Water. Water 9/13/2016. Molecular Water a great solvent. Molecular Water
 Hydrology and Water Quality Water Molecular Water Exists as an equilibrium But equilibrium altered by what is dissolved in it Water Molecular Water a great solvent In reality, water in the environment
Hydrology and Water Quality Water Molecular Water Exists as an equilibrium But equilibrium altered by what is dissolved in it Water Molecular Water a great solvent In reality, water in the environment
Unit III Nutrients & Biomes
 Unit III Nutrients & Biomes Nutrient Cycles Carbon Cycle Based on CO 2 cycling from animals to plants during respiration and photosynthesis. Heavy deposits are stored in wetland soils, oceans, sedimentary
Unit III Nutrients & Biomes Nutrient Cycles Carbon Cycle Based on CO 2 cycling from animals to plants during respiration and photosynthesis. Heavy deposits are stored in wetland soils, oceans, sedimentary
Guide 34. Ecosystem Ecology: Energy Flow and Nutrient Cycles. p://www.mordantorange.com/blog/archives/comics_by_mike_bannon/mordant_singles/0511/
 Guide 34 Ecosystem Ecology: Energy Flow and Nutrient Cycles p://www.mordantorange.com/blog/archives/comics_by_mike_bannon/mordant_singles/0511/ Overview: Ecosystems, Energy, and Matter An ecosystem consists
Guide 34 Ecosystem Ecology: Energy Flow and Nutrient Cycles p://www.mordantorange.com/blog/archives/comics_by_mike_bannon/mordant_singles/0511/ Overview: Ecosystems, Energy, and Matter An ecosystem consists
Introduction. Methods -6-
 S/OG Dissolved Oxygen and Carbonate-Carbon Dioxide in the Sea Water of the South China Sea, Area I: Gulf of Thailand and East Coast of Peninsular Malaysia Penjan Rojana-anawat and Anond Snidvongs
S/OG Dissolved Oxygen and Carbonate-Carbon Dioxide in the Sea Water of the South China Sea, Area I: Gulf of Thailand and East Coast of Peninsular Malaysia Penjan Rojana-anawat and Anond Snidvongs
Lakes, Primary Production, Budgets and Cycling Schlesinger and Bernhardt (2013): Chapter 8, p
 OCN 401-Biogeochemical Systems Lecture #12 (10.8.13) Angelos Hannides, hannides@hawaii.edu Lakes, Primary Production, Budgets and Cycling Schlesinger and Bernhardt (2013): Chapter 8, p. 288-308 1. Physical
OCN 401-Biogeochemical Systems Lecture #12 (10.8.13) Angelos Hannides, hannides@hawaii.edu Lakes, Primary Production, Budgets and Cycling Schlesinger and Bernhardt (2013): Chapter 8, p. 288-308 1. Physical
BAEN 673 / February 18, 2016 Hydrologic Processes
 BAEN 673 / February 18, 2016 Hydrologic Processes Assignment: HW#7 Next class lecture in AEPM 104 Today s topics SWAT exercise #2 The SWAT model review paper Hydrologic processes The Hydrologic Processes
BAEN 673 / February 18, 2016 Hydrologic Processes Assignment: HW#7 Next class lecture in AEPM 104 Today s topics SWAT exercise #2 The SWAT model review paper Hydrologic processes The Hydrologic Processes
Cycles of Ma,er. Lesson Overview. Lesson Overview. 3.4 Cycles of Matter
 Lesson Overview Cycles of Ma,er Lesson Overview 3.4 Cycles of Matter THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these
Lesson Overview Cycles of Ma,er Lesson Overview 3.4 Cycles of Matter THINK ABOUT IT A handful of elements combine to form the building blocks of all known organisms. Organisms cannot manufacture these
East Maui Watershed Partnership Adapted from Utah State University and University of Wisconsin Ground Water Project Ages 7 th -Adult
 INTRODUCTION What is groundwater? Water contained in saturated soil and rock materials below the surface of the earth. It is not NEW water, but is recycled water through the hydraulic cycle. The source
INTRODUCTION What is groundwater? Water contained in saturated soil and rock materials below the surface of the earth. It is not NEW water, but is recycled water through the hydraulic cycle. The source
Interpretation of Chemistry Data from Seepage, Observation Well, and Reservoir Water Samples collected at Horsetooth Dam during July 2004
 Interpretation of Chemistry Data from Seepage, Observation Well, and Reservoir Water Samples collected at Horsetooth Dam during July 2004 TSC Technical Memorandum No. 8290-04-02 by Doug Craft, Research
Interpretation of Chemistry Data from Seepage, Observation Well, and Reservoir Water Samples collected at Horsetooth Dam during July 2004 TSC Technical Memorandum No. 8290-04-02 by Doug Craft, Research
Keywords: rivers, major ions, nutrients, chemical weathering, global distribution, human impacts.
 CHEMICAL CHARACTERISTICS OF RIVERS M. Meybeck, CNRS, University of Paris VI, France Keywords: rivers, major ions, nutrients, chemical weathering, global distribution, human impacts. Contents 1. Introduction
CHEMICAL CHARACTERISTICS OF RIVERS M. Meybeck, CNRS, University of Paris VI, France Keywords: rivers, major ions, nutrients, chemical weathering, global distribution, human impacts. Contents 1. Introduction
Ocean Production and CO 2 uptake
 Ocean Production and CO 2 uptake Fig. 6.6 Recall: Current ocean is gaining Carbon.. OCEAN Reservoir size: 38000 Flux in: 90 Flux out: 88+0.2=88.2 90-88.2 = 1.8 Pg/yr OCEAN is gaining 1.8 Pg/yr Sum of the
Ocean Production and CO 2 uptake Fig. 6.6 Recall: Current ocean is gaining Carbon.. OCEAN Reservoir size: 38000 Flux in: 90 Flux out: 88+0.2=88.2 90-88.2 = 1.8 Pg/yr OCEAN is gaining 1.8 Pg/yr Sum of the
Country Profile - Republic of Korea INFORMATION
 INFORMATION Geography The Republic of Korea occupies the southern portion of the Korean Peninsula, which extends some 1,100 km from the Asian mainland. This mountainous peninsula is flanked by the Yellow
INFORMATION Geography The Republic of Korea occupies the southern portion of the Korean Peninsula, which extends some 1,100 km from the Asian mainland. This mountainous peninsula is flanked by the Yellow
Water Quality. CE 370 Lecture 1. Global Distribution of Earth s s Water
 Water Quality CE 370 Lecture 1 Global Distribution of Earth s s Water Water Demand and Supply in Saudi Arabia Total Water Consumption = 22 billion m 3 /Year Water Demand Water Supply Industrial Domestic
Water Quality CE 370 Lecture 1 Global Distribution of Earth s s Water Water Demand and Supply in Saudi Arabia Total Water Consumption = 22 billion m 3 /Year Water Demand Water Supply Industrial Domestic
Session 14 Unit VI CLIMATIC CHANGE AND GLOBAL WARMING
 Session 14 Unit VI CLIMATIC CHANGE AND GLOBAL WARMING Dr. H.S. Ramesh Professor of Environmental Engineering S.J. College of Engineering, Mysore 570 006 Carbon di-oxide is a natural constituent of atmosphere,
Session 14 Unit VI CLIMATIC CHANGE AND GLOBAL WARMING Dr. H.S. Ramesh Professor of Environmental Engineering S.J. College of Engineering, Mysore 570 006 Carbon di-oxide is a natural constituent of atmosphere,
NCERT solution for Natural Resources
 1 NCERT solution for Natural Resources Question 1 How is our atmosphere different from the atmospheres on Venus and Mars? Earth's atmosphere is a mixture of nitrogen (79%), oxygen (20%), and a small fraction
1 NCERT solution for Natural Resources Question 1 How is our atmosphere different from the atmospheres on Venus and Mars? Earth's atmosphere is a mixture of nitrogen (79%), oxygen (20%), and a small fraction
Impact of Damming and Eutrophication on DSi:DIN Variation in River Water, a case study of Yahagi River, Japan
 Impact of Damming and Eutrophication on DSi:DIN Variation in River Water, a case study of Yahagi River, Japan Masashi Kodama* * & Katsuhisa Tanaka** *National Research Institute of Fisheries Science, Fisheries
Impact of Damming and Eutrophication on DSi:DIN Variation in River Water, a case study of Yahagi River, Japan Masashi Kodama* * & Katsuhisa Tanaka** *National Research Institute of Fisheries Science, Fisheries
Cycling and Biogeochemical Transformations of N, P and S
 Cycling and Biogeochemical Transformations of N, P and S OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions of N gases from soils
Cycling and Biogeochemical Transformations of N, P and S OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 6 1. Nitrogen cycle Soil nitrogen cycle Nitrification Emissions of N gases from soils
AP Environmental Science
 AP Environmental Science Types of aquatic life zones MARINE Estuaries coral reefs mangrove swamps neritic zone pelagic zone FRESHWATER lakes and ponds streams and rivers wetlands Distribution of aquatic
AP Environmental Science Types of aquatic life zones MARINE Estuaries coral reefs mangrove swamps neritic zone pelagic zone FRESHWATER lakes and ponds streams and rivers wetlands Distribution of aquatic
Sustainability in Plastics. Calcium Carbonate [in nature, in life]
![Sustainability in Plastics. Calcium Carbonate [in nature, in life] Sustainability in Plastics. Calcium Carbonate [in nature, in life]](/thumbs/77/76282811.jpg) Sustainability in Plastics Calcium Carbonate [in nature, in life] Omya Group Omya is a leading global producer of industrial minerals, mainly fillers and pigments derived from calcium carbonate and dolomite
Sustainability in Plastics Calcium Carbonate [in nature, in life] Omya Group Omya is a leading global producer of industrial minerals, mainly fillers and pigments derived from calcium carbonate and dolomite
BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through living organisms and the physical environment.
 BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through living organisms and the physical environment. BIOCHEMIST: Scientists who study how LIFE WORKS at a CHEMICAL level. The work of biochemists has
BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through living organisms and the physical environment. BIOCHEMIST: Scientists who study how LIFE WORKS at a CHEMICAL level. The work of biochemists has
Where have we been. Where are we going today? Lecture Outline. Geoengineering. What is a system?
 Where have we been 1. Course Introduction 2. What is the environment 3. Examined in some detail weather disasters for 2010 and 2011 4. Touched on Scale 5. Touched on borders. Examples a) Air pollution
Where have we been 1. Course Introduction 2. What is the environment 3. Examined in some detail weather disasters for 2010 and 2011 4. Touched on Scale 5. Touched on borders. Examples a) Air pollution
Evaluation copy. Total Dissolved Solids. Computer INTRODUCTION
 Total Dissolved Solids Computer 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Total Dissolved Solids Computer 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
ECOSYSTEMS. Follow along in chapter 54. *Means less important
 ECOSYSTEMS Follow along in chapter 54 *Means less important How do ecosystems function? What is an ecosystem? All living things in an area and their abiotic environment Ecosystem function can be easily
ECOSYSTEMS Follow along in chapter 54 *Means less important How do ecosystems function? What is an ecosystem? All living things in an area and their abiotic environment Ecosystem function can be easily
Chapter 3 Ecosystem Ecology. Tuesday, September 19, 17
 Chapter 3 Ecosystem Ecology Reversing Deforestation in Haiti Answers the following: Why is deforestation in Haiti so common? What the negative impacts of deforestation? Name three actions intended counteract
Chapter 3 Ecosystem Ecology Reversing Deforestation in Haiti Answers the following: Why is deforestation in Haiti so common? What the negative impacts of deforestation? Name three actions intended counteract
Earth s Pools of Water What is the residence time of these pools?
 (1) The Hydrologic Cycle What are the major pools (reservoirs) of water on EARTH? More than enough water on our planet to sustain life. Why then do we have water shortages? And, why are we concerned about
(1) The Hydrologic Cycle What are the major pools (reservoirs) of water on EARTH? More than enough water on our planet to sustain life. Why then do we have water shortages? And, why are we concerned about
1) The Changing Carbon Cycle
 1) The Changing Carbon Cycle WG1 Chapter 6, figure 1 The numbers represent carbon reservoirs in Petagrams of Carbon (PgC; 10 15 gc) and the annual exchanges in PgC/year. The black numbers and arrows show
1) The Changing Carbon Cycle WG1 Chapter 6, figure 1 The numbers represent carbon reservoirs in Petagrams of Carbon (PgC; 10 15 gc) and the annual exchanges in PgC/year. The black numbers and arrows show
Battle for the Biosphere
 Battle for the Biosphere Biome: A large community of plants and animals that occupies a distinct region e.g. the Tropical rainforest. Biosphere: The parts of the land, sea and atmosphere where organisms
Battle for the Biosphere Biome: A large community of plants and animals that occupies a distinct region e.g. the Tropical rainforest. Biosphere: The parts of the land, sea and atmosphere where organisms
THE IMPORTANCE OF WATER
 THE IMPORTANCE OF WATER 2/3rds of the Earth s surface is covered in. water Earth s aquatic ecosystems contain biomass more (living mass) than its terrestrial ecosystems. Phytoplankton are microscopic producers
THE IMPORTANCE OF WATER 2/3rds of the Earth s surface is covered in. water Earth s aquatic ecosystems contain biomass more (living mass) than its terrestrial ecosystems. Phytoplankton are microscopic producers
from volcanoes; carbonate (CaCO 3 + CO 2 + H 2 . The sinks are carbonate rock weathering + SiO2. Ca HCO
 The Carbon Cycle Chemical relations We would like to be able to trace the carbon on Earth and see where it comes and where it goes. The sources are CO 2 from volcanoes; carbonate (CaCO 3 ) formation in
The Carbon Cycle Chemical relations We would like to be able to trace the carbon on Earth and see where it comes and where it goes. The sources are CO 2 from volcanoes; carbonate (CaCO 3 ) formation in
Earth Science Chapter 6 Section 1 Review
 Name: Class: Date: Earth Science Chapter 6 Section 1 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. What process is illustrated by the arrows labeled
Name: Class: Date: Earth Science Chapter 6 Section 1 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. What process is illustrated by the arrows labeled
Chapter Concepts LIFE IN WATER. The Hydrologic Cycle. The Hydrologic Cycle
 Chapter Concepts Chapter 3 LIFE IN WATER The hydrologic cycle exchanges water among reservoirs The biology of aquatic environments corresponds broadly to variations in physical factors such as light, temperature,
Chapter Concepts Chapter 3 LIFE IN WATER The hydrologic cycle exchanges water among reservoirs The biology of aquatic environments corresponds broadly to variations in physical factors such as light, temperature,
ì<(sk$m)=cdegfd< +^-Ä-U-Ä-U
 Standards Preview Earth Sciences Standard Set 3. Earth Sciences 3. Water on Earth moves between the oceans and land through the processes of evaporation and condensation. As a basis for understanding this
Standards Preview Earth Sciences Standard Set 3. Earth Sciences 3. Water on Earth moves between the oceans and land through the processes of evaporation and condensation. As a basis for understanding this
Salt-Clay Interactions: Reducing Sediment Loads and Erosion at Snow Storage Sites
 Salt-Clay Interactions: Reducing Sediment Loads and Erosion at Snow Storage Sites 17 th Street Snow Storage Site Wanda Goulden, P.Eng., P.Geol. General Supervisor, GeoEnvironmental Engineering Engineering
Salt-Clay Interactions: Reducing Sediment Loads and Erosion at Snow Storage Sites 17 th Street Snow Storage Site Wanda Goulden, P.Eng., P.Geol. General Supervisor, GeoEnvironmental Engineering Engineering
Climate Change: Implications for Hydropower Sustainability. HSAF Meeting 5 Itaipu Dec. 8, 2008
 Climate Change: Implications for Hydropower Sustainability HSAF Meeting 5 Itaipu Dec. 8, 2008 Without climate change, we might not be in this room: Renewed interest in renewable energy for climate change
Climate Change: Implications for Hydropower Sustainability HSAF Meeting 5 Itaipu Dec. 8, 2008 Without climate change, we might not be in this room: Renewed interest in renewable energy for climate change
THE WATER CYCLE IN GREATER VICTORIA
 THE WATER CYCLE IN GREATER VICTORIA KEY CONCEPTS THE GREATER VICTORIA DRINKING WATER SUPPLY SYSTEM IS DEPENDENT ON ANNUAL PRECIPITATION, WHICH FALLS MOSTLY AS RAIN. RESPONSIBILITY FOR WATER IS EVERYONE
THE WATER CYCLE IN GREATER VICTORIA KEY CONCEPTS THE GREATER VICTORIA DRINKING WATER SUPPLY SYSTEM IS DEPENDENT ON ANNUAL PRECIPITATION, WHICH FALLS MOSTLY AS RAIN. RESPONSIBILITY FOR WATER IS EVERYONE
Lecture 1 Integrated water resources management and wetlands
 Wetlands and Poverty Reduction Project (WPRP) Training module on Wetlands and Water Resources Management Lecture 1 Integrated water resources management and wetlands 1 Water resources and use The hydrological
Wetlands and Poverty Reduction Project (WPRP) Training module on Wetlands and Water Resources Management Lecture 1 Integrated water resources management and wetlands 1 Water resources and use The hydrological
Total Dissolved Solids
 Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Climate Change: Background and Implications
 Climate Change: Background and Implications American Public Works Association Conference on Climate Change Tempe, AZ April 9, 2008 Peter Schultz U.S. Climate Change Science Program Office, Director Outline
Climate Change: Background and Implications American Public Works Association Conference on Climate Change Tempe, AZ April 9, 2008 Peter Schultz U.S. Climate Change Science Program Office, Director Outline
Yoshiteru Tsuchiya Department of Applied Chemistry, Faculty of Engineering, Kogakuin University, Japan
 NATURAL WATERS Yoshiteru Tsuchiya Department of Applied Chemistry, Faculty of Engineering, Kogakuin University, Japan Keywords: natural water, water quality, Ocean, rain, precipitation, river, lake, groundwater,
NATURAL WATERS Yoshiteru Tsuchiya Department of Applied Chemistry, Faculty of Engineering, Kogakuin University, Japan Keywords: natural water, water quality, Ocean, rain, precipitation, river, lake, groundwater,
Atmosphere, the Water Cycle and Climate Change
 Atmosphere, the Water Cycle and Climate Change OCN 623 Chemical Oceanography 16 April 2013 (Based on previous lectures by Barry Huebert) 2013 F.J. Sansone 1. The water cycle Outline 2. Climate and climate-change
Atmosphere, the Water Cycle and Climate Change OCN 623 Chemical Oceanography 16 April 2013 (Based on previous lectures by Barry Huebert) 2013 F.J. Sansone 1. The water cycle Outline 2. Climate and climate-change
Water Quality of Fayoum Surface Water, Fayoum Province, Egypt
 Deltas in Times of Climate Change II, Rotterdam, the Netherlands, 24-26 Sept. 2014 Water Quality of Fayoum Surface Water, Fayoum Province, Egypt Mahmoud S.M. Abdel Wahed a,b,*, Essam A. Mohamed b, Christian
Deltas in Times of Climate Change II, Rotterdam, the Netherlands, 24-26 Sept. 2014 Water Quality of Fayoum Surface Water, Fayoum Province, Egypt Mahmoud S.M. Abdel Wahed a,b,*, Essam A. Mohamed b, Christian
CHAPTER 6: GEOCHEMICAL CYCLES Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017
 CHAPTER 6: GEOCHEMICAL CYCLES Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017 THE EARTH: ASSEMBLAGE OF ATOMS OF THE 92 NATURAL ELEMENTS Most abundant elements: oxygen (in solid
CHAPTER 6: GEOCHEMICAL CYCLES Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017 THE EARTH: ASSEMBLAGE OF ATOMS OF THE 92 NATURAL ELEMENTS Most abundant elements: oxygen (in solid
FAPESP s Research Program for Global Climate Change (PFPMCG) annual workshop São Paulo, February 18-19, 2016
 FAPESP s Research Program for Global Climate Change (PFPMCG) annual workshop São Paulo, February 18-19, 2016 PIs: Maria Victoria R. Ballester; Reynaldo L. Victoria; Alex V. Krusche & Jefrey E. Richey Co-
FAPESP s Research Program for Global Climate Change (PFPMCG) annual workshop São Paulo, February 18-19, 2016 PIs: Maria Victoria R. Ballester; Reynaldo L. Victoria; Alex V. Krusche & Jefrey E. Richey Co-
The Returning Raindrop
 Did you know that some of the water molecules we drink today may have been around when dinosaurs walked the Earth thousands of years ago? Water is continuously recycling in a process called the hydrologic
Did you know that some of the water molecules we drink today may have been around when dinosaurs walked the Earth thousands of years ago? Water is continuously recycling in a process called the hydrologic
Unit 2: Weather Dynamics Chapter 1: Hydrological Cycle
 UNIT 2 Chapter 1: Inquiring about Weather Unit 2: Weather Dynamics Chapter 1: Hydrological Cycle Science 10 Mrs. Purba Importance of H 2 O From the beginning of time when water first appeared, it has been
UNIT 2 Chapter 1: Inquiring about Weather Unit 2: Weather Dynamics Chapter 1: Hydrological Cycle Science 10 Mrs. Purba Importance of H 2 O From the beginning of time when water first appeared, it has been
National Wildlife Federation Eco-Schools USA
 ATMOSPHERE GLOBE student data within the Atmosphere investigation aids scientific understanding of spatial gaps in air temperature and precipitation coverage by weather monitoring stations, important data
ATMOSPHERE GLOBE student data within the Atmosphere investigation aids scientific understanding of spatial gaps in air temperature and precipitation coverage by weather monitoring stations, important data
Geochemical Nutrient Scavenging. High mountain lakes as model systems. Research Questions
 Research Questions Geochemical Nutrient Scavenging High mountain lakes as model systems Kurt Hanselmann, Microbiology: Ecology: Biogeochemistry: Geobiology: How do microorganisms cope with low nutrient
Research Questions Geochemical Nutrient Scavenging High mountain lakes as model systems Kurt Hanselmann, Microbiology: Ecology: Biogeochemistry: Geobiology: How do microorganisms cope with low nutrient
The unnatural carbon dioxide cycle and oceanic processes over the last few hundred years. OCN 623 Chemical Oceanography
 The unnatural carbon dioxide cycle and oceanic processes over the last few hundred years OCN 623 Chemical Oceanography In the 19th century, scientists realized that gases in the atmosphere cause a "greenhouse
The unnatural carbon dioxide cycle and oceanic processes over the last few hundred years OCN 623 Chemical Oceanography In the 19th century, scientists realized that gases in the atmosphere cause a "greenhouse
Environmental Principles & Concepts (EP&C)/COSA Correlation of 4 th - 7 th Grade CA Science Standards
 Environmental Principles & Concepts (EP&C)/COSA Correlation of 4 th - 7 th Grade CA Science Standards For ROSS Certifications at least nine of the 14 science standards are addressed during a 5-day program,
Environmental Principles & Concepts (EP&C)/COSA Correlation of 4 th - 7 th Grade CA Science Standards For ROSS Certifications at least nine of the 14 science standards are addressed during a 5-day program,
Chapter 6. Aquatic Biodiversity. Chapter Overview Questions
 Chapter 6 Aquatic Biodiversity Chapter Overview Questions Ø What are the basic types of aquatic life zones and what factors influence the kinds of life they contain? Ø What are the major types of saltwater
Chapter 6 Aquatic Biodiversity Chapter Overview Questions Ø What are the basic types of aquatic life zones and what factors influence the kinds of life they contain? Ø What are the major types of saltwater
The national-level nutrient loading estimation tool for Finland: Watershed Simulation and Forecasting System WSFS-Vemala
 The national-level nutrient loading estimation tool for Finland: Watershed Simulation and Forecasting System WSFS-Vemala Markus Huttunen, Finnish Environment Institute SYKE, HELCOM workshop on transboundary
The national-level nutrient loading estimation tool for Finland: Watershed Simulation and Forecasting System WSFS-Vemala Markus Huttunen, Finnish Environment Institute SYKE, HELCOM workshop on transboundary
Generation & management of effluents in cane sugar production
 Generation & management of effluents in cane sugar production Graham Kingston 1 & Samira Daroub 2 1 BSES Limited, Bundaberg AUSTRALIA 2 IFAS, University of Florida, USA Outline Which effluents? Perspective
Generation & management of effluents in cane sugar production Graham Kingston 1 & Samira Daroub 2 1 BSES Limited, Bundaberg AUSTRALIA 2 IFAS, University of Florida, USA Outline Which effluents? Perspective
Climate change science, knowledge and impacts on water resources in South Asia
 Climate change science, knowledge and impacts on water resources in South Asia DIAGNOSTIC PAPER 1 GUILLAUME LACOMBE, PENNAN CHINNASAMY Regional Conference on Risks and Solutions: Adaptation Frameworks
Climate change science, knowledge and impacts on water resources in South Asia DIAGNOSTIC PAPER 1 GUILLAUME LACOMBE, PENNAN CHINNASAMY Regional Conference on Risks and Solutions: Adaptation Frameworks
The Hydrological Cycle. Hydrological Cycle. Definition of Terms. Soils and Water, Spring Lecture 7, The Hydrological Cycle 1
 The Hydrological Cycle Water vapor helps warm up the earth Evaporation+Transpiration Chemical Spill Runoff To Oceans Water potential, atmosphere Hydrological Cycle Transpiration, T Atmospheric pool Interception
The Hydrological Cycle Water vapor helps warm up the earth Evaporation+Transpiration Chemical Spill Runoff To Oceans Water potential, atmosphere Hydrological Cycle Transpiration, T Atmospheric pool Interception
Carbon Dioxide, Alkalinity and ph
 Carbon Dioxide, Alkalinity and ph OCN 623 Chemical Oceanography 31 January 2013 Reading: Libes, Chapter 15, pp. 383 394 (Remainder of chapter will be used with the lecture: Biogenic production, carbonate
Carbon Dioxide, Alkalinity and ph OCN 623 Chemical Oceanography 31 January 2013 Reading: Libes, Chapter 15, pp. 383 394 (Remainder of chapter will be used with the lecture: Biogenic production, carbonate
The effect of Hilti / Salahi Recharge Dam in Batineh Area, Sultanate of Oman on the Quality of Groundwater Using GIS
 The effect of Hilti / Salahi Recharge Dam in Batineh Area, Sultanate of Oman on the Quality of Groundwater Using GIS William Bajjali University of Wisconsin Superior, Department of Biology & Earth Sciences,
The effect of Hilti / Salahi Recharge Dam in Batineh Area, Sultanate of Oman on the Quality of Groundwater Using GIS William Bajjali University of Wisconsin Superior, Department of Biology & Earth Sciences,
What does each part of the equation mean? q=cm T
 Assignment #10 Energy Pyramids LO: I can define trophic levels and explain the energy flow. I can apply those ideas to food webs EQ: Where does all the energy from the sun go? (4-5 sentences) LEVEL ZERO
Assignment #10 Energy Pyramids LO: I can define trophic levels and explain the energy flow. I can apply those ideas to food webs EQ: Where does all the energy from the sun go? (4-5 sentences) LEVEL ZERO
Life in Water. Chapter 3
 Life in Water Chapter 3 Outline Hydrologic Cycle Oceans Shallow Marine Waters Marine Shores Estuaries, Salt Marshes, and Mangrove Forests Rivers and Streams Lakes 2 The Hydrologic Cycle Over 71% of the
Life in Water Chapter 3 Outline Hydrologic Cycle Oceans Shallow Marine Waters Marine Shores Estuaries, Salt Marshes, and Mangrove Forests Rivers and Streams Lakes 2 The Hydrologic Cycle Over 71% of the
The modification of global atmospheric Nitrogen cycling by human activities. David Fowler Centre for Ecology and Hydrology Edinburgh UK
 The modification of global atmospheric Nitrogen cycling by human activities David Fowler Centre for Ecology and Hydrology Edinburgh UK Background The N cycle Global N emissions Oxidised Nitrogen Reduced
The modification of global atmospheric Nitrogen cycling by human activities David Fowler Centre for Ecology and Hydrology Edinburgh UK Background The N cycle Global N emissions Oxidised Nitrogen Reduced
Climate change and groundwater resources in Lao PDR
 Journal of Groundwater Science and Engineering Vol.5 Vol.4 No.1 No.2 Mar. Jun. 2016 2017 Climate change and groundwater resources in Lao PDR Khongsab Somphone 1*, OunakoneKone Xayviliya 1 1 Division of
Journal of Groundwater Science and Engineering Vol.5 Vol.4 No.1 No.2 Mar. Jun. 2016 2017 Climate change and groundwater resources in Lao PDR Khongsab Somphone 1*, OunakoneKone Xayviliya 1 1 Division of
Environmental Geography
 Environmental Geography Lecture 13 Water Pollution Lecture 13: Water Pollution I. Water Pollution A. Groundwater B. Surface Water C. Regulation II. Water Use in California 1 I. Water Pollution Types of
Environmental Geography Lecture 13 Water Pollution Lecture 13: Water Pollution I. Water Pollution A. Groundwater B. Surface Water C. Regulation II. Water Use in California 1 I. Water Pollution Types of
Climate: describes the average condition, including temperature and precipitation, over long periods in a given area
 Ch. 6 - Biomes Section 6.1: Defining Biomes Biome: a group of ecosystems that share similar biotic and abiotic conditions, large region characterized by a specific type of climate, plants, and animals
Ch. 6 - Biomes Section 6.1: Defining Biomes Biome: a group of ecosystems that share similar biotic and abiotic conditions, large region characterized by a specific type of climate, plants, and animals
Ecology, the Environment, and Us
 BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 23 Ecology, the Environment, and Us Lecture Presentation Anne Gasc Hawaii Pacific University and University
BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 23 Ecology, the Environment, and Us Lecture Presentation Anne Gasc Hawaii Pacific University and University
WHY CARBON? The Carbon Cycle 1/17/2011. All living organisms utilize the same molecular building blocks. Carbon is the currency of life
 The Carbon Cycle WHY CARBON? Inventories: black text Fluxes: purple arrows Carbon dioxide (+4) AN = 6 (6P/6N) AW = 12.011 Oxidation: -4 to +4 Isotopes: 11 C, 12 C, 1 C, 14 C Methane (-4) Carbon is the
The Carbon Cycle WHY CARBON? Inventories: black text Fluxes: purple arrows Carbon dioxide (+4) AN = 6 (6P/6N) AW = 12.011 Oxidation: -4 to +4 Isotopes: 11 C, 12 C, 1 C, 14 C Methane (-4) Carbon is the
Carbon Dioxide, Alkalinity and ph
 Carbon Dioxide, Alkalinity and ph OCN 62 Chemical Oceanography Reading: Libes, Chapter 15, pp. 8 94 (Remainder of chapter: Biogenic production, carbonate saturation and sediment distributions ) 1. CO 2
Carbon Dioxide, Alkalinity and ph OCN 62 Chemical Oceanography Reading: Libes, Chapter 15, pp. 8 94 (Remainder of chapter: Biogenic production, carbonate saturation and sediment distributions ) 1. CO 2
1.6 Influence of Human Activities and Land use Changes on Hydrologic Cycle
 1.6 Influence of Human Activities and Land use Changes on Hydrologic Cycle Watersheds are subjected to many types of changes, major or minor, for various reasons. Some of these are natural changes and
1.6 Influence of Human Activities and Land use Changes on Hydrologic Cycle Watersheds are subjected to many types of changes, major or minor, for various reasons. Some of these are natural changes and
Ocean Carbon Sequestration
 Aspen Global Change Institute Ocean Carbon Sequestration Ken Caldeira DOE Center for Research on Ocean Carbon Sequestration and Lawrence Livermore National Laboratory Outline Carbon management and ocean
Aspen Global Change Institute Ocean Carbon Sequestration Ken Caldeira DOE Center for Research on Ocean Carbon Sequestration and Lawrence Livermore National Laboratory Outline Carbon management and ocean
Hydrology and Water Management. Dr. Mujahid Khan, UET Peshawar
 Hydrology and Water Management Dr. Mujahid Khan, UET Peshawar Course Outline Hydrologic Cycle and its Processes Water Balance Approach Estimation and Analysis of Precipitation Data Infiltration and Runoff
Hydrology and Water Management Dr. Mujahid Khan, UET Peshawar Course Outline Hydrologic Cycle and its Processes Water Balance Approach Estimation and Analysis of Precipitation Data Infiltration and Runoff
GLOBAL WARMING IS HAPPENING GLOBAL WARMING WILL BE VERY HARD TO STOP (By John B. Wheeler, member Potomac River Association)
 GLOBAL WARMING IS HAPPENING GLOBAL WARMING WILL BE VERY HARD TO STOP (By John B. Wheeler, member Potomac River Association) READ ON AND SEE WHY YOU NEED TO BE WORRIED The Outline of what follows: 1 The
GLOBAL WARMING IS HAPPENING GLOBAL WARMING WILL BE VERY HARD TO STOP (By John B. Wheeler, member Potomac River Association) READ ON AND SEE WHY YOU NEED TO BE WORRIED The Outline of what follows: 1 The
Basics Course. Montana. Introduction to Drinking Water Systems. for small public drinking water systems serving less than 3,300 people
 Basics Course for small public drinking water systems serving less than 3,300 people Montana Montana Basics Course for Small Public Drinking Water Systems -1 Introduction to Drinking Water Systems Welcome
Basics Course for small public drinking water systems serving less than 3,300 people Montana Montana Basics Course for Small Public Drinking Water Systems -1 Introduction to Drinking Water Systems Welcome
Fossil Fuels: Natural Gas. Outline: Formation Global supply and Distribution NGCC Carbon management Biological Artificial
 Fossil Fuels: Natural Gas Outline: Formation Global supply and Distribution NGCC Carbon management Biological Artificial Formation of Natural Gas Migration Phases separate according to density, with the
Fossil Fuels: Natural Gas Outline: Formation Global supply and Distribution NGCC Carbon management Biological Artificial Formation of Natural Gas Migration Phases separate according to density, with the
People, Oceans and Climate Change
 People, Oceans and Climate Change A deeper look at the carbon dioxide cycle, greenhouse gases, and oceanic processes over the last 150 years OCN 623 Chemical Oceanography 18 April 2013 Reading: Libes,
People, Oceans and Climate Change A deeper look at the carbon dioxide cycle, greenhouse gases, and oceanic processes over the last 150 years OCN 623 Chemical Oceanography 18 April 2013 Reading: Libes,
BIOMES. Living World
 BIOMES Living World Biomes Biomes are large regions of the world with distinctive climate, wildlife and vegetation. They are divided by terrestrial (land) or aquatic biomes. Terrestrial Biomes Terrestrial
BIOMES Living World Biomes Biomes are large regions of the world with distinctive climate, wildlife and vegetation. They are divided by terrestrial (land) or aquatic biomes. Terrestrial Biomes Terrestrial
SPE DISTINGUISHED LECTURER SERIES is funded principally through a grant of the SPE FOUNDATION
 SPE DISTINGUISHED LECTURER SERIES is funded principally through a grant of the SPE FOUNDATION The Society gratefully acknowledges those companies that support the program by allowing their professionals
SPE DISTINGUISHED LECTURER SERIES is funded principally through a grant of the SPE FOUNDATION The Society gratefully acknowledges those companies that support the program by allowing their professionals
Threats to Forest Ecosystem Health Activities together influence ecosystem structure & function
 Threats to Forest Ecosystem Health Activities together influence ecosystem structure & function introduced species poor management air pollution global warming habitat fragmentation American Chestnut From
Threats to Forest Ecosystem Health Activities together influence ecosystem structure & function introduced species poor management air pollution global warming habitat fragmentation American Chestnut From
Class IX Chapter 14 Natural Resources Science
 Question 1: How is our atmosphere different from the atmospheres on Venus and Mars? Earth s atmosphere is different from those of Venus and Mars. This difference lies essentially in their compositions.
Question 1: How is our atmosphere different from the atmospheres on Venus and Mars? Earth s atmosphere is different from those of Venus and Mars. This difference lies essentially in their compositions.
Lecture 1: Hydrologic cycle
 1-1 GEOG415 Lecture 1: Hydrologic cycle Hydrologic cycle is ultimately driven by solar radiation, which evaporates water from the ocean and lift it up in the atmosphere. Dunne & Leopold, 1978, Fig. 1-1
1-1 GEOG415 Lecture 1: Hydrologic cycle Hydrologic cycle is ultimately driven by solar radiation, which evaporates water from the ocean and lift it up in the atmosphere. Dunne & Leopold, 1978, Fig. 1-1
How Salty Is Our Water?
 Title: How Salty Is Our Water? (Water Chemistry) Grade(s): 6-7 Introduction: Corals require a relatively constant environment. However, due to the proximity to the surface of the ocean, the salinity of
Title: How Salty Is Our Water? (Water Chemistry) Grade(s): 6-7 Introduction: Corals require a relatively constant environment. However, due to the proximity to the surface of the ocean, the salinity of
WHY DO WE NEED NITROGEN?? Nitrogen is needed to make up DNA and protein!
 Nitrogen Cycle 2.2 WHY DO WE NEED NITROGEN?? Nitrogen is needed to make up DNA and protein! In animals, proteins are vital for muscle function. In plants, nitrogen is important for growth. NITROGEN Nitrogen
Nitrogen Cycle 2.2 WHY DO WE NEED NITROGEN?? Nitrogen is needed to make up DNA and protein! In animals, proteins are vital for muscle function. In plants, nitrogen is important for growth. NITROGEN Nitrogen
