JANUARY 20, 2006 VOLUME 281 NUMBER 3 JOURNAL OF BIOLOGICAL CHEMISTRY 1765
|
|
|
- Bennett Newton
- 6 years ago
- Views:
Transcription
1 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 281, NO. 3, pp , January 20, 2006 Printed in the U.S.A. RhoA Modulates Smad Signaling during Transforming Growth Factor- -induced Smooth Muscle Differentiation * Received for publication, July 18, 2005, and in revised form, November 21, 2005 Published, JBC Papers in Press, November 28, 2005, DOI /jbc.M Shiyou Chen 1, Michelle Crawford, Regina M. Day, Victorino R. Briones, Jennifer E. Leader, Pedro A. Jose, and Robert J. Lechleider 2 From the Department of Cell Biology and Department of Pediatrics, Georgetown University Medical School, Washington, D. C and the Department of Pharmacology, Uniformed Services University of the Health Sciences, Bethesda, Maryland We recently reported that transforming growth factor (TGF)- induced the neural crest stem cell line Monc-1 to differentiate into a spindle-like contractile smooth muscle cell (SMC) phenotype and that Smad signaling played an important role in this phenomenon. In addition to Smad signaling, other pathways such as mitogenactivated protein kinase (MAPK), phosphoinositol-3 kinase, and RhoA have also been shown to mediate TGF- actions. The objectives of this study were to examine whether these signaling pathways contribute to TGF- -induced SMC development and to test whether Smad signaling cross-talks with other pathway(s) during SMC differentiation induced by TGF-. We demonstrate here that RhoA signaling is critical to TGF- -induced SMC differentiation. RhoA kinase (ROCK) inhibitor Y27632 significantly blocks the expression of multiple SMC markers such as smooth muscle -actin, SM22, and calponin in TGF- -treated Monc-1 cells. In addition, Y27632 reversed the cell morphology and abolished the contractility of TGF- -treated cells. RhoA signaling was activated as early as 5 min following TGF- addition. Dominant negative RhoA blocked nuclear translocation of Smad2 and Smad3 because of the inhibition of phosphorylation of both Smads and inhibited Smaddependent SBE promoter activity, whereas constitutively active RhoA significantly enhanced SBE promoter activity. Consistent with these results, C3 exotoxin, an inhibitor of RhoA activation, significantly attenuated SBE promoter activity and inhibited Smad nuclear translocation. Taken together, these data point to a new role for RhoA as a modulator of Smad activation while regulating TGF- -induced SMC differentiation. Vascular smooth muscle cell (VSMC) 3 differentiation is an important process during vasculogenesis and angiogenesis. It is well known that alterations of VSMC phenotype play a role in the progression of several cardiovascular disorders including atherosclerosis, hypertension, and restenosis (1 3). The developmental origins of VSMC are diverse, including both embryonic mesoderm precursors and the neural crest, an ectodermal cell population (4, 5). However, the molecular mechanisms controlling the differentiation and development of VSMC, * This work was supported by American Heart Association Scientist Development grant (to S. C.) and Grants R01HL65681 (to R. J. L.) and R01HL (to R. M. D.) from the National Institutes of Health. The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. 1 To whom correspondence should be addressed: Dept. of Pediatrics, Georgetown University Medical School, Bldg. D, Rm. 363, 4000 Reservoir Rd. NW, Washington, DC Tel.: ; Fax: ; sc229@georgetown.edu. 2 Current address: Medical Oncology Research Unit, National Cancer Institute, NNMC, 8901 Wisconsin Ave., Bldg. 8, Rm. 5101, Bethesda, MD The abbreviations used are: VSMC, vascular smooth muscle cell; TGF, transforming growth factor; MAPK, mitogen-activated protein kinase; PI, phosphatidylinositol; DNRhoA, dominant negative RhoA; DAPI, 4,6-diamidino-2-phenylindole. including the intracellular signaling pathways and transcriptional regulation of VSMC-specific gene expression, are largely unknown. It is clear that the identification of these mechanisms may enhance our understanding of VSMC phenotypic regulation and further provide potential therapeutic or preventive targets for cardiovascular diseases. In vivo studies have shown that TGF- signaling plays a vital role in VSMC function. TGF- 1, TGF- receptors (including endoglin, T R- II, T R-I and Alk1), and signaling intermediates have all been identified by homologous recombination in the mouse as critical for embryonic angiogenesis. Loss of any of these components leads to defects in smooth muscle cell recruitment and/or differentiation during angiogenesis in the developing embryo and extraembryonic structures (6). In vitro studies have shown that TGF- 1 can induce differentiation of C3H10T1/2 mouse embryonic fibroblasts to a smooth muscle phenotype directly (7). We recently reported that TGF- induced a contractile VSMC phenotype from neural crest stem cell line Monc-1 and that Smad proteins appeared to be critical in TGF- -induced VSMC differentiation (8). TGF- signaling is initiated by ligand binding to the transmembrane receptors, T R-I and T R-II, and transduced predominantly by phosphorylation of receptor-associated Smad proteins (R-Smads) (9, 10). R-Smads combine with the common Smad, Smad4, and translocate into the nucleus where they function as transcription factors alone or in association with other DNA binding factors (10). We have demonstrated that Smads play a critical role in TGF- -induced VSMC differentiation from neural crest cells in vitro (8). Much work clearly demonstrates, however, that other pathways including those activated by MAP and JNK kinases, PI3 kinase and RhoA also mediate TGF- signaling (11, 12). The small GTP-binding protein RhoA is expressed in VSMC. RhoA activation was observed in cultured VSMC cells with a contractile phenotype (13, 14). Other studies have shown that signaling through RhoA controls VSMC function, at least in part, through modulating myocardin/srf-dependent transcription of SMC marker genes (15, 16). Recent studies suggest that RhoA downstream target PKN interacts with p38 MAPK to regulate the SMC marker promoter activity in rat pulmonary arterial smooth muscle cell line (17). It has been suggested also that the loss of RhoA expression observed in pulmonary arteries from rats with chronic pulmonary hypertension could be involved in pulmonary artery VSMC dedifferentiation and pulmonary artery remodeling (18, 19). Similarly, coronary VSMC differentiation from proepicardial cells also requires RhoA-mediated actin reorganization and Rho kinase activity (19). However, It is not known whether RhoA cross-talks with Smad signaling in TGF- -induced VSMC differentiation. In the present studies, we have evaluated the function of non-canonical pathways in the differentiation of TGF- -treated Monc-1 cells. We demonstrate that RhoA signaling plays a critical role in TGF- -induced VSMC differentiation. RhoA kinase (ROCK) inhibitor significantly JANUARY 20, 2006 VOLUME 281 NUMBER 3 JOURNAL OF BIOLOGICAL CHEMISTRY 1765
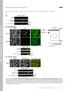
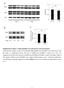
2 blocked VSMC maker gene expression and abolished the contractility of VSMC induced by TGF-. In addition, we demonstrate for the first time that RhoA signaling modulates Smad function during VSMC differentiation through enhancing Smad phosphorylation and regulating Smadmediated gene transcription. MATERIALS AND METHODS Reagents -SMA, -tubulin monoclonal antibodies, and secondary antibodies were purchased from Sigma. Affinity-purified rabbit polyclonal SM22 antiserum was generously provided by Dr. Mario Gimona (Salzburg, Austria). Smad2 monoclonal antibody was obtained from BD Bioscience. Rabbit anti-smad3 antibody was purchased from Zymed Laboratories Inc.. Phospho-Smad2 antibody was from Upstate Biotechnology. Phospho-Smad3 antibody was a generous gift from Dr. Ed Leof (Mayo Clinic). Monoclonal RhoA antibody was purchased from Pierce. Clostridium botulinum C3 exotoxin was from List Biological Laboratories, Inc. Dominant negative mutant Rho A T19N adenovirus (DNRhoA) was a gift from Dr. Tetsuaki Hirase (Kobe University Graduate School of Medicine, Kobe, Japan) (20). Constitutively activated RhoA G14V adenovirus (ACRhoA) was generously provided by Dr. Aviv Hassid (University of Tennessee Health Science Center, Memphis, Tennessee) (21). SBE promoter construct was a gift from Dr. Peter ten Dijke (Leiden University Medical Center, Leiden, The Netherlands). Cell Culture, Adenovirus Infection, and Transfection Monc-1 and C3H10T1/2 cell culture were performed as previously described (8, 22). Ligand-induced contractility of TGF- -induced Monc-1 cells was monitored as follows: Monc-1 cells were treated under indicated conditions for 3 days, and then washed with phosphate-buffered saline, followed by stimulation with DM containing 1 mm carbachol for 1 min. Contractility of SMCs was observed with Nikon Microscope, and the same fields before and after carbachol treatment were imaged (8). DNRhoA, ACRhoA, or green fluorescent protein adenovirus infection was performed following published procedures (20, 21). Monc-1 cells were infected at 200 plaque-forming units/cell (23), and C3H10T1/2 cells were infected at 100 plaque-forming units/cell. The infection was maintained for 48 h prior to TGF- treatment. C3H10T1/2 cell transfection of SBE plasmid and luciferase assay were performed as previously described (22). RhoA Activation Assay RhoA activation was determined by affinity precipitation of the active GTP-bound RhoA using a glutathione S-transferase (GST) fusion protein of the Rho-binding domain of the Rho effector rhotekin (GST-RBD) following the manufacturer s instruction as described in RhoA activation kit (Pierce). Detection of RhoA was performed by Western blot using anti-rhoa antibody. Immunocytofluorescence, Western Blot Analysis, and Reverse Transcription-PCR All analyses were performed as previously described (8). PCR primer sequences for -SMA, S 22, and calponin have been previously published (8). Statistics All values are expressed as mean S.E. Data were analyzed using analysis of variance with pairwise comparisons between relevant groups. A level of p values 0.05 was considered statistically significant. RESULTS To study the molecular mechanism of VSMC differentiation, we previously developed an in vitro model showing that TGF- induced the neural crest stem cell line Monc-1 to differentiate into VSMC (8). Further experiments demonstrated that Smad signaling was essential for smooth muscle differentiation. In addition to Smads, other signaling pathways such as p38 MAPK, p42/44 MAPK, PI3 kinase, and RhoA have FIGURE 1. Effect of ROCK inhibitor Y27632 on TGF- -induced VSMC differentiation. A, effect of Y27632 and PI3 kinase inhibitor LY on TGF- or serum-induced VSMC marker expression. Monc-1 neural crest stem cells were treated without ( ) or with ( ) TGF- (5 ng/ml) or cultured with 10% serum in M199 medium. Cells were simultaneously treated with ( ) or without ( ) Y27632 (10 M) or LY (20 M) for 2 days. Cell lysates were prepared, and Western blotting was performed using indicated antibodies. B, effect of Y27632 on TGF- -induced expression of other VSMC markers. Monc-1 cells were treated with ( ) or without ( ) TGF- (5 ng/ml) in the absence ( ) or presence ( ) of Y27632 (10 M) for 2 days. Total RNA was prepared, and reverse transcription-pcr was performed using primers for -SMA, SM22, or calponin. Cyclophilin serves as an internal control. C, the synergistic effect of Y27632 and AlK5 inhibitor SB on seruminduced -SMA expression. Monc-1 cells were cultured in medium with ( ) or without ( ) serum and treated with ( ) or without ( ) Y27632 (10 M) or SB (10 M) for 2 days. Western blotting was performed. also been shown to mediate TGF- function (11, 12). We sought to determine whether these pathways were important for TGF- -induced VSMC differentiation. We used kinase inhibitors to individually block each signaling pathway in Monc-1 cells treated by TGF-, and then examined the expression of VSMC-specific markers. p38 MAPK, p44/42 MAPK, and PI3 kinase inhibitors did not inhibit TGF- induction of smooth muscle -actin ( -SMA), suggesting that p38 MAPK, p44/42 MAPK (data not shown), or PI3 kinase signaling (Fig. 1A) are not important for VSMC differentiation from Monc-1 cells. The ROCK inhibitor Y27632, however, significantly blocked TGF- -induced -SMA expression (Fig. 1A). To determine the extent of this effect, reverse transcription-pcr analysis of additional markers was performed. We found that Y27632 inhibited the expression of VSMC markers -SMA, SM22 and calponin (Fig. 1B). These results suggest that RhoA signaling plays an important role in TGF- -induced VSMC differentiation. We and others previously reported that treatment of Monc-1 cells with serum also induced VSMC markers (8, 24), although a contractile phenotype was not observed with serum treatment (8). Similar to results obtained with TGF-, VSMC marker expression induced by serum was also blocked following Y27632 treatment (Fig. 1A), suggesting that RhoA signaling plays a role in serum-induced VSMC differentiation also. To detect whether RhoA and TGF- have a synergistic role in serum-induced SMC differentiation, we used Y27632 and TGF- type I receptor Alk5 inhibitor, SB431542, to treat serum-induced Monc-1 cells individually or in combination. We found that the maker gene expression was significantly inhibited by Y27632 or SB SB had a much greater effect than Y27632 (Fig. 1C). In addition, the combinatorial use of two inhibitors completely blocked the marker gene expres JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 281 NUMBER 3 JANUARY 20, 2006
3 FIGURE 2. Effect of ROCK inhibitor Y27632 on TGF- -induced VSMC morphology and contractility. Monc-1 cells were treated without (DM) or with TGF- (5 ng/ml) or TGF- and Y27632 (10 M) as indicated for 3 days. Cells were washed two times with phosphate-buffered saline and treated with 1 mm carbachol for 1 min. Images were acquired before (A, C, and E) and after (B, D, and F) carbachol addition. Arrows and numbers show representative contracted cells in TGF- -treated group. Scale bar 50 m. sion (Fig. 1C), suggesting that both RhoA and TGF- are critical and that they have a synergistic role in serum-induced SMC differentiation. Our previous studies showed that TGF- induced a spindle-like contractile VSMC phenotype (8). To determine whether RhoA signaling is important in this phenotypic conversion, we performed carbachol-induced contraction assay in Monc-1 cells treated with or without TGF- and Y In this assay, cells were treated with carbachol, a muscarinic agonist, and imaged before and after treatment. Cells responsive to carbachol contract during the course of the assay (8). We found that RhoA inhibitor completely reversed TGF- -induced spindle-like morphology of Monc-1 cells (Fig. 2E). Carbachol induced a contraction in cells treated with TGF- (Fig. 2, compare C with D). The addition of Y27632, however, abolished the contractility (Fig. 2, compare E with F). These results suggest that RhoA signaling is necessary for TGF- -induced contractility of the VSMC phenotype. To further investigate how RhoA signaling regulates TGF- -induced VSMC differentiation, we first determined whether TGF- indeed activates RhoA in Monc-1 cells. We performed RhoA activation assay by treating Monc-1 cells with TGF- for various times, and measuring GTP-bound RhoA. As shown in Fig. 3A, GTP-RhoA can be detected as early as 5 min following TGF- treatment. The activation can last up to 16 h, the longest time point measured. There was little change in the level of RhoA expression in TGF- -treated cells (Fig. 3A). C3H10T1/2 cells have been used as an in vitro cell model to study VSMC differentiation (7, 25). Our results showed that TGF- also induced RhoA activation in C3H10T1/2 cells (Fig. 3B). Because both Smad and RhoA signals play important roles in TGF- -induced VSMC differentiation, we sought to determine whether RhoA could cross-talk with Smad signals. We used ROCK inhibitor Y27632 to treat Monc-1 cells before, after, or at same time of TGF- addition. We did not detect the alteration of Smad activation induced by TGF-, although the expression of the SMC marker -SMA was blocked (Fig. 4A). These results suggest that the ROCK may not affect Smad activation in our system. To test whether RhoA affects Smad signaling directly, we infected Monc-1 cells with an adenoviral vector expressing DNRhoA and treated the cells with TGF- for short (30 min and 1 h) or longer (24 h) times and analyzed C-terminal phosphorylation by Western blot. We found that DNRhoA was expressed at a high level in adenovirus-infected cells (Fig. 4B). TGF- induced phosphorylation of both Smad2 and Smad3 at 30 min after TGF- addition (Fig. 4B). In DNRhoA-expressing cells, however, the phosphorylation of Smad2 and Smad3 was reduced significantly at 30 min or 1 h after TGF- induction (Fig. 4B). There was no apparent difference in the level of phosphorylation at 24 h, but the phosphorylation of both Smad proteins was very low at this time even FIGURE 3. TGF- -induced RhoA activation. Monc-1 (A) and C3H10T1/2 (B) cells were treated with TGF- (5 ng/ml) for various times as indicated. Cell lysates were prepared, and GTP-bound RhoA was enriched by absorbing cell lysates to GST-RBD beads and measured by Western blotting using RhoA antibody. Total RhoA and -tubulin in cell lysates were detected by Western blotting as controls. FIGURE 4. RhoA modulates Smad phosphorylation. A, Monc-1 cells were treated with ( ) or without TGF- and simultaneously treated with ( ) or without ( ) Y27632 (10 M) for the times indicated. Cell lysates were prepared, and Western blotting was performed using antibodies for phospho-smad (p-smad), total Smad (Smad), -SMA (indicating SMC differentiation). B, Monc-1 cells were infected with ( ) DNRhoA adenovirus for 48 h or left uninfected ( ). Cells were then treated with ( ) or without ( ) TGF- (5 ng/ml) for various times as indicated. Western blotting was performed using antibodies as indicated. -Tubulin serves as an internal control for equal loading for both A and B. with TGF- alone. Similar to the inhibition of VSMC marker expression by ROCK inhibitor Y27632 (Figs. 1A and 4A), DNRhoA also blocked SMC marker -SMA expression while inhibiting Smad phosphorylation (Fig. 4B). These results demonstrated that RhoA is required for Smad phosphorylation during TGF- -induced VSMC differentiation. To further determine the role of RhoA in Smad activation, we JANUARY 20, 2006 VOLUME 281 NUMBER 3 JOURNAL OF BIOLOGICAL CHEMISTRY 1767
4 FIGURE 5. Effect of DNRhoA on Smad nuclear translocation. Monc-1 cells were plated on coverslips and infected with adenovirus expressing dominant negative RhoA (DNRhoA, 200 plaque-forming units/cell) or left uninfected for 48 h. Cells were then treated without (DM) or with TGF- (5 ng/ml) for 30 min. Immunostaining was performed using Smad2 (A) or Smad3 (B) antibody. 4,6-Diamidino-2-phenylindole (DAPI) stains nuclei of cells. Colocalization of Smads with DAPI (Merge) indicates the nuclear translocation of Smads. Bar 100 m. infected Monc-1 cells with DNRhoA adenovirus followed by TGF- treatment. Immunocytofluorescence staining showed that both Smad2 and Smad3 were translocated into nuclei within 30 min after TGF- treatment (Fig. 5). However, Smad2 and Smad3 nuclear translocation was significantly inhibited in most of the cells infected with DNRhoA adenovirus, suggesting a role of RhoA in regulating Smad location or function (Fig. 5). Following receptor association, Smad proteins are phosphorylated on C-terminal serine residues, and this phosphorylation is essential for nuclear translocation and transcriptional activity (12). To determine whether RhoA regulates Smad functional activity, we used transient transfection assays of a Smad-specific reporter SBE along with adenoviral expression of dominant negative and activating RhoA proteins in C3H10T1/2 cells. We could not use Monc-1 cells in this functional assay because we found that combination of transfection and virus infection in Monc-1 caused significant cell death. C3H10T1/2 cells have been shown to differentiate to SMCs with TGF- (7), to activate RhoA with TGF- stimulation as do Monc-1 cells (Fig. 3B), and to have a significantly higher transfection efficiency than Monc-1 cells. C3H10T1/2 cells were transfected with the reporter, infected with adenovirus expressing green fluorescent protein, dominant negative RhoA (DNRhoA) or constitutively activated RhoA (ACRhoA), and then treated with TGF- or left untreated. Luciferase assays showed that DNRhoA significantly attenuated Smad-mediated promoter activation, whereas ACRhoA significantly enhanced Smad-mediated gene transcription (Fig. 6A). To confirm the role of RhoA in regulating functional activity of Smads, we transfected the SBE reporter into C3H10T1/2 cells, and the cells were then treated with a RhoA specific inhibitor C3 exotoxin followed by TGF- induction (26). Luciferase assay showed that C3 exotoxin also significantly inhibited TGF- -induced SBE promoter activity (Fig. 6B). To test C3 exotoxin function on Smad activation, we treated Monc-1 cells with C3 exotoxin followed by TGF- addition and performed FIGURE 6. RhoA modulates Smad-mediated gene transcription in C3H10T1/2 cells. A, effect of RhoA on SBE promoter activity. C3H10T1/2 cells were transfected with SBE luciferase reporter construct followed by infection of adenovirus (100 plaque-forming units/ml) expressing green fluorescent protein (GFP), DNRhoA, or ACRhoA for 48 h. Cells were then treated without (control) or with TGF- (5 ng/ml) for 16 h, and luciferase assay was performed. Data shown is one of the three independent experiments. *, p 0.01 for comparison to green fluorescent protein expressing cells. B, effect of C3 exotoxin on SBE promoter activity. C3H10T1/2 cells were transfected with SBE luciferase reporter construct followed by addition of C3 exotoxin (5 g/ml) for 16 h. Cells were then left untreated (control) or treated with TGF- (5 ng/ml) for 8 h, and luciferase assay was performed. Data shown is a representative of two independent experiments. *, p 0.05 for comparison to C3 untreated group. immunostaining with Smad antibodies. We found that TGF- induced Smad3 nuclear translocation, indicating Smad phosphorylation. C3 exotoxin treatment, however, significantly decreased the nuclear accumulation of Smad3 (Fig. 7), indicating that C3 exotoxin inhibited Smad3 phosphorylation. Similarly, C3 exotoxin also inhibited Smad2 phosphorylation (data not shown). Taken together, these results demonstrate that RhoA modulates Smad-mediated gene transcription through modifying Smad phosphorylation. Previous studies have shown that Rho signaling is important for the down-regulation of c-myc protein and up-regulation of p21 protein in TGF- -mediated growth inhibition of breast cancer cell MCF10CA1h (32). To determine whether these cell cycle regulators are involved in TGF- -induced SMC differentiation and whether they are regulated in a RhoA-dependent manner in our system, we overexpressed DNRhoA in Monc-1 cells with adenovirus infection and then treated the cells with TGF-. Similar to the previous report (32), we found that TGF- significantly up-regulated p21 but down-regulated c-myc expression in Monc-1 cells. Overexpression of DNRhoA, however, blocked p21 but increased c-myc protein expression (Fig. 8). These data suggest that as in other cell types (32), TGF- regulates p21 and c-myc and that these factors may play a role in TGF- -induced SMC differentiation from neural crest cells. RhoA signaling plays an important role in regulating the expression of these cell cycle regulators. DISCUSSION We previously developed an in vitro VSMC differentiation model for the study of TGF- induction of a contractile VSMC phenotype (8). In 1768 JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 281 NUMBER 3 JANUARY 20, 2006
5 FIGURE 7. Effect of RhoA inhibitor C3 exotoxin on Smad3 activation. Monc-1 cells were plated on coverslips and treated with (C3) or without C3 exotoxin (5 g/ml) for 16 h. Cells were then treated without (DM) or with TGF- (5 ng/ml) for 30 min. Immunostaining was performed using Smad3 antibody. Colocalization of Smad3 with DAPI (Merge) indicates the nuclear translocation of Smad3. Bar 100 m. FIGURE 9. Schematic overview of the role of Smad and RhoA in TGF- -induced VSMC differentiation from neural crest cells. FIGURE 8. Expression of cell cycle regulators p21 and c-myc in Monc-1 cells. Monc-1 cells were infected with ( ) DNRhoA adenovirus for 48 h or left uninfected ( ). Cells were then treated with ( ) or without ( ) TGF- (5 ng/ml) for various times (hours) as indicated. Western blotting was performed using antibodies as indicated. -Tubulin serves as an internal control for equal loading. this model, Monc-1 neural crest stem cells are treated with TGF-, and a contractile SMC phenotype results. We further demonstrated that Smad signaling played an important role in this process (8). Because recent studies suggest that other signaling pathways are important for TGF- function also, the goal of this study was to identify other pathway(s) important for TGF- -induced VSMC differentiation. To this end, our data demonstrated that in addition to Smads, RhoA signaling is another pathway critical to TGF- induction of VSMC differentiation. The blockade of RhoA signaling with ROCK inhibitor Y27632 significantly inhibited expression of the VSMC markers -SMA, SM22, and calponin. ROCK inhibitor Y27632 also blocked serum-induced VSMC marker expression. We previously reported that TGF- played a critical role in serum-induced VSMC differentiation, which was demonstrated by the blockade of serum effect with type I receptor Alk5-specific inhibitor as well as TGF- -neutralizing antibodies (8). In the present studies, we further demonstrated that RhoA and TGF- have a synergistic effect on serum-induced SMC differentiation. Contraction is a primary function of VSMC. VSMC induced from Monc-1 cells by TGF- displayed contraction upon treatment with the muscarinic agonist carbachol. ROCK inhibitor Y27632 completely reversed the morphological change and abolished the carbachol-induced contraction, suggesting that RhoA plays an important role in the contractility of VSMC induced by TGF-. This finding is consistent with the previous studies of RhoA activity on SMC function (14, 15). RhoA has been shown to regulate coronary or cultured SMC differentiation via cytoskeleton reorganization or actin polymerization through SRF-dependent transcription (19). RhoA is also critical in the regulation of contractility of cultured SMC cells (14). Our results provide direct evidence for the role of RhoA signaling in differentiation and contraction of VSMC developed from neural crest stem cells. The Smad signaling pathway is the canonical transduction route downstream of TGF- receptors. However, understanding cross-talk between Smad and other signaling pathways is necessary for determining how TGF- regulates cellular function. Many studies have tried to link Smad signaling with RhoA activity, especially in TGF- -induced cytoskeletal organization and epithelial mesenchymal transdifferentiation (EMT) (27 33). Although most of the studies showed that actin reorganization and EMT were mediated through Smad-independent RhoA signaling, several evidences showed that Smad was involved in RhoA function. Shen et al. (33) reported that dominant negative Smad3 inhibited the induction of guanine exchange factor NET and the activation of Rho activity. Edlund et al. (29) found that Smad7 was required for activation of Cdc42, a small GTPase that plays a role in cytoskeleton rearrangement. It is possible that in our system Smad signaling also plays a role in regulating RhoA function. The rapid production of GTPbound RhoA by TGF-, however, suggests that the initial activation of RhoA is likely to be Smad-independent. Whether later RhoA-specific effects are dependent on Smad signaling remains to be determined. Other evidence suggests that the Rho pathway may also regulate Smad activation. Recent studies by Kamaraju et al. (32) revealed that the Rho/ROCK pathway affects the linker region phosphorylation of Smad2/3 in human breast carcinoma cells. The linker region of Smad2/3 was shown to contain phosphorylation sites for MAPK or Cdk2/4. However, C-terminal phosphorylation of Smad2 and Smad3 and Smad-mediated promoter activation were not altered by ROCK inhibitor in these cells (32). Smads are known to be activated by receptor kinases through C-terminal phosphorylation, followed by nuclear translocation and regulation of gene transcription (12). Our studies demonstrate for the first time that RhoA directly modulates Smad activity during TGF- -induced VSMC differentiation. Dominant negative RhoA as well as RhoA inhibitor C3 exotoxin inhibited Smad2 and Smad3 C-terminal phosphorylation or nuclear translocation. DNRhoA and C3 exotoxin also significantly attenuated Smad-mediated gene activation. Furthermore, constitutively activated RhoA enhanced Smadmediated promoter activity. Most importantly, VSMC marker expression was regulated simultaneously with the modulation of Smad activity, indicating that RhoA may regulate VSMC differentiation through altering Smad activation. Together, these data demonstrate that RhoA can influence Smad signaling either by modulating phosphorylation in the linker region (32) or in the C terminus. The effects may be JANUARY 20, 2006 VOLUME 281 NUMBER 3 JOURNAL OF BIOLOGICAL CHEMISTRY 1769
6 cell type-specific and may serve to help RhoA signaling modulate TGF- function. The fact that Smad and RhoA signals are both necessary for VSMC marker induction further suggests that both pathways must be activated for smooth muscle differentiation from neural crest cells (Fig. 9). RhoA function in VSMC contractility appears to be independent of its role in modulating Smad activity. This hypothesis is supported by our data showing that ROCK inhibitor Y37632 blocked SMC marker gene activation (Fig. 1) and abolished SMC contraction (Fig. 2) but did not affect Smad phosphorylation (Fig. 4A). In addition to Rho kinase ROCK, another key downstream target of RhoA is a member of the protein kinase PKN family (17). Because the affinity of Y27632 for ROCK is at least times higher than that for PKN (34), Y27632 likely has little effect on PKN while sufficiently blocking ROCK activity. ROCK appears to be important for SMC contraction (14, 15), whereas PKN could mediate RhoA function in modulating Smad activation. PKN has been found to play a role in TGF- -induced promoter activities of SMC marker genes (17). An alternative pathway for RhoA function is myocardin/srf-dependent mechanism (15). How RhoA, TGF-, and myocardin pathways converge to regulate VSMC differentiation and contractility, and whether RhoA modulates Smad activity through PKN remains to be determined. Future work will be directed toward understanding the mechanism by which RhoA induces VSMC contraction and the role of RhoA in modulating Smad activity, especially how RhoA interacts with other components in the Smad signaling pathway. Acknowledgments We thank Dr. Aviv Hassid for generously providing the adenovirus vector expressing activated RhoA, Dr. Tetsuaki Hirase for providing us with the dominant negative RhoA, and others as indicated in the text for sharing reagents. REFERENCES 1. Kocher, O., and Gabbiani, G. (1986) Hum. Pathol. 17, Ross, R. (1993) Nature 362, Kocher, O., Gabbiani, F., Gabbiani, G., Reidy, M. A., Cokay, M. S., Peters, H., and Huttner, I. (1991) Lab. Investig. 65, Hungerford, J. E., and Little, C. D. (1999) J. Vasc. Res. 36, Christiansen, J. H., Coles, E. G., and Wilkinson, D. G. (2000) Curr. Opin. Cell Biol. 12, Goumans, M. J., and Mummery, C. (2000) Int. J. Dev. Biol. 44, Hirschi, K. K., Rohovsky, S. A., and D Amore, P. A. (1998) J. Cell Biol. 141, Chen, S., and Lechleider, R. J. (2004) Circ. Res. 94, Piek, E., Heldin, C. H., and Ten Dijke, P. (1999) FASEB J 13, Massague, J., and Wotton, D. (2000) EMBO J. 19, Mulder, K. M. (2000) Cytokine Growth Factor Rev. 11, Derynck, R., and Zhang, Y. E. (2003) Nature 425, Worth, N. F., Campbell, G. R., and Rolfe, B. E. (2001) Ann. N. Y. Acad. Sci. 947, Bi, D., Nishimura, J., Niiro, N., Hirano, K., and Kanaide, H. (2005) Circ. Res 96, Wamhoff, B. R., Bowles, D. K., McDonald, O. G., Sinha, S., Somlyo, A. P., Somlyo, A. V., and Owens, G. K. (2004) Circ. Res. 95, Mack, C. P., Somlyo, A. V., Hautmann, M., Somlyo, A. P., and Owens, G. K. (2001) J. Biol. Chem. 276, Deaton, R. A., Su, C., Valencia, T. G., and Grant, S. R. (2005) J. Biol. Chem. 280, Sauzeau, V., Rolli-Derkinderen, M., Lehoux, S., Loirand, G., and Pacaud, P. (2003) Circ. Res. 93, Lu, J., Landerholm, T. E., Wei, J. S., Dong, X. R., Wu, S. P., Liu, X., Nagata, K., Inagaki, M., and Majesky, M. W. (2001) Dev. Biol. 240, Liu, Y., Suzuki, Y. J., Day, R. M., and Fanburg, B. L. (2004) Circ. Res. 95, Desai, L. P., Aryal, A. M., Ceacareanu, B., Hassid, A., and Waters, C. M. (2004) Am. J. Physiol. 287, L1134 L Chen, S., Kulik, M., and Lechleider, R. J. (2003) Nucleic Acids Res. 31, Klesse, L. J., and Parada, L. F. (1998) J. Neurosci. 18, Jain, M. K., Layne, M. D., Watanabe, M., Chin, M. T., Feinberg, M. W., Sibinga, N. E., Hsieh, C. M., Yet, S. F., Stemple, D. L., and Lee, M. E. (1998) J. Biol. Chem. 273, Bostrom, K., Tintut, Y., Kao, S. C., Stanford, W. P., and Demer, L. L. (2000) J. Cell. Biochem. 78, Watts, K. L., and Spiteri, M. A. (2004) Am. J. Physiol. 287, L1323 L Edlund, S., Landstrom, M., Heldin, C. H., and Aspenstrom, P. (2002) Mol. Biol. Cell 13, Nawshad, A., LaGamba, D., and Hay, E. D. (2004) Arch. Oral Biol. 49, Edlund, S., Landstrom, M., Heldin, C. H., and Aspenstrom, P. (2004) J. Cell Sci. 117, Bhowmick, N. A., Ghiassi, M., Bakin, A., Aakre, M., Lundquist, C. A., Engel, M. E., Arteaga, C. L., and Moses, H. L. (2001) Mol. Biol. Cell 12, Blindt, R., Bosserhoff, A. K., Dammers, J., Krott, N., Demircan, L., Hoffmann, R., Hanrath, P., Weber, C., and Vogt, F. (2004) Cardiovasc. Res. 62, Kamaraju, A. K., and Roberts, A. B. (2005) J. Biol. Chem. 280, Shen, X., Li, J., Hu, P. P., Waddell, D., Zhang, J., and Wang, X. F. (2001) J. Biol. Chem. 276, Ishizaki, T., Uehata, M., Tamechika, I., Keel, J., Nonomura, K., Maekawa, M., and Narumiya, S. (2000) Mol. Pharmacol. 57, JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 281 NUMBER 3 JANUARY 20, 2006
During vascular development, endothelial cells form a
 Transforming Growth Factor- Induced Differentiation of Smooth Muscle From a Neural Crest Stem Cell Line Shiyou Chen, Robert J. Lechleider Abstract During vascular development, nascent endothelial networks
Transforming Growth Factor- Induced Differentiation of Smooth Muscle From a Neural Crest Stem Cell Line Shiyou Chen, Robert J. Lechleider Abstract During vascular development, nascent endothelial networks
Ma, et al. Supplemental Data
 Ma, et al Supplemental Data Title: Calpain mediates pulmonary vascular remodeling in rodent models of pulmonary hypertension and its inhibition attenuates pathologic features of disease Authors: Wanli
Ma, et al Supplemental Data Title: Calpain mediates pulmonary vascular remodeling in rodent models of pulmonary hypertension and its inhibition attenuates pathologic features of disease Authors: Wanli
Sarker et al. Supplementary Material. Subcellular Fractionation
 Supplementary Material Subcellular Fractionation Transfected 293T cells were harvested with phosphate buffered saline (PBS) and centrifuged at 2000 rpm (500g) for 3 min. The pellet was washed, re-centrifuged
Supplementary Material Subcellular Fractionation Transfected 293T cells were harvested with phosphate buffered saline (PBS) and centrifuged at 2000 rpm (500g) for 3 min. The pellet was washed, re-centrifuged
CDK5 is essential for TGF-β1-induced epithelial-mesenchymal transition and breast cancer progression
 Supplementary information for: CDK5 is essential for TGF-β1-induced epithelial-mesenchymal transition and breast cancer progression Qian Liang, Lili Li, Jianchao Zhang, Yang Lei, Liping Wang, Dong-Xu Liu,
Supplementary information for: CDK5 is essential for TGF-β1-induced epithelial-mesenchymal transition and breast cancer progression Qian Liang, Lili Li, Jianchao Zhang, Yang Lei, Liping Wang, Dong-Xu Liu,
Post-translational modification
 Protein expression Western blotting, is a widely used and accepted technique to detect levels of protein expression in a cell or tissue extract. This technique measures protein levels in a biological sample
Protein expression Western blotting, is a widely used and accepted technique to detect levels of protein expression in a cell or tissue extract. This technique measures protein levels in a biological sample
Input. Pulldown GST. GST-Ahi1
 A kd 250 150 100 75 50 37 25 -GST-Ahi1 +GST-Ahi1 kd 148 98 64 50 37 22 GST GST-Ahi1 C kd 250 148 98 64 50 Control Input Hap1A Hap1 Pulldown GST GST-Ahi1 Hap1A Hap1 Hap1A Hap1 Figure S1. (A) Ahi1 western
A kd 250 150 100 75 50 37 25 -GST-Ahi1 +GST-Ahi1 kd 148 98 64 50 37 22 GST GST-Ahi1 C kd 250 148 98 64 50 Control Input Hap1A Hap1 Pulldown GST GST-Ahi1 Hap1A Hap1 Hap1A Hap1 Figure S1. (A) Ahi1 western
Species predicted to react based on 100% sequence homology: Chicken, Bovine, Dog.
 1 of 5 11/1/2013 10:25 PM Product Pathways - Jak/Stat Pathway Phospho-Stat3 (Tyr705) Antibody #9131 Have you tried your application using our XP monoclonal antibodies? Try products: 9145 PhosphoSitePlus
1 of 5 11/1/2013 10:25 PM Product Pathways - Jak/Stat Pathway Phospho-Stat3 (Tyr705) Antibody #9131 Have you tried your application using our XP monoclonal antibodies? Try products: 9145 PhosphoSitePlus
Mechanisms of embryonic stem cell division and differentiation
 Mechanisms of embryonic stem cell division and differentiation Josephine Wbite.'.... Department of Molecular Biosciences (Biochemistry) The University of Adelaide Adelaide, South Australia Submitted for
Mechanisms of embryonic stem cell division and differentiation Josephine Wbite.'.... Department of Molecular Biosciences (Biochemistry) The University of Adelaide Adelaide, South Australia Submitted for
Wnt16 smact merge VK/AB
 A WT Wnt6 smact merge VK/A KO ctrl IgG WT KO Wnt6 smact DAPI SUPPLEMENTAL FIGURE I: Wnt6 expression in MGP-deficient aortae. Immunostaining for Wnt6 and smooth muscle actin (smact) in aortae from 7 day
A WT Wnt6 smact merge VK/A KO ctrl IgG WT KO Wnt6 smact DAPI SUPPLEMENTAL FIGURE I: Wnt6 expression in MGP-deficient aortae. Immunostaining for Wnt6 and smooth muscle actin (smact) in aortae from 7 day
Fig. S1. Effect of p120-catenin overexpression on the interaction of SCUBE2 with E-cadherin. The expression plasmid encoding FLAG.
 Fig. S1. Effect of p120-catenin overexpression on the interaction of SCUBE2 with E-cadherin. The expression plasmid encoding FLAG.SCUBE2, E-cadherin.Myc, or HA.p120-catenin was transfected in a combination
Fig. S1. Effect of p120-catenin overexpression on the interaction of SCUBE2 with E-cadherin. The expression plasmid encoding FLAG.SCUBE2, E-cadherin.Myc, or HA.p120-catenin was transfected in a combination
Supplemental Information. Pacer Mediates the Function of Class III PI3K. and HOPS Complexes in Autophagosome. Maturation by Engaging Stx17
 Molecular Cell, Volume 65 Supplemental Information Pacer Mediates the Function of Class III PI3K and HOPS Complexes in Autophagosome Maturation by Engaging Stx17 Xiawei Cheng, Xiuling Ma, Xianming Ding,
Molecular Cell, Volume 65 Supplemental Information Pacer Mediates the Function of Class III PI3K and HOPS Complexes in Autophagosome Maturation by Engaging Stx17 Xiawei Cheng, Xiuling Ma, Xianming Ding,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3240 Supplementary Figure 1 GBM cell lines display similar levels of p100 to p52 processing but respond differentially to TWEAK-induced TERT expression according to TERT promoter mutation
DOI: 10.1038/ncb3240 Supplementary Figure 1 GBM cell lines display similar levels of p100 to p52 processing but respond differentially to TWEAK-induced TERT expression according to TERT promoter mutation
Polyclonal ARHGAP25 antibody was prepared from rabbit serum after intracutaneous
 Preparation and purification of polyclonal antibodies Polyclonal ARHGAP25 antibody was prepared from rabbit serum after intracutaneous injections of glutathione S-transferase-ARHGAP25-(509-619) (GST-coiled
Preparation and purification of polyclonal antibodies Polyclonal ARHGAP25 antibody was prepared from rabbit serum after intracutaneous injections of glutathione S-transferase-ARHGAP25-(509-619) (GST-coiled
Nature Biotechnology: doi: /nbt Supplementary Figure 1
 Supplementary Figure 1 Schematic and results of screening the combinatorial antibody library for Sox2 replacement activity. A single batch of MEFs were plated and transduced with doxycycline inducible
Supplementary Figure 1 Schematic and results of screening the combinatorial antibody library for Sox2 replacement activity. A single batch of MEFs were plated and transduced with doxycycline inducible
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2271 Supplementary Figure a! WM266.4 mock WM266.4 #7 sirna WM266.4 #10 sirna SKMEL28 mock SKMEL28 #7 sirna SKMEL28 #10 sirna WM1361 mock WM1361 #7 sirna WM1361 #10 sirna 9 WM266. WM136
DOI: 10.1038/ncb2271 Supplementary Figure a! WM266.4 mock WM266.4 #7 sirna WM266.4 #10 sirna SKMEL28 mock SKMEL28 #7 sirna SKMEL28 #10 sirna WM1361 mock WM1361 #7 sirna WM1361 #10 sirna 9 WM266. WM136
Cancer cells that survive radiation therapy acquire HIF-1 activity and translocate toward tumor blood vessels Supplementary Information
 Cancer cells that survive radiation therapy acquire HIF-1 activity and translocate toward tumor blood vessels Supplementary Information 1. Supplementary Figure S1-S10: Pages 2-11 2. Supplementary References:
Cancer cells that survive radiation therapy acquire HIF-1 activity and translocate toward tumor blood vessels Supplementary Information 1. Supplementary Figure S1-S10: Pages 2-11 2. Supplementary References:
Comparison of Commercial Transfection Reagents: Cell line optimized transfection kits for in vitro cancer research.
 Comparison of Commercial Transfection Reagents: Cell line optimized transfection kits for in vitro cancer research. by Altogen Labs, 11200 Manchaca Road, Suite 203 Austin TX 78748 USA Tel. (512) 433-6177
Comparison of Commercial Transfection Reagents: Cell line optimized transfection kits for in vitro cancer research. by Altogen Labs, 11200 Manchaca Road, Suite 203 Austin TX 78748 USA Tel. (512) 433-6177
At E17.5, the embryos were rinsed in phosphate-buffered saline (PBS) and immersed in
 Supplementary Materials and Methods Barrier function assays At E17.5, the embryos were rinsed in phosphate-buffered saline (PBS) and immersed in acidic X-gal mix (100 mm phosphate buffer at ph4.3, 3 mm
Supplementary Materials and Methods Barrier function assays At E17.5, the embryos were rinsed in phosphate-buffered saline (PBS) and immersed in acidic X-gal mix (100 mm phosphate buffer at ph4.3, 3 mm
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Thompson et al., http://www.jcb.org/cgi/content/full/jcb.200909067/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Modification-specific antibodies do not detect unmodified
Supplemental material Thompson et al., http://www.jcb.org/cgi/content/full/jcb.200909067/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Modification-specific antibodies do not detect unmodified
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/9/eaat5401/dc1 Supplementary Materials for GLK-IKKβ signaling induces dimerization and translocation of the AhR-RORγt complex in IL-17A induction and autoimmune
advances.sciencemag.org/cgi/content/full/4/9/eaat5401/dc1 Supplementary Materials for GLK-IKKβ signaling induces dimerization and translocation of the AhR-RORγt complex in IL-17A induction and autoimmune
Supplementary Table 1. The Q-PCR primer sequence is summarized in the following table.
 Supplementary Table 1. The Q-PCR primer sequence is summarized in the following table. Name Sequence (5-3 ) Application Flag-u ggactacaaggacgacgatgac Shared upstream primer for all the amplifications of
Supplementary Table 1. The Q-PCR primer sequence is summarized in the following table. Name Sequence (5-3 ) Application Flag-u ggactacaaggacgacgatgac Shared upstream primer for all the amplifications of
Data Sheet. SBE Reporter Kit (TGFβ/SMAD signaling pathway) Catalog #: 60654
 Data Sheet SBE Reporter Kit (TGFβ/SMAD signaling pathway) Catalog #: 60654 Background The transforming growth factor beta (TGFβ) signaling pathway is involved in a diverse range of cell processes such
Data Sheet SBE Reporter Kit (TGFβ/SMAD signaling pathway) Catalog #: 60654 Background The transforming growth factor beta (TGFβ) signaling pathway is involved in a diverse range of cell processes such
Nature Structural & Molecular Biology: doi: /nsmb.1583
 Acetylation by GCN5 regulates CDC6 phosphorylation in the S-phase of the cell cycle Roberta Paolinelli 1,2, Ramiro Mendoza-Maldonado 2, Anna Cereseto 1 and Mauro Giacca 2 1 Molecular Biology Laboratory,
Acetylation by GCN5 regulates CDC6 phosphorylation in the S-phase of the cell cycle Roberta Paolinelli 1,2, Ramiro Mendoza-Maldonado 2, Anna Cereseto 1 and Mauro Giacca 2 1 Molecular Biology Laboratory,
3 P p25. p43 p41 28 FADD. cflips. PE-Cy5 [Fluorescence intensity]
![3 P p25. p43 p41 28 FADD. cflips. PE-Cy5 [Fluorescence intensity] 3 P p25. p43 p41 28 FADD. cflips. PE-Cy5 [Fluorescence intensity]](/thumbs/87/97394430.jpg) L S p4 3 D3 76 N S L Ve ct or p4 3 D3 76 N S L Ve ct or A aspase 8 FADD TRAF2 D95-R - + Vector D95L TL I S L D376N T RAIL-R1 T RAIL-R2 D95-R E-y5 [Fluorescence intensity] Supplemental Fig. 1 Different
L S p4 3 D3 76 N S L Ve ct or p4 3 D3 76 N S L Ve ct or A aspase 8 FADD TRAF2 D95-R - + Vector D95L TL I S L D376N T RAIL-R1 T RAIL-R2 D95-R E-y5 [Fluorescence intensity] Supplemental Fig. 1 Different
Supplementary Figure 1 Phosphorylated tau accumulates in Nrf2 (-/-) mice. Hippocampal tissues obtained from Nrf2 (-/-) (10 months old, 4 male; 2
 Supplementary Figure 1 Phosphorylated tau accumulates in Nrf2 (-/-) mice. Hippocampal tissues obtained from Nrf2 (-/-) (10 months old, 4 male; 2 female) or wild-type (5 months old, 1 male; 11 months old,
Supplementary Figure 1 Phosphorylated tau accumulates in Nrf2 (-/-) mice. Hippocampal tissues obtained from Nrf2 (-/-) (10 months old, 4 male; 2 female) or wild-type (5 months old, 1 male; 11 months old,
CIGNAL REPORTER ASSAYS & ARRAYS
 CIGNAL REPORTER ASSAYS & ARRAYS TM Cell-Based for Measuring Activity Cancer Cell Cycle Cytokines / Inflammation Neuroscience Stem Cell / Development Toxicology Focus on your CIGNAL TM REPORTER ASSAY SYSTEM
CIGNAL REPORTER ASSAYS & ARRAYS TM Cell-Based for Measuring Activity Cancer Cell Cycle Cytokines / Inflammation Neuroscience Stem Cell / Development Toxicology Focus on your CIGNAL TM REPORTER ASSAY SYSTEM
AIP1 functions as an endogenous inhibitor of VEGFR2-mediated signaling and inflammatory angiogenesis
 SUPPLEMENTAL MATERIALS functions as an endogenous inhibitor of VEGFR2-mediated signaling and inflammatory angiogenesis Haifeng Zhang 1*, Yun He 1*, Shengchuan Dai 1*, Zhe Xu 2*, Yan Luo 2, Ting Wan 2,
SUPPLEMENTAL MATERIALS functions as an endogenous inhibitor of VEGFR2-mediated signaling and inflammatory angiogenesis Haifeng Zhang 1*, Yun He 1*, Shengchuan Dai 1*, Zhe Xu 2*, Yan Luo 2, Ting Wan 2,
Accelerating skin wound healing by M-CSF through generating SSEA-1 and -3 stem cells. in the injured sites
 Accelerating skin wound healing by M-CSF through generating SSEA-1 and -3 stem cells in the injured sites Yunyuan Li, Reza Baradar Jalili, Aziz Ghahary Department of Surgery, University of British Columbia,
Accelerating skin wound healing by M-CSF through generating SSEA-1 and -3 stem cells in the injured sites Yunyuan Li, Reza Baradar Jalili, Aziz Ghahary Department of Surgery, University of British Columbia,
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Nakajima and Tanoue, http://www.jcb.org/cgi/content/full/jcb.201104118/dc1 Figure S1. DLD-1 cells exhibit the characteristic morphology
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Nakajima and Tanoue, http://www.jcb.org/cgi/content/full/jcb.201104118/dc1 Figure S1. DLD-1 cells exhibit the characteristic morphology
OriGene GFC-Arrays for High-throughput Overexpression Screening of Human Gene Phenotypes
 OriGene GFC-Arrays for High-throughput Overexpression Screening of Human Gene Phenotypes High-throughput Gene Function Validation Tool Introduction sirna screening libraries enable scientists to identify
OriGene GFC-Arrays for High-throughput Overexpression Screening of Human Gene Phenotypes High-throughput Gene Function Validation Tool Introduction sirna screening libraries enable scientists to identify
Supplementary Figures and supplementary figure legends
 Supplementary Figures and supplementary figure legends Figure S1. Effect of different percentage of FGF signaling knockdown on TGF signaling and EndMT marker gene expression. HUVECs were subjected to different
Supplementary Figures and supplementary figure legends Figure S1. Effect of different percentage of FGF signaling knockdown on TGF signaling and EndMT marker gene expression. HUVECs were subjected to different
Supplement Figure S1:
 Supplement Figure S1: A, Sequence of Xcadherin-11 Morpholino 1 (Xcad-11MO) and 2 (Xcad-11 MO2) and control morpholino in comparison to the Xcadherin-11 sequence. The Xcad-11MO binding sequence spans the
Supplement Figure S1: A, Sequence of Xcadherin-11 Morpholino 1 (Xcad-11MO) and 2 (Xcad-11 MO2) and control morpholino in comparison to the Xcadherin-11 sequence. The Xcad-11MO binding sequence spans the
gacgacgaggagaccaccgctttg aggcacattgaaggtctcaaacatg
 Supplementary information Supplementary table 1: primers for cloning and sequencing cloning for E- Ras ggg aat tcc ctt gag ctg ctg ggg aat ggc ttt gcc ggt cta gag tat aaa gga agc ttt gaa tcc Tpbp Oct3/4
Supplementary information Supplementary table 1: primers for cloning and sequencing cloning for E- Ras ggg aat tcc ctt gag ctg ctg ggg aat ggc ttt gcc ggt cta gag tat aaa gga agc ttt gaa tcc Tpbp Oct3/4
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Legends for Supplementary Tables. Supplementary Table 1. An excel file containing primary screen data. Worksheet 1, Normalized quantification data from a duplicated screen: valid
SUPPLEMENTARY INFORMATION Legends for Supplementary Tables. Supplementary Table 1. An excel file containing primary screen data. Worksheet 1, Normalized quantification data from a duplicated screen: valid
SUPPLEMETARY INFORMATION. Cdk7 mediates RPB1-driven mrna synthesis in Toxoplasma gondii. Abhijit S. Deshmukh 1 *, Pallabi Mitra 2 and Mulaka Maruthi 3
 SUPPLEMETARY INFORMATION Cdk7 mediates RPB1-driven mrna synthesis in Toxoplasma gondii Abhijit S. Deshmukh 1 *, Pallabi Mitra 2 and Mulaka Maruthi 3 1 National Institute of Animal Biotechnology, Hyderabad,
SUPPLEMETARY INFORMATION Cdk7 mediates RPB1-driven mrna synthesis in Toxoplasma gondii Abhijit S. Deshmukh 1 *, Pallabi Mitra 2 and Mulaka Maruthi 3 1 National Institute of Animal Biotechnology, Hyderabad,
SUPPLEMENTARY INFORMATION. Small molecule activation of the TRAIL receptor DR5 in human cancer cells
 SUPPLEMENTARY INFORMATION Small molecule activation of the TRAIL receptor DR5 in human cancer cells Gelin Wang 1*, Xiaoming Wang 2, Hong Yu 1, Shuguang Wei 1, Noelle Williams 1, Daniel L. Holmes 1, Randal
SUPPLEMENTARY INFORMATION Small molecule activation of the TRAIL receptor DR5 in human cancer cells Gelin Wang 1*, Xiaoming Wang 2, Hong Yu 1, Shuguang Wei 1, Noelle Williams 1, Daniel L. Holmes 1, Randal
Inhibition of Twist1 mediated invasion by Chk2 promotes premature senescence in p53 defective cancer cells
 Inhibition of Twist1 mediated invasion by Chk2 promotes premature senescence in p53 defective cancer cells Debasis Nayak, a,1 Anmol Kumar, b Souneek Chakraborty, a,1 Reyaz ur Rasool, a,1 Hina Amin, a Archana
Inhibition of Twist1 mediated invasion by Chk2 promotes premature senescence in p53 defective cancer cells Debasis Nayak, a,1 Anmol Kumar, b Souneek Chakraborty, a,1 Reyaz ur Rasool, a,1 Hina Amin, a Archana
Cytotoxicity of Botulinum Neurotoxins Reveals a Direct Role of
 Supplementary Information Cytotoxicity of Botulinum Neurotoxins Reveals a Direct Role of Syntaxin 1 and SNAP-25 in Neuron Survival Lisheng Peng, Huisheng Liu, Hongyu Ruan, William H. Tepp, William H. Stoothoff,
Supplementary Information Cytotoxicity of Botulinum Neurotoxins Reveals a Direct Role of Syntaxin 1 and SNAP-25 in Neuron Survival Lisheng Peng, Huisheng Liu, Hongyu Ruan, William H. Tepp, William H. Stoothoff,
The role of erna in chromatin looping
 The role of erna in chromatin looping Introduction Enhancers are cis-acting DNA regulatory elements that activate transcription of target genes 1,2. It is estimated that 400 000 to >1 million alleged enhancers
The role of erna in chromatin looping Introduction Enhancers are cis-acting DNA regulatory elements that activate transcription of target genes 1,2. It is estimated that 400 000 to >1 million alleged enhancers
Supplemental Data Supplementary Figure Legends and Scheme Figure S1.
 Supplemental Data Supplementary Figure Legends and Scheme Figure S1. UTK1 inhibits the second EGF-induced wave of lamellipodia formation in TT cells. A and B, EGF-induced lamellipodia formation in TT cells,
Supplemental Data Supplementary Figure Legends and Scheme Figure S1. UTK1 inhibits the second EGF-induced wave of lamellipodia formation in TT cells. A and B, EGF-induced lamellipodia formation in TT cells,
SUPPLEMENTARY INFORMATION
 doi:.38/nature899 Supplementary Figure Suzuki et al. a c p7 -/- / WT ratio (+)/(-) p7 -/- / WT ratio Log X 3. Fold change by treatment ( (+)/(-)) Log X.5 3-3. -. b Fold change by treatment ( (+)/(-)) 8
doi:.38/nature899 Supplementary Figure Suzuki et al. a c p7 -/- / WT ratio (+)/(-) p7 -/- / WT ratio Log X 3. Fold change by treatment ( (+)/(-)) Log X.5 3-3. -. b Fold change by treatment ( (+)/(-)) 8
Supplementary Fig. 1. Schematic structure of TRAIP and RAP80. The prey line below TRAIP indicates bait and the two lines above RAP80 highlight the
 Supplementary Fig. 1. Schematic structure of TRAIP and RAP80. The prey line below TRAIP indicates bait and the two lines above RAP80 highlight the prey clones identified in the yeast two hybrid screen.
Supplementary Fig. 1. Schematic structure of TRAIP and RAP80. The prey line below TRAIP indicates bait and the two lines above RAP80 highlight the prey clones identified in the yeast two hybrid screen.
Supplementary Figure 1. ERK signaling is not activated at early hypertension. a, Western blot analysis for the level of phospho-erk (perk) and total
 Supplementary Figure 1. ERK signaling is not activated at early hypertension. a, Western blot analysis for the level of phospho-erk (perk) and total ERK in the aortic tissue from the saline- or AngII-infused
Supplementary Figure 1. ERK signaling is not activated at early hypertension. a, Western blot analysis for the level of phospho-erk (perk) and total ERK in the aortic tissue from the saline- or AngII-infused
Supplementary information Activation of AMP-activated protein kinase
 Supplementary information Activation of AMP-activated protein kinase 2 by nicotine instigates formation of abdominal aortic aneurysms in mice in vivo Shuangxi Wang 1,2,5, Cheng Zhang 1,2,5, Miao Zhang
Supplementary information Activation of AMP-activated protein kinase 2 by nicotine instigates formation of abdominal aortic aneurysms in mice in vivo Shuangxi Wang 1,2,5, Cheng Zhang 1,2,5, Miao Zhang
SureSilencing sirna Array Technology Overview
 SureSilencing sirna Array Technology Overview Pathway-Focused sirna-based RNA Interference Topics to be Covered Who is SuperArray? Brief Introduction to RNA Interference Challenges Facing RNA Interference
SureSilencing sirna Array Technology Overview Pathway-Focused sirna-based RNA Interference Topics to be Covered Who is SuperArray? Brief Introduction to RNA Interference Challenges Facing RNA Interference
HPV E6 oncoprotein targets histone methyltransferases for modulating specific. Chih-Hung Hsu, Kai-Lin Peng, Hua-Ci Jhang, Chia-Hui Lin, Shwu-Yuan Wu,
 1 HPV E oncoprotein targets histone methyltransferases for modulating specific gene transcription 3 5 Chih-Hung Hsu, Kai-Lin Peng, Hua-Ci Jhang, Chia-Hui Lin, Shwu-Yuan Wu, Cheng-Ming Chiang, Sheng-Chung
1 HPV E oncoprotein targets histone methyltransferases for modulating specific gene transcription 3 5 Chih-Hung Hsu, Kai-Lin Peng, Hua-Ci Jhang, Chia-Hui Lin, Shwu-Yuan Wu, Cheng-Ming Chiang, Sheng-Chung
Life Sciences WNT PATHWAY. Integrated solutions for screening Wnt regulators. Image: TR Arnett, UCL
 Life Sciences WNT PATHWAY Integrated solutions for screening regulators Image: TR Arnett, UCL 3Find Your Total Solution SOLUTIONS FOR WNT PATHWAY SCREENING High-throughput Assays for Canonical Pathway
Life Sciences WNT PATHWAY Integrated solutions for screening regulators Image: TR Arnett, UCL 3Find Your Total Solution SOLUTIONS FOR WNT PATHWAY SCREENING High-throughput Assays for Canonical Pathway
Four different active promoter genes were chosen, ATXN7L2, PSRC1, CELSR2 and
 SUPPLEMENTARY MATERIALS AND METHODS Chromatin Immunoprecipitation for qpcr analysis Four different active promoter genes were chosen, ATXN7L2, PSRC1, CELSR2 and IL24, all located on chromosome 1. Primer
SUPPLEMENTARY MATERIALS AND METHODS Chromatin Immunoprecipitation for qpcr analysis Four different active promoter genes were chosen, ATXN7L2, PSRC1, CELSR2 and IL24, all located on chromosome 1. Primer
Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section B and ONE question from Section C.
 UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2017-18 CELL BIOLOGY BIO-5005B Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section
UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2017-18 CELL BIOLOGY BIO-5005B Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section
cells (MLEC) that produce luciferase under the control of the PAI-1 promoter in response to
 Supplemental Materials and Methods TGF bioassay. To quantify the levels of active and total TGF, we used mink lung epithelial cells (MLEC) that produce luciferase under the control of the PAI-1 promoter
Supplemental Materials and Methods TGF bioassay. To quantify the levels of active and total TGF, we used mink lung epithelial cells (MLEC) that produce luciferase under the control of the PAI-1 promoter
Supplementary data. sienigma. F-Enigma F-EnigmaSM. a-p53
 Supplementary data Supplemental Figure 1 A sienigma #2 sienigma sicontrol a-enigma - + ++ - - - - - - + ++ - - - - - - ++ B sienigma F-Enigma F-EnigmaSM a-flag HLK3 cells - - - + ++ + ++ - + - + + - -
Supplementary data Supplemental Figure 1 A sienigma #2 sienigma sicontrol a-enigma - + ++ - - - - - - + ++ - - - - - - ++ B sienigma F-Enigma F-EnigmaSM a-flag HLK3 cells - - - + ++ + ++ - + - + + - -
transcription and the promoter occupancy of Smad proteins. (A) HepG2 cells were co-transfected with the wwp-luc reporter, and FLAG-tagged FHL1,
 Supplementary Data Supplementary Figure Legends Supplementary Figure 1 FHL-mediated TGFβ-responsive reporter transcription and the promoter occupancy of Smad proteins. (A) HepG2 cells were co-transfected
Supplementary Data Supplementary Figure Legends Supplementary Figure 1 FHL-mediated TGFβ-responsive reporter transcription and the promoter occupancy of Smad proteins. (A) HepG2 cells were co-transfected
Supplementary Figure 1. The Hsp70 acetylation level is related to the co-chaperone binding of Hsp70 under various stress conditions.
 Supplementary Figure 1. The Hsp70 acetylation level is related to the co-chaperone binding of Hsp70 under various stress conditions. 1 (a) Etoposide treatment gradually changes acetylation level and co-chaperone
Supplementary Figure 1. The Hsp70 acetylation level is related to the co-chaperone binding of Hsp70 under various stress conditions. 1 (a) Etoposide treatment gradually changes acetylation level and co-chaperone
Regulating Bone Growth and Development with Bone Morphogenetic Proteins
 Regulating Bone Growth and Development with Bone Morphogenetic Proteins PHOEBE S. LEBOY Department of Biochemistry, School of Dental Medicine, University of Pennsylvania, Philadelphia, Pennsylvania 19104-6030,
Regulating Bone Growth and Development with Bone Morphogenetic Proteins PHOEBE S. LEBOY Department of Biochemistry, School of Dental Medicine, University of Pennsylvania, Philadelphia, Pennsylvania 19104-6030,
A RRM1 H2AX DAPI. RRM1 H2AX DAPI Merge. Cont. sirna RRM1
 A H2AX DAPI H2AX DAPI Merge Cont sirna Figure S1: Accumulation of RRM1 at DNA damage sites (A) HeLa cells were subjected to in situ detergent extraction without IR irradiation, and immunostained with the
A H2AX DAPI H2AX DAPI Merge Cont sirna Figure S1: Accumulation of RRM1 at DNA damage sites (A) HeLa cells were subjected to in situ detergent extraction without IR irradiation, and immunostained with the
Gα i Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα i Activation Assay Kit Catalog Number 80301 20 assays NewEast Biosciences, Inc 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα i Activation Assay Kit Catalog Number 80301 20 assays NewEast Biosciences, Inc 1 Table of Content Product
Data Sheet. TCF/LEF Reporter Kit Wnt / -catenin signaling pathway Catalog #: 60500
 Data Sheet TCF/LEF Reporter Kit Wnt / -catenin signaling pathway Catalog #: 60500 Background The Wnt / -catenin signaling pathway controls a large and diverse set of cell fate decisions in embryonic development,
Data Sheet TCF/LEF Reporter Kit Wnt / -catenin signaling pathway Catalog #: 60500 Background The Wnt / -catenin signaling pathway controls a large and diverse set of cell fate decisions in embryonic development,
Gα 13 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα 13 Activation Assay Kit Catalog Number: 80401 20 assays NewEast Biosciences 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα 13 Activation Assay Kit Catalog Number: 80401 20 assays NewEast Biosciences 1 Table of Content Product
Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or
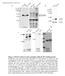 Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or absence of the acetyltransferase CBP and acetylated TDP-43
Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or absence of the acetyltransferase CBP and acetylated TDP-43
Supplementary Figure 1, related to Figure 1. GAS5 is highly expressed in the cytoplasm of hescs, and positively correlates with pluripotency.
 Supplementary Figure 1, related to Figure 1. GAS5 is highly expressed in the cytoplasm of hescs, and positively correlates with pluripotency. (a) Transfection of different concentration of GAS5-overexpressing
Supplementary Figure 1, related to Figure 1. GAS5 is highly expressed in the cytoplasm of hescs, and positively correlates with pluripotency. (a) Transfection of different concentration of GAS5-overexpressing
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/146/ra80/dc1 Supplementary Materials for DNMT1 Stability Is Regulated by Proteins Coordinating Deubiquitination and Acetylation-Driven Ubiquitination Zhanwen
www.sciencesignaling.org/cgi/content/full/3/146/ra80/dc1 Supplementary Materials for DNMT1 Stability Is Regulated by Proteins Coordinating Deubiquitination and Acetylation-Driven Ubiquitination Zhanwen
Transcription Factor Runx3 Is Induced by Influenza A Virus and Double-Strand RNA and
 Transcription Factor Is Induced by Influenza A Virus and Double-Strand RNA and Mediates Airway Epithelial Cell Apoptosis Huachen Gan 1, Qin Hao 1, Steven Idell 1,2 & Hua Tang 1 1 Department of Cellular
Transcription Factor Is Induced by Influenza A Virus and Double-Strand RNA and Mediates Airway Epithelial Cell Apoptosis Huachen Gan 1, Qin Hao 1, Steven Idell 1,2 & Hua Tang 1 1 Department of Cellular
Supplemental Online Material. The mouse embryonic fibroblast cell line #10 derived from β-arrestin1 -/- -β-arrestin2 -/-
 #1074683s 1 Supplemental Online Material Materials and Methods Cell lines and tissue culture The mouse embryonic fibroblast cell line #10 derived from β-arrestin1 -/- -β-arrestin2 -/- knock-out animals
#1074683s 1 Supplemental Online Material Materials and Methods Cell lines and tissue culture The mouse embryonic fibroblast cell line #10 derived from β-arrestin1 -/- -β-arrestin2 -/- knock-out animals
Culture media, trypsin, penicillin and streptomycin were from Invitrogen (Breda, the Netherlands).
 Methods Materials Culture media, trypsin, penicillin and streptomycin were from Invitrogen (Breda, the Netherlands). Bovine fibroblast growth factor (BFGF), thrombin, forskolin, IBMX, H-89, BAPTA-AM and
Methods Materials Culture media, trypsin, penicillin and streptomycin were from Invitrogen (Breda, the Netherlands). Bovine fibroblast growth factor (BFGF), thrombin, forskolin, IBMX, H-89, BAPTA-AM and
The Transfection Collection TCF/LEF Transient Pack Wnt / -catenin Signaling Pathway Catalog #: 79273
 Data Sheet The Transfection Collection TCF/LEF Transient Pack Wnt / -catenin Signaling Pathway Catalog #: 79273 Background The Wnt / -catenin signaling pathway controls a large and diverse set of cell
Data Sheet The Transfection Collection TCF/LEF Transient Pack Wnt / -catenin Signaling Pathway Catalog #: 79273 Background The Wnt / -catenin signaling pathway controls a large and diverse set of cell
Supplementary Figure 1. Soft fibrin gels promote growth and organized mesodermal differentiation. Representative images of single OGTR1 ESCs cultured
 Supplementary Figure 1. Soft fibrin gels promote growth and organized mesodermal differentiation. Representative images of single OGTR1 ESCs cultured in 90-Pa 3D fibrin gels for 5 days in the presence
Supplementary Figure 1. Soft fibrin gels promote growth and organized mesodermal differentiation. Representative images of single OGTR1 ESCs cultured in 90-Pa 3D fibrin gels for 5 days in the presence
Supplementary Figure 1. Nur77 and leptin-controlled obesity. (A) (B) (C)
 Supplementary Figure 1. Nur77 and leptin-controlled obesity. (A) Effect of leptin on body weight and food intake between WT and KO mice at the age of 12 weeks (n=7). Mice were i.c.v. injected with saline
Supplementary Figure 1. Nur77 and leptin-controlled obesity. (A) Effect of leptin on body weight and food intake between WT and KO mice at the age of 12 weeks (n=7). Mice were i.c.v. injected with saline
Supplementary Data. Supplementary Table S1.
 Supplementary Data Supplementary Table S1. Primer Sequences for Real-Time Reverse Transcription-Polymerase Chain Reaction Gene Forward 5?3 Reverse 3?5 Housekeeping gene 36B4 TCCAGGCTTTGGGCATCA CTTTATCAGCTGCACATCACTCAGA
Supplementary Data Supplementary Table S1. Primer Sequences for Real-Time Reverse Transcription-Polymerase Chain Reaction Gene Forward 5?3 Reverse 3?5 Housekeeping gene 36B4 TCCAGGCTTTGGGCATCA CTTTATCAGCTGCACATCACTCAGA
Reproductive Biology and Endocrinology BioMed Central Review Smad signalling in the ovary Noora Kaivo-oja, Luke A Jeffery, Olli Ritvos and David G Mot
 Reproductive Biology and Endocrinology BioMed Central Review Smad signalling in the ovary Noora Kaivo-oja, Luke A Jeffery, Olli Ritvos and David G Mottershead* Open Access Address: Programme for Developmental
Reproductive Biology and Endocrinology BioMed Central Review Smad signalling in the ovary Noora Kaivo-oja, Luke A Jeffery, Olli Ritvos and David G Mottershead* Open Access Address: Programme for Developmental
Neural Stem Cell Characterization Kit
 Neural Stem Cell Characterization Kit Catalog No. SCR019 FOR RESEARCH USE ONLY Not for use in diagnostic procedures USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23 8026 2233
Neural Stem Cell Characterization Kit Catalog No. SCR019 FOR RESEARCH USE ONLY Not for use in diagnostic procedures USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23 8026 2233
Figure S1. Figure S2. Figure S3 HB Anti-FSP27 (COOH-terminal peptide) Ab. Anti-GST-FSP27(45-127) Ab.
 / 36B4 mrna ratio Figure S1 * 2. 1.6 1.2.8 *.4 control TNFα BRL49653 Figure S2 Su bw AT p iw Anti- (COOH-terminal peptide) Ab Blot : Anti-GST-(45-127) Ab β-actin Figure S3 HB2 HW AT BA T Figure S4 A TAG
/ 36B4 mrna ratio Figure S1 * 2. 1.6 1.2.8 *.4 control TNFα BRL49653 Figure S2 Su bw AT p iw Anti- (COOH-terminal peptide) Ab Blot : Anti-GST-(45-127) Ab β-actin Figure S3 HB2 HW AT BA T Figure S4 A TAG
supplementary information
 DOI: 10.1038/ncb2172 Figure S1 p53 regulates cellular NADPH and lipid levels via inhibition of G6PD. (a) U2OS cells stably expressing p53 shrna or a control shrna were transfected with control sirna or
DOI: 10.1038/ncb2172 Figure S1 p53 regulates cellular NADPH and lipid levels via inhibition of G6PD. (a) U2OS cells stably expressing p53 shrna or a control shrna were transfected with control sirna or
Chinese Academy of Sciences, Datun Road, Chaoyang District, Beijing, 10101, China
 SUPPLEMENTARY INFORMATION Title: Bi-directional processing of pri-mirnas with branched terminal loops by Arabidopsis Dicer-like1 Hongliang Zhu 1,2,3, Yuyi Zhou 1,2,4, Claudia Castillo-González 1,2, Amber
SUPPLEMENTARY INFORMATION Title: Bi-directional processing of pri-mirnas with branched terminal loops by Arabidopsis Dicer-like1 Hongliang Zhu 1,2,3, Yuyi Zhou 1,2,4, Claudia Castillo-González 1,2, Amber
Supplementary Figure 1.
 Supplementary Figure 1. Quantification of western blot analysis of fibroblasts (related to Figure 1) (A-F) Quantification of western blot analysis for control and IR-Mut fibroblasts. Data are expressed
Supplementary Figure 1. Quantification of western blot analysis of fibroblasts (related to Figure 1) (A-F) Quantification of western blot analysis for control and IR-Mut fibroblasts. Data are expressed
Supplementary Material
 Supplementary Material Supplementary Methods Cell synchronization. For synchronized cell growth, thymidine was added to 30% confluent U2OS cells to a final concentration of 2.5mM. Cells were incubated
Supplementary Material Supplementary Methods Cell synchronization. For synchronized cell growth, thymidine was added to 30% confluent U2OS cells to a final concentration of 2.5mM. Cells were incubated
Thermo Scientific GTPase Research Tools
 Thermo Scientific Research Tools Active Pull-Down Assays We offer two different tools to study biology, one for active monitoring and one for global profiling. The Thermo Scientific Pierce Active Pull-Down
Thermo Scientific Research Tools Active Pull-Down Assays We offer two different tools to study biology, one for active monitoring and one for global profiling. The Thermo Scientific Pierce Active Pull-Down
Supplementary Figure 1. Immunoprecipitation of synthetic SUMOm-remnant peptides using UMO monoclonal antibody. (a) LC-MS analyses of tryptic
 Supplementary Figure 1. Immunoprecipitation of synthetic SUMOm-remnant peptides using UMO 1-7-7 monoclonal antibody. (a) LC-MS analyses of tryptic digest from HEK293 cells spiked with 6 SUMOmremnant peptides
Supplementary Figure 1. Immunoprecipitation of synthetic SUMOm-remnant peptides using UMO 1-7-7 monoclonal antibody. (a) LC-MS analyses of tryptic digest from HEK293 cells spiked with 6 SUMOmremnant peptides
Supplementary Figure 1 Collision-induced dissociation (CID) mass spectra of peptides from PPK1, PPK2, PPK3 and PPK4 respectively.
 Supplementary Figure 1 lision-induced dissociation (CID) mass spectra of peptides from PPK1, PPK, PPK3 and PPK respectively. % of nuclei with signal / field a 5 c ppif3:gus pppk1:gus 0 35 30 5 0 15 10
Supplementary Figure 1 lision-induced dissociation (CID) mass spectra of peptides from PPK1, PPK, PPK3 and PPK respectively. % of nuclei with signal / field a 5 c ppif3:gus pppk1:gus 0 35 30 5 0 15 10
Stargazin regulates AMPA receptor trafficking through adaptor protein. complexes during long term depression
 Supplementary Information Stargazin regulates AMPA receptor trafficking through adaptor protein complexes during long term depression Shinji Matsuda, Wataru Kakegawa, Timotheus Budisantoso, Toshihiro Nomura,
Supplementary Information Stargazin regulates AMPA receptor trafficking through adaptor protein complexes during long term depression Shinji Matsuda, Wataru Kakegawa, Timotheus Budisantoso, Toshihiro Nomura,
SUPPLEMENTARY INFORMATION FIGURE LEGENDS
 SUPPLEMENTARY INFORMATION FIGURE LEGENDS Fig. S1. Radiation-induced phosphorylation of Rad50 at a specific site. A. Rad50 is an in vitro substrate for ATM. A series of Rad50-GSTs covering the entire molecule
SUPPLEMENTARY INFORMATION FIGURE LEGENDS Fig. S1. Radiation-induced phosphorylation of Rad50 at a specific site. A. Rad50 is an in vitro substrate for ATM. A series of Rad50-GSTs covering the entire molecule
System. Dynamic Monitoring of Receptor Tyrosine Kinase Activation in Living Cells. Application Note No. 4 / March
 System Application Note No. 4 / March 2008 Dynamic Monitoring of Receptor Tyrosine Kinase Activation in Living Cells www.roche-applied-science.com Dynamic Monitoring of Receptor Tyrosine Kinase Activation
System Application Note No. 4 / March 2008 Dynamic Monitoring of Receptor Tyrosine Kinase Activation in Living Cells www.roche-applied-science.com Dynamic Monitoring of Receptor Tyrosine Kinase Activation
Charles Shuler, Ph.D. University of Southern California Los Angeles, California Approved for Public Release; Distribution Unlimited
 AD Award Number: DAMD17-01-1-0100 TITLE: Smad-Mediated Signaling During Prostate Growth and Development PRINCIPAL INVESTIGATOR: Charles Shuler, Ph.D. CONTRACTING ORGANIZATION: University of Southern California
AD Award Number: DAMD17-01-1-0100 TITLE: Smad-Mediated Signaling During Prostate Growth and Development PRINCIPAL INVESTIGATOR: Charles Shuler, Ph.D. CONTRACTING ORGANIZATION: University of Southern California
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/310/5753/1487/dc1 Supporting Online Material for Stem Cell Self-Renewal Controlled by Chromatin Remodeling Factors Rongwen Xi and Ting Xie* *To whom correspondence should
www.sciencemag.org/cgi/content/full/310/5753/1487/dc1 Supporting Online Material for Stem Cell Self-Renewal Controlled by Chromatin Remodeling Factors Rongwen Xi and Ting Xie* *To whom correspondence should
Cdc42 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
ASPP1 Fw GGTTGGGAATCCACGTGTTG ASPP1 Rv GCCATATCTTGGAGCTCTGAGAG
 Supplemental Materials and Methods Plasmids: the following plasmids were used in the supplementary data: pwzl-myc- Lats2 (Aylon et al, 2006), pretrosuper-vector and pretrosuper-shp53 (generous gift of
Supplemental Materials and Methods Plasmids: the following plasmids were used in the supplementary data: pwzl-myc- Lats2 (Aylon et al, 2006), pretrosuper-vector and pretrosuper-shp53 (generous gift of
Supplementary Information (Ha, et. al) Supplementary Figures Supplementary Fig. S1
 Supplementary Information (Ha, et. al) Supplementary Figures Supplementary Fig. S1 a His-ORMDL3 ~ 17 His-ORMDL3 GST-ORMDL3 - + - + IPTG GST-ORMDL3 ~ b Integrated Density (ORMDL3/ -actin) 0.4 0.3 0.2 0.1
Supplementary Information (Ha, et. al) Supplementary Figures Supplementary Fig. S1 a His-ORMDL3 ~ 17 His-ORMDL3 GST-ORMDL3 - + - + IPTG GST-ORMDL3 ~ b Integrated Density (ORMDL3/ -actin) 0.4 0.3 0.2 0.1
RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the
 Supplementary Methods RT-PCR and real-time PCR analysis RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the manufacturer s protocol and quantified by measuring the
Supplementary Methods RT-PCR and real-time PCR analysis RNA was isolated using NucleoSpin RNA II (Macherey-Nagel, Bethlehem, PA) according to the manufacturer s protocol and quantified by measuring the
Supporting Information
 Supporting Information Su et al. 10.1073/pnas.1211604110 SI Materials and Methods Cell Culture and Plasmids. Tera-1 and Tera-2 cells (ATCC: HTB- 105/106) were maintained in McCoy s 5A medium with 15% FBS
Supporting Information Su et al. 10.1073/pnas.1211604110 SI Materials and Methods Cell Culture and Plasmids. Tera-1 and Tera-2 cells (ATCC: HTB- 105/106) were maintained in McCoy s 5A medium with 15% FBS
Flow cytometry Stained cells were analyzed and sorted by SORP FACS Aria (BD Biosciences).
 Mice C57BL/6-Ly5.1 or -Ly5.2 congenic mice were used for LSK transduction and competitive repopulation assays. Animal care was in accordance with the guidelines of Keio University for animal and recombinant
Mice C57BL/6-Ly5.1 or -Ly5.2 congenic mice were used for LSK transduction and competitive repopulation assays. Animal care was in accordance with the guidelines of Keio University for animal and recombinant
TITLE: Epithelin/Granulin Precursor Expression in Human Breast Carcinoma
 AD GRANT NUMBER DAMD17-96-1-6072 TITLE: Epithelin/Granulin Precursor Expression in Human Breast Carcinoma PRINCIPAL INVESTIGATOR: Ginette Serrero, Ph.D. CONTRACTING ORGANIZATION: University of Maryland
AD GRANT NUMBER DAMD17-96-1-6072 TITLE: Epithelin/Granulin Precursor Expression in Human Breast Carcinoma PRINCIPAL INVESTIGATOR: Ginette Serrero, Ph.D. CONTRACTING ORGANIZATION: University of Maryland
SUPPLEMENTARY INFORMATION
 The Supplementary Information (SI) Methods Cell culture and transfections H1299, U2OS, 293, HeLa cells were maintained in DMEM medium supplemented with 10% fetal bovine serum. H1299 and 293 cells were
The Supplementary Information (SI) Methods Cell culture and transfections H1299, U2OS, 293, HeLa cells were maintained in DMEM medium supplemented with 10% fetal bovine serum. H1299 and 293 cells were
Flag-Rac Vector V12 V12 N17 C40. Vector C40 pakt (T308) Akt1. Myc-DN-PAK1 (N-SP)
 a b FlagRac FlagRac V2 V2 N7 C4 V2 V2 N7 C4 p (T38) p (S99, S24) p Flag (Rac) NIH 3T3 COS c +Serum p (T38) MycDN (NSP) Mycp27 3 6 2 3 6 2 3 6 2 min p Myc ( or p27) Figure S (a) Effects of Rac mutants on
a b FlagRac FlagRac V2 V2 N7 C4 V2 V2 N7 C4 p (T38) p (S99, S24) p Flag (Rac) NIH 3T3 COS c +Serum p (T38) MycDN (NSP) Mycp27 3 6 2 3 6 2 3 6 2 min p Myc ( or p27) Figure S (a) Effects of Rac mutants on
Marilyn G. Rimando, Hao-Hsiang Wu, Yu-An Liu, Chien-Wei Lee, Shu-Wen Kuo, Yin-
 Supplementary Information Glucocorticoid receptor and Histone Deacetylase 6 mediate the differential effect of dexamethasone during osteogenesis of Mesenchymal stromal cells (MSCs) Marilyn G. Rimando,
Supplementary Information Glucocorticoid receptor and Histone Deacetylase 6 mediate the differential effect of dexamethasone during osteogenesis of Mesenchymal stromal cells (MSCs) Marilyn G. Rimando,
Regulation of hepcidin expression by inflammation-induced activin B
 Regulation of hepcidin expression by inflammation-induced activin B Yohei Kanamori, Makoto Sugiyama, Osamu Hashimoto, Masaru Murakami, Tohru Matsui and Masayuki Funaba Supplemental methods Liver cell separation
Regulation of hepcidin expression by inflammation-induced activin B Yohei Kanamori, Makoto Sugiyama, Osamu Hashimoto, Masaru Murakami, Tohru Matsui and Masayuki Funaba Supplemental methods Liver cell separation
Transcriptional regulation of IFN-l genes in Hepatitis C virus-infected hepatocytes via IRF-3 IRF-7 NF- B complex
 POSTER PRESENTATION Transcriptional regulation of IFN-l genes in Hepatitis C virus-infected hepatocytes via IRF-3 IRF-7 NF- B complex Hai-Chon Lee *, Je-In Youn, Kyungwha Lee, Hwanyul Yong, Seung-Yong
POSTER PRESENTATION Transcriptional regulation of IFN-l genes in Hepatitis C virus-infected hepatocytes via IRF-3 IRF-7 NF- B complex Hai-Chon Lee *, Je-In Youn, Kyungwha Lee, Hwanyul Yong, Seung-Yong
Index 273. polyclonal sera characterization, 179 steric hindrance by adjacent phosphate, 184
 Index 269 Index A Adenovirus E1B, see E1B-55 kda oncoprotein Adenovirus-expressed p53, bacterial transformation and recombination, plasmid purification, 7 plasmid verification and preparation for transfection,
Index 269 Index A Adenovirus E1B, see E1B-55 kda oncoprotein Adenovirus-expressed p53, bacterial transformation and recombination, plasmid purification, 7 plasmid verification and preparation for transfection,
Notch1 (forward: 5 -TGCCTGTGCACACCATTCTGC-3, reverse: Notch2 (forward: 5 -ATGCACCATGACATCGTTCG-3, reverse:
 Supplementary Information Supplementary Methods. RT-PCR. For mouse ES cells, the primers used were: Notch1 (forward: 5 -TGCCTGTGCACACCATTCTGC-3, reverse: 5 -CAATCAGAGATGTTGGAATGC-3 ) 1, Notch2 (forward:
Supplementary Information Supplementary Methods. RT-PCR. For mouse ES cells, the primers used were: Notch1 (forward: 5 -TGCCTGTGCACACCATTCTGC-3, reverse: 5 -CAATCAGAGATGTTGGAATGC-3 ) 1, Notch2 (forward:
Methanol fixation allows better visualization of Kal7. To compare methods for
 Supplementary Data Methanol fixation allows better visualization of Kal7. To compare methods for visualizing Kal7 in dendrites, mature cultures of dissociated hippocampal neurons (DIV21) were fixed with
Supplementary Data Methanol fixation allows better visualization of Kal7. To compare methods for visualizing Kal7 in dendrites, mature cultures of dissociated hippocampal neurons (DIV21) were fixed with
Supplementary Information
 Supplementary Information Sam68 modulates the promoter specificity of NF-κB and mediates expression of CD25 in activated T cells Kai Fu 1, 6, Xin Sun 1, 6, Wenxin Zheng 1, 6, Eric M. Wier 1, Andrea Hodgson
Supplementary Information Sam68 modulates the promoter specificity of NF-κB and mediates expression of CD25 in activated T cells Kai Fu 1, 6, Xin Sun 1, 6, Wenxin Zheng 1, 6, Eric M. Wier 1, Andrea Hodgson
Supplementary Information
 Supplementary Information Figure S1. Transiently overexpressed ECFP-TIAF1/EYFP-TIAF1 causes apoptosis of Mv1Lu cells. Mv1Lu cells were transfected by electroporation with ECFP, EYFP, ECFP-TIAF1, EYFP-
Supplementary Information Figure S1. Transiently overexpressed ECFP-TIAF1/EYFP-TIAF1 causes apoptosis of Mv1Lu cells. Mv1Lu cells were transfected by electroporation with ECFP, EYFP, ECFP-TIAF1, EYFP-
