A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition.
|
|
|
- Melanie Booker
- 6 years ago
- Views:
Transcription
1 Minerals
2 Objectives List the 5 traits of minerals Identify minerals Describe characteristics of minerals Know how minerals form Describe ways that we use minerals
3 A mineral is a naturally occurring, inorganic solid that has a crystal structure and a definite chemical composition.
4 Naturally Occurring Formed by processes that occur in the natural world
5 Inorganic Not formed from materials that were once part of a living thing.
6 Solid Solid, with a definite volume and shape.
7 Crystal structure Particles of a mineral line up in a pattern that repeats over and over again. Flat sides are called faces
8 Definite Chemical Composition Always contains certain elements in definite proportions. Example quartz has one atom of silicon for every two atoms of oxygen (SiO 2 )
9 Bell Question 1/10/11 What are the 5 criteria that a mineral has to meet in order to be called a mineral?
10 Identifying Minerals Each mineral has characteristic properties that can be used to identify it.
11 Color Color is easily observed but alone provides too little information to make an identification Observe surface of mineral Mercury Ore
12 Streak The color of its powder Streak color and mineral color are often different Observe color of powder on unglazed tile
13 Luster How light is reflected from a mineral s surface. Observe how mineral reflects light
14 Density Mass in a given space No matter the sample size, density is the same Find mass per unit volume Galena
15 Hardness Mohs hardness scale 10 minerals from softest to hardest Perform a scratch test
16 Crystal Systems Crystals grow atom by atom to form that mineral s crystal structure Geometric shape Observe number and angle of crystal faces
17 Cleavage and Fracture The way a mineral breaks apart Break mineral apart to see if it splits along flat surfaces.
18 Special properties Special or unique physical property Examples include magnetism, fluorescence, optical properties
19 Luster Metallic - Galena Glassy - Topaz Waxy, Greasy or Pearly Talc
20 Luster Submetallic or dull - Graphite Silky - Malachite Earthy - Hematite
21 Key Terms Book F, Chapter 4 write down a definition for the following words 1. Mineral 2. inorganic 3. crystal 4. streak 5. luster 6. Mohs hardness scale 7. cleavage 8. fracture 9. geode 10.crystallization 11.solution 12.gemstone 13.ore 14.smelting 15.alloy Read pages F F122 Page F122 questions 1-2
22 Bell Questions 1/11/11 1. True or False, Color is the best way to identify a mineral. 2. What test can you perform to find the streak of a mineral? 3. What is luster?
23 Mohs Hardness Scale 1. Talc flakes when scratched by a fingernail 2. Gypsum a fingernail can easily scratch it 3. Calcite a fingernail can t scratch it but a penny can
24 Mohs Hardness Scale 4. Fluorite a steel knife can easily scratch it 5. Apatite a steel knife can scratch it 6. Feldspar It can t be scratched by a knife but it can scratch window glass
25 Mohs Hardness Scale 7. Quartz it can scratch steel and hard glass easily 8. Topaz It can scratch quartz easily 9. Corundum It can scratch topaz
26 Mohs Hardness Scale 10.Diamond The hardest known material. It can scratch all other materials
27 Bell Questions 1/12/11 1. The softest mineral on the Mohs hardness scale is. 2. What mineral will scratch all others? 3. Shiny minerals, such as galena, are said to have metallic.
28 Cubic Examples: Magnetite, Halite, Garnet
29 Hexagonal Six sided Examples: Beryl, apatite, Quartz
30 Tetragonal Two sides the same, one different Examples: Zircon, Rutile
31 Orthorhombic Three different Lengths Example: Sulfur
32 Monoclinic (Rectangle pushed over) Example: Azurite, Gypsum
33 Triclinic (rectangle pushed over twice) Example: Feldspar
34 How Minerals Form Minerals from magma and lava Magma below the surface Deep down it cools slowly large crystals Toward surface it cools rapidly small crystals Lava on surface Cools quickly small crystals
35
36 Minerals From Solutions Solution a mixture in which one substance is dissolved in another
37 Minerals From Solutions Evaporation water evaporates and the crystals are left behind Examples: salt, halite, gypsum, calcite
38
39 Minerals From Solutions Hot water solutions: Magma heats water, when water cools the elements leave the solution and crystallize as minerals Example: silver
Properties of Minerals
 What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
There are about 3,000 known minerals, only about 30 are common. The most common are quartz,feldspar,mica, and calcite.
 There are about 3,000 known minerals, only about 30 are common. The most common are quartz,feldspar,mica, and calcite. These minerals make up most of the rocks found in the Earth s crust. Most of the Earth
There are about 3,000 known minerals, only about 30 are common. The most common are quartz,feldspar,mica, and calcite. These minerals make up most of the rocks found in the Earth s crust. Most of the Earth
Student Exploration: Mineral Identification
 Name: Date: Student Exploration: Identification Vocabulary: crystal, density, hardness, homogeneous, luster, mass, mineral, streak, volume Prior Knowledge Questions (Do these BEFORE using the Gizmo.) 1.
Name: Date: Student Exploration: Identification Vocabulary: crystal, density, hardness, homogeneous, luster, mass, mineral, streak, volume Prior Knowledge Questions (Do these BEFORE using the Gizmo.) 1.
page - Labs 09 & 10 - Mineral Identification
 Labs 09 & 10 - Mineral Identification page - All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the
Labs 09 & 10 - Mineral Identification page - All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the
page - 1 Lab 9 - Mineral Identification
 Lab 9 - Mineral Identification page - 1 All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the bulk
Lab 9 - Mineral Identification page - 1 All rocks are composed of one or more minerals. In order to be able to identify rocks you have to know how to recognize those key minerals that make of the bulk
What is a Mineral? A Mineral is: Pure Naturally occurring Crystalline Solid
 Mineral Properties What is a Mineral? A Mineral is: Pure Naturally occurring Crystalline Solid Mineral Properties Color Streak Luster Hardness Density Crystal Shape Cleavage Fracture Fluorescence Reaction
Mineral Properties What is a Mineral? A Mineral is: Pure Naturally occurring Crystalline Solid Mineral Properties Color Streak Luster Hardness Density Crystal Shape Cleavage Fracture Fluorescence Reaction
WELCOME TO THE GLENDALE COMMUNITY COLLEGE MINERAL IDENTIFICATION PROGRAM
 presented by: WELCOME TO THE GLENDALE COMMUNITY COLLEGE MINERAL IDENTIFICATION PROGRAM Susan Celestian - Curator of the Arizona Mining and Mineral Museum Stan Celestian - Photographer copyright 2002 Where
presented by: WELCOME TO THE GLENDALE COMMUNITY COLLEGE MINERAL IDENTIFICATION PROGRAM Susan Celestian - Curator of the Arizona Mining and Mineral Museum Stan Celestian - Photographer copyright 2002 Where
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
The Study of Minerals (Chapter 1)
 GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
The Study of Minerals (Chapter 1)
 GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
GEOLOGY 306 Laboratory Instructor: TERRY J. BOROUGHS NAME: The Study of Minerals (Chapter 1) For this assignment you will require: a streak plate, glass plate, magnet, dilute hydrochloric (HCl) acid, microscope,
1. Hardness 2. Streak 3. Density 4. Crystal structure 5. A mineral must be a naturally occurring, inorganic. 7. c 8. f 9. a 10. d 11. g 12. e 13.
 Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Mineral Identification
 Name: Geosc 001 Spring 2008 Lab 3 Mineral Identification The basic goal of this lab is to introduce you to some basic ideas and strategies for identifying minerals, which are the building blocks of rocks
Name: Geosc 001 Spring 2008 Lab 3 Mineral Identification The basic goal of this lab is to introduce you to some basic ideas and strategies for identifying minerals, which are the building blocks of rocks
GEOLOGY 284: MINERALOGY
 Dr. Helen Lang Dept. of Geology & Geography West Virginia University FALL 2015 GEOLOGY 284: MINERALOGY Mineral Properties in Hand Specimen Luster Metallic Sub-metallic Non-metallic Vitreous Adamantine
Dr. Helen Lang Dept. of Geology & Geography West Virginia University FALL 2015 GEOLOGY 284: MINERALOGY Mineral Properties in Hand Specimen Luster Metallic Sub-metallic Non-metallic Vitreous Adamantine
Physical Properties of Minerals Part II
 GEOLOGY 333 LAB 2 PHYSICAL PROPERTIES OF MINERALS PART II Physical Properties of Minerals covered last week: 1. Hardness 2. Cleavage 3. Crystal Shape 4. Specific Gravity Physical Properties of Minerals
GEOLOGY 333 LAB 2 PHYSICAL PROPERTIES OF MINERALS PART II Physical Properties of Minerals covered last week: 1. Hardness 2. Cleavage 3. Crystal Shape 4. Specific Gravity Physical Properties of Minerals
Physical Properties of Minerals
 1 of 8 9/20/2002 9:30 AM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy Physical Properties of Minerals This document last updated on 20-Sep-2002 Although we have discussed x-ray identification
1 of 8 9/20/2002 9:30 AM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy Physical Properties of Minerals This document last updated on 20-Sep-2002 Although we have discussed x-ray identification
Unit 2 6 th Grade PHYSICAL PROPERTIES OF MATTER
 Unit 2 6 th Grade PHYSICAL PROPERTIES OF MATTER Elements can be classified as: Metals Good conductors of heat. Good conductors of electricity. Good reflectors of light when polished (shiny luster) Malleable
Unit 2 6 th Grade PHYSICAL PROPERTIES OF MATTER Elements can be classified as: Metals Good conductors of heat. Good conductors of electricity. Good reflectors of light when polished (shiny luster) Malleable
Make Five. A game about the chemical formulas of some common minerals
 Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
5. MINERAL PROPERTIES, IDENTIFICATION, & USES
 LAST NAME (ALL IN CAPS): FIRST NAME: 5. MINERAL PROPERTIES, IDENTIFICATION, & USES Instructions: Refer to Laboratory 3 in your Lab Manual on pages 73-110 to answer the questions in this work sheet. Your
LAST NAME (ALL IN CAPS): FIRST NAME: 5. MINERAL PROPERTIES, IDENTIFICATION, & USES Instructions: Refer to Laboratory 3 in your Lab Manual on pages 73-110 to answer the questions in this work sheet. Your
Ch. 2. Categories. Hand Sample ID. Crystal Shape. Crystal Habit. Interactions with light. Geo 271, Spring Elizabeth Goeke 1
 Categories Hand Sample ID Spring 2011 Klein & Dutrow appearance xtal form habit interaction with light diaphaneity luster color streak luminescence mechanical properties hardness tenacity cleavage fracture
Categories Hand Sample ID Spring 2011 Klein & Dutrow appearance xtal form habit interaction with light diaphaneity luster color streak luminescence mechanical properties hardness tenacity cleavage fracture
GLY 4200C LAB 1. 1 Talc 6 Orthoclase 2 Gypsum 7 Quartz 3 Calcite 8 Topaz 4 Fluorite 9 Corundum 5 Apatite 10 Diamond
 GLY 4200C LAB 1 PHYSICAL AND CHEMICAL PROPERTIES OF MINERALS IN HAND SPECIMEN There are a number of physical properties which are suitable for field use on hand specimens. One chemical test also falls
GLY 4200C LAB 1 PHYSICAL AND CHEMICAL PROPERTIES OF MINERALS IN HAND SPECIMEN There are a number of physical properties which are suitable for field use on hand specimens. One chemical test also falls
Minerals of Earth s Crust
 Chapter5 Chapter Outline Minerals of Earth s Crust 1 What Is a Mineral? Characteristics of Minerals Kinds of Minerals Crystalline Structure Crystalline Structure of Silicate Minerals Crystalline Structure
Chapter5 Chapter Outline Minerals of Earth s Crust 1 What Is a Mineral? Characteristics of Minerals Kinds of Minerals Crystalline Structure Crystalline Structure of Silicate Minerals Crystalline Structure
2.2 Physical Properties. Physical Properties. Notes due EOC
 Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
ENGINEERING GEOLOGY PROF. DEBASIS ROY DEPARTMENT OF CIVIL ENGINEERING. INDIAN INSTITUTE OF TECHNOLOGY, Kharagpur LECTURE - 5
 ENGINEERING GEOLOGY PROF. DEBASIS ROY DEPARTMENT OF CIVIL ENGINEERING INDIAN INSTITUTE OF TECHNOLOGY, Kharagpur LECTURE - 5 Physical Properties of Minerals Hello everyone and welcome back to the class
ENGINEERING GEOLOGY PROF. DEBASIS ROY DEPARTMENT OF CIVIL ENGINEERING INDIAN INSTITUTE OF TECHNOLOGY, Kharagpur LECTURE - 5 Physical Properties of Minerals Hello everyone and welcome back to the class
Elements and Compounds
 Chemistry Name: Partners: Date: Elements and Compounds Introduction Most of the materials you have used so far in this course were mixtures. If you could take them apart physically, you would find that
Chemistry Name: Partners: Date: Elements and Compounds Introduction Most of the materials you have used so far in this course were mixtures. If you could take them apart physically, you would find that
GEOLOGY 284: MINERALOGY
 Dr. Helen Lang Dept. of Geology & Geography West Virginia University FALL 2014 GEOLOGY 284: MINERALOGY Ore Deposits and Economic Minerals An ore is anything that can be mined from the Earth at a profit,
Dr. Helen Lang Dept. of Geology & Geography West Virginia University FALL 2014 GEOLOGY 284: MINERALOGY Ore Deposits and Economic Minerals An ore is anything that can be mined from the Earth at a profit,
Physical Characteristics of Minerals
 Physical Characteristics of Minerals 1. Introduction 2. Color Idiochromatism and the Chromophores Allochromatism 3. Streak 4. Luster 5. Density 6. Hardness The Mohs Scale The Diamond Indentation Method
Physical Characteristics of Minerals 1. Introduction 2. Color Idiochromatism and the Chromophores Allochromatism 3. Streak 4. Luster 5. Density 6. Hardness The Mohs Scale The Diamond Indentation Method
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter.
 1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
Minerals Basic Building Blocks Elements Compounds Definition of Minerals naturally occurring inorganic solid chemical formula regular structure
 Minerals Rocks are made of minerals. Minerals are made of chemical compounds. Compounds are made of elements. Basic Building Blocks The smallest piece of a chemical compound that still has the properties
Minerals Rocks are made of minerals. Minerals are made of chemical compounds. Compounds are made of elements. Basic Building Blocks The smallest piece of a chemical compound that still has the properties
GY 302: Crystallography & Mineralogy
 UNIVERSITY OF SOUTH ALABAMA GY 302: Crystallography & Mineralogy Lecture 6: Polymorphism and Crystal Habit Instructor: Dr. Douglas Haywick Online Lecture Review 1. Polymorphs and Polymorphism 2. Pseudomorphs
UNIVERSITY OF SOUTH ALABAMA GY 302: Crystallography & Mineralogy Lecture 6: Polymorphism and Crystal Habit Instructor: Dr. Douglas Haywick Online Lecture Review 1. Polymorphs and Polymorphism 2. Pseudomorphs
CHAPTER 6. Natural Mineral Mineral Content Elements In The Minerals Bauxite Aluminium oxide Aluminium, oxygen. Cassiterite Tin oxide Tin, oxygen
 CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
PHEN 612 SPRING 2008 WEEK 13 LAURENT SIMON
 PHEN 612 SPRING 2008 WEEK 13 LAURENT SIMON Crystallization Crystallization is a common separation process in Commodity inorganic industry (e.g., salts) Food industry (e.g., sugars) Pharmaceutical manufacturing
PHEN 612 SPRING 2008 WEEK 13 LAURENT SIMON Crystallization Crystallization is a common separation process in Commodity inorganic industry (e.g., salts) Food industry (e.g., sugars) Pharmaceutical manufacturing
be replaced or replenished in the same amount or less time as it takes to use up the supply. not be replaced as fast as it is used.
 Earth's Resources S6E6: Obtain, evaluate and communicate information about the uses & conservation of various natural resources and how they impact the Earth. A. Ask questions to determine differences
Earth's Resources S6E6: Obtain, evaluate and communicate information about the uses & conservation of various natural resources and how they impact the Earth. A. Ask questions to determine differences
Classification and Nomenclature of Minerals. Unit 3
 Classification and Nomenclature of Minerals Unit 3 Introduction to Berzelian Classification The Berzelian mineral classification system was named in honor of the Swedish chemist and mineralogist Jons Jakob
Classification and Nomenclature of Minerals Unit 3 Introduction to Berzelian Classification The Berzelian mineral classification system was named in honor of the Swedish chemist and mineralogist Jons Jakob
CARBON. Carbon is an element of symbol C, with atomic number 6 (this means it has 6. available to form covalent bonds (tetravalent).
 CARBON Carbon is an element of symbol C, with atomic number 6 (this means it has 6 protons in its core or nuclei), is a non-metallic element, with available electrons available to form covalent bonds (tetravalent).
CARBON Carbon is an element of symbol C, with atomic number 6 (this means it has 6 protons in its core or nuclei), is a non-metallic element, with available electrons available to form covalent bonds (tetravalent).
A few silicate minerals
 A few silicate minerals Stony materials and conservation of the built heritage Natural Stone _Mineralogy_ BR /18 Nesosilicates / Garnets (X 3 2+ Y 2 3+ [SiO 4 ] 3 ) Use: gemstone, abrasive Stony materials
A few silicate minerals Stony materials and conservation of the built heritage Natural Stone _Mineralogy_ BR /18 Nesosilicates / Garnets (X 3 2+ Y 2 3+ [SiO 4 ] 3 ) Use: gemstone, abrasive Stony materials
System/ Cleavage. Native Elements. Sulfides and Sulfosalts
 Element Formula System/ Cleavage Streak Miscellaneous Native Elements Copper Cu Isometric/ irregular masses Brownish High SG, metal sound Sulfur S Orthorhombic/ Rotten egg smell, bright Light yellow massive
Element Formula System/ Cleavage Streak Miscellaneous Native Elements Copper Cu Isometric/ irregular masses Brownish High SG, metal sound Sulfur S Orthorhombic/ Rotten egg smell, bright Light yellow massive
Physical Geology, 15/e
 Lecture Outlines Physical Geology, 15/e Plummer, Carlson & Hammersley Resources Physical Geology 15/e, Chapter 22 Types of Geologic Resources Energy resources petroleum (oil and natural gas), coal, uranium,
Lecture Outlines Physical Geology, 15/e Plummer, Carlson & Hammersley Resources Physical Geology 15/e, Chapter 22 Types of Geologic Resources Energy resources petroleum (oil and natural gas), coal, uranium,
A SAMPLER OF SELECTED 2017 BMC AUCTION SPECIMENS (2017 Auction Date is Saturday, 21 January) Volume 4
 A SAMPLER OF SELECTED 2017 BMC AUCTION SPECIMENS (2017 Auction Date is Saturday, 21 January) Volume 4 Fluorapatite [Ca 5 (PO 4 ) 3 F] Wilberforce area Haliburton Co. Ontario, Canada 18.8 x 8.2 x 7.2 cm
A SAMPLER OF SELECTED 2017 BMC AUCTION SPECIMENS (2017 Auction Date is Saturday, 21 January) Volume 4 Fluorapatite [Ca 5 (PO 4 ) 3 F] Wilberforce area Haliburton Co. Ontario, Canada 18.8 x 8.2 x 7.2 cm
AN INTRODUCTION TO MINERALS
 AN INTRODUCTION TO MINERALS Vivien Gornitz New York City August 1998 Copyright 1998 The New York Mineralogical Club The image below is from the Bulletin of the New York Mineralogical Club Volume 3, No.
AN INTRODUCTION TO MINERALS Vivien Gornitz New York City August 1998 Copyright 1998 The New York Mineralogical Club The image below is from the Bulletin of the New York Mineralogical Club Volume 3, No.
Review key concepts from last lecture (lattice + basis = unit cell) Bravais lattices Important crystal structures Intro to miller indices
 Outline: Review key concepts from last lecture (lattice + basis = unit cell) Bravais lattices Important crystal structures Intro to miller indices Review (example with square lattice) Lattice: square,
Outline: Review key concepts from last lecture (lattice + basis = unit cell) Bravais lattices Important crystal structures Intro to miller indices Review (example with square lattice) Lattice: square,
Energy and Packing. Materials and Packing
 Energy and Packing Non dense, random packing Energy typical neighbor bond length typical neighbor bond energy r Dense, regular packing Energy typical neighbor bond length typical neighbor bond energy r
Energy and Packing Non dense, random packing Energy typical neighbor bond length typical neighbor bond energy r Dense, regular packing Energy typical neighbor bond length typical neighbor bond energy r
Extracting and using metals. ores. native. Only the most unreactive metals such as gold and platinum are found as native metals.
 Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
Color in materials KJM3100 V2006. Pigments
 Color in materials Pigments Black body radiation Incandescence Luminescence Triboluminescence The colour of silver nanoparticles depends on the shape of the particles. A collaborative group of DuPont-led
Color in materials Pigments Black body radiation Incandescence Luminescence Triboluminescence The colour of silver nanoparticles depends on the shape of the particles. A collaborative group of DuPont-led
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Understanding Materials Flowability (Part I) BSH&E SEMINAR
 Understanding Materials Flowability (Part I) Agenda Session I Corporate Introduction Session II Understanding Materials Flowability I Session III Understanding Materials Flowability II Session IV Pneumatic
Understanding Materials Flowability (Part I) Agenda Session I Corporate Introduction Session II Understanding Materials Flowability I Session III Understanding Materials Flowability II Session IV Pneumatic
Ceramic Science 4RO3. Lecture 2. Tannaz Javadi. September 16, 2013
 Ceramic Science 4RO3 Lecture 2 September 16, 2013 Tannaz Javadi Rule 5: In an Ionic structure the chemical environment (cations) that is surrounding an anion should be at least uniform and similar. Although
Ceramic Science 4RO3 Lecture 2 September 16, 2013 Tannaz Javadi Rule 5: In an Ionic structure the chemical environment (cations) that is surrounding an anion should be at least uniform and similar. Although
THE PERIODIC TABLE. Chapter 15
 THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
Free Electron Model What kind of interactions hold metal atoms together? How does this explain high electrical and thermal conductivity?
 Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Mechanical Lustrous
Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Mechanical Lustrous
Tensile Testing for Sheet Metal Formability. Engineering Quality Solutions, Inc. / 4M Partners, LLC PMA Die Design & Simulation Technology
 Tensile Testing for Sheet Metal Formability Daniel J. Schaeffler, Ph.D. President, Engineering Quality Solutions, Inc., www.eqsgroup.com Chief Content Officer, 4M Partners, LLC, www.learning4m.com May
Tensile Testing for Sheet Metal Formability Daniel J. Schaeffler, Ph.D. President, Engineering Quality Solutions, Inc., www.eqsgroup.com Chief Content Officer, 4M Partners, LLC, www.learning4m.com May
Chapter 20 CHEMISTRY. Metallurgy and the Chemistry of Metals. Dr. Ibrahim Suleiman
 CHEMISTRY Chapter 20 Metallurgy and the Chemistry of Metals Dr. Ibrahim Suleiman GENERAL PROPERTIES AND STRUCTURE OF METALS opaque good conductors of heat and electricity high malleability and ductility
CHEMISTRY Chapter 20 Metallurgy and the Chemistry of Metals Dr. Ibrahim Suleiman GENERAL PROPERTIES AND STRUCTURE OF METALS opaque good conductors of heat and electricity high malleability and ductility
Gravity Concentration
 .............. CHAPTER 6 Gravity Concentration Frank F. Aplan INTRODUCTION Gravity concentration is a process in which particles of mixed sizes, shapes, and specific gravities are separated from each other
.............. CHAPTER 6 Gravity Concentration Frank F. Aplan INTRODUCTION Gravity concentration is a process in which particles of mixed sizes, shapes, and specific gravities are separated from each other
Oxides and Hydroxides
 Descriptive Mineralogy Oxides and Hydroxides Classification of the Minerals Non-Silicates Native Elements Halides Sulfides Oxides Hydroxides Carbonates Sulfates Phosphates Silicates Orthosilicates Sorosilicates
Descriptive Mineralogy Oxides and Hydroxides Classification of the Minerals Non-Silicates Native Elements Halides Sulfides Oxides Hydroxides Carbonates Sulfates Phosphates Silicates Orthosilicates Sorosilicates
CHAPTER 9. The Uses of Sulphuric Acid
 CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
SAIOH Interpretation of Elemental Analyses Results. By: Jaco van Rensburg
 SAIOH 2013 Interpretation of Elemental Analyses Results By: Jaco van Rensburg WHY AEROSOL SAMPLING? Air sampling for aerosols, to determine the airborne concentrations of particulate matter, forms a major
SAIOH 2013 Interpretation of Elemental Analyses Results By: Jaco van Rensburg WHY AEROSOL SAMPLING? Air sampling for aerosols, to determine the airborne concentrations of particulate matter, forms a major
MAKING SOLID SOLUTIONS WITH ALKALI HALIDES (AND BREAKING THEM) John B. Brady
 MAKING SOLID SOLUTIONS WITH ALKALI HALIDES (AND BREAKING THEM) John B. Brady Department of Geology Smith College Northampton, MA jbrady@science.smith.edu INTRODUCTION When two cations have the same charge
MAKING SOLID SOLUTIONS WITH ALKALI HALIDES (AND BREAKING THEM) John B. Brady Department of Geology Smith College Northampton, MA jbrady@science.smith.edu INTRODUCTION When two cations have the same charge
Features & Benefits. Provides Objectives-based practice questions Formatted to match the Oklahoma PASS
 eatures & Benefits Provides Objectives-based practice questions ormatted to match the Oklahoma PASS Copyright by The McGraw-Hill Companies, Inc. All rights reserved. Permission is granted to reproduce
eatures & Benefits Provides Objectives-based practice questions ormatted to match the Oklahoma PASS Copyright by The McGraw-Hill Companies, Inc. All rights reserved. Permission is granted to reproduce
LESSON 3 OTHER LAND RESOURCES C H A P T E R 6, C O N S E R V I N G O U R R E S O U R C E S
 LESSON 3 OTHER LAND RESOURCES C H A P T E R 6, C O N S E R V I N G O U R R E S O U R C E S OBJECTIVES Compare renewable and nonrenewable resources. Describe how human activities affect the environment.
LESSON 3 OTHER LAND RESOURCES C H A P T E R 6, C O N S E R V I N G O U R R E S O U R C E S OBJECTIVES Compare renewable and nonrenewable resources. Describe how human activities affect the environment.
GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY
 GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY 1. What is matrix? The ore is generally associated with rock impurities like clay, sand etc. called gangue or matrix 2. What is mineral? The natural
GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY 1. What is matrix? The ore is generally associated with rock impurities like clay, sand etc. called gangue or matrix 2. What is mineral? The natural
X-Ray Diffraction Analysis
 162402 Instrumental Methods of Analysis Unit III X-Ray Diffraction Analysis Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
162402 Instrumental Methods of Analysis Unit III X-Ray Diffraction Analysis Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
Dr. James D. Bittner, Separation Technologies, LLC. Triboelectric separator for beneficiation of fine minerals
 Dr. James D. Bittner, Separation Technologies, LLC Triboelectric separator for beneficiation of fine minerals March 2012 Separation Technologies LLC - ATit Titan America Business Who we are Developers
Dr. James D. Bittner, Separation Technologies, LLC Triboelectric separator for beneficiation of fine minerals March 2012 Separation Technologies LLC - ATit Titan America Business Who we are Developers
SMITHSONIAN CRYSTAL GROWING SET CRYSTAL GROWING PROCEDURES
 Item #28929 Item #2891-08 Ages 10 AND UP WARNING: Only for use by children over 8 years old. To be used solely under the strict supervision of adults that have studied the precautions given in the experimental
Item #28929 Item #2891-08 Ages 10 AND UP WARNING: Only for use by children over 8 years old. To be used solely under the strict supervision of adults that have studied the precautions given in the experimental
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
1 Crystallization: Introduction
 j1 1 Crystallization: Introduction Wolfgang Beckmann The beauty of crystals can be found in both the naturally appearing minerals such as diamonds or quartzite crystals and the industrial products such
j1 1 Crystallization: Introduction Wolfgang Beckmann The beauty of crystals can be found in both the naturally appearing minerals such as diamonds or quartzite crystals and the industrial products such
MINISTER OF ENERGY AND MINERAL RESOURCES OF THE REPUBLIC OF INDONESIA REGULATION OF THE MINISTER OF ENERGY AND MINERAL RESOURCES OF
 MINISTER OF ENERGY AND MINERAL RESOURCES OF THE REPUBLIC OF INDONESIA REGULATION OF THE MINISTER OF ENERGY AND MINERAL RESOURCES OF THE REPUBLIC OF INDONESIA NUMBER: 07 OF 2012 ON IMPROVEMENT OF ADDED
MINISTER OF ENERGY AND MINERAL RESOURCES OF THE REPUBLIC OF INDONESIA REGULATION OF THE MINISTER OF ENERGY AND MINERAL RESOURCES OF THE REPUBLIC OF INDONESIA NUMBER: 07 OF 2012 ON IMPROVEMENT OF ADDED
Covered with a thin layer of oxide at ordinary temperatures.
 1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
Free Electron Model What kind of interactions hold metal atoms together? How does this explain high electrical and thermal conductivity?
 Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Free Electron
Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Free Electron
INDIAN INSTITUTE OF TECHNOLOGY ROORKEE NPTEL NPTEL ONLINE CERTIFICATION COURSE Mechanical Operations. Lecture-10 Size reduction
 INDIAN INSTITUTE OF TECHNOLOGY ROORKEE NPTEL NPTEL ONLINE CERTIFICATION COURSE Mechanical Operations Lecture-10 Size reduction With Dr. Shabina Khanam Department of Chemical Engineering India Institute
INDIAN INSTITUTE OF TECHNOLOGY ROORKEE NPTEL NPTEL ONLINE CERTIFICATION COURSE Mechanical Operations Lecture-10 Size reduction With Dr. Shabina Khanam Department of Chemical Engineering India Institute
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
1. What do you think a resource is? 2. Some resources occur naturally as part of our planet; do you know any?
 Bellringer-Day 01 1. What do you think a resource is? 2. Some resources occur naturally as part of our planet; do you know any? 3. Do you think the resources we use to live today are the same as those
Bellringer-Day 01 1. What do you think a resource is? 2. Some resources occur naturally as part of our planet; do you know any? 3. Do you think the resources we use to live today are the same as those
Extraction of rare earth elements from mine tailings
 Extraction of rare earth elements from mine tailings Sebastiaan Peelman Delft University of Technology Department of Material Science and Engineering Metals Production, Refining and recycling s.peelman@tudelft.nl
Extraction of rare earth elements from mine tailings Sebastiaan Peelman Delft University of Technology Department of Material Science and Engineering Metals Production, Refining and recycling s.peelman@tudelft.nl
5th Grade Science 2nd Nine Weeks Assessment. Name
 5th rade Science 2nd Nine Weeks ssessment Name irections: The photograph below shows several sand dunes in a desert. Use the photograph and your knowledge of science to answer any questions that follow.
5th rade Science 2nd Nine Weeks ssessment Name irections: The photograph below shows several sand dunes in a desert. Use the photograph and your knowledge of science to answer any questions that follow.
FLUORSPAR. Despite its beauty and physical properties, fluorspar is primarily valuable for its fluorine content.
 FLUORSPAR INTRODUCTION When found in nature, fluorspar is known by the mineral name fluorite. Fluorspar (fluorite) is calcium fluoride (CaF 2 ). It is found in a variety of geologic environments. Fluorspar
FLUORSPAR INTRODUCTION When found in nature, fluorspar is known by the mineral name fluorite. Fluorspar (fluorite) is calcium fluoride (CaF 2 ). It is found in a variety of geologic environments. Fluorspar
Guidance document for the Registration of Complex Inorganic Coloured Pigments
 Status: September 2017 Guidance document for the Registration of Complex Inorganic Coloured Pigments 1. Introduction Complex Inorganic Colored Pigments (CICPs) are chemical substances manufactured by means
Status: September 2017 Guidance document for the Registration of Complex Inorganic Coloured Pigments 1. Introduction Complex Inorganic Colored Pigments (CICPs) are chemical substances manufactured by means
The State Law and Order Restoration Council The Myanmar Mines Law (The State Law and Order Restoration Council The Myanmar Mines Law No.
 The State Law and Order Restoration Council The Myanmar Mines Law (The State Law and Order Restoration Council The Myanmar Mines Law No. 8/94) The 2nd Waxing of Tawthalin, 1356 M.E. ( 6th September, 1994)
The State Law and Order Restoration Council The Myanmar Mines Law (The State Law and Order Restoration Council The Myanmar Mines Law No. 8/94) The 2nd Waxing of Tawthalin, 1356 M.E. ( 6th September, 1994)
Constraints in Using Manufactured Sands in Concrete Pavements
 Constraints in Using Manufactured Sands in Concrete Pavements Ion Dumitru Manager Tony Song Senior Development Engineer May 2012 Shortage of natural sands (Eastern coast) Manufactured sands, recycled products
Constraints in Using Manufactured Sands in Concrete Pavements Ion Dumitru Manager Tony Song Senior Development Engineer May 2012 Shortage of natural sands (Eastern coast) Manufactured sands, recycled products
Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons.
 Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons. A.(16) An electrochemical cell is prepared with a strip of manganese metal dipping in to a 1.0 M MnSO 4 solution
Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons. A.(16) An electrochemical cell is prepared with a strip of manganese metal dipping in to a 1.0 M MnSO 4 solution
THERMOLUMINESCENCE DOSIMETRY STUDIES OF INDUSTRIALLY IMPORTANT MINERALS CHAPTER-1 GENERAL INTRODUCTION
 THERMOLUMINESCENCE DOSIMETRY STUDIES OF INDUSTRIALLY IMPORTANT MINERALS CHAPTER-1 GENERAL INTRODUCTION 1.1 INTRODUCTION It has been more than 400 years since Robert Boyle incidentally discovered the thermoluminescence
THERMOLUMINESCENCE DOSIMETRY STUDIES OF INDUSTRIALLY IMPORTANT MINERALS CHAPTER-1 GENERAL INTRODUCTION 1.1 INTRODUCTION It has been more than 400 years since Robert Boyle incidentally discovered the thermoluminescence
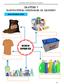
Saskatchewan s Mineral Resources Lesson: Rocks and Minerals in Your Life
 Saskatchewan s Mineral Resources Lesson: Rocks and Minerals in Your Life Overview Students identify products they use in their daily activities, and discover the rocks and minerals used to make them. Source:
Saskatchewan s Mineral Resources Lesson: Rocks and Minerals in Your Life Overview Students identify products they use in their daily activities, and discover the rocks and minerals used to make them. Source:
Diffraction: Powder Method
 Diffraction: Powder Method Diffraction Methods Diffraction can occur whenever Bragg s law λ = d sin θ is satisfied. With monochromatic x-rays and arbitrary setting of a single crystal in a beam generally
Diffraction: Powder Method Diffraction Methods Diffraction can occur whenever Bragg s law λ = d sin θ is satisfied. With monochromatic x-rays and arbitrary setting of a single crystal in a beam generally
Iron filings (Fe) 56g IRON + SULPHUR IRON SULPHIDE
 W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
Mat E 272 Lecture 19: Cast Irons
 Mat E 272 Lecture 19: Cast Irons November 8, 2001 Introduction: One reason steels and cast iron alloys find such wide-ranging applications and dominate industrial metal production is because of how they
Mat E 272 Lecture 19: Cast Irons November 8, 2001 Introduction: One reason steels and cast iron alloys find such wide-ranging applications and dominate industrial metal production is because of how they
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Skills in Science. Lab equipment. (Always draw 2D) Drawings below are NOT to scale. Beaker - A general purpose container with a pouring lip.
 Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
A Classification of Crystalline Models. Harold M. Summers, III
 A Classification of Crystalline Models By Harold M. Summers, III A senior thesis submitted to fulfill the requirements for the degree of B.S. in Geology, 1995 The Ohio State University ~~T- Rodney T..
A Classification of Crystalline Models By Harold M. Summers, III A senior thesis submitted to fulfill the requirements for the degree of B.S. in Geology, 1995 The Ohio State University ~~T- Rodney T..
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Q. 1 Q. 25 carry ONE mark each
 Q. 1 Q. 25 carry ONE mark each Q.1 Consider the following five readings from an experiment: 19, 17, 15, 13, 11. The standard deviation of the readings is. Q.2 yy(xx+h) yy(xx) h (A) dddd dddd is a numerical
Q. 1 Q. 25 carry ONE mark each Q.1 Consider the following five readings from an experiment: 19, 17, 15, 13, 11. The standard deviation of the readings is. Q.2 yy(xx+h) yy(xx) h (A) dddd dddd is a numerical
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet
 Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
2.3 Chemical Changes corrosion Kinds of Corrosion
 2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
Mixtures in Industry
 3.7 Mixtures in Industry Key Question: How are mixtures separated in industry? Many industries separate mixtures to make pure products. In this section, you will learn about three industries that separate
3.7 Mixtures in Industry Key Question: How are mixtures separated in industry? Many industries separate mixtures to make pure products. In this section, you will learn about three industries that separate
Analytical Methods for Materials
 Analytical Methods for Materials Lesson 13 Crystallography and Crystal Structures continued Chapter 6 in Waseda Excerpt from ASM Metals Handbook. Suggested Reading 337 Notation for crystal structures 1.
Analytical Methods for Materials Lesson 13 Crystallography and Crystal Structures continued Chapter 6 in Waseda Excerpt from ASM Metals Handbook. Suggested Reading 337 Notation for crystal structures 1.
Ore sorting is the separation of a target mineralogy, ore or element from waste at an individual particle level.
 TOMRA Ore Sorting What is ore sorting? Ore sorting is the separation of a target mineralogy, ore or element from waste at an individual particle level. The ore is sorted to remove waste and improve the
TOMRA Ore Sorting What is ore sorting? Ore sorting is the separation of a target mineralogy, ore or element from waste at an individual particle level. The ore is sorted to remove waste and improve the
Cu/Ag Eutectic System
 Eutectic Systems The simplest kind of system with two solid phases is called a eutectic system. A eutectic system contains two solid phases at low temperature. These phases may have different crystal structures,
Eutectic Systems The simplest kind of system with two solid phases is called a eutectic system. A eutectic system contains two solid phases at low temperature. These phases may have different crystal structures,
true grit minerals CUMI lative range of EMD PRODUCT CATALOG 100 years US $ 3 billion 29 companies +30,000 people MURUGAPPA GROUP
 MURUGAPPA GROUP 100 years US $ 3 billion 29 companies +30,000 people EMD ELECTRO MINERALS DIVISION CUMI lative range of true grit minerals PRODUCT CATALOG FUSED PRODUCTS MACRO REGULAR BROWN FUSED ALUMINA
MURUGAPPA GROUP 100 years US $ 3 billion 29 companies +30,000 people EMD ELECTRO MINERALS DIVISION CUMI lative range of true grit minerals PRODUCT CATALOG FUSED PRODUCTS MACRO REGULAR BROWN FUSED ALUMINA
UTILIZATION OF ALUMINIUM PROCESSING WASTE AS RAW MATERIALS IN THE MANUFACTURE OF CEMENT
 UTILIZATION OF ALUMINIUM PROCESSING WASTE AS RAW MATERIALS IN THE MANUFACTURE OF CEMENT M Pareek, Digvijay Singh, D Yadav*, S K Chaturvedi* and M M Ali* Century Metal Recycling Pvt. Ltd., Palwal, Haryana
UTILIZATION OF ALUMINIUM PROCESSING WASTE AS RAW MATERIALS IN THE MANUFACTURE OF CEMENT M Pareek, Digvijay Singh, D Yadav*, S K Chaturvedi* and M M Ali* Century Metal Recycling Pvt. Ltd., Palwal, Haryana
WF VAN DER VYVER
 DETERMINATION OF FACTORS INFLUENCING THE DEGREE OF REDUCTION DISINTEGRATION IN NORTHERN CAPE LUMP ORE AND THE ROLE OF GANGUE MINERALS IN THE PROPAGATION OF CRACKS WF VAN DER VYVER Dissertation submitted
DETERMINATION OF FACTORS INFLUENCING THE DEGREE OF REDUCTION DISINTEGRATION IN NORTHERN CAPE LUMP ORE AND THE ROLE OF GANGUE MINERALS IN THE PROPAGATION OF CRACKS WF VAN DER VYVER Dissertation submitted
Slidmekanismer og slidforebyggelse
 Slidmekanismer og slidforebyggelse Niels Bay DTU-Mekanik FMV Temadag om Slid på Metaller Scandic Hotel, København 15. november 212. Mechanisms of wear Primary mechanisms 1. Adhesive wear 2. Abrasive wear
Slidmekanismer og slidforebyggelse Niels Bay DTU-Mekanik FMV Temadag om Slid på Metaller Scandic Hotel, København 15. november 212. Mechanisms of wear Primary mechanisms 1. Adhesive wear 2. Abrasive wear
4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. sulfur 16
 4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. silver 47 sulfur 16 copper 29 Some are named from the natural substance that they are
4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. silver 47 sulfur 16 copper 29 Some are named from the natural substance that they are
Lesson note 5: Separation of Mixtures. Mixtures. Mixtures. Separation Methods: Many different types of mixtures Examples:
 Lesson note 5: Separation of Mixtures Mixtures Many different types of mixtures Examples: - air (gas-gas mixture), - sea water(liquid-solid mixture), - crude oil (liquid-liquid mixture) The mixtures can
Lesson note 5: Separation of Mixtures Mixtures Many different types of mixtures Examples: - air (gas-gas mixture), - sea water(liquid-solid mixture), - crude oil (liquid-liquid mixture) The mixtures can
Physical Behavior of Metals
 Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
