The octavolateralis sensory system of an aquatic vertebrate
|
|
|
- Myles McDonald
- 6 years ago
- Views:
Transcription
1 Supernumerary neuromasts in the posterior lateral line of zebrafish lacking peripheral glia Hernán López-Schier and A. J. Hudspeth* Howard Hughes Medical Institute and Laboratory of Sensory Neuroscience, The Rockefeller University, 1230 York Avenue, New York, NY Contributed by A. J. Hudspeth, December 21, 2004 The lateral line and its associated sensory nerves develop from cephalic epithelial thickenings called neurogenic placodes. In the zebrafish, the transcription factor neurogenin 1 is essential for the generation of the sensory ganglion from the placode, but is dispensable for the migration of the primordium and the initial development of neuromasts. We find that inactivation of the gene encoding neurogenin 1 leads to the development of over twice the normal number of neuromasts along the posterior lateral line of zebrafish larvae. Mutation of the gene encoding another transcription factor, sox10, has a similar effect. After a normal number of proneuromasts is initially deposited by the migrating primordia, interneuromast cells divide and differentiate to form the extra neuromasts. Our results indicate that the development of these intercalary neuromasts occurs principally because of the absence of neural crest-derived peripheral glia, which evidently inhibit the assembly of interneuromast cells into neuromasts. acousticolateralis system hair cell neurogenin The octavolateralis sensory system of an aquatic vertebrate includes both the internal ear and the lateral-line system, the latter serving to detect mechanical disturbances in the surrounding water (1 3). The lateral line comprises a stereotyped array of sensory structures called neuromasts, each containing mechanoreceptive hair cells as well as supporting cells. During the embryonic development of the zebrafish, cephalic ectodermal placodes generate both the neuromasts and their sensory ganglia (4). The posterior lateral-line placode contains 120 cells, the caudal three-quarters of which constitute the first primordium that initiates a posteriorward migration 20 h postfertilization (hpf) (5, 6). The first primordium completes its journey to the animal s tail at 40 hpf, leaving behind seven to nine proneuromasts and a trail of interneuromast cells connecting them (7, 8). Soon thereafter, a second, smaller primordium begins to migrate along the same path, depositing two or three proneuromasts over the rostral part of the trunk (8, 9). Neuromasts of the posterior lateral line mature progressively from head to tail; by the fifth day of development, 9 11 neuromasts contain functional hair cells. The system later gains complexity through the development of additional neuromasts and the growth and elongation of existing clusters (10). The rostral portion of the posterior lateral-line placode forms a sensory ganglion (6), whose development depends on the activity of the transcription factor neurogenin 1 (11). The somata of neurons in this ganglion remain near the origin of the placode, but extend axons along the path followed by the lateral-line primordia (6, 12, 13). The lateral-line nerve, in turn, serves as a scaffold to guide the migration of a subset of neural crest-derived peripheral glia associated with the lateral line (14). Although sensory axons are dispensable for the migration of the primordia and the initial development of neuromasts, the consequences of an absence of glia on the postembryonic development of the lateral-line organ are not known. Here, we take advantage of mutations that alter the number of glial cells to address this issue. Materials and Methods Zebrafish Strains and Husbandry. Zebrafish were maintained in our facility under standard conditions. Wild-type fish were of the Tup Longfin or Tübingen strains. Animals mutant for the gene encoding neurogenin 1, a gift of N. Hopkins (Massachusetts Institute of Technology, Cambridge), carried the hi1059 allele (15). KC66 transgenic animals (16) expressing histone 2B-GFP and mutants bearing the m241 allele of the gene encoding sox10 (17) were obtained from the Zebrafish International Resource Center (Eugene, OR). Morpholino Oligonucleotides. Morpholinos (11) directed against the mrna encoding neurogenin 1 (5 -ACGATCTCCATTGT- TGATAACCTGG-3 ) were purchased from Gene Tools (Corvalis, OR). Diluted to a concentration of 500 M in 1 Danieau s solution, the morpholinos were injected into one- to four-cell embryos of three different wild-type strains: Tup Longfin, Tübingen, and the histone 2B-GFP transgenics in an AB background strain. Uninjected embryos maintained identically served as controls. Labeling Procedures and Imaging. Immunohistochemical and vital labeling of hair cells were conducted as described (8). Antisera and monoclonal antibodies were used at the following dilutions: rabbit anti-claudin b, 1 500; rabbit anti-parvalbumin 3, 1 2,000; rabbit anti-phosphohistone H3 (Upstate Biotechnology), 1 1,000; mouse monoclonal antibody 6D2, 1 5; mouse monoclonal antibody HCS1, 1 20; and rat anti- -tubulin (AbCam), 1 1,000. Fluorescein- and Texas red-labeled donkey anti-mouse, -rat, and -rabbit immunoglobin secondary antibodies (The Jackson Laboratory) were used at a dilution of TO-PRO 3 was purchased from Molecular Probes. Specimens were examined with a Zeiss Axioplan II microscope and images were acquired with a Nikon Coolpix-995 digital camera. Confocal images were obtained with a BioRad MRC-1024ES scan head on a Zeiss Axiovert microscope or with a Zeiss Meta510 system. For live imaging, animals mutant for the gene encoding sox10 were incubated in fluorescein-labeled ceramide (Molecular Probes) for 12 h at 28.5 C in the dark, then rinsed and kept in system water until analysis. Each larva was mounted on a glass coverslip, covered with 1% low-melting-point agarose in a 610 M solution of the anesthetic 3-aminobenzoic acid ethyl ester, and imaged with a confocal system mounted on an inverted microscope. Statistical results are provided as means standard deviations for the indicated number of observations. Results Mutant Fish Develop Supernumerary Neuromasts. The posterior lateral-line placode gives rise to the first migratory primordium and a sensory ganglion. Although blocking the production of the basic helix-loop-helix transcription factor neurogenin 1 with morpholinos prevents the formation of the sensory ganglion, this procedure does not affect the initial development of neuromasts (11). However, it is not known how a lack of innervation affects Abbreviations: hpf, hours postfertilization; dpf, days postfertilization. *To whom correspondence should be addressed. hudspaj@rockefeller.edu by The National Academy of Sciences of the USA PNAS February 1, 2005 vol. 102 no. 5 cgi doi pnas
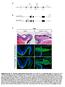
2 Fig. 1. ngn1 mutant zebrafish develop extra neuromasts postembryonically. (A) When treated with the fluorescent compound 4-Di-2-ASP at 6 dpf, a wild-type larva displays only seven to nine neuromasts with functional hair cells on one side of its trunk. The neuromasts on the animal s opposite side appear as diffuse spots of fluorescence. (B) Anngn1 mutant at 6 dpf possesses more than twice as many neuromasts in its posterior lateral line. The number and pattern of neuromasts on the head is essentially normal, as is the animal s pigmentation. (C) At 6 dpf, an animal injected with a morpholino directed against the ngn1 transcript displays a pattern of supernumerary neuromasts comparable to that of an ngn1 mutant. Tilting of the larva separates the images of the neuromasts on the animal s two sides. (D) In a wild-type larva, an antibody against claudin b (green) labels the first primordium and the trail of cells left behind it; the nuclear stain TO-PRO 3 (red) marks all cells. (E) The primordium of an ngn1 mutant does not contain significantly more cells. (F) In a wild-type animal at 24 hpf, antiserum against -tubulin strongly labels primordial cells (green), whereas that against phosphohistone H3 marks cells in mitosis (red). (G) The primordium is not more active mitotically in a homozygous ngn1 mutant larva. (H) When treated with 4-Di-2-ASP at 48 hpf, a wild-type animal shows only five or six neuromasts with functional hair cells on each side of its trunk. (I) At the same stage, an ngn1 mutant bears an equivalent number of mature neuromasts. In these and all subsequent illustrations, the animal s anterior is oriented to the left and its dorsum is situated to the top. (Scale bars, 50 m for D G and 100 m for A C, H, and I.) the postembryonic development of neuromasts. We observed that a mutation in the neurogenin 1 (ngn1) gene leads to the formation of supernumerary neuromasts along the posterior lateral line of zebrafish larvae (Fig. 1 A and B). The average number of neuromasts along one side of an ngn1 mutant at 6 days postfertilization (dpf) is (n 6), over twice the complement of (n 10) characteristic of wild-type animals. We reproduced this phenotype by injecting morpholinos directed against the ngn1 transcript (Fig. 1C). One possible explanation for the presence of supernumerary neuromasts is that the lack of neurogenin 1 function increases the number of primordial cells before migration begins, for example by biasing the development of the entire placode into nonneuronal cell fates. To address this point, we doubly labeled wild-type and ngn1 mutant embryos at 24 hpf with an antibody to claudin b to identify the primordium (8) and with TO-PRO 3 to mark cellular nuclei. We found only a modest increase in the number of primordial cells in the mutants: (n 10) for ngn1 larvae compared to (n 10) for wild-type animals (Fig. 1 D and E). The observed increment in cell number cannot by itself account for a complete duplication of the neuromasts in ngn1 mutants. Cells in the first primordium of the posterior lateral line characteristically undergo mitosis during migration; even after depositing approximately six neuromasts with 30 cells apiece, the primordium still contains cells when it reaches the tail of the fish, where it produces two or three terminal neuromasts (18). A second possibility to explain the elevated number of neuromasts in ngn1 mutants is that their primordia are mitotically more active. We tested this possibility in two complementary ways. First, we used an antibody against the phosphorylated form of histone H3 to score the number of primordial cells entering mitosis in wild-type and ngn1 mutant animals (Fig. 1 F and G). Second, we injected ngn1 morpholinos into the eggs of animals expressing a histone 2B-GFP transgene that allows the observation of mitotic events in primordial cells in vivo (12, 16) DEVELOPMENTAL BIOLOGY López-Schier and Hudspeth PNAS February 1, 2005 vol. 102 no
3 Fig. 2. The HCS1 antiserum marks mature hair cells in neuromasts. (A) In a triply labeled wild-type larva, -tubulin is present throughout the neuromast, whereas parvalbumin 3 and HCS1 occur in mature hair cells. (B) In a wild-type larva at 48 hpf, a neuromast deposited by the first primordium already bears mature hair cells labeled for HCS1 (red). The migrating second primordium is negative for this marker. Both structures are labeled by immunoreactivity against claudin b (green). (Scale bars, 10 m.) (data not shown). Neither experiment revealed an augmented mitotic rate within the migrating primordium of fish lacking neurogenin 1. We also analyzed animals at 48 hpf and found no difference in the size or behavior of the second primordium between wild-type and ngn1 mutant animals (data not shown). Furthermore, larvae of both types bore an equivalent number of neuromasts at this stage (Fig. 1 H and I). Therefore, we conclude that the extra neuromasts in ngn1 mutants do not originate during the initial development of the lateral line, and in particular from larger or mitotically more active primordia. Additional Neuromasts Develop from Interneuromast Cells. The observation that wild-type and ngn1 mutant animals are indistinguishable until 48 hpf indicates that additional neuromasts develop after the entire initial set of neuromasts has been deposited. Therefore, we used antibodies to claudin b and to HCS1, a hair cell-specific marker (Fig. 2) (19), to analyze ngn1 mutant animals at 6-h intervals commencing at 48 hpf. Around 52 hpf, the interneuromast cells left behind by the migrating first primordium began to coalesce in small groups between primary neuromasts (Fig. 3 A and B). These interneuromast cells divided mitotically (Fig. 3C) and, by 56 hpf, formed clusters resembling small proneuromasts (Fig. 3D). These clusters had already constituted several new neuromasts by 72 hpf (Fig. 3E). At 120 hpf, ngn1 mutants bore twice the number of mature neuromasts found in wild-type animals (Fig. 3F). These results indicate that proliferating interneuromast cells are the source of the additional, intercalary neuromasts in ngn1 mutant zebrafish. Lack of Glia Antecedes the Development of Extra Neuromasts. Our previous results suggest that the development of extra neuromasts in ngn1 mutant fish stems from the absence of sensory innervation to the posterior lateral line. However, in an independent line of investigation, we discovered that colourless (cls) mutant zebrafish also form supernumerary neuromasts along the posterior lateral line. The time course of development of additional neuromasts in cls larvae matches that of ngn1 animals (Fig. 4 A F and Movie 1, which is published as supporting information on the PNAS web site). The cls gene encodes sox10, a transcription factor associated with the development of nonectomesenchymal derivatives of the neural crest (17, 20, 21). The absence of sox10 leads to deficient pigmentation and defective development of peripheral glia in the zebrafish, but does not affect the development of sensory axons innervating the lateral line (14, 17). We used the monoclonal antibody 6D2, which recognizes a carbohydrate epitope of the piscine P 0 -like myelin glycoproteins IP1 and IP2 (22, 23), to confirm that glial cells are absent along the horizontal myoseptum in both cls (Fig. 4 G and H) and ngn1 mutant larvae (Fig. 4 I and J). Together, these observations indicate that the absence of glia, rather than that of sensory axons, is the most likely cause of supernumerary neuromasts in these mutant zebrafish. Discussion The number of neuromasts is highly regulated in wild-type zebrafish larvae (18, 24). However, in ngn1 and cls mutants, more than twice the normal complement of neuromasts forms in the posterior lateral line. Because the posterior lateral-line ganglion fails to develop normally in ngn1 mutants, a possible explanation for the presence of extra neuromasts is that the entire posterior lateral-line placode constitutes a primordium that is consequently endowed with more cells before its migration commences. However, our results indicate that the primordia in ngn1 mutant fish are not significantly more populated than those in wild-type animals. Moreover, primordial cells in ngn1 mutant embryos proliferate at a normal rate. Finally, wild-type and mutant animals are indistinguishable at 48 hpf, indicating that additional neuromasts do not originate during the migration of the first and second primordia. Therefore, the formation of additional neuromasts cannot be explained by the presence of a larger pool of neuromast precursor cells. We confirmed this inference by analysis of cls mutant zebrafish, which form sensory ganglia and primordia of a normal size, but also develop twice the normal number of neuromasts in the posterior lateral line. We used a combination of specific molecular markers and live imaging to establish that ngn1 and cls mutants begin to form extrasensory clusters after the primary set of neuromasts has been deposited, at 52 hpf. Additional neuromasts originate through the coalescence of mitotically active interneuromast cells in ngn1 and cls larvae. This finding suggests that the absence of a subset of neural crest-derived cells, rather than of sensory innervation, underlies the development of supernumerary neuromasts in these mutants. The production of supernumerary neuromasts in the two mutant lines under investigation represents only a partial failure of the mechanisms that normally determine the distribution of these sensory organs. It is noteworthy that, despite their excess number, the resultant neuromasts are spaced in a highly regular array. Notwithstanding a deficit in the inhibitory influence that neural-crest derivatives ordinarily exert on the differentiation of interneuromast cells, an intact signaling system evidently continues to regulate the multiplication or coalescence of these cells. In fishes and amphibians, neuromasts may have a dual origin from placodes and the neural crest (25). Although the fate of neural-crest cells in the lateral line remains uncertain, these cells could associate with the interneuromast cells and prevent the later from forming more neuromasts. One likely possibility is that these cells differentiate as peripheral glia that traverse the path formed by the lateral-line nerve and accumulate along the posterior lateral-line nerve adjacent to the interneuromast cells cgi doi pnas López-Schier and Hudspeth
4 Fig. 3. Interneuromast cells form extra neuromasts in ngn1 mutant larvae. (A) In a wild-type larva at 52 hpf, an antiserum against claudin b (green) reveals cells derived from the posterior lateral-line placode. HCS1 labeling (red) marks mature hair cells; the arrowhead designates the migrating second primordium. (B) At the same stage, a homozygous ngn1 larva displays claudin b-positive cell clusters (asterisks) between the mature neuromasts. The arrowhead marks the second primordium. (C) Labeling of an ngn1 mutant embryo at 52 hpf for phosphohistone H3 (red) shows that interneuromast cells remain mitotically active. Labeling for -tubulin (green) delineates all cells. (D) Labeling for claudin b demonstrates that the interneuromast cells of an ngn1 mutant begin to coalesce at 52 hpf. (E) By 72 hpf, these cell clusters resolve progressively into new neuromasts. (F) Anngn1 mutant larva labeled for HCS1 (red) and TO-PRO 3 (blue) confirms the presence of mature hair cells within the additional neuromasts at 120 hpf. (Scale bars, 10 m for C E and 50 m for A, B, and F.) The initial neuromasts form in the absence of these glia, because the initial patterning of the lateral line is established within the moving primordium, ahead of the neural crest-derived glia (26). At later stages of development, zebrafish normally produce numerous neuromasts in addition to those deposited by the primordia. If interneuromast cells are the source of these new neuromasts, the proliferation observed in the mutant animals represents an acceleration of the normal developmental schedule triggered by the absence of neural crest-derived cells. The phenomenon reported here may thus represent an example of heterochrony, a change in the developmental timing of a particular structure relative to that of the remainder of a developing organism. Occurring in a sensory system, such a heterochronic change could contribute to a mutant animal s ability to colonize new ecological niches, avoid predators, or capture novel prey. By allowing the animal to outperform its predecessors, these traits could contribute to speciation. Although variations in developmental timing are evident during the formation of the lateral lines in various fish species, the cellular mechanisms underlying these changes remain unknown (9, 27). We suspect that changes in the ability of neural crest cells to migrate, multiply, and survive influence the timing of development of neuromasts, thus underlying heterochrony at the organismal level (27, 28). Various Danio species display unique patterns of pigmentation that stem from changes in the development, migration, or survival of neural-crest derivatives such as melanocytes. By the same token, it may be valuable to analyze the development of neuromasts across the genus. The identification of the genes mutated in zebrafish with defects in the development and behavior of the neural crest and in the number of neuromasts DEVELOPMENTAL BIOLOGY López-Schier and Hudspeth PNAS February 1, 2005 vol. 102 no
5 Fig. 4. Additional neuromasts also develop in cls mutant larvae. (A) A bright-field micrograph shows a wild-type larva at 48 hpf. (B) A homozygous cls larva displays a similar pace of development. (C) Exposure of the wild-type animal to the fluorescent compound 4-Di-2-ASP reveals the pattern of functional neuromasts. (D) The mutant larva bears an equivalent number of neuromasts at this stage. (E) A bright-field image of a cls mutant larva at 6 dpf demonstrates the absence of pigmentation on the animal s head and along its body. (F) Treated with 4-Di-2-ASP, the same animal displays 25 neuromasts on the left side of its trunk. However, the number of neuromasts on the head resembles that of a wild-type larva. (G) The marker 6D2 delineates the distribution of glia in the posterior lateral line of a wild-type larva at 72 hpf. (H) At the same stage, a cls mutant lacks peripheral glia along its trunk. (I) 6D2-positive cells occur along the nerve contacting a neuromast in a wild-type larva. (J) No cells of the posterior lateral-line nerve label with the 6D2 antibody in an ngn1 mutant fish. (Scale bars, 100 m for A H and 20 m for I and J.) should allow experimental exploration of the genetic basis of heterochrony (29, 30). We thank N. Hopkins (Massachusetts Institute of Technology, Cambridge) and the Zebrafish International Resource Center for zebrafish lines, J. Corwin (University of Virginia School of Medicine, Charlottesville) and G. Jeserich (University of Osnabrück, Osnabrück, Germany) for antisera, D. Elreda and A. North (The Rockefeller University Imaging Center) for advice on microscopy, and A. Afolalu and P. Espitia for expert maintenance of the zebrafish colony. We also acknowledge the kindness of T. Piotrowski and D. Raible for communicating their results before publication. A. Ghysen, R. G. Northcutt, T. Whitfield, and the members of our research group provided valuable comments on the manuscript. This research was supported by National Institutes of Health Grant DC During this investigation, H.L.-S. was a fellow of the Wellcome Trust and subsequently a Howard Hughes Medical Institute fellow of the Life Sciences Research Foundation. A.J.H. is an Investigator of Howard Hughes Medical Institute. 1. Lowenstein, O. (1967) in Lateral Line Detectors, ed. Cahn, P. (Indiana Univ. Press, Bloomington), pp Dijkgraaf, S. (1989) in The Mechanosensory Lateral Line: Neurobiology and Evolution, eds. Coombs, S., Görner, P. & Münz, H. (Springer, New York), pp Montgomery, J., Baker, C. & Carton, G. (1997) Nature 389, Ghysen, A. & Dambly-Chaudière, C. (2004) Curr. Opin. Neurobiol. 14, Metcalfe, W. K., Kimmel, C. B. & Schabtach, E. (1985) J. Comp. Neurol. 233, Metcalfe, W. K. (1985) J. Comp. Neurol. 238, David, N. B., Sapède, D., Saint-Etienne, L., Thisse, C., Thisse, B., Dambly- Chaudière, C., Rosa, F. M. & Ghysen, A. (2002) Proc. Natl. Acad. Sci. USA 99, López-Schier, H., Starr, C. J., Kappler, J. A., Kollmar, R. & Hudspeth, A. J. (2004) Dev. Cell. 7, Sapède, D., Gompel, N., Dambly-Chaudière, C. & Ghysen, A. (2002) Development (Cambridge, U.K.) 129, Ledent, V. (2002) Development (Cambridge, U.K.) 129, Andermann, P., Ungos, J. & Raible, D. W. (2002) Dev. Biol. 251, Gilmour, D., Knaut, H., Maischein, H.M., Nüsslein-Volhard, C. (2004) Nat. Neurosci. 7, Li, Q., Shirabe, K. & Kuwada, J. Y. (2004) Dev. Biol. 269, Gilmour, D. T., Maischein, H. M. & Nüsslein-Volhard, C. (2002) Neuron 34, Golling, G., Amsterdam, A., Sun, Z., Antonelli, M., Maldonado, E., Chen, W., Burgess, S., Haldi, M., Artzt, K., Farrington, S., et al. (2002) Nat. Genet. 31, Pauls, S., Geldmacher-Voss, B. & Campos-Ortega, J. A. (2001) Dev. Genes Evol. 211, cgi doi pnas López-Schier and Hudspeth
6 17. Kelsh, R. N. & Eisen, J. S. (2000) Development (Cambridge, U.K.) 127, Gompel, N., Cubedo, N., Thisse, C., Thisse, B., Dambly-Chaudière, C. & Ghysen, A. (2001) Mech. Dev. 105, Gale, J. E., Meyers, J. R. & Corwin, J. T. (2000) J. Assoc. Res. Otolaryngol. 1, Dutton, K., Dutton, J. R., Pauliny, A. & Kelsh, R. N. (2001) Genesis 30, Dutton, K. A., Pauliny, A., Lopes, S. S., Elworthy, S., Carney, T. J., Rauch, J., Geisler, R., Haffter, P. & Kelsh, R. N. (2001) Development (Cambridge, U.K.) 128, Jeserich, G. & Rauen, T. (1990) Glia 3, Bastmeyer, M., Jeserich, G. & Stuermer, C. A. (1994) Glia 11, Pichon, F. & Ghysen, A. (2004) Evol. Dev. 6, Collazo, A., Fraser, S. E. & Mabee, P. M. (1994) Science 264, Itoh, M. & Chitnis, A. B. (2001) Mech. Dev. 102, Webb, J. F. (1989) in The Mechanosensory Lateral Line: Neurobiology and Evolution, eds. Coombs, S., Görner, P. & Münz, H. (Springer, New York), pp Parichy, D. M. (2001) in Beyond Heterochrony: The Evolution of Development, ed. Zelditch, M. L. (Wiley, New York), pp Kelsh, R. N., Brand, M., Jiang, Y. J., Heisenberg, C. P., Lin, S., Haffter, P., Odenthal, J., Mullins, M. C., van Eeden, F. J., Furutani-Seiki, M., et al. (1996) Development (Cambridge, U.K.) 123, Whitfield, T. T., Granato, M., van Eeden, F. J., Schach, U., Brand, M., Furutani-Seiki, M., Haffter, P., Hammerschmidt, M., Heisenberg, C. P., Jiang, Y. J., et al. (1996) Development (Cambridge, U.K.) 123, DEVELOPMENTAL BIOLOGY López-Schier and Hudspeth PNAS February 1, 2005 vol. 102 no
Chemokine Signaling Mediates Self-Organizing Tissue Migration in the Zebrafish Lateral Line
 Developmental Cell 10, 673 680, May, 2006 ª2006 Elsevier Inc. DOI 10.1016/j.devcel.2006.02.019 Chemokine Signaling Mediates Self-Organizing Tissue Migration in the Zebrafish Lateral Line Short Article
Developmental Cell 10, 673 680, May, 2006 ª2006 Elsevier Inc. DOI 10.1016/j.devcel.2006.02.019 Chemokine Signaling Mediates Self-Organizing Tissue Migration in the Zebrafish Lateral Line Short Article
Supplementary Fig. S1. Schematic representation of mouse lines Pax6 fl/fl and mrx-cre used in this study. (A) To generate Pax6 fl/ fl
 Supplementary Fig. S1. Schematic representation of mouse lines Pax6 fl/fl and mrx-cre used in this study. (A) To generate Pax6 fl/ fl, loxp sites flanking exons 3-6 (red arrowheads) were introduced into
Supplementary Fig. S1. Schematic representation of mouse lines Pax6 fl/fl and mrx-cre used in this study. (A) To generate Pax6 fl/ fl, loxp sites flanking exons 3-6 (red arrowheads) were introduced into
Lef1 regulates Dusp6 to influence neuromast formation and spacing in the zebrafish posterior lateral line primordium
 RESEARCH ARTICLE 2387 Development 140, 2387-2397 (2013) doi:10.1242/dev.091348 2013. Published by The Company of Biologists Ltd Lef1 regulates Dusp6 to influence neuromast formation and spacing in the
RESEARCH ARTICLE 2387 Development 140, 2387-2397 (2013) doi:10.1242/dev.091348 2013. Published by The Company of Biologists Ltd Lef1 regulates Dusp6 to influence neuromast formation and spacing in the
7.22 Example Problems for Exam 1 The exam will be of this format. It will consist of 2-3 sets scenarios.
 Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 10 7.22 Fall 2005 sample exam
Massachusetts Institute of Technology Department of Biology 7.22, Fall 2005 - Developmental Biology Instructors: Professor Hazel Sive, Professor Martha Constantine-Paton 1 of 10 7.22 Fall 2005 sample exam
Trasposable elements: Uses of P elements Problem set B at the end
 Trasposable elements: Uses of P elements Problem set B at the end P-elements have revolutionized the way Drosophila geneticists conduct their research. Here, we will discuss just a few of the approaches
Trasposable elements: Uses of P elements Problem set B at the end P-elements have revolutionized the way Drosophila geneticists conduct their research. Here, we will discuss just a few of the approaches
Lecture 20: Drosophila melanogaster
 Lecture 20: Drosophila melanogaster Model organisms Polytene chromosome Life cycle P elements and transformation Embryogenesis Read textbook: 732-744; Fig. 20.4; 20.10; 20.15-26 www.mhhe.com/hartwell3
Lecture 20: Drosophila melanogaster Model organisms Polytene chromosome Life cycle P elements and transformation Embryogenesis Read textbook: 732-744; Fig. 20.4; 20.10; 20.15-26 www.mhhe.com/hartwell3
Pioneer neurons in the mouse trigeminal sensory system
 Proc. Nadl. Acad. Sci. USA Vol. 87, pp. 923-927, February 1990 Neurobiology Pioneer neurons in the mouse trigeminal sensory system (mesencephalic trigeminal nucleus/monoclonal antibody/specific neuronal
Proc. Nadl. Acad. Sci. USA Vol. 87, pp. 923-927, February 1990 Neurobiology Pioneer neurons in the mouse trigeminal sensory system (mesencephalic trigeminal nucleus/monoclonal antibody/specific neuronal
A CRISPR/Cas9 Vector System for Tissue-Specific Gene Disruption in Zebrafish
 Developmental Cell Supplemental Information A CRISPR/Cas9 Vector System for Tissue-Specific Gene Disruption in Zebrafish Julien Ablain, Ellen M. Durand, Song Yang, Yi Zhou, and Leonard I. Zon % larvae
Developmental Cell Supplemental Information A CRISPR/Cas9 Vector System for Tissue-Specific Gene Disruption in Zebrafish Julien Ablain, Ellen M. Durand, Song Yang, Yi Zhou, and Leonard I. Zon % larvae
Culture and differentiation of embryonic stem cells. Hong-Lin Su Department of Life Sciences, National Chung-Hsing University
 Culture and differentiation of embryonic stem cells Hong-Lin Su Department of Life Sciences, National Chung-Hsing University Topics-ES cell maintenance Establishment Culture condition, including the serum,
Culture and differentiation of embryonic stem cells Hong-Lin Su Department of Life Sciences, National Chung-Hsing University Topics-ES cell maintenance Establishment Culture condition, including the serum,
ANAT2341 Embryology Introduction: Steve Palmer
 ANAT2341 Embryology Introduction: Steve Palmer Course overview Course lecturers specializations and roles Steve Palmer 9385 2957 Course overview Summary, aims and expected outcomes Course overview Graduate
ANAT2341 Embryology Introduction: Steve Palmer Course overview Course lecturers specializations and roles Steve Palmer 9385 2957 Course overview Summary, aims and expected outcomes Course overview Graduate
Cell autonomous vs. cell non-autonomous gene function
 Cell autonomous vs. cell non-autonomous gene function In multicellular organisms, it is important to know in what cell(s) the activity of a gene is required. - while RNA expression can be highly informative,
Cell autonomous vs. cell non-autonomous gene function In multicellular organisms, it is important to know in what cell(s) the activity of a gene is required. - while RNA expression can be highly informative,
Dynamic visualization of nervous system in live Drosophila
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 10438 10443, August 1999 Neurobiology Dynamic visualization of nervous system in live Drosophila BANGHUA SUN*, PEIZHANG XU*, AND PAUL M. SALVATERRA* *Division of
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 10438 10443, August 1999 Neurobiology Dynamic visualization of nervous system in live Drosophila BANGHUA SUN*, PEIZHANG XU*, AND PAUL M. SALVATERRA* *Division of
To assess the localization of Citrine fusion proteins, we performed antibody staining to
 Trinh et al 1 SUPPLEMENTAL MATERIAL FlipTraps recapitulate endogenous protein localization To assess the localization of Citrine fusion proteins, we performed antibody staining to compare the expression
Trinh et al 1 SUPPLEMENTAL MATERIAL FlipTraps recapitulate endogenous protein localization To assess the localization of Citrine fusion proteins, we performed antibody staining to compare the expression
Guided differentiation of ES cell into mesenchymal stem cell
 Guided differentiation of ES cell into mesenchymal stem cell Takumi Era Division of Molecular Neurobiology Institute of Molecular Embryology and Genetics, Kumamoto University Differentiation pathways of
Guided differentiation of ES cell into mesenchymal stem cell Takumi Era Division of Molecular Neurobiology Institute of Molecular Embryology and Genetics, Kumamoto University Differentiation pathways of
SUPPLEMENTARY INFORMATION
 a before amputation regeneration regenerated limb DERMIS SKELETON MUSCLE SCHWANN CELLS EPIDERMIS DERMIS SKELETON MUSCLE SCHWANN CELLS EPIDERMIS developmental origin: lateral plate mesoderm presomitic mesoderm
a before amputation regeneration regenerated limb DERMIS SKELETON MUSCLE SCHWANN CELLS EPIDERMIS DERMIS SKELETON MUSCLE SCHWANN CELLS EPIDERMIS developmental origin: lateral plate mesoderm presomitic mesoderm
Neural Induction. Chapter One
 Neural Induction Chapter One Fertilization Development of the Nervous System Cleavage (Blastula, Gastrula) Neuronal Induction- Neuroblast Formation Cell Migration Mesodermal Induction Lateral Inhibition
Neural Induction Chapter One Fertilization Development of the Nervous System Cleavage (Blastula, Gastrula) Neuronal Induction- Neuroblast Formation Cell Migration Mesodermal Induction Lateral Inhibition
Iridophores and their interactions with other chromatophores are required for stripe formation in zebrafish
 RESEARCH ARTICLE 2997 Development 140, 2997-3007 (2013) doi:10.1242/dev.096719 2013. Published by The Company of Biologists Ltd Iridophores and their interactions with other chromatophores are required
RESEARCH ARTICLE 2997 Development 140, 2997-3007 (2013) doi:10.1242/dev.096719 2013. Published by The Company of Biologists Ltd Iridophores and their interactions with other chromatophores are required
Reading. Lecture III. Nervous System Embryology. Biology. Brain Diseases. September 5, Bio 3411 Lecture III. Nervous System Embryology
 Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 Bio 3411 Wednesday 2 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 Bio 3411 Wednesday 2 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Gene Expression: Transcription
 Gene Expression: Transcription The majority of genes are expressed as the proteins they encode. The process occurs in two steps: Transcription = DNA RNA Translation = RNA protein Taken together, they make
Gene Expression: Transcription The majority of genes are expressed as the proteins they encode. The process occurs in two steps: Transcription = DNA RNA Translation = RNA protein Taken together, they make
Cell position and developmental fate in leech embryogenesis
 Proc. Nati. Acad. Sci. USA Vol. 87, pp. 8457-8461, November 1990 Developmental Biology Cell position and developmental fate in leech embryogenesis (cell lineage/transfating/equivalence group/determination)
Proc. Nati. Acad. Sci. USA Vol. 87, pp. 8457-8461, November 1990 Developmental Biology Cell position and developmental fate in leech embryogenesis (cell lineage/transfating/equivalence group/determination)
Lecture III. Nervous System Embryology
 Bio 3411 Wednesday Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 2 1 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Bio 3411 Wednesday Reading NEUROSCIENCE: 5 th ed, pp. 477-506 NEUROSCIENCE: 4 th ed, pp. 545-575 2 1 Summary from Lecture II Biology Understanding the brain is THE major question in biology and science.
Do Sensory Neurons Secrete an Anti-Inhibitory Factor that Promotes Regeneration?
 Kaleidoscope Volume 11 Article 30 July 2014 Do Sensory Neurons Secrete an Anti-Inhibitory Factor that Promotes Regeneration? Azita Bahrami Follow this and additional works at: https://uknowledge.uky.edu/kaleidoscope
Kaleidoscope Volume 11 Article 30 July 2014 Do Sensory Neurons Secrete an Anti-Inhibitory Factor that Promotes Regeneration? Azita Bahrami Follow this and additional works at: https://uknowledge.uky.edu/kaleidoscope
Developmental Biology. Cell Fate, Potency, and Determination
 Developmental Biology Cell Fate, Potency, and Determination Definitions Fate The sum of all structures that the cell or its descendants will form at a later stage of normal development. Cell Fate & Fate
Developmental Biology Cell Fate, Potency, and Determination Definitions Fate The sum of all structures that the cell or its descendants will form at a later stage of normal development. Cell Fate & Fate
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/331/6018/753/dc1 Supporting Online Material for Embryological Evidence Identifies Wing Digits in Birds as Digits 1, 2, and 3 Koji Tamura,* Naoki Nomura, Ryohei Seki,
www.sciencemag.org/cgi/content/full/331/6018/753/dc1 Supporting Online Material for Embryological Evidence Identifies Wing Digits in Birds as Digits 1, 2, and 3 Koji Tamura,* Naoki Nomura, Ryohei Seki,
! Allele Interactions
 Chapter 4!Extensions to Mendelian Genetics! Allele Interactions 1 INTRODUCTION Mendelian inheritance describes inheritance patterns that obey two laws Law of segregation Law of independent assortment Simple
Chapter 4!Extensions to Mendelian Genetics! Allele Interactions 1 INTRODUCTION Mendelian inheritance describes inheritance patterns that obey two laws Law of segregation Law of independent assortment Simple
(pp , these PROCEEDINGS) suggested to him to test the effects of the antiserum DESTRUCTION OF THE SYMPATHETIC GANGLIA IN MAMMALS BY
 384 ZOOLOGY: LEVI-MONTALC'INI AND BOOKER PROC. N. A. S. * This work has been supported by a grant from the National Science Foundation and by a contribution from an Institutional grant of the American
384 ZOOLOGY: LEVI-MONTALC'INI AND BOOKER PROC. N. A. S. * This work has been supported by a grant from the National Science Foundation and by a contribution from an Institutional grant of the American
Supplementary Figure S1. Immunodetection of full-length XA21 and the XA21 C-terminal cleavage product.
 Supplementary Information Supplementary Figure S1. Immunodetection of full-length XA21 and the XA21 C-terminal cleavage product. Total protein extracted from Kitaake wild type and rice plants carrying
Supplementary Information Supplementary Figure S1. Immunodetection of full-length XA21 and the XA21 C-terminal cleavage product. Total protein extracted from Kitaake wild type and rice plants carrying
SUPPLEMENTARY INFORMATION. Transcriptional output transiently spikes upon mitotic exit
 SUPPLEMENTARY INFORMATION Transcriptional output transiently spikes upon mitotic exit Viola Vaňková Hausnerová 1, 2, Christian Lanctôt 1* 1 BIOCEV and Department of Cell Biology, Faculty of Science, Charles
SUPPLEMENTARY INFORMATION Transcriptional output transiently spikes upon mitotic exit Viola Vaňková Hausnerová 1, 2, Christian Lanctôt 1* 1 BIOCEV and Department of Cell Biology, Faculty of Science, Charles
GM130 Is Required for Compartmental Organization of Dendritic Golgi Outposts
 Current Biology, Volume 24 Supplemental Information GM130 Is Required for Compartmental Organization of Dendritic Golgi Outposts Wei Zhou, Jin Chang, Xin Wang, Masha G. Savelieff, Yinyin Zhao, Shanshan
Current Biology, Volume 24 Supplemental Information GM130 Is Required for Compartmental Organization of Dendritic Golgi Outposts Wei Zhou, Jin Chang, Xin Wang, Masha G. Savelieff, Yinyin Zhao, Shanshan
SOX2 antibody - Embryonic Stem Cell Marker (ab15830) datasheet
 -Embryonic Stem Cell Marker (ab15830) 1/8 ページ d Products: Stem Cells >> Germline Stem Cells >> Embryonic Germ Cells SOX2 antibody - Embryonic Stem Cell Marker (ab15830) datasheet Product Name Product type
-Embryonic Stem Cell Marker (ab15830) 1/8 ページ d Products: Stem Cells >> Germline Stem Cells >> Embryonic Germ Cells SOX2 antibody - Embryonic Stem Cell Marker (ab15830) datasheet Product Name Product type
Developmental Biology 3230 Exam 1 (Feb. 6) NAME
 DevelopmentalBiology3230Exam1(Feb.6)NAME 1. (10pts) What is a Fate Map? How would you experimentally acquire the data to draw a Fate Map? Explain what a Fate Map does and does not tell you about the mechanisms
DevelopmentalBiology3230Exam1(Feb.6)NAME 1. (10pts) What is a Fate Map? How would you experimentally acquire the data to draw a Fate Map? Explain what a Fate Map does and does not tell you about the mechanisms
Supplemental Information. Glutamylation Regulates Transport, Specializes. Function, and Sculpts the Structure of Cilia
 Current Biology, Volume 27 Supplemental Information Glutamylation Regulates Transport, Specializes Function, and Sculpts the Structure of Cilia Robert O'Hagan, Malan Silva, Ken C.Q. Nguyen, Winnie Zhang,
Current Biology, Volume 27 Supplemental Information Glutamylation Regulates Transport, Specializes Function, and Sculpts the Structure of Cilia Robert O'Hagan, Malan Silva, Ken C.Q. Nguyen, Winnie Zhang,
I (ALLEN 1965; GIBSON 1970). In Tetrahymena pyriformis, syngen 1, a genetically
 SHORT PAPERS MUTANT SELECTION IN TETRAHYMENA PYRIFORMIS PETER S. CARLSON' Department of Biology, Wesleyan University, Middletown, Connecticut 06457 Received June 18, 1971 NTRACLONAL variation is a well
SHORT PAPERS MUTANT SELECTION IN TETRAHYMENA PYRIFORMIS PETER S. CARLSON' Department of Biology, Wesleyan University, Middletown, Connecticut 06457 Received June 18, 1971 NTRACLONAL variation is a well
unc-33/crmp and ankyrin organize microtubules and localize kinesin to polarize
 Supplementary Information unc-33/crmp and ankyrin organize microtubules and localize kinesin to polarize axon-dendrite sorting Tapan A. Maniar 1, Miriam Kaplan 1, George J. Wang 2, Kang Shen 2, Li Wei
Supplementary Information unc-33/crmp and ankyrin organize microtubules and localize kinesin to polarize axon-dendrite sorting Tapan A. Maniar 1, Miriam Kaplan 1, George J. Wang 2, Kang Shen 2, Li Wei
Supporting Figure 1: Meis2 and Pax6 transcripts in the adult murine brain detected by in situ
 DEVELOP2013097295_supplementary figures Supporting Figure 1: Meis2 and Pax6 transcripts in the adult murine brain detected by in situ hybridization on vibratome sections. (A) Meis2-specific transcripts
DEVELOP2013097295_supplementary figures Supporting Figure 1: Meis2 and Pax6 transcripts in the adult murine brain detected by in situ hybridization on vibratome sections. (A) Meis2-specific transcripts
Fluorescence Microscopy. Terms and concepts to know: 10/11/2011. Visible spectrum (of light) and energy
 Fluorescence Microscopy Louisiana Tech University Ruston, Louisiana Microscopy Workshop Dr. Mark DeCoster Associate Professor Biomedical Engineering 1 Terms and concepts to know: Signal to Noise Excitation
Fluorescence Microscopy Louisiana Tech University Ruston, Louisiana Microscopy Workshop Dr. Mark DeCoster Associate Professor Biomedical Engineering 1 Terms and concepts to know: Signal to Noise Excitation
Nature Immunology: doi: /ni Supplementary Figure 1
 Supplementary Figure 1 BALB/c LYVE1-deficient mice exhibited reduced lymphatic trafficking of all DC subsets after oxazolone-induced sensitization. (a) Schematic overview of the mouse skin oxazolone contact
Supplementary Figure 1 BALB/c LYVE1-deficient mice exhibited reduced lymphatic trafficking of all DC subsets after oxazolone-induced sensitization. (a) Schematic overview of the mouse skin oxazolone contact
Neural Induction. Steven McLoon Department of Neuroscience University of Minnesota
 Neural Induction Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour (with Dr. Nakagawa) Friday, Sept 22 8:30-9:30am Surdyks Café in Northrop Auditorium Stop by for
Neural Induction Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour (with Dr. Nakagawa) Friday, Sept 22 8:30-9:30am Surdyks Café in Northrop Auditorium Stop by for
Methods of Characterizing Neural Networks
 Methods of Characterizing Neural Networks Ashley Nord University of Minnesota Minneapolis, MN 55414 Advisors: Katsushi Arisaka, Adrian Cheng University of California Los Angeles Los Angeles, CA 90024 September
Methods of Characterizing Neural Networks Ashley Nord University of Minnesota Minneapolis, MN 55414 Advisors: Katsushi Arisaka, Adrian Cheng University of California Los Angeles Los Angeles, CA 90024 September
GENESDEV/2007/ Supplementary Figure 1 Elkabetz et al.,
 GENESDEV/2007/089581 Supplementary Figure 1 Elkabetz et al., GENESDEV/2007/089581 Supplementary Figure 2 Elkabetz et al., GENESDEV/2007/089581 Supplementary Figure 3 Elkabetz et al., GENESDEV/2007/089581
GENESDEV/2007/089581 Supplementary Figure 1 Elkabetz et al., GENESDEV/2007/089581 Supplementary Figure 2 Elkabetz et al., GENESDEV/2007/089581 Supplementary Figure 3 Elkabetz et al., GENESDEV/2007/089581
sides of the aleurone (Al) but it is excluded from the basal endosperm transfer layer
 Supplemental Data. Gómez et al. (2009). The maize transcription factor MRP-1 (Myb-Related-Protein-1) is a key regulator of the differentiation of transfer cells. Supplemental Figure 1. Expression analyses
Supplemental Data. Gómez et al. (2009). The maize transcription factor MRP-1 (Myb-Related-Protein-1) is a key regulator of the differentiation of transfer cells. Supplemental Figure 1. Expression analyses
Genetic Basis of Development & Biotechnologies
 Genetic Basis of Development & Biotechnologies 1. Steps of embryonic development: cell division, morphogenesis, differentiation Totipotency and pluripotency 2. Plant cloning 3. Animal cloning Reproductive
Genetic Basis of Development & Biotechnologies 1. Steps of embryonic development: cell division, morphogenesis, differentiation Totipotency and pluripotency 2. Plant cloning 3. Animal cloning Reproductive
Visualizing Cells Molecular Biology of the Cell - Chapter 9
 Visualizing Cells Molecular Biology of the Cell - Chapter 9 Resolution, Detection Magnification Interaction of Light with matter: Absorbtion, Refraction, Reflection, Fluorescence Light Microscopy Absorbtion
Visualizing Cells Molecular Biology of the Cell - Chapter 9 Resolution, Detection Magnification Interaction of Light with matter: Absorbtion, Refraction, Reflection, Fluorescence Light Microscopy Absorbtion
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Kanaani et al., http://www.jcb.org/cgi/content/full/jcb.200912101/dc1 Figure S1. The K2 rabbit polyclonal antibody is specific for GAD67,
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Kanaani et al., http://www.jcb.org/cgi/content/full/jcb.200912101/dc1 Figure S1. The K2 rabbit polyclonal antibody is specific for GAD67,
Materials and Methods: All strains were derivatives of SK1 and are listed in Supplemental Table 1.
 Supplemental Online Material: Materials and Methods: All strains were derivatives of SK1 and are listed in Supplemental Table 1. Constructs: pclb2-3ha-cdc5 and pclb2-3ha-cdc2 strains were constructed by
Supplemental Online Material: Materials and Methods: All strains were derivatives of SK1 and are listed in Supplemental Table 1. Constructs: pclb2-3ha-cdc5 and pclb2-3ha-cdc2 strains were constructed by
elegans controls the time of expression of
 The h'n-14 locus of Caenorhabditis. elegans controls the time of expression of specific postembryonic developmental events Victor Ambros 1 and H. Robert Horvitz Department of Biology, Massachusetts nstitute
The h'n-14 locus of Caenorhabditis. elegans controls the time of expression of specific postembryonic developmental events Victor Ambros 1 and H. Robert Horvitz Department of Biology, Massachusetts nstitute
Additional Practice Problems for Reading Period
 BS 50 Genetics and Genomics Reading Period Additional Practice Problems for Reading Period Question 1. In patients with a particular type of leukemia, their leukemic B lymphocytes display a translocation
BS 50 Genetics and Genomics Reading Period Additional Practice Problems for Reading Period Question 1. In patients with a particular type of leukemia, their leukemic B lymphocytes display a translocation
The regulation of mesodermal progenitor cell commitment to somitogenesis subdivides the zebrafish body musculature into distinct domains
 The regulation of mesodermal progenitor cell commitment to somitogenesis subdivides the zebrafish body musculature into distinct domains Daniel P. Szeto 1 and David Kimelman 2 Department of Biochemistry,
The regulation of mesodermal progenitor cell commitment to somitogenesis subdivides the zebrafish body musculature into distinct domains Daniel P. Szeto 1 and David Kimelman 2 Department of Biochemistry,
Afferent Neurons of the Zebrafish Lateral Line Are Strict Selectors of Hair-Cell Orientation
 Afferent Neurons of the Zebrafish Lateral Line Are Strict Selectors of Hair-Cell Orientation Adèle Faucherre 1, Jesús Pujol-Martí 1, Koichi Kawakami 2, Hernán López-Schier 1 * 1 Laboratory of Sensory Cell
Afferent Neurons of the Zebrafish Lateral Line Are Strict Selectors of Hair-Cell Orientation Adèle Faucherre 1, Jesús Pujol-Martí 1, Koichi Kawakami 2, Hernán López-Schier 1 * 1 Laboratory of Sensory Cell
Role of Bmi-1 in Epigenetic Regulation. During Early Neural Crest Development
 Role of Bmi-1 in Epigenetic Regulation During Early Neural Crest Development Thesis by Jane I. Khudyakov In Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy California Institute
Role of Bmi-1 in Epigenetic Regulation During Early Neural Crest Development Thesis by Jane I. Khudyakov In Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy California Institute
SUPPLEMENTARY FIGURES AND VIDEOS
 SUPPLEMENTARY FIGURES AND VIDEOS Supplementary Figure 1. Generation of Human Intestinal Organoids (HIOs). (a) Method for generating HIOs through directed differentiation of human pluripotent stem cells.
SUPPLEMENTARY FIGURES AND VIDEOS Supplementary Figure 1. Generation of Human Intestinal Organoids (HIOs). (a) Method for generating HIOs through directed differentiation of human pluripotent stem cells.
Lecture 2-3: Using Mutants to study Biological processes
 Lecture 2-3: Using Mutants to study Biological processes Objectives: 1. Why use mutants? 2. How are mutants isolated? 3. What important genetic analyses must be done immediately after a genetic screen
Lecture 2-3: Using Mutants to study Biological processes Objectives: 1. Why use mutants? 2. How are mutants isolated? 3. What important genetic analyses must be done immediately after a genetic screen
Concepts and Methods in Developmental Biology
 Biology 4361 Developmental Biology Concepts and Methods in Developmental Biology June 16, 2009 Conceptual and Methodological Tools Concepts Genomic equivalence Differential gene expression Differentiation/de-differentiation
Biology 4361 Developmental Biology Concepts and Methods in Developmental Biology June 16, 2009 Conceptual and Methodological Tools Concepts Genomic equivalence Differential gene expression Differentiation/de-differentiation
Enhancers mutations that make the original mutant phenotype more extreme. Suppressors mutations that make the original mutant phenotype less extreme
 Interactomics and Proteomics 1. Interactomics The field of interactomics is concerned with interactions between genes or proteins. They can be genetic interactions, in which two genes are involved in the
Interactomics and Proteomics 1. Interactomics The field of interactomics is concerned with interactions between genes or proteins. They can be genetic interactions, in which two genes are involved in the
Cycles of vascular plexus formation within the nephrogenic zone of the developing mouse kidney
 1 Supplementary text and data for: 2 3 4 5 Cycles of vascular plexus formation within the nephrogenic zone of the developing mouse kidney Authors: David A. D. Munro 1*, Peter Hohenstein 2, and Jamie A.
1 Supplementary text and data for: 2 3 4 5 Cycles of vascular plexus formation within the nephrogenic zone of the developing mouse kidney Authors: David A. D. Munro 1*, Peter Hohenstein 2, and Jamie A.
Chapter 11. How Genes Are Controlled. Lectures by Edward J. Zalisko
 Chapter 11 How Genes Are Controlled PowerPoint Lectures for Campbell Essential Biology, Fifth Edition, and Campbell Essential Biology with Physiology, Fourth Edition Eric J. Simon, Jean L. Dickey, and
Chapter 11 How Genes Are Controlled PowerPoint Lectures for Campbell Essential Biology, Fifth Edition, and Campbell Essential Biology with Physiology, Fourth Edition Eric J. Simon, Jean L. Dickey, and
HuC-eGFP mosaic labelling of neurons in zebrafish enables in vivo live cell imaging of growth cones
 HuC-eGFP mosaic labelling of neurons in zebrafish enables in vivo live cell imaging of growth cones Author St John, James, Key, Brian Published 2012 Journal Title Journal of Molecular Histology DOI https://doi.org/10.1007/s10735-012-9462-7
HuC-eGFP mosaic labelling of neurons in zebrafish enables in vivo live cell imaging of growth cones Author St John, James, Key, Brian Published 2012 Journal Title Journal of Molecular Histology DOI https://doi.org/10.1007/s10735-012-9462-7
Fundamental properties of Stem Cells
 Stem cells Learning Goals: Define what a stem cell is and describe its general properties, using hematopoietic stem cells as an example. Describe to a non-scientist the current progress of human stem cell
Stem cells Learning Goals: Define what a stem cell is and describe its general properties, using hematopoietic stem cells as an example. Describe to a non-scientist the current progress of human stem cell
Supplementary Figure 1: Derivation and characterization of RN ips cell lines. (a) RN ips cells maintain expression of pluripotency markers OCT4 and
 Supplementary Figure 1: Derivation and characterization of RN ips cell lines. (a) RN ips cells maintain expression of pluripotency markers OCT4 and SSEA4 after 10 passages in mtesr 1 medium. (b) Schematic
Supplementary Figure 1: Derivation and characterization of RN ips cell lines. (a) RN ips cells maintain expression of pluripotency markers OCT4 and SSEA4 after 10 passages in mtesr 1 medium. (b) Schematic
2004 Debye Lecture 4 C. B. Murray. Quantum Dot Applications: Sun Screen. Solar Cells. Bio-tagging. Solid State Lighting?
 2004 Debye Lecture 4 C. B. Murray Quantum Dot Applications: Sun Screen Solar Cells Bio-tagging Solid State Lighting? Quantum Dot Solar cells Nanocrystal Solar Cells Double-labeling of mitochondria
2004 Debye Lecture 4 C. B. Murray Quantum Dot Applications: Sun Screen Solar Cells Bio-tagging Solid State Lighting? Quantum Dot Solar cells Nanocrystal Solar Cells Double-labeling of mitochondria
Non-mammalian animal models in developmental toxicology. Dr. Michael Oelgeschläger
 FEDERAL INSTITUTE FOR RISK ASSESSMENT Non-mammalian animal models in developmental toxicology Dr. Michael Oelgeschläger Nick Hobgood, wikipedia Emergence of animal models in experimental developmental
FEDERAL INSTITUTE FOR RISK ASSESSMENT Non-mammalian animal models in developmental toxicology Dr. Michael Oelgeschläger Nick Hobgood, wikipedia Emergence of animal models in experimental developmental
Assessing welfare and severity of GA mice under Directive 2010/63/EU. James Bussell Wellcome Trust Sanger Institute
 Assessing welfare and severity of GA mice under Directive 2010/63/EU James Bussell Wellcome Trust Sanger Institute Varese, Italy 3 rd December 2012 David Anderson Assessing welfare and severity of GA mice
Assessing welfare and severity of GA mice under Directive 2010/63/EU James Bussell Wellcome Trust Sanger Institute Varese, Italy 3 rd December 2012 David Anderson Assessing welfare and severity of GA mice
DNA Transcription. Visualizing Transcription. The Transcription Process
 DNA Transcription By: Suzanne Clancy, Ph.D. 2008 Nature Education Citation: Clancy, S. (2008) DNA transcription. Nature Education 1(1) If DNA is a book, then how is it read? Learn more about the DNA transcription
DNA Transcription By: Suzanne Clancy, Ph.D. 2008 Nature Education Citation: Clancy, S. (2008) DNA transcription. Nature Education 1(1) If DNA is a book, then how is it read? Learn more about the DNA transcription
The zebrafish issue of Development
 SPOTLIGHT 4099 Development 139, 4099-4103 (2012) doi:10.1242/dev.085217 2012. Published by The Company of Biologists Ltd The zebrafish issue of Development Christiane Nüsslein-Volhard* Summary In December
SPOTLIGHT 4099 Development 139, 4099-4103 (2012) doi:10.1242/dev.085217 2012. Published by The Company of Biologists Ltd The zebrafish issue of Development Christiane Nüsslein-Volhard* Summary In December
Supplemental Information. Boundary Formation through a Direct. Threshold-Based Readout. of Mobile Small RNA Gradients
 Developmental Cell, Volume 43 Supplemental Information Boundary Formation through a Direct Threshold-Based Readout of Mobile Small RNA Gradients Damianos S. Skopelitis, Anna H. Benkovics, Aman Y. Husbands,
Developmental Cell, Volume 43 Supplemental Information Boundary Formation through a Direct Threshold-Based Readout of Mobile Small RNA Gradients Damianos S. Skopelitis, Anna H. Benkovics, Aman Y. Husbands,
Cell culture. HeLa cells were cultured as monolayers in Dulbecco s Minimal Essential
 Supporting Online Material Materials and methods Cell culture. HeLa cells were cultured as monolayers in Dulbecco s Minimal Essential Medium (Gibco BRL, Invitrogen Corporation, Carlsbad, CA, USA), supplemented
Supporting Online Material Materials and methods Cell culture. HeLa cells were cultured as monolayers in Dulbecco s Minimal Essential Medium (Gibco BRL, Invitrogen Corporation, Carlsbad, CA, USA), supplemented
QImaging Camera Application Notes Multicolor Immunofluorescence Imaging
 QImaging Camera Application Notes Multicolor Immunofluorescence Imaging In order to image localization of intracellular proteins with high specificity, it is frequently necessary to multiplex antibody
QImaging Camera Application Notes Multicolor Immunofluorescence Imaging In order to image localization of intracellular proteins with high specificity, it is frequently necessary to multiplex antibody
Student Manual. pglo Transformation
 Student Manual pglo Transformation Lesson 1 Introduction to Transformation In this lab you will perform a procedure known as genetic transformation. Remember that a gene is a piece of DNA which provides
Student Manual pglo Transformation Lesson 1 Introduction to Transformation In this lab you will perform a procedure known as genetic transformation. Remember that a gene is a piece of DNA which provides
BIO 315 Lab Exam I. Section #: Name:
 Section #: Name: Also provide this information on the computer grid sheet given to you. (Section # in special code box) BIO 315 Lab Exam I 1. In labeling the parts of a standard compound light microscope
Section #: Name: Also provide this information on the computer grid sheet given to you. (Section # in special code box) BIO 315 Lab Exam I 1. In labeling the parts of a standard compound light microscope
Readings. Lecture IV. Mechanisms of Neural. Neural Development. September 10, Bio 3411 Lecture IV. Mechanisms of Neural Development
 Readings Lecture IV. Mechanisms of Neural NEUROSCIENCE: References : 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994). EMBO J, 13(21),
Readings Lecture IV. Mechanisms of Neural NEUROSCIENCE: References : 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994). EMBO J, 13(21),
Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
1. Immunization. What is Immunization? 12/9/2016. Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
The Human Protein PRR14 Tethers Heterochromatin to the Nuclear Lamina During Interphase and Mitotic Exit
 Cell Reports, Volume 5 Supplemental Information The Human Protein PRR14 Tethers Heterochromatin to the Nuclear Lamina During Interphase and Mitotic Exit Andrey Poleshko, Katelyn M. Mansfield, Caroline
Cell Reports, Volume 5 Supplemental Information The Human Protein PRR14 Tethers Heterochromatin to the Nuclear Lamina During Interphase and Mitotic Exit Andrey Poleshko, Katelyn M. Mansfield, Caroline
Confocal Microscopy Analyzes Cells
 Choosing Filters for Fluorescence A Laurin Publication Photonic Solutions for Biotechnology and Medicine November 2002 Confocal Microscopy Analyzes Cells Reprinted from the November 2002 issue of Biophotonics
Choosing Filters for Fluorescence A Laurin Publication Photonic Solutions for Biotechnology and Medicine November 2002 Confocal Microscopy Analyzes Cells Reprinted from the November 2002 issue of Biophotonics
To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well
 Supplemental Information: Supplemental Methods: Cell culture To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well in 96 well Primaria plates in GNS media and incubated at
Supplemental Information: Supplemental Methods: Cell culture To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well in 96 well Primaria plates in GNS media and incubated at
Use of Phase Contrast Imaging to Track Morphological Cellular Changes due to Apoptotic Activity
 A p p l i c a t i o n N o t e Use of Phase Contrast Imaging to Track Morphological Cellular Changes due to Apoptotic Activity Brad Larson and Peter Banks, Applications Department, BioTek Instruments, Inc.,
A p p l i c a t i o n N o t e Use of Phase Contrast Imaging to Track Morphological Cellular Changes due to Apoptotic Activity Brad Larson and Peter Banks, Applications Department, BioTek Instruments, Inc.,
Genetics - Problem Drill 19: Dissection of Gene Function: Mutational Analysis of Model Organisms
 Genetics - Problem Drill 19: Dissection of Gene Function: Mutational Analysis of Model Organisms No. 1 of 10 1. The mouse gene knockout is based on. (A) Homologous recombination (B) Site-specific recombination
Genetics - Problem Drill 19: Dissection of Gene Function: Mutational Analysis of Model Organisms No. 1 of 10 1. The mouse gene knockout is based on. (A) Homologous recombination (B) Site-specific recombination
Enhancer genetics Problem set E
 Enhancer genetics Problem set E How are specific cell types generated during development? There are thought to be 10,000 different types of neurons in the human nervous system. How are all of these neural
Enhancer genetics Problem set E How are specific cell types generated during development? There are thought to be 10,000 different types of neurons in the human nervous system. How are all of these neural
A novel stem cell for auditory neuron regeneration.
 A novel stem cell for auditory neuron regeneration. M.A.Huisman*, R. el Seady*, C.F.H.M.Löwik # en J.H.M.Frijns* Departments of Ear, Nose & Throat* and Molecular Imaging #, Leiden University Medical Centre,
A novel stem cell for auditory neuron regeneration. M.A.Huisman*, R. el Seady*, C.F.H.M.Löwik # en J.H.M.Frijns* Departments of Ear, Nose & Throat* and Molecular Imaging #, Leiden University Medical Centre,
brain's soluble proteins. Antiserum to this protein, isolated from beef brain,
 A GLIAL PROTEIN SPECIFIC FOR THE NERVOUS SYSTEM* BY H. HYD*N AND B. MCEWENt INSTITUTE OF NEUROBIOLOGY, FACULTY OF MEDICINE, UNIVERSITY OF G6TEBORG, SWEDEN Communicated by A. E. Mirsky, December 9, 1965
A GLIAL PROTEIN SPECIFIC FOR THE NERVOUS SYSTEM* BY H. HYD*N AND B. MCEWENt INSTITUTE OF NEUROBIOLOGY, FACULTY OF MEDICINE, UNIVERSITY OF G6TEBORG, SWEDEN Communicated by A. E. Mirsky, December 9, 1965
AGRO/ANSC/BIO/GENE/HORT 305 Fall, 2016 Overview of Genetics Lecture outline (Chpt 1, Genetics by Brooker) #1
 AGRO/ANSC/BIO/GENE/HORT 305 Fall, 2016 Overview of Genetics Lecture outline (Chpt 1, Genetics by Brooker) #1 - Genetics: Progress from Mendel to DNA: Gregor Mendel, in the mid 19 th century provided the
AGRO/ANSC/BIO/GENE/HORT 305 Fall, 2016 Overview of Genetics Lecture outline (Chpt 1, Genetics by Brooker) #1 - Genetics: Progress from Mendel to DNA: Gregor Mendel, in the mid 19 th century provided the
Introduction to Microarray Analysis
 Introduction to Microarray Analysis Methods Course: Gene Expression Data Analysis -Day One Rainer Spang Microarrays Highly parallel measurement devices for gene expression levels 1. How does the microarray
Introduction to Microarray Analysis Methods Course: Gene Expression Data Analysis -Day One Rainer Spang Microarrays Highly parallel measurement devices for gene expression levels 1. How does the microarray
Cytomics in Action: Cytokine Network Cytometry
 Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
Computers in Biology and Bioinformatics
 Computers in Biology and Bioinformatics 1 Biology biology is roughly defined as "the study of life" it is concerned with the characteristics and behaviors of organisms, how species and individuals come
Computers in Biology and Bioinformatics 1 Biology biology is roughly defined as "the study of life" it is concerned with the characteristics and behaviors of organisms, how species and individuals come
Figure S2. Response of mouse ES cells to GSK3 inhibition. Mentioned in discussion
 Stem Cell Reports, Volume 1 Supplemental Information Robust Self-Renewal of Rat Embryonic Stem Cells Requires Fine-Tuning of Glycogen Synthase Kinase-3 Inhibition Yaoyao Chen, Kathryn Blair, and Austin
Stem Cell Reports, Volume 1 Supplemental Information Robust Self-Renewal of Rat Embryonic Stem Cells Requires Fine-Tuning of Glycogen Synthase Kinase-3 Inhibition Yaoyao Chen, Kathryn Blair, and Austin
OPTICAL CONTROL OF TUMOR INDUCTION IN THE ZEBRAFISH
 OPTICAL CONTROL OF TUMOR INDUCTION IN THE ZEBRAFISH Zhiping Feng, 1,12 Suzy Nam, 2 Fatima Hamouri, 3,4 Isabelle Aujard, 5,6 Bertrand Ducos, 3,4 Sophie Vriz, 7,8 Michel Volovitch, 7,9 Ludovic Jullien, 5,6
OPTICAL CONTROL OF TUMOR INDUCTION IN THE ZEBRAFISH Zhiping Feng, 1,12 Suzy Nam, 2 Fatima Hamouri, 3,4 Isabelle Aujard, 5,6 Bertrand Ducos, 3,4 Sophie Vriz, 7,8 Michel Volovitch, 7,9 Ludovic Jullien, 5,6
mcherry Monoclonal Antibody (16D7) Catalog Number M11217 Product data sheet
 Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 mcherry Monoclonal Antibody (16D7) Catalog Number M11217 Product data sheet Details
Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 mcherry Monoclonal Antibody (16D7) Catalog Number M11217 Product data sheet Details
Requirement for a Functional int Product in Temperature Inductions of
 JOURNAL OF VIROLOGY, SePt. 1970, p. 320-325 Vol. 6, No. 3 Copyright 1970 American Society for Microbiology Prinited in U.S.A. Requirement for a Functional int Product in Temperature Inductions of Prophage
JOURNAL OF VIROLOGY, SePt. 1970, p. 320-325 Vol. 6, No. 3 Copyright 1970 American Society for Microbiology Prinited in U.S.A. Requirement for a Functional int Product in Temperature Inductions of Prophage
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/30138 holds various files of this Leiden University dissertation Author: Driessen, Marja Title: Evaluation of the zebrafish embryo as an alternative model
Cover Page The handle http://hdl.handle.net/1887/30138 holds various files of this Leiden University dissertation Author: Driessen, Marja Title: Evaluation of the zebrafish embryo as an alternative model
Nucleofector Technology in. Somatic Stem Cell Research. gene transfer begins here
 Nucleofector Technology in Somatic Stem Cell Research The Nucleofector technology The first highly efficient method for non-viral gene transfer into primary cells and difficult-to-transfect cell lines
Nucleofector Technology in Somatic Stem Cell Research The Nucleofector technology The first highly efficient method for non-viral gene transfer into primary cells and difficult-to-transfect cell lines
Description: Nuclear morphology and dynamics in nontargeting sirna transfected cells. HeLa Kyoto
 Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Tables Title of file for HTML: Supplementary Movie 1 Description: Nuclear morphology and dynamics
Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Tables Title of file for HTML: Supplementary Movie 1 Description: Nuclear morphology and dynamics
Microarrays and Stem Cells
 Microarray Background Information Microarrays and Stem Cells Stem cells are the building blocks that allow the body to produce new cells and repair tissues. Scientists are actively investigating the potential
Microarray Background Information Microarrays and Stem Cells Stem cells are the building blocks that allow the body to produce new cells and repair tissues. Scientists are actively investigating the potential
Beta3 integrin promotes long-lasting activation and polarization of Vascular Endothelial Growth Factor Receptor 2 by immobilized ligand
 SUPPLEMENTAL FIGURES Beta3 integrin promotes long-lasting activation and polarization of Vascular Endothelial Growth Factor Receptor 2 by immobilized ligand C. Ravelli et al. FIGURE S. I Figure S. I: Gremlin
SUPPLEMENTAL FIGURES Beta3 integrin promotes long-lasting activation and polarization of Vascular Endothelial Growth Factor Receptor 2 by immobilized ligand C. Ravelli et al. FIGURE S. I Figure S. I: Gremlin
E. Incorrect! The four different DNA nucleotides follow a strict base pairing arrangement:
 AP Biology - Problem Drill 10: Molecular and Human Genetics Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully, (2) Work the problems on paper as 1. Which of the following
AP Biology - Problem Drill 10: Molecular and Human Genetics Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully, (2) Work the problems on paper as 1. Which of the following
SUPPLEMENTARY INFORMATION
 b Supplementary Figure 1. Analysis of Vkg-Dpp interactions. a, In vitro pulldown showing binding of Dpp-HA to the GST-VkgC fusion protein. No interaction is detected between denatured Dpp-HA and GST-Vkg.
b Supplementary Figure 1. Analysis of Vkg-Dpp interactions. a, In vitro pulldown showing binding of Dpp-HA to the GST-VkgC fusion protein. No interaction is detected between denatured Dpp-HA and GST-Vkg.
DEPArray Technology. Sorting and Recovery of Rare Cells
 DEPArray Technology Sorting and Recovery of Rare Cells Delivering pure, single, viable cells The DEPArray system from Silicon Biosystems is the only automated instrument that can identify, quantify, and
DEPArray Technology Sorting and Recovery of Rare Cells Delivering pure, single, viable cells The DEPArray system from Silicon Biosystems is the only automated instrument that can identify, quantify, and
Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling
 Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling Glendining KA 1, Markie D 2, Gardner RJM 4, Franz EA 3, Robertson SP 4, Jasoni CL 1 Supplementary
Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling Glendining KA 1, Markie D 2, Gardner RJM 4, Franz EA 3, Robertson SP 4, Jasoni CL 1 Supplementary
AP Biology. Scoring Guidelines
 2017 AP Biology Scoring Guidelines College Board, Advanced Placement Program, AP, AP Central, and the acorn logo are registered trademarks of the College Board. AP Central is the official online home for
2017 AP Biology Scoring Guidelines College Board, Advanced Placement Program, AP, AP Central, and the acorn logo are registered trademarks of the College Board. AP Central is the official online home for
Activity 47.1 What common events occur in the early development of animals? 1. What key events occur at each stage of development?
 Notes to Instructors Chapter 47 Animal Development What is the focus of this activity? Chapter 21 provided a review of how genes act to control development. Chapter 47 reviews some of the major morphological
Notes to Instructors Chapter 47 Animal Development What is the focus of this activity? Chapter 21 provided a review of how genes act to control development. Chapter 47 reviews some of the major morphological
